Abstract
The global use of a capsular polysaccharide-based pneumococcal vaccine has been limited because of serotype-specific protection and poor effectiveness in individuals with low immunocompetency. The mucosal immune system develops earlier in infants and lasts longer in the elderly than does the systemic immune system. Furthermore, mucosal immunization is beneficial for AIDS patients, because human immunodeficiency virus-infected subjects can develop normal mucosal antibody responses even in late clinical phases. For these reasons, we evaluated recombinant pneumococcal surface adhesin A (rPsaA) of Streptococcus pneumoniae in terms of cross-protective immune responses after oral delivery. Encapsulated rPsaA provided higher immunogenicity than naked rPsaA. Coencapsulation or codelivery of the cholera toxin (CT) B subunit (CTB) and CT also increased the immunogenicity of rPsaA. Cross-protective immunities against five strains of S. pneumoniae (types 4, 6B, 14, 19F, and 23F) were induced after oral immunization with microencapsulated rPsaA. Lung colonization and septicemia caused by the five serotypes were significantly inhibited by oral immunization with microencapsulated rPsaA. These results suggest that rPsaA coencapsulated with CTB can be used as an oral vaccine to induce cross-protective immunity for the prevention of pneumococcal infection.
Streptococcus pneumoniae is an important respiratory pathogen that causes pneumonia, meningitis, otitis media, and bacteremia (9). Although polysaccharide (PS) vaccines can elicit a protective immune response against pneumococcal infection in adults, those have little effect in young children (<2 years), in the elderly, and in patients with immunodeficiencies, such as AIDS (12, 18, 24, 36, 43). In efforts to overcome these limitations, protein-PS conjugate vaccines are being evaluated (19, 21, 35). The conjugate PS appear to activate T-helper cells, thereby eliciting T-cell-dependent responses that provide a long-term immunological memory. Although protein-PS conjugate vaccines were effective in eliminating nasopharyngeal carriage of vaccine serotypes, they increased carriage of nonvaccine serotypes causing invasive disease (30). Therefore, conjugate vaccines seem to be less effective in reducing the overall incidence of pneumococcal disease than expected. Pneumococcal proteins eliciting cross-protective immunity might provide alternative approaches (34).
Several pneumococcal proteins, such as pneumolysin, neuraminidase, autolysin, pneumococcal surface protein A, and pneumococcal surface adhesin A (PsaA), are known to elicit protective immunity (1, 8, 23, 45). Recently, it has been also reported that combinations of pneumococcal virulence proteins, pneumolysin, pneumococcal surface protein A, and PsaA can elicit enhanced protection (7, 31).
PsaA, a 37-kDa metal-binding lipoprotein, plays a critical role in bacterial adherence to respiratory mucosa and in virulence (6). Mice systemically immunized with PsaA were significantly protected against heterologous challenge with type 3 pneumococcal strain WU2 (45). Intranasal immunization of mice with PsaA conferred resistance against nasopharyngeal carriage (7). Immunization of humans with heat-killed bacteria resulted in elevation of salivary antibodies to PsaA (13). Amino acid sequences of PsaA are highly conserved within all 90 pneumococcal serotypes (13, 38). Taken together, these findings indicate that PsaA might be useful as a vaccine which confers cross-protective immunity against various serotypes.
The local secretory immunoglobulin A (secretory IgA) has been shown to prevent both bacterial colonization at the mucosal surfaces and spread into the systemic circulation (41). Moreover, mucosal immunization could be a better alternative for young children and for the elderly, since the mucosal immune system develops earlier in infants and lasts longer in the elderly compared with the systemic immune system (16, 37, 44). Mucosal immunization is also beneficial for human immunodeficiency virus patients (41), because human immunodeficiency virus-infected subjects can develop normal mucosal antibody responses even in late clinical phases (15, 22, 29, 33).
Oral immunization has been limited because of inefficient antigen uptake, induction of tolerance, and proteolytic degradation of the antigens before they reach immune cells (42). To overcome these limitations, numerous approaches using biodegradable microspheres are currently being developed (25). The encapsulated antigens may resist digestion by gastric acid and enzymes, be absorbed via M cells, and thus potentiate immune responses in the common mucosal immune systems (25, 26, 28, 32). Unlike other microspheres, water-soluble microspheres such as chitosan, starch, dextran, and alginate can be prepared easily in aqueous solutions at room temperature and therefore are very useful in encapsulating antigens (4, 17, 20, 39). Especially, small alginate microspheres were considered to be an efficient carrier of antigens to the Payer's patches and through the lymphatic system (40).
In this study, we have evaluated whether the oral administration of a recombinant PsaA (rPsaA) encapsulated in alginate microspheres induces mucosal as well as systemic immune responses and whether oral immunization confers to mice cross-protective immunity against respiratory infection by S. pneumoniae serotypes 4, 6B, 14, 19F, and 23F.
MATERIALS AND METHODS
Mice.
Female BALB/c mice, 6 to 8 weeks old, were purchased from DAEHAN Laboratory Animal Research Center Co. (Seoul, Korea) and were maintained in the facility under standard specific-pathogen-free conditions. All mice were provided sterile food and water ad libitum and were fasted for 4 h before immunization.
Antigens.
A DNA fragment (877 bp) carrying the functional psaA gene (i.e., without its signal sequence) was amplified by PCR from S. pneumoniae type 19F (American Type Culture Collection, Manassas, Va.) genomic DNA. The sense primer sequence was 5′-TCACCATGGTAGCATGTGCTAGCGG-3′. An NcoI site (underlined) was introduced. The reverse primer, complementary to the 3′ sequence, 5′-TGCCAATCCTTCAGCAATCTTGTC A-3′, had a SmaI site. The PCR fragment was then digested with NcoI and SmaI, purified after agarose gel electrophoresis, and ligated with the intein sequence in the expression plasmid pCYB4 (New England Biolabs, Inc., Beverly, Mass.). The pCYB4 DNA was digested with NcoI and SmaI and was ligated with the psaA DNA fragment. Escherichia coli competent cells (XL1-Blue) were transformed with a recombinant plasmid containing a functional psaA gene. To identify E. coli clones expressing PsaA, several colonies were screened by Western blot analysis using polyclonal sera (1:100 dilution) from rabbits immunized with S. pneumoniae. One stable recombinant clone (designated pCA77) expressing chimeric PsaA was selected for further studies.
