Abstract
Choline is a required nutrient, and some humans deplete quickly when fed a low-choline diet, whereas others do not. Endogenous choline synthesis can spare some of the dietary requirement and requires one-carbon groups derived from folate metabolism. We examined whether major genetic variants of folate metabolism modify susceptibility of humans to choline deficiency. Fifty-four adult men and women were fed diets containing adequate choline and folate, followed by a diet containing almost no choline, with or without added folate, until they were clinically judged to be choline-deficient, or for up to 42 days. Criteria for clinical choline deficiency were a more than five times increase in serum creatine kinase activity or a >28% increase of liver fat after consuming the low-choline diet that resolved when choline was returned to the diet. Choline deficiency was observed in more than half of the participants, usually within less than a month. Individuals who were carriers of the very common 5,10-methylenetetrahydrofolate dehydrogenase-1958A gene allele were more likely than noncarriers to develop signs of choline deficiency (odds ratio, 7.0; 95% confidence interval, 2.0-25; P < 0.01) on the low-choline diet unless they were also treated with a folic acid supplement. The effects of the C677T and A1298C polymorphisms of the 5,10-methylene tetrahydrofolate reductase gene and the A80C polymorphism of the reduced folate carrier 1 gene were not statistically significant. The most remarkable finding was the strong association in premenopausal women of the 5,10-methylenetetrahydrofolate dehydrogenase-1958A gene allele polymorphism with 15 times increased susceptibility to developing organ dysfunction on a low-choline diet.
Keywords: genetic polymorphism, methylene tetrahydrofolate dehydrogenase, methylene tetrahydrofolate reductase, reduced folate carrier, nutrient requirement
Choline or its metabolites are needed for the structural integrity and signaling functions of cell membranes; it is the major source of methyl groups in the diet (one of choline's metabolites, betaine, participates in the methylation of homocysteine to form methionine), and it directly affects cholinergic neurotransmission, transmembrane signaling, and lipid transport/metabolism (1). Choline is a required nutrient, and the Institute of Medicine and the National Academy of Sciences of the USA set an adequate intake level for choline of 550 mg/day for men and 425 mg/day for women (2).
One of the clinical consequences of dietary choline deficiency can be the development of fatty liver (hepatosteatosis) (3, 4), because a lack of phosphatidylcholine limits the export of excess triglyceride from liver (5, 6). Also, choline deficiency induces hepatocyte apoptosis with leakage of alanine aminotransferase from liver into blood (4, 7, 8). Some people, when deprived of choline, develop muscle damage and increased creatine kinase (CK) activity in blood (9). This effect may be attributable to impaired membrane stability as a consequence of diminished availability of phosphatidylcholine. The health significance of this rise in CK activity is unknown, but the rise is certainly an important surrogate marker for choline depletion status.
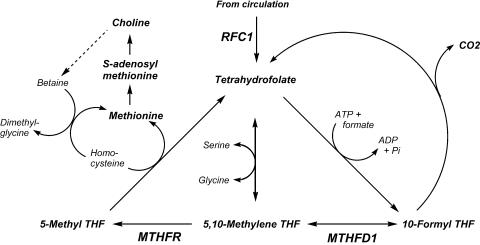
Little is understood about the factors that influence dietary requirements for choline in humans. Endogenous production of choline during phosphatidylcholine biosynthesis (through the methylation of phosphatidylethanolamine by phosphatidylethanolamine N-methyltransferase) is most active in liver but has been identified in many other tissues, including the brain and the mammary gland (10-12). This synthesis of choline provides some but not all of the choline required to sustain normal organ function in humans (4). The use of choline as a methyl-group donor also influences the dietary requirement for choline. The metabolism of choline, methionine, and methylfolate are closely interrelated and intersect at the formation of methionine from homocysteine (Fig. 1). Betaine:homocysteine methyltransferase catalyzes the remethylation of homocysteine by using the choline metabolite betaine as the methyl donor (13, 14). In an alternative pathway, 5-methyltetrahydrofolate:homocysteine S-methyltransferase (also known as methionine synthase) regenerates methionine by using a methyl group derived de novo from the one-carbon pool (15). Perturbing the metabolism of one of the methyl donors results in compensatory changes in the other methyl donors because of the intermingling of these metabolic pathways (16-19). Rats ingesting a low-choline diet showed diminished tissue concentrations of methionine and S-adenosylmethionine (SAM) (19) and of total folate (17). Humans deprived of dietary choline have difficulty removing homocysteine after a methionine load and develop elevated plasma homocysteine concentrations (20). Methotrexate, which is widely used in the treatment of cancer, psoriasis, and rheumatoid arthritis, limits the availability of methyl groups by competitively inhibiting dihydrofolate reductase, a key enzyme in intracellular folate metabolism. Rats treated with methotrexate have diminished pools of all choline metabolites in liver (21). Choline supplementation reverses the fatty liver caused by methotrexate administration (22-24). Genetically modified mice with defective 5,10-methylene tetrahydrofolate reductase (MTHFR) activity become choline-deficient (25), an important observation because many humans have genetic polymorphisms that alter the activity of this enzyme (26, 27). Thus, common SNPs in genes of folate metabolism could increase the demands for choline as a methyl-group donor, thereby increasing dietary requirements for this essential nutrient.
