Abstract
West Nile virus (WNV) is a mosquito-borne flavivirus that is neurotropic in humans, birds, and other animals. While adaptive immunity plays an important role in preventing WNV spread to the central nervous system (CNS), little is known about how alpha/beta interferon (IFN-α/β) protects against peripheral and CNS infection. In this study, we examine the virulence and tropism of WNV in IFN-α/β receptor-deficient (IFN- α/βR−/−) mice and primary neuronal cultures. IFN-α/βR−/− mice were acutely susceptible to WNV infection through subcutaneous inoculation, with 100% mortality and a mean time to death (MTD) of 4.6 ± 0.7 and 3.8± 0.5 days after infection with 100 and 102 PFU, respectively. In contrast, congenic wild-type 129Sv/Ev mice infected with 102 PFU showed 62% mortality and a MTD of 11.9 ± 1.9 days. IFN-α/βR−/− mice developed high viral loads by day 3 after infection in nearly all tissues assayed, including many that were not infected in wild-type mice. IFN-α/βR−/− mice also demonstrated altered cellular tropism, with increased infection in macrophages, B cells, and T cells in the spleen. Additionally, treatment of primary wild-type neurons in vitro with IFN-β either before or after infection increased neuronal survival independent of its effect on WNV replication. Collectively, our data suggest that IFN-α/β controls WNV infection by restricting tropism and viral burden and by preventing death of infected neurons.
West Nile virus (WNV) is a single-stranded, positive-sense RNA virus of the Flaviviridae family that is maintained in nature through a mosquito-bird-mosquito transmission cycle. Humans, horses, and other animals are incidental hosts. WNV is endemic in Africa, Asia, Europe, and Australia and has spread rapidly across the United States following its introduction in 1999 (31). Humans develop a febrile illness, which may progress to a lethal encephalitis with symptoms including cognitive dysfunction, muscle weakness, flaccid paralysis, and seizures (6, 44, 54). No specific therapy for WNV is currently approved for use in humans.
In humans, depressed immunity and increased age correlate with greater risk for neurological disease (7, 20, 26, 29). Recent studies have begun to elucidate the immunological basis for control of WNV. In rodent models, an intact adaptive immune response is required for resistance to and control of WNV infection. Humoral responses and cytotoxic T lymphocytes play critical roles in controlling WNV as both B-cell-deficient and CD8-deficient mice show increased lethality and viral burden (15, 56, 65). CD8-deficient mice also show delayed clearance of virus from the central nervous system (CNS), demonstrating that cytotoxic T lymphocyte responses are important for recovery from WNV infection (56). At present, less is known regarding the role of innate immunity in controlling WNV infection in both the periphery and the CNS. Interestingly, a recent paper demonstrated that a deficiency in Toll-like receptor 3 (TLR3) was associated with enhanced survival, presumably because the TLR3-dependent innate immune response in the periphery caused a loss of integrity of the blood-brain barrier (64).
Alpha/beta interferon (IFN-α/β) comprises an important innate immune system control against viral infections (reviewed in references 21 and 43). In general, IFN-α/β induces an antiviral state within cells through the upregulation and activation of antiviral proteins (e.g., RNA-activated protein kinase, RNaseL, and Mx) and by modulating adaptive immune responses (12, 51). IFNs also independently affect the survival of diverse cell populations. For example, IFN-α/β prolongs neuron and astrocyte survival following growth factor deprivation or serum starvation, yet can also induce apoptosis of tumor cells (2, 8, 52). It has been hypothesized that the ability of IFNs to inhibit cell death may preserve neuronal populations and limit disease in the CNS induced by either viral infection or inflammation (66).
Pretreatment of cells in vitro with IFN-α/β potently inhibits flaviviruses (1, 13, 14, 19, 25). However, the inhibitory effect of IFN is significantly attenuated after viral replication has begun (14, 34). Several groups have demonstrated that flavivirus nonstructural proteins specifically inhibit IFN-α/β signaling by preventing JAK1 and Tyk2 phosphorylation and IFN-β gene transcription (24, 34-36, 41). Although flaviviruses resist IFN-α/β-induced responses following infection, additional experiments suggest that IFNs still play a significant role in immune system defense. IFN-α has been successfully used to treat WNV encephalitis in two human cases and resulted in an improvement of CNS disease (28). Limited in vivo experiments have demonstrated that mice lacking the IFN-α/β receptor (IFN-α/βR−/−) show increased lethality and viral burden after Murray Valley encephalitis virus (MVE) infection (37). In contrast, infection of IFN-α/βR−/− mice with dengue virus (DEN) resulted in earlier dissemination and increased viral burden but did not affect lethality (55). However, the studies with MVE and DEN did not address the mechanisms by which IFN-α/β controls flavivirus infection in the periphery or the CNS. In this study, we evaluated the role of IFN-α/β in controlling WNV replication, spread, and tropism in the periphery, as well as its ability to inhibit replication in the CNS and directly modulate neuronal survival.
MATERIALS AND METHODS
Mice.
Wild-type 129Sv/Ev and C57BL/6J mice were obtained commercially. IFN-α/βR−/− mice with 129Sv/Ev and C57BL/6J backgrounds were obtained from H. Virgin (Washington University, St. Louis, MO) and J. Sprent (Scripps Institute, San Diego, CA), respectively. All mice were genotyped and bred in the animal facility of Washington University School of Medicine, and experiments were approved and performed in accordance with Washington University animal studies guidelines.
Mouse experiments and quantitation of viral burden.
The WNV strain (3000.0259) was isolated in New York in 2000 and passaged once in C6/36 cells to generate a stock virus that was used in all experiments. Eight- to 10-week-old mice were used for all in vivo studies. Peripheral infection was performed by footpad inoculation of 100 to 102 PFU of virus diluted in Hanks balanced salt solution (HBSS) with 1% heat-inactivated fetal bovine serum (FBS). Intracranial inoculation was performed using 101 PFU of virus diluted in 10 μl HBSS with 1% heat-inactivated FBS. To analyze the kinetics of viral production, mice were infected by footpad inoculation with 102 PFU of virus and euthanized on day 1, 2, 3, 6, 8, or 10. Blood was collected by phlebotomy of the axillary vein, and serum was recovered following centrifugation, aliquoted, and stored at −80°C. Mice were perfused with 20 ml PBS, and organs were removed, weighed, and homogenized. Plaque assays were performed as previously described using BHK21-15 cells (15). Viral RNA was prepared from aliquots of serum using a Qia-Amp RNA recovery kit (QIAGEN, Valencia, VA) and quantitated by real-time fluorogenic reverse transcription-PCR (RT-PCR) as previously described (15) using primers and probes corresponding to the E gene of WNV: forward, 5′-TCAGCGATCTCTCCACCAAAG-3′; reverse, 5′-GGGTCAGCACGTTTGTCATTG-3′; probe, 5′-FAM-TGCCCGACCATG GGAGAAGCTC-3′-TAMRA (30). (FAM is 6-carboxyfluorescein; TAMRA is 6-carboxytetramethylrhodamine.)
IFN detection and quantitation.
Total RNA was prepared from brain regions of uninfected and infected C57BL/6J mice with the RNEasy kit per the manufacturer's instructions (QIAGEN, Valencia, VA). Samples were quantitated via spectrophotometry and DNase treated (Invitrogen, Carlsbad, CA). Primers and probes used for fluorogenic quantitative RT-PCR for IFN-α and IFN-β have been described and validated previously (64). IFN-α primers were as follows: forward, 5′-CTTCCACAGGATCACTGTGTACCT-3′; reverse, 5′-TTCTGCTCTGACCACCTCCC-3′; and probe, 5′-FAM-AGAGAGAAGAAACACAGCCCCTGTGCC-TAMRA-3′. IFN-β primers were as follows: forward, 5′-CTGGAGCAGCTGAATGGAAAG-3′; reverse, 5′-CTTCTCCGTCATCTCCATAGGG-3′; and probe, 5′-FAM-CAACCTCACCTACAGGGCGGACTTCAAG-TAMRA-3′. Each 25-μl reaction mixture contained 2 μl RNA, 12.5 μl 2× RT-PCR mixture, 0.625 μl 40× Multiscribe (Applied Biosystems), 25 pmol of each primer and 5 pmol probe. RT-PCR was performed in 96-well optical reaction plates (Applied Biosystems, Foster City, CA) with an ABI 7000 sequence detection system. The thermal cycling reaction was as follows: 48°C for 30 min, 95°C for 10 min, and 40 cycles, each consisting of 95°C for 15 s and 60°C for 1 min. To normalize the samples, fluorogenic RT-PCR was performed in parallel on 18S rRNA (Applied Biosystems). For each sample, the normalized value was obtained by subtracting the amount of amplified gene from the amount of 18S rRNA. Relative increases (n-fold) were determined by calculating the difference between the normalized amplified gene in infected and uninfected samples. IFN-α enzyme-linked immunosorbent assay was performed with serum harvested from uninfected and infected C57BL/6J mice with the Mouse Interferon Alpha ELISA kit per the manufacturer's instructions (PBL Biomedical Laboratories, Piscataway, NJ).
Immunohistochemistry. (i) Tissue preparation.