The DNA sequence of the cloned psaA was determined using an ABI PRISM dye terminator cycle sequencing kit (Applied Biosystems, Foster City, Calif.) with primers compatible with the Ptac promoter sequence or the intein sequence (New England Biolabs, Inc.). The products were analyzed with a DNA sequencer (model 373; Applied Biosystems).
The purification of rPsaA was performed as described by the manufacturer (New England Biolabs, Inc.). The rPsaA was eluted with column buffer. The purified rPsaA was concentrated with an Amicon Centriprep (molecular mass cutoff, 10 kDa) and dialyzed with the distilled water. The purity of rPsaA was also analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Western blotting. The Bradford assay kit was used to determine the total rPsaA protein content.
Microencapsulation of antigens.
Antigens were microencapsulated as described earlier (10). The size and surface morphology of the microspheres were examined using a scanning electron microscope (S-2460N; Hitachi Ltd., Tokyo, Japan) as described previously (11). To analyze the contents of encapsulated antigen, the microspheres were dissolved in sodium citrate buffer (0.1 M, pH 7.8) for 3 h. The protein concentration and immunogenicity of rPsaA were evaluated with a Bradford assay kit or by enzyme-linked immunosorbent assay (ELISA), respectively.
Immunization.
Mice were immunized orally with rPsaA (40 μg) three times at intervals of 2 weeks. Various antigen formulations were used in the immunization. The antigen formulations were as follows: group 1, naked rPsaA (nPsaA); group 2, microencapsulated rPsaA (ePsaA); group 3, nPsaA mixed with 5 μg of cholera toxin (CT) (nPsaA-CT); group 4, ePsaA mixed with 5 μg of CT (ePsaA-CT); group 5, nPsaA mixed with 10 μg of CT B subunit (CTB) (nPsaA-CTB); group 6, ePsaA mixed with 10 μg of CTB (ePsaA-CTB); and group 7, coencapsulated rPsaA with CTB [e(PsaA-CTB)]. Various amounts of CTB were coencapsulated in fabricating e(PsaA-CTB): 1.6 μg of CTB [e(PsaA-CTB)1] for group 8, 8.4 μg of CTB [e(PsaA-CTB)2] for group 9, and 20.3 μg of CTB [e(PsaA-CTB)3] for group 10. CTB was purchased from List Biologicals Laboratory, Inc. (Campbell, Calif.) and was shown by supplier to contain no detectable CT based on the results of an ADP-ribosylation assay. Mock microspheres or microencapsulated bovine serum albumin (eBSA) was also used for the control experiments (group 1). Oral immunizations of mice and collections of sera, intestinal fluid, and bronchoalveolar lavage fluid were performed as described previously (10, 14). Blood contamination of the bronchoalveolar lavage fluid was assessed by detecting hemoglobin at by measurement of optical density at 575 nm. Murine blood diluted serially in PBS served as a standard. The mean blood contamination was 4% ± 0.7%.
ELISA.
Anti-PsaA antibodies were measured by ELISA as described previously (40). Briefly, polystyrene microtiter plates (Corning, Cambridge, Mass,) were coated with 3.2 μg of rPsaA per ml in bicarbonate coating buffer. Serum specimens were diluted 50 times, and intestinal or bronchoalveolar lavage samples diluted twofold were used as primary antibodies. The secondary antibody used was horseradish peroxidase-conjugated goat anti-mouse immunoglobulin (Organon Teknika Corp., Durham, N.C.). To calibrate the specific IgA, IgG, or IgM level, a purified respective immunoglobulin with known concentrations of isotype served as a standard. The absorbance values of the standard immunoglobulin were determined by using sandwich ELISA. The antibody concentration was expressed as the geometric mean.
Challenge.
The mice were immunized orally three times as described above. Protection of mice against intranasal challenge with live pneumococci was analyzed by recovering viable organisms from lungs and blood of mice immunized with various vaccines (40). Two weeks after the last immunization, mice were anesthetized and challenged intranasally with S. pneumoniae in 20 μl of medium as described previously (47). After 18 h, both blood and bronchoalveolar lavage solutions were serially diluted and plated on sheep blood agar plates. Bacterial cultures were incubated at 37°C in a 5% CO2 incubator, and colonies were counted after 18 h. Five strains, which are components of a seven-valent polysaccharide vaccine, were used for challenging infection. The virulence of pneumococci was enhanced by passage through mice. The challenging inocula for each serotype were predetermined in the range showing reproducible linear dose-response (CFU in samples) curves. After the mice were immunized orally with e(PsaA-CTB)1, they were challenged intranasally with 2 × 106 CFU of serotype 4 (ATCC 6304), 1 × 1010 CFU of serotype 6B (ATCC 6326), 1 × 109 CFU of serotype 14 (ATCC 6314), 1 × 106 CFU of serotype 19F (ATCC 6319), or 1 × 109 CFU of serotype 23F (ATCC 6323) at 2 weeks after the last immunization.
Statistics.
Unpaired, one-tailed Student t tests were used to compare the mean antibody levels and the CFU between groups of mice. The values were considered statistically significant at a P value of <0.05.
RESULTS
Expression and purification of rPsaA.
