Abstract
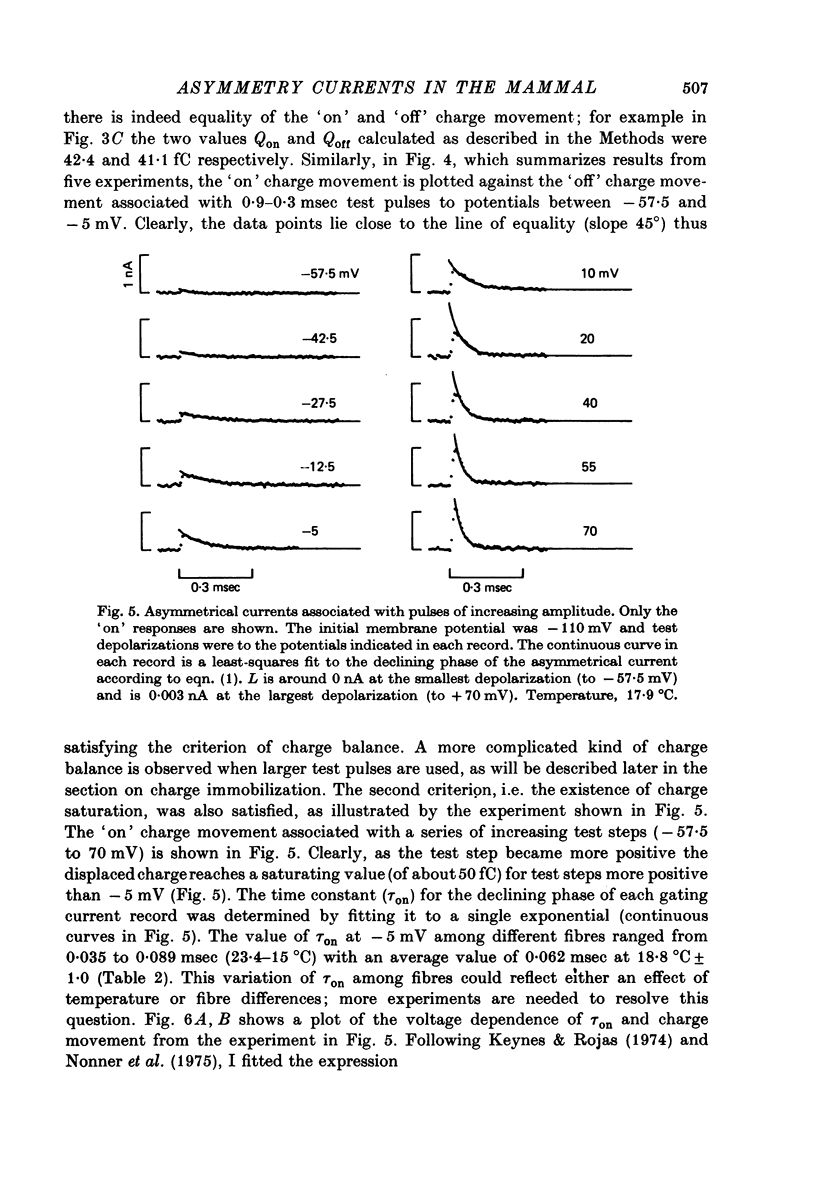
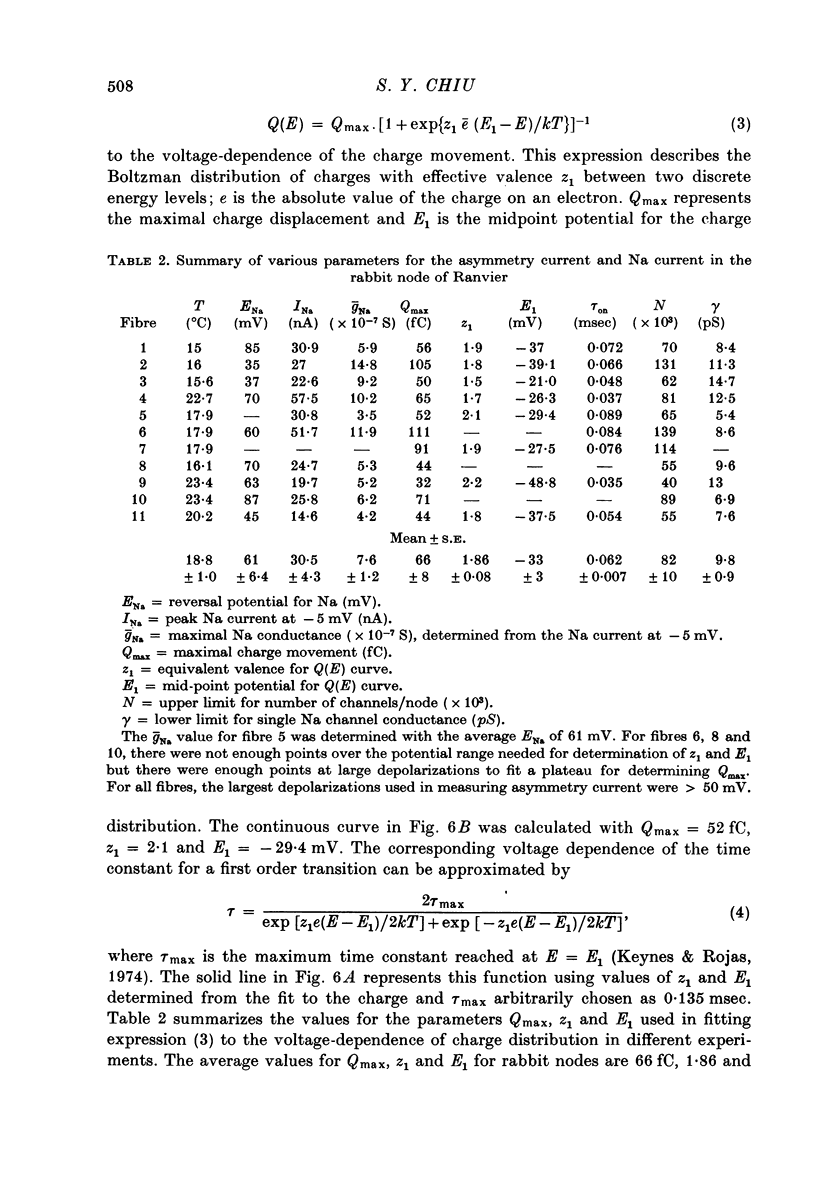
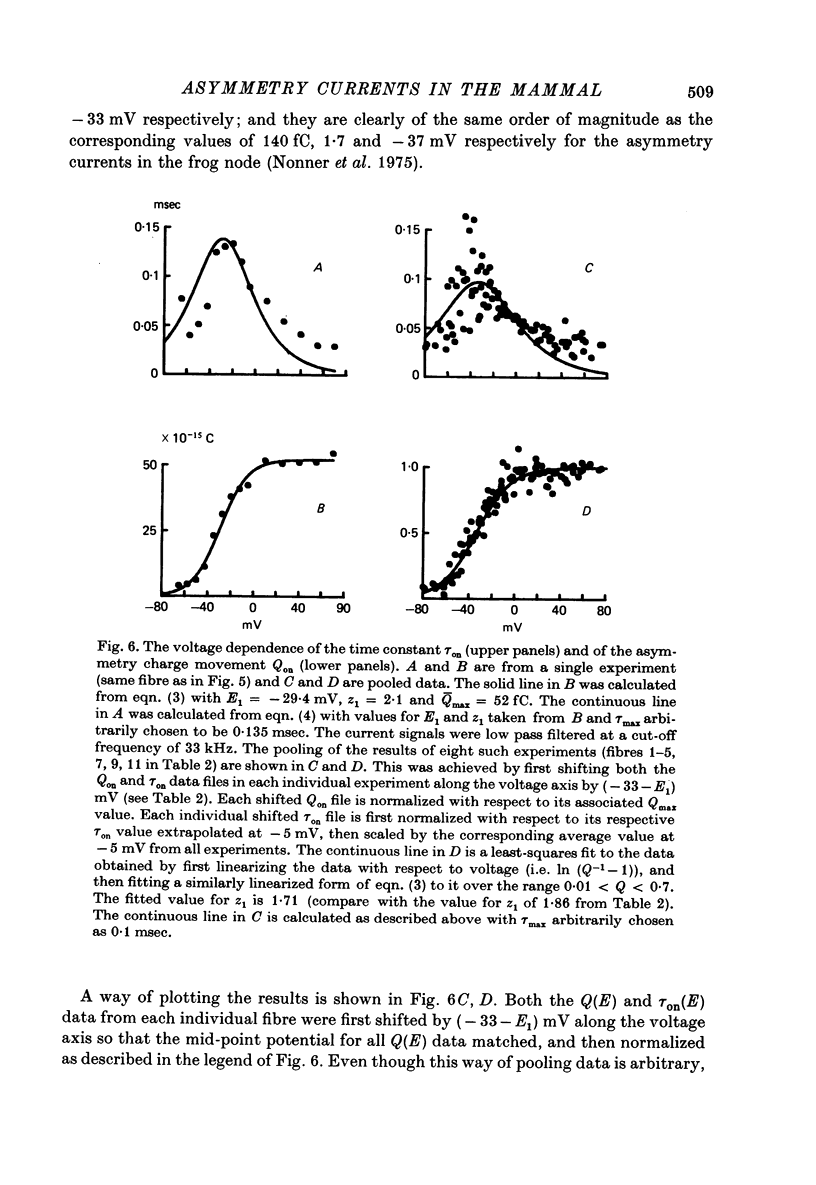
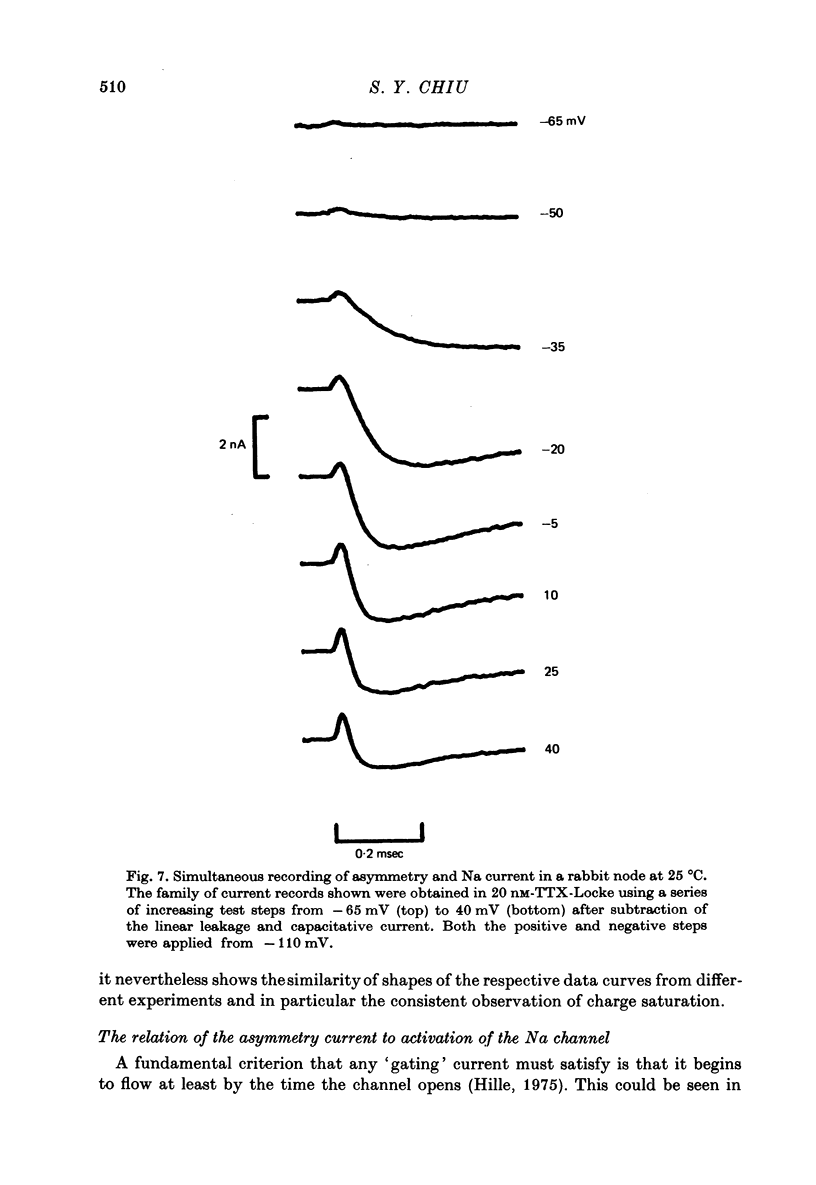
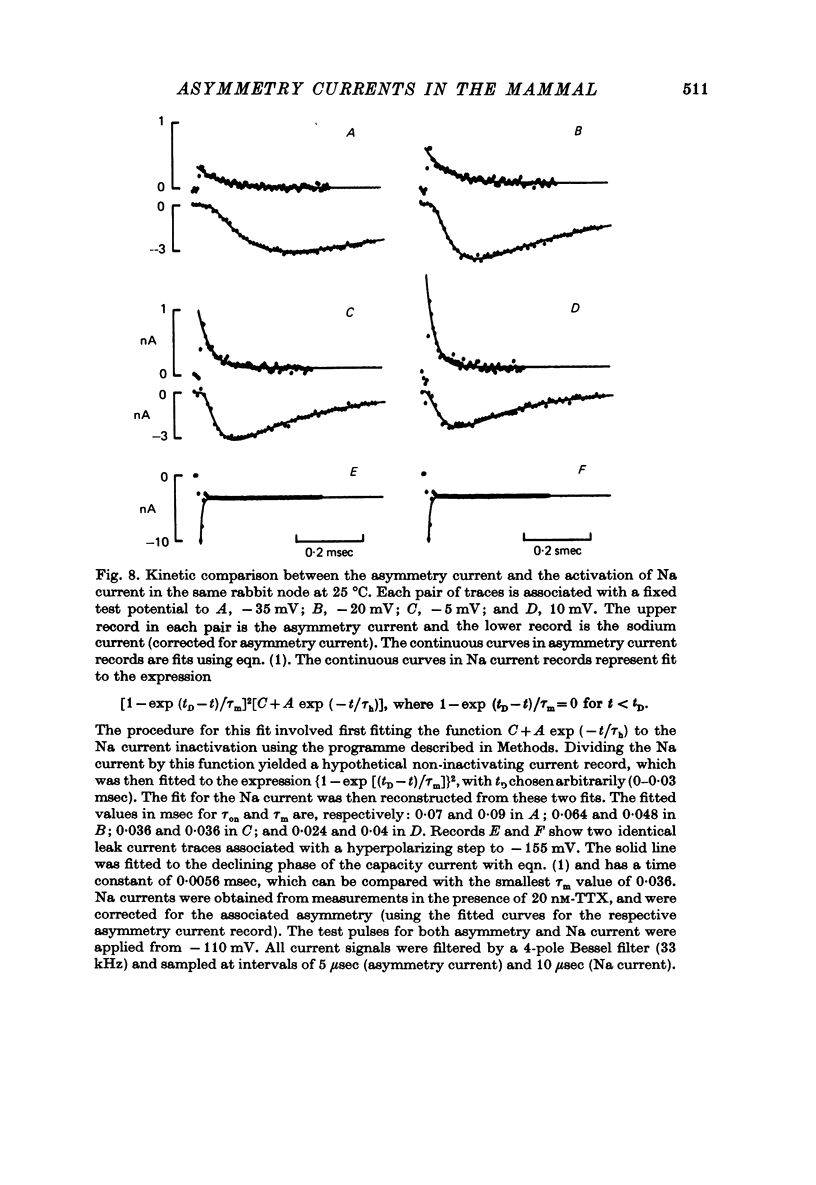
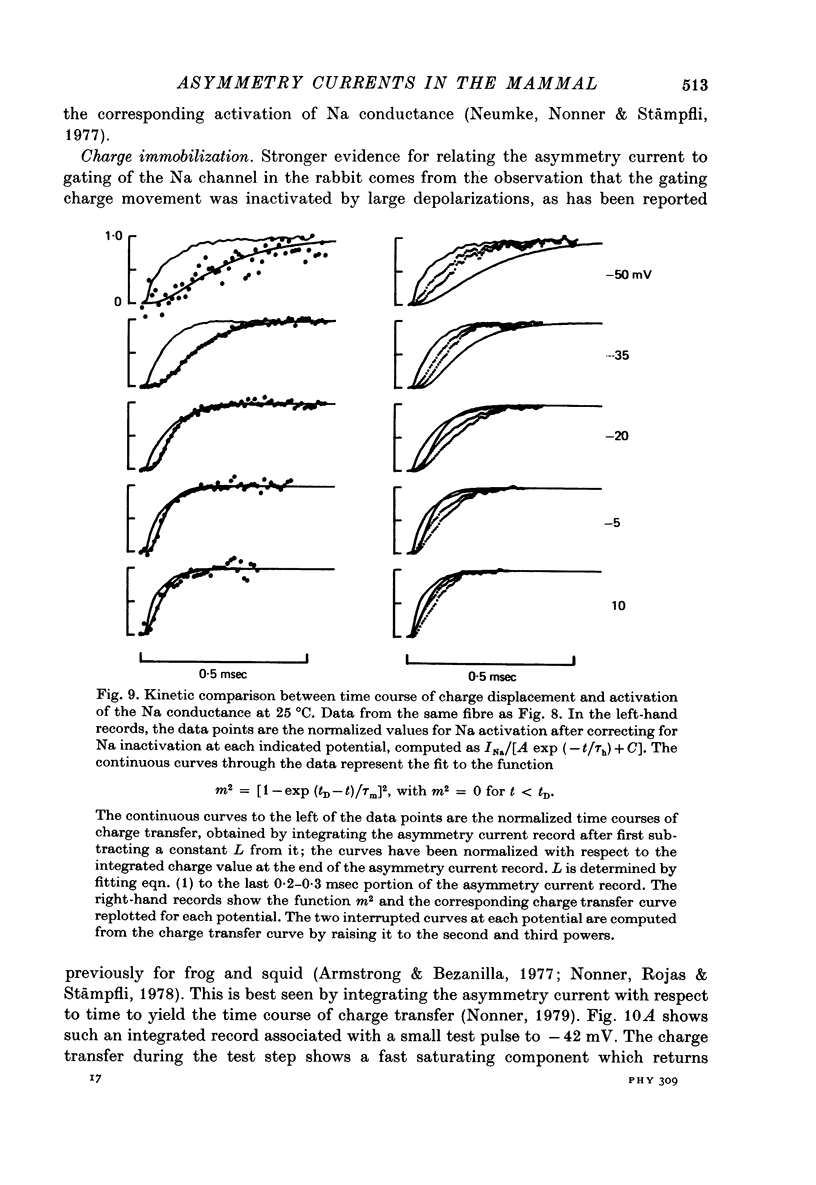
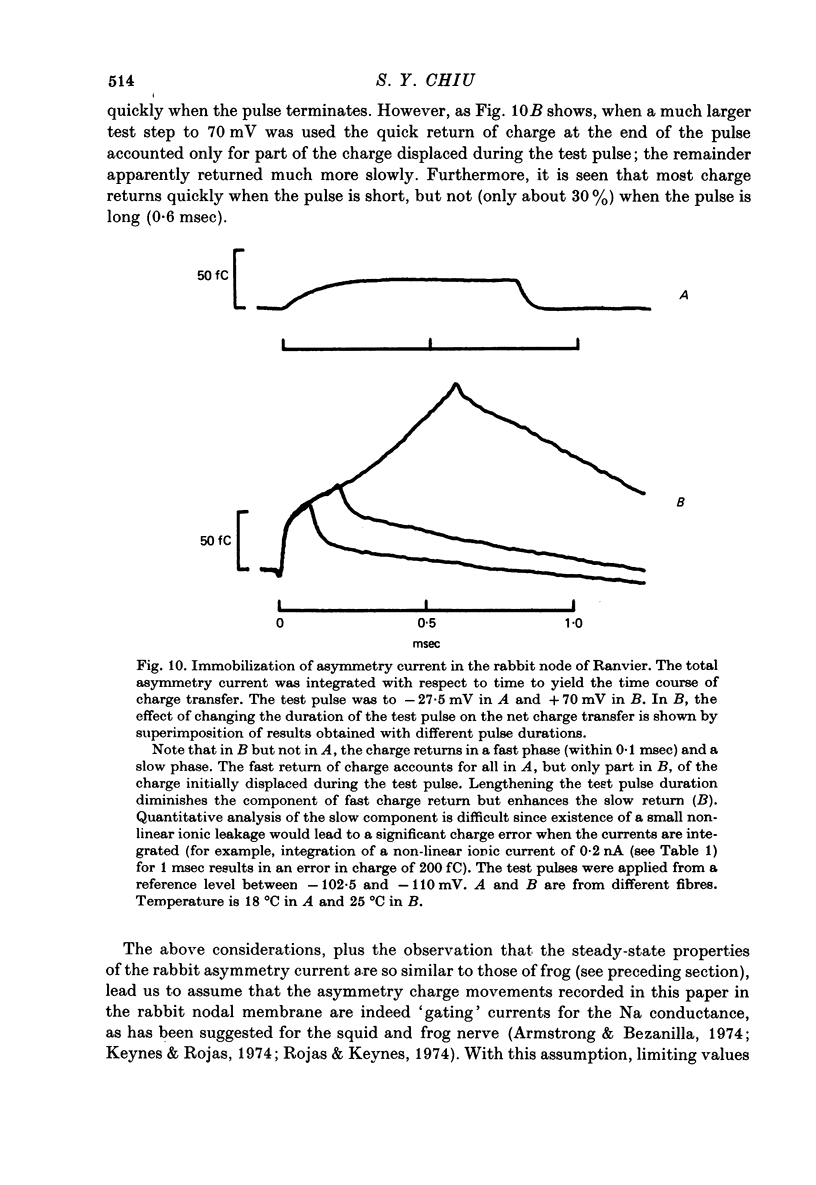
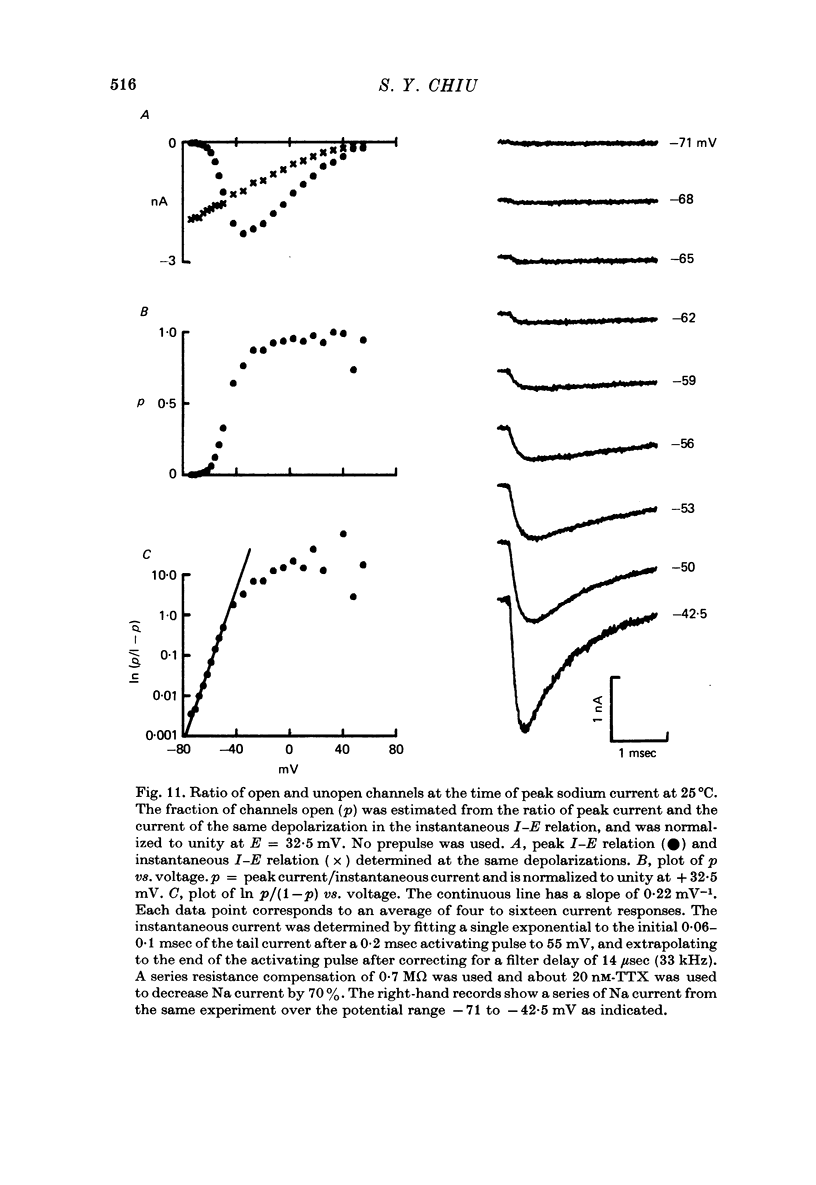
1. Asymmetrical displacement currents were recorded in the rabbit node of Ranvier by averaging the currents associated with depolarizing and hyperpolarizing pulses in the temperature range 15-25 degrees C with the ends of the fibre cut in 160 mM-CsCl. 2. The identification of the asymmetrical currents as intramembranous current is supported by the findings that (a) the "on' and "off' current transients were equal for short and small depolarizing pulses and (b) the total charge displaced reached saturation at (32-111 x 10(-15) C/node) when sufficiently large depolarizations were applied. 