Abstract
Alcoholic liver disease is associated with abnormal hepatic methionine metabolism and folate deficiency. Because folate is integral to the methionine cycle, its deficiency could promote alcoholic liver disease by enhancing ethanol-induced perturbations of hepatic methionine metabolism and DNA damage. We grouped 24 juvenile micropigs to receive folate-sufficient (FS) or folate-depleted (FD) diets or the same diets containing 40% of energy as ethanol (FSE and FDE) for 14 wk, and the significance of differences among the groups was determined by ANOVA. Plasma homocysteine levels were increased in all experimental groups from 6 wk onward and were greatest in FDE. Ethanol feeding reduced liver methionine synthase activity, S-adenosylmethionine (SAM), and glutathione, and elevated plasma malondialdehyde (MDA) and alanine transaminase. Folate deficiency decreased liver folate levels and increased global DNA hypomethylation. Ethanol feeding and folate deficiency acted together to decrease the liver SAM/S-adenosylhomocysteine (SAH) ratio and to increase liver SAH, DNA strand breaks, urinary 8-oxo-2′-deoxyguanosine [oxo(8)dG]/mg of creatinine, plasma homocysteine, and aspartate transaminase by more than 8-fold. Liver SAM correlated positively with glutathione, which correlated negatively with plasma MDA and urinary oxo(8)dG. Liver SAM/SAH correlated negatively with DNA strand breaks, which correlated with urinary oxo(8)dG. Livers from ethanol-fed animals showed increased centrilobular CYP2E1 and protein adducts with acetaldehyde and MDA. Steatohepatitis occurred in five of six pigs in FDE but not in the other groups. In summary, folate deficiency enhances perturbations in hepatic methionine metabolism and DNA damage while promoting alcoholic liver injury.
Folate deficiency is among the most common nutritional abnormalities in chronic alcoholic patients, especially in those who have developed alcoholic liver injury (1–5). In addition to poor diet, folate deficiency in chronic alcoholism can be ascribed to decreased intestinal absorption and hepatic uptake, increased renal excretion, and increased oxidative cleavage of the folate molecule (6–12). Folate in its 5-methyltetrahydrofolate (5-MTHF) form is integral to methionine metabolism. Folate deficiency perturbs hepatic methionine metabolism (13, 14), which is associated with DNA nucleotide imbalance and increased hepatocellular apoptosis in experimental animals fed folate-deficient (FD) diets or exposed to chronic ethanol (15, 16).
Hepatic methionine metabolism is regulated by the availability of dietary and endogenous folate that appears in the circulation as 5-MTHF and is the substrate with cofactor vitamin B12 for the methionine synthase (MS) reaction that generates methionine from homocysteine (Hcy) (see supporting information, which is published on the PNAS web site, www.pnas.org). In the alternate salvage pathway for methionine synthesis, choline is the precursor of betaine, which is the substrate for betaine homocysteine methyltransferase (BHMT). The methionine adenosyl transferase (MAT) reaction adds ATP to methionine for generation of S-adenosylmethionine (SAM). Two different genes express different isoforms of MAT. MAT1A encodes the catalytic unit that is expressed as a dimer, MATIII, and as a tetramer, MATI, in the liver and is capable of converting liver methionine to 6–8 g of SAM per day (17, 18). MAT2A encodes a different catalytic subunit and expresses MATII in fetal and extrahepatic tissues. SAM is the principal methyl donor in methylation reactions, including DNA methyltransferases. By its methyl donation, SAM is converted to S-adenosylhomocysteine (SAH), which is substrate for reversible SAH hydrolase in the generation of Hcy. Therefore, Hcy can increase SAH, and elevations in liver Hcy and SAH, together with decreased SAM synthesis, reduce the SAM/SAH ratio. Although not an independent indicator, the SAM/SAH ratio can be used as an adjunctive descriptor of opposing changes in SAM and SAH (18). SAH levels correlate closely with Hcy, and SAH is an effective inhibitor of methylation, such that global DNA hypomethylation increases in direct proportion to plasma SAH and Hcy levels (19). SAM regulates total or reduced glutathione (GSH) by its up-regulation of cystathionine β synthase and the transsulfuration pathway (20). GSH is oxidized to its oxidized form (GSSG), and the GSH/GSSG ratio is considered an accurate measure of overall oxidative state (21). In addition, SAM provides negative feedback to the methylenetetrahydrofolate reductase (MTHFR) reaction that converts 5,10-methylenetetrahydrofolate (5,10-MTHF) to 5-MTHF (22). Thus, SAM deficiency accelerates the use of 5,10-MTHF for the MTHFR reaction but decreases its availability for the alternate thymidylate synthetase reaction, which normally maintains the nucleotide balance of deoxyuridine monophosphate (dUMP) and deoxythymidine monophosphate (dTMP) (15).
Prior studies in animal models demonstrated multiple effects of chronic ethanol feeding on methionine metabolism. Three different ethanol feeding studies in rodents described reduced MS and SAM with compensatory increase in BHMT (23–25). Rats fed ethanol by intragastric tube and then challenged with lipopolysaccharide endotoxin showed reduced liver MATI/III and increased MATII together with DNA hypomethylation and increased DNA strand breaks (26). Provision of SAM attenuated liver injury, raised hepatic GSH levels, and improved hepatic mitochondrial histology and function in ethanol-fed rats and baboons (27, 28). In the rat model, SAM prevented ethanol-induced reductions in mitochondrial membrane fluidity and enhanced the transport of GSH to its effective mitochondrial site (29).
