Abstract
In gram-negative bacterial pathogens, such as Pseudomonas aeruginosa and Yersinia pseudotuberculosis, cell-to-cell communication via the N-acylhomoserine lactone (AHL) signal molecules is involved in the cell population density-dependent control of genes associated with virulence. This phenomenon, termed quorum sensing, relies upon the accumulation of AHLs to a threshold concentration at which target structural genes are activated. By using biosensors capable of detecting a range of AHLs we observed that, in cultures of Y. pseudotuberculosis and P. aeruginosa, AHLs accumulate during the exponential phase but largely disappear during the stationary phase. When added to late-stationary-phase, cell-free culture supernatants of the respective pathogen, the major P. aeruginosa [N-butanoylhomoserine lactone (C4-HSL) and N-(3-oxododecanoyl)homoserine lactone (3-oxo-C12-HSL)] and Y. pseudotuberculosis [N-(3-oxohexanoyl)homoserine lactone (3-oxo-C6-HSL) and N-hexanoylhomoserine lactone (C6-HSL)] AHLs were inactivated. Short-acyl-chain compounds (e.g., C4-HSL) were turned over more extensively than long-chain molecules (e.g., 3-oxo-C12-HSL). Little AHL inactivation occurred with cell extracts, and no evidence for inactivation by specific enzymes was apparent. This AHL turnover was discovered to be due to pH-dependent lactonolysis. By acidifying the growth media to pH 2.0, lactonolysis could be reversed. By using carbon-13 nuclear magnetic resonance spectroscopy, we found that the ring opening of homoserine lactone (HSL), N-propionyl HSL (C3-HSL), and C4-HSL increased as pH increased but diminished as the N-acyl chain was lengthened. At low pH levels, the lactone rings closed but not via a simple reversal of the ring opening reaction mechanism. Ring opening of C4-HSL, C6-HSL, 3-oxo-C6-HSL, and N-octanoylhomoserine lactone (C8-HSL), as determined by the reduction of pH in aqueous solutions with time, was also less rapid for AHLs with more electron-donating longer side chains. Raising the temperature from 22 to 37°C increased the rate of ring opening. Taken together, these data show that (i) to be functional under physiological conditions in mammalian tissue fluids, AHLs require an N-acyl side chain of at least four carbons in length and (ii) that the longer the acyl side chain the more stable the AHL signal molecule.
Many gram-negative bacteria regulate gene expression in a cell-density-dependent manner by using N-acyl homoserine lactone (AHL) quorum-sensing signal molecules. AHLs diffuse into and out of bacterial cells and, as the population of bacteria increases, so does the AHL concentration. Once the AHLs reach a threshold concentration, they act as coinducers, usually by activating LuxR-type transcriptional regulators to induce target gene expression. AHL-dependent quorum sensing is known to regulate many different physiological processes, including the production of secondary metabolites, swimming, swarming, biofilm maturation, and virulence in human, plant, and animal pathogens (for reviews, see references 43 and 47). In addition, certain AHLs have been shown to possess pharmacological (22, 13) and immunomodulatory activity (44, 40) such that they may function as virulence determinants per se.
AHLs consist of five-membered homoserine lactone (HSL) rings containing varied amide-linked side chains. The N-acyl moieties of the naturally occurring AHLs identified to date range from 4 to 14 carbons in length and may be saturated or unsaturated, with or without a substituent (usually hydroxy or oxo) on the carbon at the C3 position of the N-linked acyl chain. Three types of AHL synthases have been identified, corresponding to LuxI, LuxM, and HtdS types (reviewed in reference 43). Often, the enzyme that is involved in AHL production can be induced by the AHL it produces, thereby creating a positive-feedback loop that induces yet higher levels of AHLs. The biochemistry of AHL formation via LuxI-type proteins, including TraI (Agrobacterium tumefaciens), LuxI (Vibrio fischeri), and RhlI (Pseudomonas aeruginosa) has been thoroughly investigated in vitro (30, 39, 16). In each case, the data obtained indicate that the appropriately charged acyl carrier protein (acyl-ACP) is the major acyl chain donor, whereas S-adenosyl methionine provides the HSL moiety.
AHL-dependent quorum sensing is employed by the human pathogens P. aeruginosa and Yersinia pseudotuberculosis. P. aeruginosa contains the LuxRI homologues LasRI (31) and RhlRI (19). LasI directs the synthesis of N-(3-oxododecanoyl)homoserine lactone (3-oxo-C12-HSL) (32), whereas RhlI directs the synthesis of N-butanoyl-homoserine lactone (C4-HSL) (48). Each system modulates a regulon comprising an overlapping set of genes. However, the las and the rhl systems are not independent of each other but form a regulatory hierarchy where LasR/3-oxo-C12-HSL activates the transcription of rhlR (20, 36). Recently, a third LuxR homologue termed QscR has been identified, which has been shown to regulate the transcription of both lasI and rhlI (9). Genes regulated by either LasR/3-oxo-C12-HSL or RhlR/C4-HSL include those coding for elastase (lasB), LasA protease (lasA), alkaline protease (apr), exotoxin A (toxA), the cytotoxic PA-IL lectin (lecA), chitinase, hydrogen cyanide, and pyocyanin (3, 17, 19, 37, 48, 50). In addition to controlling the expression of many genes for exoproteins and secondary metabolites, the quorum-sensing machinery in P. aeruginosa influences the xcp secretion pathway (6) and biofilm maturation (11). Indeed, Whiteley et al. (46) have estimated that between 1 and 4% of all genes in P. aeruginosa may be controlled, to some extent, by quorum sensing.
Like P. aeruginosa, Y. pseudotuberculosis possesses two LuxRI pairs, YpsRI and YtbRI, which control a temperature-dependent hierarchical quorum-sensing system involved in the regulation of motility and clumping (1). YpsI directs the synthesis of N-(3-oxohexanoyl)HSL (3-oxo-C6-HSL) and N-hexanoyl-HSL (C6-HSL), whereas YtbI is responsible for the production of C6-HSL and N-octanoyl-HSL (C8-HSL). Other pathogenic yersiniae, including Yersinia enterocolitica and Yersinia pestis, also produce 3-oxo-C6-HSL and C6-HSL (45, 42).
One fundamental aspect of AHL-dependent quorum sensing is that the signal molecule is able to move from inside the cell to outside the cell and back. Using tritiated 3-oxo-C6-HSL, Kaplan and Greenberg (18) showed that in V. fischeri the AHL moved across both cell membranes by free diffusion. This also appears to be the case for C4-HSL in P. aeruginosa, which moves into and out of cells by free diffusion and equilibrates rapidly between intra- and extracellular compartments (33). In contrast, 3-oxo-C12-HSL is concentrated threefold within the cell, possibly because of partitioning into bacterial membranes. Furthermore, although the influx of 3-oxo-C12-HSL into P. aeruginosa appears to be via diffusion, efflux from the bacterial cells is dependent upon an active MexAB-OprM pump (33).
Despite the extensive investigation of AHL production and action in various gram-negative bacteria, relatively little attention has been paid to the turnover and fate of AHL molecules as a function of growth either within the producer organism or in its local environment. A soil-borne bacterium, Variovorax paradoxus, capable of degrading AHLs for use as a sole carbon source has been identified (23). Furthermore, Dong et al. (12) obtained an enzyme from Bacillus sp. strain 240B1, termed AiiA that inactivates AHLs by hydrolysis of the lactone ring. When expressed in the plant pathogen Erwinia carotovora, AiiA attenuated virulence by inactivating the AHL-dependent expression of plant wall-degrading exoenzymes.
We sought here to investigate the fate of AHLs as a function of growth in cultures of P. aeruginosa and Y. pseudotuberculosis and to determine how this correlates with chemical studies on the stability of the open- and closed-ring forms of AHLs as a function of pH, temperature, and acyl chain length. The implications of our findings with respect to quorum sensing by pathogens in vivo in human body fluids is discussed.
MATERIALS AND METHODS
Bacterial strains and culture conditions.
Y. pseudotuberculosis (YPIII (pIB1)NalR and P. aeruginosa PAO1 were routinely grown at 37°C on Luria-Bertani (LB) agar plates or with shaking at 200 rpm in LB broth (1 liter) at 22, 28, or 37°C for 24 to 30 h depending on the organism. At regular intervals, samples were removed for the measurement of growth (at an optical density at 600 nm [OD600]) and pH and AHL analysis. For some experiments, Y. pseudotuberculosis was grown as described above in LB medium but supplemented with 50 mM morpholinepropanesulfonic acid (MOPS) buffer (working range pH of 6.5 to 7.9).
Detection of AHLs produced by Y. pseudotuberculosis.