Fig. 1.
Three polymorphic genes are critical for folate-mediated one-carbon transfer. THF, tetrahydrofolate; MTHFR, 5,10-methylene tetrahydrofolate reductase; MTHFD1, cytosolic 5,10-methylene tetrahydrofolate dehydrogenase; RFC1, reduced folate carrier 1.
In this study, we examine whether SNPs in genes of folate metabolism increase the susceptibility of humans to developing signs of organ dysfunction when fed a low-choline diet.
Methods
Subjects. Healthy adults were recruited by advertising. Both males (n = 31) and females (n = 31) were included, with ages ranging from 18 to 70 years. Inclusion was contingent on age-typical good state of health as determined by physical examination and standard clinical laboratory tests. Of the originally recruited 62 subjects, 58 completed at least the initial baseline phase and the depletion phase. Of these 58, 1 subject was excluded because of a 9-kg weight loss during the study, and 3 subjects were excluded because they did not comply with diet restrictions, leaving 54 subjects included in all analyses. Subject characteristics were as follows: 28 women and 26 men; 34 Caucasians, 14 African-Americans, 3 Asians, and 3 of other ethnicity; mean age was 38.7 ± 15.4 (SD) years; mean body mass index was 25.0 ± 3.7 kg/m2. The ethnicity of the participants reflects the local population characteristics of the Raleigh-Durham-Chapel Hill area of North Carolina. The criteria for subject selection and all details of the clinical protocol were approved by the institutional review board of the University of North Carolina at Chapel Hill.
Clinical Studies. The participants stayed at the University of North Carolina at Chapel Hill General Clinical Research Center for the entire duration of the study and could leave only for brief periods under the direct supervision of study staff. All foods were prepared in-house to protocol specifications (28). Total food intake was adjusted to be isocaloric and to provide adequate intake levels of macro- and micronutrients. Individual energy requirements were estimated by using the Harris-Benedict equation, and individual adjustments were made during the first week on the basal diet, if necessary, to achieve participants' satiety. Once individual needs had been determined, daily energy intakes were kept at a constant level, ranging between 35 and 45 kcal/kg of body weight. The diets, which provided 0.8 g/kg high biologic value protein, with 30% energy coming from fat and the remaining energy from carbohydrate, met or exceeded the estimated average requirement for methionine plus cysteine and the recommended dietary allowances for vitamin B12 and all other vitamins except folic acid (diets contained 100 μg/day folate, see below). During the initial 10 days (baseline), the participants consumed normal foods containing 550 mg of choline per 70 kg of body weight per day, which approximates the current adequate intake level (550 mg/day for men and 425 mg/day for women; ref. 2) and 400 μg of folic acid per day as a supplement (General Nutrition Center, Pittsburgh). The actual choline content of a sampling of duplicate portions was assayed by our laboratory (29). Subjects were then switched to a choline-depletion diet containing <50 mg of choline per 70 kg of body weight per day by eliminating choline-rich foods, as confirmed by analysis of duplicate food portions (32, 33). Details of diet formulations were previously published (28). Furthermore, participants were randomly assigned to receive either placebo or 400 μg of folic acid per day as a supplement in addition to the amount of folate consumed with food (100 μg/day). Diets were well tolerated.
Periodic determinations of urinary choline and betaine concentrations were used to confirm compliance with the dietary restrictions. Subjects remained on this depletion diet until they developed signs of organ dysfunction associated with choline deficiency, or for 42 days if they did not. Humans were deemed to have signs of organ dysfunction associated with choline deficiency if they had more than a 5-fold increase of serum CK activity while on the choline depletion diet and if this increased CK resolved when they were returned to the repletion diet (9), or if they had an increase of liver fat content by 28% or more while on the choline depletion diet and if this increased liver fat resolved when they were returned to the repletion or ad libitum diet (20). After the depletion period, subjects were repleted by gradually increasing choline intake and were maintained at a final level of >550 mg of choline per day for at least 3 days.
Change in liver fat content was estimated by MRI with a clinical MR system (Vision 41.5-T, Siemens, Iselin, NJ) using a modified “In and Out of Phase” procedure (20, 30). This approach utilizes the differences in transverse magnetization intensity after an ultrabrief time interval [fast low-angle shot (FLASH); echo time (TE) = 2.2 msec and 4.5 msec, with a flip angle of 80°, and relaxation time (TR) = 140 msec]. Processing of successive FLASH MRI images with software from Siemens Medical Solutions (Malvern, PA) was used to estimate fat content. Organ content was derived from measurements across five liver slices per subject and standardized by relating the results to the similarly measured fat content of spleen. We assumed that spleen signal would be largely invariant and used this value to calculate the outcome variable liver-to-spleen fat ratio.
Fasting blood samples were taken every 3-4 days throughout the study, and in particular, after 10 days on the 550 mg/day choline diet (baseline), at the end of the low-choline diet (depletion phase), and after consuming a repletion diet with 137-550 mg/day choline (repletion phase). The metabolic response to an oral challenge with 100 mg l-methionine/kg was determined initially and after depletion. Blood for homocysteine, SAM, and S-adenosylhomocysteine (SAH) measurements was obtained before and 4 h after methionine ingestion (20).
