Abstract
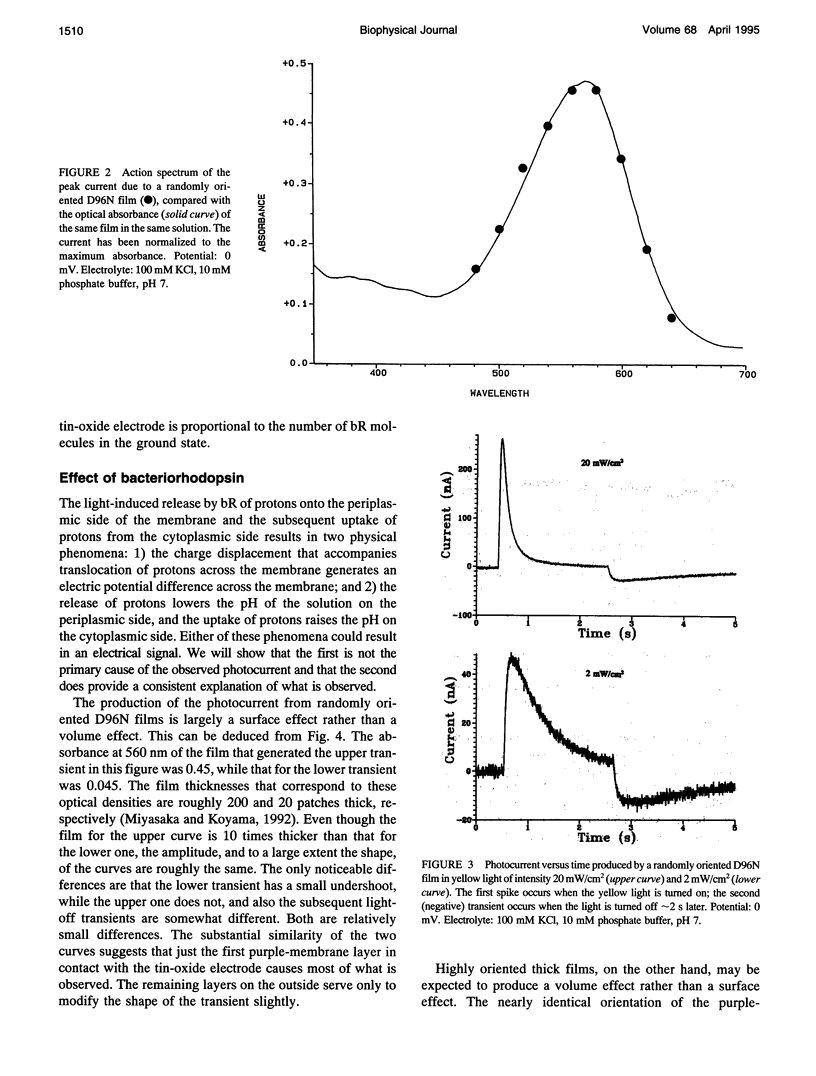
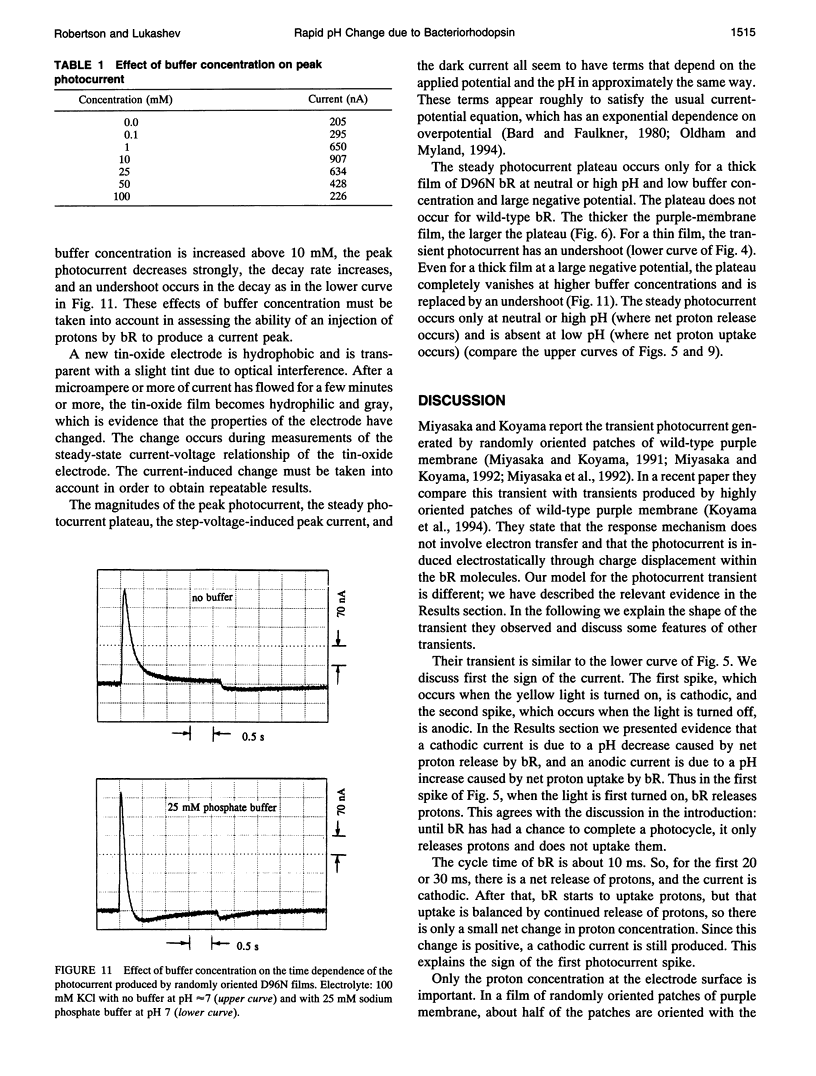
The photocurrent transient generated by bacteriorhodopsin (bR) on a tin-oxide electrode is due to pH change and not to charge displacement as previously assumed. Films of either randomly oriented or highly oriented purple membranes were deposited on transparent electrodes made of tin-oxide-coated glass. The membranes contained either wild-type or D96N-mutant bR. When excited with yellow light through the glass, the bR pumps protons across the membrane. The result is a rapid local pH change as well as a charge displacement. Experiments with these films show that it is the pH change rather than the displacement that produces the current transient. The calibration for the transient pH measurement is given. The sensitivity of a tin-oxide electrode to a transient pH change is very much larger than its sensitivity to a steady-state pH change.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balashov S. P., Govindjee R., Kono M., Imasheva E., Lukashev E., Ebrey T. G., Crouch R. K., Menick D. R., Feng Y. Effect of the arginine-82 to alanine mutation in bacteriorhodopsin on dark adaptation, proton release, and the photochemical cycle. Biochemistry. 1993 Oct 5;32(39):10331–10343. doi: 10.1021/bi00090a008. [DOI] [PubMed] [Google Scholar]
- Becher B. M., Cassim J. Y. Improved isolation procedures for the purple membrane of Halobacterium halobium. Prep Biochem. 1975;5(2):161–178. doi: 10.1080/00327487508061568. [DOI] [PubMed] [Google Scholar]
- Butt H. J., Fendler K., Bamberg E., Tittor J., Oesterhelt D. Aspartic acids 96 and 85 play a central role in the function of bacteriorhodopsin as a proton pump. EMBO J. 1989 Jun;8(6):1657–1663. doi: 10.1002/j.1460-2075.1989.tb03556.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dencher N., Wilms M. Flash photometric experiments on the photochemical cycle of bacteriorhodopsin. Biophys Struct Mech. 1975 May 30;1(3):259–271. doi: 10.1007/BF00535760. [DOI] [PubMed] [Google Scholar]
