Abstract
Thymic tissue has previously been considered a requirement for the generation of a functional and diverse population of human T cells. We report that fibroblasts and keratinocytes from human skin arrayed on a synthetic 3-dimensional matrix support the development of functional human T cells from hematopoietic precursor cells in the absence of thymic tissue. Newly generated T cells contained T cell receptor excision circles, possessed a diverse T cell repertoire, and were functionally mature and tolerant to self MHC, indicating successful completion of positive and negative selection. Skin cell cultures expressed the AIRE, Foxn1, and Hoxa3 transcription factors and a panel of autoantigens. Skin and bone marrow biopsies can thus be used to generate de novo functional and diverse T cell populations for potential therapeutic use in immunosuppressed patients.
Introduction
T cells play a central role in the human adaptive immune system, discriminating self from non-self, directing less specific elements of the immune system in the performance of effector functions, and serving as a repository for immunologic memory (1). The ability to generate T cells suitable for use in human patients could lead to significant advances in the treatment of immunodeficiencies and malignancies. Such therapies have not yet been realized because the unique requirements for T cell development are difficult to replicate in vitro.
T cell differentiation involves an obligate period of development and education in the thymus, an organ that undergoes involution in late adolescence and maintains only a low level of activity in normal adults (2, 3). Current culture systems that support the development of human T cells from bone marrow precursor cells use animal or unrelated human tissues, making them less desirable for the production of T cells for use in humans (4–16).
The features of the thymus that endow it with the capacity to generate T cells have been studied extensively. Epithelial cells, stromal cells, DCs, and lymphopoietic cytokines all appear to be required for the differentiation of bone marrow hematopoietic precursor cells (HPCs) into mature, functional T cells (17).
There are a remarkable number of similarities between the epithelial and stromal cells of the thymus and the keratinocytes and fibroblasts of the skin. Keratinocytes express the FOXN1 transcription factor that is crucial for thymic epithelial cell development (18) as well as many of the same keratins as thymic epithelial cells (19) and delta-like Notch ligands (20). Hassall corpuscles, a product of medullary thymic epithelial cells, contain keratins identical to those seen in the stratum corneum of skin (19). Similarly, thymic stromal cells bear a strong resemblance to the fibroblasts of skin. In fact, the major difference between thymus and skin is the distinct 3-dimensional architecture of each organ. Most epithelial cells are arrayed along a basement membrane and thereby segregated from many other cell types, but the epithelial cells of the thymus are arrayed in an open net-like structure that allows for maximal contact with thymocytes. In the present study, we tested the hypothesis that cellular elements of skin, reconfigured in a different 3-dimensional arrangement, can support the differentiation of T cells from HPCs. This hypothesis could be readily tested because human skin is accessible for biopsy with minimal morbidity, and methods for culturing and expanding keratinocytes and fibroblasts in vitro are well established. Similarly, HPCs are readily obtained with minimal injury to donors.
We report here that keratinocytes and fibroblasts from normal adult human skin supported the full development of T cells from HPCs. Thus, human autologous T cells can be produced using only samples of skin and bone marrow.
Results
Design of the skin cell construct.
Thymic epithelial cells are thought to provide the crucial signals for positive selection that allow double-positive T cell precursors to mature into single-positive T cells (17). It was therefore important to ensure that the construct contained large numbers of healthy epithelial keratinocytes. Initial experiments in which fragments of human skin were allowed to grow into artificial matrices produced constructs with mostly fibroblasts; additionally, these cultures were contaminated with T cells that were present in the skin samples.
We found the most successful approach was to expand the fibroblasts and keratinocytes separately and then combine them together onto artificial 3-dimensional Cellfoam cell growth matrices (Cytomatrix). Cellfoam is 3-dimensional tantalum-coated carbon matrix originally designed as an artificial bone matrix and used in previous xenogenic in vitro thymus explant cultures (Figure 1A) (8). Matrices seeded with keratinocytes and fibroblasts were cultured together using an intermediate cell culture medium until the cells reached confluence on the matrices (5–6 days), as assessed by immunofluorescence microscopy (IF). Human bone marrow–derived AC133+ hematopoietic precursors were then introduced into the colonized matrices. AC133+ input cell populations were at least 95% AC133+ and invariably negative for CD3+ T cells by flow cytometry analysis. These complete constructs were maintained in culture for 3–4 weeks in the presence of the prolymphopoietic cytokines IL-7 and IL-15, as well as Flt-3 ligand.
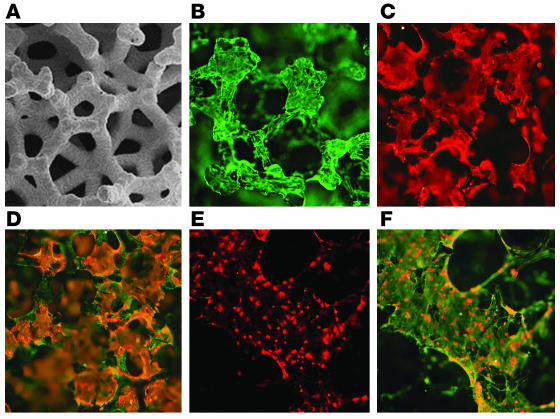
Figure 1.
Structure of 3-dimensional skin cell cultures. (A) Scanning electron micrograph of the Cellfoam 3-dimensional matrix. Image courtesy of Cytomatrix LLC. (B and C) IF demonstrating the morphologies of (B) fibroblasts stained with vimentin antibody and (C) keratinocytes stained with antibodies to cytokeratins when these cells were grown alone on matrices. (D) When grown together, keratinocytes (orange) and fibroblasts (green) occupied distinct sites on the matrices. (E) DCs, identified by intense staining with HLA-DR antibodies, were observed only if bone marrow progenitor cells were added to the matrices. (F) DCs were often found adherent to the surface of vimentin+ fibroblasts. Magnification: ×2 (E and F), ×5 (A), ×10 (B–D).
IF of the constructs showed that fibroblasts grew as adherent cells on the matrices and also formed thin cellular projections that spanned adjacent matrix components, whereas keratinocytes grew as flattened, nonstratified, plate-like cells (Figure 1, B and C). When cultured together, keratinocytes and fibroblasts occupied nonoverlapping sites on the matrices (Figure 1D). Viable populations of both keratinocytes and fibroblasts were present on the matrices after 3 weeks of culture in Iscove’s medium, a medium well suited to T cell development but not normally supportive of fibroblast and keratinocyte growth (Figure 1D).
A viable population of epithelial keratinocytes was found to be required for successful T cell development. IF was used to estimate the proportion of fibroblasts and keratinocytes occupying sites on the matrix. In a particular focal plane, the proportion of adherent cells staining for both vimentin and epithelial keratins (Figure 1D, orange) was estimated and compared to the proportion of cells staining only for vimentin (Figure 1D, green). Optimal T cell production was observed when keratinocytes composed at least 40% of the adherent cell types on the matrices. Multiple experiments (8 experiments, using 2 different skin donors and 4 different HPC donors) confirmed that T cells were never produced from HPCs in matrices colonized only with fibroblasts (data not shown). In contrast, matrices colonized with keratinocytes alone produced low numbers of T cells (mean, 9% of T cells produced from matrices containing both keratinocytes and fibroblasts and using identical skin and HPCs; n = 3; data not shown). However, keratinocytes grown in the absence of feeder fibroblasts were smaller and fewer in number by the end of the culture period. Thus, the lack of efficient T cell differentiation may be a result of low keratinocyte viability as opposed to an intrinsic inability of keratinocytes to support T cell development.
DCs, distinguished by their high expression of MHC class II and low expression of CD14, were demonstrable by IF only if HPCs were added to the construct, indicating that these cells are of bone marrow origin (Figure 1, E and F).
Characterization of T cells produced in the skin cell cultures.
Cells harvested from the constructs described above were analyzed by flow cytometry. Matrices colonized with both keratinocytes and fibroblasts robustly supported the development of cells that ultimately expressed T lymphocyte cell surface markers, including the CD3/T cell receptor complex. Optimal T cell production was observed when matrices contained at least 40–50% keratinocytes, as described above. From 1 × 104 precursor cells, individual 9-mm × 9-mm × 1.5-mm constructs produced 7.8 × 105 ± 2.4 × 105 total cells (mean ± SD; n = 6), 5–33% of which were CD3+ T cells. Individual constructs yielded between 4 × 104–1.7 × 105 T cells (mean, 7.9 × 104 ± 2.8 × 104; n = 6 representative experiments, each containing 12–40 individual matrices). In addition to T cells, we observed production of CD14loHLA-DRhi DCs (approximately 20–40%), CD14+ myeloid cells, and a variable number of CD56+ cells (data not shown).