Laboratory Analyses. Plasma folate concentrations in fasted samples were measured by using a microbiological assay (31). Serum was analyzed by using a dry-slide colorimetric method for CK activity by the McClendon Clinical Laboratories at University of North Carolina Hospitals, which is both Clinical Laboratory Improvement Act (CLIA)- and College of American Pathologists (CAP)-accredited. Total plasma homocysteine concentration was measured in fasted samples by using a HPLC method (20, 32). SAM and SAH levels in plasma were measured by HPLC with fluorescence detection after conversion into their fluorescent isoindoles (33).
Genotyping. Genomic DNA was prepared according to manufacturer's instructions from peripheral blood with a commercial extraction kit (PureGene, Gentra Systems) and diluted to a standard concentration of 1 μg/ml. The polymorphic sites of MTHFR (MTHFR-C677T and -A1298C), cytosolic C-1-THF synthase (MTHFD1-G1958A), and reduced folate carrier 1 (RFC1-G80A) were studied (Fig. 1). The targeted DNA sequences were amplified by multiplex PCR, purified, and then analyzed with matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (34). For all subjects, duplicate samples were genotyped. In the few instances of amplification failure, new DNA was prepared from backup blood samples.
Statistical Analysis. Genotype-related differences in dichotomous outcomes were calculated with two-sided Fisher's exact probability test (http://faculty.vassar.edu/lowry/webtext.html) to determine statistical significance (GraphPad, San Diego). Odds ratios for depletion by presence vs. absence of the predominant alleles were calculated as the odds to deplete for subjects with the allele divided by odds for subjects without the allele (35). Statistical significance of odds ratios was again calculated by using two-sided Fisher's exact probability test. The statistical significance of group differences for continuous variables was assessed with Student's t test, and differences between subjects on different diets were assessed by using pair t tests. A two-sample t test based on the differences between homocysteine concentrations in subjects on the two diets was used to compare the clinically depleted and not-depleted groups (GraphPad).
Results
Genetic Variation. The distribution of the polymorphic variants of MTHFR and cytosolic MTHFD1 (Table 1) largely agreed with that of larger North European populations [Norwegian Colorectal Cancer Prevention study (NORCCAP)] that were analyzed previously with the same genotyping methodology (K. Meyer and P. M. Ueland, personal communication). Within our group, however, fewer African-Americans than Caucasians had the variant allele MTHFD1 1958A (allele frequency 0.18 vs. 0.50). The RFC1 80G allele was slightly underrepresented in our subjects (0.47 vs. 0.58), but we considered this the reference allele, nonetheless. The difference is mostly attributable to the presence of many non-Caucasians in our regionally representative population sample.
Table 1. Effect of genotype on susceptibility to organ dysfunction in humans eating low-choline diets.
| Polymorphism | Genotype (n) | % subjects with signs of choline deficiency | P | Odds ratio and 95% CI |
|---|---|---|---|---|
| MTHFR 677 | CC (28) | 61 | 0.63 | CC vs. CT/TT |
| CT (22) | 73 | Odds ratio, 1.76 | ||
| TT (4) | 75 | 95% CI, 0.56–5.6 | ||
| MTHFR 1298 | AA (28) | 64 | 0.90 | AA vs AC/CC |
| AC (22) | 68 | Odds ratio, 1.25 | ||
| CC (4) | 75 | 95% CI, 0.40–3.9 | ||
| MTHFD1 1958 | GG (20) | 40 | 0.007 | GG vs GA/AA |
| GA (28) | 82 | Odds ratio, 7.0 | ||
| AA (6) | 83 | 95% CI, 2.0–25 | ||
| RFC1 80 | AA (19) | 56 | 0.59 | AA vs AG/GG |
| AG (20) | 70 | Odds ratio, 1.82 | ||
| GG (15) | 73 | 95% CI, 0.56–5.9 |
Significance was calculated with 2×3 Fisher's exact test. Application of Bonferroni's correction for multiple testing lowers the threshold for statistical significance to 0.0125.
Folate Status. Twenty-six participants were assigned to receive placebo, and 28 subjects received an additional 400 μg of folic acid per day as a supplement. Average serum folate concentration at the end of the depletion phase was lower in subjects with the lower folate intake (22.1 ± 1.3 nmol/liter vs. 28.3 ± 1.2 nmol/liter; P < 0.01 by Student's t test). None of the investigated polymorphisms had a statistically significant effect on serum folate concentrations at any time point.
Signs of Organ Dysfunction Associated with Choline Deficiency. Twelve subjects responded to the low-choline diet with an increase in serum CK activity, all but 1 within a month on the depletion diet. A significant increase in liver fat content was observed in another 24 participants, usually within a month on the depletion diet. In six of these participants, however, it took up to 42 days to accumulate the additional 28% or more of liver fat. Eighteen subjects did not show signs of organ dysfunction in response to the low-choline diet. Daily supplementation with folic acid did little to affect the likelihood of developing signs of choline deficiency [odds ratio, absence over presence of signs of choline deficiency in subjects with supplementation vs. without supplementation 0.8, 95% confidence interval (CI), 0.26-2.5].
