Abstract
The Nonsense-Mediated mRNA Decay (NMD) pathway mediates the rapid degradation of mRNAs that contain premature stop mutations in eukaryotic organisms. It was recently shown that mutations in three yeast genes that encode proteins involved in the NMD process, UPF1, UPF2, and UPF3, also reduce the efficiency of translation termination. In the current study, we compared the efficiency of translation termination in a upf1Δ strain and a [PSI+] strain using a collection of translation termination reporter constructs. The [PSI+] state is caused by a prion form of the polypeptide chain release factor eRF3 that limits its availability to participate in translation termination. In contrast, the mechanism by which Upf1p influences translation termination is poorly understood. The efficiency of translation termination is primarily determined by a tetranucleotide termination signal consisting of the stop codon and the first nucleotide immediately 3′ of the stop codon. We found that the upf1Δ mutation, like the [PSI+] state, decreases the efficiency of translation termination over a broad range of tetranucleotide termination signals in a unique, context-dependent manner. These results suggest that Upf1p may associate with the termination complex prior to polypeptide chain release. We also found that the increase in readthrough observed in a [PSI+]/upf1Δ strain was larger than the readthrough observed in strains carrying either defect alone, indicating that the upf1Δ mutation and the [PSI+] state influence the termination process in distinct ways. Finally, our analysis revealed that the mRNA destabilization associated with NMD could be separated into two distinct forms that correlated with the extent the premature stop codon was suppressed. The minor component of NMD was a 25% decrease in mRNA levels observed when readthrough was ≥0.5%, while the major component was represented by a larger decrease in mRNA abundance that was observed only when readthrough was ≤0.5%. This low threshold for the onset of the major component of NMD indicates that mRNA surveillance is an ongoing process that occurs throughout the lifetime of an mRNA.
Keywords: UPF1, [PSI+], NMD, translation termination, mRNA stability, readthrough
INTRODUCTION
Translation termination in eubacteria is mediated by two class I release factors. RF1 decodes UAG and UAA stop codons, while RF2 decodes UGA and UAA codons. Studies in bacteria have provided evidence that RF1 and RF2 associate with the ribosomal A site, where they recognize the stop codon directly through the action of a “peptide anticodon” sequence (Ito et al. 2000). After the nascent polypeptide chain is released, the class II release factor RF3 facilitates recycling of RF1 or RF2 from the termination complex by a GTP-dependent mechanism (Zavialov et al. 2001, 2002). In contrast, eukaryotic organisms mediate translation termination through the action of a single class I release factor (eRF1) that recognizes all three stop codons (UAG, UAA, and UGA; Bertram et al. 2001; Kisselev et al. 2003). A class II release factor (eRF3) forms a complex with eRF1 and assists in termination through a GTP-dependent mechanism. Further mechanistic details of eukaryotic translation termination remain to be elucidated.
Translation termination is normally a highly efficient process. Previous studies have shown that stop codons are normally suppressed at a frequency of only 0.001%–0.1% in eukaryotes (Loftfield and Vanderjagt 1972; Mori et al. 1985; Stansfield et al. 1998). The suppression of a stop codon occurs when a near-cognate aminoacyl-tRNA, which can base pair with a stop codon at two of the three positions of a codon–anticodon complex, is accommodated into the ribosomal A site. This allows its amino acid to be erroneously incorporated into the polypeptide chain and the subsequent continuation of translation beyond the stop codon (Fearon et al. 1994). The process of stop codon suppression is frequently referred to as translational readthrough.
Mutations in the yeast genes encoding eRF1 (SUP45) or eRF3 (SUP35) lead to an “omnipotent suppressor” phenotype, which is characterized by the suppression of translation termination at all three stop codons. Another condition that reduces the efficiency of translation termination in yeast is the cytoplasmically inherited [PSI+] state. This novel condition is caused by the conversion of eRF3 to a nonfunctional prion form that associates in large aggregates within yeast cells (Serio and Lindquist 1999). As a result, [PSI+] strains experience an increased level of readthrough due to the limitation of functional eRF3. In addition, mutations within the small subunit (18S) and large subunit (25S) ribosomal RNAs have also been shown to cause an increased rate of translational readthrough in yeast (Chernoff et al. 1994; Liu and Liebman 1996; Velichutina et al. 2000, 2001).
Previous studies have shown that the sequence context surrounding a stop codon can have a large effect on the efficiency of translation termination in bacteria, yeast, and mammals. For example, it was shown that changes in the immediate sequence context surrounding a stop codon can alter the efficiency of translation termination by >100-fold. Proximal sequences both upstream and downstream of the stop codon contribute to this effect (Bonetti et al. 1995). Sequence analysis has also shown that the distribution of nucleotides that surround naturally occurring stop codons is nonrandom. This bias is most evident for the first nucleotide following a stop codon, where a purine nucleotide is usually observed (Brown et al. 1990). Based upon these results, it was proposed that translation termination is mediated by a tetranucleotide termination signal that contains the stop codon and the first downstream nucleotide. More recently, it was shown that the bacterial release factor RF2 directly contacts the first nucleotide following the stop codon (Poole et al. 1998).
In eukaryotic organisms, mRNA molecules that contain a premature stop mutation frequently undergo rapid turnover by a mechanism termed Nonsense-Mediated mRNA Decay (NMD). Unlike normal 5′ → 3′ mRNA turnover, which requires removal of the poly(A) tail prior to decapping and degradation by the Xrn1 exonuclease (Decker and Parker 1993; Muhlrad and Parker 1994), the NMD pathway decaps the mRNA and facilitates its degradation without prior removal of the poly(A) tail (Hagan et al. 1995; Beelman et al. 1996). This NMD process is mediated by the trans-acting factors Upf1p, Upf2p, and Upf3p (Leeds et al. 1991, 1992). A significant fraction of each of these proteins is normally found in association with 80S ribosomes and polyribosomes (Atkin et al. 1995, 1997). Upf1 is an RNA-binding protein that exhibits ATP-dependent 5′ → 3′ helicase activity (Czaplinski et al. 1998), while Upf2p and Upf3p have been proposed to regulate the function of Upf1p (Maderazo et al. 2000). Upf3p has also been shown to shuttle between the cytoplasm and the nucleus (Shirley et al. 1998, 2002).
The mechanism that couples premature translation termination to the process of NMD remains obscure. In one model, it has been proposed that Upf1p binds to eRF1 and eRF3 while the termination complex is still bound to the stop codon in the ribosomal A site (Czaplinski et al. 1998). The association of Upf1p with Upf2p and Upf3p then results in the formation of a “surveillance complex” that scans the mRNA downstream of the stop codon following release of the nascent polypeptide chain to determine whether termination has occurred at the proper location in the mRNA (Wang et al. 2001). In another model, it was proposed that the spatial relationship between the stop codon and the 3′ UTR determines whether NMD is activated via a kinetic proofreading mechanism (Hilleren and Parker 1999). When a premature stop mutation alters this spatial relationship, the rate of translation termination is reduced beyond a crucial threshold and NMD is induced. In this model, the ATPase activity of Upf1p serves as the kinetic clock that determines the time frame in which a termination event must be completed to avoid the initiation of NMD.
Previous studies have provided strong evidence that a “downstream sequence element” (DSE) must be located within the 3′ UTR of a yeast mRNA for NMD to occur (Peltz et al. 1993; Ruiz-Echevarria and Peltz 1996; Mendell et al. 2000; He and Jacobson 2001). A somewhat different situation occurs in mammalian cells, where NMD is induced only when the premature stop codon is located >50–55 nucleotides upstream of the 3′-most exon–exon junction (Nagy and Maquat 1998; Culbertson 1999). An explanation for this observation arose when it was shown that an exon–exon junction complex (EJC) involved in mRNA splicing is deposited 20–24 nucleotides upstream of each exon–exon junction (Le Hir et al. 2000). The presence of this complex on newly synthesized mammalian mRNAs immediately following mRNA export from the nucleus is consistent with the proposal that the initial or “pioneer” round of translation may remove factors and thereby remodel the mRNA (Ishigaki et al. 2001; Lejeune et al. 2002). This remodeling process is thought to prevent the initiation of NMD in subsequent rounds of translation.
