Abstract
Antibodies to the lipopolysaccharide O antigen of Pseudomonas aeruginosa mediate high-level immunity, but protective epitopes have proven to be poorly immunogenic, while nonprotective or minimally protective O-antigen epitopes often elicit the best immune responses. With the goal of developing a broadly protective P. aeruginosa vaccine, we used a gene replacement system based on the Flp recombinase to construct an unmarked aroA deletion mutant of the P. aeruginosa serogroup O2/O5 strain PAO1. The resultant aroA deletion mutant of PAO1 is designated PAO1ΔaroA. The aroA deletion was confirmed by both PCR and failure of the mutant to grow on minimal media lacking aromatic amino acids. When evaluated for safety and immunogenicity in mice, PAO1ΔaroA could be applied either intranasally or intraperitoneally at doses up to 5 × 109 CFU per mouse without adverse effects. No dissemination of PAO1ΔaroA to blood, liver, or spleen was detected after intranasal application, and histological evidence of pneumonia was minimal. Intranasal immunization of mice and rabbits elicited high titers of immunoglobulin G to whole bacterial cells and to heat-stable bacterial antigens of all seven prototypic P. aeruginosa serogroup O2/O5 strains. The mouse antisera mediated potent phagocytic killing of most of the prototypic serogroup O2/O5 strains, while the rabbit antisera mediated phagocytic killing of several serogroup-heterologous strains in addition to killing all O2/O5 strains. This live, attenuated P. aeruginosa strain PAO1ΔaroA appears to be safe for potential use as an intranasal vaccine and elicits high titers of opsonic antibodies against multiple strains of the P. aeruginosa O2/O5 serogroup.
Lipopolysaccharide (LPS)-smooth strains of Pseudomonas aeruginosa are significant pathogens for immunocompromised (11) and critically ill (52) patients and are the initial colonizing strains in children with cystic fibrosis (3). In the setting of nosocomial pneumonia, P. aeruginosa has a strikingly high attributable mortality (51). The prevalence and importance of P. aeruginosa in human infections, coupled with the increasingly frequent presence of antibiotic resistance in clinical isolates (10, 16), have generated a pressing need for an effective vaccine.
For many years, it has been clear that high-level immunity to P. aeruginosa infections can be mediated by antibodies to the LPS O antigen (also known as O side chain) (13). However, protective epitopes have proven to be poorly immunogenic, while nonprotective or minimally protective O-antigen epitopes often elicit the best immune responses. P. aeruginosa is currently classified into 20 serogroups based on LPS O-antigen determinants, with most serogroups possessing subtype strains having subtle variations in the O antigen. Thus, there are over 30 subtypes based on LPS O-antigen chemical structure (27). Although O-antigen-based vaccines can elicit antibodies that are protective in animal models, this protection is generally seen only when the strains used to isolate the vaccine antigen are also used in the challenge studies (4, 5, 47, 48). Broad-based protection against other strains, even subtypes within the same serogroup, is not reliably generated (18, 19). With these observations in mind, an O-antigen-based vaccine would need to be more than 20-valent (probably more than 30-valent, due to additional subtypes). However, previous efforts to make even a divalent vaccine have been unsuccessful. When related O antigens (in the form of purified high-molecular-weight O-polysaccharide) from subtype strains within serogroup O2/O5 were combined, the immune response to each individual component was diminished (19). Furthermore, Cryz and colleagues have shown that an octavalent O-antigen-toxin A conjugate vaccine engendered opsonic antibody responses only against strains used to manufacture the vaccine and did not protect humans at risk for nosocomial P. aeruginosa infections after passive transfer of immunoglobulin G (IgG) isolated from vaccinated individuals (5, 6, 8).
The inability to harness the protective efficacy of LPS O-antigen-elicited antibodies into an effective, broadly protective vaccine suggests an important role for cellular immunity in the control of P. aeruginosa infections, as does recent evidence (14, 21, 46) that P. aeruginosa readily enters lung and corneal epithelial cells during infection. This cellular invasion is mediated by interactions with the cystic fibrosis transmembrane conductance regulator (44, 45, 60) and appears to result in apoptosis of infected cells (12, 20). We hypothesized that live, attenuated P. aeruginosa vaccine strains could exploit this intracellular phase in the pathogenesis of P. aeruginosa infections and elicit a broadly protective immune response.
We used a gene replacement system based on the Flp recombinase to construct an unmarked aroA deletion mutant of the P. aeruginosa serogroup O2/O5 strain PAO1. This live, attenuated strain was used to immunize mice and rabbits via the intranasal (i.n.) route, and the antisera were assessed by enzyme-linked immunosorbent assay (ELISA) and for opsonic killing activity. We have chosen to focus on i.n. immunization because it has been shown to be highly effective with a wide variety of pathogens in stimulating both local and systemic immunity (17, 59) as well as immunity at distant mucosal sites (26). The results show that intranasal immunization of mice and rabbits with this live, attenuated P. aeruginosa vaccine elicits opsonic antibodies against multiple strains of the P. aeruginosa serogroup O2/O5 and, in rabbits, against several serogroup-heterologous strains as well, indicating the significant potential of using such live, attenuated strains for vaccination against LPS-smooth strains of P. aeruginosa.
MATERIALS AND METHODS
Bacterial strains.
The bacterial strains and plasmids used in these experiments, along with their phenotypes and sources, are listed in Table 1.
TABLE 1.
Strains and plasmids used in this study
| Strain or plasmid | Descriptiona | Reference or source |
|---|---|---|
| P. aeruginosa | ||
| PAO1 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitopes O2a, O2d | M. Vasil |
| PAO1ΔaroA | aroA deletion mutant of PAO1, LPS smooth, serogroup O2/O5, subtype epitopes O2a,O2d | This study |
| PAO1ΔaroA (pUCP18) | PAO1ΔaroA containing the empty vector pUCP18, CbR | This study |
| PAO1ΔaroA (pMB1) | PAO1ΔaroA containing the intact aroA gene on plasmid pMB1, CbR | This study |
| AK44 | LPS-defective derivative of PAO1 (absent O antigen, complete outer core) | 30 |
| AK1012 | LPS-defective derivative of PAO1 (absent O antigen, incomplete core) | 25 |
| PAC1R | Wild-type strain, LPS smooth, serogroup O3 | 53 |
| PAC557 | LPS-rough derivative of PAC1R (absent O antigen, complete outer core) | 53 |
| 170003 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitopes O2a, O2b | 19 |
| IATS O16 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitopes O2a, O2b, O2e | 19 |
| Fisher IT-3 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitopes O2a, O2c | 19 |
| 170006 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitopes O2a, O2d, O2e | 19 |
| 170007 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitopes O2a, O2d, O2f | 19 |
| Fisher IT-7 | Wild-type strain, LPS smooth, serogroup O2/O5, subtype epitope O2a | 19 |
| Fisher IT-4 | Clinical isolate (bacteremia) designated Rhodes, LPS smooth, serogroup O1 | 50 |
| 170001 | Wild-type strain, LPS smooth, serogroup O3, subtype epitopes O3a, O3b | 27, 28 |
| IATS O3 | Wild-type strain, LPS smooth, serogroup O3, subtype epitopes O3a, O3b, O3c | 27, 28 |
| 6294 | Clinical isolate (corneal infection), LPS smooth, noncytotoxic, serogroup O6 | BPEIb; 15, 50 |
| 6073 | Clinical isolate (corneal infection), LPS smooth, cytotoxic, serogroup O11 | BPEI; 15, 50 |
| 6077 | Clinical isolate (corneal infection), LPS smooth, cytotoxic, serogroup O11 | BPEI; 15, 50 |
| 6206 | Clinical isolate (corneal infection), LPS smooth, cytotoxic, serogroup O11 | BPEI; 15, 50 |
| E. coli | ||
| HB101 | supE44 hsdS20(r−Bm−B) recA13 ara-14 proA2 lacY1 galK2 rpsL20 xyl-5 mtl-1, | 2 |
| S17-1 | thi pro hsdR recA RP4-2-Tc::Mu Km::Tn7 | 57 |
| SM10 | thi-1, thr leu tonA lacY supE recA::RP4-2-Tc::Mu | 7 |
| Plasmids | ||
| pMB1 | pUCP18-based plasmid containing the intact aroA gene, ApR | This study |
| pUCP18 | Broad host range shuttle vector, ApR | 55 |
| pMB4 | Derivative of pMB1 containing the aroA gene disrupted by the aacC1 cassette from pPS856, ApR/GmR | This study |
| pEX100T | oriT+sacB+ gene-replacement vector | 23, 56 |
| pMB11 | pEX 100T-based derivative of pMB4 containing the sacB sucrose-sensitivity gene, CbR/GmR/sucS | This study; 23 |
| pPS856 | Plasmid containing the aacC1 gentamicin-resistance cassette flanked by the Flp-recombinase target sequences, ApR/GmR | 23 |
| pFLP2 | Plasmid containing the Flp recombinase, ApR | 23 |
Abbreviations: Ap, ampicillin; Gm, gentamicin; Cb, carbenicillin; Km, kanamycin; suc, sucrose; Tc, tetracycline; superscript R, resistant; superscript S, sensitive.
