Abstract
DNA vaccination has emerged as a powerful approach in the search for a more efficacious vaccine against tuberculosis. In this study, we evaluated the effectiveness of immunizing with combinations of 10 different tuberculosis DNA vaccines that expressed mycobacterial proteins fused at the N terminus to eukaryotic intracellular targeting sequences. In one vaccine combination, the genes were fused to the tissue plasminogen activator signal sequence (TPA), while in a second combination the same 10 genes were expressed as ubiquitin (Ub)-conjugated proteins. In ex vivo studies in which the secretion of gamma interferon was measured, cellular immune responses were detected in mice vaccinated with either the TPA DNA vaccine combination or the Ub DNA vaccine combination at 7 and 14 days following a low-dose Mycobacterium tuberculosis challenge. Moreover, mice vaccinated with the TPA combination, the Ub combination, and Mycobacterium bovis BCG were able to limit the growth of tubercle bacilli in the lung and spleen after a virulent tuberculous aerosol challenge. Histopathological analyses also showed that mice immunized with the DNA vaccine combinations had substantially improved postinfection lung pathology relative to the naïve controls. Finally, in three different long-term experiments, the survival periods following aerogenic challenge were extended as much as sevenfold for vaccinated mice compared to naïve controls. Interestingly, in all three experiments, no significant differences were detected in the mean times to death for mice immunized with the TPA combination or the Ub combination relative to the BCG controls. In conclusion, these studies demonstrate the effectiveness of immunization with DNA vaccine combinations against tuberculosis and suggest that further testing of these plasmid cocktails is warranted.
Tuberculosis (TB) is a major global threat to human health, with more than 2 million people dying each year from Mycobacterium tuberculosis infections (39). Although the current TB vaccine (Mycobacterium bovis BCG) has been widely used throughout the world for many decades, its efficacy has been shown to be highly variable in several well-controlled clinical trials (5). Moreover, since BCG is a live attenuated vaccine, its use is contraindicated in human immunodeficiency-infected and other immunocompromised patients, who ironically have the highest risk of developing TB. Clearly, the development of a new, more effective vaccine would greatly facilitate worldwide control of this ancient plague. As a consequence, the search for a new vaccine against TB has greatly intensified over the last decade, with several experimental vaccines already shown to have promise in preclinical testing (30).
Since DNA vaccines can induce substantial cellular immunity and evoke both CD4 and CD8 T-cell responses, genetic immunization has become a viable strategy for developing new vaccines against intracellular parasites (18). Several recent studies carried out with animal models of TB have suggested that DNA vaccines against this disease may be effective when tested in the clinic (12, 20, 21, 24, 34, 35). In a number of these studies with TB DNA vaccines expressing single antigens, relevant cellular immune responses have been generated and outcomes associated with protection (i.e., bacterial growth reduction and improved lung pathology) have been detected. However, in most of these experiments, the single vaccines have elicited only partial protection against tuberculous challenge, and these single vaccines have not generated protective immunity that has been equivalent to or exceeded the level of protection produced by vaccination with live BCG vaccine (3).
To augment the effectiveness of TB DNA vaccine preparations, we have been investigating a combination DNA vaccine approach. In earlier studies from our laboratory, we validated the combination strategy by demonstrating that a combination of four TB DNA vaccines elicited a more substantial protective immune response than any of the four single components (28). In this report, we extend our combination vaccine studies by evaluating the protection produced by two different combinations of DNA constructs. One set of 10 DNA vaccines generally expresses TB antigens fused at the N terminus to the tissue plasminogen activator signal sequence (TPA), while the other combination encodes the same TB proteins linked at the N terminus to ubiquitin (Ub). The TPA-fused TB antigens are highly expressed secreted proteins that have been shown to induce significant humoral and cell-mediated responses (24). In contrast, the Ub-conjugated proteins generally undergo rapid intracellular degradation and can elicit cytokine responses in the absence of specific antibody production (12). In this study, we demonstrate that the DNA vaccine combinations induce sustained protective responses and that the protective immunity evoked by both the TPA and Ub combinations of TB DNA vaccines was essentially equivalent to the protection induced by live M. bovis BCG.
MATERIALS AND METHODS
Animals.
Specific-pathogen-free C57BL/6 female mice were obtained from the Jackson Laboratories (Bar Harbor, Maine). The mice were 8 weeks old at the time of the vaccination. They were maintained under barrier conditions and fed commercial mouse chow and water ad libitum.
Microorganisms.
M. tuberculosis Erdman (TMC 107) and M. bovis BCG Pasteur (TMC1011) were obtained from the Trudeau Mycobacterial Culture Collection, Saranac Lake, N.Y. The Escherichia coli JM109 and TOP10 strains (Invitrogen, San Diego, Calif.) were used for cloning. For expression of histidine-tagged antigens, an E. coli BL21(DE3)/pLysS strain (Invitrogen) was transformed with pET15b- or pET24b-based expression plasmids (Novagen, Madison, Wis.).
DNA vaccine preparation.
The pJW4303 vector, kindly provided by Jim Arthos (National Institutes of Allergy and Infectious Disease, National Institutes of Health, Bethesda, Md.), was used as backbone for all the DNA vaccine constructs used in this study. The DNA vaccines encoding the ESAT-6 and MPT64 proteins, either in the TPA- and UbGR-fused forms, were generated as indicated in an earlier report (12). The MPT63 (26), MPT83 (36), and KatG (25) antigen encoding genes were cloned as fusions to the TPA sequence as described earlier (28). The antigen 85B (35) gene was cloned in its native form. To create Ub fusion constructs, all these genes were amplified using a direct primer with NheI or XbaI restriction site adapters. Using the standard methods described earlier, these genes were then cloned into the same vector immediately downstream from Ub sequences (UbA and UbGR) (12). The genes encoding the MTB8.4 (7), MTB39A (13), and MTB12 (38) antigens were also amplified by PCR and cloned into the vectors that express TPA fusion or Ub-conjugated proteins as described above. The DNA plasmid encoding the N-terminal region of the Rv1818c gene (PE domain) was created as described previously (11).
Evaluation of the protective activity of DNA vaccines against aerogenic challenge.
Endotoxin-free plasmid DNA was prepared and purified with Qiagen EndoFree Plasmid Maxi Kit (Qiagen, Chatsworth, Calif.). Each plasmid encoding a single M. tuberculosis antigen was purified separately. The two cocktails of DNA vaccines (TPA combination and Ub combination), each containing an equal amount of the 10 plasmids, were prepared by combining the plasmids to a final concentration of 1 mg/ml of DNA in phosphate-buffered saline (PBS). Groups of C57BL/6 mice were injected intramuscularly in all four limbs on days 1, 21, and 42 with 200 μg (20 μg per individual constituent) of the plasmid combination. Other groups of mice were immunized with the TPA-ESAT-6 construct or the empty vector at 200 μg/dose of the single plasmid. As controls, mice were vaccinated subcutaneously with 5 × 106 CFU of BCG on day 1. Either 5 or 16 weeks after receiving the final DNA immunization, the mice were challenged with M. tuberculosis Erdman by the aerogenic route. For the challenge, a frozen ampoule of the Erdman strain was thawed and subjected to 10 s of vibration on a vortex mixer. The suspensions were then diluted to a concentration known to introduce either 30 to 50 CFU, 500 CFU, or 3,000 CFU into the mouse lung over a 30 min exposure period using a Middlebrook chamber (Glas-Col, Terre Haute, Ind.) (8). Five mice were sacrificed after 24 h to confirm the size of the challenge dose.
