Abstract
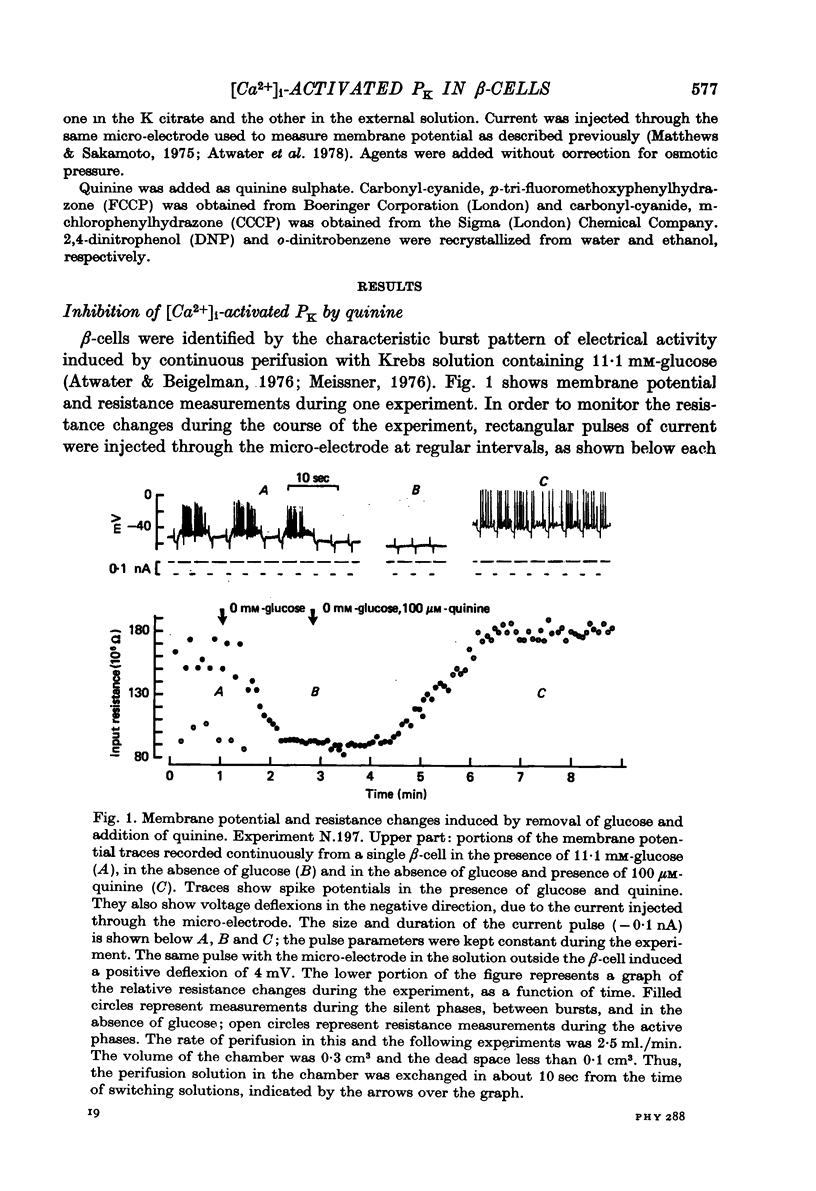
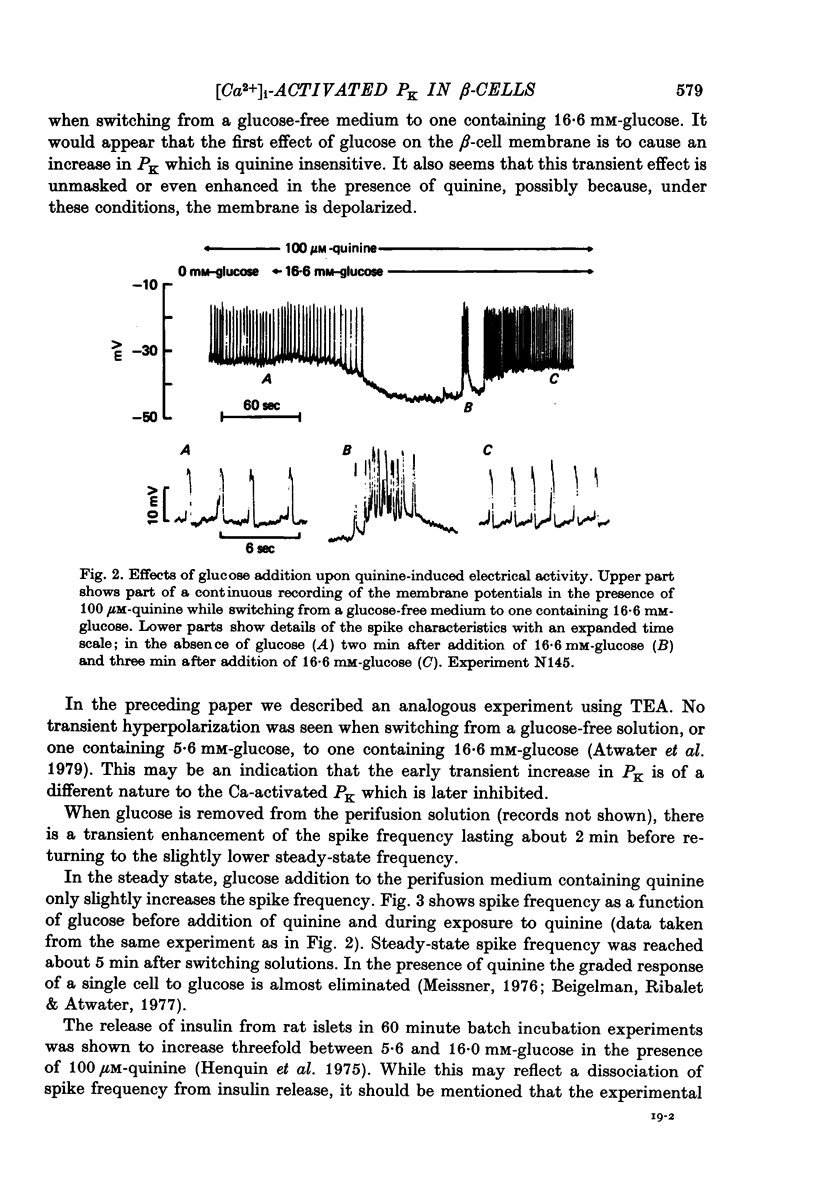
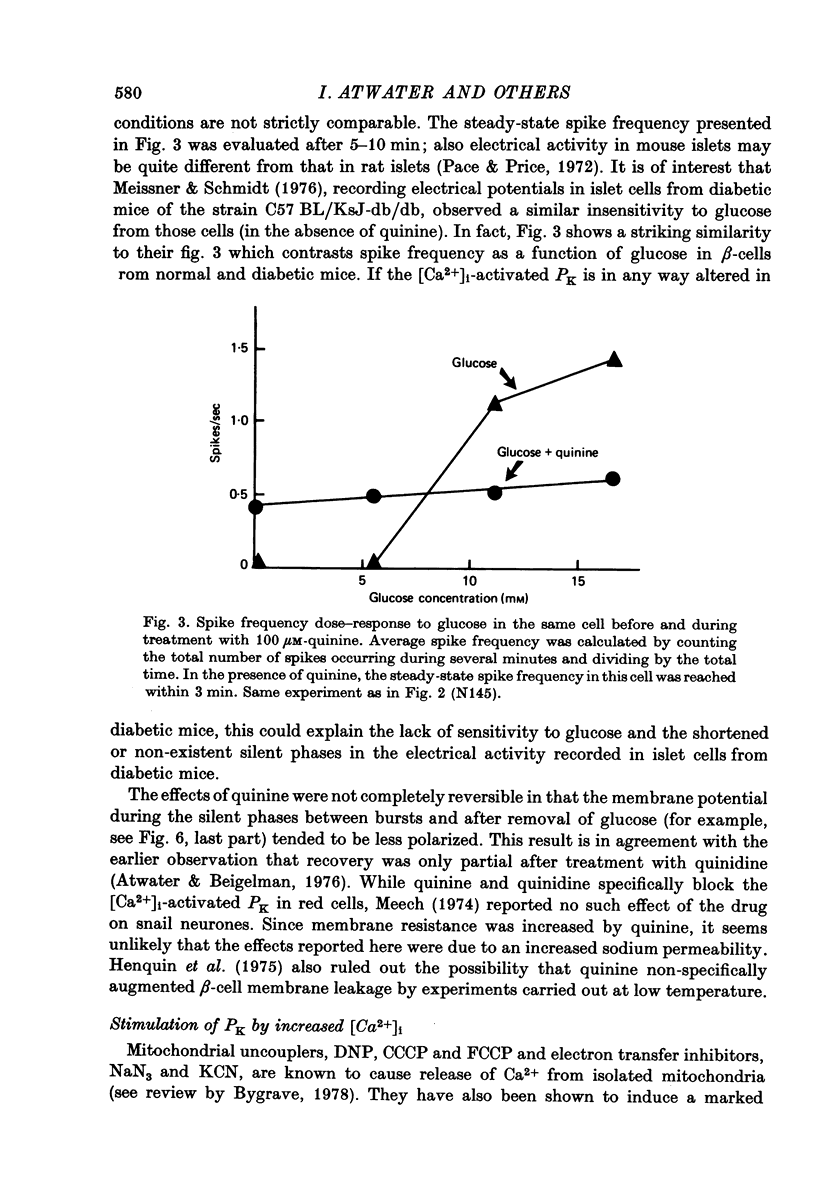
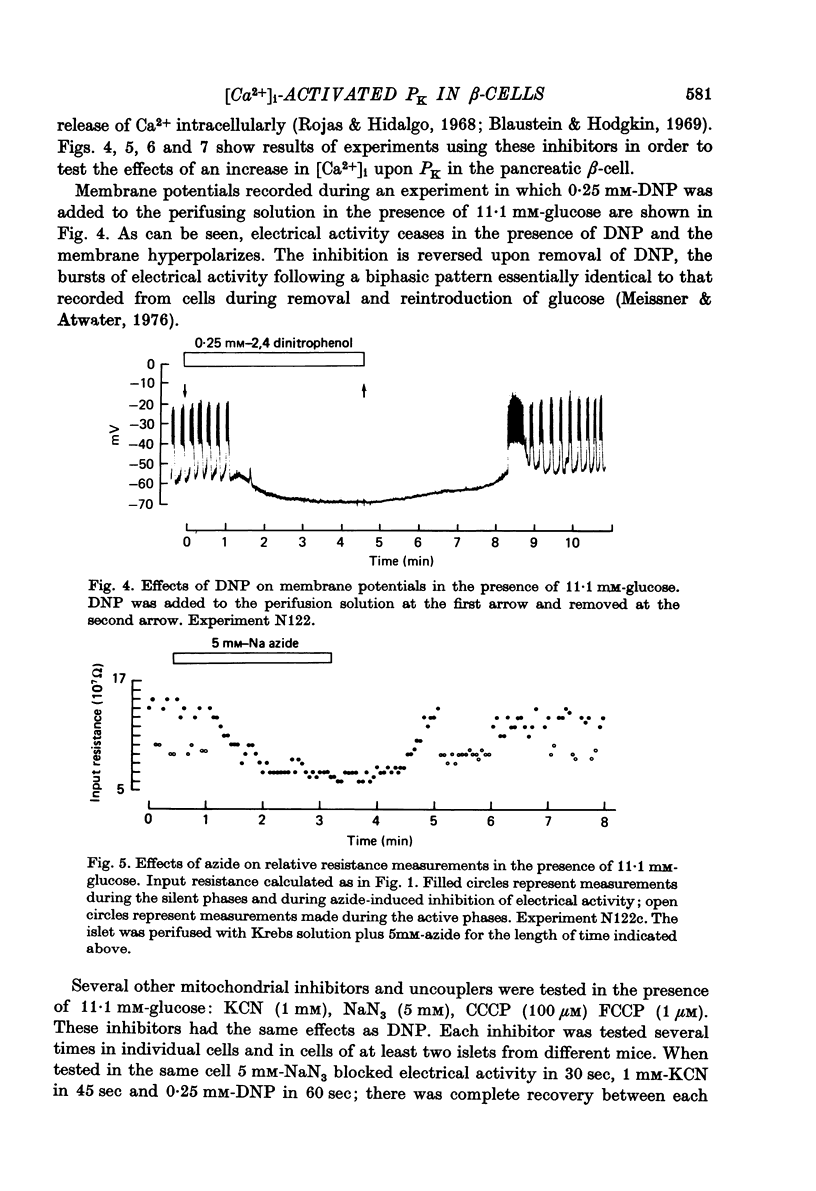
1. Membrane potentials and input resistance were measured in beta-cells from mouse pancreatic islets of Langerhans in a study designed to assess the role of a K permeability specifically blocked by quinine or quinidine and activated by intracellular calcium ion concentration ([Ca2+])i-activated PK). 2. Addition of 100 microM-quinine to the perifusion medium resulted in a 10--30 mV depolarization of the membrane and an increase in the input resistance of ca. 4.10(7) omega. 3. In the absence of glucose, 100 microM-quinine induced electrical activity. 4. In the presence of glucose, 100 microM-quinine abolished the burst pattern of electrical activity and very much reduced the graded response of spike frequency normally seen with different concentrations of glucose. 5. Addition of mitochondrial inhibitors, KCN, NaN3, DNP, CCCP, FCCP, to the perifusion medium containing glucose rapidly hyperpolarized the beta-cell membrane, inducing a concomitant decrease in input resistance. 6. In the presence of glucose, these mitochondrial inhibitors reversibly blocked electrical activity; upon removal of the inhibitor, recovery of electrical activity followed a biphasic pattern. 7. The effects of mitochondrial inhibitors were partially reversed by 100 microM-quinine. 8. It is proposed that the membrane potential of the beta-cell in the absence of glucose is predominantly controlled by the [Ca2+]i-activated PK. It is further suggested that this permeability to K controls the level for glucose stimulation and leads to the generation of the burst pattern.