We next studied the kinetics of T cell maturation. During development in the coculture system, progenitor cells initially expressed CD34 but lost expression of this marker as increasing levels of CD3 were acquired (Figure 2A). This loss of CD34 and gain of CD3 is also seen during normal T cell development in the thymus (21). Progenitor cells in our system were initially negative for CD3, CD4, and CD8. Transient production of double-positive CD4+CD8+ T cells expressing CD3 was noted between days 7 and 12 of culture; after day 14 of culture, double-positive cells were not detectable. By day 21, single-positive CD4+ and CD8+ T cells were present (Figure 2A). Normally developing thymocytes also progress from double-negative CD4–CD8– cells to double-positive CD4+CD8+ cells and subsequently to mature single-positive CD4+ or CD8+ cells (21). Double-positive CD4+CD8+ T cells, demonstrably present in our skin cell construct, are a cell population normally found exclusively in the thymus (21). By varying the culture conditions, we were able to influence the final production of CD4+ versus CD8+ cells. Irradiation of the construct prior to precursor addition, performed initially to prevent stromal overgrowth, produced larger numbers of single-positive CD4+ T cells (Figure 2B; duplicate conditions shown in Figure 2A). Pretreatment of the construct with IFN-γ prior to the addition of HPCs, performed initially to increase MHC class II expression by epithelial cells, led to increased production of single-positive CD8+ cells (Figure 2C). A more equal distribution of CD4+ and CD8+ cells was produced in the absence of these treatments (Figure 2D). Production of double-positive intermediate CD4+CD8+ cells was similar under all 3 culture conditions (data not shown).
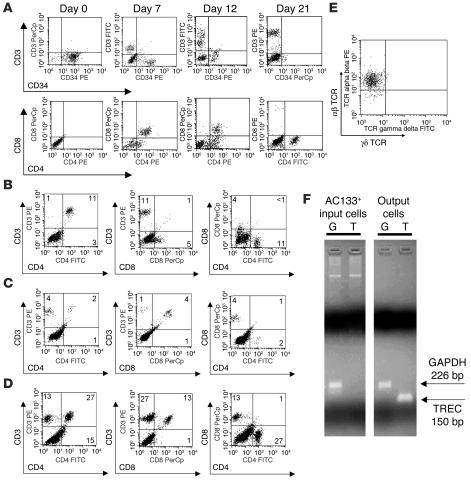
Figure 2.
HPCs differentiate into T cells in skin cell cultures. (A) Maturation of surface markers during T cell development in skin cell cultures. Matrices were irradiated prior to the addition of HPCs and produced primarily CD4+ T cells. (B–D) Alterations in culture conditions influenced the production of CD4+ versus CD8+ cells. (B) Irradiation of the construct prior to the addition of HPCs supported differentiation of proportionately more CD4+ cells. (C) Treatment of the skin cell construct with IFN-γ before the addition of HPCs increased the percentage of CD8+ cells produced. (D) In the absence of these treatments, a more equal distribution of CD4+ and CD8+ cells was observed. Cells were harvested from the matrices at 28 days. Similar results were seen in duplicate experiments. The percentage of cells in each quadrant is shown. (E) Expression of αβ TCR versus γδ TCR by CD3+ cells produced in skin cell cultures. (F) Qualitative RT-PCR for expression of TREC (T) and GAPDH (G) of input AC133+ HPCs and output T cells produced in skin cell cultures.
Skin and gut have been reported to support the development of T cells with the γδ TCR, whereas αβ T cells are primarily produced by the thymus (22). We therefore examined newly generated T cells for expression of the αβ versus γδ TCR. Flow cytometry analysis showed that consistently more than 95% of the CD3+ cells produced in our construct were αβ TCR T cells (Figure 2E).
Newly produced T cells contain T cell receptor excision circles.
The T cells observed in our cocultures could have arisen from either de novo T cell differentiation or simply from expansion of very low numbers of mature T cells contaminating the skin cell or HPC populations. To test whether the T cells produced in our system are in fact naive, newly produced T cells, we tested output cells from our construct for the presence of T cell receptor excision circles (TRECs). TRECs, episomal circles of DNA that are produced as a byproduct of the recombination of TCR genes (23), are found in mature thymocytes and naive T cells recently released from normal thymus. Proliferation of T cells in the periphery leads to dilution of TRECs, because these DNA episomes do not replicate during cell division. TRECs have therefore been used to identify naive T cells and recent thymic emigrants (23). We performed RT-PCR analysis on both input HPCs and lymphocytes produced in the construct. Figure 2F shows the absence of TRECs in the HPC population (AC133+ input cells) and the presence of TRECs in lymphocytes produced in the construct. These findings indicate that a subset of HPCs underwent rearrangement of the TCR genes during coculture with skin cells, as would be expected during normal T cell development.
Newly produced T cells express a diverse TCR repertoire.
To examine the TCR diversity of cells produced in the thymus construct, newly produced T cells were subjected to spectratyping via TCR-CDR3 length analysis. This technique allows identification of Vβ usage as well as diversity within each Vβ family. The number of peaks within a Vβ family is an index of the complexity of the TCR repertoire utilizing that Vβ. Two spectratypes are shown (Figure 3), both derived from the same HPC inoculum but differentiated in the presence of skin cells from 2 different donors. T cells produced in skin cell cultures from the first donor were significantly more diverse (Figure 3A). T cells of all 26 Vβ subfamilies tested were represented in the small population of newly generated T cells analyzed. Fully 85% of these Vβ families (22 of 26) displayed significant TCR diversity. T cells produced in cultures from the second skin donor were less diverse, suggesting that skin donors may vary in their ability to support diverse T cell differentiation (Figure 3B).
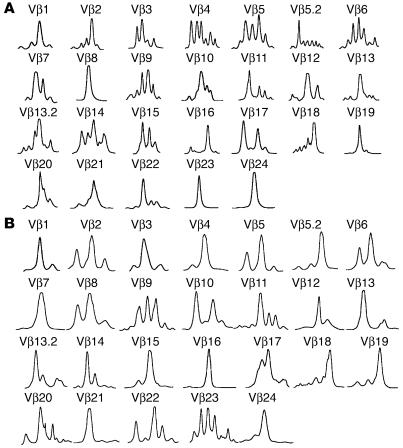
Figure 3.
Spectratype analysis of T cells produced in skin cell cultures. T cells generated in skin cell cultures were subjected to TCR-CDR3 length analysis. Diversity within each Vβ family is signified by multiple peaks. Precursor cells from 1 bone marrow donor were matured in skin cell cultures from 2 different skin donors. T cells produced in cultures from the first skin donor (A; 8 × 105 cells analyzed) were more diverse and had a different T cell repertoire than T cells produced in cultures from the second skin donor (B; 5 × 105 cells analyzed).
Epithelial cells mediate positive selection in the thymus, thereby determining the TCR repertoire of developing thymocytes (22). If epithelial cells in skin cell cultures are mediating positive selection, different skin donors would be expected to produce different patterns of oligoclonality. We have found that T cells developing from the same HPC inoculum in cultures using different skin cell donors do have differing repertoire patterns. For example, the first skin donor we tested produced clonal populations in subfamilies Vβ8, Vβ19, Vβ23, and Vβ24, and the second produced completely clonal populations in a distinct subset (Vβ7, Vβ21, and Vβ16; Figure 3, A and B). Also, the greatest diversity was seen in largely nonoverlapping subsets of Vβ subfamilies: Vβ4, Vβ5, Vβ6, and Vβ9 versus Vβ9, Vβ11, Vβ22, and Vβ23. Thus, not only does the ability to support diverse T cell production vary among donors, but T cells from different Vβ subfamilies are preferentially produced from different skin cell donors.
Newly produced T cells are mature and functional.
Mature and functional T cells are distinguished by their ability to proliferate, express the activation antigen CD69, and produce cytokines in response to stimulation through the TCR-CD3 complex. To determine whether the T cells produced in our system were mature and functional, we evaluated the response of these cells to T cell mitogens and alloantigens. T cells produced in the thymus construct proliferated in response to treatment with phytohemagglutinin (Figure 4, A and B). Proliferation was comparable to that seen using PBLs (construct-derived T cells, 28% ± 2.6%, versus PBLs, 34% ± 7.9%, mean ± SD, n = 3; PBL data not shown). Newly produced CD4+ and CD8+ single-positive T cells stimulated with concanavalin A expressed robust levels of the early activation marker CD69 (Figure 4, C–F). CD69 upregulation of newly produced T cells was also comparable to results obtained using PBLs (construct-derived T cells, 82% ± 2.6%, versus PBLs, 89% ± 2.9%; PBL data not shown). A subset of these activated cells also produced IL-2, TNF-α, and IFN-γ, as demonstrated by intracellular flow cytometry (Figure 4, G–J). Significant proliferation was also seen in response to allogeneic stimulator cells in mixed leukocyte reactions (MLRs; Figure 4K). Newly generated T cells proliferated in response to allogeneic stimulator cells at levels comparable to those seen in mature T cells isolated from peripheral blood, providing additional evidence for the functional maturity of these cells.
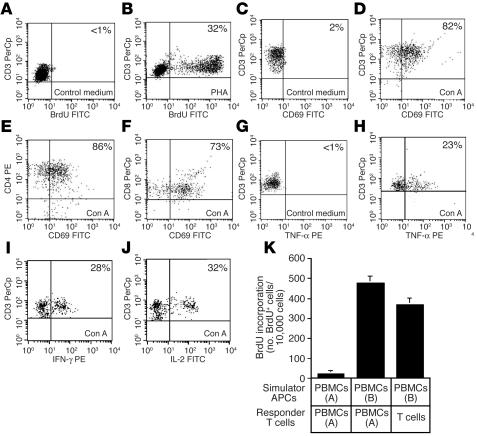
Figure 4.