The present study explores the hypothesis that folate deficiency promotes the development of alcoholic liver injury in the micropig by enhancing ethanol-induced perturbations of hepatic methionine metabolism. The bases for this hypothesis include the critical interaction of folate in methionine metabolism and experimental evidence for a variety of perturbations in methionine metabolism after development of alcoholic liver injury or clinical disease. Proof of this hypothesis would provide greater insights into the significance of altered methionine metabolism in development of alcoholic liver injury and could contribute to recommendations on the use of folate, or SAM intervention in the prevention or treatment of alcoholic liver disease.
Methods
Animals and Diets.
Twenty-four noncastrated male Yucatan micropigs 6 mo of age weighing ≈20 kg each were obtained from Sinclair Farms (Columbia, MO) and were housed in individual kennels at the University of California, Davis (UCD) Animal Resources Center. After a 1-mo period of dietary adaptation on the folate-sufficient (FS) control diet, the pigs were placed in groups of six each to receive one of four different diets. The FS diet provided 90 kcal (1 kcal = 4.18 kJ)/kg of body weight, distributed as vitamin-free casein at 11% of calories or 2 g/kg of body weight, 34% of calories as polyunsaturated corn oil, 55% of calories as cornstarch, and vitamin and mineral mix to provide all essential nutrients for growing swine, including levels that were in excess of requirements for folate at 14.5 μg, choline at 60.3 mg, and methionine at 675 mg/kg of body weight (30) (Dyets, Bethlehem, PA). Dietary total folate levels were confirmed in aliquots of each diet by the microbiological assay (31). The FD diet was formulated similarly but with complete deletion of folate from the vitamin mix. The FS/ethanol diet (FSE) and the combined FD/ethanol (FDE) diets were identical to the FS and FD diets except for the substitution of 40% of calories as ethanol for cornstarch. The diets were provided as a slurry in three equal daily portions at 0800, 1200, and 1600 hr. Mean daily intakes and body weights were determined weekly for each group, and mean caloric intakes per kg of body weight in the FDE group were used as the basis for dietary provision to each animal in the other three groups. The UCD Animal Welfare Committee approved housing, care, and all procedures performed on the micropigs. The UCD Animal Resources Center facilities are approved by the National Institutes of Health, and care of the micropigs followed the standards and procedures outlined in the National Academy of Sciences “Guide for the Care and Use of Laboratory Animals.” Micropigs were bled in the fasting state before the morning meal by central venous puncture every 2 wk for assays of plasma Hcy. After 14 wk, the micropigs were fasted overnight and anesthetized by ketamine and isoflurane inhalation, followed by laparotomy and recovery of blood samples by venipuncture, urine by bladder puncture, excision of liver samples and total liver, then killing by terminal exsanguination. Terminal blood samples were kept on ice until plasma separation. Liver samples were immediately freeze-clamped and frozen in liquid nitrogen. Terminal liver, plasma, and urine samples were frozen at −70°C until further use. Liver specimens were also placed in formalin for paraffin blocking and subsequent assessment of hepatic histology and immunohistochemical analyses.
Tissue Folate, Methionine Metabolites, and Enzymes.
After homogenization of terminal liver samples in 0.1 M Na2HPO4, pH 6.0, containing 1% ascorbic acid, polyglutamyl folates were hydrolyzed to their monoglutamyl derivatives by using hog kidney conjugase and quantified by standard microbiological assay with Lactobacillus casei (31). Plasma Hcy and liver homogenate levels of Hcy, methionine, SAM, SAH, GSH, and GSSG were measured by using an HPLC coulometric electrochemical method (32). MS activity was measured in liver cytosol fractions by a method that includes incubation with a mixture of l-Hcy and [14C]5-MTHF substrates and vitamin B12 cofactor (33). BHMT activity was measured in liver homogenates by using pure Hcy and [14C]betaine substrate and Dowex column separation of labeled methionine product (34).
DNA Oxidation and Lipid Peroxidation.
8-Oxo-2′-deoxyguanosine [oxo(8)dG] was measured in terminal bladder urine samples by using isotope-dilution HPLC-electrospray ionization tandem mass spectrometry analysis (35), and the results were standardized to urinary creatinine concentrations (36). MDA was quantified in terminal plasma samples by derivatization with pentafluorophenylhydrazine to the stable adduct N-pentafluorophenylpyrazole followed by gas chromatography/mass spectrometry (37). To determine whether MDA values were affected by circulating plasma lipids, they were regressed against plasma triglyceride levels that were obtained by routine clinical chemistry in the same blood samples.
DNA Methylation and Strand Breaks.
Global hypomethylation was quantified in genomic DNA extracts from liver by using the cytosine extension assay in which unmethylated CpG sites are digested with HpaI, followed by single nucleotide extension with [3H]dCTP, such that the extent of its incorporation is directly proportional to the number of unmethylated sites (38). DNA strand breaks were quantified by the random oligonucleotide-primed synthesis assay, in which single-stranded DNA fragments serve as primers during strand reassociation such that the incorporation of [32P]dCTP is proportional to the number of 3′-OH breaks (39).
Liver Histology and Immunochemistry.
Liver histology was interpreted and scored in terminal samples that were formalin-fixed, sectioned, and stained with hematoxylin/eosin. Histopathology was evaluated blindly by two authors (S.P. and O.N.) and scored for steatosis, necrosis, and inflammation as follows: 0, normal; 1, mild changes; >2, mild to moderate severity; >3, moderate severity; 5, maximum severity. The scores were summed for each animal to arrive at composite scores. As detailed previously (40), immunohistochemical techniques were used to identify and semiquantify protein adducts of acetaldehyde (AA), MDA, and the microsomal ethanol oxidizing enzyme CYP2E1. These procedures used polyclonal rabbit antisera against AA-modified BSA, guinea pig antisera against MDA–low-density lipoprotein (gift of S. Ylä-Herttuala, University of Kuopio, Finland), and monoclonal antibody to rat CYP2E1 (gift of M. Pasanen, University of Oulu, Finland). Liver sections were stained by biotin–streptavidin and, after incubation with primary antibody, were incubated with appropriate secondary antibody linked to peroxidase-conjugated streptavidin before development with 3,3′-diaminobenzidine tetrahydrochloride. Staining intensity was scored independently by the same two investigators who were blind to the knowledge of the histopathology scores as follows: 0, no reaction; 1, weak reaction; 3, moderate reaction; 5, strong reaction. Sections were photographed with a Leitz Aristoplan microscope (Wetzlar, Germany).