Full text
PDF



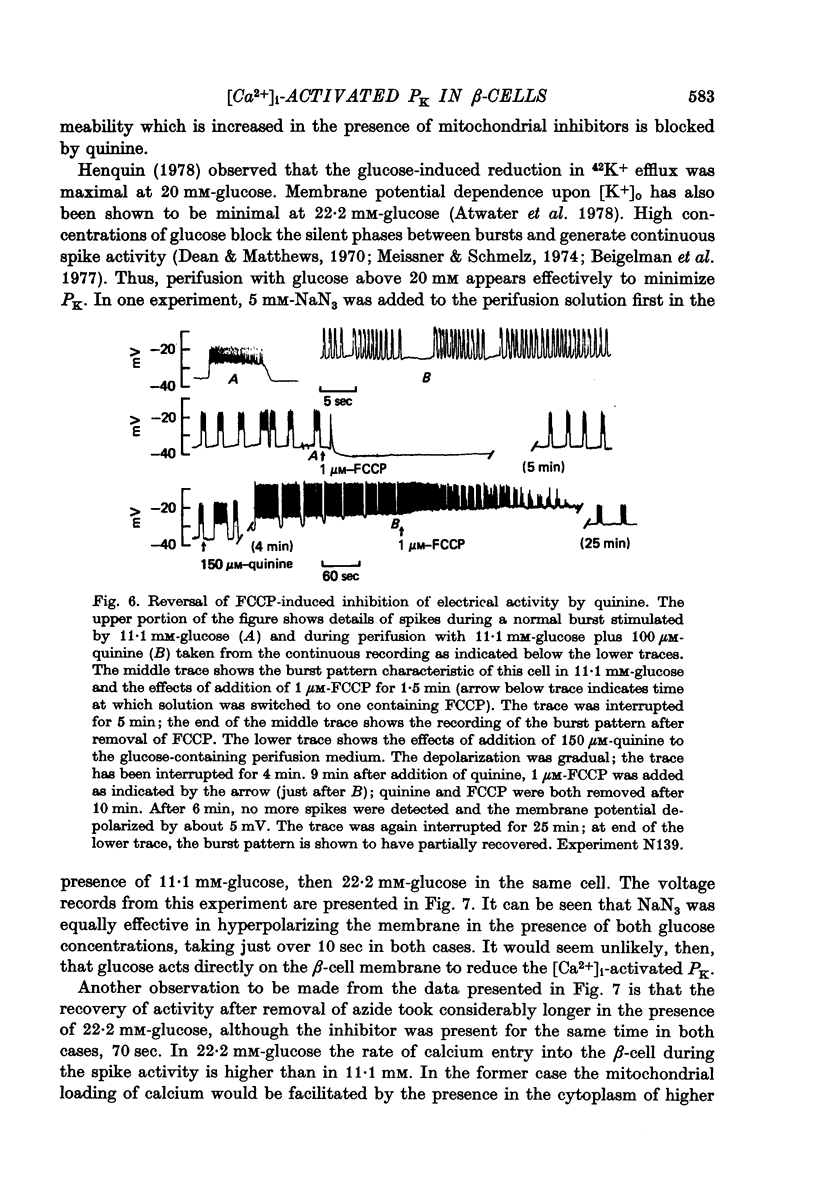
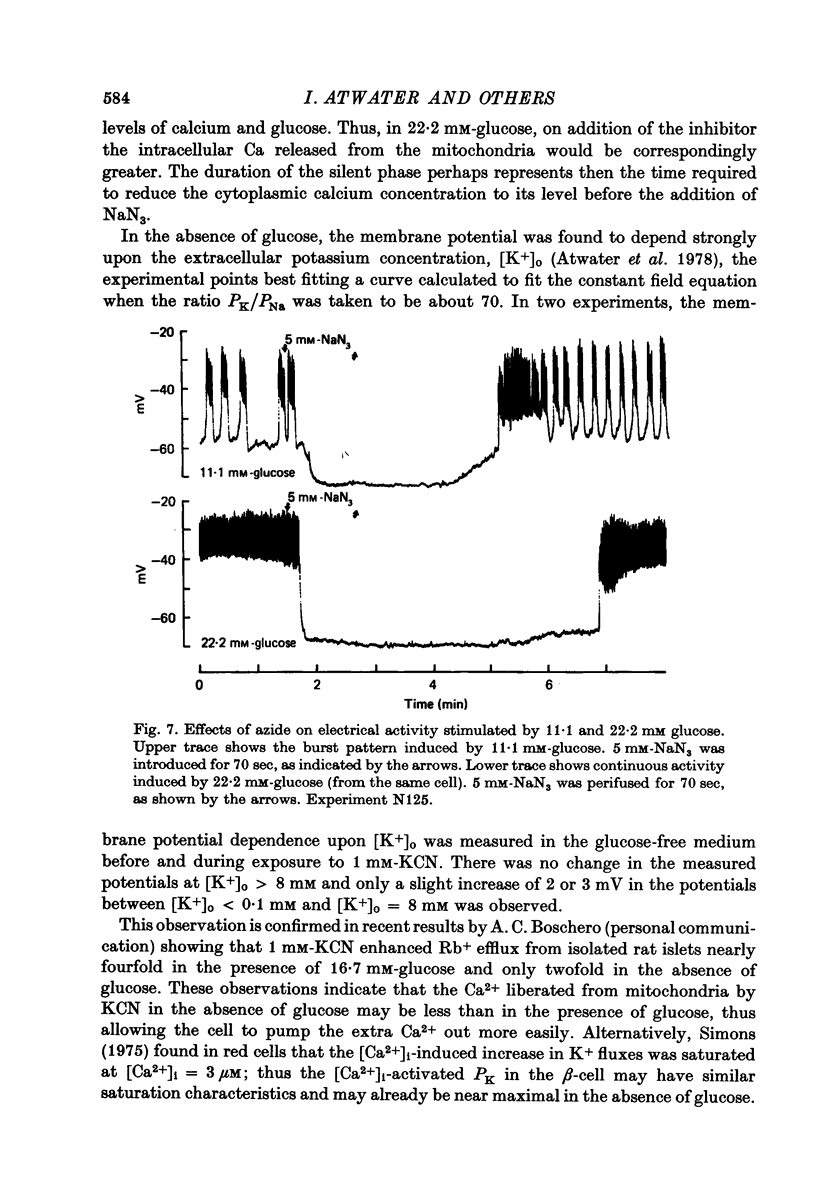




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ARMSTRONG C. M., BINSTOCK L. ANOMALOUS RECTIFICATION IN THE SQUID GIANT AXON INJECTED WITH TETRAETHYLAMMONIUM CHLORIDE. J Gen Physiol. 1965 May;48:859–872. doi: 10.1085/jgp.48.5.859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armando-Hardy M., Ellory J. C., Ferreira H. G., Fleminger S., Lew V. L. Inhibition of the calcium-induced increase in the potassium permeability of human red blood cells by quinine. J Physiol. 1975 Aug;250(1):32P–33P. [PubMed] [Google Scholar]
- Ashcroft S. J., Weerasinghe L. C., Randle P. J. Interrelationship of islet metabolism, adenosine triphosphate content and insulin release. Biochem J. 1973 Feb;132(2):223–231. doi: 10.1042/bj1320223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atwater I., Beigelman P. M. Dynamic characteristics of electrical activity in pancreatic beta-cells. I. - Effects of calcium and magnesium removal. J Physiol (Paris) 1976 Nov;72(6):769–786. [PubMed] [Google Scholar]
- Atwater I., Ribalet B., Rojas E. Cyclic changes in potential and resistance of the beta-cell membrane induced by glucose in islets of Langerhans from mouse. J Physiol. 1978 May;278:117–139. doi: 10.1113/jphysiol.1978.sp012296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atwater I., Ribalet B., Rojas E. Mouse pancreatic beta-cells: tetraethylammonium blockage of the potassium permeability increase induced by depolarization. J Physiol. 1979 Mar;288:561–574. [PMC free article] [PubMed] [Google Scholar]
- Beigelman P. M., Ribalet B., Atwater I. Electric activity of mouse pancreatic beta-cells. II. Effects of glucose and arginine. J Physiol (Paris) 1977 Jul;73(2):201–217. [PubMed] [Google Scholar]
- Blaustein M. P., Hodgkin A. L. The effect of cyanide on the efflux of calcium from squid axons. J Physiol. 1969 Feb;200(2):497–527. doi: 10.1113/jphysiol.1969.sp008704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boschero A. C., Kawazu S., Duncan G., Malaisse W. J. Effect of glucose on K+ handling by pancreatic islets. FEBS Lett. 1977 Nov 1;83(1):151–154. doi: 10.1016/0014-5793(77)80662-5. [DOI] [PubMed] [Google Scholar]