BPEI, Bascom-Palmer Eye Institute, Miami, Fla.
Construction of aroA deletion mutants.
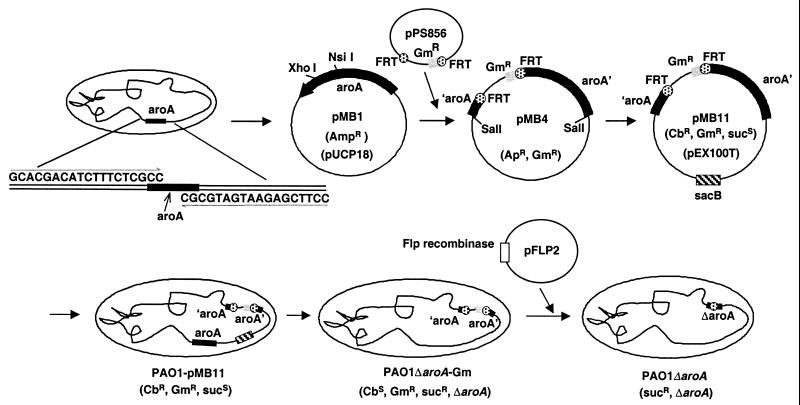
The aroA gene of P. aeruginosa PAO1 was recognized to be present from the Pseudomonas Genome Project (http://www.pseudomonas.com). As depicted in Fig. 1, primers aroA-F2 (5′-GCACGACATCTTTCTCGCC-3′) and aroA-R2 (5′-CCTTCGAGAATGATGCGC-3′) were used to amplify DNA from PAO1 by PCR. The resultant 1.7-kb PCR product was ligated into plasmid pUCP18, which was then used to construct an integration-excision vector based on the gene replacement system of Hoang et al. and Schweizer (23, 55), which initially results in a P. aeruginosa strain having a deletion for the aroA gene with an inserted gentamicin resistance marker (aacC1, the gentamicin acetyltransferase 3-1 gene) flanked by the Flp recombinase target (FRT) sequences. Briefly, the plasmid pMB11 was generated by insertional inactivation of the aroA gene with the FRT-aacC1-FRT cassette followed by ligation into the gene replacement vector pEX100T (55) (a gift of H. P. Schweizer). The pEX100T vector carries a sacB sucrose sensitivity gene, confers carbenicillin resistance, and requires a recombination event to be replicated. Plasmid pMB11 was transferred to P. aeruginosa PAO1 from Escherichia coli S17-1 (57) by biparental mating. Recipient cells were plated on cetrimide agar containing aromatic amino acids (50 μg/ml) with gentamicin (250 μg/ml) to select for the acquisition of gentamicin resistance encoded by aacC1. Colonies appearing on this medium were then replated on the same medium containing carbenicillin (300 μg/ml) to confirm that the entire plasmid was inserted into the chromosome. Surviving colonies were then plated on L agar containing aromatic amino acids, gentamicin, and 5% sucrose, which induces loss of the pEX100T-encoded sacB gene. Colonies that grew on this medium were tested for carbenicillin sensitivity to ensure loss of the plasmid and were assessed for auxotrophy for aromatic amino acids by using MSM (a minimal salts medium containing 0.5% glucose [42]) and MSM supplemented with aromatic amino acids. The resultant strain, PAO1ΔaroA-Gm, was confirmed by PCR with primers aroA-F3 (5′-CCTGATTTATCTGGCCCAGC-3′) and aroA-R3 (5′-GCGCTCAACTTGTGCCCGG-3′).
FIG. 1.
Construction of unmarked P. aeruginosa PAO1 with deletion of aroA. See Table 1 for a description of plasmids. Ap, ampicillin; Gm, gentamicin; Cb, carbenicillin; suc, sucrose.
To remove the gentamicin resistance marker and delete the aroA gene, plasmid pFLP2, which contains the Flp recombinase, was transferred from E. coli SM10 to PAO1ΔaroA-Gm. Colonies were grown on MSM containing aromatic amino acids and carbenicillin (500 μg/ml) to select for pFLP2 (23) in P. aeruginosa. Resultant colonies were screened for gentamicin sensitivity (to confirm the deletion of the gentamicin resistance cassette); the gentamicin-sensitive colonies were then plated onto MSM containing aromatic amino acids and 5% sucrose to select for loss of the pFLP2 plasmid. Loss of the plasmid and deletion of aroA were confirmed by PCR and sequencing and by failure to grow on MSM in the absence of aromatic amino acids. The resultant unmarked deletion mutant of PAO1 is referred to as PAO1ΔaroA. Auxotrophism due to deletion of aroA was confirmed by transferring in either a control plasmid (pUCP18) or a plasmid (pMB1) containing the intact aroA gene by electroporation.
Preparation of bacterial inocula.
Frozen bacterial stocks were plated and grown overnight on tryptic soy agar at 37°C. For immunization, bacteria were suspended in either normal saline or phosphate-buffered saline (PBS). Concentrations were adjusted spectrophotometrically and confirmed by viable counts after serial dilution and plating on tryptic soy agar. For i.n. immunization studies using heat-killed bacteria, the inoculum was prepared in PBS as described above, heated at 60°C for 1 h, cooled, resuspended by vortexing, and then used for i.n. application without washing.
Immunization of mice and rabbits.
Six- to 8-week-old female C3H/HeN or BALB/c mice (Harlan Sprague-Dawley Farms, Chicago, Ill.) were housed under virus-free conditions. Before immunization, mice were first anesthetized with 0.2 ml of a mixture of ketamine (6.7 mg/ml) and xylazine (1.3 mg/ml) in 0.9% saline injected intraperitoneally (i.p.). Immunization consisted of placing 10 μl of the bacterial inoculum on each nare (total, 20 μl per mouse). Escalating doses of 1 × 108, 5 × 108, and 109 CFU were administered at weekly intervals. New Zealand White rabbits (Millbrook Breeding Labs, Amherst, Mass.) were immunized on a similar schedule, followed by repeated intranasal boosting with doses of 109 CFU every 2 to 4 weeks, all using inocula of 100 μl (50 μl per nare). Rabbits were anesthetized with 2 ml of a mixture of atropine (0.4 mg), ketamine (80 mg), and xylazine (10 mg) injected subcutaneously prior to each immunization. Mice and rabbits were immunized with E. coli HB101 as a control, using identical schedules and doses. All animal experiments complied with institutional and federal guidelines regarding the use of animals in research.
Determination of internalized bacteria in infected lungs.
Using methods previously described (1), adult BALB/c mice were sacrificed at various times after i.n. inoculation. After removal of the lungs in a sterile fashion, single-cell suspensions were obtained by passage through wire screens. Total bacterial numbers were determined by lysis of cells in Triton X-100 followed by serial dilution and plating. The numbers of internalized bacteria were determined by incubation of the lung suspensions in gentamicin at 37°C for 1 h followed by washing, lysing with Triton X-100, diluting, and plating.
Histological analysis of lungs after i.n. application.
Adult C3H/HeN mice were sacrificed at 0.5, 1.5, 3, 6, 24, and 48 h after i.n. inoculation with either PAO1 or PAO1ΔaroA. The lungs were immediately instilled with 1 ml of PBS containing 1% paraformaldehyde by means of a catheter placed directly into the trachea after exposure with a midline neck incision. The lungs were removed, fixed in PBS with 1% paraformaldehyde for 1 h at room temperature, and then placed in 70% ethanol in water at 4°C overnight prior to paraffin embedding. Sections were stained with hematoxylin and eosin.
