Abstract
Dickkopf-1 (Dkk-1) has been shown to be a potent inhibitor of Wnt/β-catenin signaling in a variety of assays and organisms. In this study, we show that expression of Dkk-1 overlaps significantly with the sites of programmed cell death in normal as well as mutant vertebrate limb development, and identify several of its upstream regulators, one of which is Bmp-4. Interestingly, Bmp-4 only activates Dkk-1 when it concomitantly induces apoptosis. Moreover, Dkk-1 is heavily up-regulated by UV irradiation and several other genotoxic stimuli. We further show that normal expression of Dkk-1 is dependent on the Ap-1 family member c-Jun and that overexpression of Dkk-1 enhances Bmp-triggered apoptosis in the vertebrate limb. Taken together, our results provide evidence for an important role of Dkk-1-mediated inhibition of Wnt/β-catenin signaling in response to different stress signals that all converge on the activation of c-Jun in vivo.
Keywords: apoptosis/Bmp/c-Jun/Dkk-1/limb development
Introduction
Secreted glycoproteins of the Wnt family of signaling molecules are important regulators of cell growth and survival. Wnts are expressed in a tissue-specific manner, and mice harboring mutations in genes, the products of which are involved in Wnt signal transduction, show severe defects in the development of a variety of organs (for a review see Cadigan and Nusse, 1997). Wnts bind to a receptor complex consisting of a member of the Frizzled family of seven transmembrane proteins and the LDL-receptor-related proteins Lrp-5 or Lrp-6 (for a review see Bejsovec, 2000). In the canonical Wnt/β-catenin pathway, receptor activation leads to a stabilization of β-catenin, which accumulates and translocates into the nucleus to activate target gene expression in concert with transcription factors such as Tcf and Lef. Recently, a biochemical link between the receptor complex and the cytoplasmic components that rapidly degrade β-catenin in the absence of a Wnt signal has been established by the finding that Lrp-5 binds to Axin (J.Mao et al., 2001). The ability of many Wnts to stabilize β-catenin seems to be the basis for their proliferation-promoting effect. This proto-oncogenic potential is reflected in frequently found mutations in components of the Wnt pathway in human cancers (for a review see Polakis, 2000). Dickkopf-1 (Dkk-1) is a secreted protein that specifically inhibits Wnt/β-catenin signaling by interacting with the co-receptor Lrp-6 (B.Mao et al., 2001; Zorn, 2001). Dkk-1 was originally identified as a strong head inducer in Xenopus, due to its potent anti-Wnt effect (Glinka et al., 1998). Analyses carried out in zebrafish and chick embryos showed that the Wnt-antagonizing function of Dkk-1 is conserved in other vertebrates (Shinya et al., 2000; Marvin et al., 2001). However, less is known about the regulation of Dkk-1 expression and possible functions outside head development.
Vertebrate limb development provides a paradigm for developmental apoptosis. Programmed cell death (PCD) occurs in well-defined domains and sculpts the shape of the limb by eliminating cells between the differentiating cartilages (Hurle et al., 1996). In the early chicken limb bud, the most prominent sites of apoptosis are located in the anterior (ANZ) and posterior (PNZ) necrotic zones and in the apical ectodermal ridge (AER). Later, massive cell death takes place in the mesodermal web (interdigital necrotic zone, INZ) separating the digits. The pattern of PCD is very similar in mouse limb development, but ANZ and PNZ are less pronounced compared with in chick. Bone morphogenetic proteins (Bmp) have been identified as important signals triggering cell death in these zones (Yokouchi et al., 1996; Pizette and Niswander, 1999). In sharp contrast, Bmps also promote the formation of bone (Duprez et al., 1996; Buckland et al., 1998). These opposing activities reside in close vicinity to each other in the developing limb, namely in the interdigits versus the digital rays. The downstream mechanisms exerting this dual function are poorly understood. Bmps, as members of the transforming growth factor (TGF)-β superfamily, transmit their signal via at least two distinct pathways. One involves Smad-1, -5 or -8, which are phosphorylated by activated type I Bmp-2/4 receptors and then associate with a common signaling mediator, Smad-4. The heteromeric complex translocates into the nucleus and activates target genes together with different co-factors (for a review see Massagué et al., 2000). A mitogen-activated protein kinase (MAPK) cascade represents an alternative way for Bmp signal transduction. Several MAPKs like, for example, Jnk can be activated depending on cell type and experimental conditions (for a review see Massagué et al., 2000). The Jnk protein kinases phosporylate serine residues 63 and 73 of the c-Jun activation domain leading to increased Ap-1 transcriptional activity (Derijard et al., 1994; Kyriakis et al., 1994). Accumulating evidence suggests that the MAPKKK family member TAK-1 provides the biochemical link between the TGF-β receptor and the MAPK pathway (Takatsu et al., 2000). Smads can interact in vitro with the Jnk substrate c-Jun, indicating that Bmps might simultanously activate the Smad and MAPK pathways, which then physically converge on target genes (Zhang et al., 1998; Wong et al., 1999). Recent evidence suggests that these two pathways can also counteract each other (Kimura et al., 2000; Pessah et al., 2001), raising the possibility that the balance of these two intracellular pathways is a key to co-ordinated cellular response to Bmp in the physiological context.
We have previously shown that Dkk-1 is expressed in a dynamic pattern during mouse limb development (Grotewold et al., 1999). Here, we show that these expression domains significantly overlap with the sites of PCD, indicating a potential function of Dkk-1 in this process. Another implication for Dkk-1 in PCD comes from a recent study providing evidence for Dkk-1 being a target of p53, a checkpoint protein controling cell cycle progression and apoptosis (Wang et al., 2000). Therefore, we were interested to determine whether Dkk-1 might be involved in controling PCD in development. Furthermore, we asked whether signals triggering apoptosis, like Bmp signaling, are involved in the regulation of Dkk-1 expression.
Results
Expression pattern of Dkk-1
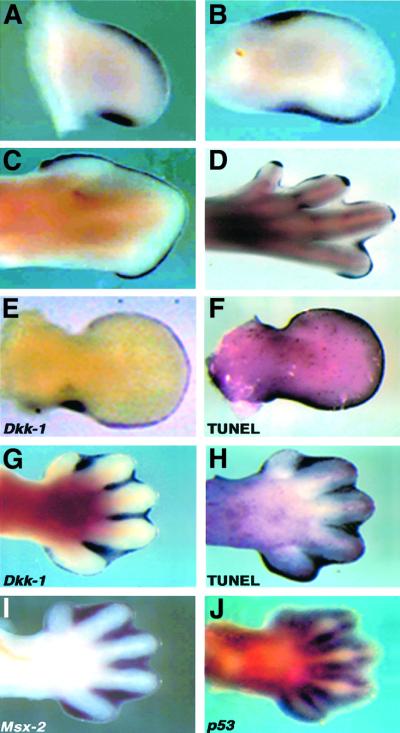
We have recently described the dynamic expression pattern of Dkk-1 in mouse limb development (Grotewold et al., 1999). We extended this expression study to the chicken embryo and observed a very similar pattern. At HH23, Dkk-1 is expressed in a posterior and an anterior mesenchymal domain, as it is at HH25 (Figure 1A and B). At later stages it starts to be expressed in the AER and weakly in the interdigital mesenchyme (Figure 1C and D). At HH32 transcripts can also be detected in the developing joints (Figure 1D). These sites of expression overlap to a high degree with the sites of PCD, as exemplified in Figure 1E–H for mouse limb buds (data not shown for the chick). At embryonic day (E) 11.5, Dkk-1 expression is confined to the AER and a posterior mesenchymal domain (Figure 1E) corresponding to the PNZ. TUNEL staining of an age-matched limb bud reveals that these areas undergo massive PCD at this time point (Figure 1F). A similar coincidence is observed at E13.5 (Figure 1G and H). In the interdigital mesenchyme, Dkk-1 is also co-expressed with the pro-apoptotic genes Msx-2 (Figure 1I), which has been suggested to mediate Bmp-triggered PCD (Marazzi et al., 1997), and p53 (Figure 1J).