Cell-free culture supernatants (20 ml) were extracted twice with dichloromethane (20 ml). After removal of the solvent under reduced pressure, the residues were suspended in acetonitrile (20 μl). The AHLs were separated by thin-layer chromatography (TLC) on reversed-phase RP-18 F254s plates (BDH/Poole, Dorset, United Kingdom) by using methanol-water (60:40 [vol/vol]) essentially as described previously with Chromobacterium violaceum CV026 as the AHL biosensor (1, 28). After overnight incubation of the TLC plates at 30°C, AHLs were located as purple spots on a white background. For some experiments, culture supernatants prepared from Y. pseudotuberculosis grown in LB medium for 24 h at 37°C were filter sterilized, and the pH was adjusted with concentrated HCl to 6.8, 4.0, 3.0, or 2.0. After 24 h of incubation at 37°C, each sample was extracted with dichloromethane as described above and subjected to TLC analysis. In addition to some stationary-phase culture supernatants, either C6-HSL or 3-oxo-C6-HSL was added. After incubation for 24 h, each sample was extracted and subjected to TLC with either CV026 or Escherichia coli(pSB401) (45, 49) for AHL detection.
Detection of AHLs produced by P. aeruginosa.
AHLs in cell-free culture supernatants were detected by using the E. coli biosensor strains E. coli(pSB536) for C4-HSL and E. coli(pSB1705) for 3-oxo-C12-HSL, respectively (49). For assay of AHLs in liquid culture, exponentially growing cells of the appropriate E. coli biosensor were mixed with filter-sterilized supernatants from P. aeruginosa cultures and inoculated into a combined luminometer-spectrophotometer (LUCY1; Anthos, Salzburg, Austria) at 30°C for 24 h as described by Winzer et al. (50). Luminescence and OD495 values were recorded every 30 min, and the bioluminescence was calculated as the relative light units per cell (RLU/OD495) to account for the growth of the sensor strains. Cell-free culture supernatant samples were also examined after separation of AHLs by TLC on RP-18 F254s plates by using methanol-water (60:40 [vol/vol]) as described above for C4-HSL. For 3-oxo-C12-HSL, RP-2 F254s reversed-phase TLC plates (Macherey & Nagel, Düren, Germany) were used with a methanol-water (45:55 [vol/vol]) solvent system. After chromatography, TLC plates were dried and then overlaid with LB medium containing 0.75% (wt/vol) agar seeded with the appropriate AHL biosensor. After 14 to 16 h at 30°C, AHLs were visualized as bright spots in a dark background when viewed with a Luminograph LB980 (Berthold, Pforzheim, Germany) photon video camera. For some experiments, C4-HSL or 3-oxo-C12-HSL (at 100 μM) were added to stationary-phase P. aeruginosa cell-free supernatants or to supernatants adjusted with concentrated HCl to pH 7.0. After incubation for 24 h, each sample was subjected to TLC with either E. coli(pSB536) or E. coli(pSB1075) for AHL detection (49).
AHL synthesis.
C4-HSL, C5-HSL, C6-HSL, 3-oxo-C6-HSL, C7-HSL, C8-HSL, 3-oxo-C10-HSL, and 3-oxo-C12-HSL were synthesized essentially as previously described by Chhabra et al. (8). Each synthetic AHL was purified to homogeneity by preparative high-pressure liquid chromatography (HPLC), and each structure was confirmed by mass spectrometry (MS) and proton nuclear magnetic resonance (NMR) spectroscopy. The HSL was prepared by treating aqueous l-homoserine (100 mg; Aldrich, Milwaukee, Wis.) with HCl (5 M, 2.5 ml), followed by freeze drying, extraction with CHCl3, and evaporation of the solvent under reduced pressure. C3-HSL was prepared by treating a solution of HSL (100 mg) in saturated aqueous NaHCO3 (4 ml) at 0°C with propionic anhydride (0.20 ml; four times over a 2-h period). After it was stirred overnight at 20°C, the solution was dried under reduced pressure, coevaporated with ethanol (three 5-ml applications) until dry, acidified (pH <1) with HCl (5 M), and freeze-dried. The residues were extracted with CHCl3 (three times, 2 ml each time) and the solvent removed under reduced pressure.
Preparation of cell extracts.
P. aeruginosa cells were harvested after growth to stationary phase, washed with phosphate buffer (50 mM, pH 7.1), and disrupted by two passages through a French press at 138 MPa. Cell debris was removed by centrifugation, and the extract was used immediately or stored at −20°C.
HPLC and LC-MS.
To investigate the alkali-driven lactonolysis of 3-oxo-C12-HSL, the HPLC and MS (LC-MS) methodology for AHLs described by Laue et al. (21) was used. NaOH-treated 3-oxo-C12-HSL samples were subjected to LC-MS (Micromass Instruments, Manchester, United Kingdom). This technique couples the resolving power of C8 reversed-phase HPLC directly with MS such that the mass of the molecular ion (M+H) and its major component fragments can be determined for a compound with a given retention time. Samples eluting from the HPLC column were ionised by positive-ion electrospray MS (ES-MS), and the spectra obtained were compared to those of the synthetic AHL standard subjected to the same LC-MS conditions.
Determination of ring opening of HSL, C3-HSL, and C4-HSL by 13C NMR.
HSL (20 mg) was dissolved in D2O at pH <1 (adjusted with HCl). For ring opening, the pH was gradually increased by adding aqueous NaOH, and spectra were recorded at the chosen pH values until the ring opening was complete. For ring closure, the pH was lowered by adding HCl (5 M), and spectra were recorded together with pH until the ring was fully closed. Spectra were measured at 75 MHz immediately after sample preparation by using a Bruker AC300 NMR spectrometer equipped with a 5-mm probe operating at 24°C. Each spectrum was completed in less than 1 h. Estimates of the ring opening were obtained from the relative magnitudes of the signals (averaged over the ring, or open ring, carbon signals, excluding the carbonyl) derived from open and closed forms. The chemical shifts in D2O (in ppm [relative to external TSP]) were as follows: (i) for HSL, δ177.0 C-1, 60.5 C-4, 51.1 C-2, and 29.4 C-3; (ii) for l-homoserine, δ175.0 C-1, 60.5 C-4, 51.1 C-2, and 29.4 C-3; (iii) for C3-HSL (ring atoms only), δ181.0 C-1, 70.0 C-4, 51.7 C-2, and 31.4 C-3; (iv) for N-propionyl-l-homoserine (open ring atoms only), δ181 C-1, 61.2 C-4, 55.1 C-2, and 36.7 C-3; (v) for C4-HSL (ring atoms only), δ176 C-1, 66.1 C-4, 48.9 C-2, 29.8 C-3; and (vi) for N-butanoyl-l-homoserine (open ring atoms only), δ179 C-1, 61.1 C-4, 54.9 C-2, and 36.5 C-3. The error in the measurement of ring opening is estimated to be <10%. At the end of the experiments, the pH values of the solutions had changed slightly due to progressive hydrolysis. However, the time needed to record a spectrum was short (<1 h), and the change in pH was small (<0.5 U).
Detection of ring opening of C4-HSL, C6-HSL, 3-oxo-C6-HSL, and C8-HSL via the change of pH occurring upon hydrolysis.
To overcome the problems associated with the reduction in AHL solubility as the N-acyl chain length increases and the longer time required to obtain acceptable 13C-NMR spectra (which would result in extensive AHL hydrolysis occurring before the spectra were completed), an alternative method for detecting lactone ring opening was developed. In unbuffered aqueous solutions of AHLs, a reduction in pH occurs as a result of hydrolysis by hydroxide ions to open-chain forms. Therefore, the rate of pH change from 7 was used as a measure of ring opening. In effect, the pH change is a logarithmic measure of the amount of hydroxide ions consumed corresponding to the amount of open-chain form released. Known quantities of AHLs were dissolved in water at pH 7 and maintained at the chosen temperature (22 or 37°C) for 7 h, whereas pH was measured as a function of time. Each point was then converted into the concentration of released open-chain form and plotted against time. The slopes of the curves for the four AHLs were calculated 1 h after the beginning of the experiment, thus representing the rate of hydrolysis at that time. The values were normalized to allow for the differences in concentrations of the various AHLs and are shown as relative rates of hydrolysis in Table 1. The estimated error in the calculation of the slopes of the curves was <20%. A measure of the effect of temperature on ring opening was obtained by conducting experiments with equal quantities of the AHLs at 22 or 37°C and calculating the ratios of the rates of hydrolysis at these temperatures. The pH of solutions was measured with a Mettler Toledo MP225 pH meter, calibrated at the appropriate temperature with standard solutions (pH 7, 4, and 10) according to the manufacturer's instructions. Calibration was carried out before and after the experiments, and in no case did the pH of the calibration solutions differ significantly (>0.05 U).
TABLE 1.