Statistical Analyses.
Data were analyzed by multivariate or two-way ANOVA. Subgroup analyses were performed by using one-way ANOVA when significant interactions were found. One-way ANOVA also was used to confirm the additive effect of folate deficiency in group FDE compared with group FSE. Correlations between variables were determined by linear regressions by using all points.
Results
Differences in weight gain were determined by regressing weight against time and calculating the individual slope for each animal. These slopes were then compared by using folate and ethanol as the main effects and including an interaction term. Over 14 wk, micropigs in the FS control group gained a mean of 11.4 kg ± 1.18 (SEM), compared with 6.1 ± 3.0 kg in the FD group, 4.5 ± 1.4 kg in the FSE group, and 3.0 ± 2.45 kg in the combined FDE group. There was an independent negative effect of ethanol feeding (P < 0.02) on weight gain, but no significant effect of folate deficiency. There were no clinical manifestations in pigs consuming the two FD diets other than roughening and scaliness of the skin. Taken 2 hr after the last daily meal, the peak blood alcohol levels in the ethanol-fed groups were similar, and combining the two groups (n = 12) was 402.6 ± 29.2 mg/dl.
The mean plasma Hcy level in the FDE group increased 4-fold to peak at 28 μM after 9 wk, followed by FSE, FD, and FS. Between wks 6–14, folate deficiency and ethanol each exerted independent effects on plasma Hcy levels (P < 0.001 for each effect). Liver folate levels were reduced by dietary folate deficiency alone, Hcy levels were increased by ethanol feeding alone, and methionine levels were reduced independently by both ethanol and folate deficiency (Table 1). The levels of folate and methionine were significantly less in group FDE than in group FSE. Ethanol feeding independently reduced hepatic MS activity, whereas BHMT activity was independently increased by folate deficiency. Linear regression analysis with all data points found a negative correlation between the activities of MS and BHMT (r = −0.445, P < 0.01).
Table 1.
Effects of diets on hepatic folate, homocysteine, methionine, MS, and BHMT
| Diet | Folate, μg/g | Homocysteine, nmol/mg protein | Methionine, nmol/mg protein | MS, pmol/mg/min | BHMT, units/mg/min |
|---|---|---|---|---|---|
| FS | 4.56 ± 1.23 | 0.77 ± 0.12 | 22.8 ± 2.54 | 8.23 ± 0.44 | 66.8 ± 7.32 |
| FD | 2.30 ± 0.49* | 0.93 ± 0.05 | 17.2 ± 2.23* | 8.88 ± 8.88 | 91.5 ± 6.02* |
| FSE | 6.83 ± 0.66 | 1.10 ± 0.05† | 13.8 ± 2.23† | 5.34 ± 0.53† | 66.6 ± 4.32 |
| FDE | 2.52 ± 0.37*‡ | 1.13 ± 0.05† | 7.4 ± .39*†‡ | 6.10 ± 0.31† | 75.6 ± 3.81* |
Mean ± SEM.
Folate effect in FD and FDE; P < 0.001, <0.01, <0.001.
Ethanol effect in FSE and FDE; P < 0.003, <0.001, <0.001.
Different from FSE, <0.01, <0.05.
Ethanol feeding independently reduced SAM levels, whereas folate deficiency and ethanol feeding each acted independently to increase levels of SAH, and the two variables interacted to produce a significant decrease in the SAM/SAH ratio (Table 2). The mean level of SAH was higher, and the SAM/SAH ratio was lower in group FDE than in group FSE.
Table 2.
Liver SAM, SAH, and their ratio
| SAM, pmol/mg protein | SAH, pmol/mg protein | SAM/SAH§ | |
|---|---|---|---|
| FS | 364 ± 30 | 136 ± 10 | 2.74 ± 0.21 |
| FD | 298 ± 10 | 243 ± 10* | 1.23 ± 0.04* |
| FSE | 261 ± 40† | 218 ± 20† | 1.18 ± 0.10† |
| FDE | 246 ± 50† | 291 ± 20*†‡ | 0.83 ± 0.11*†‡ |
Mean ± SEM.
Folate effect, P < 0.001, P < 0.001.
Ethanol effect, P < 0.03, P < 0.001, P < 0.001.
Different from FSE, P < 0.02, P < 0.04.
Interaction of folate deficiency and ethanol, P < 0.03.
Global DNA hypomethylation was increased independently by folate deficiency (Table 3). DNA oxidation as urinary oxo (8)dG was increased independently by ethanol feeding and folate deficiency and was greater in FDE than in FSE. DNA strand breaks were increased independently by ethanol feeding and folate deficiency and were increased in FDE compared with FSE.
Table 3.
DNA integrity and oxidation
| DNA hypomethylation, dpm/0.5 μg DNA | Urinary oxo(8)dG, pmol/mg creatinine | DNA strand breaks, dpm/0.25 μg DNA | |
|---|---|---|---|
| FS | 3,549.2 ± 470.7 | 7.3 ± 1.1 | 1,567 ± 41 |
| FD | 8,130.1 ± 2,947.2* | 9.1 ± 0.8* | 1,830 ± 98* |
| FSE | 5,100.3 ± 650.5 | 9.6 ± 1.2† | 1,791 ± 78† |
| FDE | 6,902.0 ± 713.3* | 14.3 ± 1.8*†‡ | 2,127 ± 48*†‡ |
Mean ± SEM.