It was recently shown that mutations in the UPF1, UPF2, and UPF3 genes increase the readthrough of stop codons (Maderazo et al. 2000; Wang et al. 2001), suggesting that these gene products also affect the efficiency of translation termination. In the current study, we compared the magnitude and context-dependence of translational readthrough in [PSI+], upf1Δ, and [PSI+]/upf1Δ strains. We found that the upf1Δ mutation, like the [PSI+] state, reduces the efficiency of translation termination over a broad range of tetranucleotide termination signals. This observation demonstrates that Upf1p influences the efficiency of translation termination in a general manner. Furthermore, the increased readthrough observed with the [PSI+]/upf1Δ strain was routinely larger than the readthrough observed in strains carrying either defect alone. By correlating the relative level of readthrough to the mRNA level, we found that the effects of NMD could be dissected into two distinct components. The major component of mRNA destabilization by NMD was induced only when translational readthrough was extremely low, indicating that NMD in yeast is not carried out solely during the first round of translation as proposed for mammalian cells. Instead, our results suggest that NMD in this simple eukaryote is an ongoing process that is carried out throughout the lifetime of an mRNA molecule.
RESULTS
The upf1Δ mutation causes a general decrease in the efficiency of translation termination that becomes more severe when combined with [PSI+]
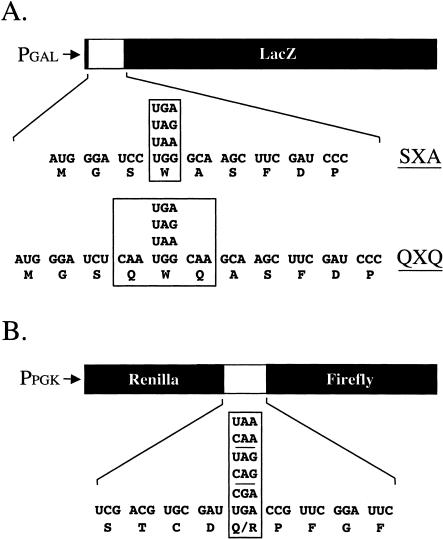
The cytoplasmically inherited [PSI+] factor has long been known to reduce the efficiency of translation termination (and thereby increase the readthrough of stop codons) in the yeast Saccharomyces cerevisiae (Serio and Lindquist 1999). More recently, it was shown that mutations that compromise the machinery involved in nonsense-mediated mRNA decay (NMD) also increase the suppression of premature stop mutations (Maderazo et al. 2000; Wang et al. 2001). To gain a better understanding of the extent and magnitude of readthrough caused by the loss of an NMD factor, we compared the efficiency of translation termination in [PSI+] and upf1Δ strains. The initial readthrough reporter system consisted of a β-galactosidase reporter protein whose expression depended upon the suppression of a UAG, UGA, or UAA stop codon (Fig. 1A ▶; Bonetti et al. 1995). As a control, a construct containing a UGG (tryptophan) codon at the same position was assayed in each strain to determine the theoretical maximal level of expression (i.e., 100% readthrough) for this reporter system. Finally, readthrough of the stop codons in this assay system was monitored in two sequence contexts. The first set, termed the SXA reporters, contains an upstream UCC (serine) codon and downstream GCA (alanine) codon on either side of the premature stop codon. This context was previously shown to yield very efficient termination at each of the three stop codons (Bonetti et al. 1995). The second set, called the QXQ reporters, differed from the SXA constructs by the addition of a CAA (glutamine) codon on either side of the premature stop codon. This modest change in the context surrounding the stop codon was previously shown to result in as much as a 100-fold reduction in the efficiency of translation termination. In particular, it was shown that the presence of a C residue at the first position following the stop codon alone could reduce the efficiency of termination by as much as 20-fold (Bonetti et al. 1995).
FIGURE 1.
Readthrough reporter systems. (A) The β-galactosidase readthrough reporter constructs containing either the SXA or QXQ readthrough cassettes. (B) The dual luciferase readthrough reporter constructs.
To compare the effects of [PSI+] and the upf1Δ mutation on the efficiency of translation termination, we introduced the SXA and QXQ reporter constructs into wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains. β-Galactosidase assays were then carried out to determine the level of full-length protein produced by the suppression of stop codons in different sequence contexts. To evaluate the level of suppression as accurately as possible, the β-galactosidase enzymatic activity was corrected for the relative level of β-galactosidase mRNA in each strain (normalized to the ACT1 mRNA as an internal loading control). This final value, referred to as the “corrected readthrough,” provides an accurate measure of the efficiency of translation termination (full-length protein produced per unit mRNA).
Northern blot analysis of the four strains expressing either set of readthrough reporter constructs revealed the presence of three LacZ mRNA species: a predominant species representing the full-length mRNA, and two higher molecular weight species (Fig. 2 ▶). The presence of multiple mRNA species was presumably due to inefficient transcription termination in these constructs. Quantitation of the total LacZ mRNA levels measured in each strain is shown in Table 1 ▶. For the wild-type strain expressing β-galactosidase from the SXA reporter constructs, we observed a significant (up to twofold) decrease in total nonsense-containing mRNA levels (relative to the sense control), indicating their destabilization by NMD. A similar decrease in the β-galactosidase mRNA level was observed for all three mRNA species, indicating that each of these LacZ species was subject to a similar level of RNA degradation by NMD. A reversal of this decrease in mRNA stability in the upf1Δ strain confirmed that this effect was due to NMD. Interestingly, this NMD effect was less pronounced in the wild-type strain when the mRNA was expressed from the QXQ reporter constructs, indicating that the context affects not only the efficiency of translation termination, but also the level of NMD observed.
FIGURE 2.
Northern blot analysis of mRNA from β-galactosidase SXA reporter constructs. Steady-state β-galactosidase mRNA levels were measured in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains. The β-galactosidase SXA reporter constructs contained either a UAA, UAG, or UGA stop codon, or a UGG tryptophan codon in the readthrough position.
TABLE 1.
Effect of stop codon context on [PSI+] and upf1Δ-mediated readthrough
| Strain | Reporter construct | Readthrough codon | Absolute β-gal activity (±SD)a | Relative β-gal mRNA (±SD)b | Corrected readthrough (%) |
| Wild type | SXA | UGG | 193.33 (±15.40) | 1 | 100 |
| Wild type | SXA | UAG | 0.023 (±0.067) | 0.66 (±0.036) | 0.018 (±0.003) |
| Wild type | SXA | UGA | 0.019 (±0.004) | 0.71 (±0.088) | 0.015 (±0.001) |
| Wild type | SXA | UAA | 0.016 (±0.007) | 0.53 (±0.025) | 0.016 (±0.004) |
| [PSI+] | SXA | UGG | 188.30 (±12.23) | 1 | 100 |
| [PSI+] | SXA | UAG | 0.088 (±0.029) | 0.44 (±0.058) | 0.11 (±0.011) |
| [PSI+] | SXA | UGA | 0.030 (±0.009) | 0.50 (±0.078) | 0.039 (±0.010) |
| [PSI+] | SXA | UAA | 0.024 (±0.010) | 0.50 (±0.052) | 0.025 (±0.006) |
| upf1Δ | SXA | UGG | 232.65 (±15.36) | 1 | 100 |
| upf1Δ | SXA | UAG | 0.184 (±0.034) | 0.85 (±0.11) | 0.056 (±0.008) |
| upf1Δ | SXA | UGA | 0.094 (±0.001) | 1.06 (±0.093) | 0.038 (±0.007) |
| upf1Δ | SXA | UAA | 0.171 (±0.029) | 1.12 (±0.11) | 0.063 (±0.005) |
| [PSI+]/upf1Δ | SXA | UGG | 163.71 (±36.33) | 1 | 100 |
| [PSI+]/upf1Δ | SXA | UAG | 0.249 (±0.025) | 1.06 (±0.062) | 0.15 (±0.022) |
| [PSI+]/upf1Δ | SXA | UGA | 0.177 (±0.027) | 1.18 (±0.11) | 0.096 (±0.009) |
| [PSI+]/upf1Δ | SXA | UAA | 0.323 (±0.037) | 1.17 (±0.041) | 0.18 (±0.025) |
| Wild type | QXQ | UGG | 549.74 (±54.17) | 1 | 100 |
| Wild type | QXQ | UAG | 3.59 (±0.18) | 0.77 (±0.050) | 0.83 (±0.054) |
| Wild type | QXQ | UGA | 3.68 (±0.21) | 0.71 (±0.11) | 1.04 (±0.15) |
| Wild type | QXQ | UAA | 1.85 (±0.15) | 0.62 (±0.083) | 0.57 (±0.085) |
| [PSI+] | QXQ | UGG | 484.91 (±41.67) | 1 | 100 |
| [PSI+] | QXQ | UAG | 16.82 (±1.21) | 0.87 (±0.12) | 4.43 (±0.47) |
| [PSI+] | QXQ | UGA | 14.63 (±1.52) | 0.76 (±0.11) | 3.97 (±0.41) |
| [PSI+] | QXQ | UAA | 9.18 (±0.64) | 0.83 (±0.11) | 2.6 (±0.22) |
| upf1Δ | QXQ | UGG | 559.10 (±34.39) | 1 | 100 |
| upf1Δ | QXQ | UAG | 10.96 (±1.13) | 1.29 (±0.18) | 1.64 (±0.25) |
| upf1Δ | QXQ | UGA | 12.70 (±1.27) | 1.16 (±0.23) | 2.33 (±0.23) |
| upf1Δ | QXQ | UAA | 9.23 (±1.23) | 0.98 (±0.058) | 1.81 (±0.14) |
| [PSI+]/upf1Δ | QXQ | UGG | 204.88 (±45.94) | 1 | 100 |
| [PSI+]/upf1Δ | QXQ | UAG | 24.60 (±2.86) | 2.02 (±0.089) | 5.75 (±0.70) |
| [PSI+]/upf1Δ | QXQ | UGA | 25.90 (±4.22) | 1.97 (±0.18) | 6.87 (±0.39) |
| [PSI+]/upf1Δ | QXQ | UAA | 7.90 (±2.12) | 1.86 (±0.057) | 2.17 (±0.28) |
aEight independent β-galactosidase assays were carried out for each construct.
bAt least three independent mRNA measurements were carried out for each construct.