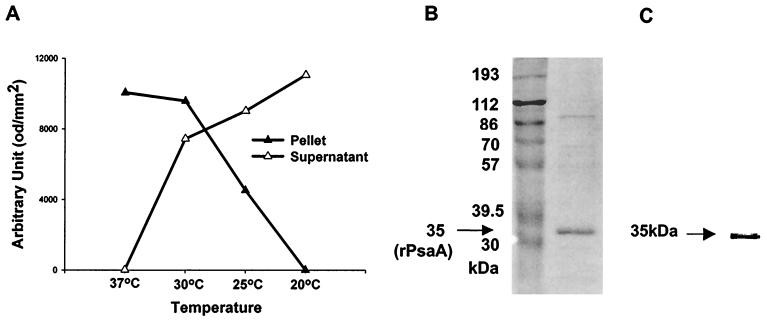
We constructed a plasmid, pCA77, expressing psaA of S. pneumoniae type 19F. DNA sequencing revealed that the psaA sequence in the plasmid was identical to that of the entire authentic psaA (870 bp) without a signal sequence. The transformed E. coli expressed a triple fusion protein (rPsaA-intein-chitin binding domain [CBD]). Densitometric analysis of Western blots was used to quantitate rPsaA-intein-CBD (Fig. 1). When induced at 37°C, most of the recombinant protein formed inclusion bodies and was detectable in pellets. However, the fusion protein was expressed in a soluble form that was detectable in supernatants when induced at 20°C.
FIG. 1.
Expression of fusion protein (rPsaA-intein-CBD) (A), identification of the purified rPsaA protein by SDS-PAGE (B), and Western blot analysis (C). (A) Fusion proteins were expressed in E. coli XL1-Blue at 37, 30, 25, or 20°C. Insoluble and soluble proteins were recovered in pellets (▴) and supernatants (▵), respectively. The density of the fusion protein band was determined with TINA software and plotted against temperature. od, optical density units. (B and C) After purification, the rPsaA solution was separated on 10% polyacrylamide gels (B) and probed with sera from rabbits immunized with S. pneumoniae (C). The numbers in panel B indicate molecular masses of marker proteins. An arrow indicates the purified rPsaA protein (35 kDa).
The rPsaA protein was purified by chitin affinity chromatography. The purified 35-kDa rPsaA was characterized by SDS-10% PAGE (Fig. 1B) and Western blotting (Fig. 1C) analysis. The purity of rPsaA was about 90%, and the average yield was approximately 10 mg of purified protein per liter of culture. The purified rPsaA appeared to be soluble only in the presence of Triton X-100 (0.1%). The purified protein formed insoluble aggregates when more than 1.0 mg of protein per ml was dialyzed with distilled water to prepare microspheres. For this reason, the antigen concentration was kept below 1.0 mg/ml for all of the procedures.
To determine whether the purified rPsaA was immunogenic, mice were immunized with 50 μg of purified rPsaA subcutaneously with Freund's adjuvant. We could detect specific antibody to rPsaA by ELISA (data not shown).
Microencapsulation of rPsaA.
For efficient oral delivery of rPsaA, alginate microspheres entrapping rPsaA were fabricated using an interfacial gelation technique as described previously (10). The microspheres were observed by scanning electron microscopy (Fig. 2). They had a smooth surface without noticeable defects, with a diameter in the range of 0.5 to 5 μm.
FIG. 2.
Microspheres entrapping rPsaA (ePsaA), examined using a scanning electron microscope.
To quantitate rPsaA contents in ePsaA, the Bradford assay was used. The average efficiency of the encapsulation of rPsaA protein (100 × encapsulated rPsaA/rPsaA in starting material) was 24%. The efficiency of the encapsulation of BSA was 42%. The percentage of rPsaA encapsulated in the microspheres (100 × weight of encapsulated rPsaA/total weight of microspheres) was 0.4% (0.8% for BSA protein).
CTB in e(PsaA-CTB) was quantitated by ELISA. The weight ratios of rPsaA to CTB were 1:0.04 in e(PsaA-CTB)1, 1: 0.2 in e(PsaA-CTB)2, and 1:0.5 in e(PsaA-CTB)3.
Antibody responses to rPsaA after oral immunization.
Two weeks after the last immunizations, the rPsaA-specific antibody responses were evaluated by ELISA (Table 1), and differences were analyzed using the one-tailed Student's t test (Table 2). nPsaA (group 2) induced systemic and mucosal antibody responses significantly (P < 0.05) higher than those induced by eBSA (group 1).
TABLE 1.
Systemic and mucosal antibody responses of mice orally immunized with rPsaA
| Group | Vaccine | No. of mice | Geometric mean concn (ng/ml) ± SEM of:
|
||||
|---|---|---|---|---|---|---|---|
| Serum IgM | Serum IgG | Serum IgA | Lung IgA | Intestine IgA | |||
| 1 | eBSA | 6 | 232.7 ± 10.6 | 17.5 ± 1.1 | 5.7 ± 3.1 | 0.7 ± 0.3 | 0.7 ± 0.2 |
| 2 | nPsaA | 7 | 439.9 ± 8.5 | 59.4 ± 1.4 | 17.1 ± 3.9 | 1.8 ± 0.1 | 2.6 ± 0.1 |
| 3 | ePsaA | 7 | 486.2 ± 12.6 | 73.0 ± 1.7 | 31.9 ± 3.1 | 2.1 ± 0.1 | 3.2 ± 0.3 |
| 4 | nPsaA−CT | 7 | 466.1 ± 23.6 | 72.9 ± 5.3 | 29.0 ± 3.9 | 2.5 ± 0.1 | 3.5 ± 0.3 |
| 5 | ePsaA−CT | 7 | 539.1 ± 13.7 | 187.9 ± 18.1 | 70.8 ± 11.8 | 3.6 ± 0.3 | 3.9 ± 0.4 |
| 6 | nPsaA−CTB | 7 | 407.4 ± 21.8 | 68.2 ± 5.9 | 18.1 ± 0.9 | 1.8 ± 0.2 | 2.8 ± 0.4 |
| 7 | ePsaA−CTB | 7 | 545.3 ± 40.1 | 126.3 ± 15.6 | 57.5 ± 1.9 | 3.9 ± 0.8 | 4.0 ± 0.2 |
| 8 | e(PsaA−CTB)1 | 7 | 1901.0 ± 141.4 | 146.5 ± 68.6 | 118.3 ± 38.9 | 5.3 ± 0.7 | 5.5 ± 0.9 |
| 9 | e(PsaA−CTB)2 | 7 | 1759.9 ± 208.1 | 53.4 ± 23.9 | 135.3 ± 96.7 | 6.0 ± 0.6 | 6.6 ± 0.4 |
| 10 | e(PsaA−CTB)3 | 9 | 1542.2 ± 222.9 | 71.1 ± 24.6 | 366.4 ± 37.6 | 16.6 ± 0.8 | 14.1 ± 1.0 |
TABLE 2.