Fig. 1. Dkk-1 is expressed at sites of apoptosis during vertebrate limb development. (A–D) Chick limb buds of different stages, (A) HH23 wing, (B) HH25 wing, (C) HH32 wing and (D) HH32 leg, show Dkk-1 transcripts in the ANZ, PNZ, the AER and INZ and the developing joints. (E) Dkk-1 expression in an E11.5 mouse forelimb bud in the AER and posterior mesenchyme and (F) whole-mount TUNEL staining of an age-matched forelimb bud indicates the co-localization of Dkk-1 transcripts and the sites of apoptosis. (G) At E13.5, Dkk-1 expression is confined to the interdigital mesenchyme, where massive apoptosis takes place as the TUNEL staining of an E13.5 limb bud indicates (H). At this time point Dkk-1 is co-expressed with the pro-apoptotic genes Msx-2 (I) and p53 (J).
Dkk-1 promotes apoptosis in limb buds
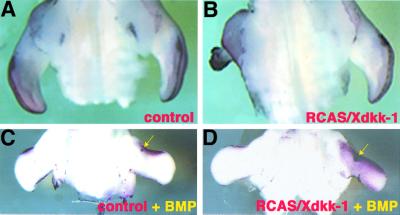
Given the correlation between Dkk-1 expression and the sites of PCD, we wanted to investigate whether overexpression of Dkk-1 might be sufficient to induce apoptosis. To this end, we injected a recombinant retrovirus containing the Xenopus Dkk-1 coding sequence (RCAS/Xdkk-1) into the mesoderm of HH18 wing buds. Viral spread was monitored by whole-mount in situ hybridization using a probe that detects viral message. Thirty-six hours after infection we observed viral transcripts in the whole wing mesoderm (data not shown). After 60–72 h, distal truncations of variable degree could be observed (42% of injected embryos, Figure 2B shows a dramatically truncated wing bud representing 29% of infected limbs) in the infected wings. TUNEL staining revealed massive ectopic PCD in the distal part of the truncated limbs (Figure 2B). As a control, we infected early wing buds with a virus expressing human alkaline phosphatase (RCAS/AP). Normal wing buds developed in 100% of these embryos, which also showed the normal pattern of PCD 60–72 h post-infection (Figure 2A). This shows that the observed phenotype after RCAS/Xdkk-1 infection is not due to non-specific cytotoxic effects of the virus.
Fig. 2. Dkk-1 promotes apoptosis in limb buds. (A and B) TUNEL stainings of RCAS-infected wing buds, ventral view. (A) Control wing infected with RCAS/AP showing the normal pattern of PCD in the ANZ and AER. (B) Dramatic truncation of a wing bud infected with RCAS/Xdkk-1. Note the massive apoptosis in the truncated wing compared with the contralateral control wing. (C and D) Dorsal views of wing buds infected with RCAS/AP (C) and RCAS/Xdkk-1 (D), which additionally received a bead soaked in rhBMP-4 (arrows). (C) Twenty hours after bead implantation the ectopic Bmp signal leads to an increased PCD in a region surrounding the bead as shown by TUNEL. (D) The overexpression of Dkk-1 dramatically enhances this effect of Bmp. The region of cells undergoing PCD covers nearly the complete wing mesoderm.
Overexpression of RCAS/Xdkk-1 resulted in only a very limited increase in TUNEL-positive cells before the drastic truncations occured (data not shown). Thus, we could not exclude the possibility that the massive PCD in these specimens (Figure 2B) occured secondary to the truncations. In order to address the question of whether Dkk-1 modulates PCD, we used another strategy. Implantation of beads soaked in Bmp protein into the undifferentiated limb mesoderm induces PCD within 20 h in a region surrounding the bead (Figure 2C; Macias et al., 1997; Zou et al., 1997; Pizette and Niswander, 1999). We asked whether the overexpression of Dkk-1 would have an effect on the extend of cell death induced by Bmp. For this purpose, wing buds were infected with RCAS/Xdkk-1 or RCAS/AP at HH18. Twenty-four hours later, we implanted a bead soaked in 100 µg/ml recombinant human BMP-4 (rhBMP-4) and analyzed the extent of PCD a further 20 h later by TUNEL staining. As shown in Figure 2D, the ectopic expression of Dkk-1 prior to the ectopic Bmp signal significantly enhanced the PCD-inducing effect of Bmp.
These results present strong evidence that Dkk-1 promotes PCD in the developing vertebrate limb.
Regulation of Dkk-1 by Bmp, Fgf and Shh
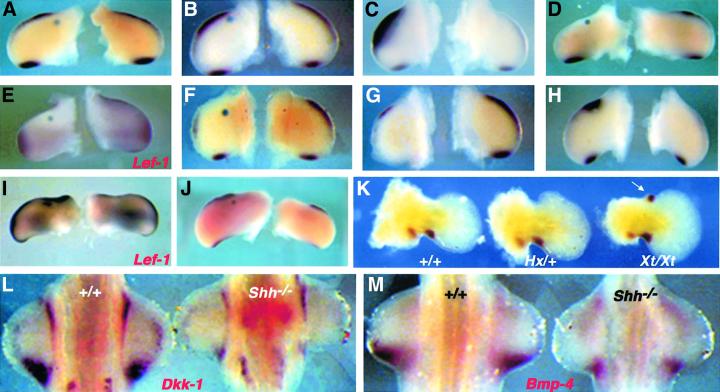
As the expression domains of Dkk-1 are remarkably similar to those of some members of the Bmp family and their target genes (Figure 1I) and Dkk-1 enhances the PCD-triggering effect of Bmp-4 in the vertebrate limb, we started to analyze the effects of enhanced Bmp signaling on Dkk-1 transcription. For this purpose, we implanted beads soaked in rhBMP-4 into the mesenchyme of HH20–23 limb buds. Control bovine serum albumin (BSA) beads and beads soaked in 1 µg/ml rhBMP-4 did not affect Dkk-1 expression levels (n = 8/8 and 5/5, respectively; Figure 3A and B). When we increased the rhBMP-4 concentration to 100 µg/ml, massive ectopic Dkk-1 expression was induced within 2 h (n = 6/6; Figure 3C), which was still evident after 8 h (n = 8/8; data not shown). To investigate whether this massive up-regulation leads to an enhanced inhibition of Wnt/β-catenin signaling, we monitored the expression of the Wnt target gene Lef-1. Indeed, Lef-1 was drastically reduced in a wide area around the bead after 8 h (n = 6/6; Figure 3E). In order to analyze whether Bmp signaling is necessary for Dkk-1 expression, we implanted beads soaked in Noggin (1 mg/ml), a potent Bmp antagonist, into the anterior mesenchyme of HH20–23 wing buds. Six, 8 and 15 h after this treatment we observed a complete loss of the endogenous Dkk-1 expression domain in vicinity to the bead (n = 16/16; shown for 8 h in Figure 3F). Only at a distance from the bead was a faint staining for Dkk-1 transcripts detectable in the very distal mesenchyme. Thus, Bmp signaling is essential for normal Dkk-1 expression. Twenty hours after implantation of a bead soaked in 100 µg/ml rhBMP-4, Dkk-1 transcription was completely abolished or drastically reduced at the implanted site (n = 7/8; Figure 3D). As this treatment induces massive ectopic cell death after 20 h (see Figure 2C), loss of these cells might provide an explanation for this down-regulation. Alternatively, this might indicate that Dkk-1 expression depends on signals from the AER, the structure and function of which has been shown to be negatively regulated by Bmp under these experimental conditions (Pizette and Niswander, 1999). In order to test for the latter possibility, the AER was surgically removed from HH20–23 limb buds. This manipulation resulted in a severe decrease of Dkk-1 transcripts in the underlying mesoderm within 6 h (n = 6/6; Figure 3G). As members of the fibroblast growth factor (Fgf) family are regarded as the main mediators of AER function, we implanted beads soaked in Fgf-8 protein to analyze whether this is sufficient to induce Dkk-1 expression. We found that Dkk-1 is up-regulated at 8 and 20 h after the surgery (n = 15/15; Figure 3H; data not shown). In spite of the forced Dkk-1 expression, ectopic Fgf did not result in an inhibition but rather in an activation of Wnt/β-catenin signaling as judged by enhanced Lef-1 transcription (n = 8/8; Figure 3I). These results show that Dkk-1 is positively regulated by Bmp and Fgf signaling.