The corrected readthrough values obtained for the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains carrying either the SXA and QXQ reporter constructs are also shown in Table 1 ▶. We found that these values ranged from 0.015% to 0.018% in the wild-type strain carrying the SXA plasmids, which represents a very efficient level of translation termination. The corrected readthrough in the wild-type strain carrying the QXQ plasmids ranged from 0.57% to 1.04%. The large differences obtained with these two sets of reporter constructs are consistent with the results obtained in a previous study (Bonetti et al. 1995) and demonstrate the large effects sequence context can play on the efficiency of translation termination.
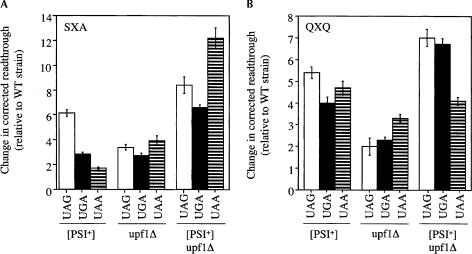
A comparison of the corrected readthrough associated with the [PSI+] state, the upf1Δ mutation, or both alterations together in the SXA context is shown in Figure 3A ▶. We found that the [PSI+] state increased readthrough in the SXA context by 1.6- to 6.1-fold (relative to the wild-type strain). The upf1Δ mutation increased readthrough in the SXA context by 2.5- to 3.9-fold. The stimulation of readthrough measured in the upf1Δ strain at the UGA codon was similar to the increase observed in the [PSI+] strain, while readthrough at the UAG stop codon was significantly lower, and readthrough at the UAA stop codon was significantly higher. When [PSI+] and the upf1Δ mutation were present together in the same strain, the readthrough observed at each stop codon was significantly higher than the level measured when either [PSI+] or the upf1Δ mutation alone were present. These results suggest that these two defects are manifested at distinct steps of the termination process that lead to larger increases in readthrough when both are present together.
FIGURE 3.
Corrected readthrough levels determined using the β-galactosidase readthrough assay. The fold increase in readthrough in the [PSI+], upf1Δ, and [PSI+]/upf1Δ strains are represented relative to the basal level of readthrough measured in the wild-type strain at the corresponding stop codon. (A) SXA β-galactosidase reporter constructs. (B) QXQ β-galactosidase reporter constructs. WT, wild type.
A comparison of the effects of [PSI+] and the upf1Δ mutation on the corrected readthrough in the QXQ context is shown in Figure 3B ▶. We found that the [PSI+] state increased readthrough in the QXQ reporters by 3.8- to 5.3-fold (relative to the wild-type strain). The upf1Δ mutation increased readthrough in this context only 2.0- to 3.2-fold, which represents a more modest increase in readthrough than [PSI+] at all three stop codons. Combining [PSI+] and the upf1Δ mutation in the same strain increased readthrough in the QXQ context by 3.8- to 6.9-fold. This represents a higher level of readthrough at the UAG and UGA stop codons than either defect alone, while the level observed at the UAA stop codon was intermediate between that observed with either defect alone. When taken together, these results indicate that both the [PSI+] state and the upf1Δ mutation can induce readthrough at each of the three stop codons in various sequence contexts. Furthermore, combining these two defects usually leads to a larger decrease in the efficiency of translation termination than either condition alone.
The context-dependent effects on translation termination caused by the upf1Δ mutation supercede the larger effects caused by [PSI+]
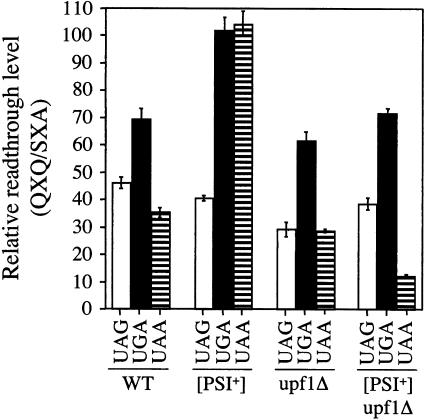
As described above, the basal levels of readthrough measured in the wild-type strain ranged from 0.015% to 0.018% with the SXA reporters and 0.57% to 1.04% with the QXQ reporters. When these context-dependent differences in readthrough (QXQ/SXA) were compared at each stop codon in the wild-type background, we found that the corrected readthrough measured using the QXQ reporters was 36- to 69-fold greater than that measured at the same stop codon in the SXA reporter constructs (Fig. 4 ▶). When the relative readthrough measured in the QXQ and SXA reporter plasmids were compared in the [PSI+] strain, we found that the context dependence was similar to the wild-type strain at the UAG codon but was significantly higher in the QXQ context at the UGA and UAA stop codons. These results suggest that the limitation of functional eRF3 that occurs in the [PSI+] strain preferentially influences the context dependence of translation termination at the UGA and UAA stop codons. Overall, these differences in readthrough measured in the [PSI+] strain ranged from 40- to 104-fold.
FIGURE 4.
Context-dependent readthrough (QXQ/SXA) determined using the β-galactosidase readthrough assay. The data is expressed as the ratio of readthrough measured with the β-galactosidase QXQ reporter relative to the readthrough measured with the β-galactosidase SXA reporter. The QXQ/SXA ratios are shown for the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ yeast strains. WT, wild type.
When we compared the relative context-dependent readthrough (QXQ/SXA) at each stop codon measured in the upf1Δ strain, we found that these ratios were smaller at each of the three stop codons than in either the wild-type and [PSI+] strains. In particular, the QXQ/SXA ratios in the upf1Δ strain were much smaller at the UGA and UAA stop codons than in the [PSI+] strain. Overall, the differences in the relative level of readthrough at the QXQ and SXA contexts measured in the upf1Δ strain ranged from 29- to 61-fold. These results suggest that the upf1Δ mutation alters the ability of the termination machinery to discriminate between different sequence contexts and may be related to the previously demonstrated ability of Upf1p to interact directly with eRF1 and eRF3 (Wang et al. 2001). When we examined the context-dependent differences in readthrough in the [PSI+]/upf1Δ strain, we found that they were generally similar to those observed in the upf1Δ strain. Overall, the QXQ/SXA ratios in the [PSI+]/upf1Δ strain ranged from 12- to 72-fold. When taken together, these results indicate that the context-dependent differences in readthrough caused by [PSI+] are much larger than those observed in either the wild-type or upf1Δ strains. Furthermore, the relatively small context-dependent differences in readthrough observed in the [PSI+]/upf1Δ strain suggest that the consequences associated with the loss of Upf1p supercede the larger context-dependent differences attributable to [PSI+].
Analysis of the effects of [PSI+] and upf1Δ on translation termination using a dual luciferase readthrough reporter system
The results described above suggest that both [PSI+] and the upf1Δ mutation reduce the efficiency of translation termination at UAG, UAA, and UGA stop codons in at least two distinct sequence contexts. However, it remained a formal possibility that these results could be an artifact caused by some limitation of the β-galactosidase reporter system. For example, it was possible that the increases in β-galactosidase activity observed could be attributable to indirect effects on the rate of translation initiation rather than a direct effect on the efficiency of translation termination. This led us to adapt another set of readthrough reporter constructs to our yeast experimental system.