Relative rates of hydrolysis of C4-HSL, C6-HSL, 3-oxo-C6, and C8 HSLs to open-chain forms at 22 and 37°Ca
| AHL | Relative rates of hydrolysis at:
|
Rate at 37°C/ rate at 22°C | |
|---|---|---|---|
| 22°C | 37°C | ||
| C4 | 130 | 400 | 3.1 |
| 3-Oxo-C6 | 70 | 170 | 2.4 |
| C6 | 40 | 110 | 2.8 |
| C8 | 1 | 1.5 | 1.5 |
The relative rates of hydrolysis were calculated as described in Materials and Methods from the changes in pH and the corresponding concentrations of open-chain forms with time in unbuffered solutions of the four AHLs (curves similar to those in Fig. 6 for C4-HSL). The quoted figures relate to a standard concentration of AHL (3.7 mM), and the errors in the calculation are estimated to be <20%.
RESULTS
The Y. pseudotuberculosis AHL profile varies as a function of growth and temperature.
When grown in LB medium at 22°C for 24 h, the AHLs 3-oxo-C6-HSL and C6-HSL are clearly visible, together with a much weaker C8-HSL spot, on TLC plates overlaid with the C. violaceum CV026 AHL biosensor (Fig. 1A). However, as the growth temperature is increased to 28°C the intensity of all three spots is substantially reduced (Fig. 1B) and, at 37°C, AHL production appears to have been virtually abolished (Fig. 1C). To investigate AHL production as a function of growth at 37°C, cell-free culture supernatant samples were taken at intervals throughout the growth curve. By using the Chromobacterium AHL biosensor, C6-HSL and 3-oxo-C6-HSL are first detected in mid-logarithmic phase (some 4 h after inoculation), reach a peak in early stationary phase (8 h), and by 14 h no 3-oxo-C6-HSL is detected and the C6-HSL levels are substantially reduced (Fig. 2A). An additional active spot migrating between C6-HSL and C8-HSL is visible in samples obtained between 10 and 16 h; we tentatively identified this by HPLC and TLC in comparison to synthetic AHL standards to be the odd-chain AHL, N-heptanoyl HSL (C7-HSL; data not shown). No AHLs were detected after 24 h of growth.
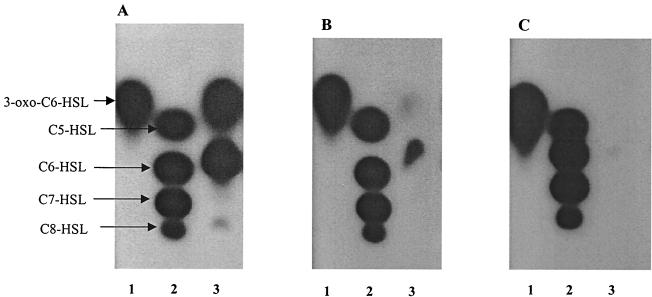
FIG. 1.
TLC chromatogram of the AHLs present in cell-free supernatants of Y. pseudotuberculosis grown with shaking in LB medium at 22°C (A), 28°C (B), and 37°C (C) and then detected by using a C. violaceum CV026 AHL biosensor overlay. Supernatants were extracted with dichloromethane and chromatographed as described in the Materials and Methods. (A) Lane 1, 3-oxo-C6-HSL; 1.4 × 10−8 mol; lane 2, the AHL standards, C5-HSL (5.4 × 10−8 mol), C6-HSL (5 × 10−9 mol), C7-HSL (4.7 × 10−7 mol), and C8-HSL (2.2 × 10−8 mol); lane 3, Y. pseudotuberculosis supernatant extract from cultures grown at 37°C. (B) Same as panel A, but lane 3 contains extract from cultures grown at 28°C. (C) Same as panel A but lane 3 contains extract from cultures grown at 37°C.
FIG. 2.
TLC chromatogram of AHL production as a function of growth in Y. pseudotuberculosis cultures grown with shaking at 37°C in LB medium alone (A) or in LB medium buffered with MOPS (50 mM) (B) and then detected by using a C. violaceum CV026 AHL biosensor overlay. Lanes 1 and 14, 3-oxo-C6-HSL (1.4 × 10−8 mol); lanes 2 and 13, C6-HSL (5 × 10−9 mol) and C8-HSL (2.2 × 10−8 mol). Lanes 3 to 12 show the AHL profiles of culture supernatants extracted at 0, 2, 4, 6, 8, 10, 12, 14, and 24 h, respectively. The respective growth curves plotted as the log OD600 versus time are superimposed on the TLC plates. For panel A the culture increased from pH 7.2 to 8.5 over the growth curve, and for panel B the pH increased from 6.7 to 7.6. The pH at each respective time point is shown on each chromatogram.
To determine whether exogenous AHLs could be inactivated in stationary-phase culture supernatants, we added concentrations of up to 100 μM of either C6-HSL or 3-oxo-C6-HSL. Both compounds were completely inactivated after 24 h of incubation, as indicated by using the LuxR-based AHL biosensor, E. coli(pSB401) (data not shown)
AHL turnover in cultures of Y. pseudotuberculosis is due to lactonolysis.
Consideration of the chemistry of lactone rings (41) suggests that AHLs may be hydrolyzed under alkaline conditions to open-chain forms. The pH of cultures of Y. pseudotuberculosis in LB medium at 37°C was determined at intervals throughout the growth curve and was found to range from pH 7.1 at the point of inoculation to pH 8.5 after 24 h of growth. Thus, the loss of detectable AHLs in stationary-phase supernatants may be a consequence of the nonenzymatic hydrolysis of the lactone ring. To explore this possibility, we first adjusted the pH of stationary-phase culture supernatants to pH 7.0 with HCl and added up to 100 μM of either C6-HSL or 3-oxo-C6-HSL. In contrast to spent stationary-phase culture supernatants at pH 8.5, no loss of AHL activity was observed after reduction of the pH to neutral (data not shown).
Cell-free supernatants were also prepared from 24-h stationary-phase Y. pseudotuberculosis cultures grown at 37°C, and their pH levels were adjusted to 6.8, 4.0, 3.0, and 2.0. Figure 3 shows that after incubation for 24 h at 37°C, reactivation of AHLs was apparent only after the pH had been reduced to <3.0. In samples incubated at pH 2.0, four active spots are clearly visible on TLC plates overlaid with C. violaceum CV026. We noted, in addition to 3-oxo-C6-HSL, C6-HSL, and C7-HSL, another spot running between C6-HSL and 3-oxo-C6-HSL (Fig. 3, lane 5); we tentatively identified this as C5-HSL after HPLC fractionation and further TLC analysis in comparison to synthetic AHL standards (data not shown). Given that the hydrolysis of AHLs in stationary-phase spent culture supernatants depends on pH, we grew Y. pseudotuberculosis at 37°C in LB medium and LB medium supplemented by 50 mM MOPS buffer and took samples at intervals for growth, pH determination, and AHL analysis. Figure 2B reveals that the growth of Y. pseudotuberculosis in MOPS-buffered LB medium was very similar to that observed for LB medium alone (Fig. 2A), with the organism reaching final OD600s of 3.7 and 3.8, respectively. In MOPS-buffered LB medium, the pH ranged from 6.7 to 7.6 over the growth curve compared with 7.16 to 8.5 for LB medium alone. For both media, AHLs were first detected after 4 h but in buffered LB medium it is clear that the levels of AHLs remain much higher throughout growth. C6-HSL and C7-HSL are also clearly present after 24 h growth but only in the MOPS-buffered medium.
FIG. 3.
TLC chromatogram showing the effect of acidification on the recovery of AHLs from Y. pseudotuberculosis cultures grown in LB medium for 24 h at 37°C and then detected by using a C. violaceum CV026 AHL biosensor overlay. The stationary-phase cell-free supernatant reached pH 8.4 after 24 h. Prior to extraction with dichloromethane, the pH of each sample was adjusted with HCl and incubated for 24 h at 37°C. Lane 1, control (pH 8.4); lane 2, pH 6.8; lane 3, pH 4.0; lane 4, pH 3.0; lane 5, pH 2.0; lane 6, C6-HSL (5 × 10−9 mol) and C8-HSL (2.2 × 10−8 mol); lane 7, 3-oxo-C6-HSL (1.4 × 10−8 mol).
Accumulation of open-ring AHLs in P. aeruginosa culture supernatants.
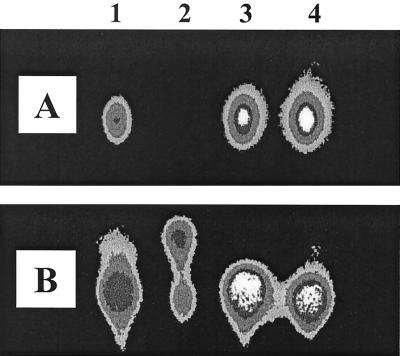
P. aeruginosa produces two major AHLs: C4-HSL and 3-oxo-C12-HSL. Incubation of these AHLs with cell extracts of P. aeruginosa had little effect on activity (data not shown). However, when a 100 μM concentration of C4-HSL or 3-oxo-C12-HSL was incubated with stationary-phase cell-free culture supernatants, C4-HSL was not detectable after 6 h of incubation and the level of 3-oxo-C12-HSL was substantially reduced (Fig. 4). While neutralization of supernatants prior to AHL addition prevented their inactivation (Fig. 4), treatment of the supernatants with either proteinase K or the protease inhibitor phenylmethylsulfonyl fluoride did not affect inactivation of either AHL (data not shown). In Fig. 4B (lane 2), we noted that the addition of 3-oxo-C12-HSL to supernatants resulted in the production of a new signal with greater mobility than that of 3-oxo-C12-HSL. This molecule was subsequently identified as the open-ring form of 3-oxo-C12-HSL (see below).