To assess the bacterial growth in vivo, five mice per group were sacrificed, and the lungs and spleens were removed aseptically and homogenized separately in 5 ml of 0.04%% Tween 80-PBS using a Seward Stomacher 80 blender (Tekmar, Cincinnati, Ohio) as described earlier (12, 24). The homogenates were diluted serially in the Tween-PBS solution, and 50-μl aliquots were plated on Middlebrook 7H11 agar (Difco, Detroit, Mich.). Samples from BCG-vaccinated mice were plated on Middlebrook 7H11 agar (9) containing 2-thiophenecarboxylic acid hydrazide (2 μg/ml). 2-Thiophenecarboxylic acid hydrazide inhibits the proliferation of BCG but does not retard the growth of M. tuberculosis. The number of CFU in the infected organs was determined after 14 to 21 days of incubation at 37° in sealed plastic bags. For the survival studies, 5 to 10 animals per group were maintained until they became moribund and had to be euthanatized.
Evaluation of the cytokine immune responses.
To assess vaccine-induced cytokine production, groups of vaccinated and control mice were sacrificed 30 days after the third vaccination and at 7 and 14 days following a low-dose aerogenic challenge with M. tuberculosis. Spleens from four or five mice were pooled, and the splenocytes were restimulated in vitro with primed mouse bone marrow macrophage cultures. These macrophage cultures were established by flushing the femurs of C57BL/6 mice and culturing the cells in Dulbecco”s modified Eagle medium containing 10% fetal bovine serum (HyClone, Logan, Utah), 2 mM glutamine, 10 mM HEPES, 0.1 mM nonessential amino acids, 50 μg of gentamicin per ml, and 10% L-929 conditioned medium. The bone marrow macrophages were primed 24 h prior to the incubation with splenocytes with one of the following preparations: 1 μg of purified protein derivative (PPD) (Center for Biologics Evaluation and Research reference preparation) per ml, 1 μg of a culture filtrate protein preparation (CFP) (provided by John Belisle, Colorado State University) per ml, or a mixture of the corresponding 10 recombinant His-tagged proteins (each at a final concentration of 1 μg/ml, and prepared by Ni-affinity chromatography) as indicated earlier (24). Supernatants were collected 72 h later, and the amount of gamma interferon (IFN-γ) secreted was analyzed by a cytokine specific enzyme-linked immunosorbent assay (ELISA) using immunoglobulin specific for IFN-γ (PharMingen, San Diego, Calif.), as described previously (12).
Histopathologic analyses.
Lung tissues were perfused and fixed with 10% paraformaldehyde in PBS and then embedded in paraffin for sectioning. The tissue sections were stained with hematoxylin and eosin (H&E) reagent or with Ziehl-Neelsen acid-fast stain and were evaluated by light microscopy.
Statistical analysis.
The CFU results, the cytokine determinations, and the survival data were evaluated using the one-way analysis of variance (ANOVA) of the Graph Pad InStat program.
RESULTS
Composition of the combination DNA vaccines.
To identify active DNA constructs for inclusion into a combination TB DNA vaccine, we generated and screened DNA vaccines expressing 32 different TB antigens. Most of these antigens were selected for evaluation in the context of DNA vaccination because of their documented immunogenicity. Initially, constructs were designed to express TB antigens as N-terminal TPA signal sequence fusions (except for antigen 85B) because high expression rates and efficient secretion of these fusion proteins had been demonstrated in in vitro transfection assays (24). The constructs listed in Table 1 induced short-term protective responses (when administered separately) following a low-dose aerosol challenge as demonstrated by significant reductions in mycobacterial growth in the lungs of vaccinated mice and improved lung pathology, relative to nonvaccinated controls. To assess the impact of increased intracellular degradation on long-term protective responses, the 10 active genes were also cloned as fusions to modified Ub sequences (UbA or UbGR) as described previously (12). Expression of transgene antigens as Ub conjugates often accelerates intracellular protein turnover and may enhance the induction of cellular immune responses (33, 40). The UbA and UbGR constructs for each gene were transfected into rhabdomyosarcoma cells and the relative intracellular concentrations of the Ub conjugates in cell lysates were determined by immunoblotting. The Ub construct within the antigen pair that yielded the highest apparent rate of intracellular turnover was selected for further study (data not shown).
TABLE 1.
M. tuberculosis genes included in the DNA vaccine combinations
| MTB Ag | Genea | Molecular mass(kDa) | Featuresb | Construct | Reference(s) |
|---|---|---|---|---|---|
| ESat-6 | Rv3875 | 6 | CFP | TPA UbGR | 12, 24 |
| mpt64 | Rv1980c | 26 | CFP | TPA UbGR | 12, 24 |
| MPT63 | Rv1926C | 17 | CFP | TPA UbGR | 26, 28 |
| mpt8e | Rv2873 | 23 | CFP | TPA UbGR | 36 |
| Ag85b | Rv1886c | 32 | Mycolyl transferase | NatcUbGR | 28, 35 |
| katG | Rv1908c | 84 | Catalase-peroxidase | TPA UbGR | 25, 24, 28 |
| mtb12 | Rv2376c | 12 | CFP | TPA UbA | 39 |
| mtb8.4 | Rv1174c | 8 | CFP | TPA UbGR | 7 |
| MTB39 | RV1361c | 39 | PPE family | TPA UbGR | 13 |
| 1818CPE | RV1818C | 14 | PE family | Nat UbGR | 11 |
Gene annotation number as indicated in the H37Rv genome sequence (http://www.sanger.ac.uk/Projects/M_tuberculosis/).
The function of only two of these antigens is known.
Nat, native protein.
Cytokine responses in vaccinated animals.
