Abstract
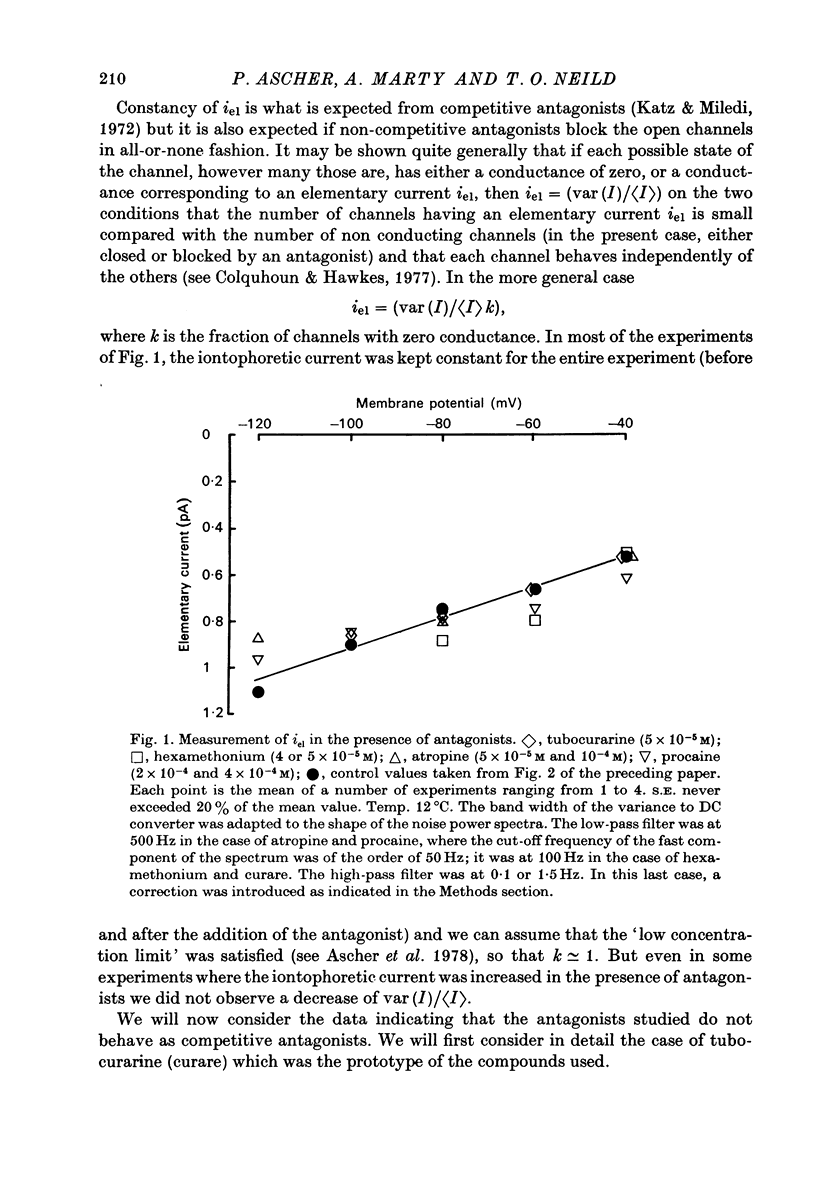
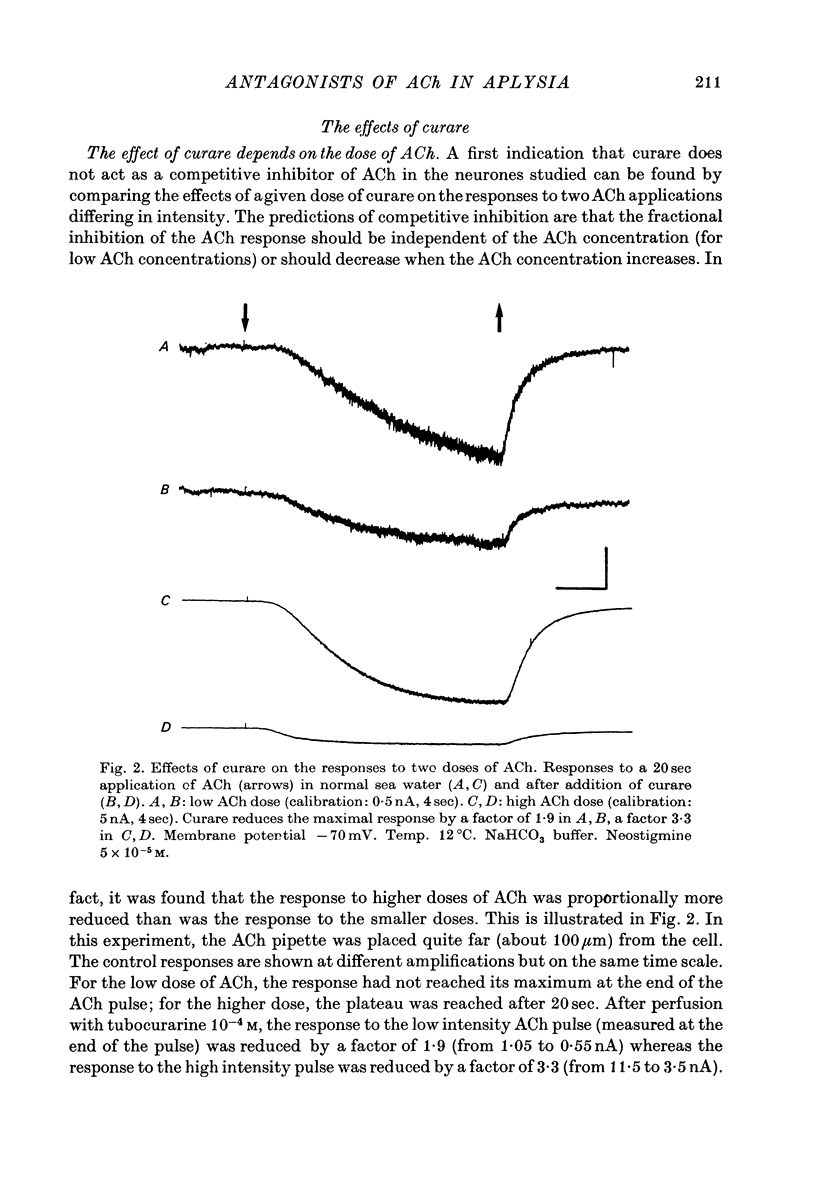
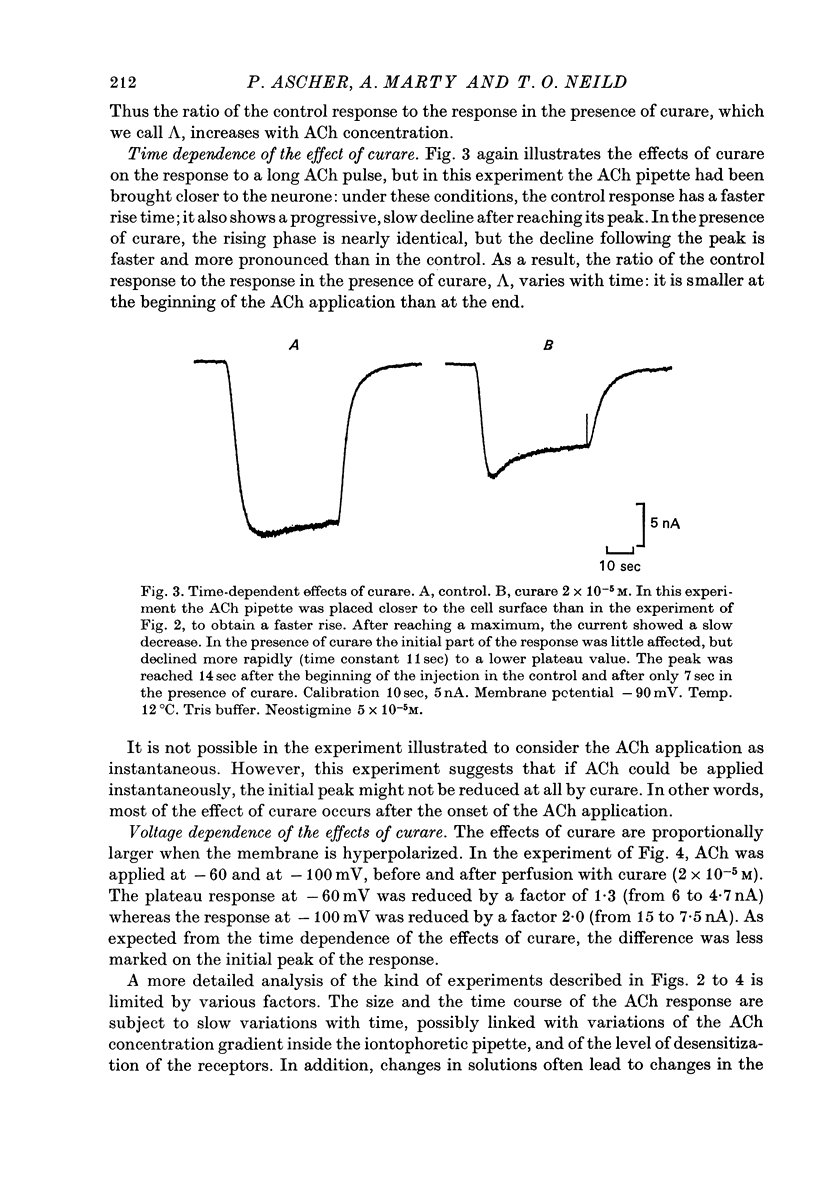
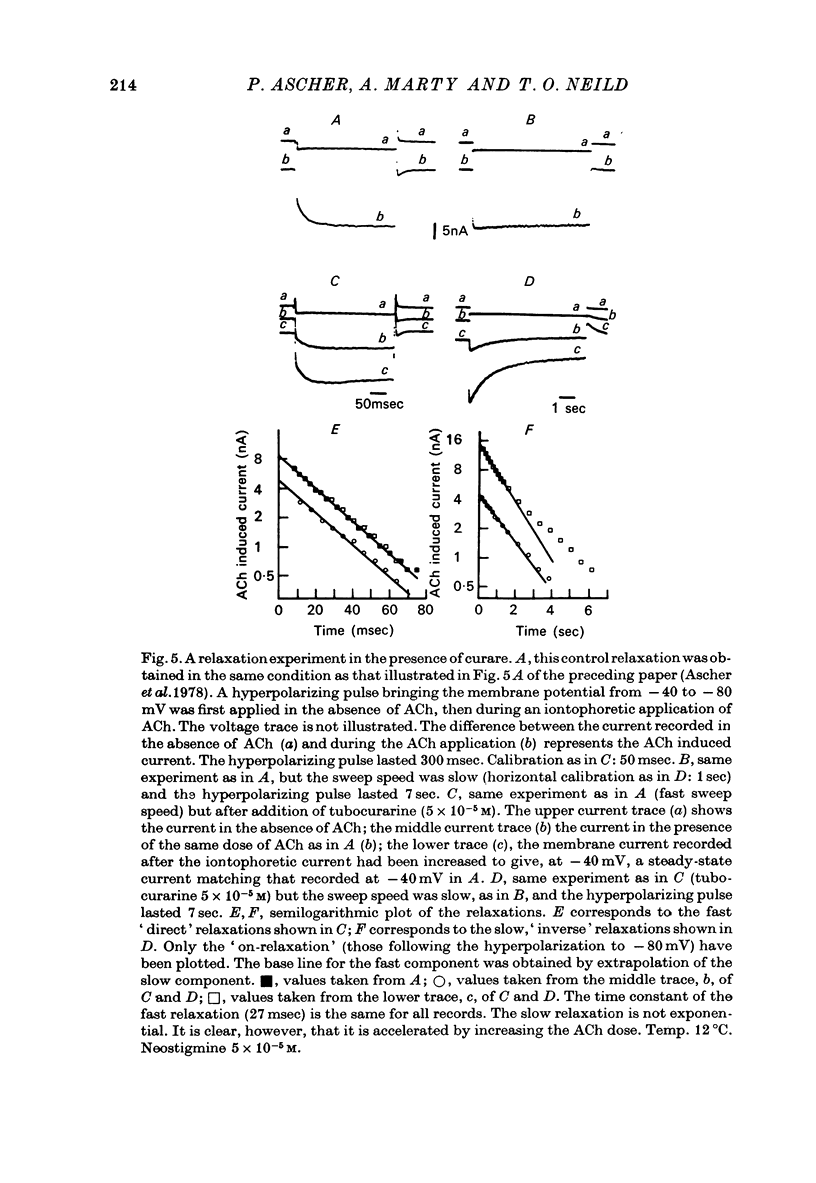
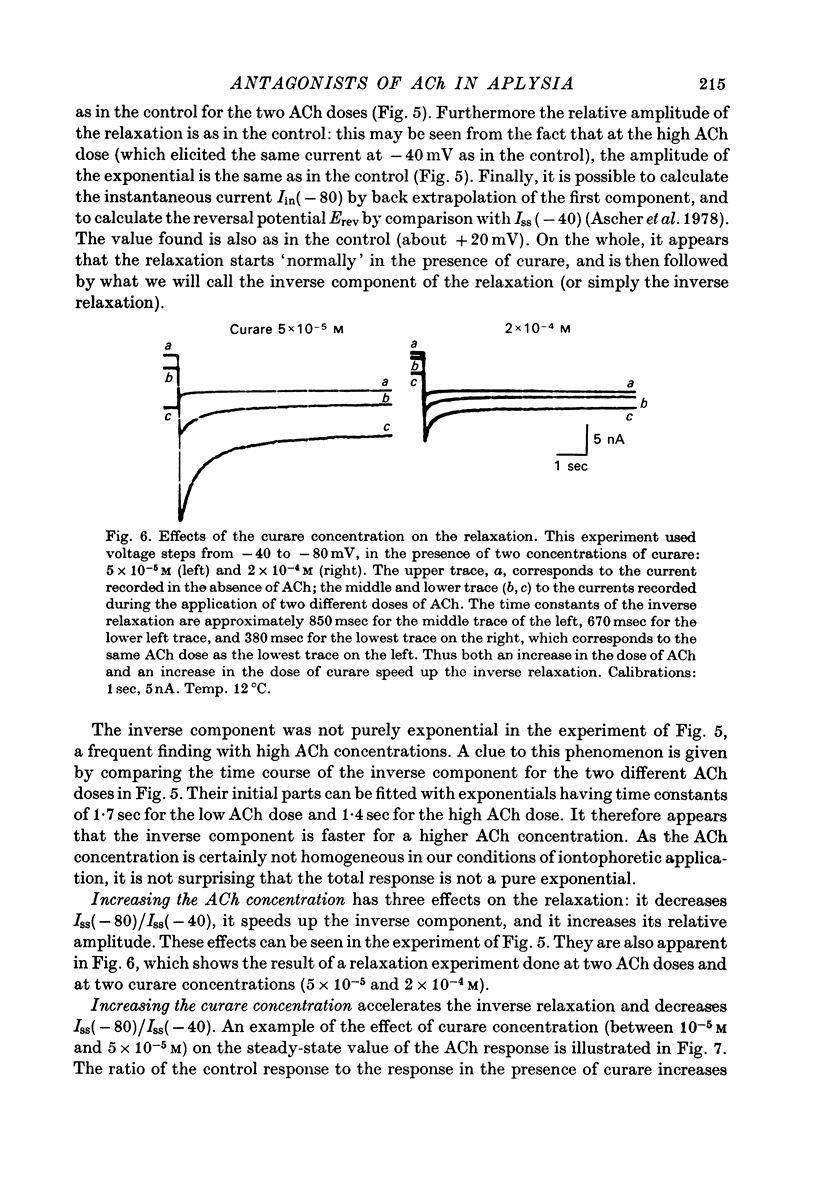
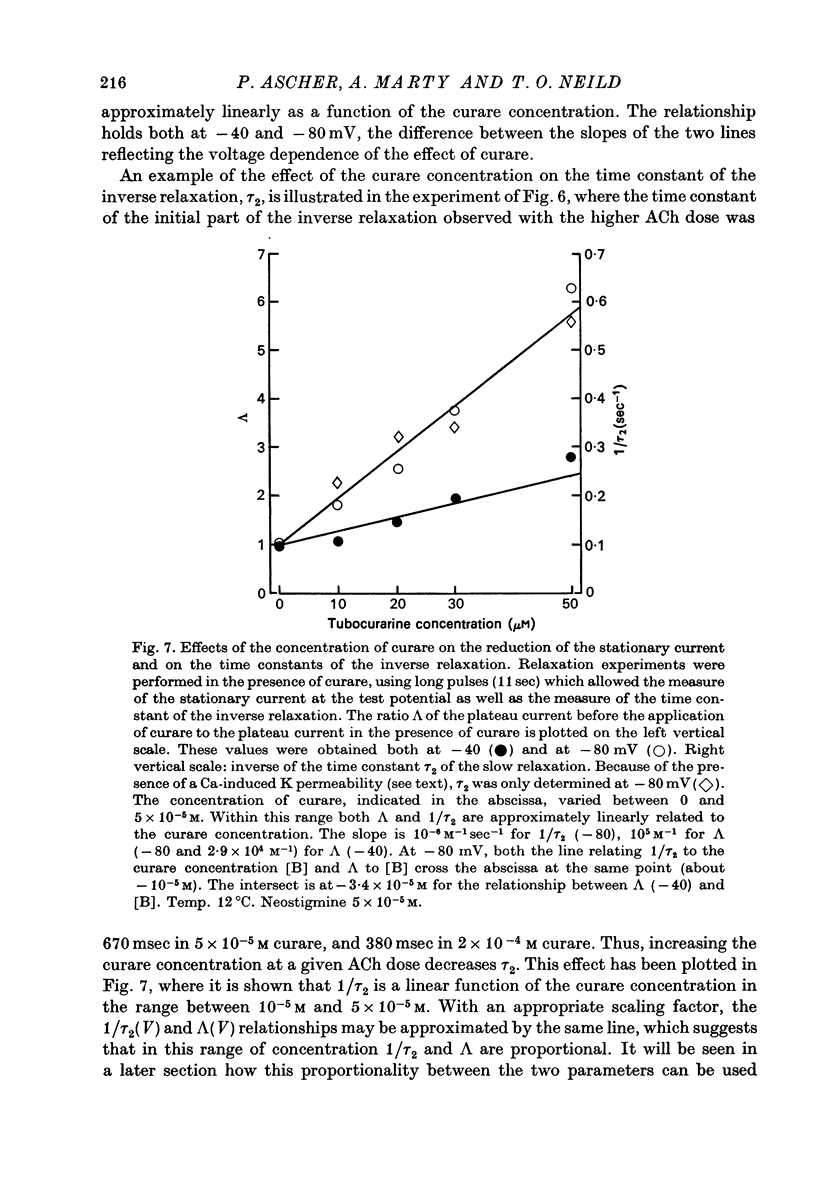
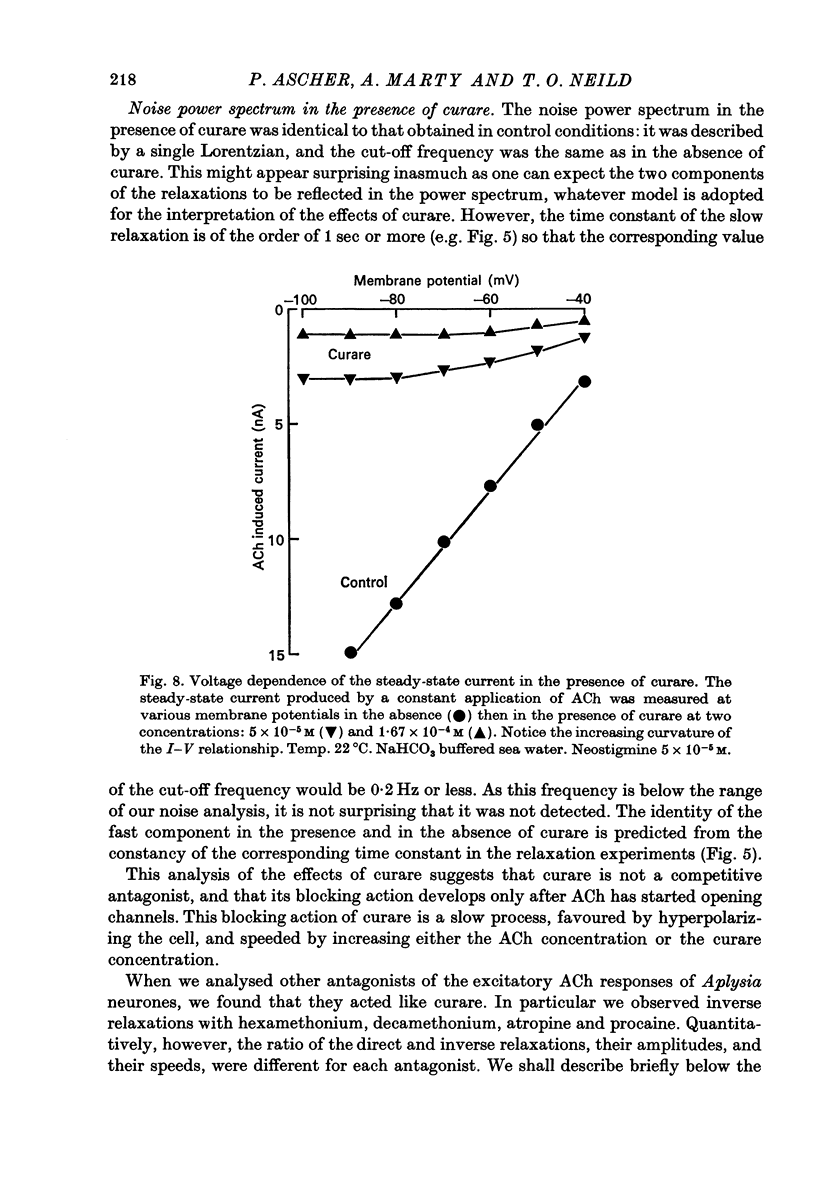
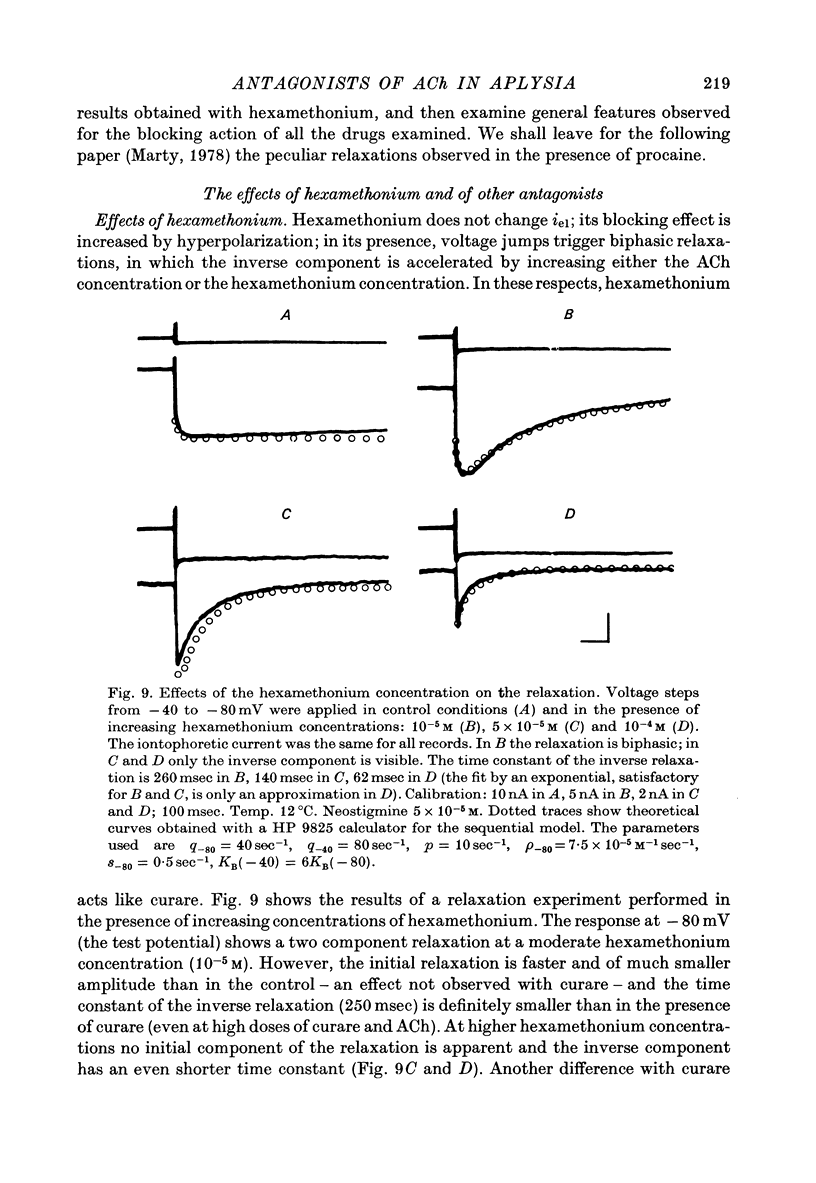
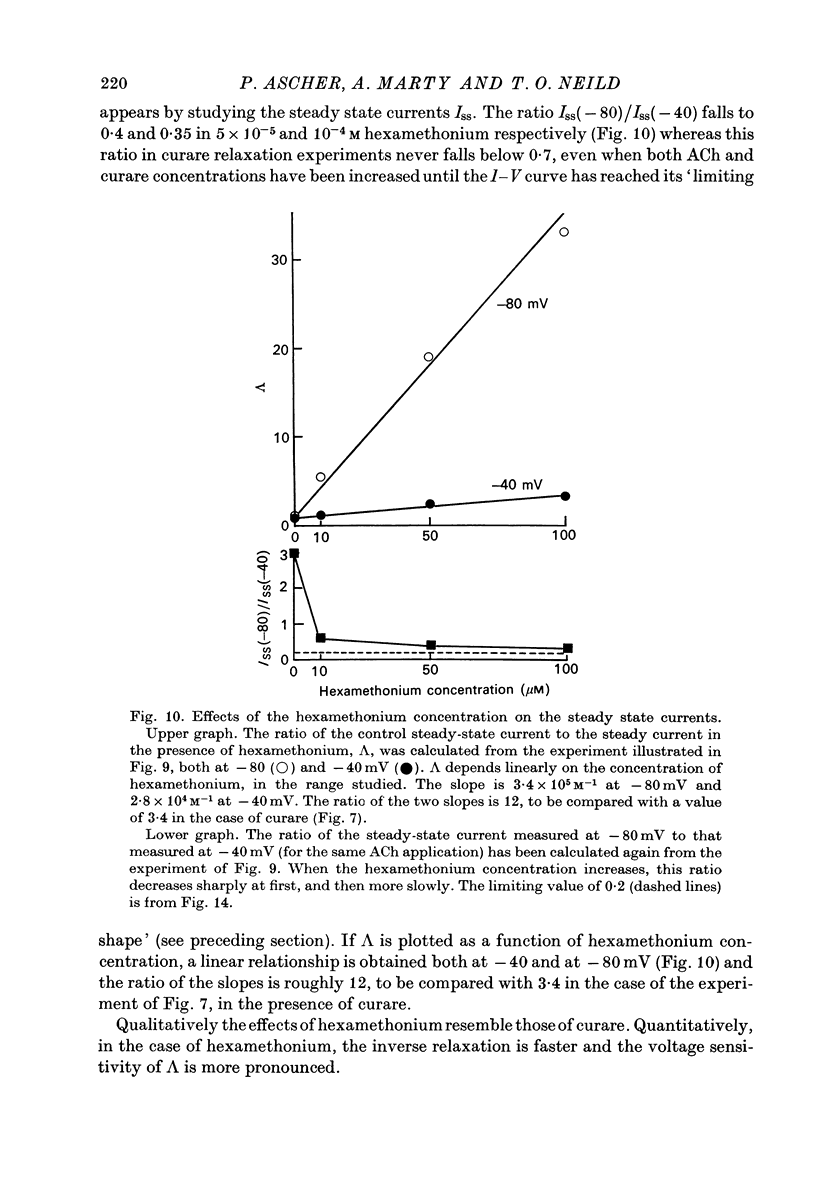