In recent studies, a dual luciferase reporter system was shown to provide an effective means to monitor the efficiency of translation termination in mammalian cells (Grentzmann et al. 1998; Howard et al. 2000). This system utilizes tandem Renilla and firefly luciferase genes that are separated by a single in-frame stop codon (Fig. 1B ▶). Thus, it uses the relative abundance of these light-emitting proteins to monitor the efficiency of translation termination. The activity of firefly luciferase, encoded by the distal open reading frame, provides a quantitative measure of the suppression of the stop codon that separates the two open reading frames. The activity of Renilla luciferase, encoded by the proximal open reading frame, serves as an internal control for mRNA abundance and precludes the need for mRNA determinations. In addition, this system also avoids possible artifacts associated with changes in the efficiency of translation initiation associated with the function of the NMD machinery (Muhlrad and Parker 1999), because both the Renilla and firefly enzymes initiate translation from the same AUG codon.
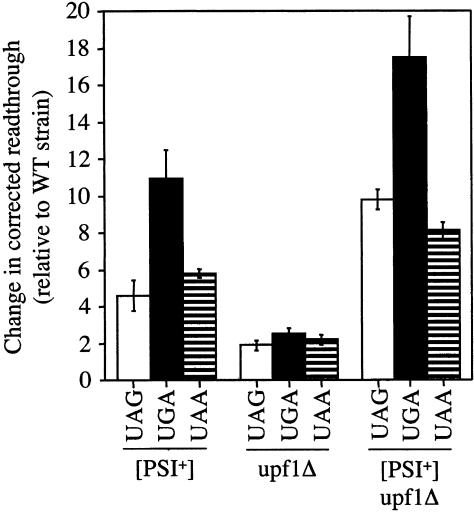
We found that the corrected readthrough measured in the wild-type strain using the dual luciferase system ranged from 0.31% to 0.79% (Table 2 ▶). These levels were very similar to those measured in the wild-type strain using the β-galactosidase QXQ reporter plasmids. This result is consistent with the presence of a C residue immediately following the stop codon in both reporter systems, which results in the presence of identical tetranucleotide termination signals. We next determined the effect of the [PSI+] state and the upf1Δ mutation on the corrected readthrough measured with the dual luciferase system. We found that the presence of [PSI+] increased readthrough in the dual luciferase plasmids by 4.6- to 10.9-fold, while the upf1Δ mutation increased readthrough in this reporter system only 1.9- to 2.5-fold (Fig. 5 ▶). The combined effects of [PSI+] and upf1Δ increased readthrough from 8.1- to 17.5-fold, indicating that the presence of these two alterations together increased readthrough more than either one alone. Overall, these results indicate that the corrected readthrough levels measured in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains using the dual luciferase system were qualitatively similar to the values obtained with the β-galactosidase reporters with the QXQ context (cf. Figs. 5 ▶ and 3B ▶), confirming that the differences in corrected readthrough efficiencies measured in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains using the β-galactosidase reporter plasmids reflect real differences in the efficiency of translation termination.
TABLE 2.
Readthrough measured with the dual luciferase reporter system
| Strain | Readthrough codon | Firefly/renilla stop codon (±SD) | Firefly/renilla sense codon (±SD) | Corrected readthrough (%) |
| Wild type | UAG | 0.0057 (±0.00015) | 1.38 (±0.065) | 0.41 (±0.012) |
| Wild type | UGA | 0.0124 (±0.0018) | 1.57 (±0.11) | 0.79 (±0.047) |
| Wild type | UAA | 0.0038 (±0.00013) | 1.21 (±0.059) | 0.31 (±0.016) |
| [PSI+] | UAG | 0.025 (±0.0014) | 1.33 (±0.059) | 1.88 (±0.025) |
| [PSI+] | UGA | 0.121 (±0.0019) | 1.40 (±0.050) | 8.60 (±0.120) |
| [PSI+] | UAA | 0.022 (±0.0024) | 1.23 (±0.087) | 1.79 (±0.038) |
| upf1Δ | UAG | 0.010 (±0.0010) | 1.31 (±0.037) | 0.76 (±0.012) |
| upf1Δ | UGA | 0.023 (±0.0025) | 1.15 (±0.056) | 2.00 (±0.040) |
| upf1Δ | UAA | 0.093 (±0.0027) | 1.39 (±0.041) | 0.67 (±0.016) |
| [PSI+]/upf1Δ | UAG | 0.050 (±0.0049) | 1.25 (±0.025) | 4.00 (±0.056) |
| [PSI+]/upf1Δ | UGA | 0.16 (±0.029) | 1.16 (±0.11) | 13.80 (±0.78) |
| [PSI+]/upf1Δ | UAA | 0.032 (±0.0050) | 1.27 (±0.053) | 2.52 (±0.096) |
aEight independent luciferase assays were carried out for each construct.
FIGURE 5.
Corrected readthrough levels determined using the dual luciferase readthrough assay. The fold increase in readthrough measured in the [PSI+], upf1Δ, and [PSI+]/upf1Δ strains are represented relative to the basal level of readthrough measured in the wild-type strain at the corresponding stop codon.
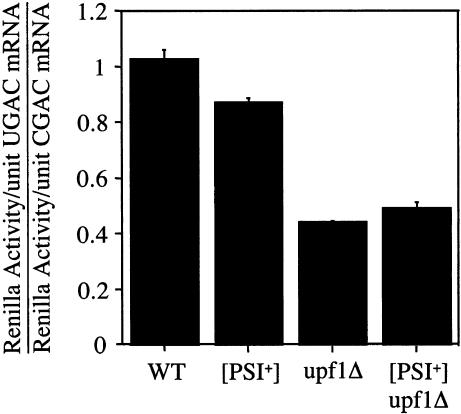
It was previously shown that the recognition of a premature stop codon not only induces NMD, but also results in a decrease in the rate of translation initiation (Muhlrad and Parker 1999). Because the expression of both the Renilla and firefly luciferase in our system is initiated at a single AUG codon in our reporter system, such an effect could not influence our corrected readthrough results. As such, any changes observed in the firefly luciferase activity could be attributed specifically to suppression of the premature stop codon. However, the Renilla protein expressed before the premature stop codon could also be used to compare the translational efficiency of nonsense mRNA and control (sense) mRNAs in this system. To determine whether translational efficiency of the dual luciferase mRNA was influenced by the presence of a premature stop codon, we compared the Renilla translational efficiency (the Renilla activity per unit mRNA) in nonsense (UGAC) and control (CGAC) constructs in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains (Fig. 6 ▶). We found that the translational efficiency (protein specific activity/unit mRNA) of Renilla luciferase was similar in nonsense (UGAC) and sense (CGAC) reporter plasmids in a wild-type strain, resulting in a translational efficiency ratio (UGAC/CGAC) of 1.0. A slightly lower ratio of 0.87 was observed in the [PSI+] strain. Interestingly, we observed a much larger decrease in this ratio in both the upf1Δ and [PSI+]/upf1Δ strains. We found that the translational efficiency ratio (UGAC/CGAC) was 0.44 in the upf1Δ strain and 0.49 in the [PSI+]/upf1Δ strain. These results suggest that the loss of Upf1p leads to a twofold reduction in the efficiency of translation initiation of the Renilla open reading frame in the dual luciferase reporter mRNA.
FIGURE 6.
Effect of a premature stop codon on the translation efficiency of Renilla luciferase. Renilla specific activity (RLUs/sec/ng protein) per unit mRNA was determined for the indicated strains using dual luciferase reporter plasmids containing either the UGAC or CGAC tetranucleotides. The ratio of these values (UGAC/CGAC) serves as a measure of the relative translational efficiency. Assuming translation elongation rates are constant, this ratio should provide a measure of the relative levels of translation initiation in the UGAC and CGAC reporter plasmids.
A low level of translational readthrough is required to activate NMD
The data obtained with the β-galactosidase reporter system also allowed us to determine how differences in the efficiency of translation termination influence mRNA stability. The corrected readthrough levels in the wild-type and [PSI+] strains ranged from 0.015% to 4.43%, which represents a 295-fold range in the efficiency of translation termination using these closely related mRNA species. To determine how the level of nonsense suppression influences mRNA stability, we correlated the corrected readthrough levels to the relative steady-state LacZ mRNA level in each strain. Quantitation of the relative mRNA level (the abundance of nonsense-containing mRNA relative to the abundance of the corresponding control mRNA containing a sense codon) revealed as much as 2.3-fold differences (Table 1 ▶).