T cells produced in skin cell cultures are mature and functional. (A and B) Proliferation of T cells in response to (A) control medium and (B) phytohemagglutinin (PHA). (C–F) Expression of CD69 activation antigen in response to (C) control medium and (D–F) concanavalin A (Con A) by (C and D) total T cells and (E) CD4+ and (F) CD8+ subsets. (G and H) Production of TNF-α by CD3+ cells in response to (G) control medium and (H) concanavalin A treatment. (I and J) Production of (I) IFN-γ and (J) IL-2 in response to concanavalin A treatment. Treatment with control medium is not shown for IFN-γ and IL-2 samples but was identical to TNF-α control shown in G. The percentage of positive cells is shown. (K) Proliferation of T cells generated in skin cell cultures (T cells) in response to allogeneic PBMCs. Proliferation was assayed by BrdU incorporation on day 6 of the MLR. BrdU incorporation was detected via flow cytometry with gating on CD3+ T cells. PBMCs were derived from allogeneic, unrelated donors [PBMCs (A), donor A; PBMCs (B), donor B]. Error bars represent the SD of 3 measurements.
Delta-like Notch ligands and T cell production.
The Notch ligand delta-like 1 has been shown to enhance T cell development via maintenance and expansion of progenitor cells, induction of lymphoid differentiation at the expense of myeloid differentiation, and the biasing of lymphocyte production toward T and away from the B lineage (24–26). OP9 monolayers expressing delta-like 1 support the full development of murine CD8+ T cells, but support human T cell development only to the double-positive stage (27, 28).
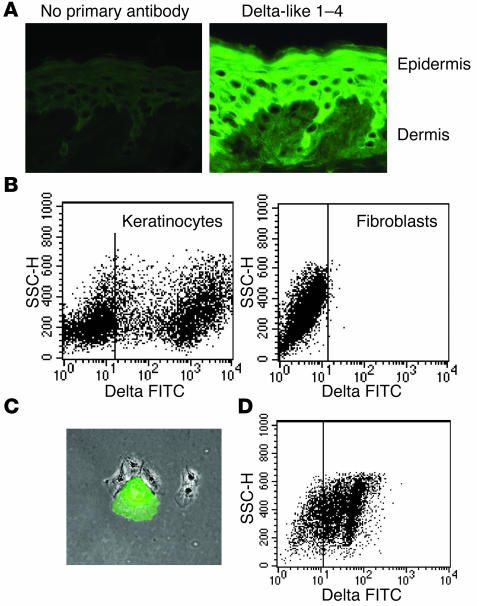
In order to examine skin cells for the presence of delta-like ligands, we stained human skin and cultured skin cells with an antibody that recognizes epitopes common to human delta-like 1–4. Suprabasal keratinocytes expressed delta-like Notch ligands (Figure 5A), and more than 50% of keratinocytes freshly disaggregated from skin expressed high levels of delta-like ligands (Figure 5B). Keratinocytes grown in low-density cultures were a mix of basaloid, small proliferating cells that did not express delta-like ligands and larger, differentiating cells that expressed high levels of delta-like molecules (Figure 5C). Culture of keratinocytes under high-density conditions that encouraged differentiation and stratification induced expression of delta-like ligands. Greater than 50% of keratinocytes grown in high-density cultures expressed delta-like ligands by day 6 after confluence of the cultures (Figure 5D). These cells maintained delta-like expression when subsequently plated on matrices (data not shown). In contrast, fibroblasts in the dermis of human skin and fibroblasts grown in culture did not express delta-like ligands (Figure 5, A and B).
Figure 5.
Expression of delta-like Notch ligands in human keratinocytes. (A) Sections of normal human skin stained for delta-like Notch ligands demonstrated abundant staining of suprabasal epidermal keratinocytes but no staining of dermal fibroblasts. Magnification: ×10. (B) Keratinocytes freshly isolated from skin expressed delta-like ligands, but fibroblasts lacked expression. SSC-H, side scatter. (C) Cultured keratinocytes contained a mixture of large differentiating cells that expressed high levels of delta-like Notch ligands (green) and smaller, basaloid cells that were negative for delta-like ligand expression. Magnification: ×4. (D) Keratinocytes cultured under high-density conditions that encouraged differentiation expressed delta-like ligands.
T cells undergo negative selection in the construct, rendering them tolerant to self MHC.
Negative selection during intrathymic development leads to clonal deletion of autoreactive T cells, a process mediated most efficiently by bone marrow–derived DCs (22). In our skin cell cultures, we observed that DCs were produced in parallel with T cells. Many of these cells, distinguished by high HLA-DR expression and low CD14 expression, were adherent to the surface of fibroblasts in the constructs (Figure 1, E and F). Thus, developing T cells likely come into contact with autologous DCs during development in the skin cell cultures. We performed a series of MLRs to determine whether these DCs could induce tolerance in developing T cells. DCs, either autologous or allogeneic, were produced from different bone marrow donors and used as stimulator cells in the MLRs.
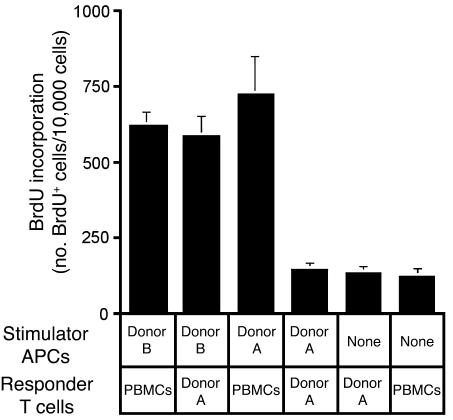
DCs were produced by culturing HPCs for 21 days in the presence of matrices colonized only with dermal fibroblasts. These cultures produced a combination of monocytes (CD14hiHLA-DRlo) and DCs (CD14loHLA-DRhi), in agreement with a previous report that dermal fibroblasts support the differentiation of monocytes and DCs from HPCs (29). These cells were cultured for 5 days in GM-CSF and IL-4, then matured for 48 hours with LPS. Output cells were at least 80% mature myeloid-lineage DCs (CD14loHLA-DRhiCD86hi). DCs were irradiated and added to cultures of T cells generated in skin cell cultures from autologous and allogeneic bone marrow samples. T cells produced in the skin construct proliferated in response to allogeneic DCs but failed to respond to autologous DCs (Figure 6). PBMCs proliferated strongly in response to DCs from both donors. These results show that T cells developing in the skin cell construct do not proliferate in response to self MHC and other antigens carried by the DCs, providing evidence that negative selection occurs within the skin cell culture microenvironment.
Figure 6.
Newly generated T cells are tolerant to challenge with autologous, but not allogeneic, DCs. T cells produced in skin cell cultures from HPCs of bone marrow donor A were exposed to donor A–derived DCs during the process of T cell development. Donor A–derived lymphocytes were therefore examined for their ability to respond to DCs derived from the same donor (donor A) and a second, unrelated donor (donor B) in the MLR. Responses of PBMCs to the DCs of both donors are included to demonstrate the immunogenicity of both DC populations. Proliferation was assayed by BrdU incorporation on day 6 of the MLR. BrdU incorporation was detected via flow cytometry with gating on CD3+ T cells. PBMCs were derived from a third, unrelated donor. Error bars represent the SD of 3 measurements. A duplicate set of experiments produced similar results.
Expression of the AIRE gene and autoantigens.
The thymus also has the ability to eliminate T cells reactive to proteins normally expressed only in specific peripheral organs. Expression of the AIRE transcription factor induces a subset of medullary epithelial cells to express high levels of proteins normally limited to specialized tissues, and this expression has been shown to be crucial for the removal of autoreactive T cells (30, 31).