Folate effect, P < 0.05, <0.03, <0.01.
Ethanol effect, P < 0.02, <0.02.
Different from FSE, P < 0.004, <0.05.
Ethanol feeding acted alone to reduce antioxidant GSH and reduce the GSH/GSSG ratio, an index of oxidative stress (Table 4). Ethanol feeding increased lipid peroxidation according to terminal plasma MDA and liver injury according to mean plasma alanine transaminase levels. Although plasma triglyceride levels were increased by ethanol feeding (P < 0.03, data not shown), the differences in MDA levels remained significant when accounting for triglyceride levels. Ethanol feeding increased plasma AST, whereas folate deficiency interacted with ethanol to further increase AST in group FDE to a mean level that was 8-fold greater than the control value.
Table 4.
Effects of diets on oxidative liver injury
| Diet | Liver GSH, nmol/mg protein | GSH/GSSG | Plasma MDA, pmol/ml | Plasma ALT, units/liter | Plasma AST‡, units/liter |
|---|---|---|---|---|---|
| FS | 46.03 ± 2.74 | 31.81 ± 6.40 | 20.7 ± 1.35 | 20.0 ± 1.2 | 42.0 ± 2.7 |
| FD | 42.92 ± 1.38 | 35.55 ± 3.98 | 21.9 ± 0.77 | 22.8 ± 2.8 | 47.7 ± 2.4 |
| FSE | 34.37 ± 1.21* | 19.60 ± 2.31* | 26.2 ± 1.84* | 41.6 ± 4.8* | 108.6 ± 22.2* |
| FDE | 29.58 ± 1.95* | 14.01 ± 2.28* | 25.3 ± 1.08*§ | 56.0 ± 6.0* | 350.8 ± 97.4*† |
Mean ± SEM.
Ethanol effect, P < 0.001, <0.001, <0.05, <0.01, <0.01.
Different from FSE, P < 0.04.
Interaction of folate and ethanol, P < 0.05.
Significance adjusted for plasma triglyceride.
Relationships among variables were assessed by linear regression and significance determined by ANOVA by using values from all micropigs. In keeping with an interaction between SAM and the level of oxidant stress, liver SAM was correlated positively with GSH (r = 0.479, P < 0.02), which in turn correlated negatively with plasma MDA (r = −0.609, P < 0.002) and urinary oxo (8)dG (r = −0.612, P < 0.005). DNA hypomethylation correlated with hepatic SAH (r = 0.418, P < 0.05) and urinary oxy (8)dG (r = 0.473, P < 0.03). DNA strand breaks correlated with urinary oxo (8)dG (r = 0.519, P < 0.02) and negatively with both SAM (r = −0.499, P < 0.01) and the SAM/SAH ratio (r = −0.660, P < 0.001).
Immunohistochemical staining of liver sections demonstrated enhanced centrilobular expression of the CYP2E1 microsomal ethanol oxidizing enzyme and protein adducts with AA and MDA in centrilobular regions surrounding the hepatic vein in ethanol-fed micropigs (not shown). The intensities of CYP2E1 and protein adducts with AA and MDA were increased equally by ethanol feeding in groups FSE and FDE but not by folate deficiency (Table 5).
Table 5.
Immunohistochemical scores of liver CYP2E1, AA, and MDA
| Diet | CYP2E1 | AA | MDA |
|---|---|---|---|
| FS | 0.88 ± 0.11 | 0.08 ± 0.05 | 0.33 ± 0.11 |
| FD | 0.80 ± 0.28 | 0.10 ± 0.09 | 0.20 ± 0.11 |
| FSE | 2.33 ± 0.41* | 0.75 ± 0.22* | 0.88 ± 0.09* |
| FDE | 1.67 ± 0.18* | 1.04 ± 0.23* | 1.13 ± 0.23* |
Mean ± SEM.
Ethanol effect, P < 0.001, <0.001, <0.001.
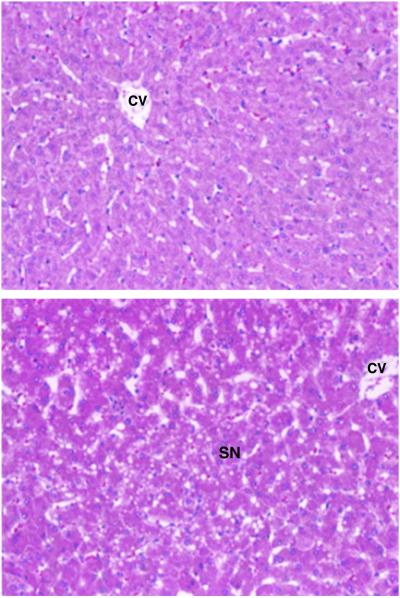
The effects of the different diets on terminal hepatic histology are illustrated in Fig. 1. Compared with unchanged histology in livers from pigs fed FS, FD and FSE diets, five of the six pigs fed the combined FDE diet demonstrated moderately severe to severe midlobular steatonecrosis. Minimal steatosis was noted in two of the six animals receiving the ethanol-containing (FSE) diet alone.
Figure 1.
Liver histology. (Upper) Representative normal histology as seen in FS, FD, and FSE. (Lower) Representative abnormal histopathology as seen in five of six pigs in FDE, demonstrating features of steatosis, necrosis, and inflammation. CV, central vein; SN, steatonecrosis with inflammation.