The results of this analysis using the data obtained with the wild-type and [PSI+] strains that contain an intact NMD pathway are shown in Figure 7A ▶. For comparison, the results of the data from the upf1Δ strain that lacks an NMD pathway are presented in Figure 7B ▶. We found that the mean steady-state level of the six mRNAs examined in the upf1Δ strain was 1.08 ± 0.15, indicating that the presence of a premature stop mutation did not alter the abundance of these mRNAs in a strain lacking an intact NMD pathway. In contrast, we observed two distinct effects of NMD on the abundance of mRNAs that contain a premature stop mutation (Fig. 7A ▶). First, the mean steady-state level of the six mRNAs that were measured in the wild-type and [PSI+] strains with a corrected readthrough level ≥0.5% was 0.76 ± 0.098. This represented a statistically significant difference from the mean mRNA level observed in the upf1Δ strain (P value <0.01). In addition, we found that the mean steady-state level of the six mRNAs examined from the wild-type and [PSI+] strains with a corrected readthrough level ≤0.5% was 0.56 ± 0.105. This greater decrease in the steady-state mRNA levels was statistically significant when compared to the mRNA levels measured when readthrough was ≥0.5% (P value <0.02). These results suggest that the consequences of NMD are exerted on these similar LacZ mRNAs in two distinct ways. The first is a modest (25%) reduction in the steady-state mRNA level that is detected at higher readthrough levels (≥0.5%). The second manifestation of NMD is a more severe mRNA destabilization that occurs only when readthrough drops below a surprisingly low threshold (≤0.5%).
FIGURE 7.
Correlation between mRNA levels and corrected readthrough of stop codons using the β-galactosidase readthrough assay. The corrected readthrough values obtained in both the β-galactosidase SXA and QXQ contexts were plotted against the relative level of LacZ mRNA for each construct as determined by northern blot analysis. (A) Plot of data obtained with the wild-type strain, SXA context (squares); wild-type strain, QXQ context (diamonds); [PSI+] strain, SXA context (circles); and [PSI+] strain, QXQ context (triangles). (B) Plot of data obtained with the upf1Δ strain, SXA context (squares); and the upf1Δ strain, QXQ context (triangles). WT, wild type.
Increased abundance of nonsense mRNA species as a function of [PSI+] and the upf1Δ mutation
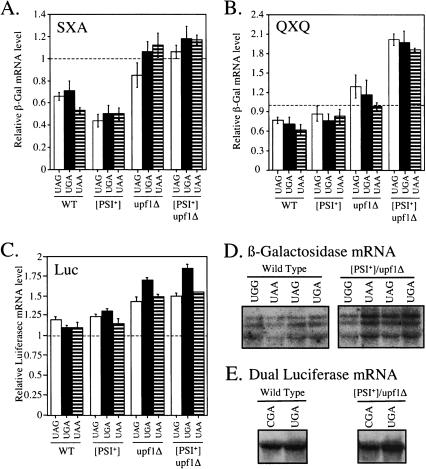
While examining the relative steady-state mRNA levels in the various strains expressing each reporter construct, we found evidence of an additional, unexpected change in mRNA stability that was dependent on the presence of a premature stop codon. First, as described in the previous section, the lowest levels of nonsense-containing mRNAs in the wild-type and [PSI+] strains correlated with the lowest levels of readthrough (Fig. 8A ▶). In some cases, we also made the surprising observation that the nonsense-containing transcript was more stable than the corresponding control mRNA containing a sense codon in the readthrough position. This was particularly evident in the [PSI+]/upf1Δ strain carrying the QXQ reporter plasmids, where the nonsense mRNAs were 1.9- to 2.0-fold more abundant than the control mRNAs encoding full-length proteins (Fig. 8B,D ▶). A similar stabilization of nonsense-containing mRNAs was observed in both the upf1Δ and [PSI+]/upf1Δ strains carrying the dual luciferase reporters (Fig. 8C,E ▶). In this case, the nonsense-containing mRNAs were 1.4- to 1.7-fold more abundant than control mRNAs in the upf1Δ strain, and 1.5- to 1.9-fold more abundant than the control mRNAs in the [PSI+]/upf1Δ strain. Although these effects were observed in strains with some of the highest levels of readthrough, this trend was not observed in other strains with a high level of corrected readthrough and an intact NMD apparatus. For example, the [PSI+] strain with the QXQ reporter plasmids did not show this effect (see Table 1 ▶). Taken together, these results suggest that both a high level of readthrough and inactivation of the NMD pathway are necessary for this preferential stabilization of nonsense-containing mRNAs.
FIGURE 8.
Comparison of relative mRNA levels (nonsense/sense). (A) The relative levels of LacZ mRNA in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains expressed from the SXA reporters. (B) The relative levels of LacZ mRNA in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains expressed from the QXQ reporters. (C) The relative levels of dual luciferase mRNA in the wild-type, [PSI+], upf1Δ, and [PSI+]/upf1Δ strains. (D) Representative Northern blot data from the wild-type and [PSI+]/upf1Δ strains expressing the β-galactosidase QXQ reporters. (E) Representative Northern blot data from the wild-type and [PSI+]/upf1Δ strains expressing the dual luciferase reporters. WT, wild type.
DISCUSSION
Recent studies have shown that mutations in the UPF1, UPF2, and UPF3 genes reduce the efficiency of translation termination (Maderazo et al. 2000; Wang et al. 2001). Our systematic examination of the effects of the upf1Δ mutation on translation termination at different termination signals and in different sequence contexts revealed that the upf1Δ mutation causes a general decrease in the efficiency of translation termination at UAG, UAA, and UGA stop codons. These results are in general agreement with a previous report by Bidou et al. using a dual LacZ-Luc reporter system (Bidou et al. 2000). Overall, we found that the loss of Upf1p function conferred readthrough at levels that were generally comparable to the readthrough observed in a [PSI+] strain. Furthermore, the magnitude of these decreases in the efficiency of translation termination associated with [PSI+] and upf1Δ were larger in strains that harbored both defects than in strains carrying either defect alone. Although the context dependence of readthrough observed in both the [PSI+] and upf1Δ strains showed distinct and strong biases (ranging from 12- to 104-fold) toward certain sequence contexts, we found that the context effects associated with the upf1Δ mutation largely superceded those associated with [PSI+] when both defects were present. It was previously proposed that Upf1p associates with eRF1 and eRF3 while they are still present in the ribosome-bound termination complex (Czaplinski et al. 1998; Wang et al. 2001). Our results support this conclusion, and further suggest that this association may occur prior to polypeptide chain release, because we found that the upf1Δ mutation can influence the efficiency of translation termination as a function of both the tetranucleotide termination signal and surrounding sequence context. Further studies will be required to determine whether the upf2Δ or upf3Δ mutations can produce similar effects.
Previous studies have shown that the suppression of an amber (UAG) stop mutation in the yeast URA3 (Losson and Lacroute 1979) and SUC2 (Gozalbo and Hohmann 1990) genes due to the introduction of amber suppressor tRNA genes resulted in a significant increase in mRNA abundance. Those results provided the first evidence that a partial suppression of nonsense mutations could antagonize the effects of NMD. Our analysis of the readthrough associated with different tetranucleotide termination signals and sequence contexts in otherwise identical β-galactosidase transcripts gave us a unique opportunity to correlate the level of stop codon readthrough with mRNA stability in a more comprehensive manner. We found two distinct consequences caused by the introduction of a premature stop mutation in the β-galactosidase mRNA that can be attributed to NMD. First, the abundance of the nonsense-containing mRNA was slightly destabilized (~75% of normal) when readthrough was ≥0.5%. In addition, we observed a greater decrease in the steady-state level of β-galactosidase mRNAs (as much as 2.3-fold lower than wild type) when these mutations were associated with lower levels of readthrough (≤0.5%). Both of these effects depended on an intact NMD machinery, because both were lost when the upf1Δ mutation was present. Consistent with this conclusion, the effects we observed were comparable to the changes in mRNA abundance observed in other studies of NMD in yeast. For example, it was reported that a PGK1(cup1) mRNA carrying a premature stop mutation showed a two- to threefold decrease in stability due to NMD (Muhlrad and Parker 1999). Similarly, the can1–100 premature stop mutation led to a fourfold decrease in the abundance of the CAN1 mRNA (Maderazo et al. 2000). Thus, the changes in LacZ mRNA abundance observed in the current yeast study are comparable to NMD-induced changes in mRNA stability documented previously.
It was previously reported that LacZ mRNAs containing premature stop mutations are not susceptible to the effects of NMD in yeast (Wang et al. 2001). In contrast, we observed a destabilization of LacZ mRNAs by NMD in the current study. It is likely that subtle differences between the reporter constructs used are responsible for this discrepancy. In particular, we found that the major destabilization of mRNAs by NMD occurred in our study only when they were associated with very low levels of readthrough. The translational threshold we found for the onset of this “severe” form of NMD (≤0.5% readthrough) was significantly lower than the 1%–3% readthrough observed with the β-galactosidase constructs used in the previous study (Wang et al. 2001). Thus, it is likely that the absence of NMD in their study is consistent with our conclusion that the major form of NMD is induced only in mRNAs that exhibit a very low level of readthrough.