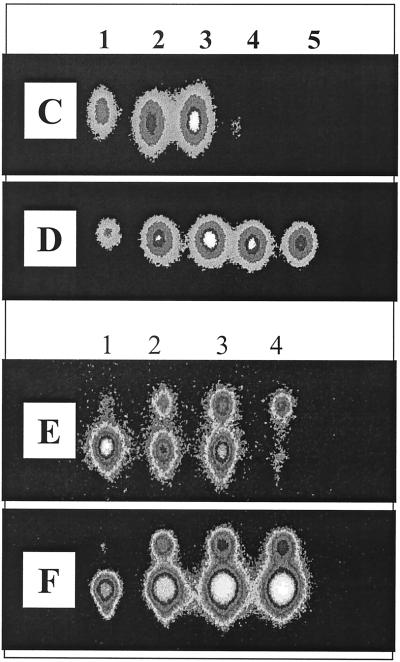
FIG. 4.
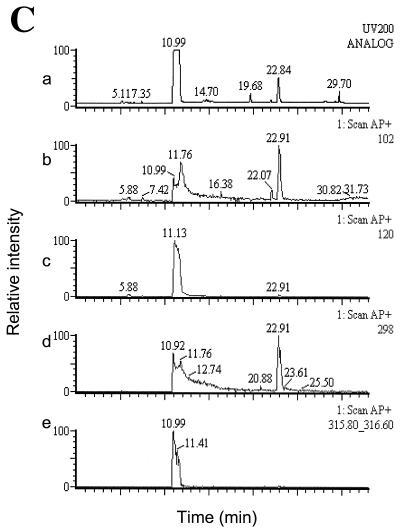
TLC chromatogram of C4-HSL (A) and 3-oxo-C12-HSL (B) after incubation in stationary-phase cell-free P. aeruginosa culture supernatants before (lanes 1 and 2) or after (lanes 3 and 4) neutralization. AHLs were added at a final concentration of 100 μM to filter-sterilized supernatants (pH 8.62) of P. aeruginosa grown in LB medium for 24 h. Samples (1 μl/spot) were spotted on TLC plates directly after the addition of AHLs to supernatants (lanes 1 and 3) and after 6 h of incubation (lanes 2 and 4); for lanes 3 and 4, the supernatant was neutralized with HCl prior to the addition of AHLs. Samples in lanes 1, 3, and 4 were diluted 1:10 with acetonitrile before spotting them on the TLC plate. C4-HSL and 3-oxo-C12-HSL were detected by overlaying TLC plates with either E. coli(pSB536) or E. coli(pSB1075). (C) LC-MS analysis of alkali-treated 3-oxo-C12-HSL (5 mM) treated with NaOH (5 mM) for 15 min prior to HPLC injection. (a) UV trace at 200 nm. The peak with a retention time of ∼11 min is the open-ring form of 3-oxo-C12-HSL and that at ∼22.9 min is the closed-ring form. (b) Scan for the positive ion (M+) at 102 Da (equivalent to HSL). (c) Scan for M+ at 120 Da (equivalent to homoserine). (d) Scan for M+ at 298 Da (equivalent to 3-oxo-C12-HSL). (e) Scan for M+ at 316 Da [equivalent to N-(3-oxododecanoyl)homoserine, the open-ring form of 3-oxo-C12-HSL].
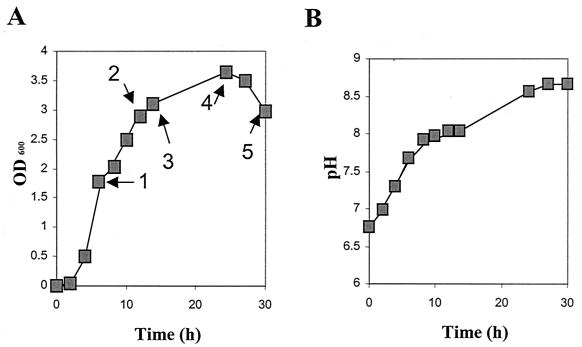
To examine the fate of C4-HSL and 3-oxo-C12-HSL as a function of growth in LB medium at 37°C, we used the lux-based AHL biosensors E. coli(pSB536) and E. coli(pSB1075), respectively. Figure 5A and B reveals that there was a steady increase in pH during growth of P. aeruginosa in LB medium. Slightly alkaline values (pH >7.5) were reached after 6 h, and at 24 h the pH was 8.5. Supernatant samples were taken after 6, 12, 14, 24, and 30 h for AHL measurements. Analysis of AHL levels by using the AHL biosensor E. coli(pSB536) in a liquid assay showed that C4-HSL levels in LB medium increased from 6 to 14 h and that C4-HSL was >90% inactivated after 24 h of growth (data not shown). Acidification of 24- and 30-h supernatant samples prior to AHL analysis restored activity. The level of 3-oxo-C12-HSL readily detected at 6 h with E. coli(pSB1075) was reduced by ∼50% after 24 h of growth (data not shown). Since 3-oxo-C12-HSL can antagonize the activity of C4-HSL (50), the results for C4-HSL were confirmed by TLC analysis, which overcomes any antagonistic activity by separating C4-HSL from 3-oxo-C12-HSL (Fig. 5C to F). For 3-oxo-C12-HSL, TLC analysis (Fig. 5E and F) shows that acidification also recovers the activity of 3-oxo-C12-HSL in stationary-phase culture supernatants. In addition, a second active spot migrating faster, i.e., it was more more hydrophilic than 3-oxo-C12-HSL, is apparent on the TLC plates. This spot is likely to contain, at least in part, N-(3-oxodecanoyl)HSL (3-oxo-C10-HSL), a minor AHL produced by P. aeruginosa (7) that comigrates with synthetic 3-oxo-C10-HSL in this TLC assay (data not shown). The appearance of a spot at the same position in the nonacidified samples (e.g., Fig. 5E, lanes 2 to 4) can also be explained by the detection of the open-ring form of 3-oxo-C12-HSL, which also comigrates with 3-oxo-C10-HSL (see below).
FIG. 5.
AHL production in P. aeruginosa cultures as a function of growth in LB medium with shaking at 37°C. (A) OD600. Numbered arrows indicate time points of sampling for AHL-analysis. (B) Variation in pH as a function of growth in LB. (C to F) TLC chromatograms of the culture supernatants prepared as described above and overlaid with E. coli(pSB536) (C and D) and E. coli(pSB1075) (E and F). Prior to TLC analysis, the samples shown in panels D and F were acidified with HCl. For panels C to F, lanes 1 to 5 correspond to the numbered arrows in panel A as follows: lane 1, 6 h of growth; lane 2, 12 h of growth; lane 3, 14 h of growth; lane 4, 24 h of growth; and lane 5, 30 h of growth.
Recyclization of the 3-oxo-C12-HSL ring during TLC analysis.
When synthetic 3-oxo-C12-HSL was added to alkaline stationary-phase P. aeruginosa supernatants, a spot that comigrates with 3-oxo-C10-HSL was observed after TLC analysis (Fig. 4B, lane 2). One possible explanation is that the new signal is related to the open-ring form of 3-oxo-C12-HSL. HPLC analysis of 3-oxo-C12-HSL (retention time, ∼23 min) treated with alkali revealed the formation of a less hydrophobic molecule with a retention time of ca. 11 min (data not shown). When the fraction eluting at 11 min was collected and subjected to TLC analysis, the E. coli(pSB1075) AHL biosensor revealed the presence of an active spot that comigrated with the novel signal observed in Fig. 4B, lane 2 (also data not shown). LC-MS analysis (Fig. 4C) revealed that this product had a molecular ion [M+H] of 316 that corresponds to N-(3-oxododecanoyl)homoserine, i.e., the open-ring form of 3-oxo-C12-HSL (which itself has a molecular ion [M+H] of 298). The spectrum also contained the characteristic fragmentation product of m/z 120 corresponding to homoserine (Fig. 4C). However, in liquid assays with the E. coli(pSB1075) biosensor, the open-ring form of 3-oxo-C12-HSL had no activity and did not antagonize the activity of 3-oxo-C12-HSL (data not shown). This suggests that the novel signal observed on the TLC plate bioassays arises via the recyclization of the lactone ring as a consequence of the open-ring form of 3-oxo-C12-HSL interacting with the hydrophobic RP2 chromatographic support matrix. This recyclization was also noted to occur after HPLC analysis of N-(3-oxododecanoyl)homoserine on reversed-phase RP8 HPLC columns, where the mass of the closed-ring form was also detected concomitant with the mass of the open-ring form.