- Bygrave F. L. Mitochondria and the control of intracellular calcium. Biol Rev Camb Philos Soc. 1978 Feb;53(1):43–79. doi: 10.1111/j.1469-185x.1978.tb00992.x. [DOI] [PubMed] [Google Scholar]
- Croghan P. C., Lea E. J. On the mechanism of uncoupling of oxidative phosphorylation. J Physiol. 1972 Jul;224(1):32P–33P. [PubMed] [Google Scholar]
- Dean P. M., Matthews E. K. Glucose-induced electrical activity in pancreatic islet cells. J Physiol. 1970 Sep;210(2):255–264. doi: 10.1113/jphysiol.1970.sp009207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GARDOS G. The function of calcium in the potassium permeability of human erythrocytes. Biochim Biophys Acta. 1958 Dec;30(3):653–654. doi: 10.1016/0006-3002(58)90124-0. [DOI] [PubMed] [Google Scholar]
- Godfraind J. M., Krnjević K., Pumain R. Unexpected features of the action of dinitrophenol on cortical neurones. Nature. 1970 Nov 7;228(5271):562–564. doi: 10.1038/228562a0. [DOI] [PubMed] [Google Scholar]
- Gorman A. L., McReynolds J. S. Control of membrane N+ permeability in a hyperpolarizing photoreceptor: similar effect of light and metabolic inhibitors. Science. 1974 Aug 16;185(4151):620–621. doi: 10.1126/science.185.4151.620. [DOI] [PubMed] [Google Scholar]
- Grabowski W., Lobsiger E. A., Lüttgau H. C. The effect of repetitive stimulation at low frequencies upon the electrical and mechanical activity of single muscle fibres. Pflugers Arch. 1972;334(3):222–239. doi: 10.1007/BF00626225. [DOI] [PubMed] [Google Scholar]
- Hanstein W. G., Hatefi Y. Trinitrophenol: a membrane-impermeable uncoupler of oxidative phosphorylation. Proc Natl Acad Sci U S A. 1974 Feb;71(2):288–292. doi: 10.1073/pnas.71.2.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henquin J. C. D-glucose inhibits potassium efflux from pancreatic islet cells. Nature. 1978 Jan 19;271(5642):271–273. doi: 10.1038/271271a0. [DOI] [PubMed] [Google Scholar]
