Abstract
Examination of the blaCTX-M-2 gene in plasmid pMAR-12 by sequencing and PCR analysis revealed that the bla gene and the surrounding DNA, which is closely related (99% homology) to the Kluyvera ascorbata chromosomal DNA that contains the blaKLUA-1 gene, are located in a complex sul1-type integron, termed In35, that includes Orf513. It is possible that blaCTX-M-2 was acquired by plasmid pMAR-12 through an uncharacterized recombinational event in which Orf513 could be involved.
Many plasmid-mediated β-lactamase genes are located within or near mobile elements, such as integrons or transposons, which enhance their rapid dissemination (1, 9, 14, 16). Also, unusual class 1 integrons containing a partial duplication of the 3′ conserved segment (3′-CS) have been described as carrying antibiotic resistance genes such as dfrA10, cat, and blaDHA-1 (15, 18, 21). Between the two 3′-CSs there is a region which includes Orf513 (previously known as Orf341) (18) GenBank accession number L06418). In Argentina, CTX-M-2 (3) is the extended-spectrum β-lactamase most frequently found in gram-negative clinical isolates (69% of all extended-spectrum β-lactamases found) (M. Galas, M. Rapoport, F. Pasteran, R. Melano, A. Petroni, P. Ceriana, A. Rossi, and the WHONET Group, Abstr. 39th Intersci. Conf. Antimicrob. Agents Chemother., abstr. 1474, 1999). blaCTX-M-2 is 99% similar to blaKLUA-1, the chromosomal class A β-lactamase from Kluyvera ascorbata (13; GenBank accession number AJ272538), but how this gene was acquired by plasmids has not been established. The goal of this work was to describe the genetic organization around blaCTX-M-2.
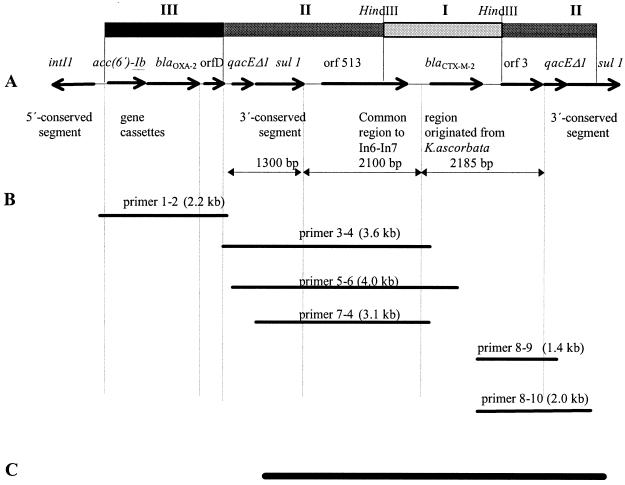
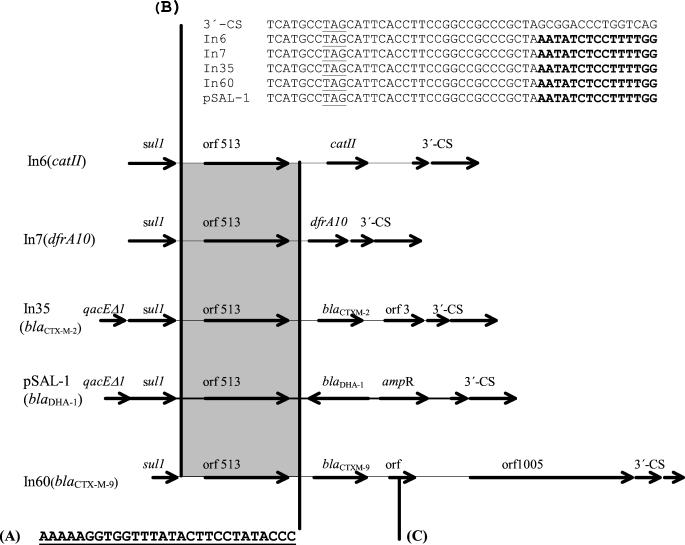
A Proteus mirabilis strain (Pm12) isolated in 1993 from the urine of a patient hospitalized in Buenos Aires, Argentina, had the following susceptibility pattern (MICs, in micrograms per milliliter): ampicillin, >512; amikacin, 0.5; aztreonam, 0.25; cefoperazone, 64; cefoperazone-sulbactam, 8; cefotaxime, >32; cefotaxime-clavulanic acid, 0.03; cefotaxime-sulbactam, 4; ceftazidime, 0.5; ciprofloxacin, 0.5; imipenem, 0.25; gentamicin, 64, piperacillin 256; and piperacillin-tazobactam, 0.5. Mating experiments with Escherichia coli J53 Azir (10) as the recipient confirmed that the cefotaxime resistance genes in Pm12 were located on a conjugative plasmid, pMAR-12 (>150 kb). Plasmid pMAR-12 DNA was digested with HindIII and ligated into pBC SK (Stratagene, La Jolla, Calif.). Recombinant plasmid pMAR-121 was introduced