Lactonolysis of HSL, C3-HSL, and C4-HSL as a function of pH.
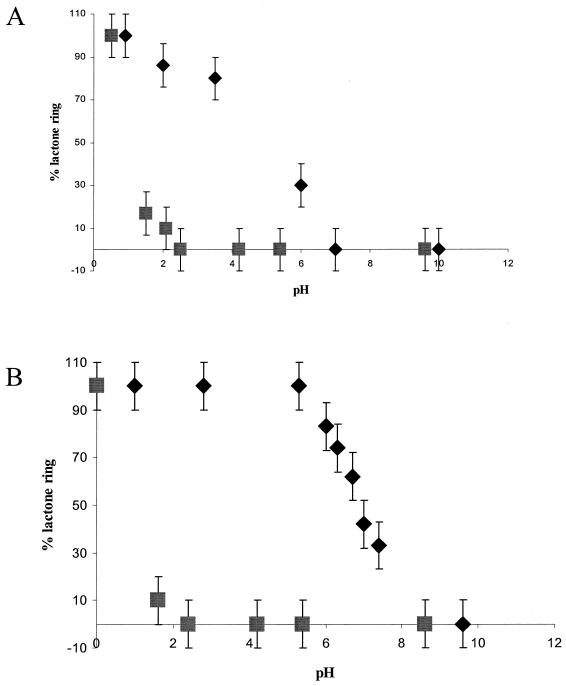
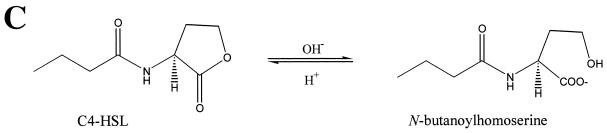
To explore the hydrolysis of HSL and the AHLs in more detail, a chemical approach using 13C NMR to investigate the influence of pH on ring opening and closing was undertaken. Figure 6A shows that the ring of HSL is largely open when the pH was raised from <1 to 2. By introducing a C3 acyl chain, the ring was largely intact at pH 2, ∼30% remained at pH 6, and a pH of 7 was required for full ring opening. Similar 13C-NMR experiments showed that the ring of C4-HSL was more stable than that of C3-HSL. As the pH was raised, the ring of C4-HSL remained largely intact until pH 5 to 6 and was only completely opened at pH 8 (Fig. 6B, compare with C3-HSL in Fig. 6A). Conversely, closure of the N-butanoylhomoserine (i.e., the open-ring form of C4-HSL) ring upon reduction of pH is not a simple reversal of the ring opening curve shown for C4-HSL as the pH is increased (Fig. 6B, diamonds). Instead, the curve obtained (Fig. 6B, boxes) follows a different route. This is because the pH must first approach the pK of the carboxyl group (ca. pH 2) before appreciable quantities of the acid (as distinct from the salt of the acid) are present. Lactonization can then occur with the hydroxyl group to form a closed ring (shown at low pH values in Fig. 6B). Thus, the ring-opening and ring-closing processes exhibit different curves (or “hysteresis”). For C4-HSL, these data show that, whereas ring opening occurred between pH 5 to 8, the ring reformed only when the pH was <2 (Fig. 6B). The structures of C4-HSL and its hydrolysis product, N-butanoylhomoserine, are shown in Fig. 6C.
FIG. 6.
Opening and closing of lactone rings of HSL, C3-HSL, and C4-HSL as determined by using 13C-NMR spectroscopy. (A) Proportion of closed-ring form persisting in aqueous solutions of HSL (▪) and C3-HSL (♦) as a function of pH. (B) Proportion of closed-ring form persisting in an aqueous solution of C4-HSL as the pH is increased (♦) and the ring closure observed as the pH was reduced (▪). (C) Structure of C4-HSL and the corresponding product (a salt) formed after lactonolysis with hydroxide ions. It is not until the pH approaches the pK of the carboxylic acid group that appreciable quantities of the closed ring can form.
pH- and temperature-dependent hydrolysis of C4-HSL, 3-oxo-C6-HSL, C6-HSL, and C8-HSL.
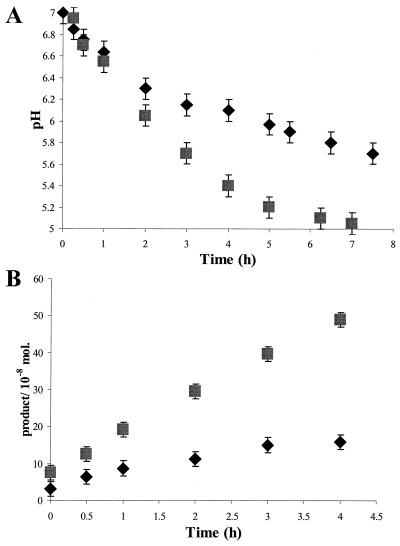
To overcome the problems associated with both the reduction in AHL solubility as the acyl chain length increases and the extended times required to obtain acceptable 13C-NMR spectra, we devised an alternative method for detecting lactone ring opening. In unbuffered aqueous solutions of AHLs, a reduction in pH occurs as a result of hydrolysis by hydroxide ions to open-chain forms. Figure 7A shows the pH reduction with time for C4-HSL at 22°C and the greater reduction at 37°C; similar curves were obtained for C6-HSL, 3-oxo-C6-HSL, and C8-HSL, but the rates of reduction varied with different AHLs (data not shown). Figure 7B shows the change with time at 22 and 37°C of the concentration of the released open-chain form of C4-HSL calculated from the pH curves. Similar curves were obtained for the other three AHLs (data not shown). The relative rates of hydrolysis of C4-HSL, 3-oxo-C6-HSL, C6-HSL, and C8-HSLs to open-chain forms at 22 and 37°C were calculated from the curves and are shown in Table 1. At both 22 and 37°C, the rate of hydrolysis is reduced as the length of the N-linked acyl side chain increases. The introduction of a 3-oxo group into the side chain of C6-HSL also increases the rate of hydrolysis (Table 1). The rates of hydrolysis are much greater at 37°C than at 22°C (Table 1), and there is a tendency for the AHL with the longest side chain to be more stable at higher temperatures.
FIG. 7.
Change in pH with time as a measure of the lactonolysis of C4-HSL in unbuffered aqueous solution. (A) Change of pH with time at 22°C (♦) and 37°C (▪). (B) Calculated concentrations of open chain form released with time at 22°C (♦) and 37°C (▪).
DISCUSSION
Since there is little information on the turnover and fate of AHL quorum-sensing signal molecules in bacterial cultures, we examined, as a function of growth, the AHL profiles of Y. pseudotuberculosis and P. aeruginosa, two human pathogens that make a range of AHLs with N-acyl side chains from C4 to C12 in length, with or without 3-oxo substituents. The growth conditions (LB medium, high aeration rate) were chosen because they have been widely used in studies of AHL-dependent quorum sensing. In Y. pseudotuberculosis supernatants prepared from cultures grown for 24 h to stationary phase at 37°C, C6-HSL and 3-oxo-C6-HSL were almost undetectable compared to mid- to late-logarithmic-phase cultures. Similarly, P. aeruginosa stationary-phase cultures grown under the same conditions also contained much lower levels of C4-HSL and 3-oxo-C12-HSL. For both organisms, we could find no evidence for the presence of AHL-inactivating enzymes and, when added to stationary-phase culture supernatants at concentrations of up to 100 μM, each of the exogenously provided AHLs was rapidly inactivated, suggesting a potential nonenzymatic basis for the AHL turnover observed. By buffering the growth medium or by acidifying the stationary-phase culture supernatants to pH <2.0, the apparent turnover of AHLs could be reduced or the AHLs could be recovered. The increase in the pH of the LB medium during the growth of Y. pseudotuberculosis and P. aeruginosa is presumably due to the release of ammonia from aerobic degradation of peptides as carbon and energy sources. The inactivation of AHLs during the growth of Yersinia and Pseudomonas in LB medium is therefore a consequence of the pH-dependent hydrolysis of the lactone ring such that, in the alkaline pHs achieved in stationary-phase LB medium, the open-ring form accumulates. In the plant pathogen, Erwinia carotovora, Holden et al. (14) noted that 3-oxo-C6-HSL levels in LB medium reach a peak at the onset of stationary phase (10 to 12 h) and fall to almost undetectable levels after 30 h. During the preparation of the present study, Byers et al. (5) reported that the microbially induced loss of 3-oxo-C6-HSL in E. carotovora LB cultures was due to pH-dependent hydrolysis, although these authors were unable to propose a breakdown mechanism. Their attempts to regain activity by acidifying the medium to pH 1.5 were probably unsuccessful because, as we have demonstrated here, prolonged incubation times are required to recyclize the HSL ring. AHL levels in the plant symbiont Rhizobium leguminosarum (2) have also been studied as a function of growth. C6-HSL, 3-oxo-C8-HSL, and N-(3-hydroxy-7-cis-tetradecenoyl)HSL (3OH,C14:1-HSL) levels are all reduced markedly in the stationary phase. This may also be a consequence of the pH-dependent hydrolysis of the lactone ring. Thus, in any studies attempting to quantify AHL production as a function of growth, it is essential to control the pH of the growth medium or to acidify spent supernatants to a pH <2 (for up to 24 h). This is essential for reversing the hydrolysis of the lactone ring as revealed by (i) the lack of ring closure observed in Y. pseudtotuberculosis stationary-phase cultures adjusted to pHs between 3.0 and 6.8 and (ii) the hysteresis in ring formation observed during the 13C-NMR experiments.
