Abstract
1. Voltage-clamp studies were carried out on single rabbit myelinated nerve fibres at 14 degrees C with the method of Dodge & Frankenhaeuser (1958). 2. A method was developed to allow the ionic currents through the modal membrane to be calibrated exactly under voltage-clamp conditions by measuring the resistance of the internode through which the current was injected. 3. The ionic currents in a rabbit node of Ranvier can be resolved into two components, a sodium current and a leak current. Potassium current is almost entirely absent. 4. The sodium currents in rabbit nodes were fitted by the Hodgkin-Huxley model using m2h kinetics. The kinetics of sodium currents in a rabbit node differ from that in a frog node under similar experimental conditions in two respects: (a) inactivation is faster, tau h for rabbit being 2-3 times smaller around -50 mV; (b) the P(Na) (E) curve for mammal is shifted 10-15 mV in the hyperpolarizing direction. 5. From the kinetics of sodium current, the non-propagating rabbit action potential was reconstructed at 14 degrees C. The transient inward sodium current is responsible for the fast initial depolarization phase of the action potential, while the repolarizing phase is accounted for by leak alone. The computed shape of the action potential was in good agreement with the experimentally obtained action potential. 6. At 14 degrees C, frog and rabbit nodes with similar diameters have similar measured gNa values.
Full text
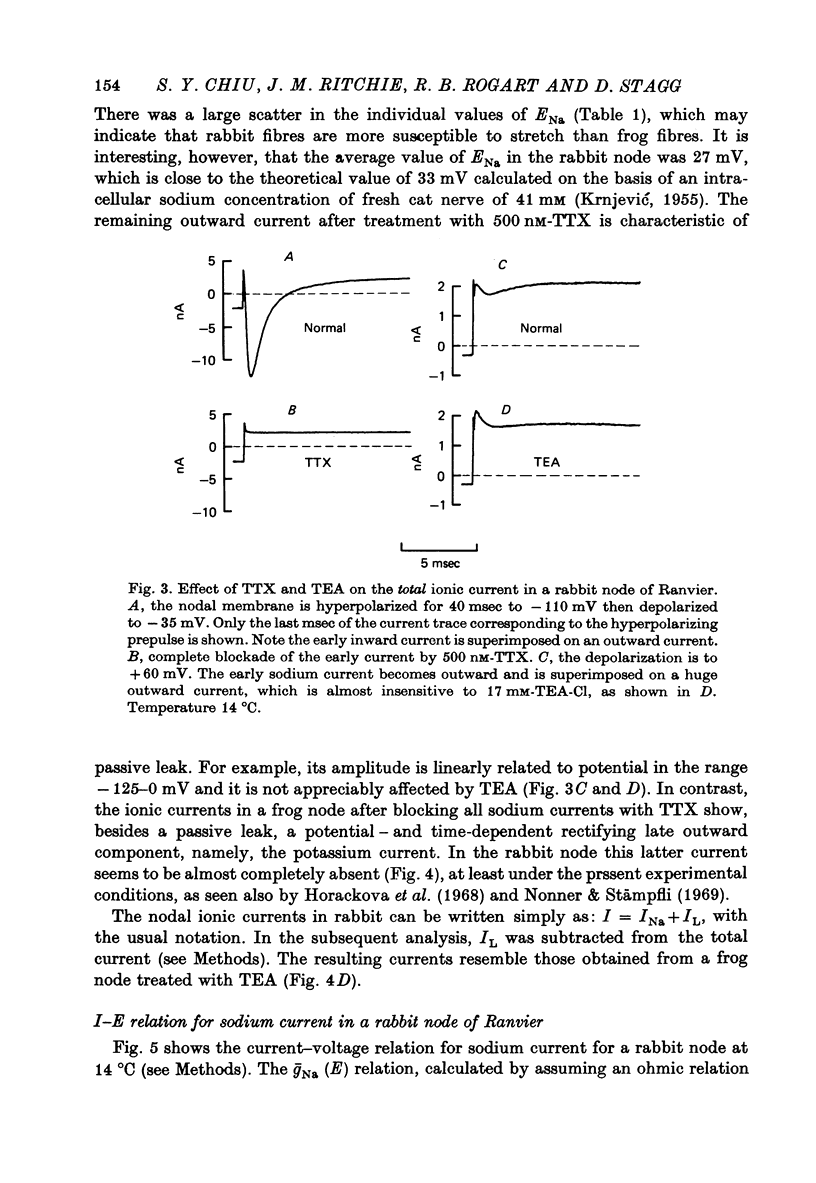
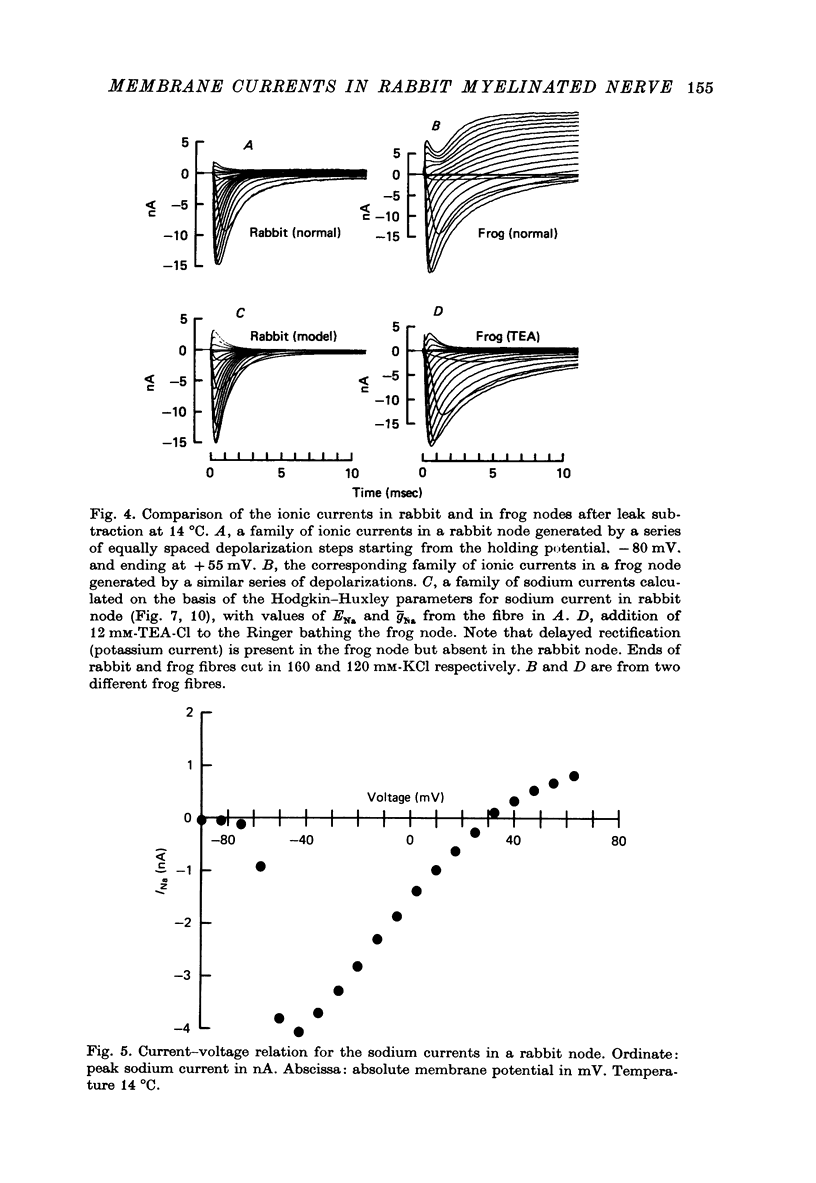
PDF



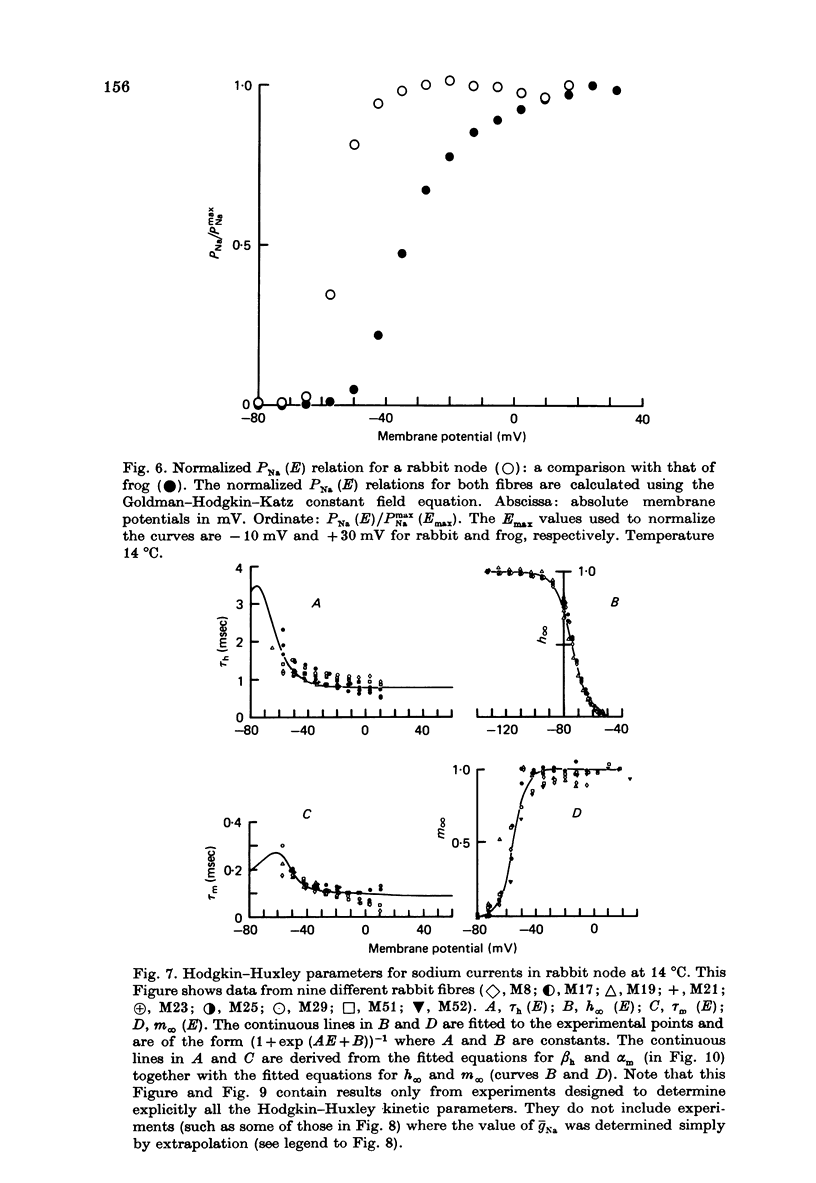






Selected References
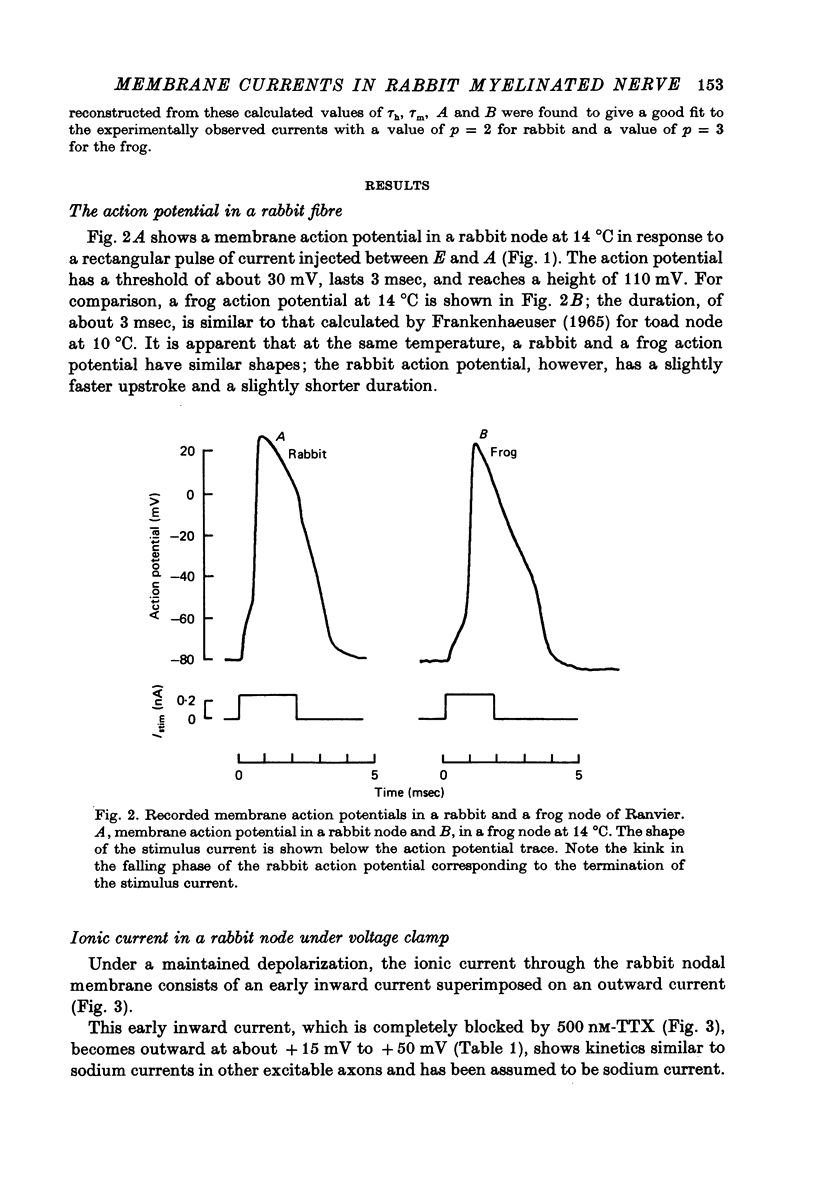
These references are in PubMed. This may not be the complete list of references from this article.