ELISA and opsonophagocytic assays.
ELISAs were performed by standard methods as described previously (18). To assess heat-stable antigens, bacteria were heated at 95°C for 45 min in PBS, followed by centrifugation for 30 min at 15,000 × g in a microcentrifuge. The pellet was resuspended in 0.01 M sodium phosphate buffer (pH 7.0) and used to coat microtiter plates for ELISA. Opsonophagocytic assays also used published methods (43), with the only differences being that infant rabbit serum (Accurate Chemical, Westbury, N.Y.) was used as the complement source and was not adsorbed with any organisms. In addition, bacteria were grown in tryptic soy broth containing 1% glycerol as a supplemental carbon source. Negative controls were antisera from mice or rabbits immunized i.n. with E. coli HB101. Tubes with PAO1ΔaroA antisera but without polymorphonuclear leukocytes (PMNs) served as additional negative controls to help distinguish killing from agglutination. The positive control antisera were 1:4 dilutions of sera from rabbits immunized intravenously with heat-killed whole bacteria of the homologous serogroup (rabbit antiserum to the heat-killed Fisher IT-7 strain was the positive control for serogroup O2/O5 strains). Antisera were adsorbed by incubating antisera with lyophilized bacteria (5 mg/ml) for 1 h at 4°C, removing the bacteria by centrifugation at 15,000 × g, and then filtering the supernatant through a 0.2-μm-pore-size filter. Antisera were each adsorbed twice by the above procedure. For all assays, mouse sera were collected and pooled (four to five C3H/HeN mice per immunization group) 3 weeks after the third immunization. Rabbit sera were collected 1 week after the seventh immunization.
Statistical analyses.
ELISA titers were calculated by linear regression analysis of duplicate or triplicate measurements of adjusted optical density values (with optical density of normal mouse or rabbit sera subtracted) versus the log10 of the serum dilutions. The x-intercept defined the endpoint titer. Calculation of 95% confidence intervals for titers was done using the formula VT = (T2VB − 2 TCAB + VA)/B2, where T is the titer (log10); VT is the variance of the titer; VB is the variance of the slope, B, of the regression analysis; CAB is the covariance of the estimated intercept and slope; and VA is the variance of the intercept coefficient, A. VA, VB, and CAB were obtained from the variance-covariance matrix of parameter estimates using SPSS statistical software (SPSS, Chicago, Ill.). The 95% confidence intervals were then calculated as T ± 1.96(VT)1/2. The significance of the percentage of organisms killed in the opsonophagocytic assay was determined by analysis of variance (ANOVA) with Fisher's protected least significant difference (PLSD) using Statview (SAS Institute, Cary, N.C.) with comparison to the E. coli HB101 control antisera. Under routine conditions, killing of >50% is considered biologically significant and therefore serves to classify a serum as positive for opsonic killing activity. Thus, although killing of <50% is sometimes statistically significant, this level of killing is not considered biologically significant.
RESULTS
Construction of the aroA deletion mutant.
Using the gene replacement system of Hoang et al. and Schweizer (23, 55), we constructed an unmarked aroA deletion mutant of the P. aeruginosa serogroup O2/O5 strain PAO1 (Fig. 1). The aroA deletion was confirmed by PCR and sequencing, and auxotrophy for aromatic amino acids was verified by the inability to grow on MSM unless supplemented with aromatic amino acids (data not shown). Introduction of a plasmid containing the intact aroA gene conferred the ability to grow on MSM without amino acids (data not shown).
Safety and biologic disposition in animals.
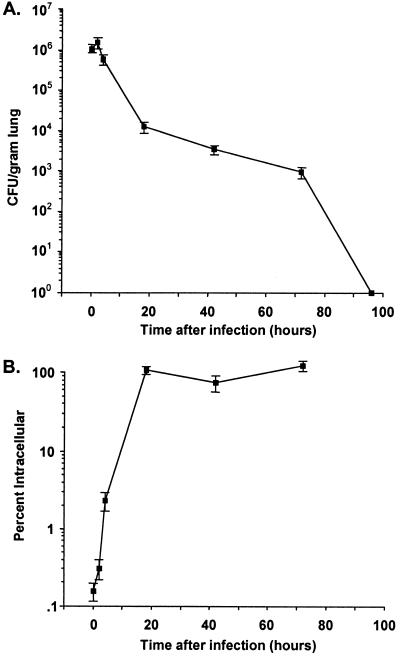
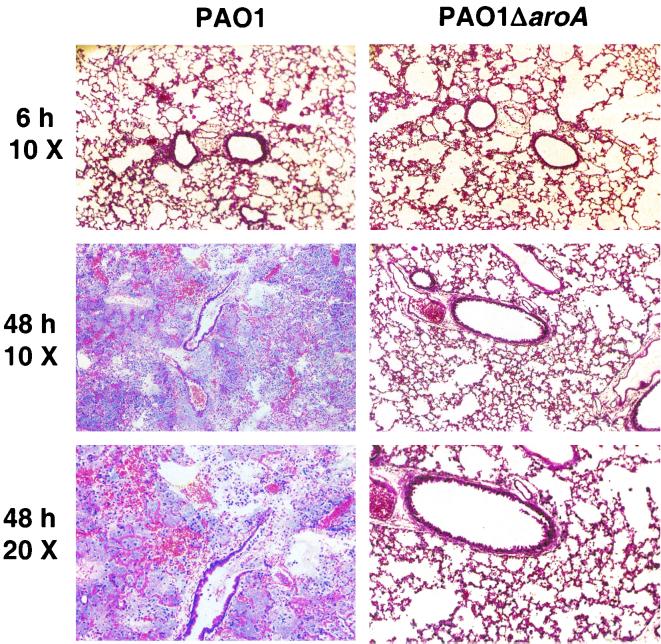
We have reported that i.n. application of P. aeruginosa on the nares of anesthetized mice reliably produces rapid translocation to the lungs and is an excellent model for pneumonia and systemic spread (1). When doses of the attenuated strain PAO1ΔaroA up to 5 × 109 CFU were administered i.n. (or up to 1 × 109 CFU i.p.) to adult BALB/c or C3H/HeN mice, there were no toxic effects. In comparison, the 50% lethal dose for PAO1 given i.n. is 3 × 107 CFU (1). Up to 109 CFU of PAO1ΔaroA given i.n. to anesthetized adult rabbits also had no apparent toxicities. As shown in Fig. 2A,when PAO1ΔaroA was inoculated i.n. at 2 × 109 CFU per mouse, about 107 CFU per gram of lung was recovered immediately postinfection (in contrast to the 67% of parental PAO1 cells that rapidly translocated to the lungs after i.n. infection [1]). The loss in viability of more than 99.9% of the inoculum is presumably due to the rapid death of the aroA deletion mutant in the absence of aromatic amino acids. Approximately 2 × 107 CFU of PAO1ΔaroA per gram of lung tissue was also recovered 4 h after infection, after which the number of recoverable CFU/gram of lung tissue dropped progressively over time. By day 4 and after, all lungs were sterile. Interestingly, as depicted in Fig. 2B, the percentage of the surviving inoculum internalized by mouse lung cells, as determined by gentamicin exclusion assays, was essentially 100% by 18 h after inoculation, probably due to the failure of the aroA deletion mutant to survive extracellularly in the absence of aromatic amino acids. No dissemination of PAO1ΔaroA to blood, liver, or spleen was detected (data not shown). Histological analysis of lungs from mice after i.n. inoculation with PAO1 or PAO1ΔaroA (Fig. 3) showed similar degrees of mild inflammation at early time points (0.5 to 6 h, with representative sections at 6 h shown). However, by 48 h after inoculation, the lungs of mice given PAO1 had evidence of severe pneumonia with extensive PMN infiltration, alveolar hemorrhage, and filling of alveoli with proteinaceous debris and bacteria. On the other hand, at 48 h, the lungs of mice given PAO1ΔaroA had only mild to moderate inflammation, with overall preservation of alveolar and airway architecture.
FIG. 2.