Y. pseudotuberculosis is a mammalian enteropathogen with a biphasic lifestyle alternating between food and water and the mammalian gastrointestinal tract and can adapt to growth at a variety of different temperatures (4). Temperature is also a key environmental parameter responsible for regulating the expression of yersinia virulence factors, including Yops (type III secreted proteins), invasin, flagellins, and lipopolysaccharide (10). Interestingly, when grown at lower temperatures, notably at 22°C, Y. pseudotuberculosis appears to produce more AHLs than at 37°C (1). These observations could be interpreted to imply that temperature influences the expression of the yersinia LuxI homologues, YpsI and YtbI, or the function of the YpsI and YtbI proteins or even the availability of substrates for AHL synthesis. However, the apparent reductions in AHL levels as the growth temperature is increased are more likely to be due to the increased rate of pH-dependent hydrolysis of AHLs at the higher temperatures. Thus, any studies of AHL production as a function of growth environment must be interpreted with caution and with due consideration for both growth temperature and pH.
By acidifying stationary-phase Y. pseudotuberculosis wild-type culture supernatants prior to dichloromethane extraction, we noted the appearance of two AHLs which migrated on TLC with different Rf values to the previously reported Y. pseudotuberculosis AHLs (1). By subjecting the extracts to HPLC prior to TLC analysis, we were able to tentatively identify these compounds by comparing them to synthetic standards as C5-HSL and C7-HSL. Most of the AHLs characterized to date have even numbers of acyl side chain carbons, although C7-HSL has also been identified in R. leguminosarum. In this plant symbiont, production of C7-HSL appears to depend on the nature of the growth medium used (26). This has led to the suggestion that for these odd-number chain length AHLs, propionyl-coenzyme A (CoA), together with acetyl-CoA and/or malonyl-CoA, could be the acyl chain starter and extender units (26). This in turn may result in the formation of precursors for acyl chains in which an odd number of carbon atoms are present.
Since gram-negative bacteria produce a range of AHLs differing in the length of the acyl side chain and the C3 substituent, we examined the fate of HSL and a range of AHLs as a function of pH, temperature, and acyl chain length. The 13C-NMR measurements of the pH-driven ring opening and closing of HSL, C3-HSL, and C4-HSL allowed us to draw two main conclusions. First, resistance to ring opening caused by an increase in pH increases with the length of the side chain. Second, on returning to acidic conditions, ring closure occurs by a mechanism that is different from a simple reversal of the mechanism for opening. Ring closure will not take place to a significant extent until the pH has been reduced to a level comparable with the pK of the acid group. Furthermore, these data also indicate that neither HSL nor C3-HSL will be useful as quorum-sensing signal molecules since they rapidly hydrolyze at pHs below the physiological level and thus suggest that C4-HSL is likely to be the shortest-chain AHL useful to a mammalian pathogen.
The rates of hydrolysis of C4-HSL, 3-oxo-C6-HSL, C6-HSL, and C8-HSL in unbuffered aqueous solution were calculated from the change of pH with time. The data obtained demonstrated that at neutral and slightly acidic pHs the rings of all AHLs opened to some extent but that the rate was influenced by the nature of the acyl side chain. This rate was slower for the AHLs with the longer side chains. As might be anticipated, the rates of hydrolysis of all of the AHLs examined increased when the temperature was raised from 22 to 37°C. These results support the conclusions derived from the 13C-NMR experiments, namely, that an increase in length of the side chain stabilizes the closed-ring form. Longer acyl side chains donate electrons more strongly to the carbonyl group than do shorter side chains, and this makes the lactone ring less less electrophilic and consequently less susceptible to attack by hydroxide ions. Electronegative substitutions in the side chain, such as in 3-oxo-C6-HSL, decrease its overall electron-donating capacity and hence lead to the reduced stability of the lactone ring (compared to the relative rates of hydrolysis of C6-HSL and 3-oxo-C6-HSL shown in Table 1). Thus, for a given growth environment, the concentration of biologically active AHL (i.e., closed ring) available will depend on a variety of parameters that will include the rate of biosynthesis, the rate of export into the medium (in the case of molecules with long acyl side chains), and the inactivation rate. Thus, the longer the AHL acyl chain, the more persistent the molecule is likely to be in mammalian tissue fluids at pH 7.4 and at 37°C. However, given their hydrophobic properties, longer-chain AHLs may be rapidly partitioned into tissue cell membranes or sequestered by serum proteins such that they are effectively removed from the immediate vicinity of the infecting microbe. It is therefore interesting in this context to note that P. aeruginosa, which is capable of infecting almost every body site, employs both short (C4-HSL)- and long (3-oxo-C12-HSL)-chain AHL to control many of the same virulence genes. In P. aeruginosa, 3-oxo-C12-HSL is produced in advance of C4-HSL and controls C4-HSL production via activation of LasR (20).
Interestingly, the open-ring form of 3-oxo-C12-HSL gave rise to a new signal detected by the E. coli(pSB1075) biosensor after separation by TLC. This signal was dependent on recyclization of the lactone ring and did not take place in liquid AHL biosensor assay medium, suggesting that the hydrophobic interactions occurring with the TLC plate RP2 matrix may lower the activation energy for ring closure and so catalyze AHL ring closure. With shorter-chain AHLs such as C4-HSL, this phenomenon did not occur, probably because their hydrophobic interactions are too weak. Thus, in addition to their higher stability at alkaline pHs, long-chain AHLs have a further advantage because, once hydrolyzed, they can regain biological activity at almost-neutral pHs. Hydrophobic surfaces capable of catalyzing lactone ring closure could be provided by any appropriate surface on which P. aeruginosa can form biofilms. These surfaces may include tissue surfaces, catheters, or implanted medical devices.
The data presented here suggest that, for bacteria employing AHL-dependent quorum sensing, the rate at which a population of bacteria become “quorate,” i.e., reach a critical AHL threshold concentration, will be significantly influenced by the local pH and temperature. This is because the local environment will influence the rate of turnover and thus the rate of AHL accumulation. This in turn will be further affected by AHL structure since molecules with longer acyl side chains and lacking a 3-oxo substituent are less prone to hydrolysis than are shorter chain signal molecules. Thus, for pathogenic bacteria that employ AHL-dependent quorum sensing to regulate virulence gene expression, the size of the quorum is likely to vary with the pH of the infected body site. In Table 2 we have summarized the pHs of mammalian body sites that are susceptible to infection by Y. pseudotuberculosis, which in humans primarily infects the gastrointestinal tract (4), and by P. aeruginosa, which, under the appropriate conditions, can infect almost any body site (27). Only three of the fluids (bile, tears, and urine) can have pHs of <8, which could result in substantial hydrolysis of AHLs. Also, only gastric juices and urine can have pHs of <5, which would stabilize the AHLs. The majority of body fluids have pHs ranging from 6 to 8, at which some AHL hydrolysis would take place, especially in the buffered conditions that occur in vivo. These conditions may provide an abundance of hydroxide ions for hydrolysis of the relatively small amounts of AHLs produced by an infecting bacterium. Nevertheless, the fact that detectable levels of AHLs are produced in Y. pseudotuberculosis and P. aeruginosa cultures when the pH is between 6 and 8 strongly suggests that any hydrolysis that does occur during growth in vivo would be more than compensated for by the production of additional AHLs by the infecting organism. Interestingly, P. aeruginosa, which produces both short-chain (C4-HSL) and long-chain (3-oxo-C12-HSL) AHLs appears to be capable of infecting more body sites than Y. pseudotuberculosis, which to date, has been shown to produce primarily AHLs with C6 and C8 acyl side chains. Long-chain AHLs, given their more hydrophobic character, are less diffusible than short-chain AHLs and, according to our results, are more stable under slightly alkaline conditions. Also, they may be less inactivated by the rises in temperature that may accompany infection. Obviously, the production of a range of AHLs with acyl side chains of different lengths provides the pathogen with greater versatility in adapting to changing conditions in vivo. However, given that mutation of the AHL synthase genes lasI and rhlI significantly reduces the virulence of P. aeruginosa (38, 34), it is likely that AHLs will be produced in vivo in quantities that can withstand some inactivation by the environmental pH. This has been demonstrated for cystic fibrosis patients infected with P. aeruginosa and Burkholderia cepacia; in these patients, AHLs produced in sputum by the infecting bacteria have been directly detected by using AHL biosensors, TLC, and MS (12a, 29).
