Abstract
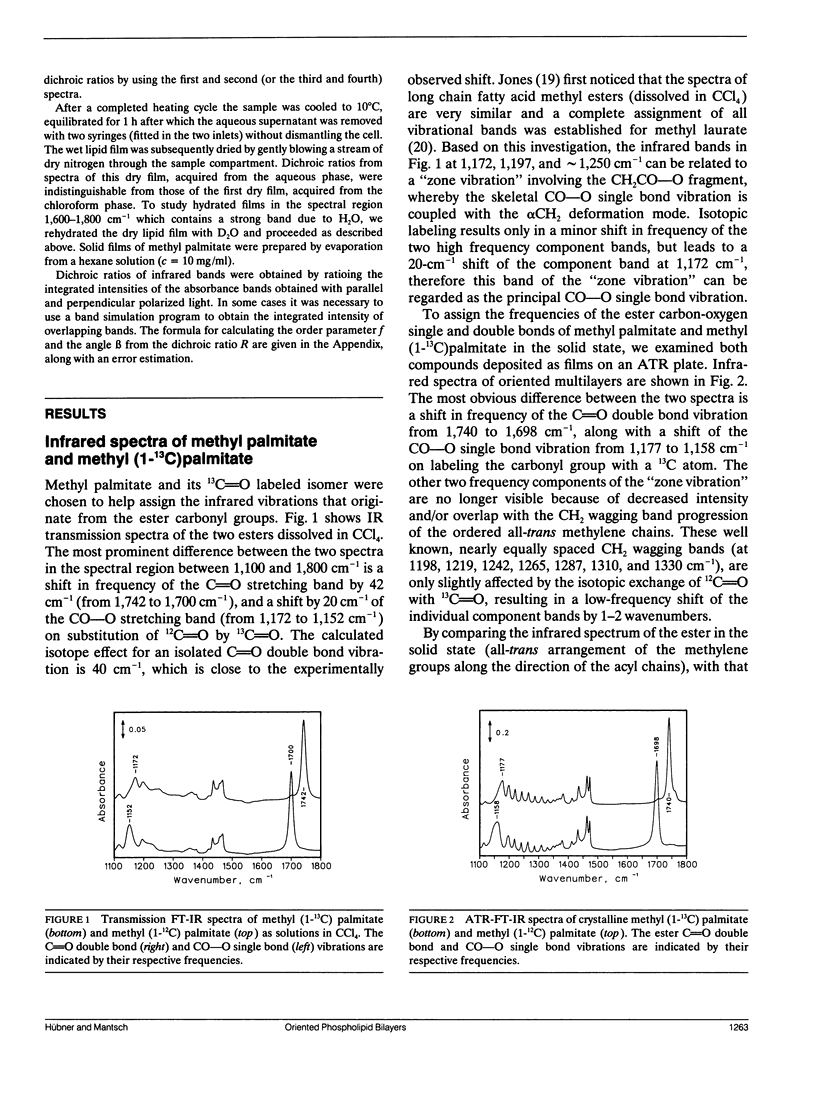
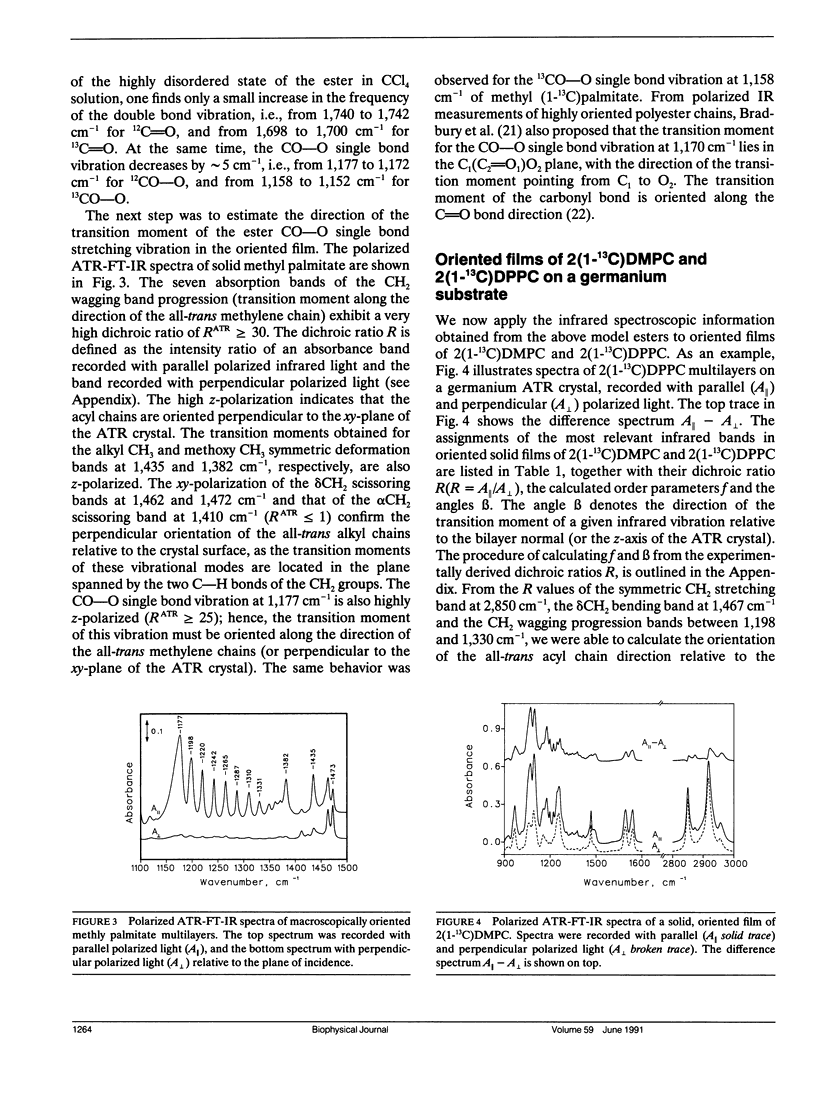
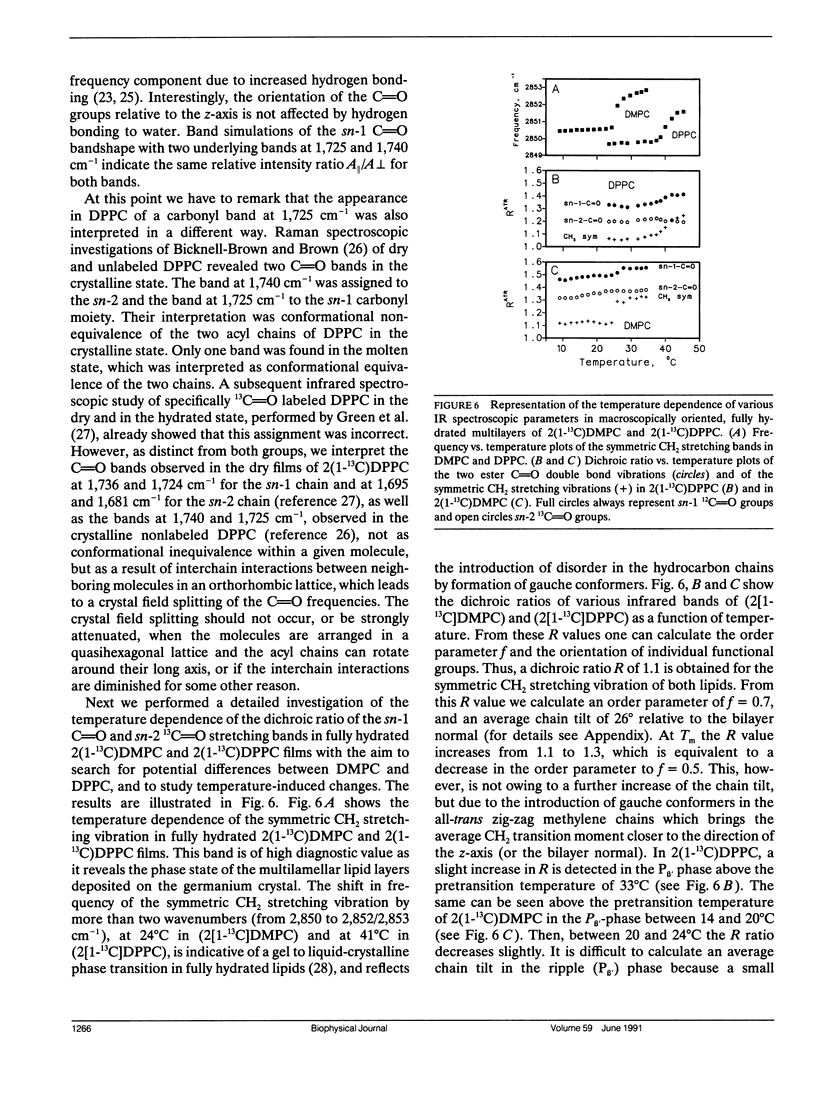
Oriented multilayers of 1-myristoyl-2(1-13C)-myristoyl-sn-glycero-3-phosphatidylcholine (2[1-13C]DMPC) and 1-palmitoyl-2(1-13C)-palmitoyl-sn-glycero-3-phosphatidylcholine (2[1-13C]DPPC) were investigated by use of attenuated total reflection infrared spectroscopy with polarized light. Experiments were performed with the aim to determine the orientation of the two ester groups in these phospholipids in the solid state and in the hydrated state at temperatures below and above the respective gel to liquid-crystalline phase transitions. Substitution of the naturally occurring 12C carbonyl carbon atom by 13C in the ester group of the sn-2 chain of DMPC and DPPC shifts the infrared absorption of the carbonyl double bond stretching vibration to lower frequency. This results in two well-resolved ester C=O bands which can be assigned unequivocally to the sn-1 and sn-2 chains as they are separated by more than 40 cm-1. The two ester CO-O single bond stretching vibrations of the molecular fragments-CH2CO-OC-are also affected and the corresponding infrared absorption band shifts by 20 cm-1 on 13C-labeling of the carbonyl carbon atom. From the dichroic ratios of the individual ester bands in 2(1-13C)DMPC and 2(1-13C)DPPC we were able to demonstrate that the sn-1 and sn-2 ester C=O groups are similarly oriented with respect to the bilayer plane, with an angle greater than or equal to 60 degrees relative to the bilayer normal. The two CO-O single bonds on the other hand have very different orientations. The CH2CO-OC fragment of the sn-1 