- Adrian R. H., Marshall M. W. Sodium currents in mammalian muscle. J Physiol. 1977 Jun;268(1):223–250. doi: 10.1113/jphysiol.1977.sp011855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M. Time course of TEA(+)-induced anomalous rectification in squid giant axons. J Gen Physiol. 1966 Nov;50(2):491–503. doi: 10.1085/jgp.50.2.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bostock H., Sherratt R. M., Sears T. A. Overcoming conduction failure in demyelinated nerve fibres by prolonging action potentials. Nature. 1978 Jul 27;274(5669):385–387. doi: 10.1038/274385a0. [DOI] [PubMed] [Google Scholar]
- Burg D. Untersuchungen am Ranvierschen Schnürring einzelner Taubennervenfasern. Pflugers Arch. 1970;317(3):278–286. doi: 10.1007/BF00586509. [DOI] [PubMed] [Google Scholar]
- Campbell D. T., Hille B. Kinetic and pharmacological properties of the sodium channel of frog skeletal muscle. J Gen Physiol. 1976 Mar;67(3):309–323. doi: 10.1085/jgp.67.3.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandler W. K., Meves H. Rate constants associated with changes in sodium conductance in axons perfused with sodium fluoride. J Physiol. 1970 Dec;211(3):679–705. doi: 10.1113/jphysiol.1970.sp009299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu S. Y. Inactivation of sodium channels: second order kinetics in myelinated nerve. J Physiol. 1977 Dec;273(3):573–596. doi: 10.1113/jphysiol.1977.sp012111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conti F., Hille B., Neumcke B., Nonner W., Stämpfli R. Measurement of the conductance of the sodium channel from current fluctuations at the node of Ranvier. J Physiol. 1976 Nov;262(3):699–727. doi: 10.1113/jphysiol.1976.sp011616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DODGE F. A., FRANKENHAEUSER B. Membrane currents in isolated frog nerve fibre under voltage clamp conditions. J Physiol. 1958 Aug 29;143(1):76–90. doi: 10.1113/jphysiol.1958.sp006045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DODGE F. A., FRANKENHAEUSER B. Sodium currents in the myelinated nerve fibre of Xenopus laevis investigated with the voltage clamp technique. J Physiol. 1959 Oct;148:188–200. doi: 10.1113/jphysiol.1959.sp006281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FRANKENHAEUSER B., HUXLEY A. F. THE ACTION POTENTIAL IN THE MYELINATED NERVE FIBER OF XENOPUS LAEVIS AS COMPUTED ON THE BASIS OF VOLTAGE CLAMP DATA. J Physiol. 1964 Jun;171:302–315. doi: 10.1113/jphysiol.1964.sp007378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FRANKENHAEUSER B. Quantitative description of sodium currents in myelinated nerve fibres of Xenopus laevis. J Physiol. 1960 Jun;151:491–501. doi: 10.1113/jphysiol.1960.sp006455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frankenhaeuser B. Computed action potential in nerve from Xenopus laevis. J Physiol. 1965 Oct;180(4):780–787. doi: 10.1113/jphysiol.1965.sp007731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman L., Schauf C. L. Quantitative description of sodium and potassium currents and computed action potentials in Myxicola giant axons. J Gen Physiol. 1973 Mar;61(3):361–384. doi: 10.1085/jgp.61.3.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952 Aug;117(4):500–544. doi: 10.1113/jphysiol.1952.sp004764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JULIAN F. J., MOORE J. W., GOLDMAN D. E. Current-voltage relations in the lobster giant axon membrane under voltage clamp conditions. J Gen Physiol. 1962 Jul;45:1217–1238. doi: 10.1085/jgp.45.6.1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shrager P. Ionic conductance changes in voltage clamped crayfish axons at low pH. J Gen Physiol. 1974 Dec;64(6):666–690. doi: 10.1085/jgp.64.6.666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulbricht W., Wagner H. H. The influence of pH on equilibrium effects of tetrodotoxin on myelinated nerve fibres of Rana esculenta. J Physiol. 1975 Oct;252(1):159–184. doi: 10.1113/jphysiol.1975.sp011139. [DOI] [PMC free article] [PubMed] [Google Scholar]