TABLE 2.
pH values for human body fluids
| Fluida | pH range |
|---|---|
| Amniotic fluid | 7.0-7.3 |
| Aqueous humour | 7.2-7.4 |
| Bile | 6.4-8.6 |
| Cerebrospinal fluid | 7.3-7.4 |
| Faeces | 5.0-8.0 |
| Gastric juice | 2.0-4.0 |
| Small intestine fluid | 5.0-7.0 |
| Large intestine fluid | 6.5-7.5 |
| Saliva | 6.8-7.5 |
| Sperm | 7.2-7.6 |
| Synovial fluid | 7.3-7.6 |
| Tears | 7.4-8.2 |
| Urine | 4.5-8.2 |
| Blood | 7.1-7.4 |
| Airway gland fluidb | ≈6.97 |
| Cystic fibrosis sputum (sol)c | 5.9-7.0 |
Acknowledgments
We thank Mavis Daykin and Catherine Ortori for technical assistance and Steve Diggle and Klaus Winzer for helpful discussions.
Work at Nottingham was funded by grants and a studentship from the Biotechnology and Biological Sciences Research Council (United Kingdom) by a Medical Research Council Cooperative Group core grant and by European Union contract BIO4-CT96-0181 (which also supported the Gif-sur-Yvette group). B.P. was supported by the Deutsche Forschungsgemeinschaft.
E.A.Y., B.P., and C.B. contributed equally to this study.
Editor: A. D. O'Brien
REFERENCES
- 1.Atkinson, S., J. P. Throup, G. S. A. B. Stewart, and P. Williams. 1999. A hierarchical quorum-sensing system in Yersinia pseudotuberculosis is involved in the regulation of motility and clumping. Mol. Microbiol. 33:1267-1277. [DOI] [PubMed] [Google Scholar]
- 2.Blosser-Middleton, R. S., and K. M. Gray. 2001. Multiple N-acylhomoserine lactone signals of Rhizobium leguminosarum are synthesized in a distinct temporal pattern. J. Bacteriol. 183:6771-6777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brint, J. M., and D. E. Ohman. 1995. Synthesis of multiple exoproducts in Pseudomonas aeruginosa is under the control of RhlR-RhlI, another set of regulators in strain PAO1 with homology to the autoinducer responsive LuxR-LuxI family. J. Bacteriol. 177:7155-7163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Butler, T. 1994. Yersinia infection. Centennial of the discovery of the plague bacillus. Clin. Infect. Dis. 19:655-661. [DOI] [PubMed] [Google Scholar]
- 5.Byers, J. T., C. Lucas, G. P. C. Salmond, and M. Welch. 2002. Nonenzymatic turnover of an Erwinia carotovora quorum sensing signaling molecule. J. Bacteriol. 184:1163-1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chapon-Hervé, V., M. Akrim, A. Latifi, P. Williams, A. Lazdunski, and M. Bally. 1997. Regulation of the xcp secretion pathway by multiple quorum-sensing modulons in Pseudomonas aeruginosa. Mol. Microbiol. 24:1169-1178. [DOI] [PubMed] [Google Scholar]
- 7.Charlton, T. S., R. de Nys, A. Netting, N. Kumar, M. Hentzer, M. Givskov, and S. Kjelleberg. 2000. A novel and sensitive method for the quantification of N-3-oxoacyl homoserine lactones using gas chromatography-mass spectrometry: application to a model bacterial biofilm. Environ. Microbiol. 2:530-541. [DOI] [PubMed] [Google Scholar]
- 8.Chhabra, S. R., P. Stead, N. J. Bainton, G. P. C. Salmond, G. S. A. B. Stewart, P. Williams, and B. W. Bycroft. 1993. Autoregulation of carbapenem biosynthesis in Erwinia carotovora by analogues of N-(3-oxohexanoyl)-l-homoserine lactone. J. Antibiot. 46:441-454. [DOI] [PubMed] [Google Scholar]
- 9.Chugani, S. A., M. Whitely, K. M. Lee, D. D'Argenio, C. Manoil, and E. P. Greenberg. 2001. QscR, a modulator of quorum-sensing signal synthesis and virulence in Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 98:2752-2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cornelis, G. R., and H. Wolf-Watz. 1997. The Yersinia Yop virulon: a bacterial system for subverting eukaryotic cells. Mol. Microbiol. 23:861-867. [DOI] [PubMed] [Google Scholar]
- 11.Davies, D. G., M. R. Parsek, J. P. Pearson, B. H. Iglewski, J. W. Costerton, and E. P. Greenberg. 1998. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 280:295-298. [DOI] [PubMed] [Google Scholar]
- 12.Dong, Y.-H., L.-H. Wang, J.-L. Xu, H.-B. Zhang, X.-F. Zhang, and L.-H. Zhang. 2001. Quenching quorum sensing-dependent bacterial infection by an N-acylhomoserine lactonase. Nature 411:813-817. [DOI] [PubMed] [Google Scholar]
- 12a.Erickson, D. L., R. Endersby, A. Kirkham, K. Stuber, D. D. Vollman, H. R. Rabin, I. Mitchell, and D. G. Storey. 2002. Pseudomonas aeruginosa quorum-sensing systems may control virulence factor expression in the lungs of patients with cystic fibrosis. Infect. Immun. 70:1783-1790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gardiner, S., S. R. Chhabra, C. Harty, D. I. Pritchard, B. W. Bycroft, P. Williams, and T. Bennett. 2001. Haemodynamic properties of bacterial quorum sensing signal molecules. Br. J. Pharmacol. 133:1047-1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Holden, M. T. G., S. J. McGowan, B. W. Bycroft, G. S. A. B. Stewart, P. Williams, and G. P. C. Salmond. 1998. Cryptic carbapenem antibiotic production genes are widespread in Erwinia carotovora: facile transactivation by the carR transcriptional regulator. Microbiology 144:1495-1508. [DOI] [PubMed] [Google Scholar]
- 15.Jayaraman, S., N. Soo Joo, B. Reitz, J. J. Wine, and A. S. Verkman. 2001. Submucosal gland secretions in airways from cystic fibrosis patients have normal [Na+] and pH but elevated viscosity. Proc. Natl. Acad. Sci. USA 98:8119-8123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jiang, Y., M. Cámara, S. R. Chhabra, K. H. Hardie, B. W. Bycroft, A. Lazdunski, G. P. Salmond, G. S. A. B. Stewart, and P. Williams. 1998. In vitro biosynthesis of the Pseudomonas aeruginosa quorum sensing signal molecule N-butanoyl-l-homoserine lactone. Mol. Microbiol. 28:193-203. [DOI] [PubMed] [Google Scholar]
- 17.Jones, S., B. Yu, N. J. Bainton, M. Birdsall, B. W. Bycroft, S. R. Chhabra, A. J. R. Cox, P. Golby, P. J. Reeves, S. Stephens, M. K. Winson, G. P. C. Salmond, G. A. S. B. Stewart, and P. Williams. 1993. The lux autoinducer regulates the production of exoenzyme virulence determinants in Erwinia carotovora and Pseudomonas aeruginosa. EMBO J. 12:2477-2482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kaplan, H. B., and E. P. Greenberg. 1985. Diffusion of autoinducer is involved in regulation of the Vibrio fischeri luminescence system. J. Bacteriol. 163:1210-1214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Latifi, A., M. K. Winson, M. Foglino, B. W. Bycroft, G. S. A. B. Stewart, A. Lazdunski, and P. Williams. 1995. Multiple homologues of LuxR and LuxI control expression of virulence determinants and secondary metabolites through quorum sensing in Pseudomonas aeruginosa PAO1. Mol. Microbiol. 17:333-343. [DOI] [PubMed] [Google Scholar]
- 20.Latifi, A., M. Foglino, K. Tanaka, P. Williams, and A. Lazdunski. 1996. A hierarchical quorum sensing cascade in Pseudomonas aeruginosa links the transcriptional activators LasR and RhlR (VsmR) to expression of the stationary phase sigma factor RpoS. Mol. Microbiol. 21:1137-1146. [DOI] [PubMed] [Google Scholar]
- 21.Laue, B. E., Y. Jiang, S. Ram Chhabra, S. Jacob, G. S. A. B. Stewart, A. Hardman, J. A. Downie, F. O'Gara, and P. Williams. 2000. The biocontrol strain Pseudomonas fluorescens F113 produces the Rhizobium small bacteriocin, N-(3-hydroxy-7-cis-tetradecenoyl)homoserine lactone, via HdtS, a putative novel N-acylhomoserine lactone synthase. Microbiology 146:2469-2480. [DOI] [PubMed] [Google Scholar]
- 22.Lawrence, R. N., W. R. Dunn, B. W. Bycroft, M. Camara, S. R. Chhabra, P. Williams, and V. G. Wilson. 1999. The Pseudomonas aeruginosa quorum sensing signal molecule, N-(3-oxododecanoyl)-l-homoserine lactone inhibits poircine arterial smooth muscle contraction. Br. J. Pharmacol. 128:845-848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Leadbetter, J. R., and E. P. Greenberg. 2000. Metabolism of acyl-homoserine lactone quorum sensing signals by Variovorax paradoxus. J. Bacteriol. 182:6921-6926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lentner, C. 1981. Geigy scientific tables, vol. 1. Units of measurements, body fluids, composition of the body, nutrition. Ciba Geigy, Ltd., Basel, Switzerland.