Methionine Metabolism. Homocysteine concentrations during both baseline and choline-depletion conditions were measured in 54 subjects, and methionine load tests were completed in 52 of these participants at the end of the baseline and depletion diet phases. Homocysteine concentration increased by 19% with the low-choline regime (P < 0.001, paired t test). Supplementation with 400 μg/day folic acid blunted this increase, compared with placebo (15% vs. 23% increase, P < 0.05, Student's t test). However, there was no significant interaction between fasting plasma homocysteine concentration and clinical status (P = 0.113, Student's t test), because there was a similar increase of this measure in subjects judged to be clinically depleted (by 1.4 μmol/liter; 95% CI, 1.1-1.7) as in subjects without clinical signs of choline deficiency (0.9 μmol/liter; 95% CI, 0.5-1.4). None of the polymorphic variants had a statistically significant influence on plasma homocysteine concentrations, at either baseline or depletion. The expected rise of plasma homocysteine concentration after methionine loading was observed on both diets. The rise in homocysteine concentration in response to the methionine challenge in subjects eating the 550-mg choline diet was significantly less in subjects without the RFC1 80G allele than in the carriers of this allele (P = 0.02, Student's t test). None of the other polymorphic variations were predictive for the metabolic response to methionine or the change of this response with choline depletion.
As was previously reported in a pilot study (20), the rise in plasma homocysteine concentration after a methionine load was greater in individuals developing signs of choline deficiency when ingesting a low-choline diet than in those that did not. In this study, after a methionine load at the end of the depletion phase, plasma homocysteine concentrations in the group with signs of choline deficiency rose 6.9 μmol/liter above that which was previously observed after a methionine load on the 550-mg choline diet (95% CI, 4.4-9.3, P = 0.0001). For subjects without signs of deficiency, plasma homocysteine did not increase significantly after the same methionine load (1.6 μmol/liter; 95% CI, -1.7-4.9, P = 0.318).
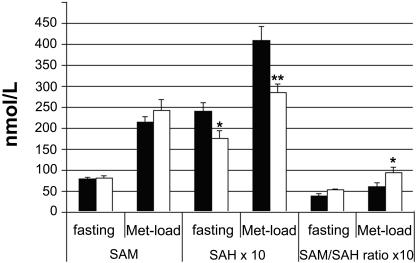
SAM and SAH concentrations were assessed in 26 individuals with MTHFD1 1958GA/AA genotype and in 15 individuals with MTHFD1 1958GG genotype. Concentrations did not change significantly upon switching from baseline to a low-choline diet. On both the baseline and the low-choline diets, SAM and SAH concentrations increased greatly after oral methionine load, as expected. The postloading concentration of SAH increased from 28.8 ± 12.8 nmol/liter on the baseline diet to 34.7 ± 12.8 nmol/liter on the low-choline diet (P < 0.05). No statistically significant change of postloading SAM concentrations in response to the low-choline diet was observed. Although SAM concentrations at depletion did not differ significantly between MTHFD1 1958 genotype groups, SAH concentrations were significantly lower in participants with the GG genotype than in those with the GA and AA genotypes, both with and without methionine loading (Fig. 2). The same pattern of genotype-SAH concentration was observed while subjects were on the baseline diet, but the contrasts did not reach statistical significance.
Fig. 2.
Increase in SAH concentrations after a methionine load was lower in MTFD1 1958 GG individuals. Subjects were treated with a low-choline diet as described in Methods. Blood for SAM and SAH measurements was obtained before (fasting) and 4 h after an oral methionine load (Met-load; 100 mg of l-methionine per kg of body weight) from 26 individuals with MTHFD1 1958 GA/AA genotype and from 15 individuals with MTHFD1 1958 GG genotype. Values are presented as mean ± standard error. Solid bars indicate means of individuals with the MTHFD1 1958 GA or AA genotypes, and open bars correspond to means from those with the GG genotype. *, P < 0.05; **, P < 0.01 different from other genotype by one-way ANOVA.
Among the examined polymorphisms, the MTHFD1 G1958A variant was the best predictor of susceptibility to choline depletion (Table 1). In light of the small subject numbers in several of the cells, the carriers of what are usually the minor alleles were grouped together for calculating odds ratios. Again, the MTHFD1 polymorphism was the only one of the four variants with a distinct impact on risk of developing clinical signs of choline deficiency. A higher percentage of the 34 carriers of the 1958A allele showed signs of choline deficiency in response to the low-choline diet (odds ratio, 7.0; two-sided, P = 0.0025; Table 2). This genotypic difference was largely attributable to the fact that none of the young women with MTHFD1 1958GG genotype showed deficiency signs, whereas seven of the eight young women carriers of the GA or AA genotypes did so. The corresponding differences were not seen in men (odds ratio, 3.0; P = 0.33) and postmenopausal women (odds ratio, 1.0; P = 0.99). However, because only four postmenopausal women had the MTHFD1 1958-GG genotype, the power to detect an odds ratio of 7.0 (the average for all subjects) at the usual level of significance was only 0.13. With the eight male carriers of the GG genotype, the corresponding statistical power also was very low. Thus, the study was clearly underpowered for the detection of an effect smaller than the one observed in the young women.