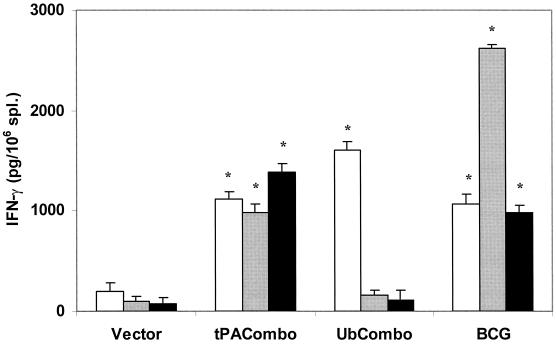
In order to assess whether vaccination with the TB DNA combinations stimulated cell-mediated immunity, splenocyte cytokine responses were evaluated by ex vivo analysis 1 month after the final immunization. Since IFN-γ is a critical component of the antimycobacterial protective response, we focused on evaluating the levels of this cytokine secreted in splenocyte cultures. As seen in Fig. 1, stimulation of the splenocytes with PPD, CFP, or a mixture of all 10 recombinant proteins generated robust IFN-γ responses in cultures of spleen cell from mice vaccinated with the TPA combination or with BCG. In this postvaccination assay, significant IFN-γ responses from mice immunized with the Ub combination were only detected after stimulation of splenocytes with PPD.
FIG. 1.
IFN-γ in vitro secretion by splenocytes from vaccinated and control mice. Splenocytes were restimulated in vitro with bone marrow macrophages that had been primed with either PPD (open bars), a CFP mixture (gray bars), or the recombinant His-tagged antigens (black bars). The amounts of cytokine secreted in the supernatant were determined by ELISA. Spleens from three to five mice per group were pooled, and the supernatants were analyzed in triplicates. Typical results from one of two separate experiments are shown. Asterisks indicate that the amount of IFN-γ secreted is statistically significant compared to the vector control group, as determined by ANOVA (P < 0.05). Error bars, standard errors.
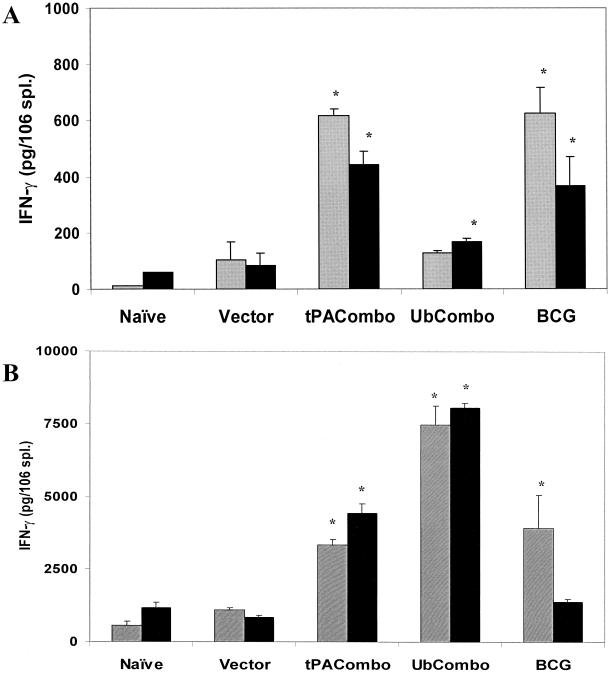
To investigate the cellular immunity induced following infection, vaccinated and control mice were challenged with a low dose of M. tuberculosis Erdman and IFN-γ responses were assessed 7 and 14 days later by ex vivo analysis. In these experiments, the vaccinated mice had been rested 16 weeks before the low-dose aerosol challenge was administered. As shown in Fig. 2A, a modest immune stimulation (probably reflecting the low numbers of bacilli directly implanted into the lung) was seen at 7 days postchallenge. Significant amounts of IFN-γ were detected after splenocytes from mice immunized with the TPA combination or with BCG were stimulated with the CFP and recombinant antigen preparations. Moreover, stimulation with the recombinant protein mixture yielded a significantly increased IFN-γ response in spleen cells taken from the Ub combination-vaccinated mice compared to naïve and vector controls. At 14 days after challenge, the overall IFN-γ production in splenocyte cultures from each group increased 5- to 50-fold (Fig. 2B). Most noticeably, the IFN-γ responses from splenocytes of mice vaccinated with the Ub combination were dramatically elevated.
FIG. 2.
IFN-γ in vitro secretion from splenocytes isolated from vaccinated mice that were aerogenically challenged with M. tuberculosis Erdman. Vaccinated mice were sacrificed 7 days (A) or 14 days (B) following aerogenic challenge, and splenocytes were restimulated in vitro using bone marrow macrophages primed with either the CFP mixture (gray boxes) or the recombinant protein mixture (black boxes). The amounts of IFN-γ that were secreted were determined by ELISA. Asterisks indicate that the amount of cytokine secreted is statistically significant compared to the vector control group, as determined by ANOVA (P < 0.05). Please note that the scales in panels A and B are different because the 14-day cytokine responses were substantially larger than the 7-day responses. Error bars, standard errors.
Bacterial growth in the lungs and spleens of vaccinated mice.
We evaluated whether the growth of tubercle bacilli was restricted in vaccinated mice by determining the number of tubercle bacilli in the lungs and spleens at 28, 63, and 112 days after they received a low-dose aerogenic challenge with M. tuberculosis Erdman (Table 2). In this experiment, the interval between vaccination and challenge was 5 weeks. The numbers of bacterial colonies in the lungs were significantly reduced 28 days postchallenge in all the vaccine groups relative to the naïve controls. Although a greater decrease in CFU values was detected in the BCG-vaccinated mice (−1.3 log10; 20-fold decrease), significant CFU reductions (P < 0.01) for the TPA (−0.8 log10; 6.5-fold) and Ub (−0.6 log10,; 4-fold) groups were also seen at this early time point. At 63 days postinfection, when the chronic infection had been established, significant (P < 0.01) decreases in bacterial CFU in the lung (relative to controls) were again detected in mice vaccinated with the TPA combination (−0.8 log10) and live BCG (−1.0 log10). Although the lung CFU values of the four test groups at day 112 were not statistically different from those of the naïve controls (primarily because of considerable variability), the trend again suggested that decreased numbers of tubercle bacilli were present in the TPA combination (−0.9 log10 reduction)- and the BCG (−0.8 log10 reduction)-vaccinated groups.
TABLE 2.
Vaccine-induced protection in mouse model of pulmonary tuberculosis
| Vaccine groupa | Mean log10 CFU of M. tuberculosis ±SDb
|
|||||
|---|---|---|---|---|---|---|
| 28 daysc
|
63 days
|
112 days
|
||||
| Lung | Spleen | Lung | Spleen | Lung | Spleen | |
| Naïve | 5.87 ± 0.16 | 5.36 ± 0.21 | 5.41 ± 0.22 | 4.98 ± 0.23 | 5.05 ± 0.20 | 4.07 ± 0.23 |
| TPA combo | 5.18 ± 0.22** | 4.20 ± 0.50** | 4.62 ± 0.20** | 4.16 ± 0.33** | 4.32 ± 0.64 | 2.78 ± 0.53* |
| Ub combo | 5.32 ± 0.13** | 4.75 ± 0.24** | 5.15 ± 0.20 | 4.64 ± 0.19 | 4.65 ± 0.27 | 4.06 ± 0.36 |
| BCG | 4.57 ± 0.19** | 4.18 ± 0.33** | 4.41 ± 0.03** | 3.72 ± 0.35** | 4.42 ± 0.39 | 3.51 ± 0.34 |
C57BL/6 mice were immunized once with BCG or injected three times with the DNA vaccine combinations.
