Abstract
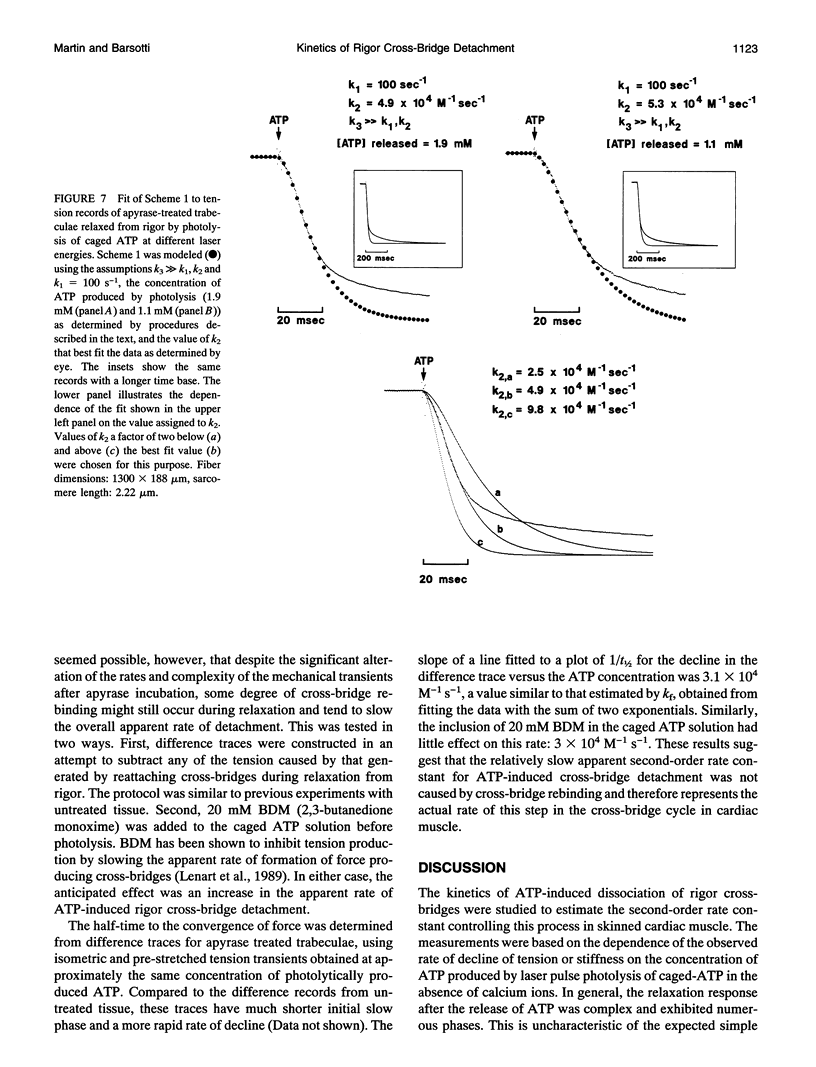
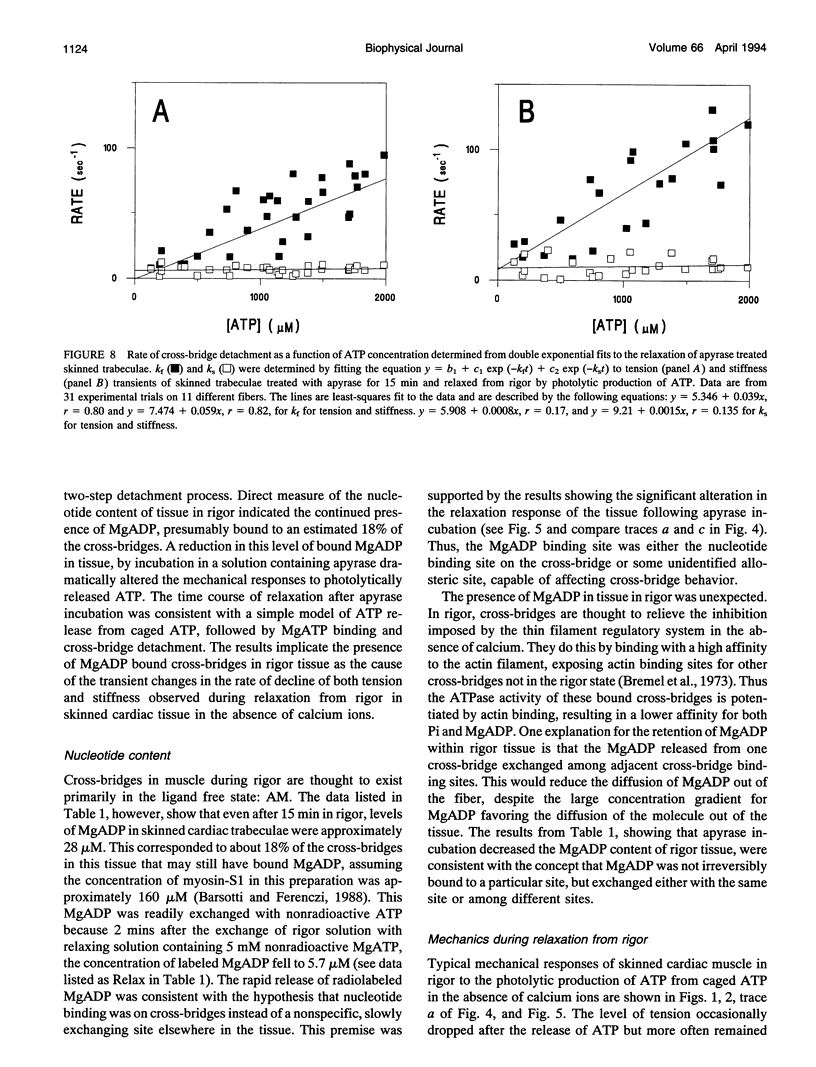
The kinetics of ATP-induced rigor cross-bridge detachment were studied by initiating relaxation in chemically skinned trabeculae of the guinea pig heart using photolytic release of ATP in the absence of calcium ions (pCa > 8). The time course of the fall in tension exhibited either an initial plateau phase of variable duration with little change in tension or a rise in tension, followed by a decrease to relaxed levels. The in-phase component of tissue stiffness initially decreased. The rate then slowed near the end of the tension plateau, indicating transient cross-bridge rebinding, before falling to relaxed levels. Estimates of the apparent second-order rate constant for ATP-induced detachment of rigor cross-bridges based on the half-time for relaxation or on the half-time to the convergence of tension records to a common time course were similar at 3 x 10(3) M-1 s-1. Because the characteristics of the mechanical transients observed during relaxation from rigor were markedly similar to those reported from studies of rabbit psoas fibers in the presence of MgADP (Dantzig, J. A., M. G. Hibberd, D. R. Trentham, and Y. E. Goldman. 1991. Cross-bridge kinetics in the presence of MgADP investigated by photolysis of caged ATP in rabbit psoas muscle fibres. J. Physiol. 432:639-680), direct measurements of MgADP using [3H]ATP in cardiac tissue in rigor were made. Results indicated that during rigor, nearly 18% of the cross-bridges in skinned trabeculae had [3H]MgADP bound. Incubation of the tissue during rigor with apyrase, an enzyme with both ADPase and ATPase activity, reduced the level of [3H]MgADP to that measured following a 2-min chase in a solution containing 5 mM unlabeled MgATP. Apyrase incubation also significantly reduced the tension and stiffness transients, so that both time courses became monotonic and could be fit with a simple model for cross-bridge detachment. The apparent second-order rate constant for ATP-induced rigor cross-bridge detachment measured in the apyrase treated tissue at 4 x 10(4) M-1 s-1 was faster than that measured in untreated tissue. Nevertheless, this rate was still over an order of magnitude slower than the analogous rate measured in previous studies of isolated cardiac actomyosin-S1. These results are consistent with the hypothesis that the presence of MgADP bound cross-bridges suppresses the inhibition normally imposed by the thin filament regulatory system in the absence of calcium ions and allows cross-bridge rebinding and force production during relaxation from rigor.