Total (A) and intracellular (B) bacteria in lungs of BALB/c mice at the indicated times following i.n. inoculation of 2 × 109 CFU of PAO1ΔaroA. Each time point represents data from five mice; error bars indicate standard errors of the means.
FIG. 3.
Low-power views of hematoxylin-and-eosin-stained sections of lungs removed at the indicated time points from C3H/HeN mice infected i.n. with PAO1 (left column) or PAO1ΔaroA (right column).
Antibody responses after i.n. immunization.
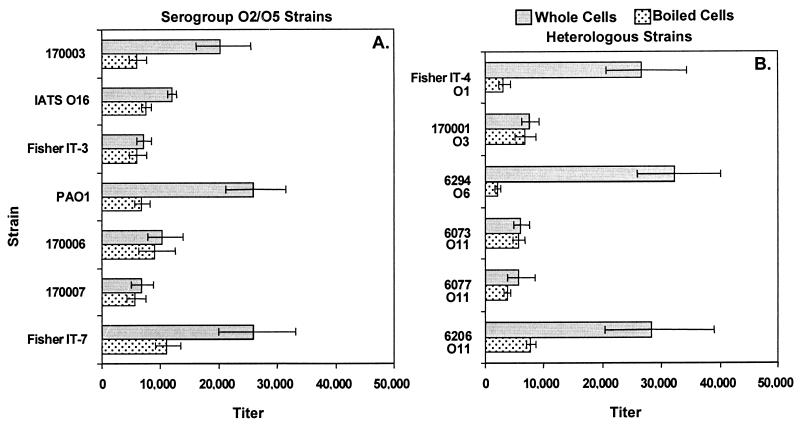
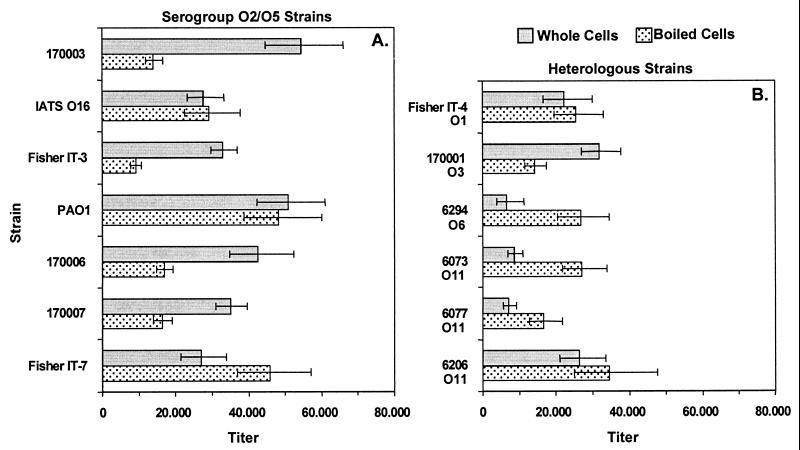
All immunizations were done without adjuvants. When adult C3H/HeN mice were immunized i.n., high titers of serum IgG to whole bacterial cells were measured for serum collected 3 weeks after the last vaccination (Fig. 4). Titers to the parental strain PAO1 were highest (>20,000) and were cross-reactive with most serogroup-homologous strains (>10,000 for five of the seven prototypic serogroup O2/O5 strains; Fig. 4A) and several serogroup-heterologous strains (Fig. 4B). Repeated i.n. immunization of rabbits with PAO1ΔaroA was also well tolerated and, after seven doses, elicited high titers of IgG (>20,000) to whole bacterial cells of all seven of the prototypic serogroup O2/O5 strains and to several serogroup-heterologous strains (Fig. 5). The antisera also possessed high IgG titers against heat-stable bacterial antigens (Fig. 4 and 5), which are predominantly composed of LPS epitopes (titers were >5,000 for heat-stable antigens of all seven prototypic serogroup O2/O5 strains for the mouse antisera and >10,000 for six of the seven strains using the rabbit antisera).
FIG. 4.
IgG titers of antisera from C3H/HeN mice against P. aeruginosa serogroup O2/O5 (A) and heterologous (B) strains by ELISA. The International Antigenic Typing Scheme (IATS) (31, 32) serogroup of each heterologous strain is indicated below the strain designation. Bars indicate endpoint titers as determined by linear regression, and error bars indicate the 95% confidence intervals.
FIG. 5.
IgG titers of rabbit antisera against P. aeruginosa serogroup O2/O5 (A) and heterologous (B) strains by ELISA. The International Antigenic Typing Scheme (IATS) (31, 32) serogroup of each heterologous strain is indicated below the strain designation. Bars indicate endpoint titers as determined by linear regression, and error bars indicate the 95% confidence intervals.
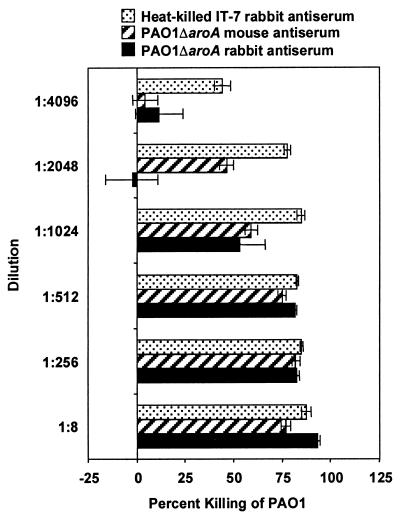
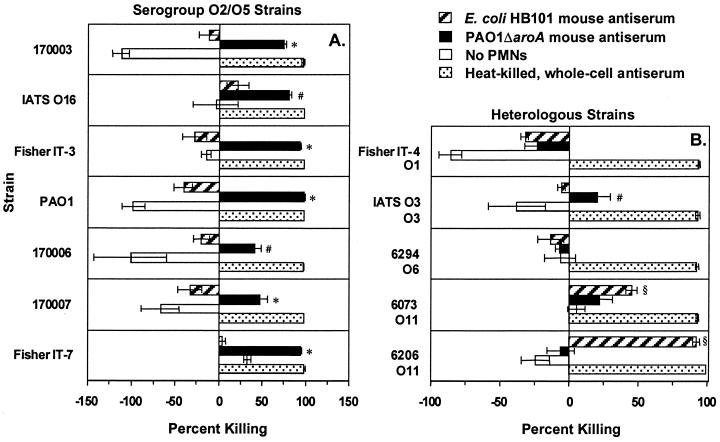
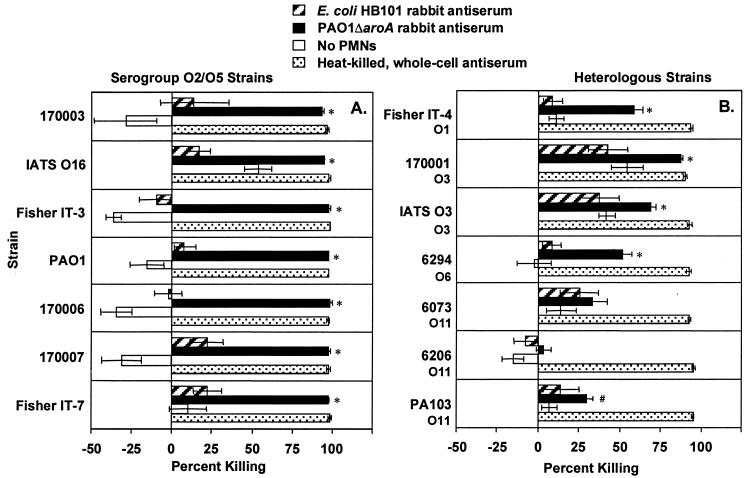
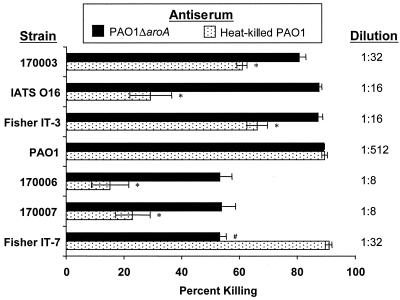
The antisera mediated phagocytic killing of ≥50% of cells of the parental strain PAO1 at serum dilutions as high as 1:2,048 for the mouse antiserum and 1:1,024 for the rabbit antiserum (Fig. 6). Remarkably, when compared with antiserum from a rabbit immunized intravenously with heat-killed (60°C for 1 h) Fisher IT-7, the antisera from these i.n.-immunized animals had only two- to fourfold-lower activity in the opsonophagocytic assays. Using serum dilutions of 1:8, five of the seven prototypic serogroup O2/O5 strains were killed (>50% killing) by the mouse antisera (Fig. 7A) and all seven were killed by the rabbit antisera (Fig. 8A). The rabbit antisera also mediated phagocytic killing (>50%) of several serogroup-heterologous strains (Fig. 8B), but the mouse antisera had no killing activity >50% against serogroup-heterologous strains (Fig. 7B). Curiously, the E. coli HB101 mouse antiserum killed two P. aeruginosa O11 strains, 6073 and 6206, while the rabbit antiserum to E. coli HB101 showed minimal killing of these P. aeruginosa strains. This cross-reactive opsonic killing was likely due to a shared epitope, which was not further characterized.