3. After a large depolarization to around 50 mV, the "off' response consisted of a fast phase followed by a slow phase. Lengthening the depolarization reduced the size of the fast response but enhanced the slow response. 4. The steady-state rearrangement of the charges can be described by a Boltzmann distribution of charges with an effective valence of 1.86 and a midpoint potential of -33 mV. The time course of rearrangement of these charges following a change in membrane potential could be fitted reasonably well with a single exponential though a double exponential might have been better at large depolarizations. 5. The steady-state activation curve for Na conductance was measured with various procedures to eliminate the effects of series resistance, which include decreasing peak Na current by TTX, and electronic compensation of the series resistance. The measured steepness of the Na-activation curve corresponds to moving a minimum charge of about 5e to open each Na channel at 22.7 degrees C. 6. The time constant (Tm) for activation of Na current and time constant (Ton) for declining phase of the asymmetry current were of the same order of magnitude but not identical over the potential range -50 to +25 mV. 7. The time course of charge displacement determined from the asymmetry current occurred earlier than did Na activation. Raising the charge displacement curve to second or third power did not yield a curve that matched the time course of Na activation. 8. If all the observed asymmetrical currents are related to the Na-gating system, an upper limit for the number of Na channels per rabbit node is 82,000 and a lower limit for the single channel conductance is 9.8 pS at 18.8 degrees C.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams D. J., Gage P. W. Gating currents associated with sodium and calcium currents in an Aplysia neuron. Science. 1976 May 21;192(4241):783–784. doi: 10.1126/science.1265479. [DOI] [PubMed] [Google Scholar]