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arner A., Goody R. S., Rapp G., Rüegg J. C. Relaxation of chemically skinned guinea pig taenia coli smooth muscle from rigor by photolytic release of adenosine-5'-triphosphate. J Muscle Res Cell Motil. 1987 Oct;8(5):377–385. doi: 10.1007/BF01578427. [DOI] [PubMed] [Google Scholar]
- Barsotti R. J., Ferenczi M. A. Kinetics of ATP hydrolysis and tension production in skinned cardiac muscle of the guinea pig. J Biol Chem. 1988 Nov 15;263(32):16750–16756. [PubMed] [Google Scholar]
- Bremel R. D., Weber A. Cooperation within actin filament in vertebrate skeletal muscle. Nat New Biol. 1972 Jul 26;238(82):97–101. doi: 10.1038/newbio238097a0. [DOI] [PubMed] [Google Scholar]
- Chiu Y. L., Ballou E. W., Ford L. E. Internal viscoelastic loading in cat papillary muscle. Biophys J. 1982 Nov;40(2):109–120. doi: 10.1016/S0006-3495(82)84465-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dantzig J. A., Goldman Y. E., Millar N. C., Lacktis J., Homsher E. Reversal of the cross-bridge force-generating transition by photogeneration of phosphate in rabbit psoas muscle fibres. J Physiol. 1992;451:247–278. doi: 10.1113/jphysiol.1992.sp019163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dantzig J. A., Hibberd M. G., Trentham D. R., Goldman Y. E. Cross-bridge kinetics in the presence of MgADP investigated by photolysis of caged ATP in rabbit psoas muscle fibres. J Physiol. 1991 Jan;432:639–680. doi: 10.1113/jphysiol.1991.sp018405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dantzig J. A., Walker J. W., Trentham D. R., Goldman Y. E. Relaxation of muscle fibers with adenosine 5'-[gamma-thio]triphosphate (ATP[gamma S]) and by laser photolysis of caged ATP[gamma S]: evidence for Ca2+-dependent affinity of rapidly detaching zero-force cross-bridges. Proc Natl Acad Sci U S A. 1988 Sep;85(18):6716–6720. doi: 10.1073/pnas.85.18.6716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg E., Greene L. E. The relation of muscle biochemistry to muscle physiology. Annu Rev Physiol. 1980;42:293–309. doi: 10.1146/annurev.ph.42.030180.001453. [DOI] [PubMed] [Google Scholar]