Fig. 3. Regulation of Dkk-1 by Bmp, Fgf and Shh. Surgical manipulations of wing buds in (A)–(K) were performed between HH20 and HH23. Operated wings (left) and contralateral controls (right) are presented in each panel. (A) A BSA-soaked control bead or (B) a bead soaked in 1 µg/ml rhBMP-4 does not affect Dkk-1 expression after 8 h. (C) Massive ectopic expression of Dkk-1 around a Bmp bead (100 µg/ml) 2 h post-implantation. Note that Dkk-1 is only induced between the bead and the AER. (D) Twenty hours after application of 100 µg/ml rhBMP-4, Dkk-1 expression is completely down-regulated. (E) Lef-1 expression is drastically reduced 8 h after the same treatment. (F) Loss of Dkk-1 transcripts in the vicinity of a bead soaked in 1 mg/ml Noggin after 8 h. (G) Removal of the AER leads to a decline of Dkk-1 transcripts in the subjacent mesenchyme after 6 h. (H) Dkk-1 is induced by 1 mg/ml Fgf-8 (8 h). (I) Lef-1 expression is also clearly enhanced following this treatment. (J) Shh (2.5 mg/ml) has no effect on Dkk-1 expression, even after 24 h. Note the increased size of the operated wing, which indicates that the Shh protein provided by the bead is functional. (K) Dkk-1 expression in E12.0 mouse forelimb buds. Wild type (left) and Hx/+ (middle) show the same pattern, whereas a strong ectopic expression domain of Dkk-1 is detectable in the anterior mesenchyme of Xt/Xt (right) limb buds (arrow). (L) Dkk-1 expression is drastically reduced in Shh–/– forelimb buds (right) at E10.5 (left side shows an age-matched wild-type control). (M) Bmp-4 is also down-regulated in Shh–/– limb buds (right) compared with wild type (left) at E10.5.
Shh is expressed in the posterior mesenchyme of the limb bud and is essential for proper limb development (Chiang et al., 1996). A recent study also implicated Shh in the regulation of PCD (Sanz-Ezquerro and Tickle, 2000). As the expression domains of Dkk-1 and Shh initially overlap and then become separated (Grotewold et al., 1999), we wanted to investigate possible effects of this signaling molecule on Dkk-1 mRNA levels. Beads soaked in 2.5 mg/ml Shh failed to induce ectopic activation of Dkk-1 at all time points investigated (4 h, n = 5/5; 10 h, n = 4/4; 24 h, n = 5/5; Figure 3J). Thus, Shh seems not to be sufficient to activate the Dkk-1 promoter. This finding in the chick is consistent with data we obtained from expression analyses in two different polydactylous mouse mutants, Extra-toes (Xt) and Hemimelic extratoe (Hx). Although Shh is strongly expressed in the anterior mesenchyme of Hx/+ limb buds (Büscher and Rüther, 1998), no ectopic activation of Dkk-1 could be observed at all developmental stages examined (E11.0–12.5; Figure 3K). Thus, also in the mouse, ectopic Shh expression is not sufficient to induce Dkk-1 expression. In the anterior mesenchyme of Xt/Xt forelimb buds, however, Dkk-1 is ectopically expressed at E12.0 (Figure 3K). In order to analyze whether Shh is necessary for the normal expression of Dkk-1, we performed in situ hybridizations on Shh–/– embryos at E10.5. Figure 3L shows that Dkk-1 is expressed in those embryos, albeit at a dramatically decreased level. This shows that Shh is not required for the initial induction of Dkk-1, but is for the maintenance of Dkk-1 expression. In addition, Bmp-4 is drastically down-regulated in Shh–/– mutant embryos (Figure 3M). These results uncover a complex regulation of Dkk-1 in the molecular network of limb development.
Bmp concomitantly induces Dkk-1 expression and apoptosis
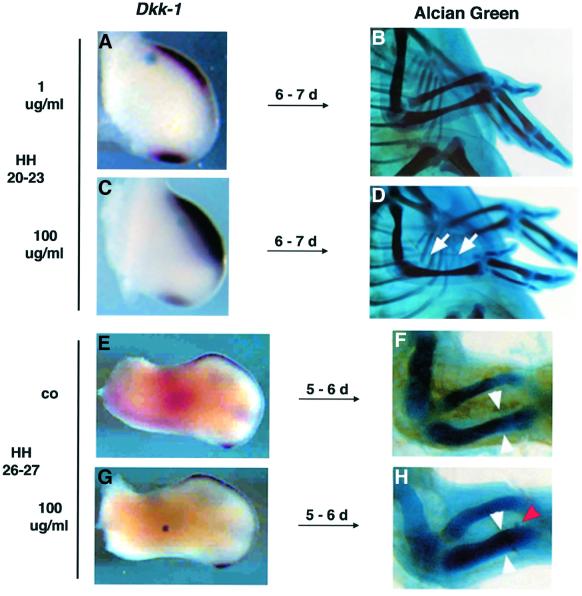
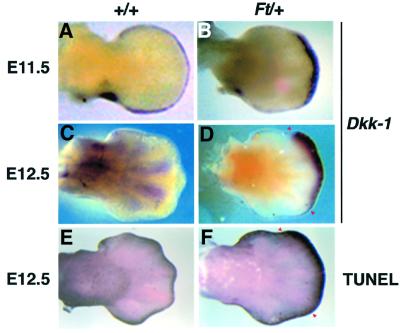
As shown in Figure 2C, a Bmp-bead implanted into the undifferentiated mesenchyme between HH20 and HH23 induces massive apoptosis within 20 h. Some embryos treated this way were allowed to develop until E9–10 to observe the morphological effects of increased cell death. Figure 4D shows that a complete loss of the radius could be detected in all of these embryos (n = 8/8). The increased cell death is preceeded by a dramatic up-regulation of Dkk-1 in the same region (Figure 4C). As shown previously, implantation of a Bmp bead of the same concentration at HH26–27 leads to the completely opposite phenotype, namely an increase in bone mass (Buckland et al., 1998). This was also the case in our experiments (n = 9/12; compare Figure 4F with H). Importantly, in none of these cases (n = 12/12) could an up-regulation of Dkk-1 expression be observed at 4–10 h around the bead (Figure 4G). This suggests that the concomitant activation of Bmp and the inhibition of Wnt/β-catenin signaling leads to apoptosis in limb mesodermal cells. Our laboratory has previously characterized the syndactylous mouse mutant Fused toes (Ft) heterozygous embryos, which exhibit ectopic activation of the Bmp signaling pathway in the anterior/distal mesenchyme of the limb (Heymer and Rüther, 1999). We were interested in whether this physiological expression of Bmps also influenced the expression of Dkk-1 and the extent of PCD. Remarkably, at E11.5–12.0, when the ectopic Bmp activation can be first observed, Dkk-1 is ectopically expressed in the distal mesenchyme of Ft/+ forelimb buds (Figure 5B). At this timepoint, no ectopic cell death could be observed by whole-mount TUNEL staining (data not shown). At E12.5, when Dkk-1 is expressed in the interdigital mesenchyme in the wild type (Figure 5C), Dkk-1 is still ectopically expressed in the region of the future fusion of the digits in Ft/+ embryos (Figure 5D). At this point, massive ectopic cell death could be detected in the Dkk-1-expressing region (compare Figure 5D with F). Thus, during normal as well as mutant limb development, Dkk-1 is expressed in regions of high Bmp signaling and apoptosis. Strikingly, Dkk-1 expression preceeds the appearance of apoptosis and is not induced by a Bmp signal that leads to enhanced chondrogenesis. This implies an important function of Dkk-1 in the modulation of Bmp-induced PCD.