The 0.5% readthrough threshold we observed for the onset of the major phase of NMD represents a ribosome progressing through the premature stop codon only once in 200 attempts. This indicates that a very low level of translational readthrough of a premature stop mutation can suppress the onset of the most severe phase of NMD in yeast cells. Such a mechanism could have evolved to protect yeast mRNAs that are weakly expressed, and possibly genes whose translation requires the suppression of a leaky stop codon (Namy et al. 2003). More importantly, this observation strongly suggests that the fate of an mRNA is not determined solely during the initial round of translation in yeast cells. NMD in mammalian cells retains many of the same features as NMD in yeast, including participation by homologs of the Upf1p, Upf2p, and Upf3p components of the surveillance complex. However, it has been proposed that NMD is exerted primarily at the first (or “pioneer”) round of translation (Ishigaki et al. 2001; Lejeune et al. 2002). Our results in the yeast system are more consistent with a model in which the mechanism controlling the onset of NMD can be activated following translation termination during each successive round of translation. Evidence supporting this conclusion was also reached in another recent study, where strains lacking the UPF1, UPF2, or UPF3 genes were used in conjunction with plasmids that allowed their expression under the control of an inducible promoter. It was shown that preexisting mRNAs containing premature stop mutations could be promptly destabilized once the expression of the components of an intact surveillance complex was restored (Maderazo et al. 2003). When taken together with our results, a growing body of evidence suggest that mRNA surveillance by NMD in yeast is an ongoing process that occurs throughout the lifetime of the mRNA molecule.
This conclusion implies that NMD in yeast has evolved to be an inherently inefficient process to discriminate between these subtle differences in the level of readthrough. It is possible that this inefficiency in NMD may be due to the limitation of one (or more) components of the surveillance complex. It has been reported that Upf1p is present in large excess over Upf3p in yeast cells (Atkin et al. 1997). More recently, it was estimated that a typical yeast cell contains roughly 1600 molecules of Upf1p, 160 molecules of Upf2p, and 80 molecules of Upf3p (Maderazo et al. 2000). These data are more consistent with the notion that the components of the NMD machinery may be recruited in a sequential manner, rather than forming as one large preassembled complex directly at the premature stop codon (Atkin et al. 1997; Shirley et al. 1998). Because the complete absence of any one of these components eliminates NMD, it is possible that the level of the least abundant of the three components (Upf3p) may determine the absolute efficiency of NMD in this simple eukaryote.
Muhlrad and Parker (1999) previously used a hybrid PGK1(cup1) reporter mRNA containing a premature stop mutation to show that a threefold increase in the rate of translation initiation occurred upon: (1) the introduction of a upf1Δ mutation, or (2) the removal of the DSE from the mRNA. However, removal of the DSE in a upf1Δ mutation resulted in a 33% decrease in translational efficiency. These results indicate that translation initiation of an mRNA increases in a upf1Δ strain when a DSE is present, but decreases in a upf1Δ strain when the DSE is absent. We observed a twofold decrease in Renilla expression (per unit mRNA) in both upf1Δ and upf1Δ/[PSI+] strains. Assuming that the upf1Δ mutation does not affect the rate of translation elongation, these results suggest that the absence of the NMD pathway function led to a twofold decrease in the efficiency of translation initiation of the dual luciferase mRNA. Because we did not observe NMD with the dual luciferase mRNA in the wild-type strain, it either lacks a DSE or the level of readthrough of the premature stop codon in that mRNA remained above the threshold level required for the induction of the major component of NMD. Thus, our observation of a twofold decrease in translation initiation is generally consistent with the previous observation that the lack of a DSE leads to a reduction of translation initiation in a upf1Δ strain. When taken together with our finding that the readthrough threshold may represent a critical determinant for the induction of the major component of NMD (as demonstrated with the β-galactosidase reporters), these results raise the intriguing possibility that factors that bind to the DSE may not only mediate the onset of NMD, but may also regulate the translational efficiency of mRNAs that contain a premature stop codon.
We also found that some nonsense-containing mRNAs from both the β-galactosidase and dual luciferase constructs become more stable than control mRNAs when expressed in the upf1Δ and/or upf1Δ/[PSI+] strains. These effects were observed in strains with some of the highest levels of readthrough. However, other strains with an intact NMD apparatus as well as a high level of corrected readthrough, such as the [PSI+] strain carrying the QXQ reporter plasmids, did not show this effect. From these results we conclude that this differential mRNA stability is normally suppressed by a Upf1p-dependent mechanism. This effect could result from either a preferential stabilization of the nonsense-containing mRNAs, or a destabilization of normal mRNAs that lack a premature stop codon. Although the components of the surveillance complex are frequently thought to be involved only in NMD, a upf1Δ mutation has also been shown to influence the abundance of a wide range of normal cellular mRNAs (Lelivelt and Culbertson 1999). Our results indicate that a differential effect between nonsense-containing and normal mRNAs are also most evident in a [PSI+] background, suggesting that the absence of Upf1p in conjunction with a reduced availability of eRF3 in the termination complex could lead to these complex effects on mRNA stability. It is also possible that this differential mRNA stability is obscured or superceded by the normal function of the surveillance complex.
MATERIALS AND METHODS
Strains and media
In a previous study, we used the yeast strain YDB108 (MATa ura2–52 his3Δ200 trp1–Δ901 ade2–101 leu2–3, 112 Δpep4 :: LEU2 Δste6 :: HIS3) to characterize the efficiency of translation termination using β-galactosidase readthrough assays (Bonetti et al. 1995). We subsequently found that the strain YDB108 is [PSI+]. This strain was cured by growth in the presence of guanidine hydrochloride, resulting in the [psi−] strain YDB342. The UPF1 gene was knocked out in strain YDB342 to create YDB343 (MATa ura2– 52 his3Δ200 trp1–Δ901 ade2–101 leu2–3, 112 Δpep4 :: LEU2 Δste6 :: HIS3 Δupf1 :: hisG). UPF1 was also knocked out in strain YDB108 to create YDB339 (MATa ura2–52 his3Δ200 trp1–Δ901 ade2–101 leu2–3, 112 Δpep4 :: LEU2 Δste6 :: HIS3 Δupf1 :: hisG [PSI+]). Cultures were grown in minimal medium containing 0.67% (w/v) yeast nitrogen base without amino acids (Difco) containing 2% (w/v) glucose (SMD plates) or 2% galactose (SMGal plates) as specified. Additional nutritional supplements were added as required (Adams et al. 1997).
Construction of readthrough reporter systems
Construction of the β-galactosidase readthrough reporter plasmids containing each of the 12 tetranucleotide termination signals and the four related (sense) control constructs was previously described (Bonetti et al. 1995). The initial construction of dual luciferase reporter plasmids used to monitor the efficiency of translation termination in mammalian cells was previously described (Grentzmann et al. 1998). The dual luciferase constructs were adapted for expression in yeast by subcloning a 2.6 kb DNA fragment into the pYEplac195 yeast expression plasmid (Gietz and Sugino 1988) that carries the yeast PGK promoter and the CYC2 transcription terminator.
β-Galactosidase readthrough assays
The β-galactosidase reporter plasmid constructs were transformed into the yeast strains described above, and transformants were selected on SMD dropout plates lacking uracil (Adams et al. 1997). Transformed strains were grown in SMGal medium to a cell density of approximately 0.75 A600 units/mL as measured using a Shimadzu UV-1201 spectrophotometer. Cells were then harvested and β-galactosidase activity was assayed using standard conditions (Guarente 1983). The final values are expressed as the mean ± the standard deviation.
In all cases, β-galactosidase activity was expressed per unit mRNA. This was done by correcting for the relative steady-state β-galactosidase mRNA level as measured by Northern blot analysis. To do this, strains were grown under the same conditions used for the readthrough assays and total cellular RNA was isolated by SDS/phenol extraction (Schmitt et al. 1990). An equal amount of RNA (10 μg) from each strain was subjected to agarose gel electrophoresis in the presence of formaldehyde. Following transfer to nitrocellulose, the blots were probed with a 2558 bp PvuII fragment from the LacZ gene. The blot was stripped and reprobed with a 420-bp fragment from the yeast ACT1 gene to serve as an internal control. The ACT1 probe was prepared by PCR amplification from genomic DNA using the primers DB154 (5′-GCGCGGAATTCAACGTTCCAGCCTTCTAC-3′) and DB155 (5′-GGATGGAACAAAGCTTCTGG-3′). All probes were labeled with [α-32P]-dATP using the random hexamer method (Sambrook and Russell 2001). Radioactivity in specific hybrids was quantified by PhosphorImager analysis (Molecular Dynamics). LacZ mRNA levels were corrected for each strain by normalizing the lacZ-specific counts to the ACT1-specific counts. These values were then normalized to the same measurements obtained with the sense control for each set of constructs (containing a UGG tryptophan codon in place of the UAG, UAA, or UGA stop codon) to determine the theoretical maximal level of expression (i.e., 100% readthrough) for this reporter system. Independent mRNA measurements were carried out multiple times for each construct with similar results, and the data is expressed as the mean ± standard deviation (SD). Statistical analysis using the Mann-Whitney nonparametric test was carried out using the InStat 3 program (GraphPad Software).