- Ferenczi M. A., Homsher E., Trentham D. R. The kinetics of magnesium adenosine triphosphate cleavage in skinned muscle fibres of the rabbit. J Physiol. 1984 Jul;352:575–599. doi: 10.1113/jphysiol.1984.sp015311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferenczi M. A. Phosphate burst in permeable muscle fibers of the rabbit. Biophys J. 1986 Sep;50(3):471–477. doi: 10.1016/S0006-3495(86)83484-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman Y. E., Hibberd M. G., McCray J. A., Trentham D. R. Relaxation of muscle fibres by photolysis of caged ATP. Nature. 1982 Dec 23;300(5894):701–705. doi: 10.1038/300701a0. [DOI] [PubMed] [Google Scholar]
- Goldman Y. E., Hibberd M. G., Trentham D. R. Initiation of active contraction by photogeneration of adenosine-5'-triphosphate in rabbit psoas muscle fibres. J Physiol. 1984 Sep;354:605–624. doi: 10.1113/jphysiol.1984.sp015395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman Y. E., Hibberd M. G., Trentham D. R. Relaxation of rabbit psoas muscle fibres from rigor by photochemical generation of adenosine-5'-triphosphate. J Physiol. 1984 Sep;354:577–604. doi: 10.1113/jphysiol.1984.sp015394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman Y. E. Kinetics of the actomyosin ATPase in muscle fibers. Annu Rev Physiol. 1987;49:637–654. doi: 10.1146/annurev.ph.49.030187.003225. [DOI] [PubMed] [Google Scholar]
- Goldman Y. E., Simmons R. M. Control of sarcomere length in skinned muscle fibres of Rana temporaria during mechanical transients. J Physiol. 1984 May;350:497–518. doi: 10.1113/jphysiol.1984.sp015215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Güth K., Potter J. D. Effect of rigor and cycling cross-bridges on the structure of troponin C and on the Ca2+ affinity of the Ca2+-specific regulatory sites in skinned rabbit psoas fibers. J Biol Chem. 1987 Oct 5;262(28):13627–13635. [PubMed] [Google Scholar]
- Hibberd M. G., Dantzig J. A., Trentham D. R., Goldman Y. E. Phosphate release and force generation in skeletal muscle fibers. Science. 1985 Jun 14;228(4705):1317–1319. doi: 10.1126/science.3159090. [DOI] [PubMed] [Google Scholar]
- Hibberd M. G., Trentham D. R. Relationships between chemical and mechanical events during muscular contraction. Annu Rev Biophys Biophys Chem. 1986;15:119–161. doi: 10.1146/annurev.bb.15.060186.001003. [DOI] [PubMed] [Google Scholar]
- Horiuti K., Somlyo A. V., Goldman Y. E., Somlyo A. P. Kinetics of contraction initiated by flash photolysis of caged adenosine triphosphate in tonic and phasic smooth muscles. J Gen Physiol. 1989 Oct;94(4):769–781. doi: 10.1085/jgp.94.4.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan J. H., Forbush B., 3rd, Hoffman J. F. Rapid photolytic release of adenosine 5'-triphosphate from a protected analogue: utilization by the Na:K pump of human red blood cell ghosts. Biochemistry. 1978 May 16;17(10):1929–1935. doi: 10.1021/bi00603a020. [DOI] [PubMed] [Google Scholar]