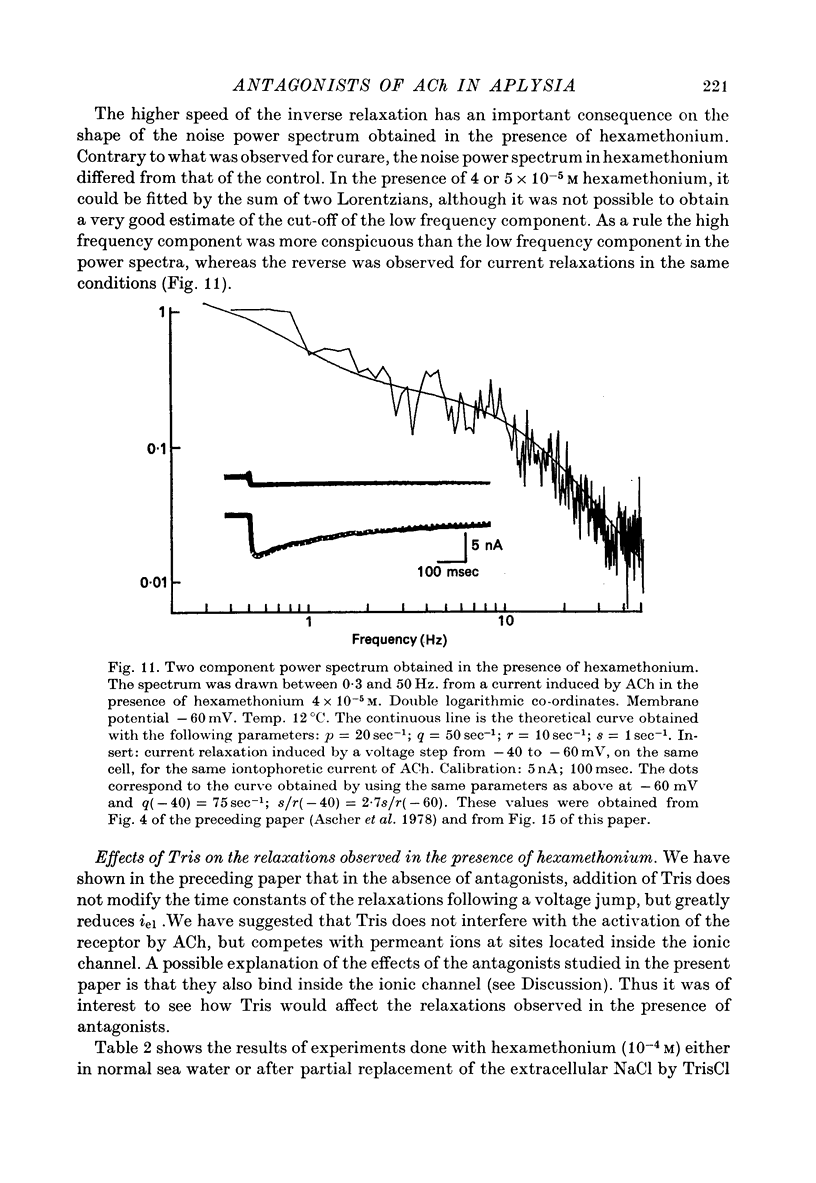
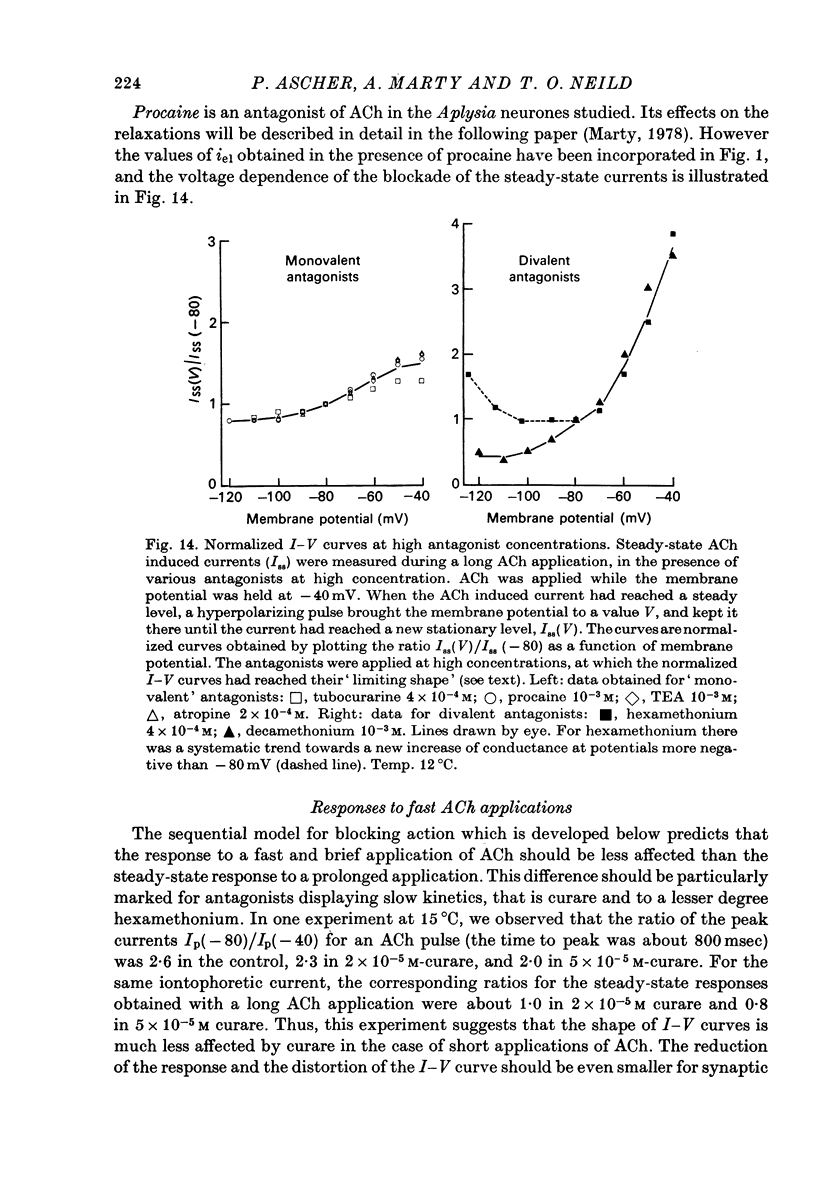
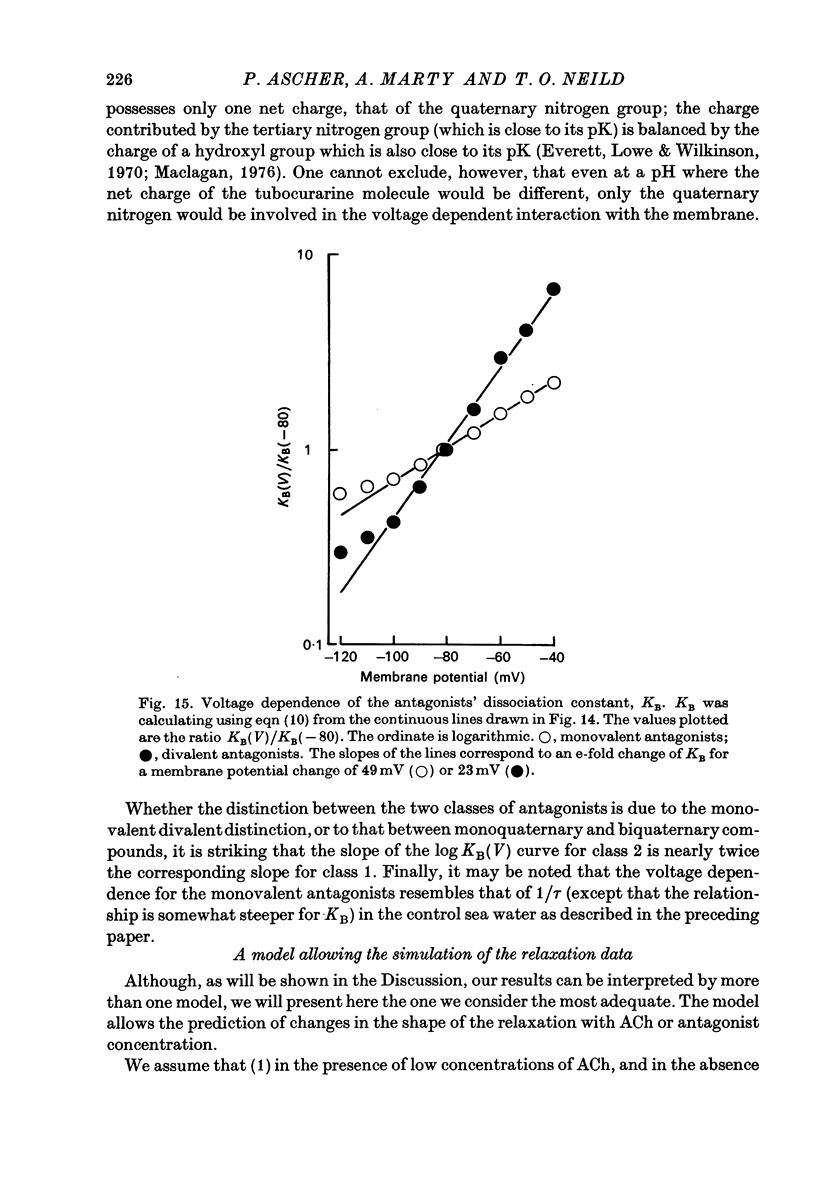
1. The mode of action of various antagonists of acetylcholine (ACh) excitatory effects on Aplysia neurones was studied under voltage clamp. ACh was applied by iontophoresis whereas antagonists were applied in the bath. Tubocurarine and hexamethonium were the most thoroughly studied compounds. 