Discussion
Unlike other animal models of alcoholism that rely on oral liquid diets or intragastric tube feeding (41, 42), the micropig consumes ethanol voluntarily and completely in the diet while achieving daily levels of blood alcohol consistent with intoxication. Our initial study demonstrated that castrated male micropigs fed diets with or without ethanol at 40% of energy and an excess of folate, methionine, and choline for 12 mo developed all of the features of alcoholic liver injury, including intralobular steatonecrosis with inflammation and centrilobular collagen deposition (43). These findings were associated with the tissue accumulation of protein adducts of AA and MDA, poor weight gain, decreased body fat, but no changes in liver folate levels (43–45). However, there was no evidence of alcoholic liver injury in a second study when noncastrated male micropigs were fed the same diets for 12 mo (16), an observation that was associated with the modulation of oxidative liver injury by testosterone-dependent carbonic anhydrase III (46). Nevertheless, the study of noncastrated male micropigs demonstrated that ethanol feeding perturbed hepatic methionine metabolism and DNA integrity, including findings of a 3-fold increase in serum Hcy levels, reduced hepatic MS activity, increased SAH, and a corresponding decrease in the SAM/SAH ratio. The dUMP/dTMP ratio was elevated 3-fold in the ethanol-fed animals and correlated negatively with MS activity in pigs from both groups. This finding was associated with a 3-fold increase in hepatocellular apoptotic bodies in the ethanol-fed pigs (16).
The present experimental design permits conclusions on the separate and combined effects of folate deficiency and chronic ethanol exposure on hepatic methionine metabolism within the context of the development of alcoholic liver disease. By deleting folate from the diet, we achieved ≈50% decrease in liver folate levels in animals fed the FD and FDE diets with or without ethanol (Table 1). Most significantly, pigs fed the combined FDE diet demonstrated an 8-fold increase in plasma AST (Table 4), whereas typical histological features of steatonecrosis were found in five of six group FDE micropigs after 14 wk (Fig. 1). This finding contrasts with the absence of significant histopathology in the other groups and in our previous 12-mo study of noncastrated male micropigs fed folate sufficient diets with ethanol (16). As shown in Tables 1–5, the independent effects of ethanol included elevated levels of liver Hcy, CYP2E1, AA, MDA, and plasma alanine transaminase and reduced levels of liver MS, SAM, GSH, and the GSH/GSSG ratio. The independent effects of folate deficiency included reduced liver folate and increased BHMT and DNA hypomethylation. Both chronic ethanol exposure and folate deficiency acted independently in elevating plasma Hcy, liver SAH, urinary oxo(8)dG, and DNA strand breaks, and in reducing liver methionine. Folate deficiency and ethanol interacted in reducing the liver SAM/SAH ratio and in elevating plasma AST levels. The significance of the combined effects of folate deficiency and ethanol feeding was confirmed by finding significant differences in each of these variables in group FDE compared with group FSE.
Alcoholic liver injury represents a combination of hepatocellular apoptosis and necrosis that may evolve from an interplay of the CYP2E1-generated free hydroxyl radical (47, 48) and from tumor necrosis factor α-mediated enhancement of the mitochondrial and NADPH oxidase-dependent generation of reactive oxidant species that overwhelm antioxidative defenses (49–53). Oxidant-induced hepatocellular necrosis is the net result of enhanced generation of reactive oxidant species and depletion of antioxidants, in particular mitochondrial GSH (54). Disordered methionine metabolism is an established feature of experimental and clinical alcoholic liver disease and could contribute to its pathogenesis through effects on oxidant stress (28) and on DNA integrity (16, 26, 55).
In the context of these processes, the linear correlations found among the measured variables were consistent with known mechanistic relationships in the methionine cycle and provide insights into the role of altered methionine metabolism in the development of alcoholic liver injury. Thus, the observed reduction in GSH can be explained by ethanol-dependent reduction in SAM, which regulates the biosynthesis of the GSH precursor cysteine (20). The observed inverse correlation between SAH and DNA hypomethylation is consistent with the inhibitory role of SAH on SAM-regulated methylation processes (19). DNA hypomethylation may in turn be linked mechanistically to the observed increase in urinary oxo (8)dG and DNA strand breaks through increased accessibility of internucleosomal sites (56). Oxidative stress causes DNA damage by one or two interactive events: intracellular hydroxyl radical formation secondary to the reaction of H2O2 with a DNA-bound metal ion and/or oxidation-induced rise in cellular calcium transport that triggers Ca+2-dependent endonuclease activity and strand breakage (57, 58). At the same time, nucleotide imbalance secondary to abnormal methionine metabolism promotes hepatocellular apoptosis, as described in our prior study of ethanol-fed micropigs (16) and in rats fed folate- and methyl-deficient diets (15). Others described increased susceptibility of oxidized DNA to strand breakage, which may be considered a prerequisite for either cellular apoptosis or necrosis (59).
In addition to the observed folate- and ethanol-induced changes cited above, other mechanisms could account for the interaction of perturbed methionine metabolism in the development of alcoholic liver injury in our micropig model. For example, the observed ethanol-related decreases in both SAM and GSH (Tables 2 and 4) are consistent with the known effects of chronic exposure to ethanol on the reduction of liver GSH and its correction by SAM in the baboon (28). The present studies demonstrated an ethanol-induced inhibition of MS activity that could explain the reduced liver methionine and SAM levels (Tables 1 and 2). Others showed that nitric oxide inactivates MS (60) because of interaction with its cobalamin cofactor (61). MAT activity is reduced posttranslationally through nitrosylation of cysteine residue 121 in MATI/III (62), whereas its activity is maintained in part by GSH (63). Recent data indicate that the transsulfuration pathway is up-regulated by oxidative stress, which could modulate the decrease in GSH associated with decreased SAM (64). However, in addition to activating the transsulfuration pathway (20), SAM also plays an antioxidative role by enhancing mitochondrial membrane fluidity and GSH transport (29). The critical linkage between liver SAM and GSH is further strengthened by the observation of reduced GSH in MAT1A knockout mice, which are defective in SAM synthesis and develop hepatic steatosis while expressing many proinflammatory cytokines (65).