by electroporation into E. coli XL1-Blue (Stratagene), and transformants were selected on Luria-Bertani agar with cefotaxime at 8 μg/ml (17). The 2,100-bp insert of pMAR-121 was sequenced in its entirety with an ABI377 sequencer (Applied Biosystems, Foster City, Calif.). The nucleotide sequence was analyzed using Genetics Computer Group programs. To assess the genetic environment of the blaCTX-M-2 gene (3), PCR analysis (12) of the sequences flanking blaCTX-M-2 was carried out by using plasmid pMAR-12 DNA and the PCR primers listed in Table 1. The sequences of four overlapping PCR fragments upstream and two fragments downstream from the blaCTX-M-2 gene yielded a continuous sequence of more than 5.6 kb around blaCTX-M-2 (Fig. 1). Upstream of blaCTX-M-2, a segment of 266 bp with 96% identity to the chromosomal DNA of K. ascorbata is preceded by a segment of 2,100 bp which is identical to the common region of the integrons In6 and In7 and which includes Orf513 (18). The common region in In35 as well as in the Orf513-associated gene for blaDHA-1 on plasmid pSAL-1 (21) ends with a 28-bp segment that is duplicated in In6 (Fig. 2A) (18). This 28-bp segment at the end of the common region has been proposed as a recognition site for the Orf513 putative transposase (20). The common region begins at the end of the first partial 3′-CS, which lies 24 bp after the stop codon of the sul1 gene, as was described for plasmid pDGO100 (Fig. 2B) (6). Downstream of blaCTX-M-2 is a segment of 1,043 bp with 99% identity with DNA downstream of blaKLUA-1 in K. ascorbata that ends 28 bp before the stop codon of Orf3 (of unknown function) and that is followed by a second copy of the 3′-CS of a typical class 1 integron.
TABLE 1.
Oligonucleotides used for PCR mapping
| Primer | Primer sequence (5′ to 3′) | Region | Accession no. |
|---|---|---|---|
| 1 | GGC ATC CAA GCA GCA AG | 5′-CS | M73819 |
| 2 | AAG CAG ACT TGA CCT GA | 3′-CS | M73819 |
| 3 | CAT TCT GCG GTC GGC TT | OrfD gene | X72585 |
| 4 | AAT AGC AGG GGT AGC GT | blaCTX-M-2 gene | X92507 |
| 5 | GCG AAG TAA TCG CAA CAT CC | 3′-CS | U49101 |
| 6 | TCA CTT TAT CGG GAC CAC | blaCTX-M-2 gene | X92507 |
| 7 | GAC GGT GTT CGG CAT TCT | 3′-CS | U49101 |
| 8 | CGT CGG GAT ATT CTG GC | blaCTX-M-2 gene | X92507 |
| 9 | AGC CCC ATA CCT ACA AAG CC | 3′-CS | U49101 |
| 10 | TTT GAA GGT TCG ACA GC | 3′-CS | U49101 |
| 11 | AAA CAC GCC AGG CAT TC | aac(6′)-Ib gene | AF231133 |
| 12 | CGC AGA TCA GTT GGA AG | aadA gene | AF326210 |
| 13 | CCG CAG CTA GAA TTT TG | aadB gene | X04555 |
FIG. 1.