2. The 'elementary current', calculated as the ratio of the variance of the ACh noise to the mean ACh induced current, was not modified by any of the antagonists tested. 3. The evolution of the ACh induced current after a voltage jump, which is normally described by a single exponential, was modified by all the antagonists tested. A common feature of the modified relaxations was the appearance, over a certain concentration range of the antagonist, of two successive and opposite exponential components. 4. The characteristics of the composite relaxations depend on the antagonist. For a given antagonist they vary with membrane potential, ACh concentration, and antagonist concentration. 5. The noise power spectra of the ACh induced current showed changes consistent with those of the relaxations. 6. In the absence of antagonists, the current induced by a steady application of ACh increases linearly with hyperpolarization. In the presence of antagonists, the I-V curve shows a marked curvature, indicating a proportionally larger reduction of the ACh response at more negative membrane potentials. 7. The voltage sensitivity of the blocking action of hexamethonium and decamethonium is noticeably stronger than that of monovalent antagonists. 8. A model is proposed which accounts for the observed effects. It assumes that the antagonists studied bind perferentially to the 'activated' ACh-receptor complex, and convert it to a non-conducting state. Kinetic constants can be calculated for this reaction; e.g. for curare, at 12 degrees C and -80 mV, the dissociation and association constants were estimated at 0.1 sec-1 and 4 X 10(5) M-1 sec-1. 9. Partial replacement of the extracellular Na by Tris modifies the relaxations observed in the presence of hexamethonium. Hexamethonium appears less effective in the presence of Tris, which supports the hypothesis that the binding site of the antagonists is linked to the ionic channel.