Number of bacteria isolated from lungs and spleens following aerogenic challenge with ∼50 CFU of M. tuberculosis Erdman per mouse. The mice (n = 4 to 6) were challenged 5 weeks after the third DNA immunization. Statistical significance: *, P < 0.05 compared to naïve controls; **, P < 0.001.
Days after aerogenic challenge.
The spleen CFU data for these experimental groups were generally consistent with the lung results (Table 2). At 28 days after the challenge, significant decreases in the spleen bacterial burden (P < 0.01) relative to the naïve controls were detected for all three vaccinated groups. Reductions in bacterial dissemination to the spleen of about 90% (−1 log10) were demonstrated for the TPA combination- and the BCG-vaccinated mice, while approximately an 80% CFU decrease (−0.7 log10) was seen in animals vaccinated with the Ub combination. Substantial decreases in spleen bacterial numbers (P < 0.01) were also detected in the TPA and BCG groups at 63 days after challenge. At 112 days postchallenge, the spleen CFU were significantly decreased compared to those of controls only in the TPA combination group (−1.5 log10, P < 0.05).
The persistence of the memory response induced by the DNA vaccine combinations was further evaluated by aerogenically challenging vaccinated and control animals 16 weeks after the final immunization. Mycobacterial growth in the lungs and spleens was then assessed at 1, 2, and 4 weeks postinfection. As seen in Table 3, no differences in the lung CFU values were detected 1 week after the low-dose challenge. However, at 2 and 4 weeks postchallenge, the lung CFU values for all three vaccinated groups were significantly reduced (P < 0.05) compared to those for naïve and vector controls. While tubercle bacilli could not be detected in the spleen 7 days after the aerosol infection, significant decreases (P < 0.05) in spleen CFU were seen for the TPA combination and BCG groups at days 14 and 28. Overall, these 16-week challenge data are consistent with the 5-week results in that reductions in bacterial growth in the lungs were detected for all vaccine groups and significant decreases in the dissemination of bacteria to spleen were primarily observed in the TPA combination- and BCG-immunized animals. It is also important to emphasize that injection of the empty vector had no significant effect on bacterial growth in this experiment.
TABLE 3.
Vaccine-induced protection in mouse model of pulmonary tuberculosis
| Vaccine groupa | Mean log10 CFU of M. tuberculosis ±SDb
|
|||||
|---|---|---|---|---|---|---|
| 7 days
|
14 days
|
28 days
|
||||
| Lung | Spleenc | Lung | Spleen | Lung | Spleen | |
| Naïve | 4.07 ± 0.08 | ND | 5.82 ± 0.08 | 3.87 ± 0.27 | 5.80 ± 0.12 | 5.03 ± 0.06 |
| Vector | 3.83 ± 0.06 | ND | 5.84 ± 0.09 | 4.20 ± 0.10 | 5.69 ± 0.14 | 5.00 ± 0.19 |
| TPA combo | 3.84 ± 0.15 | ND | 5.18 ± 0.02* | 2.56 ± 0.14* | 5.20 ± 0.16* | 4.15 ± 0.03* |
| Ub combo | 4.08 ± 0.05 | ND | 5.26 ± 0.05* | 4.05 ± 0.25 | 5.43 ± 0.07* | 4.60 ± 0.15 |
| BCG | 3.89 ± 0.14 | ND | 4.48 ± 0.13* | 3.28 ± 0.25* | 4.78 ± 0.19* | 3.91 ± 0.09* |
C57BL/6 mice were immunized once with BCG or injected three times with the DNA vaccine combinations 16 weeks before the challenge.
Number of bacteria isolated from lungs and spleens following aerogenic challenge with ∼500 CFU of M. tuberculosis Erdman per mouse (n = 4 to 6). *, P < 0.05 compared to naive controls.
Dissemination of M. tuberculosis to the spleen cannot be detected until 10 to 14 days after a low-dose aerogenic challenge. Therefore, spleen homogenates were not plated (ND) at the day 7 time point.
Histopathologic analyses.
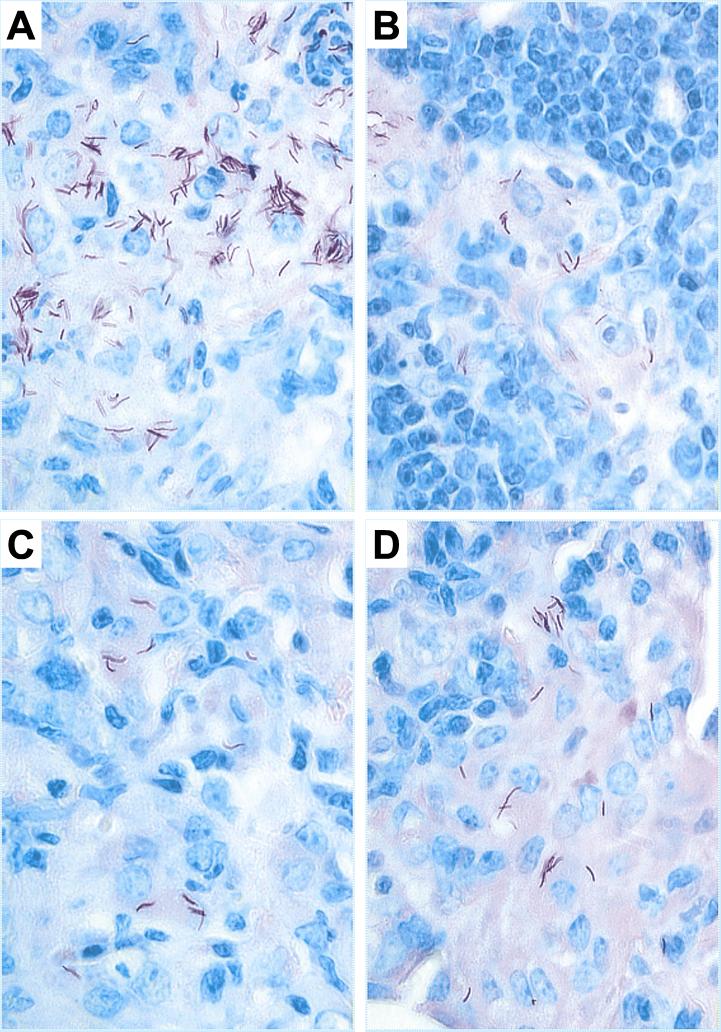
To assess the degree of lung pathology present after infection in vaccinated and control mice, lung tissues obtained from the same mice that were used to determine the bacterial CFU values were processed and then stained with either H&E stain or Ziehl-Neelsen acid-fast reagent (32). As shown in Fig. 3, the appearance of acid-fast-stained tissues recovered 28 days after challenge was consistent with the mycobacterial growth data. Numerous acid-fast bacilli were seen in the lungs of naïve mice while few acid-fast-stained organisms were apparent in lung tissues from vaccinated animals. Moreover, the H&E-stained sections showed a more severe pathology in unvaccinated mice than in immunized mice (Fig. 4). In the naïve controls examined at 63 days postchallenge, multiple coalescing granulomatous lesions involving a significant amount of lung tissue were apparent, suggesting that the functional capability of the lung had been severely compromised. The granulomas consisted mainly of macrophages and epitheloid cells surrounded by rims of lymphocytes. In contrast, the more limited overall lung inflammation and the decreased number and sizes of granulomatous lesions in lung tissues from vaccinated mice suggested a better protective response and a more benign outcome for the disease. In the vaccinated animals, extensive lymphocyte infiltration was seen 28 days after the challenge, which resulted in the earlier development of lymphocyte-rich granulomas (data not shown). At day 63 postchallenge (Fig. 4), the alveolar tissue appeared to be more intact in the vaccinated mice and only mild signs of alveolitis were apparent. Minimal amounts of consolidated tissue were detected at this time in the vaccinated animals. Taken together, these results suggest that a more effective mobilization of lymphocytes to the site of infection had occurred in mice vaccinated with the two DNA vaccine combinations, relative to controls, which resulted in better control of the bacterial growth, less inflammation, and an improved overall host response.