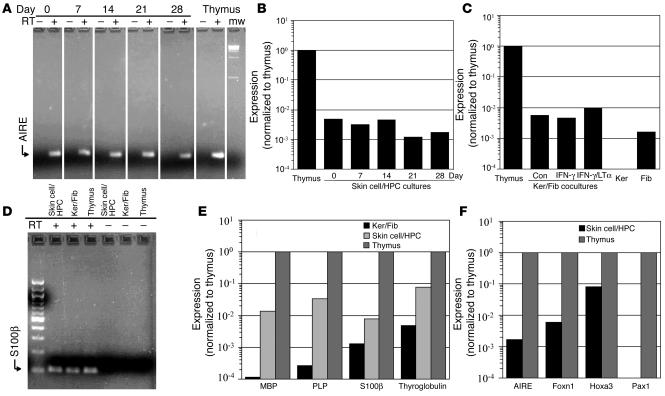
We tested skin cell cultures for expression of AIRE by real-time RT-PCR and found that combined HPC/skin cell cultures expressed AIRE, although at significantly lower levels than in the thymus (Figure 7, A and B). Expression was fairly constant during the 28-day culture period (Figure 7B). DCs in the thymus also express AIRE and a panel of autoantigens (32, 33). In order to determine whether AIRE was expressed by skin or hematopoietic cells, we next tested cultures of keratinocytes and fibroblasts grown on 3-dimensional matrices in the absence of HPCs. We found that mixed cultures of fibroblasts and keratinocytes also expressed AIRE (Figure 7C). Lymphotoxin-α (LTα) induces the expression of the AIRE gene both in intact mouse thymus and in thymic epithelial cell lines (34). LTα mediates its effect by binding to the lymphotoxin-β receptor (LTβR), and this receptor is upregulated by IFN-γ (35). Keratinocytes express LTα and upregulate LTβR after treatment with IFN-γ) (36). We observed a modest increase in AIRE expression when cocultures of keratinocytes and fibroblasts were pretreated with IFN-γ and stimulated with recombinant LTα (Figure 7C). To determine whether fibroblasts or keratinocytes expressed AIRE, we tested cells that were grown separately on 3-dimensional matrices. AIRE is expressed by several types of epithelial cells in mice, including Fallopian tube, kidney, and large airway epithelia (37). We were therefore surprised to find that AIRE was expressed by dermal fibroblasts, not by keratinocytes (Figure 3C). To our knowledge, this is the first report of AIRE expression in cells from the skin.
Figure 7.
Skin cell cultures express autoantigens and transcription factors important for thymic and T cell development. (A and B) Skin cell/HPC cultures express AIRE. Equal amounts of mRNA derived from skin cell/HPC cultures and from normal human thymus were analyzed by real-time RT-PCR for AIRE expression in the presence (+) or absence (–) of RT. RNA from skin cell–colonized matrices was isolated on days 0, 7, 14, 21, and 28 after the addition of HPCs. Signal for each sample was first normalized to cyclophilin A expression and then normalized to thymus expression. mw, molecular weight markers (100-kb ladder). (C) Fibroblasts in skin cell cultures express AIRE. AIRE expression was assayed by real-time RT-PCR in keratinocyte (Ker) and fibroblast (Fib) cocultures without HPC and in cultures of keratinocytes or fibroblasts alone. Combined cultures of skin cells were assayed after treatment with control medium (Con), IFN-γ, or IFN-γ followed by LTα stimulation. Signal for each sample was normalized to cyclophilin A and thymus expression. (D and E) Skin cell cocultures with and without HPC express the autoantigens MBP, PLP, S100β and thyroglobulin by quantitative real-time RT-PCR. RNA from skin cell/HPC cocultures harvested on day 14, matrices colonized with keratinocytes and fibroblasts alone, and thymus was analyzed in the presence (+) or absence (–) of RT. Signal for all samples was normalized to cyclophilin A and thymus expression. No product was detected in samples without RT. (F) Skin cell/HPC cultures express Foxn1 and Hoxa3, but do not express Pax1. Day-28 skin cell/HPC cultures were analyzed by real-time RT-PCR. Samples without RT had no signal in all samples. Expression was normalized to cyclophilin A, and expression of each factor was then normalized to thymus expression.
We observed relatively low levels of AIRE expression in skin cell cultures. To determine whether this expression induces autoantigen expression, we tested skin cell/HPC cultures by quantitative real-time RT-PCR for a panel of noncutaneous autoantigens which included myelin basic protein (MBP), myelin phospholipid protein (PLP), S100β, and thyroglobulin. These 4 autoantigens were detected in both human thymus and in day-14 combined skin cell/HPC cultures (Figure 7, D and E). By day 14, DCs were likely present in skin cell/HPC cultures. Thymic DCs are known to express AIRE and a subset of autoantigens (38). To determine whether skin cells alone expressed autoantigens, we tested cocultures of fibroblasts and keratinocytes in the absence of HPCs. We found that these cultures expressed lower, but detectable, levels of all 4 autoantigens (Figure 7E).
Skin cell cultures express the thymic transcription factors Hoxa3 and Foxn1.
Mutations in several transcription factors, including Foxn1, Hoxa3, and Pax1, result in impaired development or function of the thymus. Foxn1 is critical for the differentiation and survival of thymic epithelial cells. Foxn1 deficiency induces the nude phenotype in mice, rats, and humans, characterized by hairlessness and congenital athymia (39). Nude mice have a rudimentary thymus that lacks all types of thymic epithelial cells and cannot support T cell differentiation. We found significant expression levels of Foxn1 in skin cell/HPC cultures by real-time RT-PCR (Figure 7F). This result is consistent with the fact that Foxn1 is also critical for differentiation of epithelial cells in the skin (40). Foxn1-deficient mice have abnormal keratinocyte maturation and produce fragile hairs with altered keratin composition. Transient Foxn1 expression has recently been shown induce a transcriptional program leading to the differentiation of epidermal keratinocytes (41).
Hoxa3, a member of the Hox family of transcription factors, is critical for the development of structures in the pharyngeal region. Hoxa3-deficient mice are athymic and also lack parathyroid glands (42). We found significant levels of Hoxa3 expression in skin cell/HPC cultures (Figure 7F). This is consistent with the finding that Hoxa3 also plays a role in keratinocyte biology. Hoxa-3 is expressed by keratinocytes and promotes migration of these cells during wound healing (43).
Deficiency of the Pax1 transcription factor has more subtle effects on thymic function than Foxn1 and Hoxa3 deficits. Pax1-deficient mice have a hypoplastic thymus that is less efficient at supporting T cell development (44). In contrast to our findings with Foxn1 and Hoxa3, we found no evidence of Pax1 expression in skin cell/HPC cultures by real-time RT-PCR (Figure 7F).
Discussion
In this report, we present evidence that keratinocytes and fibroblasts from normal human skin can support the full process of human T cell development. T cells produced in these skin cell cultures are mature, diverse, functional, and tolerant to self MHC. We have several lines of evidence to support our contention that we are observing de novo T cell production as opposed to the expansion of small numbers of mature T cells contaminating the input cell population. First, we have used highly purified HPCs that showed no evidence of T cell contamination by flow cytometry analysis and were consistently negative for TRECs by RT-PCR. If the input precursor cell populations were contaminated with mature T cells at levels too low to detect by flow cytometry or RT-PCR, we would expect that the spectratype analysis of the output cells would contain clonal populations in only a few Vβ subfamilies. Instead, we found significant TCR diversity in T cells produced in skin cell cultures. Second, we saw clear production of double-positive T cell precursors before the appearance of mature T cells. Third, we routinely included control experiments in which only fibroblasts were used to colonize the matrices. These experiments used identical precursor cell and skin cell populations and the same cytokines. Using our highly purified, TREC-negative bone marrow HPCs, we did not see production of T cells in fibroblast-only cultures. If small numbers of contaminating mature T cells were expanding and giving rise to the T cells we observed, we should have observed these T cells in fibroblast-only cultures as well.
During the process of positive selection, developing T cells die after completion of TCRα rearrangements unless they are rescued by a low-affinity interaction of the TCR complex with MHC antigens expressed on thymic epithelial cells (22). Successful completion of positive selection is an absolute requirement for the eventual production of single-positive CD4+ and CD8+ cells (22). We observed in our system the initial production of double-positive CD4+CD8+ T cells followed by the appearance of single-positive CD4+ or CD8+ T cells. The production of single-positive, mature T cells in our system provides evidence that the skin-derived construct is capable of supporting the positive selection of human lymphocyte precursor cells.
It is informative to compare our results to recent reports using monolayers of mouse bone marrow stromal OP9 cells transduced with delta-like 1 (OP9-DL1) (28). These cells support the full development of mouse CD8+ T cells, but human cord blood precursors undergo T cell development only to the double-positive stage; further development appears to be blocked (27, 28). Transfer of these double-positive cells to human fetal thymus organ cultures permits further development to the single-positive stage. This arrest at the double-positive stage likely indicates an inability of OP9-DL1 cells to support positive selection. OP9-DL1 cells lack human MHC molecules, and this has been postulated to explain their inability to support human positive selection (45). However, this appears unlikely, because other monolayer and 3-dimensional cultures of thymic cells from mice and nonhuman primates support full development of human T cells to the mature single-positive stage, despite the fact that these tissues do not express human MHC molecules (8–10, 46). Res et al. have specifically shown that murine MHC molecules support the positive selection of human T cells (46). Therefore, although MHC molecules of some type are required for positive selection of human T cells, they are not necessarily sufficient. The inability of OP9-DL1 cells to support human single-positive CD8+ T cell development demonstrates a lack of additional factors that are present on thymic stromal cells from many species but are apparently absent on OP9 cells. We found that human skin cells supported positive selection of human T cell precursors, suggesting that these critical factors are shared between thymus and skin cells.