Fig. 4. Bmp-4 concomitantly induces Dkk-1 expression and apoptosis. The bead implantations in (A)–(D) have been carried out between HH20 and HH23, and those in (E)–(H) at HH26/27. (A) A control bead soaked in 1 µg/ml rhBMP-4 does not affect Dkk-1 expression. (B) Wild-type skeletal pattern of a wing treated the manner described in (A) after 10 days of incubation. (C) rhBMP-4 100 µg/ml rapidly induces Dkk-1 expression, resulting in the complete absence of the radius after 10 days [(D), arrows)]. (E) Control (co) wing shows the wild-type pattern of Dkk-1 expression at HH26. (F) Wild-type size of radius and ulna in an E10 embryo. (G) A Bmp bead (100 µg/ml) implanted into the center of a HH26/27 wing does not induce Dkk-1 expression after 8 h. (H) This treatment leads to a significant increase in bone mass of radius and ulna after 10 days. White arrowheads have the same distance as in (F), the red arrowhead points to the implanted bead.
Fig. 5. Dkk-1 expression and PCD in Ft/+ forelimb buds. (A) E11.5 wild-type limb bud shows Dkk-1 transcripts in the PNZ and AER. (B) Ectopic expression of Dkk-1 in the distal-most mesenchyme of an E11.5 Ft/+ limb bud. (C) Dkk-1 expression in the interdigital mesenchyme of an E12.5 wild-type limb bud. (D) Dkk-1 is ectopically activated at E12.5 in Ft/+ forelimb buds (red arrowheads). (E) TUNEL staining shows the wild-type pattern of PCD at E12.5. Apoptotic cells are restricted to the AER and the distal-most interdigital mesenchyme. (F) High ectopic cell death in Ft/+ forelimb bud (red arrowheads) in a region that significantly overlaps with that of ectopic Dkk-1 transcription [compare with (D)].
Dkk-1 is induced by several genotoxic signals
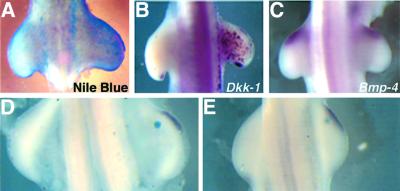
We were interested in whether Dkk-1 might also be activated by other apoptotic stimuli as Dkk-1 has recently been suggested to be a target of the tumor-suppressor gene p53, one of the most important regulators of PCD (Wang et al., 2000). Therefore, we exposed chick embryos of different developmental stages to ultraviolet (UV) light as UV-induced DNA damage results in a stabilization of p53, which then activates the cell death or cell cycle arrest program (for a review see Vogelstein et al., 2000). Figure 6A shows that this treatment induces massive cell death after 10 h (n = 16/16) in the irradiated territories as judged by Nile Blue staining. When we looked for Dkk-1 expression in these irradiated embryos, we observed high ectopic expression of the gene in a salt-and-pepper-like fashion in the whole mesenchyme as well as ectoderm (n = 12/12 after 8 h; Figure 6B), which closely resembled the pattern of apoptosis (cf. Figure 6A). Thus, Dkk-1 is induced by the UV-activated cell death pathway in vivo. As mentioned above, Bmp-4 is a strong inducer of Dkk-1 in apoptotic responses. Therefore, we wished to investigate whether the UV-induced Dkk-1 activation might be mediated by Bmp-4 or whether the two pathways are rather independent. Figure 6C shows that Bmp-4 is normally expressed in irradiated chick embryos at various time points after the treatment (1 h, n = 3/3; 4 h, n = 5/5; 8 h, n = 8/8; 16 h, n = 6/6). In the next set of experiments, we implanted beads soaked in staurosporin, a protein kinase inhibitor that has been shown to induce apoptosis when delivered to the chick limb bud (Sanz-Ezquerro and Tickle, 2000). When we monitored Dkk-1 expression following this treatment, we found a strong up-regulation of its expression 2 h (n = 5/5; Figure 6D) and 4 h (n = 6/6; Figure 6E) after bead implantation. Thus, we conclude that several different apoptotic stimuli lead to an up-regulation of Dkk-1 expression.
Fig. 6. Dkk-1 is induced by UV irradiation and staurosporin. (A) Nile Blue staining of a chick embryo 10 h after UV irradiation. Note the massive cell death in the right irradiated limb bud. The non-irradiated left side served as an internal control in all of these experiments. (B) Dkk-1 is ectopically expressed in a salt-and-pepper-like fashion 8 h after UV irradiation. (C) Bmp-4 expression is not altered after 8 h. (D and E) Beads soaked in staurosporin induce Dkk-1 expression after 2 h (D) and 4 h (E).
Dkk-1 is regulated by c-Jun
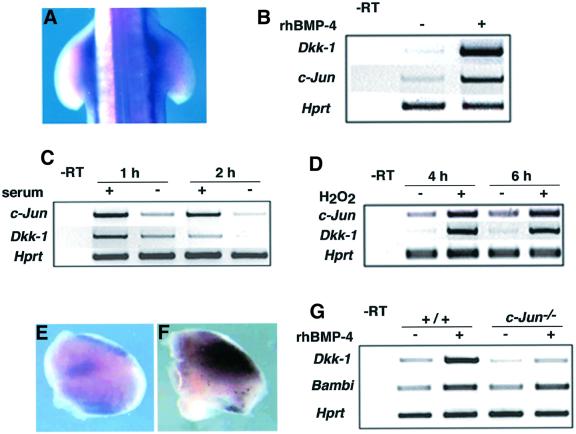
As very different stress stimuli lead to the induction of Dkk-1, we sought to determine whether these signals might activate a common transcription factor to induce Dkk-1. A good candidate for such a role was c-Jun, which has been shown to be required for UV-induced apoptosis (Shaulian et al., 2000). In addition, staurosporin was suggested to induce apoptosis via Jnk-mediated increase in Ap-1 activity (Chae et al., 2000). We performed in situ hybridizations on UV-irradiated chicks and found a massive up-regulation of chicken c-Jun after 1 h (n = 9/9; Figure 7A). This up-regulation was transient as it was no longer detectable after 4 h (data not shown). Next we were interested in determining whether Bmp can activate c-Jun expression under conditions where Dkk-1 is also induced. We could not test this by bead implantations because c-Jun is induced by embryonic wounding (L.Grotewold, unpublished observation); therefore, we decided to switch to a cell culture assay. We applied rhBMP-4 to mouse embryonic fibroblasts (MEF) and could strongly induce Dkk-1 expression after 30 min, as determined by RT–PCR (Figure 7B). Under these conditions, c-Jun was also strongly induced (Figure 7B). c-Jun has been shown to be regulated by serum factors and induced by several different stress stimuli. If Dkk-1 was regulated by c-Jun we should expect a concomitant switch of Dkk-1 expression under the respective conditions. Figure 7C shows that c-Jun and Dkk-1 expression are rapidly down-regulated after serum withdrawal, indicating that Dkk-1 expression depends on serum factors and/or c-Jun expression. Apoptosis triggered by oxidative stress involves c-Jun induction in different cell types (Janssen et al., 1997; Richter-Landsberg and Vollgraf, 1998). We applied H2O2 to MEF and observed an induction of c-Jun and Dkk-1 expression (Figure 7D). These results show that c-Jun and Dkk-1 are co-regulated under several experimental conditions. The question emerged whether c-Jun might be sufficient to induce Dkk-1 expression. To analyze this, we used the ex vivo electroporation system to deliver a c-Jun expression plasmid to the limb bud. After 6 h in culture, the explants were processed for in situ hybridization. Whereas the endogenous expression pattern of Dkk-1 was nicely recapitulated in the control limb buds, those electroporated with a c-Jun expression vector expressed Dkk-1 at high levels at ectopic sites (Figure 7E and F). This clearly indicates that c-Jun is able to induce Dkk-1 expression in vivo. To analyze whether c-Jun might also be essential for Dkk-1 expression and the Bmp-induced Dkk-1 up-regulation, we applied rhBMP-4 to MEF derived from either wild-type or c-Jun–/– embryos. Figure 7G shows that the endogenous expression of Dkk-1 (without application of rhBMP) was already severely reduced in c-Jun–/– cells. When rhBMP-4 was added to the cultures, Dkk-1 was up-regulated 5.2-fold after 4 h in wild-type MEF. In c-Jun–/– MEF, Dkk-1 was still induced by rhBMP-4, albeit at much reduced levels (only by a factor of 1.7 compared with c-Jun–/– without rhBMP-4; Figure 7G). Another gene that we have previously shown to be inducible by rhBMP-4 in this system is Bambi (Grotewold et al., 2001). The basal level of Bambi expression and its induction by rhBMP-4 are unaltered in c-Jun–/– MEF (Figure 7G), demonstrating that these cells do not harbor a general defect in Bmp signal transduction. These results show that c-Jun is indispensable for the normal expression of Dkk-1 as well as for the full induction of Dkk-1 transcription in response to Bmp-4.