chain is oriented along the direction of the all-trans methylene chain, whereas the same molecular segment of the sn-2 carbon chain is directed toward the bilayer plane. This orientation of the ester groups is retained in the liquid-crystalline phase. The tilt angle of the hydrocarbon all-trans chains, relative to the membrane normal, is 25 degrees in the solid state of DMPC and DPPC multibilayers. In the hydrated gel state this angle varies between 26 degrees and 30 degrees, depending on temperature. Neither the orientation of the phosphate group, nor that of the choline group varies significantly in the different physical states of these phospholipids.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blume A., Hübner W., Messner G. Fourier transform infrared spectroscopy of 13C = O-labeled phospholipids hydrogen bonding to carbonyl groups. Biochemistry. 1988 Oct 18;27(21):8239–8249. doi: 10.1021/bi00421a038. [DOI] [PubMed] [Google Scholar]
- Braach-Maksvytis V. L., Cornell B. A. Chemical shift anisotropies obtained from aligned egg yolk phosphatidylcholine by solid-state 13C nuclear magnetic resonance. Biophys J. 1988 May;53(5):839–843. doi: 10.1016/S0006-3495(88)83163-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Büldt G., Gally H. U., Seelig J., Zaccai G. Neutron diffraction studies on phosphatidylcholine model membranes. I. Head group conformation. J Mol Biol. 1979 Nov 15;134(4):673–691. doi: 10.1016/0022-2836(79)90479-0. [DOI] [PubMed] [Google Scholar]
- Büldt G., Seelig J. Conformation of phosphatidylethanolamine in the gel phase as seen by neutron diffraction. Biochemistry. 1980 Dec 23;19(26):6170–6175. doi: 10.1021/bi00567a034. [DOI] [PubMed] [Google Scholar]