Statistical significance of differences between the groups shown in 1.
| Vaccines compared |
P value for difference of antibody responses
|
||||
|---|---|---|---|---|---|
| Serum IgM | Serum IgG | Serum IgA | Lung IgA | Intestine IgA | |
| eBSA vs nPsaA | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 |
| nPsaA vs ePsaA | 0.005 | <0.001 | 0.006 | 0.03 | 0.03 |
| nPsaA−CT vs nPsaA | NSa | <0.01 | 0.03 | <0.001 | <0.01 |
| ePsaA−CT vs ePsaA | <0.01 | <0.001 | <0.005 | <0.005 | NS |
| ePsaA−CT vs nPsaA-CT | <0.01 | <0.001 | <0.005 | <0.005 | NS |
| nPsaA−CTB vs nPsaA | NS | NS | NS | NS | NS |
| ePsaA−CTB vs ePsaA | NS | <0.005 | <0.001 | 0.02 | 0.02 |
| ePsaA−CTB vs ePsaA-CT | NS | 0.01 | NS | NS | NS |
| e(PsaA−CTB)1 vs nPsaA-CTB | <0.001 | NS | <0.01 | <0.001 | <0.01 |
| e(PsaA−CTB)2 vs nPsaA-CTB | <0.001 | NS | NS | <0.001 | <0.001 |
| e(PsaA−CTB)3 vs nPsaA-CTB | <0.001 | NS | <0.001 | <0.001 | <0.001 |
| e(PsaA−CTB)1 vs ePsaA-CTB | <0.001 | NS | BSb | BS | BS |
| e(PsaA−CTB)2 vs ePsaA-CTB | <0.001 | <0.01 | NS | <0.05 | <0.001 |
| e(PsaA−CTB)3 vs ePsaA-CTB | <0.001 | <0.05 | <0.001 | <0.001 | <0.001 |
| e(PsaA−CTB)3 vs e(PsaA-CTB)1 | BS | BS | <0.001 | <0.001 | <0.001 |
| e(PsaA−CTB)3 vs e(PsaA-CTB)2 | NS | NS | <0.01 | <0.001 | <0.001 |
NS, not significant (P ≥ 0.05).
BS, borderline significance (0.05 < P ≤ 0.1).
The effects of microencapsulation on the immunogenicity of orally administered rPsaA were evaluated after the oral immunization of BALB/c mice with nPsaA (group 2) or with ePsaA (group 3). The ePsaA induced serum IgM (P = 0.005), serum IgG (P < 0.001), serum IgA (P = 0.006), and mucosal IgA (P = 0.03) responses that were significantly higher than those induced by nPsaA.
To determine whether CT or CTB could enhance systemic and mucosal immune responses against rPsaA, CT or CTB was coadministered with vaccines. When CT was coadministered orally with nPsaA (group 4), the rPsaA-specific serum IgG, IgA, and mucosal IgA responses of mice significantly increased (P < 0.05). Moreover, the ePsaA-CT (group 5) induced more prominent serum antibody and bronchoalveolar IgA responses than those induced by ePsaA (group 3) or by nPsaA-CT (group 4) (P < 0.005). Coadministration of CTB with nPsaA (group 6), however, did not show significant differences compared to nPsaA (group 2). Mice immunized with ePsaA-CTB (group 7) yielded higher serum IgG, IgA, and mucosal IgA responses than those immunized with ePsaA (group 3) (P < 0.05). The systemic and mucosal antibody responses were not statistically different between group 5 (ePsaA-CT) and group 7 (ePsaA-CTB).
We evaluated the effects of CTB after coencapsulation with rPsaA. All e(PsaA-CTB) elicited serum IgM, serum IgA, bronchoalveolar IgA, and intestinal IgA responses significantly higher than those induced by mixed ePsaA-CTB (Tables 1 and 2). The mucosal IgA responses induced by e(PsaA-CTB)3 were always higher than those induced by ePsaA-CTB (P < 0.001). On the other hand, serum IgG responses induced by e(PsaA-CTB)3 were significantly reduced (P < 0.05) compared with those induced by ePsaA-CTB. e(PsaA-CTB)3, which contains the highest concentration of CTB, induced the most prominent mucosal IgA and serum IgA responses among the e(PsaA-CTB) groups (P < 0.01). However, serum IgG responses induced by e(PsaA-CTB)3 were lower than those induced by e(PsaA-CTB)1 (P < 0.1).
Homotypic protection study.
Mice were immunized with various formulations of antigen, and protection was evaluated by challenge with live S. pneumoniae type 19F (Table 3). The number of viable pneumococci recovered from the lung lavage fluids or from the blood of mice in all groups was significantly reduced (<0.01) when compared with that of mice immunized with mock antigen (Table 4). The extent of inhibition (EI) [100 × (1 − CFUtest/CFUcontrol)] of bacterial colonization in the bronchoalveoli or of bacteremia was analyzed. The EIs of bacterial colonization in the lungs of mice immunized with ePsaA, nPsaA-CTB, ePsaA-CTB, e(PsaA-CTB)1, e(PsaA-CTB)2, and e(PsaA-CTB)3 were 85, 84, 88, 96, 95, and 86%, respectively. Bacteremia was also inhibited, by 91, 99, 96, 100, 97, and 83%, respectively. The differences in EIs between the groups showed no statistical significance, except for some groups shown in Table 4. The best protection was achieved by immunization with e(PsaA-CTB)1. Although the mucosal IgA responses induced by e(PsaA-CTB)1 were lower than those induced by e(PsaA-CTB)3, the mice orally immunized with e(PsaA-CTB)1 were better protected than those immunized with e(PsaA-CTB)3.