WNV infected and uninfected mice were anesthetized with xylazine and ketamine and then perfused with 20 ml of phosphate-buffered saline (PBS), followed by 20 ml of 4% paraformaldehyde (PFA) in PBS. Tissues were dissected, placed in 4% PFA at 4°C overnight, and then embedded in paraffin. Immunohistochemistry was performed with serial 6-μm sections.
(ii) Tissue staining.
WNV antigen was detected by coupling the catalyzed signal amplification and animal research kits essentially per the manufacturer's instructions (Dako Cytomation, Carpinteria, CA) with the following modifications. Paraffin-embedded sections were deparaffinized and then treated using Antigen Retrieval solution at 95°C for 20 min (DAKO Cytomation). To detect WNV infection, three anti-WNV monoclonal antibodies that recognize the WNV E protein (E18, E22, and E31) (42) were used at a 1:1:1 ratio. Antibodies were biotinylated using the animal research kit system and used at a final concentration of 1.25 μg/ml for 30 min at room temperature. Sections were developed using diaminobenzadine and then counterstained using hematoxylin QS (Vector Laboratories, Burlingame, CA).
Flow cytometry.
Spleens were dissected from WNV-infected 129Sv/Ev and congenic IFN-α/βR−/− mice on day 3; single-cell suspensions were generated. Cells were washed with DMEM, and red blood cells were depleted with ACK lysis buffer (0.15 M NH4Cl, 10 mM KHCO3, 0.1 mM Na2EDTA, pH 7.4). Splenocytes were incubated with Fcγ receptor block and stained using fluorescently conjugated antibodies for CD3, CD4, CD8, B220 (BD Pharmingen, San Diego, CA), and F4/80 (Serotec, Inc., Raleigh, NC) in Ca2+/Mg2+-free HBSS with 1% bovine serum albumin (BD Pharmingen). Stained cells were fixed with 4% PFA in PBS, followed by permeabilization with a 0.1% (wt/vol) saponin solution in HBSS and a second Fcγ receptor block. WNV was detected using a 1:1:1 mixture of three APC conjugated anti-WNV E protein monoclonal antibodies (E16, E22, and E33) (42). Data were collected using a FACScalibur flow cytometer and analyzed using CellQuest software (Becton Dickinson, Franklin Lanes, NJ).
Strand-specific real-time RT-PCR.
Spleens were dissected from WNV-infected 129Sv/Ev and congenic IFN-α/βR−/− mice on day 3, and splenocytes were isolated as described above. CD11b+ and CD19+ cells were selected using antibody-conjugated magnetic beads per the manufacturer's instructions (Miltenyi Biotec, Auburn, CA). The purity of isolated cells was assessed by flow cytometry using fluorescently conjugated antibodies against the CD11b and B220 antigens (BD Pharmingen, San Diego, CA). RNA was extracted from purified cells using an RNEasy kit per the manufacturer's instructions (QIAGEN, Valencia, VA). Strand-specific real-time RT-PCR was performed using a T7-tagged primer as a modification of a procedure previously described for hepatitis C virus (32). Each 25-μl reaction mixture contained 2 μl RNA, 12.5 μl 2× RT-PCR mixture, 0.625 μl 40× Multiscribe (Applied Biosystems), and 2 pmol of either positive-strand primer T7E1229R (5′-GCGTAATACGACTCACTATAGGGTCAGCACGTTTGTCATTG-3′) or negative-strand primer T7E1160F (5′-GCGTAATACGACTCACTATATCAGCGATCTCTCCAC CAAAG-3′). The boldface and underlined sequences correspond to the T7 tag and WNV E gene, respectively. Reaction mixtures were incubated for 30 min at 55°C and 95°C for 10 min, followed by the addition of 5 pmol of probe (5′-FAM-CAACCTCACCTACAGGGCGGACTTCAAG-TAMRA-3′) and 20 pmol each of primers T7 (5′-GCGTAATACGACTCACTATA-3′) and E1160F to positive-strand reaction mixtures and primers T7 and E1229R to negative-strand reaction mixtures. The thermal cycling reaction was then allowed to proceed (40 cycles, each consisting of 95°C for 15 s and 60°C for 1 min). Normalized values were obtained after the amount of 18S rRNA (Applied Biosystems) in each sample was determined by fluorogenic RT-PCR. Relative increases (n-fold) were determined by calculating the difference between the normalized Ct values for infected IFN-α/βR−/− and congenic wild-type 129Sv/Ev mice.
Neuronal cultures.
Primary sympathetic neuronal cultures were generated from superior cervical ganglia (SCG) of postnatal day 1 129Sv/Ev and congenic IFN-α/βR−/− mice, essentially as previously described (17). Briefly, dissected SCG were treated with 1-mg/ml collagenase for 30 min at 37°C (Worthington Biochemical Corporation, Lakewood, NJ), followed by treatment with 0.25% trypsin for 30 min at 37°C (Sigma-Aldrich, St. Louis, MO). The ganglia were dissociated using a p100 pipette tip and seeded at a density of 6,000 cells/well onto 24-well plates or 8-well chamber slides (Nunc, Naperville, IL) coated with rat tail collagen. Following dissection, SCG were cultured for 3 days in minimal essential medium (Sigma-Aldrich, St. Louis, MO) containing 10% heat-inactivated FBS, 2 mM l-glutamine, 30 μM fluorodeoxyuridine, 30 μM uridine, 50-ng/ml nerve growth factor (Harlan Bioproducts, Madison, WI), and 3.3-ng/ml aphidicolin (Fisher Scientific, Pittsburgh, PA). Neurons were then maintained in aphidicolin-free medium. Purity of SCG cultures was determined via staining with anti-NeuN and anti-glial fibrillary acidic protein antibodies (∼98 to 99%). All experiments were performed on neurons cultured 6 to 7 days prior to treatment or infection.
Virus infection of neuronal cultures and IFN treatment.
For all in vitro experiments, primary SCG neurons were infected at a multiplicity of infection (MOI) of 10 for 1 h at 37°C. Free virus was then removed by a serial wash with PBS, followed by a wash 1 h later with neuronal medium. For IFN treatment experiments, SCG were treated with 100-IU/ml of mouse IFN-α, IFN-β, IFN-γ, or IFN-γ plus IFN-β (PBL Laboratories, Madison, WI). For IFN pretreatment experiments, neurons were treated 24 h before infection at an MOI of 10. Supernatants were harvested at 24 h postinfection (p.i.) after IFN-α, IFN-β, IFN-γ, or IFN-γ plus IFN-β treatment and on days 2, 3, 4, 8, and 11 after IFN-β treatment. In all cases, free virus was removed after extensive washing 24 h prior to the indicated collection time point. Production of infectious virus was measured by plaque assay with Vero cells as described previously (15). For IFN posttreatment experiments, neurons were infected at an MOI of 10 for 24 h, followed by treatment with IFN for 12 h. Cells were then washed to remove free virus. Supernatants were harvested on day 2 following IFN-α, IFN-β, IFN-γ, or IFN-γ plus IFN-β treatment and on days 3, 4, 8, and 11 postinfection after IFN-β treatment, following extensive washing as described above. To further assess viral replication, RNA was harvested from untreated and IFN-treated SCG neurons on days 1 and 4 after infection using a RNEasy recovery kit (QIAGEN, Valencia, VA) and quantitated by strand-specific, real-time fluorogenic RT-PCR as described above.
In vitro neuronal survival studies. SCG neurons were treated with 100 IU of IFN-β/ml either 24 h prior to or after infection with an MOI of 10 or infected in the absence of IFN treatment. The medium was changed every 3 days, and IFN-treated cells were maintained in the presence of 100-IU/ml IFN-β throughout the course of the experiment. On days 4, 8, and 11 postinfection, cultures were harvested to quantitate neuronal survival. Cells were washed twice with PBS, fixed with 4% PFA in PBS overnight at 4°C, and then stained with toluidine blue (final concentration, 0.25g/liter). Neurons that were stained for Nissl substance and displayed intact nuclear and cellular morphology were considered alive and were counted as previously described (45). The data were normalized to untreated, uninfected neurons harvested at the same time points.
Immunofluorescence microscopy of neuronal cells.
SCG neurons were infected with WNV at an MOI 10 or treated with 100 IU of IFN-β 24 h prior to or after infection. At 4 days after infection, WNV-infected and control SCG were fixed overnight with 4% PFA in PBS at 4°C. Cells were permeabilized in PBS with 0.2% Triton X-100, blocked (5% normal goat serum and 0.1% Tween 20), and stained with a rat anti-WNV polyclonal antibody and a Cy3-conjugated goat anti-rat secondary antibody (Jackson Laboratories, West Grove, PA). Cells were counterstained with DAPI (4′,6′-diamidino-2-phenylindole) and visualized using a Zeiss Axiovert 200 microscope.
Statistical analysis.
For in vitro experiments, an unpaired t test was used to determine significant differences. For viral burden analysis, an unpaired t test with Welch's correction was used. For survival analysis, Kaplan-Meier curves were analyzed by the log rank test.
RESULTS
WNV infection induces IFN-α/β production.