FIG. 3.
Acid-fast bacillus staining of lung tissues from vaccinated mice infected with M. tuberculosis. Control mice (A) or mice vaccinated with BCG (B), the TPA combination (C), or the Ub combination (D) were challenged with a low-dose aerogenic challenge (50 CFU) of M. tuberculosis Erdman. Twenty-eight days later the lower left lobe of each lung was isolated, fixed, and stained with the Ziehl-Neelsen reagent for analyzing acid-fast bacilli. Five mice per group were analyzed, and representative slides are shown (magnification, × 1,000).
FIG. 4.
Histopathological analysis of lungs from vaccinated mice obtained 63 days following M. tuberculosis challenge. The left lobe of the lung was removed, fixed, and stained with H&E stain. Representative slides are shown for the naïve controls (A) and mice vaccinated with either BCG (B), the TPA combination (C), or the Ub combination (D). Five mice per group and six sections per each lung were analyzed (magnification, ×40).
Survival of vaccinated mice aerogenically challenged with different doses of M. tuberculosis.
Our initial long-term studies investigated the effectiveness of our most-potent single DNA vaccine, the TPA-ESAT-6 construct, to extend postchallenge survival periods. When aerogenically challenged with 50 CFU of M. tuberculosis Erdman, an extension of the survival period (P < 0.05) was demonstrated for mice immunized with 200 μg (per dose) of the TPA-ESAT-6 vaccine (mean time to death [MTD], 341 ± 34 days) relative to mice given the vector only (MTD, 284 ± 16 days) (unless otherwise noted, values are means ± standard errors). However, since 9 of 10 mice administered the BCG vaccine survived the entire 400-day study period (MTD > 400 days), immunization with the single TPA-ESAT-6 construct was clearly not as effective as BCG vaccination.
Concurrent with the TPA-ESAT-6 experiments, the capacity of the DNA vaccine combinations to elicit sustained protective immunity was also assessed in long-term assays. In these studies, mice were exposed to one of three different aerogenic challenge doses. For the low-dose challenge experiment (30 to 50 CFU), the vaccinated mice efficiently controlled the infection with all the BCG- and TPA combination-immunized mice and seven of eight Ub combination-vaccinated mice surviving the 400-day study period. In contrast, only three of eight naïve controls were alive after 400 days (median time to death of 345 days). In this low-dose study, the difference in survival at 400 days between the TPA combination- and BCG-vaccinated groups and the controls was of borderline significance (P = 0.05).
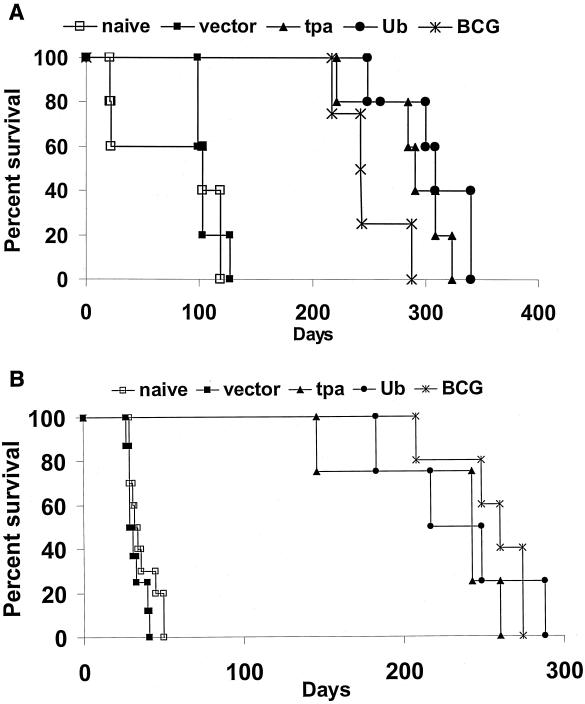
In a second study, the mice were infected with a moderate aerogenic challenge dose (500 CFU). At this challenge dose, highly significant differences (P < 0.001) in the survival periods were detected between the vaccinated and nonvaccinated mice (Fig. 5A). Although no statistically significant differences were noted between the vaccine groups, the mean survival times for the mice vaccinated with the TPA combination and the Ub combination exceeded the mean survival period for the BCG-immunized animals. Specifically, the MTD calculated for this moderate-dose challenge study were 76 ± 50 days for the naïve controls, 106 ± 11 days for the vector controls, 285 ± 38 days for mice immunized with the TPA combination, 307 ± 38 days for the Ub plasmid cocktail group, and 248 ± 30 days for the BCG vaccinated mice.
FIG. 5.
The survival of vaccinated and control mice following a moderate-dose (500 CFU) (A) or a high-dose (3,000 CFU) (B) aerogenic challenge with M. tuberculosis Erdman. The postinfection rates of survival of mice immunized with either the TPA combination vaccine (closed triangles), the Ub combination vaccine (closed circles), or BCG (×) were compared to the rates of survival of mice injected with the vector only (closed squares) and naïve mice (open squares). Five to ten animals per group were used in this analysis.
The high-dose challenge experiment (3,000 CFU) further illustrated the effectiveness of the DNA vaccine combinations (Fig. 5B). At this elevated aerogenic dose, the survival period was extended six- to sevenfold in the vaccinated mice relative to the controls. Highly significant differences were again noted (P < 0.001) between the MTD for the naïve mice (MTD = 36 ± 9 days) and vector controls (32 ± 5) compared to mice vaccinated with either the TPA combination (227 ± 54), the Ub combination (234 ± 44), or BCG (258 ± 27). However, again no differences were demonstrated in the survival periods between the three vaccine groups.