Table 2. Effect of folate and sex on effect of MTHFD1 1958 SNP on susceptibility to organ dysfunction in humans eating low-choline diets.
| Group | Genotype (n) | % subjects with signs of choline deficiency | P | Odds ratio and 95% CI |
|---|---|---|---|---|
| Premenopausal women | GG (8) | 0 | Odds ratio, 85* | |
| GA/AA (8) | 88 | 0.000 | 95% CI, 3–2418 | |
| Postmenopausal women | GG (4) | 75 | Odds ratio, 1.0 | |
| GA/AA (8) | 75 | 0.99 | 95% CI, 0.06–16 | |
| Men | GG (8) | 63 | Odds ratio, 3.0 | |
| GA/AA (18) | 83 | 0.33 | 95% CI, 0.45–20 | |
| All subjects | GG (20) | 40 | Odds ratio, 7.0 | |
| GA/AA (34) | 82 | 0.007 | 95% CI, 2.0–25 | |
| Diet folate only | GG (10) | 30 | Odds ratio, 35 | |
| GA/AA (16) | 94 | 0.00 | 95% CI, 3.0–39 | |
| Diet folate plus folic acid supplement | GG (10) | 50 | Odds ratio, 2.6 | |
| (400 μg/day) | GA/AA (18) | 72 | 0.41 | 95% CI, 0.52–13 |
The odds ratios were calculated as the odds of showing signs of deficiency for subjects without the MTHFD1 1958A allele divided by the odds of showing signs of deficiency for subjects with the A allele. Two-sided P and 95% CI were calculated with Fisher's exact test.
The odds ratio for premenopausal women was calculated by adding a value of 0.5 to each cell for premenopausal women; this value is an underestimate, because the value in the GG-YES cell was 0. Thus, in reality, the odds ratio exceeds 85
In regard to folate supplementation, the MTHFD1 1958A allele-related difference in susceptibility to developing organ dysfunction when eating a low-choline diet was greatest in the group getting folate only from the diet (no supplement; odds ratio, 35; two-sided, P = 0.001; Table 2) and was much smaller and statistically not significant in the folate-supplemented group (odds ratio, 2.5; two-sided, P = 0.41; Table 2). None of the other polymorphisms showed a similarly strong or consistent relationship to choline-depletion susceptibility, with or without folate supplementation. Power calculations based on Fisher's exact test indicate that, for an odds ratio of 1.5, >200 subjects with the least frequent genotype would be needed to give the study a power of 0.8 at the usual level of statistical significance.
Discussion
In this investigation of healthy subjects, more than half of the participants developed signs of organ dysfunction when consuming low-choline diets. This report focuses on the impact of genetic variants of folate metabolism on susceptibility to clinical choline deficiency. The most remarkable finding was the strong association of the MTHFD1 G1958A polymorphism with susceptibility to developing signs of organ dysfunction associated with choline deficiency, particularly in comparison to the very weak association with variation in the MTHFR gene. Presence of the MTHFD1 1958A allele made it much more likely that subjects developed signs of choline deficiency.
Choline-deficient individuals were found to have impaired capacity to handle a methionine load, developing elevated plasma SAH and homocysteine concentrations. This finding confirms our earlier report in a limited number of subjects (20) and highlights the importance of alternative folate-mediated pathways for homocysteine remethylation. The observation of higher SAH concentrations in carriers of the MTHFD1 1958A allele, compared with noncarriers, is particularly informative, because accumulation of this metabolite has been found to be a more sensitive indicator of disturbed methionine regeneration than is elevated homocysteine concentration (36). SAH is a potent inhibitor of phosphatidylethanolamine methyltransferase, which catalyzes the endogenous formation of choline moiety in liver (10). Phosphatidylethanolamine methyltransferase activity is increased by estrogen (37), and this mechanism probably explains why we observed that premenopausal women were relatively resistant to developing signs of organ dysfunction when fed a low-choline diet, compared with men. It is in these premenopausal women that we observed the most significant effect of the MTHFD1 1958A SNP on susceptibility to developing signs of choline deficiency (Table 2). We suggest that this SNP restricts methyl-group availability enough so that SAM availability for the phosphatidylethanolamine methyltransferase-catalyzed formation of choline moiety becomes limiting, thereby eliminating this protective mechanism for females. Our SAH data (Fig. 2) support this hypothesis. Alternatively, it is possible that men and postmenopausal women are already so susceptible to choline deficiency (80% show signs of organ dysfunction on a low-choline diet) that a further increase in susceptibility cannot be appreciated, given the small incremental effect size. In premenopausal women, in contrast, where 60% of the population was resistant to choline deficiency, there was sufficient margin for detecting an increase in susceptibility associated with the MTHFD1 1958A SNP.