- Almers W. Gating currents and charge movements in excitable membranes. Rev Physiol Biochem Pharmacol. 1978;82:96–190. doi: 10.1007/BFb0030498. [DOI] [PubMed] [Google Scholar]
- Armstrong C. M., Bezanilla F. Charge movement associated with the opening and closing of the activation gates of the Na channels. J Gen Physiol. 1974 May;63(5):533–552. doi: 10.1085/jgp.63.5.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M., Bezanilla F. Currents related to movement of the gating particles of the sodium channels. Nature. 1973 Apr 13;242(5398):459–461. doi: 10.1038/242459a0. [DOI] [PubMed] [Google Scholar]
- Bezanilla F., Armstrong C. M. Inactivation of the sodium channel. I. Sodium current experiments. J Gen Physiol. 1977 Nov;70(5):549–566. doi: 10.1085/jgp.70.5.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brismar T. Potential clamp experiments on myelinated nerve fibres from alloxan diabetic rats. Acta Physiol Scand. 1979 Mar;105(3):384–386. doi: 10.1111/j.1748-1716.1979.tb06356.x. [DOI] [PubMed] [Google Scholar]
- Cahalan M. D., Almers W. Block of sodium conductance and gating current in squid giant axons poisoned with quaternary strychnine. Biophys J. 1979 Jul;27(1):57–73. doi: 10.1016/S0006-3495(79)85202-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu S. Y., Mrose H. E., Ritchie J. M. Anomalous temperature dependence of the sodium conductance in rabbit nerve compared with frog nerve. Nature. 1979 May 24;279(5711):327–328. doi: 10.1038/279327a0. [DOI] [PubMed] [Google Scholar]
- Chiu S. Y., Ritchie J. M., Rogart R. B., Stagg D. A quantitative description of membrane currents in rabbit myelinated nerve. J Physiol. 1979 Jul;292:149–166. doi: 10.1113/jphysiol.1979.sp012843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conti F., Hille B., Neumcke B., Nonner W., Stämpfli R. Measurement of the conductance of the sodium channel from current fluctuations at the node of Ranvier. J Physiol. 