DISCUSSION
The goal of our studies has been to produce an effective combination of DNA vaccines against TB. We hypothesized that immunization protocols that employ multiple vaccines leading to the expression of multiple antigens would enhance the protective capacity of TB DNA vaccine preparations. In human immunodeficiency and malaria model systems, immune responses to DNA vaccination were amplified by using combinations of active constructs (17, 19). Our overall strategy has been to produce DNA constructs encoding genes of known TB immunogens, screen these vaccines for their capacity to induce protective immune responses in short term assays, and then combine the active constructs into a combination plasmid cocktail for testing in survival studies (12, 24, 28). Among the 10 TB DNA vaccines that we have identified in screening assays, eight encode culture filtrate proteins, the highly immunogenic antigens that are recognized early by the immune system (2). The other two components of the cocktail (MTB39 and 1818c) are members of the PE and PPE family of conserved proteins that appear to be expressed in macrophages and seem to induce strong cellular responses in patients (6, 13, 31). Overall, these combinations of DNA vaccines were formulated to generate a wide range of immune responses directed toward secreted and somatic antigens that are likely expressed in the early and chronic stages of infection (22).
In this study, we demonstrated that the combination vaccine strategy is sound. By showing reduced bacterial growth in vivo, decreased lung pathology, and increased cytokine production in vaccinated relative to control mice, we have proven that immunization with these DNA vaccine combinations induces substantial cellular immune responses. Most importantly, in separate experiments using three different challenge doses, the MTD for mice vaccinated with the DNA combinations were increased as much as sevenfold relative to naïve controls and were statistically indistinguishable from those for the BCG controls. Therefore, in survival studies using this mouse model, the protective responses evoked by the DNA cocktails and BCG were essentially equivalent. These results are of interest for several reasons. First, the TB nucleic acid preparations expressing single antigens that have been tested thus far have not generated protective immunity that was equivalent to the BCG protective response in long-term survival experiments. Our studies with immunogenic DNA vaccines expressing single antigens have yielded either a modest extension (ESAT-6) or no increase in the mean survival period (CFP-10 and TB10.4 vaccines [A. Li and S. L. Morris, unpublished results]) relative to unvaccinated controls. In contrast, the results of the combination vaccine survival studies are clearly promising and create renewed optimism regarding the potential usefulness of TB DNA vaccination strategies. Second, because of practical limitations, we administered only 20 μg of each individual vaccine, although our previous studies had indicated that injection of 20 μg of a single component was suboptimal (G. Delogu, unpublished data). Our failure to detect strong humoral responses to many of the individual components after vaccination with the TPA combination in this study supports the low-dose concern (data not shown). This apparent suboptimal dosage of individual vaccine components suggests a strategy to further increase the activity of the vaccine combinations and to possibly make them more potent than BCG. The construction of minigene (1) or multicistronic (23) plasmids in which several mycobacterial proteins or peptides are expressed from the same plasmid should permit increased dosage of individual antigens and possibly amplification of the protective immunity induced by DNA vaccine combinations. The recent report by McShane et al., which demonstrated that a DNA vaccine encoding both the ESAT-6 and MPT-63 antigens induced significant protective responses, validates this approach (27).
Interestingly, the TPA and the Ub DNA vaccine combinations that had been designed to generate different types of immune responses yielded virtually identical survival data in each of the experiments. Vaccines expressing TPA-fused proteins were developed to direct the transgene TB antigens to the secretory pathway for enhanced secretion by the host cells. An unexpected consequence of creating the TPA vectors was the increased expression (in the context of DNA vaccination) of the TPA fusion proteins relative to native mycobacterial antigens. In general, our TPA constructs express relatively high levels of protein that appear to be effectively secreted from the cells (12, 24). In contrast, the Ub fusion vaccines were designed to accelerate turnover of the transgene antigen and increase the variety and number of peptides available for major histocompatibility complex (MHC) binding. Our previous studies with the UbGR constructs illustrated that these vaccines can elicit significant cell-mediated responses in the absence of a humoral response (12). However, despite the dramatic differences in the intracellular turnover observed for the TPA- and Ub-conjugated antigens (as assessed by in vitro transfection of rhabdomyosarcoma cells), the cellular immune responses generated by the TPA and Ub vaccine combinations were usually not significantly different. In fact, comparable levels of IFN-γ have often been detected in ex vivo analyses using primed splenocytes from the TPA and Ub combination-vaccinated animals. Therefore, the simplest explanation for the nearly equivalent survival periods for the two vaccine cocktails is that the TPA and Ub constructs evoke similar levels of antimycobacterial cellular immunity and act to enhance survival via the same primary protective mechanism.
Among the three possible antigen presentation events postulated to occur after DNA vaccination (direct priming by somatic cells, direct priming by antigen-presenting cells, or cross-priming of antigen-presenting cells), activation by cross-priming seems to be the most likely immune mechanism occurring following intramuscular vaccination that could be shared by the TPA and Ub vaccines (10, 16, 18). It is well known that CD4 T-cell stimulation can result from endocytosis of exogenous peptides or proteins followed by antigenic processing via MHC class II pathways (37). Recent studies have also demonstrated that exogenous proteins or peptides, possibly complexed to heat shock proteins, can be taken up by antigen processing cells, processed through the MHC class I pathway, and ultimately stimulate naïve CD8 cells (4, 29). Thus, via cross-priming mechanisms, both secreted fusion proteins expressed from TPA plasmids or peptides released from cells transfected with the Ub constructs could induce both CD4 and CD8 T-cell populations. The importance of CD4 stimulation by TB DNA vaccines was recently demonstrated when CD4 knockout (KO) mice immunized with an antigen 85 TB DNA vaccine could not be protected against tuberculous challenge (14). In the same study, however, CD8 KO mice were protected by DNA vaccination. Thus, the role of CD8 cells in DNA vaccine-induced protective immunity is less certain. Studies in CD4 and CD8 KO mice are currently ongoing in our laboratory to determine which T-cell subsets are induced by the TPA and Ub DNA vaccine combinations to generate protective responses against tuberculous infection. A greater understanding of the immune mechanisms responsible for producing protective immunity in response to DNA vaccination should facilitate the creation of new and improved TB DNA vaccination strategies.