- Casal H. L., Mantsch H. H. Polymorphic phase behaviour of phospholipid membranes studied by infrared spectroscopy. Biochim Biophys Acta. 1984 Dec 4;779(4):381–401. doi: 10.1016/0304-4157(84)90017-0. [DOI] [PubMed] [Google Scholar]
- Fringeli U. P., Günthard H. H. Infrared membrane spectroscopy. Mol Biol Biochem Biophys. 1981;31:270–332. doi: 10.1007/978-3-642-81537-9_6. [DOI] [PubMed] [Google Scholar]
- Fringeli U. P. The structure of lipids and proteins studied by attenuated total reflection (ATR) infrared spectroscopy. II. Oriented layers of a homologous series: phosphatidylethanolamine to phosphatidylcholine. Z Naturforsch C. 1977 Jan-Feb;32(1-2):20–45. doi: 10.1515/znc-1977-1-205. [DOI] [PubMed] [Google Scholar]
- Hauser H., Pascher I., Sundell S. Preferred conformation and dynamics of the glycerol backbone in phospholipids. An NMR and X-ray single-crystal analysis. Biochemistry. 1988 Dec 27;27(26):9166–9174. doi: 10.1021/bi00426a014. [DOI] [PubMed] [Google Scholar]
- Hitchcock P. B., Mason R., Thomas K. M., Shipley G. G. Structural chemistry of 1,2 dilauroyl-DL-phosphatidylethanolamine: molecular conformation and intermolecular packing of phospholipids. Proc Natl Acad Sci U S A. 1974 Aug;71(8):3036–3040. doi: 10.1073/pnas.71.8.3036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason J. T., Broccoli A. V., Huang C. A method for the synthesis of isomerically pure saturated mixed-chain phosphatidylcholines. Anal Biochem. 1981 May 1;113(1):96–101. doi: 10.1016/0003-2697(81)90049-x. [DOI] [PubMed] [Google Scholar]