FIG. 6.
Opsonophagocytic killing of PAO1 by dilutions of mouse and rabbit antisera raised against PAO1ΔaroA after i.n. immunization. Bars represent mean percent killing of two to four replicates relative to no-serum control, and error bars represent the standard errors of the means. Negative killing indicates growth during the 90-min incubation. Rabbit antiserum raised against heat-killed Fisher IT-7 by intravenous immunization served as a positive control.
FIG. 7.
Opsonophagocytic killing of P. aeruginosa serogroup O2/O5 (A) and heterologous (B) strains by a 1:8 dilution of antisera from PAO1ΔaroA-immunized or E. coli HB101-immunized (control) C3H/HeN mice. The International Antigenic Typing Scheme (IATS) (31, 32) serogroup of each heterologous strain is indicated below the strain designation. Bars represent mean percent killing of two to four replicates relative to no-serum control, and error bars represent the standard errors of the means. Negative killing indicates growth during the 90-min incubation. Rabbit antisera raised by intravenous immunization with heat-killed whole bacteria from the same serogroup served as positive controls. Tubes with PAO1ΔaroA antiserum but without PMNs served as additional negative controls. ∗, P < 0.001; #, P < 0.05 by ANOVA with Fisher's PLSD for comparison with E. coli HB101 antiserum., P < 0.01 by ANOVA with Fisher's PLSD for comparison with PAO1ΔaroA antiserum.
FIG. 8.
Opsonophagocytic killing of P. aeruginosa serogroup O2/O5 (A) and heterologous (B) strains by a 1:8 dilution of serum from PAO1ΔaroA-immunized or E. coli HB101-immunized (control) rabbits. The International Antigenic Typing Scheme (IATS) (31, 32) serogroup of each heterologous strain is indicated below the strain designation. Bars represent mean percent killing of two to four replicates relative to no-serum control, and error bars represent the standard errors of the means. Negative killing indicates growth during the 90-min incubation. Rabbit antiserum raised by intravenous immunization with heat-killed whole bacteria from the same serogroup served as positive controls. Tubes with PAO1ΔaroA antiserum but without PMNs served as additional negative controls. ∗, P < 0.001; #, P < 0.05 by ANOVA with Fisher's PLSD for comparison with E. coli HB101 antiserum.
The level of opsonophagocytic killing activity elicited after i.n. immunization of mice with the live, attenuated strain PAO1ΔaroA was compared with that generated after i.n. immunization with heat-killed PAO1 (Fig. 9). For 5 of the 7 prototypic P. aeruginosa serogroup O2/O5 strains, opsonic killing activity engendered by i.n. immunization with PAO1ΔaroA was significantly higher than that with heat-killed PAO1. Killing of the homologous strain PAO1 was similar for the two vaccines, while killing of Fisher IT-7 was better after the heat-killed vaccine. Similarities between PAO1 and Fisher IT-7 are expected, since PAO1 is classified as an IT-7 strain when serogrouping is performed with antisera to the prototypic Fisher IT strains.
FIG. 9.
Opsonophagocytic killing of P. aeruginosa serogroup O2/O5 strains by sera at the indicated dilutions from C3H/HeN mice immunized i.n. with either PAO1ΔaroA or heat-killed PAO1. Bars represent mean percent killing of two to four replicates relative to no-serum control, and error bars represent the standard errors of the means. ∗, P < 0.001 by ANOVA with Fisher's PLSD in comparison with antiserum from mice immunized with PAO1ΔaroA; #, P < 0.001 by ANOVA with Fisher's PLSD in comparison with antiserum from mice immunized with heat-killed PAO1.
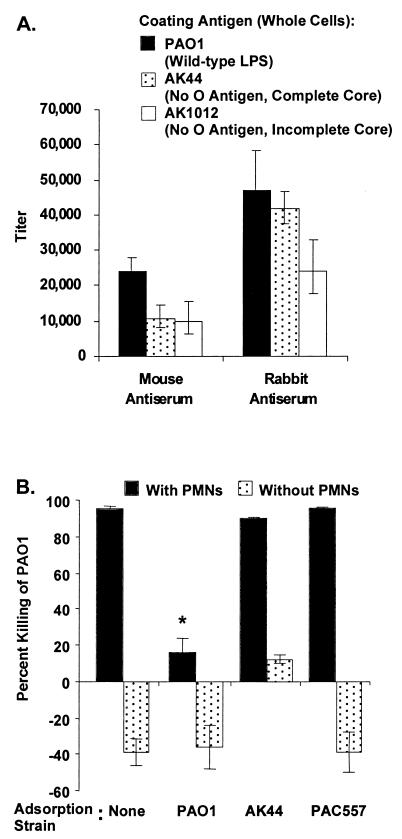
A substantial proportion of the IgG elicited by i.n. immunization with PAO1ΔaroA was directed against the LPS of P. aeruginosa, as indicated by comparing IgG titers to whole cells of the parental strain PAO1, AK44 (an O-antigen-deficient, complete-LPS-core mutant of PAO1), and AK1012 (an O-antigen-deficient, incomplete-LPS-core mutant of PAO1) (Fig. 10A). The differences in titers against AK44 between the mouse and rabbit antisera suggest that the mouse antiserum possesses relatively low amounts of antibody directed against the LPS outer core, while the rabbit antiserum has a high proportion of outer-core-directed antibody. Adsorption of the rabbit antisera with the isogenic O-antigen-deficient P. aeruginosa strain AK44 or with the O-antigen-deficient serogroup O3 strain PAC557 did not affect the level of phagocytic killing of PAO1, while adsorption with PAO1 reduced killing to <15%, thereby confirming the importance of antibody to the O antigen in phagocytic killing (Fig. 10B).
FIG. 10.
Antibodies elicited by i.n. immunization with PAO1ΔaroA are directed against the LPS of P. aeruginosa. (A) IgG titers of mouse and rabbit antisera to PAO1ΔaroA against P. aeruginosa PAO1, AK44 (an O-antigen-deficient, complete-LPS-core mutant of PAO1), and AK1012 (an O-antigen-deficient, incomplete-LPS-core mutant of PAO1). Bars represent endpoint titers as determined by linear regression, and error bars indicate the 95% confidence intervals. (B) Adsorption of rabbit antiserum to PAO1ΔaroA with the O-antigen-deficient mutants AK44 and PAC557 does not alter the opsonic activity, while adsorption with PAO1 abolishes opsonic activity. Bars represent mean percent killing of two to four replicates relative to no-serum control, and error bars represent the standard errors of the means. Negative killing indicates growth during the 90-min incubation. Tubes with the variously adsorbed PAO1ΔaroA antisera but without PMNs served as negative controls. ∗, P < 0.0001 by ANOVA with Fisher's PLSD for comparisons with either unadsorbed, AK44-adsorbed, or PAC557-adsorbed antiserum.