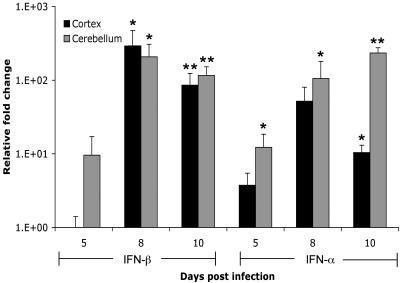
IFN-α/β is rapidly produced following infection with several RNA viruses and plays key roles in initiating antiviral defenses (12). To determine the time course and amount of IFN-α/β produced following WNV infection, we measured the levels of IFN-α in the serum of wild-type mice subcutaneously inoculated with 102 PFU. By day 2 p.i., an average of 3.0 ± 0.4 ng/ml of IFN-α was detected in serum, and production was maintained for several days following infection (days 4 to 8; range, 2.2 ± 0.3 to 3.1 ± 2.6 ng/ml). Following a primary viremia, WNV crosses the blood-brain barrier and infects the CNS, resulting in neuronal injury. However, the production and role of IFN-α/β in the CNS following RNA virus infection are largely unknown. We assessed the induction of IFN-α/β in both the cerebellum and cortex of wild-type mice, following peripheral inoculation. By day 5 p.i., IFN-α mRNA was increased 10 fold in the cortex, whereas in the cerebellum both IFN-α and IFN-β transcripts were increased 5 to 10 fold (Fig. 1). The peak induction of IFN-β (200- to 300-fold increase; P < 0.05) mRNA was observed on day 8, correlating with peak virus infection. IFN-α transcript levels also peaked in the cortex on day 8, while in the cerebellum the highest amounts were detected on day 10 (P < 0.05).
FIG. 1.
Induction of IFN-α/β mRNA in brains of mice infected with WNV. Mice were inoculated with 102 PFU of WNV by footpad injection and sacrificed on days 5, 8, and 10 p.i. Total RNA from frontal cortices (black bars) and cerebella (gray bars) was analyzed for the expression of 18S rRNA, IFN-α mRNA, and IFN-β mRNA by fluorogenic quantitative RT-PCR. Data are expressed as the relative increase (n-fold) over RNA isolated from uninfected controls. Average values are from three to four mice, and error bars indicate standard deviations. Asterisks indicate differences that are statistically significant (*, P < 0.05; **, P < 0.005).
Susceptibility of IFN-α/βR−/− mice to peripheral WNV infection.
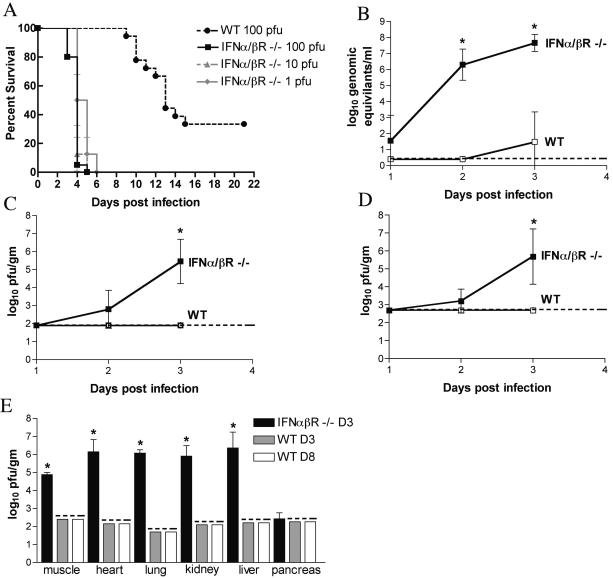
To directly investigate the role of IFN-α/β in protection against WNV in vivo, we assessed morbidity and mortality after subcutaneous infection of wild-type and congenic IFN- α/βR−/− 129Sv/Ev mice (Fig. 2A). Following footpad inoculation with 100, 101, or 102 PFU, all IFN-α/βR−/− mice showed severe clinical symptoms by day 3, including hunching, fur ruffling, and greatly reduced activity. Death rapidly followed the onset of symptoms, occurring within 12 to 48 h. In contrast, wild-type mice did not show clinical signs of infection until day 8 and experienced a more protracted illness that was typified by weight loss, fur ruffling, hunching, and occasional tremors. IFN-α/βR−/− mice showed extreme vulnerability to WNV infection, with an MTD of 3.8 ± 0.5 days and 100% lethality by day 5 following infection with 102 PFU (Fig. 2A). In contrast, congenic wild-type 129Sv/Ev mice demonstrated an MTD of 11.9 ± 1.9 days and 38% survival following infection with 102 PFU (P < 0.001). To confirm that the phenotype observed in IFN-α/βR−/− mice was independent of strain background, we also infected IFN-α/βR−/− mice on the C57BL/6 background. These mice also rapidly and uniformly succumbed to infection, showing 100% lethality and a mean survival time of 3.4 ± 0.5 days (data not shown). These data suggest that a functional IFN-α/β system is essential for protection of mice from fatal WNV infection.
FIG. 2.
Survival and virologic analysis of wild-type and IFN-αβR−/− 129Sv/Ev mice. (A) Eight- to 10-week-old mice were inoculated with 100 to 102 PFU of WNV by footpad injection and followed for 21 days. The survival curves were constructed using data from two to three independent experiments. The numbers of animals were as follows: 18 wild-type mice and 20 IFN-α/βR−/− mice infected with 102 PFU and 8 IFN-α/βR−/− mice infected with 101 and 100 PFU. Survival differences between wild-type and IFN-α/βR−/− mice were statistically significant at all viral doses (P < 0.001). (B to E) Viral burden after WNV infection. WNV RNA levels in the serum (B) and viral loads in the brain (C) and spinal cord (D) were determined from samples harvested on days 1, 2, and 3 p.i. with 102 PFU by quantitative RT-PCR or by plaque assay. (E) Viral burden in muscle, heart, lung, kidney, liver, and pancreas was determined on day 3 after infection for IFN-α/βR−/− mice and on days 3 and 8 for wild-type mice. Data are shown as the average PFU per gram of tissue and reflect four to eight mice per group. The dotted line represents the limit of sensitivity of the assay. The viral burden in IFN-α/βR−/− mice was statistically significant in tissue samples as indicated by asterisks (*, P < 0.05).
IFN-α/β is required for controlling viral replication and restricting tissue tropism.
Since IFN-α/β has an established role in controlling replication and dissemination of other viruses, we predicted that the increased susceptibility of IFN- α/βR−/− mice to WNV infection would correspond to higher viral loads in peripheral and CNS tissues. To test this, wild-type and congenic IFN-α/βR−/− mice were infected with 102 PFU, and viral loads were measured in different tissues.
(i) Viremia.
In wild-type mice, viremia was undetectable at 24 and 48 h after infection. Using a more sensitive quantitative RT-PCR assay, low-level viremia was observed at 72 h p.i. (Fig. 2B), results that agree with previous studies of WNV infection in C57BL/6 mice (15, 16). In contrast, high levels of infectious virus were detected in the serum of IFN-α/βR−/− mice (107 PFU/ml) by viral plaque assay. Using RT-PCR, viral RNA was detected in serum in IFN-α/βR−/− mice at 24 h; by 48 and 72 h, 106 and 108 viral RNA copies/ml were measured, respectively (Fig. 2B).
(ii) CNS.
Following subcutaneous infection of IFN-α/βR−/− mice, WNV rapidly disseminated to the CNS. Virus was detected by 48 h p.i. in IFN-α/βR−/− mice in both the brain and spinal cord (Fig. 2C and D). By 72 h p.i., viral loads in CNS tissues averaged 106 PFU/g, corresponding to the onset of severe symptoms and death. In marked contrast, no virus was detected in wild-type 129Sv/Ev mice by plaque assay of either the brain or spinal cord at these time points. Instead, WNV spread to the CNS in wild-type mice by day 6 p.i. At day 10 p.i., wild-type mice demonstrated high viral loads in CNS compartments, with an average of 6 × 106 PFU/g and 4.7 × 104 PFU/g in the brain and spinal cord, respectively.
(iii) Peripheral tissues.
In addition to controlling viral replication, IFN-α/β has been implicated in modulating viral tropism (38, 40, 50). To determine whether the lack of IFN- α/β-induced responses altered infection of peripheral tissues, heart, lung, kidney, liver, pancreas, and muscle were harvested from IFN-α/βR−/− mice on day 3 p.i. and from wild-type mice on days 3 and 8 p.i. Despite excessive perfusion to remove blood-borne virus, high viral loads (105 to 106.5 PFU/g) were detected in every peripheral tissue tested from the IFN- α/βR−/− mice with the exception of the pancreas. In comparison, no virus was measured in wild-type mice at either day 3 or day 8 after infection in any of these tissues (Fig. 2E). Thus, IFN-α/β restricts viral replication in peripheral tissues and alters viral tropism at the tissue level.
Immunohistochemistry of WNV infection in IFN-α/βR−/− mice.
To evaluate how an absence of IFN-α/β-induced responses alters viral infection, we examined brain, spinal cord, and liver for WNV antigen staining from IFN-α/βR−/− mice on day 3 p.i. and from symptomatic wild-type mice on day 10 p.i. Immunohistochemistry showed extensive localization of WNV antigen in neurons throughout the brain and spinal cord of IFN-α/βR−/− mice. Intense antigen staining was observed in neurons in the Purkinje layer of the cerebellum, brain stem, hippocampus, and spinal cord (Fig. 3A). In symptomatic wild-type mice, less-intense antigen staining in CNS tissues was noted, with sporadic infection of neurons in these tissues (Fig. 3A). Interestingly, abundant viral infection was observed in the epithelial cells of the choroid plexus of IFN-α/βR−/− mice; this was largely lacking in wild-type mice (Fig. 3A), suggesting an additional possible route of entry for WNV into the CNS.