Fig. 7. Dkk-1 is regulated by c-Jun. (A) c-Jun is up-regulated 1 h after UV irradiation. Note the intense c-Jun expression in the right wing bud and the flank compared with the non-irradiated left side of the embryo. (B) Dkk-1 and c-Jun are co-induced by application of rhBMP-4 to MEF after 30 min. Hprt was used for standardization in all RT–PCR experiments. (C) c-Jun and Dkk-1 are down-regulated in MEF, 1 and 2 h after serum withdrawal. (D) H2O2 strongly up-regulates c-Jun and Dkk-1. (E and F) Limb bud explants electroporated with a control vector (E) and a c-Jun expression plasmid (F). Note the intense up-regulation of Dkk-1 expression 6 h after electroporation of the c-Jun expression vector (F). (G) Dkk-1 expression is reduced by 40% in c-Jun–/– MEF compared with wild type (compare +/+ and c-Jun–/– without rhBMP-4). After the application of rhBMP-4 Dkk-1 is 5.2-fold induced in wild-type MEF, but only 1.7-fold in c-Jun–/– cells. Bambi is normally expressed and induced by the same factor in c-Jun–/– as in wild-type MEF. –RT, control without reverse transcriptase.
Discussion
Expression of Dkk-1 at sites of PCD
We show that during mouse and chicken limb development the sites of Dkk-1 expression significantly overlap with the areas of PCD. We found this co-localization in the early phase of limb development, where the most prominent apoptotic activity resides in the ANZ, PNZ and in the AER, and during the growth and differentiation phase, when the interdigital mesenchyme undergoes extensive PCD. Here, Dkk-1 expression overlaps with the expression of the pro-apoptotic genes Bmp-4, Msx-2, p53 and also c-Jun. In the late limb bud, the developing joints represent another prominent site of co-expression of Dkk-1 and c-Jun. Interestingly, the domains of Bmp-4, Dkk-1 and c-Jun also overlap at other sites in the embryo, including the otic vesicle and branchial arches, where apoptosis plays a pivotal role to elaborate the shape of the corresponding structure (Wilkinson et al., 1989; Sanz et al., 1999; data not shown). This indicates that the molecular cascade for Bmp-triggered apoptosis we propose (see below) is not limited to the developing limb but might be of general relevance during embryonic development.
Interaction of Bmp, Fgf and Wnt: the role of Dkk-1
Besides the Bmp, Fgf and Wnt pathways, Shh is also crucial for patterning the vertebrate limb. Our results from bead implantations in the chick embryo as well as from gene expression analyses in different mouse mutants, however, show that Shh plays only a minor role in Dkk-1 expression. The up-regulation of Dkk-1 expression in Xt/Xt limb buds preceeds the ectopic activation of Shh, as it can already be detected at E10.5 (data not shown), i.e. before the ectopic Shh domain emerges (Büscher and Rüther, 1998). Thus, the up-regulation of Dkk-1 in Xt/Xt limb buds is independent of Shh. Shh is neither sufficient nor necessary to activate Dkk-1; however, it seems to be needed for the maintenance of Dkk-1 transcription. This effect of Shh on Dkk-1 is likely to be mediated by Bmp-4, which is down-regulated in Shh–/– limb buds, as we show here that Dkk-1 is rapidly induced by Bmp in vivo. Application of a bead soaked in Bmp to the undifferentiated mesenchyme induces Dkk-1 within 2 h and thereby leads to a subsequent down-regulation of the Wnt/β-catenin target gene Lef-1. In the corresponding region, massive PCD is induced, resulting in the loss of the radius. In addition, Mukhopadhyay et al. (2001) recently reported the activation of Dkk-1 transcription by BMP-2. Further evidence implicating an important endogenous function of Dkk-1 in modulating Bmp-triggered apoptosis comes from its ectopic expression and the ectopic PCD in Ft/+ mutant limb buds.
Dkk-1 also underlies positive regulation by Fgf signals from the AER. Fgf released from a bead led to a rapid induction of Dkk-1. In contrast to Bmp and consistent with previous studies (Montero et al., 2001 and references therein), Fgf treatment caused a significant mesodermal outgrowth during the first 8 h after its application. Thus, although Dkk-1 has the ability to induce apoptosis in the limb bud, the Fgf treatment did not cause apoptosis even after 18 h (data not shown). Strikingly, Fgf application led to increased transcriptional activity of the Wnt/β-catenin target gene Lef-1 in our experiments. Thus, the strong proliferative signal of increased Fgf and Wnt/β-catenin signaling overrides the pro-apoptotic effect of Dkk-1, leading to a net activation of Wnt signaling following this treatment. Fgfs have classically been regarded as factors promoting mesodermal proliferation (Martin, 1998). However, a recent paper by Montero et al. (2001) now provides evidence that Fgfs are also required for PCD in limb development. Fgf beads implanted in a comparable way to in the present study inhibited physiological cell death in the first 12 h; however, this was followed by an increase of PCD after 24 h and later. Their results further suggest that Bmps are not capable of inducing apoptosis in the absence of Fgf signals. The implication of Fgfs in apoptosis appears to result from their requirement for the expression of genes of the PCD cascade, such as Msx-2. A similar mechanism might hold true for the regulation of Dkk-1. By the implantation of Noggin beads we could show that Bmp signaling is necessary for Dkk-1 expression. The fact that Dkk-1 is rapidly down-regulated after surgical removal of the AER indicates a requirement of Fgf for Dkk-1 activation. Consistent with this, the Bmp-induced ectopic expression domain of Dkk-1 is always located between the bead and the AER. Thus, the greater distance from an Fgf source might explain why a Bmp bead implanted into the center of a HH26 limb bud did not induce Dkk-1 expression, which suggests that both Bmp and Fgf signaling are essential for Dkk-1 activation. This might indicate that the inhibition of Wnt/β-catenin signaling and the presence of Fgf are required for Bmp to induce apoptosis. In conclusion, concomitant activity of Bmp and Wnt/β-catenin pathways should lead to a completely different reaction in a cell. Indeed, Wnt/β-catenin signaling is implicated in the promotion of chondrogenesis in the chick limb bud (Hartmann and Tabin, 2000) and we have shown that Dkk-1 is not activated by Bmp when it promotes bone formation in vivo. This suggests that the co-ordinated regulation of the Bmp, Wnt/β-catenin and Fgf pathways is critical for the genetic program activated in limb mesodermal cells to undergo either apoptosis or chondrogenesis.