- Druckmann S., Friedman N., Lanyi J. K., Needleman R., Ottolenghi M., Sheves M. The back photoreaction of the M intermediate in the photocycle of bacteriorhodopsin: mechanism and evidence for two M species. Photochem Photobiol. 1992;56(6):1041–1047. doi: 10.1111/j.1751-1097.1992.tb09727.x. [DOI] [PubMed] [Google Scholar]
- Garty H., Klemperer G., Eisenbach M., Caplan S. R. The direction of light-induced pH changes in purple membrane suspensions. Influence of pH and temperature. FEBS Lett. 1977 Sep 15;81(2):238–242. doi: 10.1016/0014-5793(77)80526-7. [DOI] [PubMed] [Google Scholar]
- Holz M., Drachev L. A., Mogi T., Otto H., Kaulen A. D., Heyn M. P., Skulachev V. P., Khorana H. G. Replacement of aspartic acid-96 by asparagine in bacteriorhodopsin slows both the decay of the M intermediate and the associated proton movement. Proc Natl Acad Sci U S A. 1989 Apr;86(7):2167–2171. doi: 10.1073/pnas.86.7.2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karvaly B., Dancsházy Z. Bacteriorhodopsin: a molecular photoelectric regulator. Quenching of photovoltaic effect of bimolecular lipid membranes containing bacteriorhodopsin by blue light. FEBS Lett. 1977 Apr 1;76(1):36–40. doi: 10.1016/0014-5793(77)80115-4. [DOI] [PubMed] [Google Scholar]
- Kono M., Misra S., Ebrey T. G. pH dependence of light-induced proton release by bacteriorhodopsin. FEBS Lett. 1993 Sep 27;331(1-2):31–34. doi: 10.1016/0014-5793(93)80291-2. [DOI] [PubMed] [Google Scholar]
- Koyama K., Yamaguchi N., Miyasaka T. Antibody-mediated bacteriorhodopsin orientation for molecular device architectures. Science. 1994 Aug 5;265(5173):762–765. doi: 10.1126/science.265.5173.762. [DOI] [PubMed] [Google Scholar]
- Lanyi J. K. Proton translocation mechanism and energetics in the light-driven pump bacteriorhodopsin. Biochim Biophys Acta. 1993 Dec 7;1183(2):241–261. doi: 10.1016/0005-2728(93)90226-6. [DOI] [PubMed] [Google Scholar]