- Okamura E., Umemura J., Takenaka T. Orientation studies of hydrated dipalmitoylphosphatidylcholine multibilayers by polarized FTIR-ATR spectroscopy. Biochim Biophys Acta. 1990 Jun 11;1025(1):94–98. doi: 10.1016/0005-2736(90)90195-t. [DOI] [PubMed] [Google Scholar]
- Pearson R. H., Pascher I. The molecular structure of lecithin dihydrate. Nature. 1979 Oct 11;281(5731):499–501. doi: 10.1038/281499a0. [DOI] [PubMed] [Google Scholar]
- Schindler H., Seelig J. Deuterium order parameters in relation to thermodynamic properties of a phospholiped bilayer. A statistical mechanical interpretation. Biochemistry. 1975 Jun 3;14(11):2283–2287. doi: 10.1021/bi00682a001. [DOI] [PubMed] [Google Scholar]
- Seelig A., Seelig J. Bilayers of dipalmitoyl-3-sn-phosphatidylcholine. Conformational differences between the fatty acyl chains. Biochim Biophys Acta. 1975 Sep 16;406(1):1–5. doi: 10.1016/0005-2736(75)90037-1. [DOI] [PubMed] [Google Scholar]
- Ter-Minassian-Saraga L., Okamura E., Umemura J., Takenaka T. Fourier transform infrared-attenuated total reflection spectroscopy of hydration of dimyristoylphosphatidylcholine multibilayers. Biochim Biophys Acta. 1988 Dec 22;946(2):417–423. doi: 10.1016/0005-2736(88)90417-8. [DOI] [PubMed] [Google Scholar]
- Wittebort R. J., Schmidt C. F., Griffin R. G. Solid-state carbon-13 nuclear magnetic resonance of the lecithin gel to liquid-crystalline phase transition. Biochemistry. 1981 Jul 7;20(14):4223–4228. doi: 10.1021/bi00517a042. [DOI] [PubMed] [Google Scholar]
- Zaccai G., Büldt G., Seelig A., Seelig J. Neutron diffraction studies on phosphatidylcholine model membranes. II. Chain conformation and segmental disorder. J Mol Biol. 1979 Nov 15;134(4):693–706. doi: 10.1016/0022-2836(79)90480-7. [DOI] [PubMed] [Google Scholar]