- Kawai M., Brandt P. W. Sinusoidal analysis: a high resolution method for correlating biochemical reactions with physiological processes in activated skeletal muscles of rabbit, frog and crayfish. J Muscle Res Cell Motil. 1980 Sep;1(3):279–303. doi: 10.1007/BF00711932. [DOI] [PubMed] [Google Scholar]
- Marston S. B., Taylor E. W. Comparison of the myosin and actomyosin ATPase mechanisms of the four types of vertebrate muscles. J Mol Biol. 1980 Jun 5;139(4):573–600. doi: 10.1016/0022-2836(80)90050-9. [DOI] [PubMed] [Google Scholar]
- McCray J. A., Herbette L., Kihara T., Trentham D. R. A new approach to time-resolved studies of ATP-requiring biological systems; laser flash photolysis of caged ATP. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7237–7241. doi: 10.1073/pnas.77.12.7237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millar N. C., Homsher E. Kinetics of force generation and phosphate release in skinned rabbit soleus muscle fibers. Am J Physiol. 1992 May;262(5 Pt 1):C1239–C1245. doi: 10.1152/ajpcell.1992.262.5.C1239. [DOI] [PubMed] [Google Scholar]
- Millar N. C., Homsher E. The effect of phosphate and calcium on force generation in glycerinated rabbit skeletal muscle fibers. A steady-state and transient kinetic study. J Biol Chem. 1990 Nov 25;265(33):20234–20240. [PubMed] [Google Scholar]
- Morano I., Rüegg J. C. What does TnCDANZ fluorescence reveal about the thin filament state? Pflugers Arch. 1991 May;418(4):333–337. doi: 10.1007/BF00550870. [DOI] [PubMed] [Google Scholar]
- Poole K. J., Rapp G., Maéda Y., Goody R. S. The time course of changes in the equatorial diffraction patterns from different muscle types on photolysis of caged-ATP. Adv Exp Med Biol. 1988;226:391–404. [PubMed] [Google Scholar]
- Rapp G., Poole K. J., Maeda Y., Güth K., Hendrix J., Goody R. S. Time-Resolved Structural Studies on Insect Flight Muscle after Photolysis of Caged-ATP. Biophys J. 1986 Nov;50(5):993–997. doi: 10.1016/S0006-3495(86)83540-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siemankowski R. F., Wiseman M. O., White H. D. ADP dissociation from actomyosin subfragment 1 is sufficiently slow to limit the unloaded shortening velocity in vertebrate muscle. Proc Natl Acad Sci U S A. 1985 Feb;82(3):658–662. doi: 10.1073/pnas.82.3.658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith J. P., Barsotti R. J. A computer-based servo system for controlling isotonic contractions of muscle. Am J Physiol. 1993 Nov;265(5 Pt 1):C1424–C1432. doi: 10.1152/ajpcell.1993.265.5.C1424. [DOI] [PubMed] [Google Scholar]