Structure of the integron of pMAR-12. (A) In35 containing the blaCTX-M-2 gene obtained by sequencing pMAR-121 (2,100 bp) (region I) and PCR mapping of the surrounding regions (regions II and III); (B) lengths of PCR products; (C) sequence reported here (GenBank accession number AY079169).
FIG. 2.
Comparison of the genetic structures of In6 (catII), In7(dfrA10), In35 (blaCTX-M-2), pSAL-1(blaDHA-1), and In60 (blaCTX-M-9). Sequences indicated by shaded boxes are identical. (A) A 28-bp segment at the end of the common region; (B) beginning of the common region of In6 and In7 (nucleotides in bold) that lies 24 bp beyond the stop codon of the sul1 gene (underlined) in the first 3′-CS; (C) open reading frame (orf) that is 71.6% identical to Orf3 from K. ascorbata (accession number AJ272538).
Deletions at the 5′ end of the second 3′-CS have been described previously (15, 20, 21), and they differ in length: 248 bp in In6, 76 bp in In7 (19), 179 bp in pSAL-1, and 70 bp in pMAR-12. Moreover, in In60 carrying blaCTX-M-9 (GenBank accession number AF373104), no deletion at this point has been found (Fig. 3). Although there are differences among the described genetic structures that include the common region, it is clear that in all cases there is a direct repeat of part of the 3′-CS of class 1 integrons.
FIG. 3.
Deletions at the 5′ end of the second 3′-CS in In60 (blaCTX-M-9), In35 (blaCTX-M-2), In7 (dfrA10), pSAL-1 (blaDHA-1), and In6 (catII). Base 1 corresponds to the first base of the 3′-CS.
The dissemination of blaCTX-M-2 that is potentially mediated by Orf513 has been analyzed by PCR using primers 3, 4, 11, 12, and 13 (Table 1) in 12 P. mirabilis strains, 7 Klebsiella pneumoniae strains, 1 Enterobacter cloacae strain, 1 Serratia marcescens strain, 1 Salmonella infantis strain, and 1 Acinetobacter baumannii strain which harbor blaCTX-M-2 and were obtained from patients with nosocomial infections in four hospitals in Buenos Aires, Argentina. In all isolates, blaCTX-M-2 was associated with Orf513 (Fig. 1A, regions I and II). However, in some isolates, the variable regions of class 1 integrons (Fig. 1A, region III) were different and already-described cassettes such as aac(6′)-Ib-aadA1, orfD, aadB-aadA1, aadA1, and aac(6′)Ib were found with blaCTX-M-2 (7, 12).
β-Lactamases of the CTX-M family can be divided into groups by amino acid sequence similarities (4; http://www.lahey.org/studies/webt.htm). The CTX-M-2 type of β-lactamase (CTX-M-2, CTX-M-4, CTX-M-5, CTX-M-6, CTX-M-7, and Toho-1) derives from KLUA-1 of K. ascorbata, with which it is at least 97% similar, while the Toho-2 type (Toho-2, CTX-M-9, and its derivative, CTX-M-16), the CTX-M-1 type (CTX-M-1, -3, -10, -11, and -12), and CTX-M-8 have only 77.7 to 83.8% identity with CTX-M-2 and seem to have evolved from another ancestor. Nonetheless, the genes for CTX-M-2 and CTX-M-9, which are only 80% identical, are both located in integrons which include Orf513. Furthermore, downstream of blaCTX-M-9 (GenBank accession number AF373104), there is an open reading frame that is 73% to Orf3, which is found in a similar position in In35 (Fig. 2C).
