Abstract
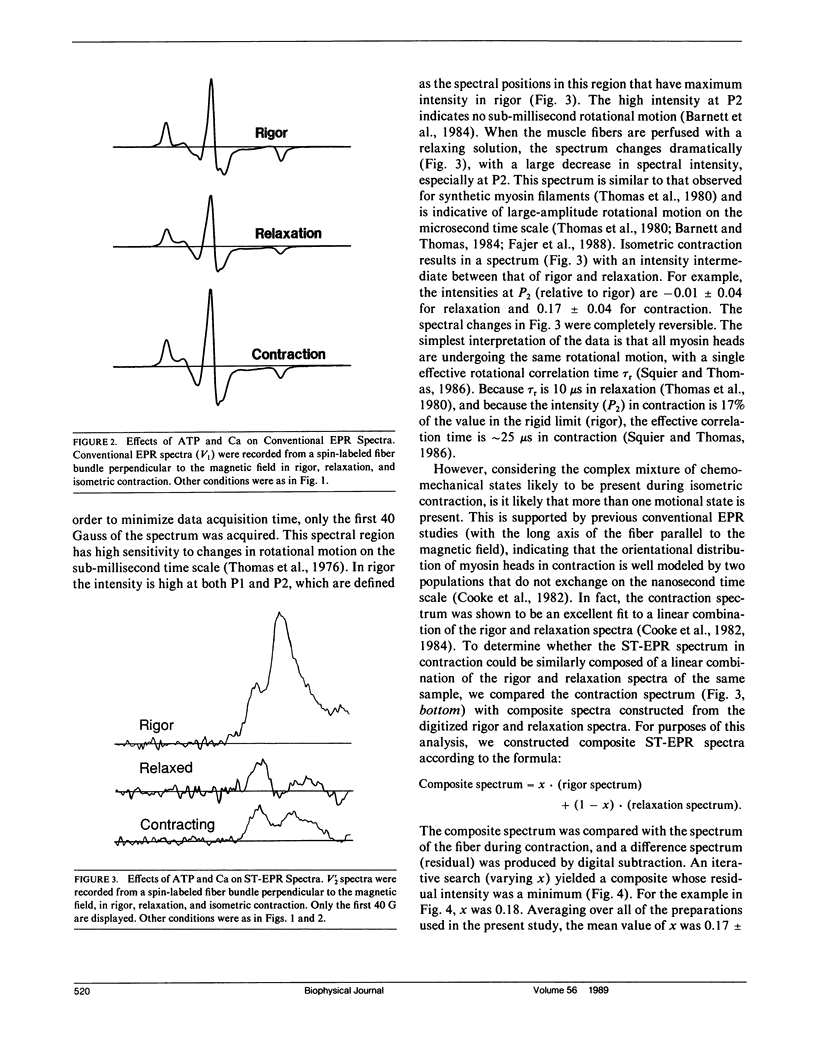
We have used saturation transfer electron paramagnetic resonance (ST-EPR) to detect the microsecond rotational motions of spin-labeled myosin heads in bundles of skinned muscle fibers, under conditions of rigor, relaxation, and isometric contraction. Experiments were performed on fiber bundles perfused continuously with an ATP-regenerating system. Conditions were identical to those we have used in previous studies of myosin head orientation, except that the fibers were perpendicular to the magnetic field, making the spectra primarily sensitive to rotational motion rather than to the orientational distribution. In rigor, the high intensity of the ST-EPR signal indicates the absence of microsecond rotational motion, showing that heads are all rigidly bound to actin. However, in both relaxation and contraction, considerable microsecond rotational motion is observed, implying that the previously reported orientational disorder under these conditions is dynamic, not static, on the microsecond time scale. The behavior in relaxation is essentially the same as that observed when myosin heads are detached from actin in the absence of ATP (Barnett and Thomas, 1984), corresponding to an effective rotational correlation time of approximately 10 microseconds. Slightly less mobility is observed during contraction. One possible interpretation is that in contraction all heads have the same mobility, corresponding to a correlation time of approximately 25 microseconds. Alternatively, more than one motional population may be present. For example, assuming that the spectrum in contraction is a linear combination of those in relaxation (mobile) and rigor (immobile), we obtained a good fit with a mole fraction of 78-88% of the heads in the mobile state. These results are consistent with previous STEPR studies on contracting myofibrils(Thomas et al., 1980). Thus most myosin heads undergo microsecond rotational motions most of the time during isometric contraction, at least in the probed region of the myosin head.These motions could arise primarily from the free rotations of heads detached from actin. However, if most of these heads are attached to actin during contraction, as suggested by stiffness measurements, this result provides support for the hypothesis that sub-millisecond rotational motions of actin-attached myosin heads play an important role in force generation.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barnett V. A., Fajer P., Polnaszek C. F., Thomas D. D. High-Resolution Detection of muscle Crossbridge Orientation by Electron Paramagnetic Resonance. Biophys J. 1986 Jan;49(1):144–147. doi: 10.1016/S0006-3495(86)83628-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnett V. A., Thomas D. D. Saturation transfer electron paramagnetic resonance of spin-labeled muscle fibers. Dependence of myosin head rotational motion on sarcomere length. J Mol Biol. 1984 Oct 15;179(1):83–102. doi: 10.1016/0022-2836(84)90307-3. [DOI] [PubMed] [Google Scholar]
- Burghardt T. P., Ajtai K. Fraction of myosin cross-bridges bound to actin in active muscle fibers: estimation by fluorescence anisotropy measurements. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8478–8482. doi: 10.1073/pnas.82.24.8478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooke R., Bialek W. Contraction of glycerinated muscle fibers as a function of the ATP concentration. Biophys J. 1979 Nov;28(2):241–258. doi: 10.1016/S0006-3495(79)85174-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooke R., Crowder M. S., Thomas D. D. Orientation of spin labels attached to cross-bridges in contracting muscle fibres. Nature. 1982 Dec 23;300(5894):776–778. doi: 10.1038/300776a0. [DOI] [PubMed] [Google Scholar]
- Cooke R., Crowder M. S., Wendt C. H., Barnett V. A., Thomas D. D. Muscle cross-bridges: do they rotate? Adv Exp Med Biol. 1984;170:413–427. doi: 10.1007/978-1-4684-4703-3_37. [DOI] [PubMed] [Google Scholar]
- Cooke R. The mechanism of muscle contraction. CRC Crit Rev Biochem. 1986;21(1):53–118. doi: 10.3109/10409238609113609. [DOI] [PubMed] [Google Scholar]