Mediation of the Bmp signal: a crucial role for c-Jun
As outlined above, Bmps can use at least two different intracellular pathways, the mutual interaction of which leads to the activation of a particular genetic program. We propose that the c-Jun-mediated activation of a Wnt/β-catenin inhibitor is fundamental for Bmp-induced apoptosis. The cytoplasmic kinase TAK-1 has been reported to be essential for Bmp-2-induced apoptosis, and Bmp-4 can also directly activate this kinase (Kimura et al., 2000 and references therein). There are several further implications for TAK-1 in apoptosis. Importantly, overexpression of TAK-1 in the Drosophila visual system leads to ectopically induced apoptosis mediated by JNK (Takatsu et al., 2000). Enhanced apoptosis has also been observed in transgenic frogs and mice overexpressing TAK-1 (Shibuya et al., 1998; Zhang et al., 2000). TAK-1 activates Jnk signaling, which in turn activates c-Jun. Transcription of c-Jun is then autoregulated by the c-Jun protein (Angel et al., 1988), the overexpression of which is sufficient to induce apoptosis (Bossy-Wetzel et al., 1997). This cascade might provide the link between Bmp and the induction of c-Jun that we report in this study. In our electroporations, c-Jun was sufficient to induce Dkk-1 expression, a potent inhibitor of the Wnt/β-catenin signaling pathway. Consistently, the TAK-1–MAPK-like pathway was recently shown to counteract the Wnt/β-catenin pathway (Ishitani et al., 1999). Based on our finding that Dkk-1 expression and induction by Bmp is severely hampered in c-Jun–/– cells, we show that c-Jun is also necessary for the full expression of Dkk-1, although the residual expression in these cells indicates that additional transcription factors contribute to Dkk-1 activation. However, our result that the induction of Dkk-1 is reduced to 22% of the wild-type level in c-Jun–/– MEF in response to Bmp highlights the importance of c-Jun for Dkk-1 activation by Bmp. The residual part of activation by Bmp could be mediated by Smads or other Ap-1 family members that are also activated by Jnk. As we were not able to detect Dkk-1 transcripts in Jnk-1–/– MEF by RT–PCR (data not shown), we conclude that the Bmp/Smad pathway plays, if any, only a minor role for transcription of Dkk-1. The Bmp-induced activation of Dkk-1 leads to an inhibition of Wnt/β-catenin signaling as shown by the dramatic down-regulation of Lef-1 transcription. Interestingly, increased Jnk activity has also been reported to cause a destabilization of β-catenin accompanied with increased apoptosis in several cell lines (Neo et al., 2000). We suggest that the predominant activation of a particular intracellular signaling cascade downstream of the Bmp receptor also contributes to the different effects that Bmps have on limb mesodermal cells. According to our model, Bmp would induce apoptosis when the Bmp/Jnk pathway dominates the Bmp/Smad pathway to activate certain genes, as is the case for Dkk-1. Further support for our model comes from a study showing that the distortion of positional information determined by dpp and wg signaling gradients leads to the activation of the DJNK apoptotic pathway, which subsequently induces cell death in the Drosophila wing (Adachi-Yamada et al., 1999). The authors suggested that this pathway is latent in normal wing development, but is activated upon abnormal dpp signaling to maintain proper development. Whether c-Jun activity is necessary for the physiological cell death occuring during vertebrate limb development is hard to judge, as c-Jun–/– embryos die at E13.5 (Hilberg et al., 1993) when the most prominent PCD in the interdigits of the limb starts. However, a recent study provided genetic evidence that c-Jun N-terminal phosphorylation is not essential for the developmental regulation of apoptosis, since it is not impaired in mice harboring a c-Jun allele that cannot be phosphorylated at serines 63 and 73 (Behrens et al., 1999). Thus, we cannot rule out the possibility that this pathway might only be used upon inappropriate signaling.
Regulation by stress signals
c-Jun activity is rapidly induced by exposure of cells to a variety of stress signals that commit a cell to apoptosis. In fact, c-Jun induction is the hallmark of the mammalian UV response and cells lacking c-Jun are less sensitive to UV-induced apoptosis (Shaulian et al., 2000). We now show that c-Jun is also immediately induced in chick embryos exposed to UV light. UV irradiation results in a rapid accumulation of p53, which leads to either cell cycle arrest or apoptosis. Recently, it was shown that constitutive expression of c-Jun is the critical event regulating p53-induced apoptosis upon UV irradiation (Shaulian et al., 2000). Remarkably, Dkk-1 has been suggested to be a p53 target gene, as it is induced by DNA damage in a p53-dependent manner in cultured cells (Wang et al., 2000). These findings, together with our findings that Dkk-1 is activated upon UV irradiation in chicken embryos and that Dkk-1 is a c-Jun target gene, provide strong evidence for an important role of Dkk-1-mediated Wnt/β-catenin inhibition downstream of p53. Recently, two papers revealed another way of linking genotoxic injury to the destruction of β-catenin. It was shown that the p53-inducible Siah-1 mediates β-catenin degradation independent of β-TrCP (Liu et al., 2001; Matsuzawa and Reed, 2001). In summary, these results suggest that blocking Wnt/β-catenin signaling might be a general requirement in apoptotic responses, as multiple ways exist to ensure degradation of β-catenin upon inappropriate stimuli.
Although p53 can induce Dkk-1 expression, three lines of evidence indicate that it is not required for it. First, staurosporin, which has been shown to induce apoptosis independently of p53 (Rocha et al., 2000), also strongly activates Dkk-1 expression. Secondly, the apoptosis-inducing ability of TGF-β family members also seems not to proceed via p53 (Selvakumaran et al., 1994; Yamamoto et al., 1996; Francis et al., 2000). Thirdly, Dkk-1 is expressed normally in limb buds of p53–/– embryos (data not shown), indicating that p53 is also dispensable for the normal expression of Dkk-1. On the other hand, the UV-induced expression of Dkk-1 is likely to be independent of Bmp signaling, as Bmp-4 is not transcriptionally induced in our assay. Thus, we conclude that Dkk-1 can be activated by multiple independent apoptotic stimuli. In contrast to p53, however, c-Jun is pivotal for the physiological expression of Dkk-1 and an important transcription factor for its induction by Bmp-4. Moreover, staurosporin-, H2O2- and UV-induced apoptosis also involve Jnk-mediated activation of Ap-1 activity and c-Jun induction (Buschmann et al., 2000; Chae et al., 2000; Shaulian et al., 2000; this study). Hence, we propose that multiple apoptotic pathways converge on c-Jun activation, resulting in increased Dkk-1 transcription.
In our experiments, overexpression of Dkk-1 with a retroviral vector led to severe distal truncations of the infected wing buds. These results are in line with those of Mukhopadhyay et al. (2001), who recently reported that ectopic Dkk-1 expression with an adenoviral vector in the chick limb bud produced distal truncations similar to those reported here. This phenotype might not result from enhanced PCD as we could not detect a significant increase in apoptotic cells before truncations were rather advanced. An alternative explanation would be that the Dkk-1 overexpression interferes with normal AER function, as Wnt/β-catenin signals have been shown to be essential for AER maintenance (Kawakami et al., 2001). This might impair distal outgrowth of the Dkk-1-infected wing buds, which then leads to more PCD.
By showing that Dkk-1 clearly promotes Bmp-triggered apoptosis, however, we provide direct evidence for the pro-apoptotic capability of Dkk-1. As Dkk-1 is induced by Bmp-4 under PCD-inducing conditions, this might suggest that antagonism of Wnt/β-catenin signals by Dkk-1 is even necessary for Bmp-triggered apoptosis to occur in limb mesodermal cells. In consequence, the loss of Dkk-1 function should lead to decreased PCD in areas where Bmps are known to be important endogenous regulators of PCD like, for example, the interdigital mesenchyme. Indeed, a strong argument in favor of this interpretation comes from the phenotype of Dkk-1–/– mice. The fusion of the digits observed in these mice is likely to result from decreased PCD (Mukhopadhyay et al., 2001).
It will be interesting to determine whether Dkk-1, as a secreted protein, exerts its pro-apoptotic function in a cell-autonomous or non-cell-autonomous manner. In conclusion, our study unravels a putative important function of Wnt/β-catenin antagonism by Dkk-1 in developmental and adaptive apoptosis. Furthermore, we show that the Ap-1 family member c-Jun is pivotal for Dkk-1 transcriptional activation.
Materials and methods
Details on Materials and methods are available at The EMBO Journal Online as Supplementary data, or on request.
Acknowledgments
Acknowledgements
We are indebted to Juan Carlos Izpisúa Belmonte for sharing reagents prior to publication with us. Thanks are due to Erwin Wagner and Uta Möhle-Steinlein, Thomas Brand, Christof Niehrs, Juan Carlos Izpisúa Belmonte, Benoît Robert, Brigid Hogan, Astrid Buchberger and Jens Böse for embryos, cells and reagents. L.G. is grateful to Juan Carlos Izpisúa Belmonte and members of his laboratory, especially Concepción Rodríguez Esteban, Dirk Büscher and Jennifer Ng, for introducing him into the chicken embryological system. We thank Michael Kessel and Andrea Vortkamp for suggestions on explant electroporation and organ culture, and Thomas Theil for critically reading this manuscript. This work was supported by a grant from the Deutsche Forschungsgemeinschaft to U.R. (Ru 376/8 and SFB 590).