Among plasmid-carried β-lactamase genes that have been described in similar structures, some encode AmpC-type enzymes (DHA-1, MOX-1, and CMY-1) (2, 8, 21) which belong to the group 1 described by Bush et al. (5). Of these structures, only the blaDHA-1 flanking sequences in pSAL-1 have been sequenced entirely. The common feature of DHA-1 and CTX-M-2 is a chromosomal DNA ancestor that has been identified in both cases. Orf513 has been proposed to encode a site-specific recombinase (20), and it may have been able to capture genes from the chromosome and insert them into a complex sul1 type integron, thus enabling them to disseminate using conjugative plasmids.
On the other hand, a putative insert sequence, ISEcp1, has recently been found located upstream from CTX-M-15, an enzyme closely related to CTX-M-3 (11). These findings suggest that more than one transposable element may be associated with different members of the CTX-M family.
By PCR analysis, integrons of class 1 have been found in 100% of 67 strains which are resistant to expanded-spectrum cephalosporins and aminoglycosides and which were isolated from patients with nosocomial infections in four hospitals in Buenos Aires, Argentina, between 1993 and 2000 (B. Orman, unpublished data). blaCTX-M-2 always has been found at the same sequence position in the unusual class 1 integrons (Fig. 1A, regions I and II), with different arrays of cassettes in the variable region (Fig. 1A, region III) in the studied strains. The dissemination of plasmids carrying these structures may explain the widespread distribution of CTX-M-2 in our bacterial population.
Nucleotide sequence accession number.
The sequence of In35 has been submitted to GenBank under accession number AY079169.
Acknowledgments
This study was financed in part by grant PEI-98 from CONICET awarded to D.C. The work in G.A.J.'s laboratory was supported in part by a Merit Review award from the VA/DoD Collaborative Research Program on Mechanisms of Emerging Pathogens. Work in P.H.R.'s laboratory was supported by Canadian Institutes of Health Research grant MT-13564.
REFERENCES
- 1.Aubert, D., L. Poirel, J. Chevalier, S. Leotard, J.-M. Pages, and P. Nordmann. 2001. Oxacillinase-mediated resistance to cefepime and susceptibility to ceftazidime in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 45:1615-1620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bauernfeind, A., I. Stemplinger, R. Jungwirth, R. Wilhelm, and Y. Chong. 1996. Comparative characterization of the cephamycinase blaCMY-1 gene and its relationship with other β-lactamase genes. Antimicrob. Agents Chemother. 40:1926-1930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bauernfeind, A., T. Stemplinger, R. Jungwirth, S. Ernst, and J. M. Casellas. 1996. Sequences of β-lactamase genes encoding CTX-M-1 (MEN-1) and CTX-M-2 and relationship of their amino acid sequences with those of other β-lactamases. Antimicrob. Agents Chemother. 40:509-513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bonnet, R., C. Dutour, J. L. M. Sampaio, C. Chanal, D. Sirot, R. Labia, C. De Champs, and J. Sirot. 2001. Novel cefotaximase (CTX-M-16) with increased catalytic efficiency due to substitution Asp-240→Gly. Antimicrob. Agents Chemother. 45:2269-2275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bush, K., G. A. Jacoby, and A. Medeiros. 1995. A functional classification scheme for β-lactamases and its correlation with molecular structure. Antimicrob Agents Chemother. 39:1211-1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hall, R. M., and H. W. Stokes. 1990. The structure of a partial duplication in the integron of plasmid pDGO100. Plasmid 23:76-79. [DOI] [PubMed] [Google Scholar]