Defining the immune responses that are required for protection against TB, however, remains a challenging task. Two results for this study illustrate the difficulties associated with identifying the protective immune mechanisms against TB. First, our bacterial proliferation data clearly indicate that the bacterial burden in relevant organs does not inversely correlate with survival in this mouse model. While the Ub DNA vaccine combination was less effective than BCG in reducing bacterial CFU in the lungs and spleens of vaccinated mice, the MTD for the Ub vaccine- and BCG-immunized animals were essentially equivalent. Similarly, Baldwin et al. showed that the decrease in the number of bacterial CFU in the lung was not a direct correlate of protection, because two TB vaccines they tested prolonged survival without reducing short-term bacterial burdens in the lung (3). These data suggest that although the reduction in bacterial growth in relevant organs is one criterion for evaluating the activity of TB vaccines, other immune parameters such as pathology are likely better measures of TB vaccine effectiveness. Second, our IFN-γ induction data confirm earlier observations which indicated that the IFN-γ response is not a direct correlate of protective immunity (15, 24). Interestingly, while the IFN-γ responses detected in splenocytes from Ub DNA-, TPA DNA-, and BCG-vaccinated mice were different, the MTD for these three vaccine groups were statistically equivalent in three separate survival studies. Therefore, although IFN-γ is obviously a key cytokine in the control of M. tuberculosis infections and is almost certainly required for an effective protective response against the pathogen, IFN-γ production alone is likely insufficient to retard proliferation of the highly virulent tubercle bacilli.
In sum, we have shown that two different DNA vaccine combinations induce sustained protective immune responses in a mouse model of pulmonary TB. These promising results indicate that further evaluation and characterization of these plasmid cocktails are warranted. Moreover, the data suggest that further optimization of the DNA constructs and of the dosing schedules may lead to nucleic acid vaccine preparations that are more potent than the current vaccine, M. bovis BCG.
Editor: J. D. Clements
REFERENCES
- 1.An, L. L., F. Rodriguez, S. Harkins, J. Zhang, and J. L. Whitton. 2000. Quantitative and qualitative analyses of the immune responses induced by a multivalent minigene DNA vaccine. Vaccine 18: 2132–2141. [DOI] [PubMed] [Google Scholar]
- 2.Andersen, P. 1997. Host responses and antigens involved in protective immunity to Mycobacterium tuberculosis. Scand. J. Immunol. 45:115–131. [DOI] [PubMed] [Google Scholar]
- 3.Baldwin, S. L., C. D’Souza, A. D. Roberts, B. P. Kelly, A. A. Frank, M. A. Lui, J. B. Ulmer, K. Huygen, D. M. McMurray, and I. M. Orme. 1998. Evaluation of new vaccines in the mouse and guinea pig model of tuberculosis. Infect. Immun. 66:2951–2959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Castellino, F., P. E. Boucher, K. Eichelberg, M. Mayhew, J. E. Rothman, A. N. Houghton, and R. N. Germain. 2000. Receptor-mediated uptake of antigen/heat shock protein complexes results in major histocompatibility complex class I antigen presentation via two distinct processing pathways. J. Exp. Med. 191:1957–1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Colditz, G. A., T. F. Brewer, C. S. Berkey, M. E. Wilson, E. Burdick, H. V. Fineberg, and F. Mosteller. 1994. Efficacy of BCG vaccine in the prevention of tuberculosis. Meta-analysis of the published literature. JAMA 271:698–702. [PubMed] [Google Scholar]
- 6.Cole, S. T., R. Brosch, J. Parkhill, T. Garnier, C. Churcher, D. Harris, S. V. Gordon, K. Eiglmeier, S. Gas, C. E. Barry III, F. Tekaia, K. Badcock, D. Basham, D. Brown, T. Chillingworth, R. Connor, R. Davies, K. Devlin, T. Feltwell, S. Gentles, N. Hamlin, S. Holroyd, T. Hornsby, K. Jagels, and B. G. Barrell. 1998. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393:537–544. [DOI] [PubMed] [Google Scholar]
- 7.Coler, R. N., Y. A. Skeiky, T. Vedvick, T. Bement, P. Ovendale, A. Campos-Neto, M. R. Alderson, and S. G. Reed. 1998. Molecular cloning and immunologic reactivity of a novel low molecular mass antigen of Mycobacterium tuberculosis. J. Immunol. 161:2356–2364. [PubMed] [Google Scholar]
- 8.Collins, F. M. 1985. Protection to mice afforded by BCG vaccines against an aerogenic challenge by three mycobacteria of decreasing virulence. Tubercle 66:267–276. [DOI] [PubMed] [Google Scholar]
- 9.Collins, F. M., and G. B. Mackaness. 1970. The relationship of delayed hypersensitivity to acquired antituberculous immunity. I. Tuberculin sensitivity and resistance to reinfection in BCG-vaccinated mice. Cell. Immunol. 1:253–265. [DOI] [PubMed] [Google Scholar]
- 10.Corr, M., A. von Damm, D. J. Lee, and H. Tighe. 1999. In vivo priming by DNA injection occurs predominantly by antigen transfer. J. Immunol. 163:4721–4727. [PubMed] [Google Scholar]
- 11.Delogu, G., and M. J. Brennan. 2001. Comparative immune response to PE and PE_PGRS antigens of Mycobacterium tuberculosis. Infect. Immun. 69:5606–5611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Delogu, G., A. Howard, F. M. Collins, and S. L. Morris. 2000. DNA vaccination against tuberculosis: expression of a ubiquitin-conjugated tuberculosis protein enhances antimycobacterial immunity. Infect. Immun. 68:3097–3102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dillon, D. C., M. R. Alderson, C. H. Day, D. M. Lewinsohn, R. Coler, T. Bement, A. Campos-Neto, Y. A. Skeiky, I. M. Orme, A. Roberts, S. Steen, W. Dalemans, R. Badaro, and S. G. Reed. 1999. Molecular characterization and human T-cell responses to a member of a novel Mycobacterium tuberculosis mtb39 gene family. Infect. Immun. 67:2941–2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.D’Souza, S., O. Denis, T. Scorza, F. Nzabintwali, H. Verschueren, and K. Huygen. 2000. CD4+ T cells contain Mycobacterium tuberculosis infection in the absence of CD8+ T cells in mice vaccinated with DNA encoding Ag85A. Eur. J. Immunol. 30:2455–2459. [DOI] [PubMed] [Google Scholar]
- 15.Flynn, J. L., and J. Chan. 2001. Immunology of tuberculosis. Annu. Rev. Immunol. 19:93–129. [DOI] [PubMed] [Google Scholar]