FIG. 3.
Immunohistochemistry of WNV-infected IFN-α/βR−/− and wild-type mice. (A) WNV antigen expression within CNS tissues of IFN-α/βR−/− and wild-type mice. Representative images from the cerebellum, brain stem, hippocampus, choroid plexus, and spinal cord are shown from day 3 p.i. for IFN-α/βR−/− and day 10 p.i. for wild- type mice after review of brains from three or four independent experiments for each group. Inset images show spinal cord staining at a higher magnification (×400). Staining in neurons (black arrows) and within cells of the choroid plexus (blue arrows) is indicated. (B) Detection of WNV antigen in livers of infected IFN-α/βR−/− and wild-type mice at day 3 and day 10 p.i., respectively. Infection of cells lining the sinusoids is indicated (red arrows). All sections were counterstained with hematoxylin; magnifications are ×200 to 400.
The presence of high viral loads in many peripheral tissues of IFN-α/βR−/− mice suggested that IFN-α/β functions to restrict replication in these tissues. To begin to define the cellular basis for the change in tissue tropism, additional immunohistochemistry was performed. Analysis of liver sections revealed WNV infection predominately in small cells lining the sinusoids that are most likely myeloid-derived Kupfer cells (Fig. 3B). In contrast, WNV antigen was largely absent from other parenchymal cells, as little to no infection of hepatocytes was observed.
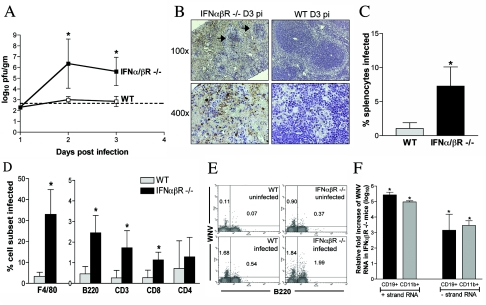
Splenic infection in IFN-α/βR−/− mice.
Our virologic studies indicated that mice lacking the IFN-α/β receptor had increased levels of infection in the spleen (Fig. 4A). Consistent with this, higher levels of viral antigen were observed with spleens from IFN-α/βR−/− mice on day 3 p.i. (Fig. 4B). Marked destruction of splenic architecture and a distinct loss of cellularity were observed in infected IFN-α/βR−/− mice compared to infected wild-type animals and uninfected IFN-α/βR−/− controls (Fig. 4B and data not shown). Quantitation confirmed this, as IFN-α/βR−/− mice contained 28% ± 12% splenocytes compared to age- and sex-matched uninfected IFN-α/βR−/− mice, whereas infected wild-type mice contained 124% ± 48% splenocytes compared to wild-type controls (P < 0.001). By immunohistochemistry, WNV antigen appeared to be present in an array of cell types in spleens from IFN-α/βR−/− mice. To more rigorously determine the cellular basis of this enhanced infection, we evaluated different splenocyte populations for WNV infection by flow cytometry. Splenocytes were costained for WNV and individual immune cell markers and analyzed. We observed both an increase in the total number of splenocytes infected in IFN-α/βR−/− mice compared to wild-type (7.3% ± 2.2% versus 1.0% ± 0.9%; P = 0.0001) and increased infection in several cell populations (Fig. 4C and D). CD3+, CD8+, and B220+ (lymphocyte) cells all showed significantly increased WNV infection in IFN-α/βR−/− mice (Fig. 4D and E). However, F4/80+ (macrophage) cells demonstrated the greatest increase in susceptibility in IFN-α/βR−/− mice (33% ± 12% versus 3.3% ± 2.0% in wild-type mice; P < 0.0001). Strand-specific RT-PCR confirmed the presence of replicating virus in myeloid and lymphoid cell populations from IFN-α/βR−/− mice (Fig. 4F). Positive-strand RNA was increased 5 to 6 logs in CD11b+ and CD19+ cells relative to levels detected in infected wild-type mice. Significant levels of negative-strand RNA were also detected, with a 3- to 4-log increase in both CD11b+ and CD19+ cells from IFN-α/βR−/− mice. Thus, an absence of IFN-α/β-induced responses altered cellular tropism in the spleen, with macrophages becoming especially sensitive to increased infection.
FIG. 4.
IFN-α/βR−/− mice show increased viral replication and altered tropism in the spleen. (A) Viral burden in the spleen of IFN-α/βR−/− and wild-type mice was determined from samples harvested on days 1, 2, and 3 p.i. by plaque assay. Data are shown as the average PFU per gram of tissue and represent average values from five mice per group. Error bars indicate standard deviations, and the dotted line represents the limit of sensitivity of the assay. (B) Immunohistochemical analysis of WNV antigen within spleen sections from WNV-infected IFN-α/βR−/− and wild-type mice at day 3 p.i. At low magnification, marked destruction of splenic architecture is observed in infected IFN-α/βR−/− mice with disrupted follicle structures (black arrows). Data are representative of staining performed on tissues from three mice per group. (C to F) Analysis of splenocytes derived from 8- to 9-week-old wild-type and IFN-α/βR−/− mice at day 3 p.i. Cells were costained for WNV antigen and cellular markers to determine the total percentage of splenocytes infected (C) and the percentage of the individual cell population infected (D). Data were averaged from four independent experiments performed on a total of five to nine age- and sex-matched mice per group. Error bars indicate standard deviations, and asterisks indicate differences that are statistically significant (*, P < 0.05). (E) Representative flow cytometry histograms of B220+ and WNV antigen-positive costaining in wild-type and IFN-α/βR−/− mice are shown. The numbers indicate the percentages of positive cells in each quadrant. (F) Total RNA from CD11b+ and CD19+ cells purified from infected spleens was analyzed for the levels of WNV-positive and -negative strands using strand-specific real-time RT-PCR. Data were normalized to 18S rRNA levels and are expressed as the relative severalfold increases in WNV RNA detected in IFN-α/βR−/− mice compared to that in infected wild-type mice. Average values are from six mice per group.
The affect of IFN-α/β in the CNS after intracranial infection.
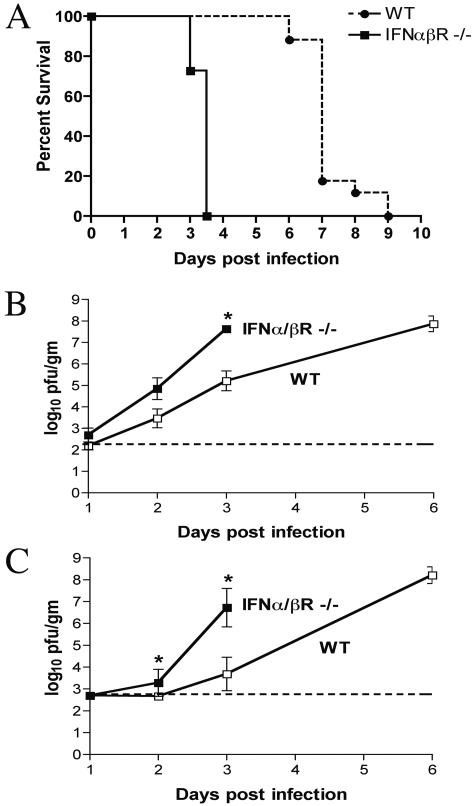
Since WNV is naturally acquired through peripheral inoculation and subsequently spreads to the CNS, an effective immune response to WNV controls viral replication both in the periphery and CNS compartments. Our experiments using a subcutaneous infection model showed that IFN-α/β is critical for controlling peripheral viral replication and spread (Fig. 2). To determine whether IFN-α/β plays an independent role in the CNS following WNV infection, we inoculated wild-type and IFN-α/βR−/− mice intracranially with 101 PFU of virus and examined lethality and viral burden in CNS tissues. IFN-α/βR−/− mice demonstrated uniform lethality by day 3.5 p.i. and an MTD of 3.4 ± 0.2 days, whereas wild-type mice had complete lethality by day 9 p.i., with an MTD of 7.2 ± 0.8 days (P < 0.0001) (Fig. 5A). Virologic analysis showed significantly increased titers in the brain and spinal cord of IFN-α/βR−/− mice by day 3 p.i. compared to those of wild-type mice (Fig. 5B and C).
FIG. 5.
Survival data for wild-type and IFN-α/βR−/− mice, following intracranial inoculation. (A) Eight- to 10-week-old mice were inoculated with 101 PFU of WNV by intracranial injection. The survival curves were constructed using data from two independent experiments with 20 wild-type mice and 11 IFN-α/βR−/− mice. The difference in the mean time to death between wild-type and IFN-α/βR−/− mice was statistically significant (P < 0.001). (B and C) Viral burden in CNS tissues after intracranial WNV infection. Viral load in the brain (B) and spinal cord (C) was determined by plaque assay from samples harvested on days 1, 2, and 3 p.i. for IFN-α/βR−/− and wild-type mice as well as day 6 p.i. for wild-type mice. Data are shown as the average PFU per gram of tissue and reflect four to eight mice per group. The dotted line represents the limit of sensitivity of the assay; asterisks indicate values that are statistically significant (*, P < 0.05) compared to data for wild-type mice.