- 7.Hall, R. M., D. E. Brookes, and H. W. Stokes. 1991. Site-specific insertion of genes into integrons: role of the 59-base element and determination of the recombination cross-over point. Mol. Microbiol. 5:1941-1959. [DOI] [PubMed] [Google Scholar]
- 8.Horii, T., Y. Arakawa, M. Ohta, T. Sugiyama, R. Wacharotayankun, I. Ito, and N. Kato. 1994. Characterization of a plasmid borne and constitutively expressed blaMOX-1 gene encoding AmpC-type β-lactamase. Gene 139:93-98. [DOI] [PubMed] [Google Scholar]
- 9.Iyobe, S., H. Kusadokoro, J. Ozaki, N. Matsumura, S. Minami, S. Haruta, T. Sawai, and K. O'Hara. 2000. Amino acid substitutions in a variant of IMP-1 metallo-β-lactamase. Antimicrob. Agents Chemother. 44:2023-2027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jacoby, G. A., and P. Han. 1996. Detection of extended-spectrum β-lactamases in clinical isolates of Klebsiella pneumoniae and Escherichia coli. J. Clin. Microbiol. 34:908-911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karim, A., L. Poirel, S. Nagarajan, and P. Nordmann. 2001. Plasmid-mediated extended-spectrum beta-lactamase (CTX-M-3-like) from India and gene association with insertion sequence ISEcp1. FEMS Microbiol. Lett. 201:237-241. [DOI] [PubMed] [Google Scholar]
- 12.Lévesque, C., L. Piché, C. Larose, and P. H. Roy. 1995. PCR mapping of integrons reveals several novel combinations of resistance genes. Antimicrob. Agents Chemother. 39:185-191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Oliver, A., J. C. Perez-Diaz, T. M. Coque, F. Baquero, and R. Cantón. 2001. Nucleotide sequence and characterization of a novel cefotaxime-hydrolyzing β-lactamase (CTX-M-10) isolated in Spain. Antimicrob. Agents Chemother. 45:616-620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pallecchi, L., M. L. Riccio, J. D. Docquier, R. Fontana, and G. M. Rossolini. 2001. Molecular heterogeneity of blaVIM-2-containing integrons from Pseudomonas aeruginosa plasmids encoding the VIM-2 metallo-β-lactamase. FEMS Microbiol. Lett. 195:145-150. [DOI] [PubMed] [Google Scholar]
- 15.Parsons, Y., R. M. Hall, and H. W. Stokes. 1991. A new trimethoprim resistance gene, dhfrX, in the In7 integron of plasmid pDGO100. Antimicrob. Agents Chemother. 35:2436-2439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Poirel, L., G. F. Weldhagen, T. Naas, C. De Champs, M. G. Dove, and P. Nordmann. 2001. GES-2, a class A β-lactamase from Pseudomonas aeruginosa with increased hydrolysis of imipenem. Antimicrob. Agents Chemother. 45:2598-2603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
- 18.Stokes, H. W., C. Tomaras, Y. Parsons, and R. M. Hall. 1993. The partial 3′-conserved segment duplications in the integron In6 from pSa and In7 from pDGO100 have a common origin. Plasmid 30:39-50. [DOI] [PubMed] [Google Scholar]
- 19.Stokes, H. W., D. B. O'Gorman, G. D. Recchia, M. Parsekhian, and R. M. Hall. 1997. Structure and function of 59-base element recombination sites associated with mobile gene cassettes. Mol. Microbiol. 26:731-745. [DOI] [PubMed] [Google Scholar]
- 20.Valentine, C. R., M. J. Heinrich, S. L. Chissoe, and B. A. Roe. 1994. DNA sequence of direct repeats of the sul1 gene of plasmid pSa. Plasmid 32:222-227. [DOI] [PubMed] [Google Scholar]
- 21.Verdet, C., G. Arlet, G. Barnaud, P. H. Lagrange, and A. Philippon. 2000. A novel integron in Salmonella enterica serovar Enteritidis, carrying the blaDHA-1 gene and its regulator gene ampR, originated from Morganella morganii. Antimicrob. Agents Chemother. 44:222-225. [DOI] [PMC free article] [PubMed] [Google Scholar]