Abstract
A ciprofloxacin-resistant, nalidixic acid-susceptible mutant of Staphylococcus aureus (F145) contained no mutations within gyrA, gyrB, grlA, and grlB or within norA or its promoter region. MICs and accumulation studies suggest the role of a novel multidrug efflux pump.
The primary target of several fluoroquinolones in Staphylococcus aureus is thought to be topoisomerase IV (encoded by the grlA and grlB genes), whereas DNA gyrase (encoded by the gyrA and gyrB genes) is regarded as the secondary target (1, 9, 13). Quinolone-resistant S. aureus which possesses mutations in one or more of the topoisomerase genes, grlA, grlB, gyrA, or gyrB, and/or which has an enhanced efflux of fluoroquinolones due to the overexpression of norA has been described (3). Other than NorA, no other efflux pumps mediating fluoroquinolone resistance have been characterized in S. aureus, although Kaatz et al. (4) provided evidence for the existence of another multidrug efflux transporter from S. aureus; however, the gene(s) and protein(s) involved were not identified. Clinical isolates have also been reported where quinolone resistance appeared to be due to efflux, but no mutations in norA or its promoter were found (8).
(This work was presented in part at the 39th Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, Calif., 1999.)
During a study to select fluoroquinolone-resistant S. aureus (10), a novel mutant, F145, was selected from S. aureus NCTC 8532 (F77) on agar containing 1.25 μg of ciprofloxacin/ml in a single step (mutation frequency, 10−9). The MICs of 21 antibiotics were determined for both strains by the agar-doubling dilution method as previously described (10). The MIC of tetracycline was also determined with decimal dilutions (using increments of 0.1 μg/ml) in order to define the MIC more accurately. F145 showed decreased susceptibility to 11 fluoroquinolones, including ciprofloxacin, but an increased susceptibility to nalidixic acid and tetracycline compared to F77 (Table 1). The MICs of enoxacin, enrofloxacin, lomefloxacin, ofloxacin, pefloxacin, rufloxacin, sparfloxacin, temafloxacin, and tosufloxacin were all increased two- to fourfold for F145 (data not shown). F145 was also more susceptible than F77 to certain dyes and detergents, including acridine orange, ethidium bromide, cetrimide, sodium dodecyl sulfate, Triton X-114, NP-40, doxorubicin, and tetraphenylphosphonium (Table 1). The MICs of novobiocin, β-lactams, chloramphenicol, erythromycin, gentamicin, polymyxin, trimethoprim, and rifampin were unchanged in the mutant (data not shown). The effects of reserpine and carbonyl cyanide m-chlorophenylhydrazone (CCCP), inhibitors of the NorA efflux system, on the MICs of a selected number of antibiotics were determined. In the presence of reserpine (20 μg/ml) or CCCP (100 μM), the MICs of nalidixic acid for F77 and F145 were reduced to 0.25 μg/ml. For F77, neither agent reduced the MIC of ciprofloxacin, whereas for F145, CCCP reduced the MIC to 0.25 μg/ml, the same as for F77.
TABLE 1.
MICs of 15 quinolones and other antibiotics, dyes, detergents, and disinfectants for S. aureus F77 and F145
| Agent(s)a | MICs (μg/ml) of S. aureus strains
|
|
|---|---|---|
| F77 | F145 | |
| Antibiotics | ||
| Nalidixic acid | 128 | 16 |
| Nalidixic acid + reserpine | 0.25 | 0.25 |
| Nalidixic acid + CCCP | 0.25 | 0.25 |
| Ciprofloxacin | 0.25 | 4 |
| Ciprofloxacin + reserpine | 0.25 | 1.6 |
| Ciprofloxacin + CCCP | 0.25 | 0.25 |
| Norfloxacin | 2 | 16 |
| Norfloxacin + reserpine | 0.2 | 0.4 |
| Tetracyclineb | 0.5 | 0.3 |
| Dyes | ||
| Acriflavine | 4 | 16 |
| Acridine orange | 8 | 4 |
| Ethidium bromide | 2 | 0.5 |
| Ethidium bromide + reserpine | 0.2 | 0.2 |
| Detergents | ||
| Dehydrocholic acid | 256 | 512 |
| Dehydrocholic acid + CCCP | 0.25 | 0.25 |
| Doxorubicin | 12.5 | 0.4 |
| CHAPS | 1024 | 1024 |
| SDS | 32 | 16 |
| TX-114 | 0.5 | 0.25 |
| NP-40 | 4 | <0.015 |
| Disinfectants | ||
| Cetrimide | 0.25 | 4 |
| Efflux pump inhibitors | ||
| TPP | 64 | 32 |
| CCCP | 64 | 512 |
| Reserpine | 128 | 512 |
CHAPS, 3-[(3-cholamidopropyl)-dimethylammonio]-1-propanesulfonate; SDS, sodium dodecyl sulfate; TX-114, Triton X-114; NP-40, Nonidet P-40; TPP, tetraphenylophosphonium.