- 16.Fu, T. M., J. B. Ulmer, M. J. Caulfield, R. R. Deck, A. Friedman, S. Wang, X. Liu, J. J. Donnelly, and M. A. Liu. 1997. Priming of cytotoxic T lymphocytes by DNA vaccines: requirement for professional antigen presenting cells and evidence for antigen transfer from myocytes. Mol. Med. 3:362–371. [PMC free article] [PubMed] [Google Scholar]
- 17.Grifantini, R., O. Finco, E. Bartolini, M. Draghi, G. Del Giudice, C. Kocken, A. Thomas, S. Abrignani, and G. Grandi. 1998. Multi-plasmid DNA vaccination avoids antigenic competition and enhances immunogenicity of a poorly immunogenic plasmid. Eur. J. Immunol. 28:1225–1232. [DOI] [PubMed] [Google Scholar]
- 18.Gurunathan, S., D. M. Klinman, and R. A. Seder. 2000. DNA vaccines: immunology, application, and optimization. Annu. Rev. Immunol. 18:927–974. [DOI] [PubMed] [Google Scholar]
- 19.Hinkula, J., P. Lundholm, and B. Wahren. 1997. Nucleic acid vaccination with HIV regulatory genes: a combination of HIV-1 genes in separate plasmids induces strong immune responses. Vaccine 15:874–878. [DOI] [PubMed] [Google Scholar]
- 20.Huygen, K., J. Content, O. Denis, D. L. Montgomery, A. M. Yawman, R. R. Deck, C. M. DeWitt, I. M. Orme, S. Baldwin, C. D’Souza, A. Drowart, E. Lozes, P. Vandenbussche, J. P. Van Vooren, M. A. Liu, and J. B. Ulmer. 1996. Immunogenicity and protective efficacy of a tuberculosis DNA vaccine. Nat. Med. 2:893–898. [DOI] [PubMed] [Google Scholar]
- 21.Kamath, A. T., C. G. Feng, M. Macdonald, H. Briscoe, and W. J. Britton. 1999. Differential protective efficacy of DNA vaccines expressing secreted proteins of Mycobacterium tuberculosis. Infect. Immun. 67:1702–1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kaufmann, S. H., and P. Andersen. 1998. Immunity to mycobacteria with emphasis on tuberculosis: implications for rational design of an effective tuberculosis vaccine. Chem. Immunol. 70:21–59. [DOI] [PubMed] [Google Scholar]
- 23.Lee, S. W., J. H. Cho, and Y. C. Sung. 1998. Optimal induction of hepatitis C virus envelope-specific immunity by bicistronic plasmid DNA inoculation with the granulocyte-macrophage colony-stimulating factor gene. J. Virol. 72:8430–8436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li, Z., A. Howard, C. Kelley, G. Delogu, F. Collins, and S. Morris. 1999. Immunogenicity of DNA vaccines expressing tuberculosis proteins fused to tissue plasminogen activator signal sequences. Infect. Immun. 67:4780–4786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li, Z., C. Kelley, F. Collins, D. Rouse, and S. Morris. 1998. Expression of katG in Mycobacterium tuberculosis is associated with its growth and persistence in mice and guinea pigs. J. Infect. Dis. 177:1030–1035. [DOI] [PubMed] [Google Scholar]
- 26.Manca, C., K. Lyashchenko, H. G. Wiker, D. Usai, R. Colangeli, and M. L. Gennaro. 1997. Molecular cloning, purification, and serological characterization of MPT63, a novel antigen secreted by Mycobacterium tuberculosis. Infect. Immun. 65:16–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McShane, H., R. Brookes, S. C. Gilbert, and A. V. Hill. 2001. Enhanced immunogenicity of CD4+ T-cell responses and protective efficacy of a DNA-modified vaccinia virus Ankara prime-boost vaccination regimen for murine tuberculosis. Infect. Immun. 69:681–686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Morris, S., C. Kelley, A. Howard, Z. Li, and F. Collins. 2000. The immunogenicity of single and combination DNA vaccines against tuberculosis. Vaccine 18:2155–2163. [DOI] [PubMed] [Google Scholar]
- 29.Norbury, C. C., M. F. Princiotta, I. Bacik, R. R. Brutkiewicz, P. Wood, T. Elliott, J. R. Bennink, and J. W. Yewdell. 2001. Multiple antigen-specific processing pathways for activating naive CD8+ T cells in vivo. J. Immunol. 166:4355–4362. [DOI] [PubMed] [Google Scholar]
- 30.Orme, I. M. 1999. New vaccines against tuberculosis. The status of current research. Infect. Dis. Clin. N. Am. 13:169–185. [DOI] [PubMed] [Google Scholar]
- 31.Ramakrishnan, L., N. A. Federspiel, and S. Falkow. 2000. Granuloma-specific expression of mycobacterium virulence proteins from the glycine-rich PE-PGRS family. Science 288:1436–1439. [DOI] [PubMed] [Google Scholar]
- 32.Rhoades, E. R., A. A. Frank, and I. M. Orme. 1997. Progression of chronic pulmonary tuberculosis in mice aerogenically infected with virulent Mycobacterium tuberculosis. Tuber. Lung Dis. 78:57–66. [DOI] [PubMed] [Google Scholar]
- 33.Rodriguez, F., J. Zhang, and J. L. Whitton. 1997. DNA immunization: ubiquitination of a viral protein enhances cytotoxic T-lymphocyte induction and antiviral protection but abrogates antibody induction. J. Virol. 71:8497–8503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tascon, R. E., M. J. Colston, S. Ragno, E. Stavropoulos, D. Gregory, and D. B. Lowrie. 1996. Vaccination against tuberculosis by DNA injection. Nat. Med. 2:888–892. [DOI] [PubMed] [Google Scholar]
- 35.Ulmer, J. B., M. A. Liu, D. L. Montgomery, A. M. Yawman, R. R. Deck, C. M. DeWitt, J. Content, and K. Huygen. 1997. Expression and immunogenicity of Mycobacterium tuberculosis antigen 85 by DNA vaccination. Vaccine 15:792–794. [DOI] [PubMed] [Google Scholar]
- 36.Vordermeier, H. M., P. J. Cockle, A. O. Whelan, S. Rhodes, M. A. Chambers, D. Clifford, K. Huygen, R. Tascon, D. Lowrie, M. J. Colston, and R. G. Hewinson. 2000. Effective DNA vaccination of cattle with the mycobacterial antigens MPB83 and MPB70 does not compromise the specificity of the comparative intradermal tuberculin skin test. Vaccine 19:1246–1255. [DOI] [PubMed] [Google Scholar]
- 37.Watts, C. 1997. Capture and processing of exogenous antigens for presentation on MHC molecules. Annu. Rev. Immunol. 15:821–850. [DOI] [PubMed] [Google Scholar]
- 38.Webb, J. R., T. S. Vedvick, M. R. Alderson, J. A. Guderian, S. S. Jen, P. J. Ovendale, S. M. Johnson, S. G. Reed, and Y. A. Skeiky. 1998. Molecular cloning, expression, and immunogenicity of MTB12, a novel low-molecular-weight antigen secreted by Mycobacterium tuberculosis. Infect. Immun. 66:4208–4214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.World Health Organization. 1999. Global tuberculosis control. W. H. O. Report 1999. World Health Organization, Geneva, Switzerland.
- 40.Wu, Y., and T. J. Kipps. 1997. Deoxyribonucleic acid vaccines encoding antigens with rapid proteasome-dependent degradation are highly efficient inducers of cytolytic T lymphocytes. J. Immunol. 159:6037–6043. [PubMed] [Google Scholar]