References
- Adachi-Yamada T., Fujimura-Kamada,K., Nishida,Y. and Matsumoto,K. (1999) Distortion of proximodistal information causes JNK-dependent apoptosis in Drosophila wing. Nature, 400, 166–169. [DOI] [PubMed] [Google Scholar]
- Angel P., Hattori,K., Smeal,T. and Karin,M. (1988) The jun proto-oncogene is positively autoregulated by its product, Jun/AP1. Cell, 55, 875–885. [DOI] [PubMed] [Google Scholar]
- Behrens A., Sibilia,M. and Wagner,E.F. (1999) Amino-terminal phosphorylation of c-Jun regulates stress-induced apoptosis and cellular proliferation. Nature Genet., 21, 326–329. [DOI] [PubMed] [Google Scholar]
- Bejsovec A. (2000) Wnt signaling: an embarrassment of receptors. Curr. Biol., 10, R919–R922. [DOI] [PubMed] [Google Scholar]
- Bossy-Wetzel E., Bakiri,L. and Yaniv,M. (1997) Induction of apoptosis by the transcription factor c-Jun. EMBO J., 16, 1695–1709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckland R.A., Collinson,J.M., Graham,E., Davidson,D.R. and Hill,R.E. (1998) Antagonistic effects of FGF4 on BMP induction of apoptosis and chondrogenesis in the chick limb bud. Mech. Dev., 71, 143–150. [DOI] [PubMed] [Google Scholar]
- Büscher D. and Rüther,U. (1998) Expression profile of Gli family members and Shh in normal and mutant mouse limb development. Dev. Dyn., 211, 88–96. [DOI] [PubMed] [Google Scholar]
- Buschmann T., Yin,Z., Bhoumik,A. and Ronai,Z. (2000) Amino-terminal-derived JNK fragment alters expression and activity of c-Jun, ATF2 and p53 and increases H2O2-induced cell death. J. Biol. Chem., 275, 16590–16596. [DOI] [PubMed] [Google Scholar]
- Cadigan K.M. and Nusse,R. (1997) Wnt signaling: a common theme in animal development. Genes Dev., 11, 3286–3305. [DOI] [PubMed] [Google Scholar]
- Chae H.J., Kang,J.S., Byun,J.O., Han,K.S., Kim,D.U., Oh,S.M., Kim,H.M., Chae,S.W. and Kim,H.R. (2000) Molecular mechanism of staurosporine-induced apoptosis in osteoblasts. Pharmacol. Res., 42, 373–381. [DOI] [PubMed] [Google Scholar]
- Chiang C., Litingtung,Y., Lee,E., Young,K.E., Corden,J.L., Westphal,H. and Beachy,P.A. (1996) Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature, 383, 407–413. [DOI] [PubMed] [Google Scholar]
- Derijard B., Hibi,M., Wu,I.H., Barrett,T., Su,B., Deng,T., Karin,M. and Davis,R.J. (1994) JNK1: a protein kinase stimulated by UV light and Ha-Ras that binds and phosphorylates the c-Jun activation domain. Cell, 76, 1025–1037. [DOI] [PubMed] [Google Scholar]
- Duprez D., Bell,E.J., Richardson,M.K., Archer,C.W., Wolpert,L., Brickell,P.M. and Francis-West,P.H. (1996) Overexpression of BMP-2 and BMP-4 alters the size and shape of developing skeletal elements in the chick limb. Mech. Dev., 57, 145–157. [DOI] [PubMed] [Google Scholar]
- Francis J.M., Heyworth,C.M., Spooncer,E., Pierce,A., Dexter,T.M. and Whetton,A.D. (2000) Transforming growth factor-β 1 induces apoptosis independently of p53 and selectively reduces expression of Bcl-2 in multipotent hematopoietic cells. J. Biol. Chem., 275, 39137–39145. [DOI] [PubMed] [Google Scholar]
- Glinka A., Wu,W., Delius,H., Monaghan,A.P., Blumenstock,C. and Niehrs,C. (1998) Dickkopf-1 is a member of a new family of secreted proteins and functions in head induction. Nature, 391, 357–362. [DOI] [PubMed] [Google Scholar]
- Grotewold L., Theil,T. and Rüther,U. (1999) Expression pattern of Dkk-1 during mouse limb development. Mech. Dev., 89, 151–153. [DOI] [PubMed] [Google Scholar]
- Grotewold L., Plum,M., Dildrop,R., Peters,T. and Rüther,U. (2001) Bambi is coexpressed with Bmp-4 during mouse embryogenesis. Mech. Dev., 100, 327–330. [DOI] [PubMed] [Google Scholar]
- Hartmann C. and Tabin,C.J. (2000) Dual roles of Wnt signaling during chondrogenesis in the chicken limb. Development, 127, 3141–3159. [DOI] [PubMed] [Google Scholar]
- Heymer J. and Rüther,U. (1999) Syndactyly of Ft/+ mice correlates with an imbalance in Bmp4 and Fgf8 expression. Mech. Dev., 88, 173–181. [DOI] [PubMed] [Google Scholar]
- Hilberg F., Aguzzi,A., Howells,N. and Wagner,E.F. (1993) c-Jun is essential for normal mouse development and hepatogenesis. Nature, 365, 179–181. [DOI] [PubMed] [Google Scholar]
- Hurle J.M., Ros,M.A., Climent,V. and Garcia-Martinez,V. (1996) Morphology and significance of programmed cell death in the developing limb bud of the vertebrate embryo. Microsc. Res. Tech., 34, 236–246. [DOI] [PubMed] [Google Scholar]
- Ishitani T. et al. (1999) The TAK1–NLK–MAPK-related pathway antagonizes signalling between β-catenin and transcription factor TCF. Nature, 399, 798–802. [DOI] [PubMed] [Google Scholar]
- Janssen Y.M., Matalon,S. and Mossman,B.T. (1997) Differential induction of c-fos, c-jun and apoptosis in lung epithelial cells exposed to ROS or RNS. Am. J. Physiol., 273, L789–L796. [DOI] [PubMed] [Google Scholar]
- Kawakami Y., Capdevila,J., Büscher,D., Itoh,T., Rodriguez Esteban,C. and Izpisua Belmonte,J.C. (2001) WNT signals control FGF-dependent limb initiation and AER induction in the chick embryo. Cell, 104, 891–900. [DOI] [PubMed] [Google Scholar]
- Kimura N., Matsuo,R., Shibuya,H., Nakashima,K. and Taga,T. (2000) BMP-2 induced apoptosis is mediated by activation of the TAK1–p38 kinase pathway that is negatively regulated by Smad6. J. Biol. Chem., 275, 17647–17652. [DOI] [PubMed] [Google Scholar]
- Kyriakis J.M., Banerjee,P., Nikolakaki,E., Dai,T., Rubie,E.A., Ahmad,M.F., Avruch,J. and Woodgett,J.R. (1994) The stress-activated protein kinase subfamily of c-Jun kinases. Nature, 369, 156–160. [DOI] [PubMed] [Google Scholar]
- Liu J., Stevens,J., Rote,C.A., Yost,H.J., Hu,Y., Neufeld,K.L., White,R.L. and Matsunami,N. (2001) Siah-1 mediates a novel β-catenin degradation pathway linking p53 to the adenomatous polyposis coli protein. Mol. Cell, 5, 927–936. [DOI] [PubMed] [Google Scholar]
- Macias D., Ganan,Y., Sampath,T.K., Peidra,M.E., Ros,M.A. and Hurle,J.M. (1997) Role of BMP-2 and OP-1 (BMP-7) in programmed cell death and skeletogenesis during chick limb development. Development, 124, 1109–1117. [DOI] [PubMed] [Google Scholar]
- Mao B., Wu,W., Li,Y., Stannek,P., Glinka,A. and Niehrs,C. (2001) LDL-receptor-related protein 6 is a receptor for Dickkopf proteins. Nature, 411, 321–325. [DOI] [PubMed] [Google Scholar]
- Mao J. et al. (2001) Low-density lipoprotein receptor-related protein-5 binds to Axin and regulates the canonical Wnt signaling pathway. Mol. Cell, 4, 801–809. [DOI] [PubMed] [Google Scholar]
- Marazzi G., Wang,Y. and Sassoon,D. (1997) Msx2 is a transcriptional regulator in the BMP4-mediated programmed cell death pathway. Dev. Biol., 186, 127–138. [DOI] [PubMed] [Google Scholar]