IFN-α/β inhibits WNV replication in neurons.
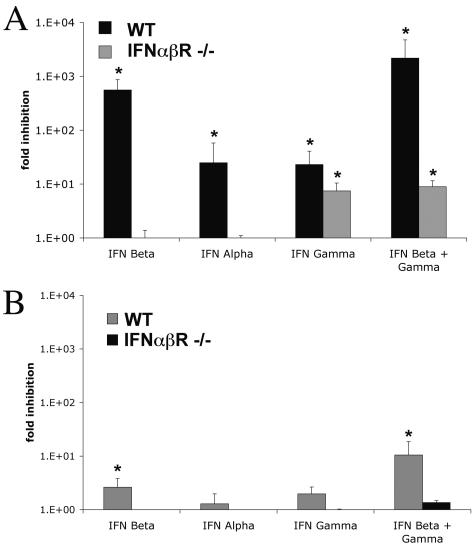
Although the in vivo studies suggested that IFN-α/β modulates viral replication within CNS tissues, it was unclear whether this was due to specific antiviral activity in neurons. To test directly whether IFN-α/β inhibited WNV replication in neurons, we utilized a primary neuron culture model for WNV infection. Neurons derived from SCG are of high purity (∼98 to 99%) and are susceptible to WNV infection (46). Pretreatment of SCG neurons with IFN-β inhibited WNV 1,000 fold, whereas treatment with IFN-α or IFN-γ decreased viral production by 100 fold (P < 0.05) (Fig. 6A). Treatment with a combination of IFN-β and IFN-γ resulted in the greatest inhibition, reducing viral production by 1,500 fold (P < 0.05). Similar results were obtained when strand-specific real-time RT-PCR was used to analyze viral RNA from untreated and IFN-β-pretreated cells: at day 1 p.i., neurons pretreated with IFN-β had 1,000-fold- and 100-fold-lower levels of negative-strand and positive-strand viral RNA, respectively (data not shown). As expected, IFN treatment following WNV infection less efficiently blocked viral replication, with 5-fold and 10-fold inhibition of viral production following IFN-β or IFN-β and IFN-γ treatment, respectively (P < 0.05) (Fig. 6B). Thus, similar to nonneuronal cells, primary neurons respond to IFN by efficiently inhibiting viral production when treated prior to but not after WNV infection.
FIG. 6.
IFN-α/β inhibits WNV infection in neurons. Primary cultures of SCG neurons were prepared from wild-type and IFN-α/βR−/− mice. (A) Neurons were pretreated 24 h prior to infection with 100 IU/ml of the indicated mouse IFN. Cells were infected at an MOI of 10 and evaluated for production of infectious WNV at 24 h. (B) Neurons were infected at an MOI of 10 for 24 h, treated with 100 IU/ml of the indicated mouse IFN for 12 h, washed, and then evaluated for WNV production at 48 h. Data represent an average of three independent experiments, and asterisks indicate values that are statistically significant (*, P < 0.05) compared to untreated cells.
WNV-induced neuronal death is inhibited by IFN-α/β.
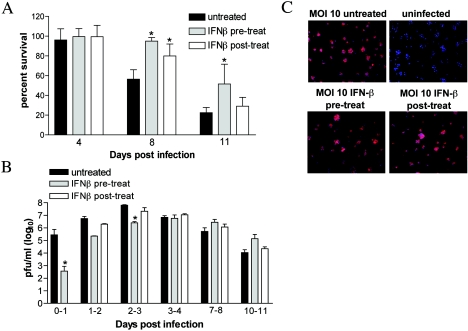
Following intracranial inoculation, wild-type mice had viral loads in the CNS tissues at day 6 p.i. that were equivalent to those detected with IFN-α/βR−/− mice at day 3 (Fig. 5B and C). However, complete lethality was not observed in wild-type mice until day 9 p.i., indicating that some mice survived for several days despite apparently high viral titers in the brain and spinal cord. We hypothesized that IFN-α/β may have an independent function that allows prolonged survival in the presence of high viral titers. To test this, SCG neurons were treated with 100 IU of IFN-β either 24 h prior to or after infection with WNV, and neuronal survival was quantitated over time. Treatment with IFN-β either prior to or after infection significantly increased neuronal survival on day 8 p.i. (pretreated 95% survival, posttreated 80% survival, and untreated 56% survival; P < 0.05). By day 11 p.i., IFN-β-pretreated cells still showed improved survival over untreated cells, demonstrating a 50% survival increase (P = 0.01) (Fig. 7A). Because increased survival of IFN-β treated neurons could be due to a difference in the relative production of WNV, we measured WNV levels in IFN-β-treated and untreated cells at 24-h intervals throughout the time course of infection. At days 2 and day 3 p.i., virus infection was slightly reduced in IFN-β-pretreated cells. Interestingly, after day 3, equivalent levels of WNV were recovered from cell supernatants, independent of IFN treatment. These data were confirmed by strand-specific RT-PCR at day 4 p.i., which showed only 0.8- and 0.7-fold inhibition of positive- and negative-strand viral RNA, respectively (data not shown). Indeed, by day 10 to 11 IFN-β-pretreated cells paradoxically produced more virus than untreated or posttreated cells; however, this may be due to the increased survival rate of IFN-treated neurons (Fig. 7B). To assess whether IFN-β treatment altered the number of neurons infected, immunofluorescent staining for WNV antigen was performed at day 4 p.i. Regardless of IFN-β treatment, virtually all neurons stained positive for WNV antigen (Fig. 7C). These data suggest that IFN-β prolongs neuronal survival following WNV infection in a manner that is at least partially independent of its antiviral effect.
FIG. 7.
IFN-β prolongs neuronal survival following WNV infection. SCG neurons from wild-type mice were infected with WNV at an MOI of 10. Neurons were either untreated (black bars) or treated 24 h prior to (gray bars) or after (white bars) infection with 100 IU IFN-β. Cells were subsequently maintained in the presence of IFN-β throughout the time course. (A) Neuronal survival was quantitated on days 4, 8, and 11 p.i. Approximately 1,000 to 2,000 cells were counted per condition in each replicate. Data are expressed relative to survival of uninfected neurons and represent the average of four independent experiments. Error bars indicate standard deviations. (B) Infectious virus production at 24 intervals following IFN-β treatment prior to or after infection was assayed on days 1 to 2, 2 to 3, 3 to 4, 7 to 8, and 10 to 11 p.i. Viral production from days 0 to 1 was assayed for IFN-β pretreatment only, as posttreatment occurred at day 1 p.i. (C) WNV antigen expression (red) was determined on day 4 p.i. by immunofluorescence microscopy. DAPI was used as a nuclear counterstain (blue). Asterisks indicate differences that are statistically significant (*, P < 0.05).
DISCUSSION
In this study, we used several experimental approaches to demonstrate that IFN-α/β plays a critical role in the early control of WNV replication and spread. IFN-α/β was produced rapidly in the serum and upregulated in the brains of wild-type mice following WNV infection. Mice that lacked a functional IFN-α/β receptor had increased viremia and viral burden in the majority of peripheral and CNS tissues and quickly succumbed to infection. Additional experiments showed that IFN-α/β had an independent role in protecting CNS tissues and functioned to control WNV replication and inhibit WNV-induced death of primary neurons.
IFN-α/β acts as a key innate defense against WNV infection.
In vitro studies have demonstrated that pretreatment with IFN-α/β can reduce flavivirus replication in several cell populations, including Vero cells and human foreskin fibroblasts, although treatment after infection is generally less effective (1, 13, 14, 25, 34). Moreover, mouse embryonic fibroblasts that lack the IFN-α/β receptor show increased susceptibility to WNV infection (10). However, the potential role of endogenous IFN-α/β in modulating WNV infection in vivo was untested. Our experiments demonstrate that IFN-α/β is essential for controlling WNV infection. IFN-α/βR−/− mice had an MTD of 3.8 ± 0.5 days and 100% lethality following peripheral inoculation, whereas wild-type mice showed 60% mortality and an MTD of 11.9 ± 1.9 days. Complete lethality was also observed with IFN-α/βR−/− mice, following inoculation of as little as 100 PFU of WNV, indicating that these mice are more sensitive to WNV infection than our plaque assay. Thus, in the absence of IFN-α/β responses, mice are acutely susceptible to WNV infection. IFN-α/β has also been shown to play a protective role for other flaviviruses, although in varying degrees. For example, the same IFN-α/βR−/− mice had no lethality following intravenous DEN infection with 108 PFU, although increased viral replication and spread were detected in some tissues (55). In contrast, intravenous infection of 6-week-old IFN-α/βR−/− mice with 102 PFU MVE resulted in uniform lethality with kinetics similar to those observed for WNV, as all mice died by day 6. Moreover, increased MVE loads were detected in the serum, brain, and spleen, although other tissues were not assayed (37). Other studies using IFN-α/βR−/− mice have also shown that IFN-α/β plays an important role in neurotropic RNA virus infections, including Sindbis virus (50), Theiler's virus (18), Venezuelan equine encephalitis virus (22), and measles virus (40). For example, infection of IFN-α/βR−/− mice with Theiler's virus resulted in increased lethality, following inoculation with both virulent and attenuated strains (18). However, unlike several of these viruses, mice absolutely require IFN-α/β for survival following WNV infection.