- Henquin J. C. Tetraethylammonium potentiation of insulin release and inhibition of rubidium efflux in pancreatic islets. Biochem Biophys Res Commun. 1977 Jul 25;77(2):551–556. doi: 10.1016/s0006-291x(77)80014-4. [DOI] [PubMed] [Google Scholar]
- Krnjević K., Lisiewicz A. Injections of calcium ions into spinal motoneurones. J Physiol. 1972 Sep;225(2):363–390. doi: 10.1113/jphysiol.1972.sp009945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lassen U. V., Pape L., Vestergaard-Bogind B. Effect of calcium on the membrane potential of Amphiuma red cells. J Membr Biol. 1976 Feb 17;26(1):51–70. doi: 10.1007/BF01868866. [DOI] [PubMed] [Google Scholar]
- Lew V. L. Effect of intracellular calcium on the potassium permeability of human red cells. J Physiol. 1970 Feb;206(2):35P–36P. [PubMed] [Google Scholar]
- Lew V. L., Ferreira H. G. Variable Ca sensitivity of a K-selective channel in intact red-cell membranes. Nature. 1976 Sep 23;263(5575):336–338. doi: 10.1038/263336a0. [DOI] [PubMed] [Google Scholar]
- Liberman E. A., Topaly V. P. Selective transport of ions through bimolecular phospholipid membranes. Biochim Biophys Acta. 1968 Sep 17;163(2):125–136. doi: 10.1016/0005-2736(68)90089-8. [DOI] [PubMed] [Google Scholar]
- Malaisse W. J., Boschero A. C., Kawazu S., Hutton J. C. The stimulus secretion coupling of glucose-induced insulin release. XXVII. Effect of glucose on K+ fluxes in isolated islets. Pflugers Arch. 1978 Mar 20;373(3):237–242. doi: 10.1007/BF00580830. [DOI] [PubMed] [Google Scholar]
- Matthews E. K., Sakamoto Y. Electrical characteristics of pancreatic islet cells. J Physiol. 1975 Mar;246(2):421–437. doi: 10.1113/jphysiol.1975.sp010897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meech R. W. The sensitivity of Helix aspersa neurones to injected calcium ions. J Physiol. 1974 Mar;237(2):259–277. doi: 10.1113/jphysiol.1974.sp010481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meissner H. P., Atwater I. J. The kinetics of electrical activity of beta cells in response to a "square wave" stimulation with glucose or glibenclamide. Horm Metab Res. 1976 Jan;8(1):11–16. doi: 10.1055/s-0028-1093685. [DOI] [PubMed] [Google Scholar]