DISCUSSION
For over 30 years, it has been clear that high-level immunity to P. aeruginosa infections can be mediated by antibodies to the LPS O antigen (13). However, just as carbohydrate-based vaccines for other bacterial pathogens have been problematic, it appears that protective epitopes are poorly immunogenic while nonprotective or minimally cross-protective O-antigen epitopes provoke the best immune responses in preclinical evaluations of vaccines for mice and rabbits (19). Thus, the poor immunogenicity of the key antigenic determinants of P. aeruginosa LPS O antigens may lie at the core of the lack of effective LPS-specific vaccines. In addition, with recent evidence from multiple investigators that P. aeruginosa readily enters lung and corneal epithelial cells during infection (14, 21, 46), a potential role for cell-mediated immunity is clear, indicating that in spite of the strong role played by antibodies to LPS in protective immunity, they may not be sufficient to control infections. Indeed, T-cell-based immune protection against P. aeruginosa infections has been demonstrated by several groups over the past 15 years (9, 29, 35-39).
Other P. aeruginosa vaccine strategies have focused on flagellar antigens (24), on outer membrane proteins F (34, 41) and I (34), and more recently, on the PcrV antigen component of the type III secretion system (54). While protection against heterologous serogroups has sometimes been seen, the protection afforded by these non-LPS-based vaccines has, as a rule, been of only modest potency. A particularly striking illustration of the remarkable protective efficacy of LPS-based vaccines is a study in which an outer membrane protein F vaccine protected 30 to 95% of burned mice against challenge doses up to 2 × 106 CFU of six different serogroups while an LPS-based vaccine protected against a challenge dose of 3 × 1011 CFU of the homologous strain (40).
Mutations in the aroA gene, which encodes an enzyme essential for the synthesis of aromatic amino acids (5-enolpyruvylshikimate 3-phosphate synthase of the shikimate pathway), have been utilized with several other pathogens, including Salmonella species (58) and Aeromonas hydrophila (22), for the production of live, attenuated vaccine strains. In fact, in the Salmonella enterica serovar Typhimurium system, aroA deletion mutants have been used as delivery vehicles to vaccinate mice against plasmid-encoded foreign proteins, with subsequent generation of broad cellular immunity (33, 49). Although single aroA deletion mutants in Salmonella enterica serovar Typhi retain sufficient virulence to make them unacceptable as human vaccines, the intrinsically lower virulence of P. aeruginosa was predicted to allow single aroA deletion mutants to be sufficiently attenuated to permit study in animal models.
In the present study, we have constructed an unmarked aroA deletion mutant of the common laboratory strain of P. aeruginosa, PAO1, and confirmed that it is auxotrophic for aromatic amino acids. The strain is highly attenuated in mice in that doses up to 5 × 109 CFU can be given i.n. or i.p. without any apparent adverse effects. We have previously shown that nasal application of a bacterial inoculum in anesthetized adult mice results in rapid translocation of two-thirds or more of the inoculum into the lungs (1). When virulent strains of P. aeruginosa are administered, the mice die of pneumonia and systemic spread within 72 h. In this murine lung infection model, the aroA deletion mutant was translocated to the lungs less well (only about 0.1% of the inoculum could be recovered from the lungs immediately after application) and was cleared from the lung by 4 days after inoculation. Notably, there was no dissemination to the blood, liver, or spleen; and there was only low-level inflammation in lungs as determined by histological evaluation at 48 h. The amount of internalized bacteria as a percentage of total bacteria recovered from the lungs was essentially 100% by 18 h after inoculation with the aroA deletion mutant strain. By comparison, the percentage of internalized bacteria in this model after challenge with the parental strain PAO1 is approximately 10% at most time points (1). This propensity of the aroA deletion mutant for intracellular localization is likely due to its inability to survive in an extracellular environment lacking aromatic amino acids. The limited but significant survival of the attenuated strain is also important for the prospect of using engineered versions of the aroA deletion mutant to overexpress protein antigens that might bolster the immune response.
We have found that i.n. immunization of mice and rabbits with the aroA deletion mutant of PAO1 elicits high titers of IgG against whole cells and boiled cells of multiple subtype- and even serogroup-heterologous strains of P. aeruginosa. These high titers were achieved without the use of adjuvants. While IgG titers determined by ELISA are useful in screening sera for possible protective serologic responses, the levels of opsonic antibodies against P. aeruginosa are the best predictors of protective efficacy in animal models. Along these lines, it is remarkable that i.n. immunization with a single aroA deletion mutant strain engenders opsonic antibodies against the parental strain as well as multiple strains within serogroup O2/O5. This is in stark contrast to our previous findings that i.p. immunization of mice with the purified high-molecular-weight O-polysaccharide from PAO1 elicited low-level opsonic titers against only two of the prototypic serogroup O2/O5 strains and did not elicit a good opsonic antibody response even to the parental strain (19). Furthermore, in challenge experiments using the mouse model of pneumonia and systemic spread after i.n. inoculation (1), mice immunized i.n. with PAO1ΔaroA were 100% protected from death while 100% of mice immunized i.n. with E. coli HB101 died within 4 days of challenge with a cytotoxic variant of PAO1 (PAO1 transfected with a plasmid expressing the cytotoxin ExoU and its chaperone [1]) at a dose 100-fold higher than the 50% lethal dose (J. Goldberg, M. Brinig, M. Grout, K. Hatano, F. Coleman, G. Priebe, and G. Pier, Abstr. 100th Gen. Meet. Am. Soc. Microbiol., abstr. D-155, 2000).
We also compared the i.n. immunization of mice with heat-killed PAO1 to that with PAO1ΔaroA and found that the live attenuated strain PAO1ΔaroA generated significantly higher levels of opsonic antibody against five of the seven prototypic P. aeruginosa serogroup O2/O5 strains (Fig. 9). This may be due to the ability of the live attenuated strain to serve as a better immunogen than heat-killed bacteria by means of simple multiplication as well as by exploitation of the natural pathways of infection, especially the intracellular phase. Presumably, modifications of bacterial antigens that occur during in vivo infection would also occur in the live attenuated strain and could thereby engender a broader immune response. Our findings that the opsonic activity was not removed by adsorption with the O-antigen-deficient strains AK44 and PAC557 (Fig. 10) indicate, as expected, that the active antibodies after i.n. immunization with PAO1ΔaroA are directed against O-antigen epitopes of LPS.
Thus, i.n. immunization of mice and rabbits with the single strain PAO1ΔaroA elicits opsonic antibodies against multiple members of the P. aeruginosa serogroup O2/O5, indicating the significant potential of using live, attenuated strains for vaccination against LPS-smooth strains of P. aeruginosa.
Acknowledgments
This work was supported by NIH grants AI22535 (G.B.P.), AI50036 (G.P.P.), and AI37632 (J.B.G.), by the University of Virginia School of Medicine and a Student Traineeship Grant from the Cystic Fibrosis Foundation (BRINIG99H0) (M.M.B.), and by the Anesthesia Department of Children's Hospital and Harvard Medical School, Boston, Mass. (G.P.P.).
We thank Vincent Carey for assistance with statistical analysis.