We observed swift dissemination following WNV infection in IFN-α/βR−/− mice, resulting in high viremia, seeding of visceral organs (such as heart, lung, and liver) not normally infected in wild-type mice, and rapid spread to CNS tissues. Thus, IFN-α/β rapidly restricts WNV replication following infection, and its absence permits uncontrolled viral replication, dissemination, and death. In vitro, WNV has been shown to be acutely sensitive to IFNs when cells are treated prior to infection, but like other flaviviruses, this sensitivity is largely lost when cells are treated after WNV infection is established (24, 36). Recent studies demonstrate that flavivirus nonstructural proteins can inhibit IFN-α/β signaling by preventing phosphorylation of JAK1 and Tyk2 and also decrease IFN gene transcription (24, 34-36, 41). However, our data suggest that despite the ability of WNV to resist the antiviral effects of IFN-α/β after infection, it still plays an essential role in inhibiting WNV replication and spread in vivo.
IFN-α/β modulates WNV tropism.
Our viral burden data suggest that WNV tropism is modulated in part by IFN-α/β-induced responses. Following infection, the early production of IFN-α/β can limit WNV infection by inducing an antiviral state in neighboring cells and distant tissues. Correspondingly, we observed that WNV spread to an array of peripheral tissues in IFN-α/βR−/− mice that are not normally infected in wild-type mice. These results are consistent with infection of IFN- α/βR−/− mice for other viruses, including herpes simplex virus (38), Sindbis virus (50), measles virus (40), and myxoma virus (62), in which the absence of downstream IFN signaling expanded the range and species of cells infected. It still remains unclear whether IFN-α/β restricts tissue tropism in WNV infection through the induction of an antiviral state or whether higher viremia increases exposure of peripheral organs to infectious virus.
IFN-α/βR−/− mice also had significantly increased viral burden in the spleen, and immunohistochemical analysis showed marked destruction of splenic architecture, as well as widespread infection compared to infected wild-type mice. Although altered tissue tropism has been reported with IFN- α/βR−/− mice, limited work has been performed to define the cell populations that are protected by IFN-α/β in vivo. Flow cytometric analysis of splenocytes demonstrated significantly increased infection in CD3+, CD8+, B220+, and F4/80+ cells in IFN-α/βR−/− mice. Thus, in the spleen IFN-α/β restricts WNV replication in a diverse array of cell populations, including B cells, T cells, and macrophages. In the livers of IFN- α/βR−/− mice, viral antigen localized predominantly to what appear to be myeloid-derived Kupfer cells lining the sinusoids. Analogously, for Sindbis virus, in the absence of the IFN-α/β receptor viral antigen colocalized with macrophage and/or dendritic cells in the spleen and F4/80+ macrophages in the liver (50). Thus, at least for WNV and Sindbis virus, IFN-α/β may modulate tropism by restricting infection in myeloid-derived cells. Although the precise mechanisms by which IFN-α/β restricts WNV cellular tropism remain unclear, given the increasing number of candidate cell entry receptors for flaviviruses (9, 11, 48, 59, 60), IFN may regulate tropism at a postentry level, perhaps by depleting and/or modifying critical host or viral factors that are necessary for viral translation or replication. Alternatively, cell-specific differences in the levels or activities of IFN-stimulated antiviral proteins (e.g., RNA-activated protein kinase or RNaseL) could determine the outcome of infection in a given cell type.
Role of IFN-α/β in CNS protection following WNV infection.
Once viral infection in the CNS occurs, effective resolution requires clearance without irreversible damage of critical nonrenewing cells such as neurons. For WNV, CD8+ T cells have been shown to play an essential role in eliminating WNV infection in CNS tissues (56, 65). Our experiments suggest that IFN-α/β has an independent role in controlling viral infection and injury in the CNS. Peripheral WNV infection induces significant expression of both IFN-α and IFN-β in the brain, indicating that IFN-α/β may act directly in the CNS to restrict viral replication. Moreover, following intracranial inoculation with WNV, IFN-α/βR−/− mice show a significantly decreased average survival time, with complete mortality observed by day 3.5 compared to day 9 in wild-type mice. Although the IFN- α/βR−/− mice likely die as a result of the observed increase in CNS infection, heightened viral burdens in peripheral tissues could contribute. Our data agree with an intracranial inoculation study with Theiler's virus that showed increased lethality and demyelization in brains of IFN-α/βR−/− mice compared to the wild type (18). In contrast, a deficiency in TLR3 was associated with increased survival following peripheral WNV infection and equivalent survival following intracerebroventricular inoculation (64). Although further study is required, because of the significant differences in phenotypes it would appear that IFN-α/β signaling in the CNS is initiated primarily through a TLR3-indepdendent pathway.
To study the function of IFN-α/β in neurons in a more controlled system, we infected primary sympathetic motor neurons (SCG) with WNV. In contrast to embryonic stem cell-derived glutaminergic neurons, which rapidly die following WNV infection (57), SCG neurons sustain viral replication for up to 12 days, similar to dorsal root ganglia sensory neurons (27). Nonetheless, as with embryonic stem cell-derived neurons (B. Shrestha and M. S. Diamond, unpublished data), pretreatment of SCG neurons with IFN-α/β markedly inhibited negative-strand accumulation and WNV production. These data are consistent with studies showing that IFN-β pretreatment of neuroblastoma cells and primary olfactory neurons strongly inhibits vesicular stomatitis virus (61). For WNV, however, treatment of neurons after infection was significantly less effective in blocking viral production. These results show that primary neurons can effectively respond to IFN, but as seen in other cells, WNV becomes resistant once infection is established.
Following intracranial inoculation, CNS tissues of wild-type mice at day 6 had equivalent viral burdens compared to IFN-α/βR−/− mice at day 3 after infection. Despite this, mortality of all the wild-type mice did not occur until day 9, suggesting that IFN-α/β may have an independent function in protecting some neuronal populations. IFN-α/β has been shown to affect survival of a number of cell types, including neurons, astrocytes, neutrophils, and peripheral B cells (2, 8, 49, 63). Indeed, IFN-α/β has been hypothesized to contribute to neuronal preservation following viral infection (23, 66). Our data with SCG neurons showed increased survival rates after exposure to IFN-β. Although this could be explained by a decrease in viral replication, viral production after day 3 p.i. was equivalent in IFN-β-treated and untreated neurons. Moreover, at late time points after infection, IFN-β-pretreated cells produced more virus than untreated cells, likely secondary to increased neuronal survival. To our knowledge, this is the first study that demonstrates the neuroprotective effects of IFN-α/β following virus infection. These studies are consistent with the neuroprotective effect of IFN-γ following Sindbis virus infection in vitro and in vivo (3, 5).
The importance of IFN-α/β in inhibiting flavivirus infection has been confirmed in other in vivo models. Pretreatment of rodents with IFN-α inhibited St. Louis encephalitis virus infection and resulted in decreased viral loads and mortality for Modoc virus and WNV (4, 33, 39). Although no large-scale trials with humans have assessed the effectiveness of IFN-α/β therapy in WNV infection, pilot studies suggest that it may have therapeutic potential. Treatment with IFN-α reduced complications in human St. Louis encephalitis virus cases and has been used successfully, albeit in an uncontrolled manner, to treat small numbers of human cases of WNV encephalitis (28, 47, 53). However, in Vietnam, a double-blinded, randomized placebo-controlled clinical trial was performed on 1,112 children with suspected or documented Japanese encephalitis virus infection; treatment with IFN-α2a failed to improve outcome (58). Although no consensus has emerged, a randomized nonblinded clinical trial of IFN-α2b has been initiated (http://nyhq.org/posting/rahal.html) for WNV infection. Given our findings that IFN-α/β inhibits infection and prolongs survival in neurons, we agree that further study of IFN therapy late in the course of infection is warranted, as it could have beneficial effects in mitigating long-term sequelae associated with neuronal death.
Acknowledgments
We thank R. Klein, E. Johnson, V. Miller, K. Blight, and members of their laboratories for experimental advice. We thank H. Virgin and J. Sprent for the IFN-α/βR−/− mice and D. Leib, Deborah Lenschowd, and M. Gale for critical reading of the manuscript.
This work was supported by a New Scholar Award in Global Infectious Disease from the Ellison Medical Foundation (M.S.D.) and by a Predoctoral Fellowship from the Howard Hughes Medical Institute (M.A.S.).