- Meissner H. P. Electrical characteristics of the beta-cells in pancreatic islets. J Physiol (Paris) 1976 Nov;72(6):757–767. [PubMed] [Google Scholar]
- Meissner H. P., Schmelz H. Membrane potential of beta-cells in pancreatic islets. Pflugers Arch. 1974;351(3):195–206. doi: 10.1007/BF00586918. [DOI] [PubMed] [Google Scholar]
- Meissner H. P., Schmidt H. The electrical activity of pancreatic beta-cells of diabetic mice. FEBS Lett. 1976 Sep 1;67(3):371–374. doi: 10.1016/0014-5793(76)80567-4. [DOI] [PubMed] [Google Scholar]
- Pace C. S., Price S. Electrical responses of pancreatic islet cells to secretory stimuli. Biochem Biophys Res Commun. 1972 Feb 25;46(4):1557–1563. doi: 10.1016/0006-291x(72)90785-1. [DOI] [PubMed] [Google Scholar]
- Ransom B. R., Barker J. L., Nelson P. G. Two mechanisms for poststimulus hyperpolarisations in cultured mammalian neurones. Nature. 1975 Jul 31;256(5516):424–425. doi: 10.1038/256424a0. [DOI] [PubMed] [Google Scholar]
- Rojas E., Hidalgo C. Effect of temperature and metabolic inhibitors on 45Ca outflow from squid giant axons. Biochim Biophys Acta. 1968 Dec 10;163(4):550–556. doi: 10.1016/0005-2736(68)90084-9. [DOI] [PubMed] [Google Scholar]
- Sehlin J., Taljedal I. B. Glucose-induced decrease in Rb+ permeability in pancreatic beta cells. Nature. 1975 Feb 20;253(5493):635–636. doi: 10.1038/253635a0. [DOI] [PubMed] [Google Scholar]
- Simons T. J. Resealed ghosts used to study the effect of intracellular calcium ions on the potassium permeability of human red cell membranes. J Physiol. 1975 Mar;246(2):52P–54P. [PubMed] [Google Scholar]
- Vassort G. Voltage-clamp analysis of transmembrane ionic currents in guinea-pig myometrium: evidence for an initial potassium activation triggered by calcium influx. J Physiol. 1975 Nov;252(3):713–734. doi: 10.1113/jphysiol.1975.sp011167. [DOI] [PMC free article] [PubMed] [Google Scholar]