- Martin G.R. (1998) The roles of FGFs in the early development of vertebrate limbs. Genes Dev., 12, 1571–1586. [DOI] [PubMed] [Google Scholar]
- Marvin M.J., Di Rocco,G., Gardiner,A., Bush,S.M. and Lassar,A.B. (2001) Inhibition of Wnt activity induces heart formation from posterior mesoderm. Genes Dev., 15, 316–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massagué J., Blain,S.W. and Lo,R.S. (2000) TGFβ signaling in growth control, cancer and heritable disorders. Cell, 103, 295–309. [DOI] [PubMed] [Google Scholar]
- Matsuzawa S.I. and Reed,J.C. (2001) Siah-1, SIP and Ebi collaborate in a novel pathway for β-catenin degradation linked to p53 responses. Mol. Cell, 5, 915–926. [DOI] [PubMed] [Google Scholar]
- Montero J.A., Gañan,Y., Macias,D., Rodriguez-Leon,J., Sanz-Ezquerro, J.J., Merino,R., Chimal-Monroy,J., Nieto,M.A. and Hurle,J.M. (2001) Role of FGFs in the control of programmed cell death during limb development. Development, 128, 2075–2084. [DOI] [PubMed] [Google Scholar]
- Mukhopadhyay M. et al. (2001) Dickkopf1 is required for embryonic head induction and limb morphogenesis in the mouse. Dev. Cell, 1, 423–434. [DOI] [PubMed] [Google Scholar]
- Neo S.Y., Zhang,Y., Yaw,L.P., Li,P. and Lin,S.C. (2000) Axin-induced apoptosis depends on the extend of its JNK activation and its ability to down-regulate β-catenin levels. Biochem. Biophys. Res. Commun., 272, 144–150. [DOI] [PubMed] [Google Scholar]
- Pessah M., Prunier,C., Marais,J., Ferrand,N., Mazars,A., Lallemand,F., Gauthier,J.M. and Atfi,A. (2001) c-Jun interacts with the corepressor TG-interacting factor (TGIF) to suppress Smad2 transcriptional activity. Proc. Natl Acad. Sci. USA, 98, 6198–6203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pizette S. and Niswander,L. (1999) BMPs negatively regulate structure and function of the limb apical ectodermal ridge. Development, 126, 883–894. [DOI] [PubMed] [Google Scholar]
- Polakis P. (2000) Wnt signaling and cancer. Genes Dev., 14, 1837–1851. [PubMed] [Google Scholar]
- Richter-Landsberg C. and Vollgraf,U. (1998) Mode of cell injury and death after hydrogen peroxide exposure in cultured oligodendroglia cells. Exp. Cell Res., 244, 218–229. [DOI] [PubMed] [Google Scholar]
- Rocha S., Soengas,M.S., Lowe,S.W., Glanzmann,C., Fabbro,D., Winterhalter,K., Bodis,S. and Pruschy,M. (2000) Protein kinase C inhibitor and irradiation-induced apoptosis: relevance of the cytochrome c-mediated caspase-9 death pathway. Cell Growth Differ., 11, 491–499. [PubMed] [Google Scholar]
- Sanz C., León,Y., Cañon,S., Alvarez,L., Giraldez,F. and Varela-Nieto,I. (1999) Pattern of expression of the Jun family of transcription factors during the early development of the inner ear: implications in apoptosis. J. Cell Sci., 112, 3967–3974. [DOI] [PubMed] [Google Scholar]
- Sanz-Ezquerro J.J. and Tickle,C. (2000) Autoregulation of Shh expression and Shh induction of cell death suggest a mechanism for modulating polarising activity during chick limb development. Development, 127, 4811–4823. [DOI] [PubMed] [Google Scholar]
- Selvakumaran M., Lin,H.K., Miyashita,T., Wang,H.G., Krajewski,S., Reed,J.C., Hoffman,B. and Liebermann,D. (1994) Immediate early up-regulation of bax expression by p53 but not TGF β 1: a paradigm for distinct apoptotic pathways. Oncogene, 6, 1791–1798. [PubMed] [Google Scholar]
- Shaulian E., Schreiber,M., Piu,F., Beeche,M., Wagner,E.F. and Karin,M. (2000) The mammalian UV response: c-Jun induction is required for exit from p53-imposed growth arrest. Cell, 103, 897–907. [DOI] [PubMed] [Google Scholar]
- Shibuya H., Iwata,H., Masuyama,N., Gotoh,Y., Yamaguchi,K., Irie,K., Matsumoto,K., Nishida,E. and Ueno,N. (1998) Role of TAK1 and TAB1 in BMP signaling in early Xenopus development. EMBO J., 17, 1019–1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinya M., Eschbach,C., Clark,M., Lehrach,H. and Furutani-Seiki,M. (2000) Zebrafish Dkk1, induced by the pre-MBT Wnt signaling, is secreted from the prechordal plate and patterns the anterior neural plate. Mech. Dev., 98, 3–17. [DOI] [PubMed] [Google Scholar]
- Takatsu Y., Nakamura,M., Stapleton,M., Danos,M.C., Matsumoto,K., O’Connor,M.B., Shibuya,H. and Ueno,N. (2000) TAK1 participates in c-Jun N-terminal kinase signaling during Drosophila development. Mol. Cell. Biol., 20, 3015–3026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogelstein B., Lane,D. and Levine,A.J. (2000) Surfing the p53 network. Nature, 408, 307–310. [DOI] [PubMed] [Google Scholar]
- Wang J., Shou,J. and Chen,X. (2000) Dickkopf-1, an inhibitor of the Wnt signaling pathway, is induced by p53. Oncogene, 19, 1843–1848. [DOI] [PubMed] [Google Scholar]
- Wilkinson D.G., Bhatt,S., Ryseck,R.P. and Bravo,R. (1989) Tissue-specific expression of c-jun and junB during organogenesis in the mouse. Development, 106, 465–471. [DOI] [PubMed] [Google Scholar]
- Wong C., Rougier-Chapman,E.M., Frederick,J.P., Datto,M.B., Liberati, N.T., Li,J.M. and Wang,X.F. (1999) Smad3–Smad4 and AP-1 complexes synergize in transcriptional activation of the c-Jun promoter by transforming growth factor β. Mol. Cell. Biol., 19, 1821–1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto M., Maehara,Y., Sakaguchi,Y., Kusumoto,T., Ichiyoshi,Y. and Sugimachi,K. (1996) Transforming growth factor-β 1 induces apoptosis in gastric cancer cells through a p53-independent pathway. Cancer, 77 (8 Suppl.), 1628–1633. [DOI] [PubMed] [Google Scholar]
- Yokouchi Y., Sakiyama,J., Kameda,T., Iba,H., Suzuki,A., Ueno,N. and Kuroiwa,A. (1996) BMP-2/-4 mediate programmed cell death in chicken limb buds. Development, 122, 3725–3734. [DOI] [PubMed] [Google Scholar]
- Zhang D., Gaussin,V., Taffet,G.E., Belaguli,N.S., Yamada,M., Schwartz, R.J., Michael,L.H., Overbeek,P.A. and Schneider,M.D. (2000) TAK1 is activated in the myocardium after pressure overload and is sufficient to provoke heart failure in transgenic mice. Nature Med., 5, 556–563. [DOI] [PubMed] [Google Scholar]
- Zhang Y., Feng,X.H. and Derynck,R. (1998) Smad3 and Smad4 cooperate with c-Jun/c-Fos to mediate TGF-β-induced transcription. Nature, 394, 909–913. [DOI] [PubMed] [Google Scholar]
- Zorn A.M. (2001) Wnt signaling: antagonistic Dickkopfs. Curr. Biol., 11, R592–R595. [DOI] [PubMed] [Google Scholar]
- Zou H., Choe,K.M., Lu,Y., Massagué,J. and Niswander,L. (1997) BMP signaling and vertebrate limb development. Cold Spring Harb. Symp. Quant. Biol., 62, 269–272. [PubMed] [Google Scholar]