Luciferase assays and Northern analysis
Dual luciferase reporter plasmids expressing either a stop codon or a sense codon were transformed into the indicated yeast strains, and transformants were selected on SMD dropout plates lacking uracil (Adams et al. 1997). Transformed strains were grown in liquid SMD medium to a cell density of 0.5–0.7 A600 units/mL as measured using a Shimadzu UV-1201 spectrophotometer. The luciferase assay was performed using the Dual-Luciferase Reporter Assay System (Promega). Approximately 104 yeast cells from each strain expressing the indicated dual luciferase reporter were lysed using 100 μL of Passive Lysis Buffer. Two microliters of the lysate were added to 10 μL of the Luciferase Assay Reagent II. Relative luminescence units (RLUs) produced by firefly luciferase activity were then measured for 10 sec using a Berthold Lumat LB9507 luminometer. Ten microliters of Stop&Glo buffer was then added to quench the firefly activity and activate the Renilla luciferase activity. RLUs were again measured for 10 sec to determine the Renilla luciferase activity. Negative controls that contained all the reaction components except cell lysates were used to determine the background for each luciferase reaction and were subtracted from the experimental values obtained. In all cases, the background units were less than 0.5% of the experimental values. The percent readthrough is expressed as the mean ± the standard deviation.
To calculate the Renilla specific activity and translational efficiency for each yeast strain, 104 cells were lysed using 100 μL of Passive Lysis Buffer. Two microliters of each lysate were used to determine the total protein concentration (in quadruplicate) using the Bio-Rad Protein Assay. The protein concentrations were then used to calculate the Renilla specific activity (RLU/min/ng total protein) and to determine the total Renilla produced per unit RNA (the translational efficiency). The translational efficiency values are expressed as the mean ± the standard deviation.
For Northern blot analysis of the dual luciferase mRNAs, strains were grown under the same conditions used as the cells harvested for readthrough assays. The RNA was isolated and analyzed as described in the preceding section. The probe for the Renilla luciferase was constructed by PCR amplification of a 935-bp fragment from the dual luciferase reporter plasmid using DB907 (5′-GGCCGGATCCATGACTTCGAAAGTTTATGA-3′) and DB908 (5′-GGCCGTCGACATTTGTTCATTTTTGAGAAC-3′). For all samples, the luciferase mRNA levels were normalized to actin mRNA on the same blot. Independent mRNA measurements were carried out multiple times for each construct with similar results, and the data is expressed as the mean ± standard deviation (SD).
Acknowledgments
The authors thank John Atkins and Lauren Petros for generously providing their extensive set of mammalian dual luciferase readthrough reporter constructs, Vytas Bankaitis for providing the PGK promoter construct, and Ambro van Hoof for helpful comments on the manuscript. This work was supported by NIH Grant RO1 GM 068854 (to D.M.B.). Postdoctoral fellowships were provided by NIH Grants T32 DK07545 (to A.K.A.), T32 AI07150 (to K.M.K.), and T32 HL07553 (to L.G.). A predoctoral fellowship was also provided by the Cystic Fibrosis Foundation Grant R464 (to J.S.M.).
The publication costs of this article were defrayed in part by payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 USC section 1734 solely to indicate this fact.
Article and publication are at http://www.rnajournal.org/cgi/doi/10.1261/rna.5147804.
REFERENCES
- Adams, A., Gottschling, D.E., Kaiser, C.A., and Stearns, T. 1997. Methods in yeast genetics: A laboratory course manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- Atkin, A.L., Altamura, N., Leeds, P., and Culbertson, M.R. 1995. The majority of yeast UPF1 co-localizes with polyribosomes in the cytoplasm. Mol. Biol. Cell 6: 611–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atkin, A.L., Schenkman, L.R., Eastham, M., Dahlseid, J.N., Lelivelt, M.J., and Culbertson, M.R. 1997. Relationship between yeast polyribosomes and Upf proteins required for nonsense mRNA decay. J. Biol. Chem. 272: 22163–22172. [DOI] [PubMed] [Google Scholar]
- Beelman, C.A., Stevens, A., Caponigro, G., LaGrandeur, T.E., Hatfield, L., Fortner, D.M., and Parker, R. 1996. An essential component of the decapping enzyme required for normal rates of mRNA turnover. Nature 382: 642–646. [DOI] [PubMed] [Google Scholar]
- Bertram, G., Innes, S., Minella, O., Richardson, J., and Stansfield, I. 2001. Endless possibilities: Translation termination and stop codon recognition. Microbiology 147: 255–269. [DOI] [PubMed] [Google Scholar]
- Bidou, L., Stahl, G., Hatin, I., Namy, O., Rousset, J.P., and Farabaugh, P.J. 2000. Nonsense-mediated decay mutants do not affect programmed—1 Frameshifting. RNA 6: 952–961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonetti, B., Fu, L., Moon, J., and Bedwell, D.M. 1995. The efficiency of translation termination is determined by a synergistic interplay between upstream and downstream sequences in Saccharomyces cerevisiae. J. Mol. Biol. 251: 334–345. [DOI] [PubMed] [Google Scholar]
- Brown, C.M., Stockwell, P.A., Trotman, C.N., and Tate, W.P. 1990. Sequence analysis suggests that tetra-nucleotides signal the termination of protein synthesis in eukaryotes. Nucleic Acids Res. 18: 6339–6345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chernoff, Y.O., Vincent, A., and Liebman, S.W. 1994. Mutations in eukaryotic 18S ribosomal RNA affect translational fidelity and resistance to aminoglycoside antibiotics. EMBO J. 13: 906–913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Culbertson, M.R. 1999. RNA surveillance. Unforeseen consequences for gene expression, inherited genetic disorders and cancer. Trends Genet. 15: 74–80. [DOI] [PubMed] [Google Scholar]
- Czaplinski, K., Ruiz-Echevarria, M.J., Paushkin, S.V., Han, X., Weng, Y., Perlick, H.A., Dietz, H.C., Ter-Avanesyan, M.D., and Peltz, S.W. 1998. The surveillance complex interacts with the translation release factors to enhance termination and degrade aberrant mRNAs. Genes & Dev. 12 1665–1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decker, C.J. and Parker, R. 1993. A turnover pathway for both stable and unstable mRNAs in yeast: Evidence for a requirement for deadenylation. Genes & Dev. 7: 1632–1643. [DOI] [PubMed] [Google Scholar]
- Fearon, K., McClendon, V., Bonetti, B., and Bedwell, D.M. 1994. Premature translation termination mutations are efficiently suppressed in a highly conserved region of yeast Ste6p, a member of the ATP-binding cassette (ABC) transporter family. J. Biol. Chem. 269: 17802–17808. [PubMed] [Google Scholar]
- Gietz, R.D. and Sugino, A. 1988. New yeast—Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene 74: 527–534. [DOI] [PubMed] [Google Scholar]
- Gozalbo, D. and Hohmann, S. 1990. Nonsense suppressors partially revert the decrease of the mRNA level of a nonsense mutant allele in yeast. Curr. Genet. 17: 77–79. [DOI] [PubMed] [Google Scholar]
- Grentzmann, G., Ingram, J.A., Kelly, P.J., Gesteland, R.F., and Atkins, J.F. 1998. A dual-luciferase reporter system for studying recoding signals. RNA 4: 479–486. [PMC free article] [PubMed] [Google Scholar]
- Guarente, L. 1983. Yeast promoters and LacZ fusions designed to study expression of cloned genes in yeast. Methods Enzymol. 101: 181–191. [DOI] [PubMed] [Google Scholar]
- Hagan, K.W., Ruiz-Echevarria, M.J., Quan, Y., and Peltz, S.W. 1995. Characterization of cis-acting sequences and decay intermediates involved in nonsense-mediated mRNA turnover. Mol. Cell. Biol. 15: 809–823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He, F. and Jacobson, A. 2001. Upf1p, Nmd2p, and Upf3p regulate the decapping and exonucleolytic degradation of both nonsense-containing mRNAs and wild-type mRNAs. Mol. Cell. Biol. 21: 1515–1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilleren, P. and Parker, R. 1999. mRNA surveillance in eukaryotes: Kinetic proofreading of proper translation termination as assessed by mRNP domain organization? RNA 5: 711–719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard, M.T., Shirts, B.H., Petros, L.M., Flanigan, K.M., Gesteland, R.F., and Atkins, J.F. 2000. Sequence specificity of aminoglycoside-induced stop codon readthrough: Potential implications for treatment of Duchenne muscular dystrophy. Ann. Neurol. 48: 164–169. [PubMed] [Google Scholar]