Negative selection is a crucial step in lymphocyte development that leads to the clonal deletion of cells with a high avidity for self-MHC molecules. DCs of bone marrow origin are the most efficient mediators of this process (22). In mouse models that have impaired negative selection in the thymus, autoreactive cells are released into the periphery and cause a variety of autoimmune disorders (30, 47). Generation of T cells in skin cell cultures would be less useful clinically if defective negative selection allowed the production of autoreactive T cells. Unlike positive selection, negative selection is not an obligate step in lymphocyte development (48). It was therefore important to determine whether skin cell cultures supported the process of negative selection. MLR studies, although not the most sensitive assay for reactivity, demonstrated clearly that newly generated T cells did not proliferate in response to DCs derived from autologous bone marrow precursors, but reacted normally to DCs derived from allogeneic, unrelated donors. These results provide strong evidence that T cells developing in skin cell cultures are tolerized to the repertoire of self proteins expressed and presented by DCs.
One remarkable feature of the thymus is its ability to eliminate T cells reactive to a variety of proteins normally expressed only in specific peripheral organs. Specialized antigens from these peripheral organs are expressed by a subset of medullary thymic epithelial cells under the control of the AIRE transcription factor. AIRE deficiency leads to autoimmune polyendocrinopathy syndrome type 1 in humans and to a group of autoimmune disorders in mice, demonstrating the importance of this expression (30). We found that dermal fibroblasts in skin cell/HPC cultures expressed AIRE. Moreover, skin cell/HPC cultures expressed a panel of noncutaneous autoantigens, including MBP, PLP, S100β, and thyroglobulin. Skin cells cultured in the presence of HPCs expressed higher levels of autoantigens than skin cells alone, although AIRE expression was comparable between the 2 groups (Figure 7, B–E). DCs express AIRE, and thus the DCs generated from HPCs in skin cell/HPC cultures may contribute to autoantigen expression. However, human DCs express only a subset of the autoantigens expressed in the thymus (38). Thymic DCs express MBP and S100β, but do not express PLP or thyroglobulin (38). The enhanced expression of PLP and thyroglobulin in skin cell/HPC cultures suggests that coculture with HPCs enhances the ability of human skin cells to express autoantigens.
Skin cell cultures also expressed Foxn1 and Hoxa3, 2 transcription factors crucial for the development and function of the thymus. Foxn1 and Hoxa3 are also implicated in the control of important aspects of cutaneous biology. Foxn1 regulates the differentiation of epidermal keratinocytes in an analogous fashion to its role in promoting the survival and differentiation of thymic epithelial cells (40). Hoxa3 mediates the migration of keratinocytes during wound repair and the development and migration of cells that give rise to the thymus (49). The analogous roles for these transcription factors in the thymus and in the skin are further examples of the remarkable similarities between these 2 tissues.
We have found that keratinocytes present in skin cell cultures are a rich source of delta-like Notch ligands, which are crucial for early T cell development. Keratinocyte expression of delta-like Notch ligands may partially explain the ability of skin cell cultures to support T cell development and likely underlies our finding that a viable population of keratinocytes was required for successful T cell differentiation. Different members of the delta-like ligand family are redundant in their ability to support T cell development. Both delta-like 1 and delta-like 4 can support T cell differentiation, and mice in which delta-like 1 is deleted after birth have normal T cell development in the thymus (50). In situ hybridization has demonstrated that delta-like 1 is expressed by epidermal stem cells, located in the basal layer of the epidermis (51). No expression of delta-like 1 was found in the suprabasal layers of the epidermis. We observed the highest levels of immunostaining in suprabasal keratinocytes, and this expression correlated with the ability of keratinocytes to support T cell differentiation. We therefore predict that the delta-like ligand we have identified on suprabasal keratinocytes is distinct from delta-like 1 and is most likely delta-like 4.
T cells produced in the skin cell cultures exhibited significant TCR diversity. Spectratype analysis of newly generated T cells showed that all 26 Vβ families analyzed were represented. In the spectratype from donor A, 85% of these Vβ families also had significant complexity. However, 4 of the 26 Vβ families (15%) had clonal or nearly clonal populations. We found that different skin donors varied in their ability to support the development of diverse T cell populations. It is likely that epithelial cells in skin cell cultures support positive selection, as they do in the thymus. The spectratypes reported in this manuscript were derived from T cells differentiating in the presence of untreated keratinocytes. Future experiments are needed to determine whether keratinocytes treated with agents that upregulate MHC II, such as IFN-γ, give rise to more diverse T cell populations. It therefore remains to be seen if the more limited repertoire we have observed in skin cultures can be improved upon or if it represents a fundamental property of skin cell cultures.
Given the potential clinical applications of newly produced autologous T cells, it is not surprising that significant efforts have been directed toward replicating the thymus microenvironment in vitro. Human T cell differentiation has been supported by a variety of systems that use xenogenic tissues or human fetal thymus (4–16, 27, 28). These systems have provided valuable information about the process of T cell development, but their use of tissues from animals or unrelated human donors makes them less suitable for the production of T cells to be administered to human patients.
The 2 main obstacles to producing T cells for patients are the need for these cells to be self-restricted and tolerant to human antigens. Xenogenic systems, such as OP9-DL1 monolayers, lack human MHC molecules and thus do not support the development of T cells that recognize antigen in the context of human MHC (27, 28). Cells in xenogenic systems could be stably transduced with human MHC molecules to enable them to produce T cells that are restricted to human MHC. However, each recipient patient has a unique repertoire of MHC molecules, and T cells selected on a different set of MHC are likely to be poorly functional and may be autoreactive. The method we describe supports T cell generation using the patient’s own skin cells to replicate the thymus microenvironment. These cells express the full complement of MHC molecules unique to an individual and therefore produce self-restricted T cells without the need for exogenous introduction of MHC genes. Moreover, they can be grown under well-established GMP conditions that may eventually allow newly generated T cells to be administered to patients.
In addition to self restriction, it is imperative that T cells for use in patients are self tolerant both to self-MHC molecules and to the broad repertoire of self antigens present in peripheral organs. It is possible that partial tolerance to human self antigens could be accomplished even in xenogenic systems. There are likely to be DCs produced in parallel with T cells that could induce tolerance to DC antigens, as we have observed in our system. However, tolerance to specialized tissue antigens is unlikely to be accomplished in systems that use animal tissues. Introduction of the AIRE gene into xenogenic cells would lead to expression of specialized species-specific antigens that are significantly different from analogous genes in humans. In contrast, the skin cell system we describe has the potential to produce self-restricted and self-tolerant T cells because all tissues used are human and autologous. Moreover, skin and bone marrow are accessible tissues that can be obtained from adult donors with minimal morbidity.
The ability to produce autologous human T cells using accessible adult human tissues could have a significant impact on the treatment of immunodeficiencies, malignancies, autoimmune diseases, and possibly transplant rejection. Patients who have lost peripheral T cells from HIV, malignancy, or recent bone marrow transplantation are susceptible to infection and some opportunistic malignancies, often with devastating results (52). Although methods exist for the in vitro expansion of pre-existing T cells, there are currently no methods that allow production of naive autologous T cells. A method such as we describe could be used to generate large numbers of naive T cells for use in these patients. T cells with engineered specificities could also be generated by transduction of stem cells with TCR genes or constructs followed by maturation in skin cell cultures. CD4+ and CD8+ T cells specific for tumor- or infection-associated antigens could be generated using this approach, and these cells could be used as immunotherapy for malignancies and infections. Tregs generated in skin cell cultures could be used for the suppression of autoimmune diseases. Lastly, the implantation of a skin construct colonized with the skin and DCs from an organ donor could potentially be used to tolerize an individual to an allograft in the manner that murine thymus grafts have been used to tolerize recipient pigs to grafts of mouse skin (53).
In conclusion, we present evidence that biopsies of human skin and bone marrow can provide the basis for the generation of a new, patient-specific T cell repertoire. T cells generated in skin cell cultures are diverse, mature, functional, and tolerant to self MHC. Ongoing studies in our laboratory are focused on the goal of refining a system that will allow the production of T cells for the purpose of treating human disease.
Methods
Isolation and culture of keratinocytes and fibroblasts.
Normal human skin was obtained with institutional review board approval from patients undergoing cosmetic procedures or excision of skin cancers and was divided into 6-mm × 6-mm portions. Acquisition of skin samples and all scientific studies were approved by the Institutional Review Board, Partners Human Research Committee (Partners Research Management, Boston, Massachusetts, USA). Keratinocytes were isolated by incubation of skin fragments in 2.4 U/ml dispase II (Roche Diagnostics Corp.) for 1 hour at 37°C and subsequent removal of the epidermal sheet with tweezers, followed by incubation for 5 minutes at 37°C in PBS containing 0.25% trypsin and 0.1% EDTA. Keratinocytes were grown in Keratinocyte-SFM (Gibco; Invitrogen Corp.) supplemented with 25 μg/ml bovine pituitary extract (Gibco; Invitrogen Corp.), 0.2 ng/ml EGF (Gibco; Invitrogen Corp.), 0.3 mM CaCl2, and 100 IU/ml penicillin and 100 μg/ml streptomycin. Keratinocytes were maintained at less than 30% confluency in this medium. Fibroblasts were isolated by mincing of dermal skin fragments and incubation in HBSS with 2.5 mg/ml trypsin and 5 mg/ml collagenase for 1 hour. Fibroblasts were cultured in a 1:1 mixture of DMEM with Ham’s F-12 (Gibco; Invitrogen Corp.) supplemented with 15% FCS (Sigma-Aldrich), 10 ng/ml EGF, and penicillin/streptomycin. Keratinocytes and fibroblasts were passaged fewer than 5 times before use in experiments.