Under standard conditions, serine provides the bulk of one-carbon groups (38). Cytosolic serine hydromethyltransferase (EC 2.1.2.1) transfers a one-carbon unit from serine to THF, and the resulting 5,10-methylene-THF can then be reduced by MTHFR to 5-methyl-THF. An alternative source for the one-carbon unit is derived from formate through mitochondrial or cytosolic reactions that can be linked to free folate by formyl-THF synthetase (EC 6.3.4.3) and generate 10-formyl-THF in an ATP-dependent reaction. This distinct reaction is only one of three that are catalyzed by the cytosolic enzyme C-1-THF synthase complex (all encoded by the MTHFD1 gene sequence). Two additional reactions, mediated by methylene-THF dehydrogenase (EC 1.5.1.5) and methenyl-THF cyclohydrolase (EC 3.5.4.9), can then convert 10-formyl-THF to 5,10-methylene-THF (Fig. 1). Although the formation of 5-methyl-THF is practically irreversible in vivo, the interconversion of 5,10-methylene-THF and 10-formyl-THF is closer to equilibrium (39). Thus, 5,10-methylene-THF may be directed either toward homocysteine remethylation or away from it. Both purine synthesis and oxidative release of carbon dioxide and THF by 10-formyl-THF dehydrogenase (EC 1.5.1.6) draw on the 10-formyl-THF pool. The irreversible and nonproductive dissipation of an excess in transferable one-carbon units is likely to be a significant regulatory factor, because the intrahepatic concentration of 10-formyl-THF exceeds the half-maximal equilibrium constant Km of 10-formyl-THF dehydrogenase (40). The 10-formyl-THF synthase activity of C-1-THF synthase, on the other hand, can add to the 5,10-methylene-THF pool by linking formate to free folate. The G-to-A transition mutation at nucleotide 1958 in MTHFD1 causes an arginine to glutamine substitution in the protein region responsible for 10-formyl-THF dehydrogenase, which is far removed from the region providing for the methenyl-THF cyclohydrolase and methylene-THF dehydrogenase activities. The MTHFD1 G1958A polymorphism may thus affect the delicately balanced flux between 5,10-methylene-THF and 10-formyl-THF and thereby influence the availability of 5-methyl-THF for homocysteine remethylation. The pattern of decreased SAM:SAH ratios among individuals with an MTHFD1 1958A allele appears to be consistent with the view that their one-carbon flux slightly tilts away from 5-methyl-THF formation. The finding of increased susceptibility to developing signs of choline deficiency coinciding with evidence of impaired 5-methyl-THF availability (increased SAH concentration) in carriers of an MTHFD1 1958A allele makes it less likely that the association is due to just random chance. If a nearby gene locus in strong linkage disequilibrium with the MTHFD1 1958A were ultimately responsible for increased susceptibility to choline deficiency, this locus also would have to explain the observed shift in methyl-group metabolism.
It is of particular interest that the gene-variant effect can be overcome if humans are supplemented with folic acid. It may be surprising that the development of choline deficiency signs was strongly favored by the presence of the MTHFD1 1958A allele but not by polymorphic variants of MTHFR or RFC1. A partial explanation may be provided by a recent investigation of folate-dependent homocysteine remethylation in young women (41). This study found that the MTHFR 677TT genotype had little detectable effect on remethylation flux. In comparison, the MTHFD1 1958A polymorphism, which has not been extensively investigated until now, may be a more potent determinant of the rate at which one-carbon units become available for methyl-group transfer reactions, such as the synthesis of phosphatidyl-choline from phosphatidylethanolamine.
Observations on genetically linked susceptibility to choline deficiency are important because they can help the Institute of Medicine refine their recommendations for dietary choline intake by taking into account the needs of sizeable population groups with greater-than-average vulnerability to low choline or folate intake. There also is a potential relevance for the prevention of neural tube defects. One of the great successes of nutrition science has been the identification of the role that folate plays in normal neural tube closure; adequate dietary folate intake by mothers during pregnancy can prevent >50% of neural tube defects in babies (42). The risk of having a child with a neural tube defect increases in mothers with the G1958A SNP in MTHFD1 (43). As discussed earlier, choline and folate metabolism are highly interrelated. Inhibition of choline uptake and metabolism was associated with the development of neural tube defects in mice (44, 45). Recent evidence suggests that availability of choline also might impact the risk of neural tube defects in humans: A retrospective case-control study (400 cases and 400 controls) of periconceptional dietary intakes of choline in women in California found that women in the lowest quartile for daily choline intake had 4× the risk of having a baby with a neural tube defect than did women in the highest quartile for intake (46). We suggest the need to focus future epidemiologic studies on interactions among dietary choline intake, folate intake, and MTHFD1 polymorphisms.
Acknowledgments
We thank Klaus Meyer and Per Magne Ueland (both of the University of Bergen, Norway) for the SNP analyses; Conrad Wagner (Vanderbilt University, Nashville) for the analysis of methionine metabolites; and Lester Kwock for assistance with MRI studies. This work was supported by National Institutes of Health Grants DK55865, DK56350, RR00046, and ES10126.
Author contributions: S.H.Z. designed research; K.D., L.M.F., and S.H.Z. performed research; M.K., K.D., and S.H.Z. analyzed data; and M.K. and S.H.Z. wrote the paper.