REFERENCES
- 1.Anderson, J. F., and J. J. Rahal. 2002. Efficacy of interferon alpha-2b and ribavirin against West Nile virus in vitro. Emerg. Infect. Dis. 8:107-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barca, O., S. Ferre, M. Seoane, J. M. Prieto, M. Lema, R. Senaris, and V. M. Arce. 2003. Interferon beta promotes survival in primary astrocytes through phosphatidylinositol 3-kinase. J. Neuroimmunol. 139:155-159. [DOI] [PubMed] [Google Scholar]
- 3.Binder, G. K., and D. E. Griffin. 2001. Interferon-gamma-mediated site-specific clearance of alphavirus from CNS neurons. Science 293:303-306. [DOI] [PubMed] [Google Scholar]
- 4.Brooks, T. J., and R. J. Phillpotts. 1999. Interferon-alpha protects mice against lethal infection with St. Louis encephalitis virus delivered by the aerosol and subcutaneous routes. Antiviral Res. 41:57-64. [DOI] [PubMed] [Google Scholar]
- 5.Burdeinick-Kerr, R., and D. E. Griffin. 2005. Gamma interferon-dependent, noncytolytic clearance of Sindbis virus infection from neurons in vitro. J. Virol. 79:5374-5385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ceausu, E., S. Erscoiu, P. Calistru, D. Ispas, O. Dorobat, M. Homos, C. Barbulescu, I. Cojocaru, C. V. Simion, C. Cristea, C. Oprea, C. Dumitrescu, D. Duiculescu, I. Marcu, C. Mociornita, T. Stoicev, I. Zolotusca, C. Calomfirescu, R. Rusu, R. Hodrea, S. Geamai, and L. Paun. 1997. Clinical manifestations in the West Nile virus outbreak. Rom. J. Virol. 48:3-11. [PubMed] [Google Scholar]
- 7.Chambers, T. J., and M. S. Diamond. 2003. Pathogenesis of flavivirus encephalitis. Adv. Virus Res. 60:273-342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chang, J. Y., D. P. Martin, and E. M. Johnson, Jr. 1990. Interferon suppresses sympathetic neuronal cell death caused by nerve growth factor deprivation. J. Neurochem. 55:436-445. [DOI] [PubMed] [Google Scholar]
- 9.Chen, Y., T. Maguire, R. E. Hileman, J. R. Fromm, J. D. Esko, R. J. Linhardt, and R. M. Marks. 1997. Dengue virus infectivity depends on envelope protein binding to target cell heparan sulfate. Nat. Med. 3:866-871. [DOI] [PubMed] [Google Scholar]
- 10.Cheng, Y., N. J. King, and A. M. Kesson. 2004. Major histocompatibility complex class I (MHC-I) induction by West Nile virus: involvement of 2 signaling pathways in MHC-I up-regulation. J. Infect. Dis. 189:658-668. [DOI] [PubMed] [Google Scholar]
- 11.Chu, J. J., and M. L. Ng. 2004. Interaction of West Nile virus with αVβ3 integrin mediates virus entry into cells. J. Biol. Chem. 279:54533-54541. [DOI] [PubMed] [Google Scholar]
- 12.Colonna, M., G. Trinchieri, and Y. J. Liu. 2004. Plasmacytoid dendritic cells in immunity. Nat. Immunol. 5:1219-1226. [DOI] [PubMed] [Google Scholar]
- 13.Crance, J. M., N. Scaramozzino, A. Jouan, and D. Garin. 2003. Interferon, ribavirin, 6-azauridine and glycyrrhizin: antiviral compounds active against pathogenic flaviviruses. Antiviral Res. 58:73-79. [DOI] [PubMed] [Google Scholar]
- 14.Diamond, M. S., T. G. Roberts, D. Edgil, B. Lu, J. Ernst, and E. Harris. 2000. Modulation of dengue virus infection in human cells by alpha, beta, and gamma interferons. J. Virol. 74:4957-4966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Diamond, M. S., B. Shrestha, A. Marri, D. Mahan, and M. Engle. 2003. B cells and antibody play critical roles in the immediate defense of disseminated infection by West Nile encephalitis virus. J. Virol. 77:2578-2586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Diamond, M. S., E. M. Sitati, L. D. Friend, S. Higgs, B. Shrestha, and M. Engle. 2003. A critical role for induced IgM in the protection against West Nile virus infection. J. Exp. Med. 198:1853-1862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Easton, R. M., T. L. Deckwerth, A. S. Parsadanian, and E. M. Johnson, Jr. 1997. Analysis of the mechanism of loss of trophic factor dependence associated with neuronal maturation: a phenotype indistinguishable from Bax deletion. J. Neurosci. 17:9656-9666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fiette, L., C. Aubert, U. Muller, S. Huang, M. Aguet, M. Brahic, and J. F. Bureau. 1995. Theiler's virus infection of 129Sv mice that lack the interferon alpha/beta or interferon gamma receptors. J. Exp. Med. 181:2069-2076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fredericksen, B. L., M. Smith, M. G. Katze, P. Y. Shi, and M. Gale, Jr. 2004. The host response to West Nile Virus infection limits viral spread through the activation of the interferon regulatory factor 3 pathway. J. Virol. 78:7737-7747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Granwehr, B. P., K. M. Lillibridge, S. Higgs, P. W. Mason, J. F. Aronson, G. A. Campbell, and A. D. Barrett. 2004. West Nile virus: where are we now? Lancet Infect. Dis. 4:547-556. [DOI] [PubMed] [Google Scholar]
- 21.Gresser, I. 1997. Wherefore interferon? J. Leukoc. Biol. 61:567-574. [DOI] [PubMed] [Google Scholar]
- 22.Grieder, F. B., and S. N. Vogel. 1999. Role of interferon and interferon regulatory factors in early protection against Venezuelan equine encephalitis virus infection. Virology 257:106-118. [DOI] [PubMed] [Google Scholar]
- 23.Griffin, D. E. 2003. Immune responses to RNA-virus infections of the CNS. Nat. Rev. Immunol. 3:493-502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guo, J. T., J. Hayashi, and C. Seeger. 2005. West Nile virus inhibits the signal transduction pathway of alpha interferon. J. Virol. 79:1343-1350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Harinasuta, C., C. Wasi, and S. Vithanomsat. 1984. The effect of interferon on Japanese encephalitis virus in vitro. Southeast Asian J. Trop. Med. Public Health 15:564-568. [PubMed] [Google Scholar]
- 26.Huhn, G. D., C. Austin, C. Langkop, K. Kelly, R. Lucht, R. Lampman, R. Novak, L. Haramis, R. Boker, S. Smith, M. Chudoba, S. Gerber, C. Conover, and M. S. Dworkin. 2005. The emergence of West Nile virus during a large outbreak in Illinois in 2002. Am. J. Trop. Med. Hyg. 72:768-776. [PubMed] [Google Scholar]
- 27.Hunsperger, E., and J. T. Roehrig. 2005. Characterization of West Nile viral replication and maturation in peripheral neurons in culture. J. Neurovirol. 11:11-22. [DOI] [PubMed] [Google Scholar]
- 28.Kalil, A. C., M. P. Devetten, S. Singh, B. Lesiak, D. P. Poage, K. Bargenquast, P. Fayad, and A. G. Freifeld. 2005. Use of interferon-alpha in patients with West Nile encephalitis: report of 2 cases. Clin. Infect. Dis. 40:764-766. [DOI] [PubMed] [Google Scholar]
- 29.Kleinschmidt-DeMasters, B. K., B. A. Marder, M. E. Levi, S. P. Laird, J. T. McNutt, E. J. Escott, G. T. Everson, and K. L. Tyler. 2004. Naturally acquired West Nile virus encephalomyelitis in transplant recipients: clinical, laboratory, diagnostic, and neuropathological features. Arch. Neurol. 61:1210-1220. [DOI] [PubMed] [Google Scholar]
- 30.Lanciotti, R. S., A. J. Kerst, R. S. Nasci, M. S. Godsey, C. J. Mitchell, H. M. Savage, N. Komar, N. A. Panella, B. C. Allen, K. E. Volpe, B. S. Davis, and J. T. Roehrig. 2000. Rapid detection of West Nile virus from human clinical specimens, field-collected mosquitoes, and avian samples by a TaqMan reverse transcriptase-PCR assay. J. Clin. Microbiol. 38:4066-4071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lanciotti, R. S., J. T. Roehrig, V. Deubel, J. Smith, M. Parker, K. Steele, B. Crise, K. E. Volpe, M. B. Crabtree, J. H. Scherret, R. A. Hall, J. S. MacKenzie, C. B. Cropp, B. Panigrahy, E. Ostlund, B. Schmitt, M. Malkinson, C. Banet, J. Weissman, N. Komar, H. M. Savage, W. Stone, T. McNamara, and D. J. Gubler. 1999. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 286:2333-2337. [DOI] [PubMed] [Google Scholar]
- 32.Lanford, R. E., C. Sureau, J. R. Jacob, R. White, and T. R. Fuerst. 1994. Demonstration of in vitro infection of chimpanzee hepatocytes with hepatitis C virus using strand-specific RT/PCR. Virology 202:606-614. [DOI] [PubMed] [Google Scholar]