Full text
PDF















Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams P. R. A model for the procaine end-plate current. J Physiol. 1975 Mar;246(2):61P–63P. [PubMed] [Google Scholar]
- Adams P. R. Drug blockade of open end-plate channels. J Physiol. 1976 Sep;260(3):531–552. doi: 10.1113/jphysiol.1976.sp011530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adams P. R. Voltage jump analysis of procaine action at frog end-plate. J Physiol. 1977 Jun;268(2):291–318. doi: 10.1113/jphysiol.1977.sp011858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M. Inactivation of the potassium conductance and related phenomena caused by quaternary ammonium ion injection in squid axons. J Gen Physiol. 1969 Nov;54(5):553–575. doi: 10.1085/jgp.54.5.553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M. Interaction of tetraethylammonium ion derivatives with the potassium channels of giant axons. J Gen Physiol. 1971 Oct;58(4):413–437. doi: 10.1085/jgp.58.4.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong C. M. Potassium pores of nerve and muscle membranes. Membranes. 1975;3:325–358. [PubMed] [Google Scholar]
- Armstrong C. M. Time course of TEA(+)-induced anomalous rectification in squid giant axons. J Gen Physiol. 1966 Nov;50(2):491–503. doi: 10.1085/jgp.50.2.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ascher P., Marty A., Neild T. O. Life time and elementary conductance of the channels mediating the excitatory effects of acetylcholine in Aplysia neurones. J Physiol. 1978 May;278:177–206. doi: 10.1113/jphysiol.1978.sp012299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barker J. L., Gainer H. Pentobarbital: selective depression of excitatory postsynaptic potentials. Science. 1973 Nov 16;182(4113):720–722. doi: 10.1126/science.182.4113.720. [DOI] [PubMed] [Google Scholar]
- Blackman J. G., Purves R. D. Ganglionic transmission in the autonomic nervous system. N Z Med J. 1968 Mar;67(429):376–384. [PubMed] [Google Scholar]
- Carpenter D. O., Swann J. W., Yarowsky P. J. Effect of curare on responses to different putative neurotransmitters in Aplysia neurons. J Neurobiol. 1977 Mar;8(2):119–132. doi: 10.1002/neu.480080204. [DOI] [PubMed] [Google Scholar]
- Cartaud J., Benedetti E. L., Cohen J. B., Meunier J. C., Changeux J. P. Presence of a lattice structure in membrane fragments rich in nicotinic receptor protein from the electric organ of Torpedo marmorata. FEBS Lett. 1973 Jun 15;33(1):109–113. doi: 10.1016/0014-5793(73)80171-1. [DOI] [PubMed] [Google Scholar]
- Colquhoun D., Hawkes A. G. Relaxation and fluctuations of membrane currents that flow through drug-operated channels. Proc R Soc Lond B Biol Sci. 1977 Nov 14;199(1135):231–262. doi: 10.1098/rspb.1977.0137. [DOI] [PubMed] [Google Scholar]
- Feltz A., Large W. A., Trautmann A. Analysis of atropine action at the frog neutromuscular junction. J Physiol. 1977 Jul;269(1):109–130. doi: 10.1113/jphysiol.1977.sp011895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gage P. W. Generation of end-plate potentials. Physiol Rev. 1976 Jan;56(1):177–247. doi: 10.1152/physrev.1976.56.1.177. [DOI] [PubMed] [Google Scholar]
- Katz B., Miledi R. The effect of procaine on the action of acetylcholine at the neuromuscular junction. J Physiol. 1975 Jul;249(2):269–284. doi: 10.1113/jphysiol.1975.sp011015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz B., Miledi R. The statistical nature of the acetycholine potential and its molecular components. J Physiol. 1972 Aug;224(3):665–699. doi: 10.1113/jphysiol.1972.sp009918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kehoe J. Three acetylcholine receptors in Aplysia neurones. J Physiol. 1972 Aug;225(1):115–146. doi: 10.1113/jphysiol.1972.sp009931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manalis R. S. Voltage-dependent effect of curare at the frog neuromuscular junction. Nature. 1977 May 26;267(5609):366–368. doi: 10.1038/267366a0. [DOI] [PubMed] [Google Scholar]
- Marty A., Neild T., Ascher P. Voltage sensitivity of acetylcholine currents in Aplysia neurones in the presence of curare. Nature. 1976 Jun 10;261(5560):501–503. doi: 10.1038/261501a0. [DOI] [PubMed] [Google Scholar]
- Marty A. Noise and relaxation studies of acetylcholine induced currents in the presence of procaine. J Physiol. 1978 May;278:237–250. doi: 10.1113/jphysiol.1978.sp012301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsegian A. Energy of an ion crossing a low dielectric membrane: solutions to four relevant electrostatic problems. Nature. 1969 Mar 1;221(5183):844–846. doi: 10.1038/221844a0. [DOI] [PubMed] [Google Scholar]
- Rang H. P., Ritter J. M. The relationship between desensitization and the metaphilic effect at cholinergic receptors. Mol Pharmacol. 1970 Jul;6(4):383–390. [PubMed] [Google Scholar]
- Ruff R. L. A quantitative analysis of local anaesthetic alteration of miniature end-plate currents and end-plate current fluctuations. J Physiol. 1977 Jan;264(1):89–124. doi: 10.1113/jphysiol.1977.sp011659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinbach A. B. A kinetic model for the action of xylocaine on receptors for acetylcholine. J Gen Physiol. 1968 Jul;52(1):162–180. doi: 10.1085/jgp.52.1.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strichartz G. R. The inhibition of sodium currents in myelinated nerve by quaternary derivatives of lidocaine. J Gen Physiol. 1973 Jul;62(1):37–57. doi: 10.1085/jgp.62.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TAUC L., GERSCHENFELD H. M. A cholinergic mechanism of inhibitory synaptic transmission in a molluscan nervous system. J Neurophysiol. 1962 Mar;25:236–262. doi: 10.1152/jn.1962.25.2.236. [DOI] [PubMed] [Google Scholar]
- Woodhull A. M. Ionic blockage of sodium channels in nerve. J Gen Physiol. 1973 Jun;61(6):687–708. doi: 10.1085/jgp.61.6.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeh J. Z., Narahashi T. Kinetic analysis of pancuronium interaction with sodium channels in squid axon membranes. J Gen Physiol. 1977 Mar;69(3):293–323. doi: 10.1085/jgp.69.3.293. [DOI] [PMC free article] [PubMed] [Google Scholar]