- Smith S. J., White H. D. Kinetic mechanism of 1-N6-etheno-2-aza-ATP and 1-N6-etheno-2-aza-ADP binding to bovine ventricular actomyosin-S1 and myofibrils. J Biol Chem. 1985 Dec 5;260(28):15156–15162. [PubMed] [Google Scholar]
- Somlyo A. V., Goldman Y. E., Fujimori T., Bond M., Trentham D. R., Somlyo A. P. Cross-bridge kinetics, cooperativity, and negatively strained cross-bridges in vertebrate smooth muscle. A laser-flash photolysis study. J Gen Physiol. 1988 Feb;91(2):165–192. doi: 10.1085/jgp.91.2.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stienen G. J., Ferenczi M. A. Relaxation from rigor by photolysis of caged-ATP in different types of muscle fibres from Xenopus laevis. J Muscle Res Cell Motil. 1991 Dec;12(6):507–516. doi: 10.1007/BF01738439. [DOI] [PubMed] [Google Scholar]
- Taylor E. W. Mechanism of actomyosin ATPase and the problem of muscle contraction. CRC Crit Rev Biochem. 1979;6(2):103–164. doi: 10.3109/10409237909102562. [DOI] [PubMed] [Google Scholar]
- Taylor R. S., Weeds A. G. The magnesium-ion-dependent adenosine triphosphatase of bovine cardiac Myosin and its subfragment-1. Biochem J. 1976 Nov;159(2):301–315. doi: 10.1042/bj1590301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker J. W., Lu Z., Moss R. L. Effects of Ca2+ on the kinetics of phosphate release in skeletal muscle. J Biol Chem. 1992 Feb 5;267(4):2459–2466. [PubMed] [Google Scholar]
- West J. M., Stephenson D. G. Contractile activation and the effects of 2,3-butanedione monoxime (BDM) in skinned cardiac preparations from normal and dystrophic mice (129/ReJ). Pflugers Arch. 1989 Mar;413(5):546–552. doi: 10.1007/BF00594187. [DOI] [PubMed] [Google Scholar]
- Yamakawa M., Goldman Y. E. Mechanical transients initiated by photolysis of caged ATP within fibers of insect fibrillar flight muscle. J Gen Physiol. 1991 Oct;98(4):657–679. doi: 10.1085/jgp.98.4.657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Tombe P. P., ter Keurs H. E. An internal viscous element limits unloaded velocity of sarcomere shortening in rat myocardium. J Physiol. 1992 Aug;454:619–642. doi: 10.1113/jphysiol.1992.sp019283. [DOI] [PMC free article] [PubMed] [Google Scholar]