TABLE 3.
Homotypic protection of mice after oral immunization and challenge infection with pneumococcus type 19F
| Vaccine | No. of mice | Concn (CFU/ml, mean ± SEM) of S. pneumoniae from:
|
|
|---|---|---|---|
| Bronchoalveoli | Blood | ||
| Mock | 5 | 4,140 ± 935 | 906 ± 188 |
| ePsaA | 15 | 554 ± 186 | 67 ± 41 |
| nPsaA−CTB | 8 | 570 ± 287 | 11 ± 7 |
| ePsaA−CTB | 8 | 425 ± 193 | 33 ± 26 |
| e(PsaA−CTB)1 | 7 | 140 ± 63 | 3 ± 1 |
| e(PsaA−CTB)2 | 7 | 176 ± 169 | 23 ± 15 |
| e(PsaA−CTB)3 | 9 | 510 ± 255 | 132 ± 79 |
TABLE 4.
Statistical significance of differences between the groups of mice shown in 3
| Vaccines compared | Significancea
|
|
|---|---|---|
| Lung | Blood | |
| ePsaA vs nPsaA−CTB | NSb | NS |
| ePsaA vs ePsaA−CTB | NS | NS |
| nPsaA−CTB vs ePsaA−CTB | NS | NS |
| e(PsaA−CTB)1 vs nPsaA−CTB | BSc | NS |
| e(PsaA−CTB)2 vs nPsaA−CTB | BS | NS |
| e(PsaA−CTB)3 vs nPsaA−CTB | NS | BS |
| e(PsaA−CTB)1 vs ePsaA−CTB | BS | NS |
| e(PsaA−CTB)2 vs ePsaA−CTB | NS | NS |
| e(PsaA−CTB)3 vs ePsaA−CTB | NS | BS |
| e(PsaA−CTB)3 vs e(PsaA−CTB)1 | BS | BS |
| e(PsaA−CTB)3 vs e(PsaA−CTB)2 | NS | BS |
The differences between the mock-immunized mice and other groups of mice were always statistically significant (P ≤ 0.01).
NS, not significant (P ≥ 0.05).
BS, borderline significance (0.05 < P ≤ 0.1).
Heterotypic protection study.
Cross-protection against challenge with five S. pneumoniae serotypes, i.e., serotypes 4, 6B, 14, 19F, and 23F, after oral vaccination was studied (Table 5). The protection against each strain was examined by comparing the number of viable pneumococci recovered from the lungs or blood of mice immunized with mock microspheres, nPsaA-CTB, or e(PsaA-CTB)1 (Table 5). The EIs for lung colonization conferred by nPsaA-CTB were 100% (serotype 4), 73% (serotype 6B), 96% (serotype 14), 84% (serotype 19F), and 100% (serotype 23F). The P values in Table 5 indicate the statistical significances of the differences in CFU between the test groups and the control group (mock-immunized mice). One hundred percent means that no colonies were recovered from lavage fluid or blood diluted 10,000-fold. The EIs for lung colonization conferred by e(PsaA-CTB)1 were 100% (serotype 4), 100% (serotype 6B), 100% (serotype 14), 96% (serotype 19F), and 100% (serotype 23F). After immunization with nPsaA-CTB, the bacteremia was inhibited by 100% (serotype 4), 83% (serotype 6B), 85% (serotype 14), 99% (serotype 19F), and 88% (serotype 23F). In addition, immunization with e(PsaA-CTB)1 conferred inhibition of bacteremia by 100% (serotype 4), 99% (serotype 6B), 95% (serotype 14), 100% (serotype 19F), and 100% (serotype 23F). Generally, e(PsaA-CTB)1 provided better heterotypic protection than nPsaA-CTB did.
TABLE 5.
Heterotypic protection of mice after oral immuization and challenge infection with various serotypes of S. pneumoniae
| Pneumococcal serotype | Vaccine | CFU/ml (mean ± SEM) from:
|
|
|---|---|---|---|
| Bronchoalveoli | Blood | ||
| 4 | Mock | (4 ± 1.6) × 107 | (7 ± 0.5) × 107 |
| nPsaA−CTB | (3 ± 2.0) × 104 (0.01)a | (5 ± 1.6) × 103 (<0.001) | |
| e(PsaA−CTB)1 | (6 ± 1.0) × 102 (0.01) | (3 ± 1.4) × 103 (<0.001) | |
| 6B | Mock | (2 ± 0.8) × 105 | (4 ± 3.0) × 104 |
| nPsaA−CTB | (3 ± 2.3) × 104 (<0.05) | (3 ± 1.8) × 103 (NSb) | |
| e(PsaA−CTB)1 | (3 ± 1.9) × 10 (0.03) | (2 ± 1.6) × 102 (BSc) | |
| 14 | Mock | (3 ± 1.2) × 107 | (3 ± 0.7) × 103 |
| nPsaA−CTB | (1 ± 0.5) × 106 (0.01) | (4 ± 1.1) × 102 (0.004) | |
| e(PsaA−CTB)1 | (8 ± 3.6) × 104 (0.01) | (1 ± 0.7) × 102 (0.002) | |
| 19F | Mock | (4 ± 0.9) × 103 | (9 ± 1.8) × 102 |
| nPsaA−CTB | (6 ± 2.9) × 102 (<0.001) | (1 ± 0.7) × 10 (<0.001) | |
| e(PsaA−CTB)1 | (1 ± 0.6) × 102 (<0.001) | (3 ± 01.8) × 1 (<0.001) | |
| 23F | Mock | (2 ± 1.1) × 108 | (1 ± 0.3) × 104 |
| nPsaA−CTB | (3 ± 2.4) × 105 (0.04) | (1 ± 1.2) × 103 (0.002) | |
| e(PsaA−CTB)1 | (2 ± 1.0) × 103 (0.04) | (2 ± 1.9) × 10 (<0.001) | |
The numbers in parentheses indicate statistical significance (P value) of the difference between the mock-immunized mice and test group of mice.
NS, not significant (P ≥ 0.1).