High-density culture of keratinocytes.
To encourage differentiation and upregulation of delta-like ligands, keratinocytes were grown in high-density cultures as follows: keratinocytes were grown as described above in supplemented Keratinocyte-SFM until they reached 30% confluence. At this point, cells were transferred to a 1:1 (vol/vol) mix of complete Keratinocyte-SFM (supplemented as above) with DF-K medium. DF-K medium is composed of a 1:1 (vol/vol) mix of calcium-free, glutamine-free DMEM (Gibco; Invitrogen Corp.) with Ham’s F12 (Gibco; Invitrogen Corp.) supplemented with 0.2 ng/ml EGF, 25 μg/ml bovine pituitary extract, 1.5 mM L-glutamine, and penicillin/streptomycin. Cells were fed daily when maintained at high densities in this medium.
The skin cell construct.
9-mm × 9-mm × 1.5-mm Cellfoam matrices (Cytomatrix) were incubated in a solution of 100 μg/ml rat tail collagen I (BD Biosciences) in PBS for 30 minutes at 37°C and rinsed twice in PBS. Keratinocytes (1 × 105) and fibroblasts (5 × 104) were added to each matrix and incubated for 4 hours at 37°C, 5% CO2. Nonadherent cells were removed by transferring the matrices to new 24-well plates. Culture medium (2 ml) was added to each matrix and consisted of a 1:1 mix of the fibroblast and keratinocyte media described above. The constructs were then cultured for 6 days, and the medium was changed every other day. Irradiated matrices were treated with 3,000 rad on day 6. For IFN-γ treated matrices, 50 ng/ml IFN-γ (R&D Systems) was included in the culture medium from plating until day 6. On day 7, 10,000 normal human bone marrow AC133+ precursor cells (AllCells) were added to each matrix, and the unit was cultured in Iscove’s modified medium (Mediatech) with 10% heat-inactivated FCS (Sigma-Aldrich), 20 ng/ml IL-7, 20 ng/ml IL-15, 100 ng/ml Flt-3 ligand (R&D Systems), and penicillin/streptomycin. One-half of the medium was aspirated and replaced 3 times weekly, and the culture was maintained for 3–4 weeks.
IF and flow cytometry analysis.
IF of thymus constructs was performed by rinsing the constructs in PBS, followed by fixation in 100% acetone for 5 minutes and rehydration in PBS. Matrices were then incubated with mixed monoclonal mouse anti-pan cytokeratin ascites (1:50 dilution; Sigma-Aldrich) and goat anti-vimentin antiserum (1:20 dilution; Sigma-Aldrich) in PBS plus 1% BSA for 45 minutes at room temperature. Matrices were rinsed twice and incubated in secondary antibodies FITC-conjugated rabbit anti-goat IgG (Zymed Laboratories Inc.) and Rhodamine Red-X–conjugated rabbit anti-mouse IgG (Jackson ImmunoResearch Laboratories Inc.) for 30 minutes at room temperature. Matrices were then mounted on microscope slides using Prolong Antifade Kit (Invitrogen Corp.) and examined by fluorescence microscopy. IF of frozen human skin sections was performed by fixation of sections in 100% acetone for 5 minutes, incubation with polyclonal rabbit anti-human delta (H-265, 1:100 dilution; Santa Cruz Biotechnology Inc.), and rinsing in PBS plus 1% BSA, followed by staining with goat anti-rabbit FITC (1:100 dilution; Zymed Laboratories Inc.) and examination under the fluorescent microscope. Flow cytometry analysis of T cells was performed using directly conjugated monoclonal antibodies (BD Biosciences) or with the anti-delta antibodies described above. αβ TCR antibody was purchased from BD (catalog no. 555548), and γδ TCR antibody was purchased from Beckman Coulter (catalog no. IM1571). Analysis of flow cytometry samples was performed on a BD FACScan instrument.
TREC analysis.
DJ signal join–type TRECs were assayed. DNA was isolated from 1 × 105 cells per the manufacturer’s protocol (QIAamp DNA Blood Mini Kit; QIAGEN). DNA (0.5 μg) was used in each GAPDH or TREC PCR reaction as previously described (23).
TCR spectratype analysis.
TCR-CDR3 length analysis was performed as previously described (54). Briefly, total RNA was isolated from T cells (SV Total RNA Isolation System; Promega) and reverse transcribed into cDNA (PowerScript Reverse Transcriptase; BD Biosciences). PCR reactions were performed using Cβ primers recognizing both the Cβ1 and Cβ2 regions and individual primers for the 26 TCR Vβ chains, as described previously (54). Additional runoff reactions were performed using fluorophore-labeled primers, and labeled products were analyzed using a 377 DNA Sequencer (Applied Biosystems) and GeneScan software version 3.0 (Applied Biosystems) (54).
T cell functional assays.
To assess mitogen-induced proliferation, cells produced in the thymus construct were incubated in RPMI plus 10% FCS for 72 hours with and without 5 μg/ml of phytohemagglutinin (Sigma-Aldrich). BrdU (1 mM; BD Biosciences) was included in the culture medium for the last 18 hours of treatment. Cells were collected and stained for surface expression of CD3 using directly conjugated anti-CD3 (BD Biosciences), then fixed in 0.5% paraformaldehyde, permeabilized, stained for intracellular BrdU per the manufacturer’s protocol (BrdU Flow Kit; BD Biosciences), and examined by flow cytometry. Scatter plots were generated by gating on CD3+ cells. To examine surface CD69 expression, T cells were incubated in RPMI plus 10% FCS, with or without 5 μg/ml concanavalin A (Calbiochem), for 4 hours. Cells were then collected, stained for CD69 expression using directly conjugated CD69 mAb (BD Biosciences), and assayed by flow cytometry with gating on CD3+ cells. Production of TNF-α, IL-2, and IFN-γ was assayed after treatment of cells with RPMI plus 10% FCS, with or without 5 μg/ml concanavalin A, for 6 hours. Brefeldin A (10 μg/ml; Calbiochem) was included in the culture medium for the last 5 hours of incubation. Cells were then stained for surface marker expression, fixed, permeabilized, stained with anti-cytokine antibodies (Fastimmune; BD Biosciences) and examined by flow cytometry. For the MLR reactions, PBMCs were isolated from 2 different donors via ficoll centrifugation. Stimulator cells were irradiated with 2,000 rad, combined in a 1:2 ratio with responder cells, and cultured in RPMI plus 10% FCS. BrdU (1 mM; BD Biosciences) was added to the culture medium for 24 hours beginning on day 5. Cells were collected on day 6, stained for surface expression of CD3 using directly conjugated anti-CD3 (BD Biosciences), fixed in 0.5% paraformaldehyde, permeabilized, stained for intracellular BrdU per the manufacturer’s protocol (BrdU Flow Kit; BD Biosciences) and examined by flow cytometry. The number of BrdU+ cells per 10,000 CD3+ cells was measured for all groups.
MLRs using DCs.
HPCs from 1 donor were used to colonize matrices with fibroblasts and keratinocytes (to produce T cells) as well as matrices with only fibroblasts (to produce autologous DCs). HPCs from a second, unrelated donor were used to colonize fibroblast-only matrices (to produce allogeneic DCs). DCs were produced as follows: HPCs were added to matrices colonized with fibroblasts alone. Nonadherent cells were harvested from the matrices on day 21 and cultured for 5 days in RPMI plus 10% FCS containing 50 ng/ml GM-CSF and 10 ng/ml IL-4 (R&D Systems). On day 5, 1 μg/ml lipopolysaccharide (Sigma-Aldrich) was added. Cells were collected on day 7, irradiated (2,000 rad), and added to MLR assays as described below. Responder T cells were produced by culturing HPCs in matrices colonized with both keratinocytes and fibroblasts as described above. Additionally, PBMCs were isolated by ficoll fractionation of human leukopheresis products. These cells were used as responder T cells to assess the stimulatory capacity of DCs derived from HPCs. Irradiated stimulator cells (2 × 105 cells) in 100 μl of RPMI plus 10% FCS were added to 1 × 105 responder T cells in 100 μl of RPMI plus 10% FCS (2:1 stimulator/responder ratio) in individual wells of a round-bottomed 96-well plate. Three to 6 duplicate wells were done for each condition. BrdU (1 mM; BD Biosciences) was added to the culture medium for 24 hours beginning on day 5. Cells were collected on day 6, stained for surface expression of CD3 using directly conjugated anti-CD3 (BD Biosciences), fixed in 0.5% paraformaldehyde, permeabilized, stained for intracellular BrdU per the manufacturer’s protocol (BrdU Flow Kit; BD Biosciences), and examined by flow cytometry. The number of BrdU+ cells per 10,000 CD3+ cells was measured for all groups.
Real-time RT-PCR.