- Ishigaki, Y., Li, X., Serin, G., and Maquat, L.E. 2001. Evidence for a pioneer round of mRNA translation: mRNAs subject to nonsense-mediated decay in mammalian cells are bound by CBP80 and CBP20. Cell 106: 607–617. [DOI] [PubMed] [Google Scholar]
- Ito, K., Uno, M., and Nakamura, Y. 2000. A tripeptide “anticodon” deciphers stop codons in messenger RNA. Nature 403: 680–684. [DOI] [PubMed] [Google Scholar]
- Kisselev, L., Ehrenberg, M., and Frolova, L. 2003. Termination of translation: Interplay of mRNA, rRNAs and release factors? EMBO J. 22: 175–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Hir, H., Izaurralde, E., Maquat, L.E., and Moore, M.J. 2000. The spliceosome deposits multiple proteins 20–24 nucleotides upstream of mRNA exon–exon junctions. EMBO J. 19: 6860–6869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leeds, P., Peltz, S.W., Jacobson, A., and Culbertson, M.R. 1991. The product of the yeast UPF1 gene is required for rapid turnover of mRNAs containing a premature translational termination codon. Genes & Dev. 5: 2303–2314. [DOI] [PubMed] [Google Scholar]
- Leeds, P., Wood, J.M., Lee, B.S., and Culbertson, M.R. 1992. Gene products that promote mRNA turnover in Saccharomyces cerevisiae. Mol. Cell. Biol. 12: 2165–2177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lejeune, F., Ishigaki, Y., Li, X., and Maquat, L.E. 2002. The exon junction complex is detected on CBP80-bound but not eIF4E-bound mRNA in mammalian cells: Dynamics of mRNP remodeling. EMBO J. 21: 3536–3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lelivelt, M.J. and Culbertson, M.R. 1999. Yeast Upf proteins required for RNA surveillance affect global expression of the yeast transcriptome. Mol. Cell. Biol. 19: 6710–6719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, R. and Liebman, S.W. 1996. A translational fidelity mutation in the universally conserved sarcin/ricin domain of 25S yeast ribosomal RNA. RNA 2: 254–263. [PMC free article] [PubMed] [Google Scholar]
- Loftfield, R.B. and Vanderjagt, D. 1972. The frequency of errors in protein biosynthesis. Biochem. J. 128: 1353–1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Losson, R. and Lacroute, F. 1979. Interference of nonsense mutations with eukaryotic messenger RNA stability. Proc. Natl. Acad. Sci. 76: 5134–5137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maderazo, A.B., He, F., Mangus, D.A., and Jacobson, A. 2000. Upf1p control of nonsense mRNA translation is regulated by Nmd2p and Upf3p. Mol. Cell. Biol. 20: 4591–4603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maderazo, A.B., Belk, J.P., He, F., and Jacobson, A. 2003. Nonsense-containing mRNAs that accumulate in the absence of a functional nonsense-mediated mRNA decay pathway are destabilized rapidly upon its restitution. Mol. Cell. Biol. 23: 842–851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mendell, J.T., Medghalchi, S.M., Lake, R.G., Noensie, E.N., and Dietz, H.C. 2000. Novel Upf2p orthologues suggest a functional link between translation initiation and nonsense surveillance complexes. Mol. Cell. Biol. 20: 8944–8957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori, N., Funatsu, Y., Hiruta, K., and Goto, S. 1985. Analysis of translational fidelity of ribosomes with protamine messenger RNA as a template. Biochemistry 24: 1231–1239. [DOI] [PubMed] [Google Scholar]
- Muhlrad, D. and Parker, R. 1994. Premature translational termination triggers mRNA decapping. Nature 370: 578–581. [DOI] [PubMed] [Google Scholar]
- Muhlrad, D. and Parker, R. 1999. Recognition of yeast mRNAs as “nonsense containing” leads to both inhibition of mRNA translation and mRNA degradation: Implications for the control of mRNA decapping. Mol. Biol. Cell 10: 3971–3978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagy, E. and Maquat, L.E. 1998. A rule for termination-codon position within intron-containing genes: When nonsense affects RNA abundance. Trends Biochem. Sci. 23: 198–199. [DOI] [PubMed] [Google Scholar]
- Namy, O., Duchateau-Nguyen, G., Hatin, I., Hermann-Le Denmat, S., Termier, M., and Rousset, J.P. 2003. Identification of stop codon readthrough genes in Saccharomyces cerevisiae. Nucleic Acids Res. 31: 2289–2296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peltz, S.W., Brown, A.H., and Jacobson, A. 1993. mRNA destabilization triggered by premature translational termination depends on at least three cis-acting sequence elements and one trans-acting factor. Genes & Dev. 7: 1737–1754. [DOI] [PubMed] [Google Scholar]
- Poole, E.S., Major, L.L., Mannering, S.A., and Tate, W.P. 1998. Translational termination in Escherichia coli: Three bases following the stop codon crosslink to release factor 2 and affect the decoding efficiency of UGA-containing signals. Nucleic Acids Res. 26: 954–960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz-Echevarria, M.J. and Peltz, S.W. 1996. Utilizing the GCN4 leader region to investigate the role of the sequence determinants in nonsense-mediated mRNA decay. EMBO J. 15: 2810–2819. [PMC free article] [PubMed] [Google Scholar]
- Sambrook, J. and Russell, D.W. 2001. Molecular cloning: A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- Schmitt, M.E., Brown, T.A., and Trumpower, B.L. 1990. A rapid and simple method for preparation of RNA from Saccharomyces cerevisiae. Nucleic Acids Res. 18: 3091–3092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serio, T.R. and Lindquist, S.L. 1999. [PSI+]: An epigenetic modulator of translation termination efficiency. Annu. Rev. Cell Dev. Biol. 15: 661–703. [DOI] [PubMed] [Google Scholar]
- Shirley, R.L., Lelivelt, M.J., Schenkman, L.R., Dahlseid, J.N., and Culbertson, M.R. 1998. A factor required for nonsense-mediated mRNA decay in yeast is exported from the nucleus to the cytoplasm by a nuclear export signal sequence. J. Cell Sci. 111: 3129–3143. [DOI] [PubMed] [Google Scholar]
- Shirley, R.L., Ford, A.S., Richards, M.R., Albertini, M., and Culbertson, M.R. 2002. Nuclear Import of Upf3p is mediated by importin-α/-β and export to the cytoplasm is required for a functional nonsense-mediated mRNA decay pathway in yeast. Genetics 161: 1465–1482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stansfield, I., Jones, K.M., Herbert, P., Lewendon, A., Shaw, W.V., and Tuite, M.F. 1998. Missense translation errors in Saccharomyces cerevisiae. J. Mol. Biol. 282: 13–24. [DOI] [PubMed] [Google Scholar]
- Velichutina, I.V., Dresios, J., Hong, J.Y., Li, C., Mankin, A., Synetos, D., and Liebman, S.W. 2000. Mutations in helix 27 of the yeast Saccharomyces cerevisiae 18S rRNA affect the function of the decoding center of the ribosome. RNA 6: 1174–1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velichutina, I.V., Hong, J.Y., Mesecar, A.D., Chernoff, Y.O., and Liebman, S.W. 2001. Genetic interaction between yeast Saccharomyces cerevisiae release factors and the decoding region of 18 S rRNA. J. Mol. Biol. 305: 715–727. [DOI] [PubMed] [Google Scholar]
- Wang, W., Czaplinski, K., Rao, Y., and Peltz, S.W. 2001. The role of Upf proteins in modulating the translation read-through of nonsense-containing transcripts. EMBO J. 20: 880–890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zavialov, A.V., Buckingham, R.H., and Ehrenberg, M. 2001. A posttermination ribosomal complex is the guanine nucleotide exchange factor for peptide release factor RF3. Cell 107: 115–124. [DOI] [PubMed] [Google Scholar]
- Zavialov, A.V., Mora, L., Buckingham, R.H., and Ehrenberg, M. 2002. Release of peptide promoted by the GGQ motif of class 1 release factors regulates the GTPase activity of RF3. Mol. Cell 10: 789– 798. [DOI] [PubMed] [Google Scholar]