In summary, the present study demonstrates that the addition of folate deficiency to a diet containing excessive ethanol both enhances abnormal hepatic methionine metabolism and promotes the early development of alcoholic liver injury. Feeding the combined FDE diet resulted in the greatest increase in plasma Hcy and liver SAH levels and the greatest reduction in the SAM/SAH ratio (Table 2), together with an 8-fold increase in plasma AST (Table 4), and the unique finding of early onset steatohepatitis (Fig. 1). The promotion of early-onset liver injury with increased abnormalities in hepatic methionine metabolism by combining folate deficiency with ethanol feeding constitutes strong evidence for the mechanistic linkage of perturbed methionine metabolism and its associated DNA damage to the development of alcoholic liver disease. Furthermore, our studies imply that the risk of developing alcoholic liver disease is magnified by concomitant folate deficiency in consumers of excessive amounts of ethanol.
Supplementary Material
Acknowledgments
We are indebted to Carmen Castro for technical assistance in the BHMT assays. C.H.H. conceived and supervised all aspects of the study and wrote the manuscript; J.A.V. was responsible for conducting the animal protocols and surgeries, tissue folate preparations, assays of MS and DNA strand breaks, and compiling data and statistics; A.M.D. was responsible for DNA hypomethylation assays and statistical analyses; S.P. and O.N. for histopathology and immunochemical studies; T.A.G. for supervising BHMT assays; L.M.W. and M.K.S. for oxidation studies; and S.M. and S.J.J. for assays of methionine metabolites. All authors shared in the final data interpretation and analysis and concurred with the final manuscript. This work was supported by National Institutes of Health Grants DK45301 and DK35747 to C.H.H. and DK52501 to T.A.G., and by a grant from the Finnish Foundation for Alcohol Studies to O.N.
Abbreviations
- FD
folate depleted
- FDE
FD/ethanol
- FS
folate sufficient
- FSE
FS/ethanol
- AA
acetaldehyde
- AST
aspartate transaminase
- BHMT
betaine homocysteine methyltransferase
- GSH
glutathione
- GSSG
oxidized form of GSH
- Hcy
homocysteine
- MAT
methionine adenosyl transferase
- MDA
malondialdehyde
- MS
methionine synthase
- SAM
S-adenosylmethionine
- SAH
S-adenosylhomocysteine
- oxo(8)dG
8-hydroxy-2′-deoxyguanosine
- 5-MTHF
5-methyltetrahydrofolate
References
- 1.Cravo M L, Gloria L M, Selhub J, Nadeau M R, Camilo M E, Resende M P, Cardoso J N, Leitao C N, Mira F C. Am J Clin Nutr. 1996;63:220–224. doi: 10.1093/ajcn/63.2.220. [DOI] [PubMed] [Google Scholar]
- 2.Herbert V, Zalusky R, Davidson C S. Ann Intern Med. 1963;58:977–988. doi: 10.7326/0003-4819-58-6-977. [DOI] [PubMed] [Google Scholar]
- 3.Eichner E R, Hillman R S. Am J Med. 1971;50:218–232. doi: 10.1016/0002-9343(71)90151-3. [DOI] [PubMed] [Google Scholar]
- 4.Wu A, Chanarin I, Slavin G, Levi A J. Br J Haematol. 1975;29:469–478. doi: 10.1111/j.1365-2141.1975.tb01844.x. [DOI] [PubMed] [Google Scholar]
- 5.Leevy C M, Cardi L, Frank O, Gellene R, Baker H. Am J Clin Nutr. 1965;17:259–271. doi: 10.1093/ajcn/17.4.259. [DOI] [PubMed] [Google Scholar]
- 6.Halsted C H, Robles E A, Mezey E. N Engl J Med. 1971;285:701–706. doi: 10.1056/NEJM197109232851301. [DOI] [PubMed] [Google Scholar]
- 7.Romero J J, Tamura T, Halsted C H. Gastroenterology. 1981;80:99–102. [PubMed] [Google Scholar]
- 8.Tamura T, Romero J J, Watson J E, Gong E J, Halsted C H. J Lab Clin Med. 1981;97:654–661. [PubMed] [Google Scholar]
- 9.Tamura T, Halsted C H. J Lab Clin Med. 1983;101:623–628. [PubMed] [Google Scholar]
- 10.McMartin K E, Collins T D, Eisenga B H, Fortney T, Bates W R, Bairnsfather L. J Nutr. 1989;119:1490–1497. doi: 10.1093/jn/119.10.1490. [DOI] [PubMed] [Google Scholar]
- 11.Russell R M, Rosenberg I H, Wilson P D, Iber F L, Oaks E B, Giovetti A C, Otradovec C L, Karwoski P A, Press A W. Am J Clin Nutr. 1983;38:64–70. doi: 10.1093/ajcn/38.1.64. [DOI] [PubMed] [Google Scholar]
- 12.Shaw S, Jayatilleke E, Herbert V, Colman N. Biochem J. 1989;257:277–280. doi: 10.1042/bj2570277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Henning S M, McKee R W, Swendseid M E. J Nutr. 1989;119:1478–1482. doi: 10.1093/jn/119.10.1478. [DOI] [PubMed] [Google Scholar]