REFERENCES
- 1.Allewelt, M., F. T. Coleman, M. Grout, G. P. Priebe, and G. B. Pier. 2000. Acquisition of expression of the P. aeruginosa ExoU cytotoxin leads to increased bacterial virulence in a murine model of acute pneumonia and systemic spread. Infect. Immun. 68:3998-4004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boyer, H. W., and D. Roulland-Dussoix. 1969. A complementation analysis of the restriction and modification of DNA in Escherichia coli. J. Mol. Biol. 41:459-472. [DOI] [PubMed] [Google Scholar]
- 3.Burns, J. L., R. L. Gibson, S. McNamara, D. Yim, J. Emerson, M. Rosenfeld, P. Hiatt, K. McCoy, R. Castile, A. L. Smith, and B. W. Ramsey. 2001. Longitudinal assessment of Pseudomonas aeruginosa in young children with cystic fibrosis. J. Infect. Dis. 183:444-452. [DOI] [PubMed] [Google Scholar]
- 4.Cryz, S. J., Jr., E. Furer, and R. Germanier. 1984. Protection against fatal Pseudomonas aeruginosa burn wound sepsis by immunization with lipopolysaccharide and high-molecular-weight polysaccharide. Infect. Immun. 43:795-799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cryz, S. J., Jr., E. Furer, J. C. Sadoff, T. Fredeking, J. U. Que, and A. S. Cross. 1991. Production and characterization of a human hyperimmune intravenous immunoglobulin against Pseudomonas aeruginosa and Klebsiella species. J. Infect. Dis. 163:1055-1061. [DOI] [PubMed] [Google Scholar]
- 6.Cryz, S. J., Jr., J. C. Sadoff, and E. Furer. 1989. Octavalent Pseudomonas aeruginosa O-polysaccharide-toxin A conjugate vaccine. Microb. Pathog. 6:75-80. [DOI] [PubMed] [Google Scholar]
- 7.DeLorenzo, V., and K. N. Timmis. 1994. Analysis and construction of stable phenotypes in Gram-negative bacteria with Tn 5 and Tn 10-derived transposons. Methods Enzymol. 235:386-405. [DOI] [PubMed] [Google Scholar]
- 8.Donta, S. T., P. Peduzzi, A. S. Cross, J. Sadoff, C. Haakenson, S. J. Cryz, Jr., C. Kauffman, S. Bradley, G. Gafford, D. Elliston, T. R. Beam, Jr., J. F. John, Jr., B. Ribner, R. Cantey, C. H. Welsh, R. T. Ellison III, E. J. Young, R. J. Hamill, H. Leaf, R. M. Schein, M. Mulligan, C. Johnson, E. Abrutyn, J. M. Griffiss, D. Slagle, et al. 1996. Immunoprophylaxis against Klebsiella and Pseudomonas aeruginosa infections. The Federal Hyperimmune Immunoglobulin Trial Study Group. J. Infect. Dis. 174:537-543. [DOI] [PubMed] [Google Scholar]
- 9.Dunkley, M. L., R. L. Clancy, and A. W. Cripps. 1994. A role for CD4+ T cells from orally immunized rats in enhanced clearance of Pseudomonas aeruginosa from the lung. Immunology 83:362-369. [PMC free article] [PubMed] [Google Scholar]
- 10.Edgeworth, J. D., D. F. Treacher, and S. J. Eykyn. 1999. A 25-year study of nosocomial bacteremia in an adult intensive care unit. Crit. Care Med. 27:1421-1428. [DOI] [PubMed] [Google Scholar]
- 11.Elishoov, H., R. Or, N. Strauss, and D. Engelhard. 1998. Nosocomial colonization, septicemia, and Hickman/Broviac catheter-related infections in bone marrow transplant recipients. A 5-year prospective study. Medicine 77:83-101. [DOI] [PubMed] [Google Scholar]
- 12.Esen, M., H. Grassme, J. Riethmuller, A. Riehle, K. Fassbender, and E. Gulbins. 2001. Invasion of human epithelial cells by Pseudomonas aeruginosa involves src-like tyrosine kinases p60Src and p59Fyn. Infect. Immun. 69:281-287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fisher, M. W., H. B. Devlin, and F. Gnabski. 1969. New immunotype schema for Pseudomonas aeruginosa based on protective antigens. J. Bacteriol. 98:835-836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fleiszig, S. M., V. Vallas, C. H. Jun, L. Mok, D. F. Balkovetz, M. G. Roth, and K. E. Mostov. 1998. Susceptibility of epithelial cells to Pseudomonas aeruginosa invasion and cytotoxicity is upregulated by hepatocyte growth factor. Infect. Immun. 66:3443-3446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fleiszig, S. M., T. S. Zaidi, M. J. Preston, M. Grout, D. J. Evans, and G. B. Pier. 1996. Relationship between cytotoxicity and corneal epithelial cell invasion by clinical isolates of Pseudomonas aeruginosa. Infect. Immun. 64:2288-2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gales, A. C., R. N. Jones, J. Turnidge, R. Rennie, and R. Ramphal. 2001. Characterization of Pseudomonas aeruginosa isolates: occurrence rates, antimicrobial susceptibility patterns, and molecular typing in the global SENTRY antimicrobial surveillance program, 1997-1999. Clin. Infect. Dis. 32(Suppl. 2):S146-S155. [DOI] [PubMed] [Google Scholar]
- 17.Guzman, C. A., S. R. Talay, G. Molinari, E. Medina, and G. S. Chhatwal. 1999. Protective immune response against Streptococcus pyogenes in mice after intranasal vaccination with the fibronectin-binding protein SfbI. J. Infect. Dis. 179:901-906. [DOI] [PubMed] [Google Scholar]
- 18.Hatano, K., S. Boisot, D. DesJardins, D. C. Wright, J. Brisker, and G. B. Pier. 1994. Immunogenic and antigenic properties of a heptavalent high-molecular-weight O-polysaccharide vaccine derived from Pseudomonas aeruginosa. Infect. Immun. 62:3608-3616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hatano, K., and G. B. Pier. 1998. Complex serology and immune response of mice to variant high-molecular-weight O polysaccharides isolated from Pseudomonas aeruginosa serogroup O2 strains. Infect. Immun. 66:3719-3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hauser, A. R., and J. N. Engel. 1999. Pseudomonas aeruginosa induces type-III-secretion-mediated apoptosis of macrophages and epithelial cells. Infect. Immun. 67:5530-5537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hauser, A. R., S. Fleiszig, P. J. Kang, K. Mostov, and J. N. Engel. 1998. Defects in type III secretion correlate with internalization of Pseudomonas aeruginosa by epithelial cells. Infect. Immun. 66:1413-1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hernanz Moral, C., E. Flano del Castillo, P. Lopez Fierro, A. Villena Cortes, J. Anguita Castillo, A. Cascon Soriano, M. Sanchez Salazar, B. Razquin Peralta, and G. Naharro Carrasco. 1998. Molecular characterization of the Aeromonas hydrophila aroA gene and potential use of an auxotrophic aroA mutant as a live attenuated vaccine. Infect. Immun. 66:1813-1821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hoang, T. T., R. R. Karkhoff-Schweizer, A. J. Kutchma, and H. P. Schweizer. 1998. A broad-host-range Flp-FRT recombination system for site-specific excision of chromosomally-located DNA sequences: application for isolation of unmarked Pseudomonas aeruginosa mutants. Gene 212:77-86. [DOI] [PubMed] [Google Scholar]
- 24.Holder, I. A., and J. G. Naglich. 1986. Experimental studies of the pathogenesis of infections due to Pseudomonas aeruginosa: immunization using divalent flagella preparations. J. Trauma 26:118-122. [DOI] [PubMed] [Google Scholar]
- 25.Jarrell, K., and A. M. Kropinski. 1977. The chemical composition of the lipopolysaccharide from Pseudomonas aeruginosa strain PAO and a spontaneously derived rough mutant. Microbios 19:103-116. [PubMed] [Google Scholar]
- 26.Klavinskis, L. S., C. Barnfield, L. Gao, and S. Parker. 1999. Intranasal immunization with plasmid DNA-lipid complexes elicits mucosal immunity in the female genital and rectal tracts. J. Immunol. 162:254-262. [PubMed] [Google Scholar]
- 27.Knirel, Y. A. 1990. Polysaccharide antigens of Pseudomonas aeruginosa. Crit. Rev. Microbiol. 17:273-304. [DOI] [PubMed] [Google Scholar]
- 28.Knirel, Y. A., N. A. Paramonov, E. V. Vinogradov, A. S. Shashkov, B. A. Dmitriev, N. K. Kochetkov, E. V. Kholodkova, and E. S. Stanislavsky. 1987. Somatic antigens of Pseudomonas aeruginosa. The structure of O-specific polysaccharide chains of lipopolysaccharides of P. aeruginosa O3 (Lanyi), O25 (Wokatsch) and Fisher immunotypes 3 and 7. Eur. J. Biochem. 167:549-561. [DOI] [PubMed] [Google Scholar]