The MIC of tetracycline was determined three times using a decimal dilution method with increments of 0.1 μg/ml.
grlA, grlB, gyrA, gyrB, and norA were amplified by PCR, and their DNA was sequenced. No changes between the published (1, 12) and amplified sequences of gyrA, grlA, and grlB were seen, other than a silent change at position 139 of grlB (C→T). We detected three mutations in gyrB of both F77 and F145 at nucleotides 1257, 1552, and 1723 which resulted in the amino acid substitutions of Glu419 to Lys419, Ile517 to Thr517, and Leu574 to Arg574, respectively.
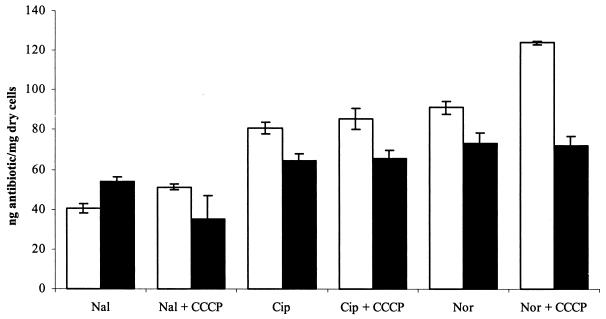
The accumulation of three quinolones (nalidixic acid, ciprofloxacin, and norfloxacin) ± 100 μM CCCP by the two strains was measured by the modified fluorescence method (7) in parallel on at least five separate occasions, and the mean values and standard deviations were determined. Statistical analysis of the accumulation data was performed using the two-tailed Student's t test, and the mean values and standard deviations were determined. A norfloxacin-resistant S. aureus mutant with an inducible norA has been described (2). Each strain was also grown in the presence of putative efflux pump gene inducers nalidixic acid (1/8 of the MIC), norfloxacin (1/10 of the MIC), and ethidium bromide (1/8 of the MIC) prior to measuring accumulation. Accumulation of ethidium bromide was also measured in both strains by the method of A. Neyfakh (personal communication). F145 accumulated significantly lower concentrations of ciprofloxacin and norfloxacin than F77 (P = 0.000047 and 0.00058, respectively) (Fig. 1). However, F145 accumulated nalidixic acid at a higher concentration than did F77 (P = 0.000048). CCCP (100 μM) increased the concentration of all three agents accumulated by F77, and for nalidixic acid and norfloxacin, this was statistically significant (P = 0.005 and 0.000015, respectively). However, CCCP did not increase the concentration of norfloxacin or ciprofloxacin accumulated by F145 (even at 500 μM [data not shown]) and actually reduced the concentration of nalidixic acid accumulated by F145 (P = 0.01). Growth in subinhibitory concentrations of quinolones had little or no effect upon the concentration of quinolone accumulated by F145 with or without CCCP. F145 consistently accumulated slightly more ethidium bromide than F77, but this difference was not statistically significant (data not shown). DNA sequencing of norA, including the promoter region, revealed no differences between F77 and F145. Northern blotting also showed no difference in the expression of norA between the two strains.
FIG. 1.
Accumulation of quinolones by F77 and F145. Data shown are mean values from three experiments; error bars indicate the standard deviation for each experiment. Abbreviations: Nal, nalidixic acid; Cip, ciprofloxacin; Nor, norfloxacin; CCCP, 100 μM CCCP. Symbols: □, F77; ▪, F145.
The combination of fluoroquinolone resistance and nalidixic acid sensitivity has not been previously described for S. aureus; however, a clinical isolate of Escherichia coli has been shown to have a similar phenotype, which can be attributed to reduced fluoroquinolone uptake and a gyrA mutation (6). Failure to detect any mutations within the quinolone resistance-determining regions of the genes coding for topoisomerase IV and DNA gyrase in F145 indicated that other mechanisms, such as efflux, might be involved. The accumulation data support this hypothesis: F145 accumulated less ciprofloxacin and norfloxacin (agents to which the strain is resistant) than F77 but accumulated more nalidixic acid and ethidium bromide, to which it was hypersusceptible. Changes in the accumulation of drugs may be related to changes in efflux pump expression; however, no changes in the expression of norA were detected in F145, suggesting that the reduced accumulation of fluoroquinolones seen in this strain may be due to the activity of another efflux system. The increased accumulation of nalidixic acid (and hypersusceptibility to tetracycline and other agents) seen in F145 is unexplained, but it is possible that this hypersusceptibility is due to the decreased expression of an efflux system and that the fluoroquinolone resistance is a result of concomitant overexpression of another efflux system, for which the latter agents are substrates. The fact that CCCP and reserpine reduce the MICs of fluoroquinolones in F145 but do not increase accumulation of these agents by this strain is unexplained but is similar to data obtained with efflux mutant Streptococcus pneumoniae (11). It may be that CCCP is a substrate for the putative transporter or that the transporter is insensitive to CCCP (e.g., an ABC transporter). Further experiments are being performed in order to understand this phenomenon. The recently published sequences of two staphylococcal genomes (5) suggest the existence of a large number of unidentified transporters, one or more of which may be responsible for the phenotype observed. Homologues of known efflux pump proteins from Bacillus subtilis and E. coli have been identified, and current work is focusing on the role of these homologues in the antibiotic resistance and susceptibility of F145.