- Mathies R. A., Lin S. W., Ames J. B., Pollard W. T. From femtoseconds to biology: mechanism of bacteriorhodopsin's light-driven proton pump. Annu Rev Biophys Biophys Chem. 1991;20:491–518. doi: 10.1146/annurev.bb.20.060191.002423. [DOI] [PubMed] [Google Scholar]
- Miyasaka T., Koyama K., Itoh I. Quantum conversion and image detection by a bacteriorhodopsin-based artificial photoreceptor. Science. 1992 Jan 17;255(5042):342–344. doi: 10.1126/science.255.5042.342. [DOI] [PubMed] [Google Scholar]
- Oesterhelt D., Stoeckenius W. Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nat New Biol. 1971 Sep 29;233(39):149–152. doi: 10.1038/newbio233149a0. [DOI] [PubMed] [Google Scholar]
- Otto H., Marti T., Holz M., Mogi T., Lindau M., Khorana H. G., Heyn M. P. Aspartic acid-96 is the internal proton donor in the reprotonation of the Schiff base of bacteriorhodopsin. Proc Natl Acad Sci U S A. 1989 Dec;86(23):9228–9232. doi: 10.1073/pnas.86.23.9228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soppa J., Oesterhelt D. Bacteriorhodopsin mutants of Halobacterium sp. GRB. I. The 5-bromo-2'-deoxyuridine selection as a method to isolate point mutants in halobacteria. J Biol Chem. 1989 Aug 5;264(22):13043–13048. [PubMed] [Google Scholar]
- Tittor J., Soell C., Oesterhelt D., Butt H. J., Bamberg E. A defective proton pump, point-mutated bacteriorhodopsin Asp96----Asn is fully reactivated by azide. EMBO J. 1989 Nov;8(11):3477–3482. doi: 10.1002/j.1460-2075.1989.tb08512.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Váró G., Lanyi J. K. Protonation and deprotonation of the M, N, and O intermediates during the bacteriorhodopsin photocycle. Biochemistry. 1990 Jul 24;29(29):6858–6865. doi: 10.1021/bi00481a015. [DOI] [PubMed] [Google Scholar]
- Zimányi L., Váró G., Chang M., Ni B., Needleman R., Lanyi J. K. Pathways of proton release in the bacteriorhodopsin photocycle. Biochemistry. 1992 Sep 15;31(36):8535–8543. doi: 10.1021/bi00151a022. [DOI] [PubMed] [Google Scholar]