BS, borderline significance (0.05 < P ≤ 0.1).
DISCUSSION
The increase of drug-resistant pneumococci poses an urgent need for new control methods. Clearly, the global prevalence of drug-resistant pneumococci demands the development and marketing of new vaccines, although more widespread use of the existing 23-valent polysaccharide vaccine among high-risk populations is essential in the interim. Immunization with a pneumococcal vaccine is important to protect immunocompromised patients from pneumonia but is often unsuccessful. A significant breakthrough in the development of pneumococcal vaccines would be a mucosal vaccine providing long-lasting mucosal as well as systemic immune responses and conferring cross-protective immunity against all of the serotypes of S. pneumoniae.
In this study, we evaluated rPsaA in terms of mucosal and systemic humoral immune responses as well as its ability to induce cross-protective immunity against several serotypes of pneumococci. Many delivery systems for oral administration of antigens are being studied (5, 27). In this study, the preparation of rPsaA in alginate microspheres resulted in higher levels of antigen-specific IgM, IgG, and IgA antibodies than those produced by nPsaA in bicarbonate buffer.
Antibody responses induced by oral immunization with rPsaA were enhanced by use of CT or CTB. Immunization with nPsaA and CT induced both systemic and mucosal immune responses significantly higher than those induced by nPsaA without CT and comparable to the immune responses induced by ePsaA. This finding suggests that the effect of microencapsulation in enhancing immunogenicity might be comparable to that of CT. nPsaA mixed with CTB did not induce antibody responses significantly higher than those of nPsaA without CTB. ePsaA mixed with CT or CTB also induced more prominent immune responses than those of ePsaA without CT or CTB. Although CTB is reportedly a poorer adjuvant than CT (46), there was no significant difference in antibody levels between ePsaA-CT and ePsaA-CTB.
Mucosal immune responses induced by coencapsulated antigens (rPsaA and CTB) were higher than those induced by mixing singly encapsulated rPsaA and CTB. The higher the CTB content in the coencapsulated microsphere, the higher the mucosal IgA response was. On the other hand, systemic IgG and IgM levels were inversely proportional to the CTB content in coencapsulated microspheres. Although we do not know the exact mechanism, protective immunity was closely related to serum IgG levels and inversely related to mucosal IgA levels.
The immune exclusion and elimination at the mucosal surfaces by secretory IgA is crucial in preventing pneumococcal bacteremia (41). In addition, it has been suggested that the most important factor contributing to protection might be the specific serum IgG levels for the prevention of septicemia (3). Although the levels of antibodies that correlate with protection against pneumococcal disease have not been clearly defined, serum antibody responses and their opsonophagocytic activity represent the major defense mechanism of the host against pneumococcal infection (2). It seems that nasal immunization would have been a preferred route of immunization, since nasal immunization typically requires less antigen and adjuvant than gastric immunization for the induction of both systemic and mucosal immune responses. It also seems that nasal immunization may induce better protection against a challenge administered via the nasal route. In a previous study, we compared vaccine efficacies after introduction by the intranasal or oral route (40). We found that lung clearance was better in the group of mice immunized nasally than in those immunized orally. Although regurgitated antigen might prime immune cells in nose-associated lymphatic tissue (NALT), pneumococcal septicemia was profoundly inhibited only in the group of mice immunized orally. Nonetheless, we could not determine which route is better. Taken together, these results indicate that pneumococcal vaccines should be carefully analyzed in terms of protective immunity by using mice immunized orally as well as intranasally.
Following oral immunization with various antigens, protection was evaluated by intranasal challenge with various S. pneumoniae serotypes, i.e., types 19F, 4, 6B, 14, and 23F. The numbers of pneumococci in the lungs and blood of mice immunized with e(PsaA-CTB)1 were profoundly reduced compared to the control.
In summary, our data suggest that oral immunization with rPsaA coencapsulated with CTB elicits specific antibody responses and provides cross-protective immunity against various serotypes of pneumococci. They imply that oral vaccination with e(rPsaA-CTB) might be useful in populations with relatively higher mucosal immunocompetency than systemic immunocompetency, such as children under 2 years of age, the elderly, and AIDS patients.
Acknowledgments
This work was supported by grant 2V00451 from the KIST 2000 program.
REFERENCES
- 1.Alexander, J. E., R. A. Lock, C. C. Peeters, J. T. Poolman, P. W. Andrew, T. J. Mitchell, D. Hansman, and J. C. Paton. 1994. Immunization of mice with pneumolysin toxoid confers a significant degree of protection against at least nine serotypes of Streptococcus pneumoniae. Infect. Immun. 62:5683-5688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alonso De Velasco, E., B. A. Dekker, A. F. Verheul, R. G. Feldman, J. Verhoef, and H. Snippe. 1995. Anti-polysaccharide immunoglobulin isotype levels and opsonic activity of antisera: relationships with protection against Streptococcus pneumoniae infection in mice. J. Infect. Dis. 172:562-565. [DOI] [PubMed] [Google Scholar]