The RNA was prepared using Trizol reagent (Invitrogen Corp.) according to the manufacturer’s instructions. Total RNA (3 μg) in 20 μl was transcribed into cDNA using OligodT (Promega) and Omniscript RT Kit (QIAGEN). The cDNA product was mixed with QIAGEN SYBR Green Reagent and forward and reverse primers. Real-time RT-PCR was performed using a Bio-Rad iCycler (Bio-Rad Laboratories). The PCR amplification was performed as follows: denaturation at 95°C for 15 minutes, 40 cycles of amplification (94°C for 15 seconds, 55°C for 30 seconds, and 72°C for 30 seconds), 1 cycle of denaturation (95°C for 60 seconds), and 70 cycles of 1°C touchdown (from 95°C to 25°C, 10 seconds per cycle), ending by holding at 20°C. All PCR runs included absence of RT as the negative control and cyclophilin A as the positive control. Signal for all samples was normalized to expression of cyclophilin A. For gels shown, 10 μl of product was loaded onto 2% agarose gel for detection. All primer sequences were designed with Primer3 software (http://frodo.wi.mit.edu/cgi-bin/primer3/primer3_www.cgi). AIRE primers were as follows: forward, 5′-GATGACCTGGAGTCCCTTCT-3′; reverse, 5′-CTCATCAGAGCTGCATGTCC-3′. AIRE primers were common to the 3 isomers of human AIRE. MBP primers were as follows: forward, 5′-AGGACTGTCCCTGAGCAGAT-3′; reverse, 5′-ACTCCCTTGAATCCCTTGTG-3′. PLP primers were as follows: forward, 5′-GGACCACCTGCCAGTCTATT-3′; reverse, 5′-AAAGCATTCCATGGGAGAAC-3′. S100β primers were as follows: forward, 5′-GGAGACAAGCACAAGCTGAA-3′; reverse, 5′-CCATGACTTTGTCCACAACC-3′. Thyroglobulin primers were as follows: forward, 5′-GATTCTGAGTTCCCCGTGAT-3′; reverse, 5′-GGCAACATTGTCACTTGTCC-3′. Foxn1 primers were as follows: forward, 5′-TCCCTCACTCACTGACTTCG-3′; reverse, 5′-GTGGCATCGAAGATGATGTC-3′. Hoxa3 primers were as follows: forward, 5′-AAGCAGAAAACCAGCAGCTC-3′; reverse, 5′-AGCGGTTGAAGTGGAACTCT-3′. Pax1 primers were as follows: forward, 5′-GGACGTTTATGGAGCAAACA-3′ reverse, 5′-GCAGGTTTCTCTAGCCCATT-3′. Cyclophilin A primers were as follows: forward, 5′-CCGAGGAAAACCGTGTACTAT-3′; reverse, 5′- CTGCAAACAGCTCAAAGGAG-3′. All primers spanned 2 exons to minimize interference from genomic DNA.
Acknowledgments
Thomas Cochran (Boston Center for Cosmetic Surgery, Boston, Massachusetts, USA) and Elof Eriksson (Brigham and Women’s Hospital Department of Plastic Surgery) generously provided adult human skin. James Rheinwald (Harvard Skin Disease Research Center and Brigham and Women’s Hospital Department of Dermatology) kindly provided advice regarding the culture of human keratinocytes. Cellfoam matrices were purchased at an initially reduced cost from Cytomatrix LLC, and Cytomatrix LLC kindly provided the scanning electron micrograph shown in Figure 1A. This research was funded in part by NIH grants P30AR42689, T32AR07098, and R37AI25082 (to T.S. Kupper) and by a Clinical Career Development Award grant from the Dermatology Foundation (to R.A. Clark).
Footnotes
Nonstandard abbreviations used: HPC, hematopoietic precursor cells; LTα, lymphotoxin-α; LTβR, lymphotoxin-β receptor; MBP, myelin basic protein; MLR, mixed leukocyte reaction; OP9-DL1, OP9 cells transduced with delta-like 1; PLP, myelin phospholipid protein; TREC, T cell receptor excision circle.
Conflict of interest: The authors have declared that no conflict of interest exists.
References
- 1.Heitger A, et al. Essential role of the thymus to reconstitute naive (CD45RA+) T-helper cells after human allogeneic bone marrow transplantation. Blood. 1997;90:850–857. [PubMed] [Google Scholar]
- 2.Simpson JG, Gray ES, Beck JS. Age involution in the normal human adult thymus. Clin. Exp. Immunol. 1975;19:261–265. [PMC free article] [PubMed] [Google Scholar]
- 3.von Gaudecker B. Ultrastructure of the age-involuted adult human thymus. Cell Tissue Res. 1978;186:507–525. doi: 10.1007/BF00224939. [DOI] [PubMed] [Google Scholar]
- 4.Galy A, Travis M, Cen D, Chen B. Human T, B, natural killer, and dendritic cells arise from a common bone marrow progenitor cell subset. Immunity. 1995;3:459–473. doi: 10.1016/1074-7613(95)90175-2. [DOI] [PubMed] [Google Scholar]
- 5.Kingston R, Jenkinson EJ, Owen JJ. A single stem cell can recolonize an embryonic thymus, producing phenotypically distinct T-cell populations. Nature. 1985;317:811–813. doi: 10.1038/317811a0. [DOI] [PubMed] [Google Scholar]
- 6.Weijer K, et al. Intrathymic and extrathymic development of human plasmacytoid dendritic cell precursors in vivo. Blood. 2002;99:2752–2759. doi: 10.1182/blood.v99.8.2752. [DOI] [PubMed] [Google Scholar]
- 7.McCune JM, et al. The SCID-hu mouse: murine model for the analysis of human hematolymphoid differentiation and function. Science. 1988;241:1632–1639. doi: 10.1126/science.241.4873.1632. [DOI] [PubMed] [Google Scholar]
- 8.Poznansky MC, et al. Efficient generation of human T cells from a tissue-engineered thymic organoid. Nat. Biotechnol. 2000;18:729–734. doi: 10.1038/77288. [DOI] [PubMed] [Google Scholar]
- 9.Gardner JP, et al. T-lymphopoietic capacity of cord blood-derived CD34+ progenitor cells. Exp. Hematol. 1998;26:991–999. [PubMed] [Google Scholar]
- 10.Rosenzweig M, et al. In vitro T lymphopoiesis of human and rhesus CD34+ progenitor cells. Blood. 1996;87:4040–4048. [PubMed] [Google Scholar]
- 11.Tagoh H, et al. Induction of recombination activating gene expression in a human lymphoid progenitor cell line: requirement of two separate signals from stromal cells and cytokines. Blood. 1996;88:4463–4473. [PubMed] [Google Scholar]
- 12.Yeoman H, Clark DR, DeLuca D. Development of CD4 and CD8 single positive T cells in human thymus organ culture: IL-7 promotes human T cell production by supporting immature T cells. Dev. Comp. Immunol. 1996;20:241–263. doi: 10.1016/0145-305x(96)00013-4. [DOI] [PubMed] [Google Scholar]
- 13.Plum J, De Smedt M, Defresne MP, Leclercq G, Vandekerckhove B. Human CD34+ fetal liver stem cells differentiate to T cells in a mouse thymic microenvironment. Blood. 1994;84:1587–1593. [PubMed] [Google Scholar]
- 14.Krowka JF, Sarin S, Namikawa R, McCune JM, Kaneshima H. Human T cells in the SCID-hu mouse are phenotypically normal and functionally competent. J. Immunol. 1991;146:3751–3756. [PubMed] [Google Scholar]
- 15.Freedman AR, Zhu H, Levine JD, Kalams S, Scadden DT. Generation of human T lymphocytes from bone marrow CD34+ cells in vitro. Nat. Med. 1996;2:46–51. doi: 10.1038/nm0196-46. [DOI] [PubMed] [Google Scholar]
- 16.Rosenzweig M, Marks DF, Hempel D, Johnson RP. In vitro T lymphopoiesis: a model system for stem cell gene therapy for AIDS. J. Med. Primatol. 1996;25:192–200. doi: 10.1111/j.1600-0684.1996.tb00016.x. [DOI] [PubMed] [Google Scholar]
- 17.Anderson G, Jenkinson EJ. Lymphostromal interactions in thymic development and function. Nat. Rev. Immunol. 2001;1:31–40. doi: 10.1038/35095500. [DOI] [PubMed] [Google Scholar]
- 18.Coffer PJ, Burgering BM. Forkhead-box transcription factors and their role in the immune system. Nat. Rev. Immunol. 2004;4:889–899. doi: 10.1038/nri1488. [DOI] [PubMed] [Google Scholar]
- 19.Laster AJ, Itoh T, Palker TJ, Haynes BF. The human thymic microenvironment: thymic epithelium contains specific keratins associated with early and late stages of epidermal keratinocyte maturation. Differentiation. 1986;31:67–77. doi: 10.1111/j.1432-0436.1986.tb00385.x. [DOI] [PubMed] [Google Scholar]
- 20.Rangarajan A, et al. Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J. 2001;20:3427–3436. doi: 10.1093/emboj/20.13.3427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Anderson G, Harman BC, Hare KJ, Jenkinson EJ. Microenvironmental regulation of T cell development in the thymus. Semin. Immunol. 2000;12:457–464. doi: 10.1006/smim.2000.0260. [DOI] [PubMed] [Google Scholar]