- 33.Leyssen, P., C. Drosten, M. Paning, N. Charlier, J. Paeshuyse, E. De Clercq, and J. Neyts. 2003. Interferons, interferon inducers, and interferon-ribavirin in treatment of flavivirus-induced encephalitis in mice. Antimicrob. Agents Chemother. 47:777-782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lin, R. J., C. L. Liao, E. Lin, and Y. L. Lin. 2004. Blocking of the alpha interferon-induced Jak-Stat signaling pathway by Japanese encephalitis virus infection. J. Virol. 78:9285-9294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liu, W. J., H. B. Chen, X. J. Wang, H. Huang, and A. A. Khromykh. 2004. Analysis of adaptive mutations in Kunjin virus replicon RNA reveals a novel role for the flavivirus nonstructural protein NS2A in inhibition of beta interferon promoter-driven transcription. J. Virol. 78:12225-12235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu, W. J., X. J. Wang, V. V. Mokhonov, P. Y. Shi, R. Randall, and A. A. Khromykh. 2005. Inhibition of interferon signaling by the New York 99 strain and Kunjin subtype of West Nile virus involves blockage of STAT1 and STAT2 activation by nonstructural proteins. J. Virol. 79:1934-1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lobigs, M., A. Mullbacher, Y. Wang, M. Pavy, and E. Lee. 2003. Role of type I and type II interferon responses in recovery from infection with an encephalitic flavivirus. J. Gen. Virol. 84:567-572. [DOI] [PubMed] [Google Scholar]
- 38.Luker, G. D., J. L. Prior, J. Song, C. M. Pica, and D. A. Leib. 2003. Bioluminescence imaging reveals systemic dissemination of herpes simplex virus type 1 in the absence of interferon receptors. J. Virol. 77:11082-11093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Morrey, J. D., C. W. Day, J. G. Julander, L. M. Blatt, D. F. Smee, and R. W. Sidwell. 2004. Effect of interferon-alpha and interferon-inducers on West Nile virus in mouse and hamster animal models. Antivir. Chem. Chemother. 15:101-109. [DOI] [PubMed] [Google Scholar]
- 40.Mrkic, B., J. Pavlovic, T. Rulicke, P. Volpe, C. J. Buchholz, D. Hourcade, J. P. Atkinson, A. Aguzzi, and R. Cattaneo. 1998. Measles virus spread and pathogenesis in genetically modified mice. J. Virol. 72:7420-7427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Munoz-Jordan, J. L., G. G. Sanchez-Burgos, M. Laurent-Rolle, and A. Garcia-Sastre. 2003. Inhibition of interferon signaling by dengue virus. Proc. Natl. Acad. Sci. USA 100:14333-14338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Oliphant, T., M. Engle, G. E. Nybakken, C. Doane, S. Johnson, L. Huang, S. Gorlatov, E. Mehlhop, A. Marri, K. M. Chung, G. D. Ebel, L. D. Kramer, D. H. Fremont, and M. S. Diamond. 2005. Development of a humanized monoclonal antibody with therapeutic potential against West Nile virus. Nat. Med. 11:522-530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pestka, S., C. D. Krause, and M. R. Walter. 2004. Interferons, interferon-like cytokines, and their receptors. Immunol. Rev. 202:8-32. [DOI] [PubMed] [Google Scholar]
- 44.Petersen, L. R., and A. A. Marfin. 2002. West Nile virus: a primer for the clinician. Ann. Intern. Med. 137:173-179. [DOI] [PubMed] [Google Scholar]
- 45.Pierchala, B. A., R. C. Ahrens, A. J. Paden, and E. M. Johnson, Jr. 2004. Nerve growth factor promotes the survival of sympathetic neurons through the cooperative function of the protein kinase C and phosphatidylinositol 3-kinase pathways. J. Biol. Chem. 279:27986-27993. [DOI] [PubMed] [Google Scholar]
- 46.Pierson, T. C., M. S. Diamond, A. A. Ahmed, L. E. Valentine, C. W. Davis, M. A. Samuel, S. L. Hanna, B. A. Puffer, and R. W. Doms. 2005. An infectious West Nile virus that expresses a GFP reporter gene. Virology 334:28-40. [DOI] [PubMed] [Google Scholar]
- 47.Rahal, J. J., J. Anderson, C. Rosenberg, T. Reagan, and L. L. Thompson. 2004. Effect of interferon-α2b therapy on St. Louis viral meningoencephalitis: clinical and laboratory results of a pilot study. J. Infect. Dis. 190: 1084-1087. [DOI] [PubMed] [Google Scholar]
- 48.Reyes-Del Valle, J., S. Chavez-Salinas, F. Medina, and R. M. Del Angel. 2005. Heat shock protein 90 and heat shock protein 70 are components of dengue virus receptor complex in human cells. J. Virol. 79:4557-4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ruuth, K., L. Carlsson, B. Hallberg, and E. Lundgren. 2001. Interferon-alpha promotes survival of human primary B-lymphocytes via phosphatidylinositol 3-kinase. Biochem. Biophys. Res. Commun. 284:583-586. [DOI] [PubMed] [Google Scholar]
- 50.Ryman, K. D., W. B. Klimstra, K. B. Nguyen, C. A. Biron, and R. E. Johnston. 2000. Alpha/beta interferon protects adult mice from fatal Sindbis virus infection and is an important determinant of cell and tissue tropism. J. Virol. 74:3366-3378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Samuel, C. E. 1991. Antiviral actions of interferon. Interferon-regulated cellular proteins and their surprisingly selective antiviral activities. Virology 183:1-11. [DOI] [PubMed] [Google Scholar]
- 52.Sangfelt, and H. Strander. 2001. Apoptosis and cell growth inhibition as antitumor effector functions of interferons. Med. Oncol. 18:3-14. [DOI] [PubMed] [Google Scholar]
- 53.Sayao, A. L., O. Suchowersky, A. Al-Khathaami, B. Klassen, N. R. Katz, R. Sevick, P. Tilley, J. Fox, and D. Patry. 2004. Calgary experience with West Nile virus neurological syndrome during the late summer of 2003. Can. J. Neurol. Sci. 31:194-203. [DOI] [PubMed] [Google Scholar]
- 54.Sejvar, J. J., M. B. Haddad, B. C. Tierney, G. L. Campbell, A. A. Marfin, J. A. Van Gerpen, A. Fleischauer, A. A. Leis, D. S. Stokic, and L. R. Petersen. 2003. Neurologic manifestations and outcome of West Nile virus infection. JAMA 290:511-515. [DOI] [PubMed] [Google Scholar]
- 55.Shresta, S., J. L. Kyle, H. M. Snider, M. Basavapatna, P. R. Beatty, and E. Harris. 2004. Interferon-dependent immunity is essential for resistance to primary dengue virus infection in mice, whereas T- and B-cell-dependent immunity are less critical. J. Virol. 78:2701-2710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shrestha, B., and M. S. Diamond. 2004. Role of CD8+ T cells in control of West Nile virus infection. J. Virol. 78:8312-8321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shrestha, B., D. Gottlieb, and M. S. Diamond. 2003. Infection and injury of neurons by West Nile encephalitis virus. J. Virol. 77:13203-13213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Solomon, T., N. M. Dung, B. Wills, R. Kneen, M. Gainsborough, T. V. Diet, T. T. Thuy, H. T. Loan, V. C. Khanh, D. W. Vaughn, N. J. White, and J. J. Farrar. 2003. Interferon alfa-2a in Japanese encephalitis: a randomised double-blind placebo-controlled trial. Lancet 361:821-826. [DOI] [PubMed] [Google Scholar]
- 59.Tassaneetrithep, B., T. H. Burgess, A. Granelli-Piperno, C. Trumpfheller, J. Finke, W. Sun, M. A. Eller, K. Pattanapanyasat, S. Sarasombath, D. L. Birx, R. M. Steinman, S. Schlesinger, and M. A. Marovich. 2003. DC-SIGN (CD209) mediates dengue virus infection of human dendritic cells. J. Exp. Med. 197:823-829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Thepparit, C., and D. R. Smith. 2004. Serotype-specific entry of dengue virus into liver cells: identification of the 37-kilodalton/67-kilodalton high-affinity laminin receptor as a dengue virus serotype 1 receptor. J. Virol. 78:12647-12656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Trottier, M. D., Jr., B. M. Palian, and C. S. Reiss. 2005. VSV replication in neurons is inhibited by type I IFN at multiple stages of infection. Virology 333:215-225. [DOI] [PubMed] [Google Scholar]
- 62.Wang, F., Y. Ma, J. W. Barrett, X. Gao, J. Loh, E. Barton, H. W. Virgin, and G. McFadden. 2004. Disruption of Erk-dependent type I interferon induction breaks the myxoma virus species barrier. Nat. Immunol. 5:1266-1274. [DOI] [PubMed] [Google Scholar]
- 63.Wang, K., D. Scheel-Toellner, S. H. Wong, R. Craddock, J. Caamano, A. N. Akbar, M. Salmon, and J. M. Lord. 2003. Inhibition of neutrophil apoptosis by type 1 IFN depends on cross-talk between phosphoinositol 3-kinase, protein kinase C-delta, and NF-κB signaling pathways. J. Immunol. 171:1035-1041. [DOI] [PubMed] [Google Scholar]
- 64.Wang, T., T. Town, L. Alexopoulou, J. F. Anderson, E. Fikrig, and R. A. Flavell. 2004. Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat. Med. 10:1366-1373. [DOI] [PubMed] [Google Scholar]
- 65.Wang, Y., M. Lobigs, E. Lee, and A. Mullbacher. 2003. CD8+ T cells mediate recovery and immunopathology in West Nile virus encephalitis. J. Virol. 77:13323-13334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yang, C. H., A. Murti, S. R. Pfeffer, L. Basu, J. G. Kim, and L. M. Pfeffer. 2000. IFN alpha/beta promotes cell survival by activating NF-κB. Proc. Natl. Acad. Sci. USA 97:13631-13636. [DOI] [PMC free article] [PubMed] [Google Scholar]