- 3.Anttila, M., M. Voutilainen, V. Jantti, J. Eskola, and H. Kayhty. 1999. Contribution of serotype-specific IgG concentration, IgG subclasses and relative antibody avidity to opsonophagocytic activity against Streptococcus pneumoniae. Clin. Exp. Immunol. 118:402-407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Artursson, P., I. L. Martensson, and I. Sjoholm. 1986. Biodegradable microspheres. III. Some immunological properties of polyacryl starch microparticles. J. Pharm. Sci. 75:697-701. [DOI] [PubMed] [Google Scholar]
- 5.Ben Ahmeida, E. T., G. Gregoriadis, C. W. Potter, and R. Jennings. 1993. Immunopotentiation of local and systemic humoral immune responses by ISCOMs, liposomes and FCA: role in protection against influenza A in mice. Vaccine 11:1302-1309. [DOI] [PubMed] [Google Scholar]
- 6.Berry, A. M., and J. C. Paton. 1996. Sequence heterogeneity of PsaA, a 37-kilodalton putative adhesin essential for virulence of Streptococcus pneumoniae. Infect. Immun. 64:5255-5262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Briles, D. E., E. Ades, J. C. Paton, J. S. Sampson, G. M. Carlone, R. C. Huebner, A. Virolainen, E. Swiatlo, and S. K. Hollingshead. 2000. Intranasal immunization of mice with a mixture of the pneumococcal proteins PsaA and PspA is highly protective against nasopharyngeal carriage of Streptococcus pneumoniae. Infect. Immun. 68:796-800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Briles, D. E., R. C. Tart, H. Y. Wu, B. A. Ralph, M. W. Russell, and L. S. McDaniel. 1996. Systemic and mucosal protective immunity to pneumococcal surface protein A. Ann. N.Y. Acad. Sci. 797:118-126. [DOI] [PubMed] [Google Scholar]
- 9.Butler, J. C., E. D. Shapiro, and G. M. Carlone. 1999. Pneumococcal vaccines: history, current status, and future directions. Am. J. Med. 107:69S-76S. [DOI] [PubMed]
- 10.Cho, N. H., S. Y. Seong, K. H. Chun, Y. H. Kim, I. C. Kwon, B. Y. Ahn, and S. Y. Jeong. 1998. Novel mucosal immunization with polysaccharide-protein conjugates entrapped in alginate microspheres. J. Control. Release 53:215-224. [DOI] [PubMed]
- 11.Chun, K. H., I. C. Kwon, Y. H. Kim, S. B. La, Y. T. Sohn, and S. Y. Jeong. 1996. Preparation of sodium alginate microspheres containing hydrophilic beta-lactam antibiotics. Arch. Pharm. Res. 19:106-116. [Google Scholar]
- 12.Cowan, M. J., A. J. Ammann, D. W. Wara, V. M. Howie, L. Schultz, N. Doyle, and M. Kaplan. 1978. Pneumococcal polysaccharide immunization in infants and children. Pediatrics 62:721-727. [PubMed] [Google Scholar]
- 13.Crook, J., J. A. Tharpe, S. E. Johnson, D. B. Williams, A. R. Stinson, R. R. Facklam, E. W. Ades, G. M. Carlone, and J. S. Sampson. 1998. Immunoreactivity of five monoclonal antibodies against the 37-kilodalton common cell wall protein (PsaA) of Streptococcus pneumoniae. Clin. Diagn. Lab Immunol. 5:205-210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Elson, C. O., W. Ealding, and J. Lefkowitz. 1984. A lavage technique allowing repeated measurement of IgA antibody in mouse intestinal secretions. J. Immunol. Methods 67:101-108. [DOI] [PubMed] [Google Scholar]
- 15.Eriksson, K., A. Kilander, L. Hagberg, G. Norkrans, J. Holmgren, and C. Czerkinsky. 1995. Intestinal immune responsiveness in HIV-infected individuals. Adv. Exp. Med. Biol. 371B:1011-1014. [PubMed]
- 16.Horan, M. A. 1993. Immunosenescence and mucosal immunity. Lancet 341:793-794. [DOI] [PubMed] [Google Scholar]
- 17.Illum, L. 1998. Chitosan and its use as a pharmaceutical excipient. Pharm. Res. 15:1326-1331. [DOI] [PubMed] [Google Scholar]
- 18.Janoff, E. N., J. M. Douglas, Jr., M. Gabriel, M. J. Blaser, A. J. Davidson, D. L. Cohn, and F. N. Judson. 1988. Class-specific antibody response to pneumococcal capsular polysaccharides in men infected with human immunodeficiency virus type 1. J. Infect. Dis. 158:983-990. [DOI] [PubMed] [Google Scholar]
- 19.Kuo, J., M. Douglas, H. K. Ree, and A. A. Lindberg. 1995. Characterization of a recombinant pneumolysin and its use as a protein carrier for pneumococcal type 18C conjugate vaccines. Infect. Immun. 63:2706-2713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kwok, K. K., M. J. Groves, and D. J. Burgess. 1991. Production of 5-15 microns diameter alginate-polylysine microcapsules by an air-atomization technique. Pharm. Res. 8:341-344. [DOI] [PubMed] [Google Scholar]
- 21.Lee, C. J., R. A. Lock, P. W. Andrew, T. J. Mitchell, D. Hansman, and J. C. Paton. 1994. Protection of infant mice from challenge with Streptococcus pneumoniae type 19F by immunization with a type 19F polysaccharide-pneumolysoid conjugate. Vaccine 12:875-878. [DOI] [PubMed] [Google Scholar]
- 22.Lewis, D. J., C. F. Gilks, S. Ojoo, L. R. Castello-Branco, G. Dougan, M. R. Evans, S. McDermott, and G. E. Griffin. 1994. Immune response following oral administration of cholera toxin B subunit to HIV-1-infected UK and Kenyan subjects. AIDS 8:779-785. [DOI] [PubMed] [Google Scholar]
- 23.Lock, R. A., J. C. Paton, and D. Hansman. 1988. Comparative efficacy of pneumococcal neuraminidase and pneumolysin as immunogens protective against Streptococcus pneumoniae. Microb. Pathog. 5:461-467. [DOI] [PubMed] [Google Scholar]
- 24.MacLeod, C. M., R. G. Hodge, M. Heidelberger, and W. G. Bernhard. 1945. Prevention of pneumococcal pneumonia by immunization with specific capsular polysaccharides. J. Exp. Med. 82:445-465. [PMC free article] [PubMed] [Google Scholar]