- 29.Kondratieva, T. K., N. V. Kobets, S. V. Khaidukov, V. V. Yeremeev, I. V. Lyadova, A. S. Apt, M. F. Tam, and M. M. Stevenson. 2000. Characterization of T cell clones derived from lymph nodes and lungs of Pseudomonas aeruginosa-susceptible and resistant mice following immunization with heat-killed bacteria. Clin. Exp. Immunol. 121:275-282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kropinski, A. M., L. C. Chan, and F. H. Milazzo. 1979. The extraction and analysis of lipopolysaccharides from Pseudomonas aeruginosa strain PAO and three rough mutants. Can. J. Microbiol. 25:390-398. [DOI] [PubMed] [Google Scholar]
- 31.Liu, P. V., H. Matsumoto, H. Kusama, and T. Bergan. 1983. Survey of heat-stable major somatic antigens of Pseudomonas aeruginosa. Int. J. Syst. Bacteriol. 33:256-264. [Google Scholar]
- 32.Liu, P. V., and S. Wang. 1990. Three new major somatic antigens of Pseudomonas aeruginosa. J. Clin. Microbiol. 28:922-925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lo-Man, R., J. P. M. Langeveld, E. Deriaud, M. Jehanno, M. Rojas, J. Clement, R. H. Meloen, M. Hofnung, and C. Leclerc. 2000. Extending the CD4+ T-cell epitope specificity of the Th1 immune response to an antigen using a Salmonella enterica serovar Typhimurium delivery vehicle. Infect. Immun. 68:3079-3089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mansouri, E., J. Gabelsberger, B. Knapp, E. Hundt, U. Lenz, K. D. Hungerer, H. E. Gilleland, Jr., J. Staczek, H. Domdey, and B. U. von Specht. 1999. Safety and immunogenicity of a Pseudomonas aeruginosa hybrid outer membrane protein F-I vaccine in human volunteers. Infect. Immun. 67:1461-1470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Markham, R. B., J. Goellner, and G. B. Pier. 1984. In vitro T cell-mediated killing of Pseudomonas aeruginosa. I. Evidence that a lymphokine mediates killing. J. Immunol. 133:962-968. [PubMed] [Google Scholar]
- 36.Markham, R. B., G. B. Pier, J. J. Goellner, and S. B. Mizel. 1985. In vitro T cell-mediated killing of Pseudomonas aeruginosa. II. The role of macrophages and T cell subsets in T cell killing. J. Immunol. 134:4112-4117. [PubMed] [Google Scholar]
- 37.Markham, R. B., G. B. Pier, and W. G. Powderly. 1988. Suppressor T cells regulating the cell-mediated immune response to Pseudomonas aeruginosa can be generated by immunization with anti-bacterial T cells. J. Immunol. 141:3975-3979. [PubMed] [Google Scholar]
- 38.Markham, R. B., G. B. Pier, and J. R. Schreiber. 1991. The role of cytophilic IgG3 antibody in T cell-mediated resistance to infection with the extracellular bacterium, Pseudomonas aeruginosa. J. Immunol. 146:316-320. [PubMed] [Google Scholar]
- 39.Markham, R. B., and W. G. Powderly. 1988. Exposure of mice to live Pseudomonas aeruginosa generates protective cell-mediated immunity in the absence of an antibody response. J. Immunol. 140:2039-2045. [PubMed] [Google Scholar]
- 40.Matthews-Greer, J. M., and H. E. Gilleland, Jr. 1987. Outer membrane protein F (porin) preparation of Pseudomonas aeruginosa as a protective vaccine against heterologous immunotype strains in a burned mouse model. J. Infect. Dis. 155:1282-1291. [DOI] [PubMed] [Google Scholar]
- 41.Matthews-Greer, J. M., D. E. Robertson, L. B. Gilleland, and H. E. Gilleland, Jr. 1990. Pseudomonas aeruginosa outer membrane protein F produced in Escherichia coli retains vaccine efficacy. Curr. Microbiol. 20:171-175.
- 42.Ohman, D. E., and A. M. Chakrabarty. 1982. Utilization of human respiratory secretions by mucoid Pseudomonas aeruginosa of cystic fibrosis origin. Infect. Immun. 37:662-669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pier, G. B. 1982. Safety and immunogenicity of high molecular weight polysaccharide vaccine from immunotype 1 Pseudomonas aeruginosa. J. Clin. Investig. 69:303-308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pier, G. B., M. Grout, and T. S. Zaidi. 1997. Cystic fibrosis transmembrane conductance regulator is an epithelial cell receptor for clearance of Pseudomonas aeruginosa from the lung. Proc. Natl. Acad. Sci. USA 94:12088-12093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pier, G. B., M. Grout, T. S. Zaidi, and J. B. Goldberg. 1996. How mutant CFTR may contribute to Pseudomonas aeruginosa infection in cystic fibrosis. Am. J. Respir. Crit. Care Med. 154:S175-S82. [DOI] [PubMed] [Google Scholar]
- 46.Pier, G. B., M. Grout, T. S. Zaidi, J. C. Olsen, L. G. Johnson, J. R. Yankaskas, and J. B. Goldberg. 1996. Role of mutant CFTR in hypersusceptibility of cystic fibrosis patients to lung infections. Science 271:64-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pier, G. B., and M. Pollack. 1989. Isolation, structure, and immunogenicity of Pseudomonas aeruginosa immunotype 4 high-molecular-weight polysaccharide. Infect. Immun. 57:426-431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pier, G. B., D. Thomas, G. Small, A. Siadak, and H. Zweerink. 1989. In vitro and in vivo activity of polyclonal and monoclonal human immunoglobulins G, M, and A against Pseudomonas aeruginosa lipopolysaccharide. Infect. Immun. 57:174-179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Poirier, T. P., M. A. Kehoe, and E. H. Beachey. 1988. Protective immunity evoked by oral administration of attenuated aroA Salmonella typhimurium expressing cloned streptococcal M protein. J. Exp. Med. 168:25-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Preston, M. J., S. M. Fleiszig, T. S. Zaidi, J. B. Goldberg, V. D. Shortridge, M. L. Vasil, and G. B. Pier. 1995. Rapid and sensitive method for evaluating Pseudomonas aeruginosa virulence factors during corneal infections in mice. Infect. Immun. 63:3497-3501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rello, J., M. Rue, P. Jubert, G. Muses, R. Sonora, J. Valles, and M. S. Niederman. 1997. Survival in patients with nosocomial pneumonia: impact of the severity of illness and the etiologic agent. Crit. Care Med. 25:1862-1867. [DOI] [PubMed] [Google Scholar]
- 52.Richards, M. J., J. R. Edwards, D. H. Culver, and R. P. Gaynes. 1999. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit. Care Med. 27:887-892. [DOI] [PubMed] [Google Scholar]
- 53.Rowe, P. S., and P. M. Meadow. 1983. Structure of the core oligosaccharide from the lipopolysaccharide of Pseudomonas aeruginosa PAC1R and its defective mutants. Eur. J. Biochem. 132:329-337. [DOI] [PubMed] [Google Scholar]
- 54.Sawa, T., T. L. Yahr, M. Ohara, K. Kurahashi, M. A. Gropper, J. P. Wiener-Kronish, and D. W. Frank. 1999. Active and passive immunization with the Pseudomonas V antigen protects against type III intoxication and lung injury. Nat. Med. 5:392-398. [DOI] [PubMed] [Google Scholar]
- 55.Schweizer, H. P. 1991. Escherichia-Pseudomonas shuttle vectors derived from pUC18/19. Gene 97:109-112. [DOI] [PubMed] [Google Scholar]
- 56.Schweizer, H. P., and T. T. Hoang. 1995. An improved system for gene replacement and xylE fusion analysis in Pseudomonas aeruginosa. Gene 158:15-22. [DOI] [PubMed] [Google Scholar]
- 57.Simon, R., U. Priefer, and A. Puhler. 1983. A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in Gram-negative bacteria. Bio/Technology 1:784-791. [Google Scholar]
- 58.Stocker, B. A. 1988. Auxotrophic Salmonella typhi as live vaccine. Vaccine 6:141-145. [DOI] [PubMed] [Google Scholar]
- 59.Sun, J. B., N. Mielcarek, M. Lakew, J. M. Grzych, A. Capron, J. Holmgren, and C. Czerkinsky. 1999. Intranasal administration of a Schistosoma mansoni glutathione S-transferase-cholera toxoid conjugate vaccine evokes antiparasitic and antipathological immunity in mice. J. Immunol. 163:1045-1052. [PubMed] [Google Scholar]
- 60.Zaidi, T. S., J. Lyczak, M. Preston, and G. B. Pier. 1999. Cystic fibrosis transmembrane conductance regulator-mediated corneal epithelial cell ingestion of Pseudomonas aeruginosa is a key component in the pathogenesis of experimental murine keratitis. Infect. Immun. 67:1481-1492. [DOI] [PMC free article] [PubMed] [Google Scholar]