- 22.Spits H. Development of alphabeta T cells in the human thymus. Nat. Rev. Immunol. 2002;2:760–772. doi: 10.1038/nri913. [DOI] [PubMed] [Google Scholar]
- 23.Douek DC, et al. Changes in thymic function with age and during the treatment of HIV infection. Nature. 1998;396:690–695. doi: 10.1038/25374. [DOI] [PubMed] [Google Scholar]
- 24.Jaleco AC, et al. Differential effects of notch ligands delta-1 and jagged-1 in human lymphoid differentiation. J. Exp. Med. 2001;194:991–1002. doi: 10.1084/jem.194.7.991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ohishi K, Varnum-Finney B, Bernstein ID. Delta-1 enhances marrow and thymus repopulating ability of human CD34+CD38– cord blood cells. J. Clin. Invest. 2002;110:1165–1174. doi:10.1172/JCI200216167. doi: 10.1172/JCI16167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Karanu FN, et al. Human homologues of Delta-1 and Delta-4 function as mitogenic regulators of primitive human hematopoietic cells. Blood. 2001;97:1960–1967. doi: 10.1182/blood.v97.7.1960. [DOI] [PubMed] [Google Scholar]
- 27.Schmitt TM, Zuniga-Pflucker JC. Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity. 2002;17:749–756. doi: 10.1016/s1074-7613(02)00474-0. [DOI] [PubMed] [Google Scholar]
- 28.La Motte-Mohs RN, Herer E, Zuniga-Pflucker JC. Induction of T cell development from human cord blood hematopoietic stem cells by Delta-like 1 in vitro. Blood. 2004;105:1431–1439. doi: 10.1182/blood-2004-04-1293. [DOI] [PubMed] [Google Scholar]
- 29.Mollah ZUA, Aiba S, Manome H, Yoshino Y, Tagami H. Cord blood CD34+ cells differentiate into dermal dendritic cells in co-culture with cutaneous fibroblasts or stromal cells. J. Invest. Dermatol. 2002;118:450–460. doi: 10.1046/j.0022-202x.2001.01692.x. [DOI] [PubMed] [Google Scholar]
- 30.Anderson MS, et al. Projection of an immunological self shadow within the thymus by the Aire protein. Science. 2002;298:1395–1401. doi: 10.1126/science.1075958. [DOI] [PubMed] [Google Scholar]
- 31.Pitkanen J, Peterson P. Autoimmune regulator: from loss of function to autoimmunity. Genes Immun. 2003;4:12–21. doi: 10.1038/sj.gene.6363929. [DOI] [PubMed] [Google Scholar]
- 32.Pugliese A, Diez J. Lymphoid organs contain diverse cells expressing self-molecules [letter] Nat. Immunol. 2002;3:335–336; author reply 336. doi: 10.1038/ni0402-335b. [DOI] [PubMed] [Google Scholar]
- 33.Pietropaolo M, Giannoukakis N, Trucco M. Cellular environment and freedom of gene expression [letter] Nat. Immunol. 2002;3:335; author reply 336. doi: 10.1038/ni0402-335a. [DOI] [PubMed] [Google Scholar]
- 34.Chin RK, et al. Lymphotoxin pathway directs thymic Aire expression. Nat. Immunol. 2003;4:1121–1127. doi: 10.1038/ni982. [DOI] [PubMed] [Google Scholar]
- 35.Aggarwal BB, Eessalu TE, Hass PE. Characterization of receptors for human tumour necrosis factor and their regulation by gamma-interferon. Nature. 1985;318:665–667. doi: 10.1038/318665a0. [DOI] [PubMed] [Google Scholar]
- 36.Middel P, et al. Expression of lymphotoxin-alpha by keratinocytes: a further mediator for the lichenoid reaction. Pathobiology. 2000;68:291–300. doi: 10.1159/000055940. [DOI] [PubMed] [Google Scholar]
- 37.Adamson KA, Pearce SH, Lamb JR, Seckl JR, Howie SE. A comparative study of mRNA and protein expression of the autoimmune regulator gene (Aire) in embryonic and adult murine tissues. J. Pathol. 2004;202:180–187. doi: 10.1002/path.1493. [DOI] [PubMed] [Google Scholar]
- 38.Gotter J, Brors B, Hergenhahn M, Kyewski B. Medullary epithelial cells of the human thymus express a highly diverse selection of tissue-specific genes colocalized in chromosomal clusters. J. Exp. Med. 2004;199:155–166. doi: 10.1084/jem.20031677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nehls M, Pfeifer D, Schorpp M, Hedrich H, Boehm T. New member of the winged-helix protein family disrupted in mouse and rat nude mutations. Nature. 1994;372:103–107. doi: 10.1038/372103a0. [DOI] [PubMed] [Google Scholar]
- 40.Baxter RM, Brissette JL. Role of the nude gene in epithelial terminal differentiation. J. Invest. Dermatol. 2002;118:303–309. doi: 10.1046/j.0022-202x.2001.01662.x. [DOI] [PubMed] [Google Scholar]
- 41.Janes SM, Ofstad TA, Campbell DH, Watt FM, Prowse DM. Transient activation of FOXN1 in keratinocytes induces a transcriptional programme that promotes terminal differentiation: contrasting roles of FOXN1 and Akt. J. Cell Sci. 2004;117:4157–4168. doi: 10.1242/jcs.01302. [DOI] [PubMed] [Google Scholar]
- 42.Manley NR, Capecchi MR. The role of Hoxa-3 in mouse thymus and thyroid development. Development. 1995;121:1989–2003. doi: 10.1242/dev.121.7.1989. [DOI] [PubMed] [Google Scholar]
- 43.Mace KA, Hansen SL, Myers C, Young DM, Boudreau N. HOXA3 induces cell migration in endothelial and epithelial cells promoting angiogenesis and wound repair. J. Cell Sci. 2005;118:2567–2577. doi: 10.1242/jcs.02399. [DOI] [PubMed] [Google Scholar]
- 44.Wallin J, et al. Pax1 is expressed during development of the thymus epithelium and is required for normal T-cell maturation. Development. 1996;122:23–30. doi: 10.1242/dev.122.1.23. [DOI] [PubMed] [Google Scholar]
- 45.Zuniga-Pflucker JC. T-cell development made simple. Nat. Rev. Immunol. 2004;4:67–72. doi: 10.1038/nri1257. [DOI] [PubMed] [Google Scholar]
- 46.Res P, Blom B, Hori T, Weijer K, Spits H. Downregulation of CD1 marks acquisition of functional maturation of human thymocytes and defines a control point in late stages of human T cell development. J. Exp. Med. 1997;185:141–152. doi: 10.1084/jem.185.1.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liston A, Lesage S, Wilson J, Peltonen L, Goodnow CC. Aire regulates negative selection of organ-specific T cells. Nat. Immunol. 2003;4:350–354. doi: 10.1038/ni906. [DOI] [PubMed] [Google Scholar]
- 48.van Ewijk W, et al. Thymic microenvironments, 3-D versus 2-D? Semin. Immunol. 1999;11:57–64. doi: 10.1006/smim.1998.0158. [DOI] [PubMed] [Google Scholar]
- 49.Manley NR, Capecchi MR. Hox group 3 paralogs regulate the development and migration of the thymus, thyroid, and parathyroid glands. Dev. Biol. 1998;195:1–15. doi: 10.1006/dbio.1997.8827. [DOI] [PubMed] [Google Scholar]
- 50.Hozumi K, et al. Delta-like 1 is necessary for the generation of marginal zone B cells but not T cells in vivo. Nat. Immunol. 2004;5:638–644. doi: 10.1038/ni1075. [DOI] [PubMed] [Google Scholar]
- 51.Lowell S, Jones P, Le Roux I, Dunne J, Watt FM. Stimulation of human epidermal differentiation by Delta-Notch signalling at the boundaries of stem-cell clusters. Curr. Biol. 2000;10:491–500. doi: 10.1016/s0960-9822(00)00451-6. [DOI] [PubMed] [Google Scholar]
- 52.Hiesse C, et al. Malignancy after renal transplantation: analysis of incidence and risk factors in 1700 patients followed during a 25-year period. Transplant. Proc. 1997;29:831–833. doi: 10.1016/s0041-1345(96)00153-4. [DOI] [PubMed] [Google Scholar]
- 53.Zhao Y, et al. Highly disparate xenogeneic skin graft tolerance induction by fetal pig thymus in thymectomized mice: conditioning requirements and the role of coimplantation of fetal pig liver. Transplantation. 2001;72:1608–1615. doi: 10.1097/00007890-200111270-00006. [DOI] [PubMed] [Google Scholar]
- 54.Pilch H, et al. Improved assessment of T-cell receptor (TCR) VB repertoire in clinical specimens: combination of TCR-CDR3 spectratyping with flow cytometry-based TCR VB frequency analysis. Clin. Diagn. Lab. Immunol. 2002;9:257–266. doi: 10.1128/CDLI.9.2.257-266.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]