- 25.Marx, P. A., R. W. Compans, A. Gettie, J. K. Staas, R. M. Gilley, M. J. Mulligan, G. V. Yamshchikov, D. Chen, and J. H. Eldridge. 1993. Protection against vaginal SIV transmission with microencapsulated vaccine. Science 260:1323-1327. [DOI] [PubMed] [Google Scholar]
- 26.Mathiowitz, E., J. S. Jacob, Y. S. Jong, G. P. Carino, D. E. Chickering, P. Chaturvedi, C. A. Santos, K. Vijayaraghavan, S. Montgomery, M. Bassett, and C. Morrell. 1997. Biologically erodable microspheres as potential oral drug delivery systems. Nature 386:410-414. [DOI] [PubMed] [Google Scholar]
- 27.Moldoveanu, Z., M. Novak, W. Q. Huang, R. M. Gilley, J. K. Staas, D. Schafer, R. W. Compans, and J. Mestecky. 1993. Oral immunization with influenza virus in biodegradable microspheres. J. Infect. Dis. 167:84-90. [DOI] [PubMed] [Google Scholar]
- 28.Morris, W., M. C. Steinhoff, and P. K. Russell. 1994. Potential of polymer microencapsulation technology for vaccine innovation. Vaccine 12:5-11. [DOI] [PubMed] [Google Scholar]
- 29.Murray, H. W., R. A. Gellene, D. M. Libby, C. D. Rothermel, and B. Y. Rubin. 1985. Activation of tissue macrophages from AIDS patients: in vitro response of AIDS alveolar macrophages to lymphokines and interferon-gamma. J. Immunol. 135:2374-2377. [PubMed] [Google Scholar]
- 30.Obaro, S. K., R. A. Adegbola, W. A. Banya, and B. M. Greenwood. 1996. Carriage of pneumococci after pneumococcal vaccination. Lancet 348:271-272. [DOI] [PubMed] [Google Scholar]
- 31.Ogunniyi, A. D., R. L. Folland, D. E. Briles, S. K. Hollingshead, and J. C. Paton. 2000. Immunization of mice with combinations of pneumococcal virulence proteins elicits enhanced protection against challenge with Streptococcus pneumoniae. Infect. Immun. 68:3028-3033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.O'Hagan, D. T., H. Jeffery, M. J. Roberts, J. P. McGee, and S. S. Davis. 1991. Controlled release of microparticles for vaccine development. Vaccine 9:768-771. [DOI] [PubMed] [Google Scholar]
- 33.Opstad, N. L., C. L. Daley, J. R. Thurn, J. B. Rubins, C. Merrifield, P. C. Hopewell, and E. N. Janoff. 1995. Impact of Streptococcus pneumoniae bacteremia and human immunodeficiency virus type 1 on oral mucosal immunity. J. Infect. Dis. 172:566-570. [DOI] [PubMed] [Google Scholar]
- 34.Paton, J. C. 1998. Novel pneumococcal surface proteins: role in virulence and vaccine potential. Trends Microbiol. 6:85-88. [DOI] [PubMed] [Google Scholar]
- 35.Paton, J. C., R. A. Lock, C. J. Lee, J. P. Li, A. M. Berry, T. J. Mitchell, P. W. Andrew, D. Hansman, and G. J. Boulnois. 1991. Purification and immunogenicity of genetically obtained pneumolysin toxoids and their conjugation to Streptococcus pneumoniae type 19F polysaccharide. Infect. Immun. 59:2297-2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Peters, V. B., E. P. Diamant, D. S. Hodes, and C. O. Cimino. 1994. Impaired immunity to pneumococcal polysaccharide antigens in children with human immunodeficiency virus infection immunized with pneumococcal vaccine. Pediatr. Infect. Dis. J. 13:933-934. [DOI] [PubMed] [Google Scholar]
- 37.Rognum, T. O., S. Thrane, L. Stoltenberg, A. Vege, P. Brandtzaeg. 1992. Development of intestinal mucosal immunity in fetal life and the first postnatal months. Pediatr. Res. 32:145-149. [DOI] [PubMed] [Google Scholar]
- 38.Sampson, J. S., Z. Furlow, A. M. Whitney, D. Williams, R. Facklam, and G. M. Carlone. 1997. Limited diversity of Streptococcus pneumoniae PsaA among pneumococcal vaccine serotypes. Infect. Immun. 65:1967-1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schroder, U., and A. Stahl. 1984. Crystallized dextran nanospheres with entrapped antigen and their use as adjuvants. J. Immunol. Methods 70:127-132. [DOI] [PubMed] [Google Scholar]
- 40.Seong, S. Y., N. H. Cho, I. C. Kwon, and S. Y. Jeong. 1999. Protective immunity of microsphere-based mucosal vaccines against lethal intranasal challenge with Streptococcus pneumoniae. Infect. Immun. 67:3587-3592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Service, R. F. 1994. Triggering the first line of defense. Science 265:1522-1524. [DOI] [PubMed] [Google Scholar]
- 42.Shalaby, W. S. 1995. Development of oral vaccines to stimulate mucosal and systemic immunity: barriers and novel strategies. Clin. Immunol. Immunopathol. 74:127-134. [DOI] [PubMed] [Google Scholar]
- 43.Siber, G. R. 1994. Pneumococcal disease: prospects for a new generation of vaccines. Science 265:1385-1387. [DOI] [PubMed] [Google Scholar]
- 44.Szewczuk, M. R., R. J. Campbell, and L. K. Jung. 1981. Lack of age-associated immune dysfunction in mucosal-associated lymph nodes. J. Immunol. 126:2200-2204. [PubMed] [Google Scholar]
- 45.Talkington, D. F., B. G. Brown, J. A. Tharpe, A. Koenig, and H. Russell. 1996. Protection of mice against fatal pneumococcal challenge by immunization with pneumococcal surface adhesin A (PsaA). Microb. Pathog. 21:17-22. [DOI] [PubMed] [Google Scholar]
- 46.Van der Heijden, P. J., A. T. Bianchi, M. Dol, J. W. Pals, W. Stok, and B. A. Bokhout. 1991. Manipulation of intestinal immune responses against ovalbumin by cholera toxin and its B subunit in mice. Immunology 72:89-93. [PMC free article] [PubMed] [Google Scholar]
- 47.Weiser, J. N., R. Austrian, P. K. Sreenivasan, and H. R. Masure. 1994. Phase variation in pneumococcal opacity: relationship between colonial morphology and nasopharyngeal colonization. Infect. Immun. 62:2582-2589. [DOI] [PMC free article] [PubMed] [Google Scholar]