- 14.Miller J W, Nadeau M R, Smith J, Smith D, Selhub J. Biochem J. 1994;298:415–419. doi: 10.1042/bj2980415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.James S J, Miller B J, Basnakian A G, Pogribny I P, Pogribna M, Muskhelishvili L. Carcinogenesis. 1997;18:287–293. doi: 10.1093/carcin/18.2.287. [DOI] [PubMed] [Google Scholar]
- 16.Halsted C H, Villanueva J, Chandler C J, Stabler S P, Allen R H, Muskhelishvili L, James S J, Poirier L. Hepatology. 1996;23:497–505. doi: 10.1002/hep.510230314. [DOI] [PubMed] [Google Scholar]
- 17.Mudd S H, Poole J R. Metabolism. 1975;24:721–735. doi: 10.1016/0026-0495(75)90040-2. [DOI] [PubMed] [Google Scholar]
- 18.Mato J M, A L, Corrales F J, Pajares M A. In: The Liver: Biology and Pathobiology. Arias I M, Boyer J L, Fausto N, Jakoby W B, Schachter D A, Shafritz D A, editors. New York: Raven; 1994. pp. 461–470. [Google Scholar]
- 19.Yi P, Melnyk S, Pogribna M, Pogribny I P, Hine R J, James S J. J Biol Chem. 2000;275:29318–29323. doi: 10.1074/jbc.M002725200. [DOI] [PubMed] [Google Scholar]
- 20.Finkelstein J D, Kyle W E, Martin J L, Pick A M. Biochem Biophys Res Commun. 1975;66:81–87. doi: 10.1016/s0006-291x(75)80297-x. [DOI] [PubMed] [Google Scholar]
- 21.Pajares M A, Duran C, Corrales F, Pliego M M, Mato J M. J Biol Chem. 1992;267:17598–605. [PubMed] [Google Scholar]
- 22.Shane B. In: Folic Acid Metabolism in Health and Disease. Picciano M, Stokstad E L R, Gregory J F, editors. Vol. 13. New York: Wiley–Liss; 1990. pp. 65–68. [Google Scholar]
- 23.Barak A J, Beckenhauer H C, Tuma D J, Badakhsh S. Biochem Cell Biol. 1987;65:230–233. doi: 10.1139/o87-029. [DOI] [PubMed] [Google Scholar]
- 24.Finkelstein J D, Cello J P, Kyle W E. Biochem Biophys Res Commun. 1974;61:525–531. doi: 10.1016/0006-291x(74)90988-7. [DOI] [PubMed] [Google Scholar]
- 25.Trimble K C, Molloy A M, Scott J M, Weir D G. Hepatology. 1993;18:984–989. doi: 10.1002/hep.1840180433. [DOI] [PubMed] [Google Scholar]
- 26.Lu S C, Huang Z Z, Yang H, Mato J M, Avila M A, Tsukamoto H. Am J Physiol Gastrointest Liver Physiol. 2000;279:G178–G185. doi: 10.1152/ajpgi.2000.279.1.G178. [DOI] [PubMed] [Google Scholar]
- 27.Garcia-Ruiz C, Morales A, Colell A, Ballesta A, Rodes J, Kaplowitz N, Fernandez-Checa J C. Hepatology. 1995;21:207–214. doi: 10.1002/hep.1840210133. [DOI] [PubMed] [Google Scholar]
- 28.Lieber C S, Casini A, DeCarli L M, Kim C I, Lowe N, Sasaki R, Leo M A. Hepatology. 1990;11:165–172. doi: 10.1002/hep.1840110203. [DOI] [PubMed] [Google Scholar]
- 29.Colell A, García-Ruiz C, Morales A, Ballesta A, Ookhtens M, Rodés J, Kaplowitz N, Fernández-Checa J C. Hepatology. 1997;26:699–708. doi: 10.1002/hep.510260323. [DOI] [PubMed] [Google Scholar]
- 30.Committee on Animal Nutrition, National Research Council. Nutrient Requirements of Swine. Washington, DC: Natl. Acad. Press; 1998. , 10th Rev. Ed., pp. 71–89. [Google Scholar]
- 31.Tamura T. In: Folic Acid Metabolism in Health and Disease. Picciano M, Stokstad E L R, Gregory J F III, editors. Vol. 13. New York: Wiley–Liss; 1990. pp. 121–138. [Google Scholar]
- 32.Melnyk S, Pogribna M, Pogribny I P, Yi P, James S J. Clin Chem. 2000;46:265–272. [PubMed] [Google Scholar]
- 33.Kondo H, Osborne M L, Kolhouse J F, Binder M J, Podell E R, Utley C S, Abrams R S, Allen R H. J Clin Invest. 1981;67:1270–1283. doi: 10.1172/JCI110155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Garrow T A. J Biol Chem. 1996;271:22831–22838. doi: 10.1074/jbc.271.37.22831. [DOI] [PubMed] [Google Scholar]
- 35.Ravanat J L, Duretz B, Guiller A, Douki T, Cadet J. J Chromatogr B Biomed Sci Appl. 1998;715:349–356. doi: 10.1016/s0378-4347(98)00259-x. [DOI] [PubMed] [Google Scholar]
- 36.Helbock H J, Beckman K B, Shigenaga M K, Walter P B, Woodall A A, Yeo H C, Ames B N. Proc Natl Acad Sci USA. 1998;95:288–293. doi: 10.1073/pnas.95.1.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yeo H C, Liu J, Helbock H J, Ames B N. Methods Enzymol. 1999;300:70–78. doi: 10.1016/s0076-6879(99)00115-9. [DOI] [PubMed] [Google Scholar]
- 38.Pogribny I, Yi P, James S J. Biochem Biophys Res Commun. 1999;262:624–628. doi: 10.1006/bbrc.1999.1187. [DOI] [PubMed] [Google Scholar]