Conflict of interest statement: No conflicts declared.
This paper was submitted directly (Track II) to the PNAS office.
Abbreviations: THF, tetrahydrofolate; MTHFD1, cytosolic 5,10-methylene tetrahydrofolate dehydrogenase; MTHFR, 5,10-methylene tetrahydrofolate reductase; RFC1, reduced folate carrier 1; SAH, S-adenosylhomocysteine; SAM, S-adenosylmethionine; CI, confidence interval; CK, creatine kinase.
References
- 1.Zeisel, S. H. & Blusztajn, J. K. (1994) Ann. Rev. Nutr. 14, 269-296. [DOI] [PubMed] [Google Scholar]
- 2.Institute of Medicine and National Academy of Sciences of the USA (1998) in Dietary Reference Intakes for Folate, Thiamin, Riboflavin, Niacin, Vitamin B12, Panthothenic Acid, Biotin, and Choline (Natl. Acad. Press, Washington, DC), Vol. 1, pp. 390-422. [PubMed] [Google Scholar]
- 3.Buchman, A., Dubin, M., Moukarzel, A., Jenden, D., Roch, M., Rice, K., Gornbein, J. & Ament, M. (1995) Hepatology 22, 1399-1403. [PubMed] [Google Scholar]
- 4.Zeisel, S. H., da Costa, K.-A., Franklin, P. D., Alexander, E. A., Lamont, J. T., Sheard, N. F. & Beiser, A. (1991) FASEB J. 5, 2093-2098. [PubMed] [Google Scholar]
- 5.Yao, Z. M. & Vance, D. E. (1988) J. Biol. Chem. 263, 2998-3004. [PubMed] [Google Scholar]
- 6.Yao, Z. M. & Vance, D. E. (1989) J. Biol. Chem. 264, 11373-11380. [PubMed] [Google Scholar]
- 7.Albright, C. D., Lui, R., Bethea, T. C., da Costa, K.-A., Salganik, R. I. & Zeisel, S. H. (1996) FASEB J. 10, 510-516. [DOI] [PubMed] [Google Scholar]
- 8.Albright, C. D., da Costa, K.-A., Craciunescu, C. N., Klem, E., Mar, M. H. & Zeisel, S. H. (2005) Cell. Physiol. Biochem. 15, 59-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.da Costa, K.-A., Badea, M., Fischer, L. M. & Zeisel, S. H. (2004) Am. J. Clin. Nutr. 80, 163-170. [DOI] [PubMed] [Google Scholar]
- 10.Vance, D. E., Walkey, C. J. & Cui, Z. (1997) Biochim. Biophys. Acta 1348, 142-150. [DOI] [PubMed] [Google Scholar]
- 11.Blusztajn, J. K., Zeisel, S. H. & Wurtman, R. J. (1985) Biochem. J. 232, 505-511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang, E. K., Blusztajn, J. K., Pomfret, E. A. & Zeisel, S. H. (1988) Biochem. J. 256, 821-828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sunden, S., Renduchintala, M., Park, E., Miklasz, S. & Garrow, T. (1997) Arch. Biochem. Biophys. 345, 171-174. [DOI] [PubMed] [Google Scholar]
- 14.Millian, N. S. & Garrow, T. A. (1998) Arch. Biochem. Biophys. 356, 93-98. [DOI] [PubMed] [Google Scholar]
- 15.Bailey, L. B. & Gregory, J. F., III (1999) J. Nutr. 129, 779-782. [DOI] [PubMed] [Google Scholar]
- 16.Kim, Y.-I., Miller, J. W., da Costa, K.-A., Nadeau, M., Smith, D., Selhub, J., Zeisel, S. H. & Mason, J. B. (1995) J. Nutr. 124, 2197-2203. [DOI] [PubMed] [Google Scholar]
- 17.Selhub, J., Seyoum, E., Pomfret, E. A. & Zeisel, S. H. (1991) Cancer Res. 51, 16-21. [PubMed] [Google Scholar]
- 18.Varela-Moreiras, G., Selhub, J., da Costa, K.-A. & Zeisel, S. H. (1992) J. Nutr. Biochem. 3, 519-522. [Google Scholar]
- 19.Zeisel, S. H., Zola, T., da Costa, K.-A. & Pomfret, E. A. (1989) Biochem. J. 259, 725-729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.da Costa, K.-A., Gaffney, C. E., Fischer, L. M. & Zeisel, S. H. (2005) Am. J. Clin. Nutr. 81, 440-444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pomfret, E. A., da Costa, K.-A. & Zeisel, S. H. (1990) J. Nutr. Biochem. 1, 533-541. [DOI] [PubMed] [Google Scholar]
- 22.Freeman-Narrod, M., Narrod, S. A. & Yarbro, J. W. (1977) Med. Pediatr. Oncol. 3, 9-14. [DOI] [PubMed] [Google Scholar]