- Crowder M. S., Cooke R. The effect of myosin sulphydryl modification on the mechanics of fibre contraction. J Muscle Res Cell Motil. 1984 Apr;5(2):131–146. doi: 10.1007/BF00712152. [DOI] [PubMed] [Google Scholar]
- Fajer P. G., Fajer E. A., Brunsvold N. J., Thomas D. D. Effects of AMPPNP on the orientation and rotational dynamics of spin-labeled muscle cross-bridges. Biophys J. 1988 Apr;53(4):513–524. doi: 10.1016/S0006-3495(88)83131-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman Y. E., Simmons R. M. Active and rigor muscle stiffness [proceedings]. J Physiol. 1977 Jul;269(1):55P–57P. [PubMed] [Google Scholar]
- Haselgrove J. C. X-ray evidence for conformational changes in the myosin filaments of vertebrate striated muscle. J Mol Biol. 1975 Feb 15;92(1):113–143. doi: 10.1016/0022-2836(75)90094-7. [DOI] [PubMed] [Google Scholar]
- Huxley A. F., Simmons R. M. Proposed mechanism of force generation in striated muscle. Nature. 1971 Oct 22;233(5321):533–538. doi: 10.1038/233533a0. [DOI] [PubMed] [Google Scholar]
- Huxley H. E., Faruqi A. R., Bordas J., Koch M. H., Milch J. R. The use of synchrotron radiation in time-resolved X-ray diffraction studies of myosin layer-line reflections during muscle contraction. Nature. 1980 Mar 13;284(5752):140–143. doi: 10.1038/284140a0. [DOI] [PubMed] [Google Scholar]
- Huxley H. E., Kress M. Crossbridge behaviour during muscle contraction. J Muscle Res Cell Motil. 1985 Apr;6(2):153–161. doi: 10.1007/BF00713057. [DOI] [PubMed] [Google Scholar]
- Huxley H. E. The mechanism of muscular contraction. Science. 1969 Jun 20;164(3886):1356–1365. doi: 10.1126/science.164.3886.1356. [DOI] [PubMed] [Google Scholar]
- Ip W., Heuser J. Direct visualization of the myosin crossbridge helices on relaxed rabbit psoas thick filaments. J Mol Biol. 1983 Nov 25;171(1):105–109. doi: 10.1016/s0022-2836(83)80317-9. [DOI] [PubMed] [Google Scholar]
- Ishiwata S., Manuck B. A., Seidel J. C., Gergely J. Saturation transfer electron paramagnetic resonance study of the mobility of myosin heads in myofibrils under conditions of partial dissociation. Biophys J. 1986 Apr;49(4):821–828. doi: 10.1016/S0006-3495(86)83711-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manuck B. A., Seidel J. C., Gergely J. Single-headed binding of a spin-labeled-HMM-ADP complex to F-actin. Saturation transfer electron paramagnetic resonance and sedimentation studies. Biophys J. 1986 Aug;50(2):221–230. doi: 10.1016/S0006-3495(86)83456-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pate E., Cooke R. Energetics of the actomyosin bond in the filament array of muscle fibers. Biophys J. 1988 Apr;53(4):561–573. doi: 10.1016/S0006-3495(88)83136-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reedy M. K., Holmes K. C., Tregear R. T. Induced changes in orientation of the cross-bridges of glycerinated insect flight muscle. Nature. 1965 Sep 18;207(5003):1276–1280. doi: 10.1038/2071276a0. [DOI] [PubMed] [Google Scholar]
- Squier T. C., Thomas D. D. Methodology for increased precision in saturation transfer electron paramagnetic resonance studies of rotational dynamics. Biophys J. 1986 Apr;49(4):921–935. doi: 10.1016/S0006-3495(86)83720-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svensson E. C., Thomas D. D. ATP induces microsecond rotational motions of myosin heads crosslinked to actin. Biophys J. 1986 Nov;50(5):999–1002. doi: 10.1016/S0006-3495(86)83541-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas D. D., Cooke R. Orientation of spin-labeled myosin heads in glycerinated muscle fibers. Biophys J. 1980 Dec;32(3):891–906. doi: 10.1016/S0006-3495(80)85024-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas D. D., Fajer P. G., Fajer E. A., Brunsvold N. J., Svensson E. C. EPR evidence for nucleotide effects on attached cross-bridges. Adv Exp Med Biol. 1988;226:241–253. [PubMed] [Google Scholar]
- Thomas D. D., Ishiwata S., Seidel J. C., Gergely J. Submillisecond rotational dynamics of spin-labeled myosin heads in myofibrils. Biophys J. 1980 Dec;32(3):873–889. doi: 10.1016/S0006-3495(80)85023-5. [DOI] [PMC free article] [PubMed] [Google Scholar]