REFERENCES
- 1.Ferrero, L., B. Cameron, B. Manse, D. Lagneaux, J. Crouzet, A. Famechon, and F. Blanche. 1994. Cloning and primary structure of Staphylococcus aureus DNA topoisomerase IV: a primary target of fluoroquinolones. Mol. Microbiol. 13:641-653. [DOI] [PubMed] [Google Scholar]
- 2.Kaatz, G. W., and S. M. Seo. 1997. Mechanisms of fluoroquinolone resistance in genetically related strains of Staphylococcus aureus. Antimicrob. Agents Chemother. 41:2733-2737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kaatz, G. W., and S. M. Seo. 1995. Inducible NorA-mediated multidrug resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 39:2650-2655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kaatz, G. W., S. M. Seo, L. O'Brien, M. Wahiduzzaman, and T. J. Foster. 2000. Evidence for the existence of a multidrug efflux transporter distinct from NorA in Staphylococcus aureus. Antimicrob. Agents Chemother. 44:1404-1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kuruda, M., T. Ohta, I. Uchiyama, T. Baba, H. Yuzawa, I. Kobayashi, L. Cui, A. Oouchi, K.-I. Aoki, Y. Nagai, J. Lian, T. Ito, M. Kanamori, H. Matsumaru, A. Maruyama, H. Marukami, A. Hosoyama, Y. Mizutani-Uj, M. K. Takahashi, T. Sawano, R.-I. Inoue, C. Kaito, K. Sekimizu, H. Hirakawa, S. Kubara, S. Goto, J. Yabuzaki, M. Kanehisa, A. Yamashita, K. Oshima, K. Furuwa, C. Yoshino, S. Tadayoshi, M. Hattori, N. Ogasawara, H. Hayashi, and K. Hiramatsu. 2001. Whole genome sequencing of methicillin-resistant Stapyhlococcus aureus. Lancet 357:1225-1240. [DOI] [PubMed] [Google Scholar]
- 6.Moniot-Ville, N., J. Guibert, N. Moreau, J. F. Acar, E. Collatz, and L. Gutmann. 1991. Mechanism of quinolone resistance in a clinical isolate of Escherichia coli highly resistant to fluoroquinolone but susceptible to nalidixic acid. Antimicrob. Agents Chemother. 35:519-523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mortimer, P. G., and L. J. V. Piddock. 1991. A comparison of methods used for measuring the accumulation of quinolones by Enterobacteriaceae. Pseudomonas aeruginosa and Staphylococcus aureus. J. Antimicrob. Chemother. 28:639-653. [DOI] [PubMed] [Google Scholar]
- 8.Mũnoz-Bellido, J. L., M. A. Alonso Manzanares, J. A. Martínez Andrés, M. N. Gutiérrez Zufiaurre, G. Ortiz, M. Segovia Hernández, and J. A. García-Rodríguez. 1999. Efflux pump-mediated quinolone resistance in Staphylococcus aureus strains wild type for gyrA, gyrB, grlA, and norA. Antimicrob. Agents Chemother. 43:354-356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ng, E. Y., M. Trucksis, and D. C. Hooper. 1996. Quinolone resistance mutations in topoisomerase IV: relationship to the flqA locus and genetic evidence that topoisomerase IV is the primary target and DNA gyrase is the secondary target of fluoroquinolones in Staphylococcus aureus. Antimicrob. Agents Chemother. 40:1881-1888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Piddock, L. J. V., and M. Zhu. 1991. Mechanism of action of sparfloxacin against and mechanism of resistance in gram-negative and gram-positive bacteria. Antimicrob. Agents Chemother. 35:2423-2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Piddock, L. J. V., and M. M. Johnson. 2002. Accumulation of 10 fluoroquinolones by wild-type or efflux mutant Streptococcus pneumoniae. Antimicrob. Agents Chemother. 46:813-820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sreedharan, S., L. R. Peterson, and L. M. Fisher. 1991. Ciprofloxacin resistance in coagulase-positive and -negative staphylococci: role of mutations at serine 84 in the DNA gyrase A protein of Staphylococcus aureus and Staphylococcus epidermidis. Antimicrob. Agents Chemother. 35:2151-2154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tanaka, M., Y. Onodera, Y. Uchida, K. Sato, and I. Hayakawa. 1997. Inhibitory activities of quinolones against DNA gyrase and topoisomerase IV purified from Staphylococcus aureus. Antimicrob. Agents Chemother. 41:2362-2366. [DOI] [PMC free article] [PubMed] [Google Scholar]