1976 Nov;262(3):699–727. doi: 10.1113/jphysiol.1976.sp011616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952 Aug;117(4):500–544. doi: 10.1113/jphysiol.1952.sp004764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hille B. Gating in sodium channels of nerve. Annu Rev Physiol. 1976;38:139–152. doi: 10.1146/annurev.ph.38.030176.001035. [DOI] [PubMed] [Google Scholar]
- Keynes R. D., Rojas E. Kinetics and steady-state properties of the charged system controlling sodium conductance in the squid giant axon. J Physiol. 1974 Jun;239(2):393–434. doi: 10.1113/jphysiol.1974.sp010575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura J. E., Meves H. The effect of temperature on the asymmetrical charge movement in squid giant axons. J Physiol. 1979 Apr;289:479–500. doi: 10.1113/jphysiol.1979.sp012748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostyuk P. G., Krishtal O. A., Pidoplichko V. I. Asymmetrical displacement currents in nerve cell membrane and effect of internal fluoride. Nature. 1977 May 5;267(5606):70–72. doi: 10.1038/267070a0. [DOI] [PubMed] [Google Scholar]
- Meves H. The effect of holding potential on the asymmetry currents in squid gaint axons. J Physiol. 1974 Dec;243(3):847–867. doi: 10.1113/jphysiol.1974.sp010780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumcke B., Nonner W., Stämpfli R. Asymmetrical displacement current and its relation with the activation of sodium current in the membrane of frog myelinated nerve. Pflugers Arch. 1976 Jun 22;363(3):193–203. doi: 10.1007/BF00594601. [DOI] [PubMed] [Google Scholar]
- Nonner W. Relations between the inactivation of sodium channels and the immobilization of gating charge in frog myelinated nerve. J Physiol. 1980 Feb;299:573–603. doi: 10.1113/jphysiol.1980.sp013143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nonner W., Rojas E., Stämpfli H. Displacement currents in the node of Ranvier. Voltage and time dependence. Pflugers Arch. 1975;354(1):1–18. doi: 10.1007/BF00584499. [DOI] [PubMed] [Google Scholar]
- Nonner W., Rojas E., Stämpfli R. Asymmetrical displacement currents in the membrane of frog myelinated nerve: early time course and effects of membrane potential. Pflugers Arch. 1978 Jun 21;375(1):75–85. doi: 10.1007/BF00584151. [DOI] [PubMed] [Google Scholar]
- Ritchie J. M., Rogart R. B. Density of sodium channels in mammalian myelinated nerve fibers and nature of the axonal membrane under the myelin sheath. Proc Natl Acad Sci U S A. 1977 Jan;74(1):211–215. doi: 10.1073/pnas.74.1.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigworth F. J. Sodium channels in nerve apparently have two conductance states. Nature. 1977 Nov 17;270(5634):265–267. doi: 10.1038/270265a0. [DOI] [PubMed] [Google Scholar]
- Sigworth F. J. The variance of sodium current fluctuations at the node of Ranvier. J Physiol. 1980 Oct;307:97–129. doi: 10.1113/jphysiol.1980.sp013426. [DOI] [PMC free article] [PubMed] [Google Scholar]