- 39.Basnakian A G, James S J. DNA Cell Biol. 1996;15:255–262. doi: 10.1089/dna.1996.15.255. [DOI] [PubMed] [Google Scholar]
- 40.Niemelä O, Parkkila S, Pasanen M, Viitala K, Villanueva J A, Halsted C H. Hepatology. 1999;30:1011–1017. doi: 10.1002/hep.510300413. [DOI] [PubMed] [Google Scholar]
- 41.Lieber C S. Gastroenterology. 1994;106:1085–1105. doi: 10.1016/0016-5085(94)90772-2. [DOI] [PubMed] [Google Scholar]
- 42.Tsukamoto H, Gaal K, French S W. Hepatology. 1990;12:599–608. doi: 10.1002/hep.1840120325. [DOI] [PubMed] [Google Scholar]
- 43.Halsted C H, Villanueva J, Chandler C J, Ruebner B, Munn R J, Parkkila S, Niemelä O. Hepatology. 1993;18:954–960. doi: 10.1002/hep.1840180429. [DOI] [PubMed] [Google Scholar]
- 44.Nakamura M T, Tang A B, Villanueva J, Halsted C H, Phinney S D. Metabolism. 1993;42:1340–1350. doi: 10.1016/0026-0495(93)90136-c. [DOI] [PubMed] [Google Scholar]
- 45.Niemelä O, Parkkila S, Yla-Herttuala S, Villanueva J, Ruebner B, Halsted C H. Hepatology. 1995;22:1208–1214. [PubMed] [Google Scholar]
- 46.Parkkila S, Halsted C H, Villanueva J A, Vaananen H K, Niemelä O. Dig Dis Sci. 1999;44:2205–2213. doi: 10.1023/a:1026640317233. [DOI] [PubMed] [Google Scholar]
- 47.Albano E, Clot P, Morimoto M, Tomasi A, Ingelman-Sundberg M, French S W. Hepatology. 1996;23:155–163. doi: 10.1002/hep.510230121. [DOI] [PubMed] [Google Scholar]
- 48.Román J, Colell A, Blasco C, Caballeria J, Parés A, Rodés J, Fernández-Checa J C. Hepatology. 1999;30:1473–1480. doi: 10.1002/hep.510300623. [DOI] [PubMed] [Google Scholar]
- 49.Tilg H, Diehl A M. N Engl J Med. 2000;343:1467–1476. doi: 10.1056/NEJM200011163432007. [DOI] [PubMed] [Google Scholar]
- 50.Tsukamoto H, Lu S C. FASEB J. 2001;15:1335–1349. doi: 10.1096/fj.00-0650rev. [DOI] [PubMed] [Google Scholar]
- 51.Garcia-Ruiz C, Morales A, Ballesta A, Rodes J, Kaplowitz N, Fernandez-Checa J C. J Clin Invest. 1994;94:193–201. doi: 10.1172/JCI117306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hannun Y A. Science. 1996;274:1855–1859. doi: 10.1126/science.274.5294.1855. [DOI] [PubMed] [Google Scholar]
- 53.Wheeler M D, Kono H, Yin M, Nakagami M, Uesugi T, Arteel G E, Gabele E, Rusyn I, Yamashina S, Froh M, et al. Free Radical Biol Med. 2001;31:1544–1549. doi: 10.1016/s0891-5849(01)00748-1. [DOI] [PubMed] [Google Scholar]
- 54.Fernandez-Checa J C, Garcia-Ruiz C, Ookhtens M, Kaplowitz N. J Clin Invest. 1991;87:397–405. doi: 10.1172/JCI115010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chawla R K, Watson W H, Eastin C E, Lee E Y, Schmidt J, McClain C J. Am J Physiol. 1998;275:G125–G129. doi: 10.1152/ajpgi.1998.275.1.G125. [DOI] [PubMed] [Google Scholar]
- 56.Pogribny I P, Basnakian A G, Miller B J, Lopatina N G, Poirier L A, James S J. Cancer Res. 1995;55:1894–1901. [PubMed] [Google Scholar]
- 57.Bertoncini C R, Meneghini R. Nucleic Acids Res. 1995;23:2995–3002. doi: 10.1093/nar/23.15.2995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Halliwell B, Aruoma O I. FEBS Lett. 1991;281:9–19. doi: 10.1016/0014-5793(91)80347-6. [DOI] [PubMed] [Google Scholar]
- 59.Cantoni O, Sestili P, Guidarelli A, Palomba L, Brambilla L, Cattabeni F. Arch Toxicol Suppl. 1996;18:223–235. doi: 10.1007/978-3-642-61105-6_23. [DOI] [PubMed] [Google Scholar]
- 60.Nicolaou A, Kenyon S H, Gibbons J M, Ast T, Gibbons W A. Eur J Clin Invest. 1996;26:167–170. doi: 10.1046/j.1365-2362.1996.122254.x. [DOI] [PubMed] [Google Scholar]
- 61.Danishpajooh I O, Gudi T, Chen Y, Kharitonov V G, Sharma V S, Boss G R. J Biol Chem. 2001;276:27296–27303. doi: 10.1074/jbc.M104043200. [DOI] [PubMed] [Google Scholar]
- 62.Avila M A, Mingorance J, Martinez-Chantar M L, Casado M, Martin-Sanz P, Bosca L, Mato J M. Hepatology. 1997;25:391–396. doi: 10.1002/hep.510250222. [DOI] [PubMed] [Google Scholar]
- 63.Corrales F, Ochoa P, Rivas C, Martin-Lomas M, Mato J M, Pajares M A. Hepatology. 1991;14:528–533. [PubMed] [Google Scholar]
- 64.Mosharov E, Cranford M R, Banerjee R. Biochemistry. 2000;39:13005–13011. doi: 10.1021/bi001088w. [DOI] [PubMed] [Google Scholar]
- 65.Lu S C, Alvarez L, Huang Z Z, Chen L, An W, Corrales F J, Avila M A, Kanel G, Mato J M. Proc Natl Acad Sci USA. 2001;98:5560–5565. doi: 10.1073/pnas.091016398. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.