- 23.Aarsaether, N., Berge, R. K., Aarsland, A., Svardal, A. & Ueland, P. M. (1988) Biochim. Biophys. Acta 958, 70-80. [DOI] [PubMed] [Google Scholar]
- 24.Svardal, A. M., Ueland, P. M., Berge, R. K., Aarsland, A., Aarsaether, N., Lonning, P. E. & Refsum, H. (1988) Cancer Chemother. Pharmacol. 21, 313-318. [DOI] [PubMed] [Google Scholar]
- 25.Schwahn, B. C., Chen, Z., Laryea, M. D., Wendel, U., Lussier-Cacan, S., Genest, J., Jr., Mar, M. H., Zeisel, S. H., Castro, C., Garrow, T. & Rozen, R. (2003) FASEB J. 17, 512-514. [DOI] [PubMed] [Google Scholar]
- 26.Rozen, R. (1996) Clin. Invest. Med. 19, 171-178. [PubMed] [Google Scholar]
- 27.Wilcken, D., Wang, X., Sim, A. & McCredie, R. (1996) Arterioscler. Thromb. Vasc. Biol. 16, 878-882. [DOI] [PubMed] [Google Scholar]
- 28.Busby, M. G., Fischer, L., da Costa, K.-A., Thompson, D., Mar, M. H. & Zeisel, S. H. (2004) J. Am. Diet Assoc. 104, 1836-1845. [DOI] [PubMed] [Google Scholar]
- 29.Zeisel, S. H., Mar, M. H., Howe, J. C. & Holden, J. M. (2003) J. Nutr. 133, 1302-1307. [DOI] [PubMed] [Google Scholar]
- 30.Fishbein, M., Gardner, K., Potter, C., Schmalbrock, P. & Smith, M. (1997) Magn. Reson. Imaging 15, 287-293. [DOI] [PubMed] [Google Scholar]
- 31.Horne, D. W. & Patterson, D. (1988) Clin. Chem. 34, 2357-2359. [PubMed] [Google Scholar]
- 32.Ubbink, J. B., Hayward Vermaak, W. J. & Bissbort, S. (1991) J. Chromatogr. 565, 441-446. [DOI] [PubMed] [Google Scholar]
- 33.Davis, S. R., Quinlivan, E. P., Shelnutt, K. P., Maneval, D. R., Ghandour, H., Capdevila, A., Coats, B. S., Wagner, C., Selhub, J., Bailey, L. B., et al. (2005) J. Nutr. 135, 1040-1044. [DOI] [PubMed] [Google Scholar]
- 34.Meyer, K., Fredriksen, A. & Ueland, P. M. (2004) Clin. Chem. 50, 391-402. [DOI] [PubMed] [Google Scholar]
- 35.Bland, J. M. & Altman, D. G. (2000) Br. Med. J. 320, 1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kerins, D. M., Koury, M. J., Capdevila, A., Rana, S. & Wagner, C. (2001) Am. J. Clin. Nutr. 74, 723-729. [DOI] [PubMed] [Google Scholar]
- 37.Drouva, S. V., LaPlante, E., Leblanc, P., Bechet, J. J., Clauser, H. & Kordon, C. (1986) Endocrinol. 119, 2611-2622. [DOI] [PubMed] [Google Scholar]
- 38.Davis, S. R., Stacpoole, P. W., Williamson, J., Kick, L. S., Quinlivan, E. P., Coats, B. S., Shane, B., Bailey, L. B. & Gregory, J. F., III (2004) Am. J. Physiol. 286, E272-E279. [DOI] [PubMed] [Google Scholar]
- 39.Horne, D. W. (2003) J. Nutr. 133, 476-478. [DOI] [PubMed] [Google Scholar]
- 40.Gregory, J. F., III, Cuskelly, G. J., Shane, B., Toth, J. P., Baumgartner, T. G. & Stacpoole, P. W. (2000) Am. J. Clin. Nutr. 72, 1535-1541. [DOI] [PubMed] [Google Scholar]
- 41.Davis, S. R., Quinlivan, E. P., Shelnutt, K. P., Ghandour, H., Capdevila, A., Coats, B. S., Wagner, C., Shane, B., Selhub, J., Bailey, L. B., et al. (2005) J. Nutr. 135, 1045-1050. [DOI] [PubMed] [Google Scholar]
- 42.Shaw, G. M., Schaffer, D., Velie, E. M., Morland, K. & Harris, J. A. (1995) Epidemiology 6, 219-226. [DOI] [PubMed] [Google Scholar]
- 43.Brody, L. C., Conley, M., Cox, C., Kirke, P. N., McKeever, M. P., Mills, J. L., Molloy, A. M., O'Leary, V. B., Parle-McDermott, A., Scott, J. M. & Swanson, D. A. (2002) Am. J. Hum. Genet. 71, 1207-1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fisher, M. C., Zeisel, S. H., Mar, M. H. & Sadler, T. W. (2001) Teratology 64, 114-122. [DOI] [PubMed] [Google Scholar]
- 45.Fisher, M. C., Zeisel, S. H., Mar, M. H. & Sadler, T. W. (2002) FASEB J. 16, 619-621. [DOI] [PubMed] [Google Scholar]
- 46.Shaw, G. M., Carmichael, S. L., Yang, W., Selvin, S. & Schaffer, D. M. (2004) Am. J. Epidemiol. 160, 102-109. [DOI] [PubMed] [Google Scholar]