- 25.Lentner, C. 1984. Geigy scientific tables, vol. 3. Physical chemistry, composition of the blood, hematology, somatometric data. Ciba Geigy, Ltd., Basel, Switzerland.
- 26.Lithgow, J. K., A. Wilkinson, A. Hardman, B. Rodelas, F. Wisniewski-Dye, P. Williams, and J. A. Downie. 2000. The regulatory locus cinRI in Rhizobium leguminosarum controls a network of quorum-sensing loci. Mol. Microbiol. 37:81-97. [DOI] [PubMed] [Google Scholar]
- 27.Lyczak, J. B., C. L. Cannon, and G. B. Pier. 2000. Establishment of Pseudomonas aeruginosa infection: lessons from a versatile opportunist. Microb. Infect. 2:1051-1060. [DOI] [PubMed] [Google Scholar]
- 28.McClean, K. H., M. K. Winson, L. Fish, A. Taylor, S. R. Chhabra, M. Camara, M. Daykin, J. H. Lamb, S. Swift, B. W. Bycroft, G. S. A. B. Stewart, and P. Williams. 1997. Quorum sensing and Chromobacterium violaceum: exploitation of violacein production and inhibition for the detection of N-acylhomoserine lactones. Microbiology 143:3703-3711. [DOI] [PubMed] [Google Scholar]
- 29.Middleton, B., H. C. Rodgers, M. Camara, A. J. Knox, P. Williams, and A. Hardman. 2002. Direct detection of N-acylhomoserine lactones in cystic fibrosis sputum. FEMS Microbiol. Lett. 207:1-7. [DOI] [PubMed] [Google Scholar]
- 30.Moré, M. I., L. D. Finger, J. L. Stryker, C. Fuqua, A. Eberhard, and S. C. Winans. 1996. Enzymatic synthesis of a quorum-sensing autoinducer through use of defined substrates. Science 272:1655-1658. [DOI] [PubMed] [Google Scholar]
- 31.Passador, L., J. M. Cook, M. J. Gambello, L. Rust, and B. H. Iglewski. 1993. Expression of Pseudomonas aeruginosa virulence genes requires cell-to-cell communication. Science 260:1127-1130. [DOI] [PubMed] [Google Scholar]
- 32.Pearson, J. P., K. M. Gray, L. Passador, K. D. Tucker, A. Eberhard, B. H. Iglewski, and E. P. Greenberg. 1994. Structure of the autoinducer required for expression of P. aeruginosa virulence genes. Proc. Natl. Acad. Sci. USA 91:197-201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pearson, J. P., C. Van Delden, and B. H. Iglewski. 1999. Active efflux and diffusion are involved in transport of Pseudomonas aeruginosa cell-to-cell signals. J. Bacteriol. 181:1203-1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pearson, J. P., M. Feldman, B. H. Iglewski, and A. Prince. 2000. Pseudomonas aeruginosa cell-to-cell signaling is required for virulence in a model of acute pulmonary infection. Infect. Immun. 68:4331-4334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Perricone, M. A., D. D. Rees, C. R. Sacks, K. A. Smith, J. M. Kaplan and J. A. St. George. 2000. Inhibitory effect of cystic fibrosis sputum on adenovirus-mediated gene transfer in cultured epithelial cells. Hum. Gene Ther. 11:1997-2008. [DOI] [PubMed] [Google Scholar]
- 36.Pesci, E. C., J. P. Pearson, P. C. Seed, and B. H. Iglewski. 1997. Regulation of las and rhl quorum sensing in Pseudomonas aeruginosa. J. Bacteriol. 179:3127-3132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pessi, G., and D. Haas. 2000. Transcriptional control of the hydrogen cyanide biosynthetic genes hcnABC by the anaerobic regulator ANR and the quorum sensing regulators LasR and RhlR in Pseudomonas aeruginosa. J. Bacteriol. 182:6940-6949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rumbaugh, K. P., J. A. Griswold, B. H. Iglewski, and A. N. Hamood. 1999. Contribution of quorum sensing to the virulence of Pseudomonas aeruginosa in burn wound infections. Infect. Immun. 67:5854-5862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schaefer, A. L., D. L. Val, B. L. Hanzelka, J. E. Cronan, Jr., and E. P. Greenberg. 1996. Generation of cell-to-cell signals in quorum sensing: acyl homoserine lactone synthase activity of a purified Vibrio fischeri LuxI protein. Proc. Natl. Acad. Sci. USA 93:9505-9509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Smith, R. S., S. G. Harris, R. Phipps, and B. Iglewski. 2002. The Pseudomonas aeruginosa quorum sensing molecule N-(3-oxododecanoyl)homoserine lactone contributes to virulence and induces inflammation in vivo. J. Bacteriol. 184: 1132-1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Streitwiezer, J., and C. Heathcock. 1985. Introduction to organic chemistry, p. 859-861. MacMillan Press, New York, N.Y.
- 42.Swift, S., K. E. Isherwood, S. Atkinson, P. C. F. Oyston, and G. S. A. B. Stewart. 1999. Quorum sensing in Aeromonas and Yersinia, p. 85-104. In R. England, G. Hobbs, N. Bainton, and D. M. Roberts (ed.), Microbial signalling and communication. Society for General Microbiology symposium 57. Cambridge University Press, Cambridge, United Kingdom.
- 43.Swift, S., J. A. Downie, N. Whithead, A. M. L. Barnard, G. P. C. Salmond, and P. Williams. 2001. Quorum sensing as a population density dependent determinant of bacterial physiology. Adv. Microb. Physiol. 45:199-270. [DOI] [PubMed] [Google Scholar]
- 44.Telford, G., D. Wheeler, P. Williams, P. T. Tomkins, P. Appleby, H. Sewell, G. S. A. B. Stewart, B. W. Bycroft, and D. I. Pritchard. 1998. The Pseudomonas aeruginosa quorum sensing signal molecule, N-(3-oxododecanoyl)-l-homoserine lactone has immunomodulatory activity. Infect. Immun. 66:36-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Throup, J. P., M. Camara, G. Briggs, M. K. Winson, B. W. Bycroft, P. Williams, and G. S. A. B. Stewart. 1995. Characterisation of the yenI/yenR locus from Yersinia enterocolitica mediating the synthesis of two quorum sensing signal molecules. Mol. Microbiol. 17:345-356. [DOI] [PubMed] [Google Scholar]
- 46.Whiteley, M., K. M. Lee, and E. P. Greenberg. 1999. Identification of genes controlled by quorum sensing in Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 96:13904-13909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Williams, P., M. Camara, A. Hardman, S. Swift, D. Milton, V. J. Hope, K. Winzer, B. Middleton, D. I. Pritchard, and B. W. Bycroft. 2000. Quorum sensing and the population-dependent control of virulence. Phil. Trans. R. Soc. B 355:667-680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Winson, M. K., M. Camara, A. Latifi, M. Foglino, S. R. Chhabra, M. Daykin, M. Bally, V. Chapon, G. P. C. Salmond, B. W. Bycroft, A. Lazdunski, G. S. A. B. Stewart, and P. Williams. 1995. Multiple N-acyl-l-homoserine lactone signal molecules regulate production of virulence determinants and secondary metabolites in Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 92:9427-9431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Winson, M. K., S. Swift, L. Fish, J. P. Throup, F. Jorgensen, S. R. Chhabra, B. W. Bycroft, P. Williams, and G. S. A. B. Stewart. 1998. Construction and analysis of luxCDABE-based plasmid sensors for investigating N-acylhomoserine lactone-mediated quorum sensing. FEMS Microbiol. Lett. 163:185-192. [DOI] [PubMed] [Google Scholar]
- 50.Winzer, K., C. Falconer, N. C. Garber, S. P. Diggle, M. Cámara, and P. Williams. 2000. The Pseudomonas aeruginosa lectins PA-IL and PA-IIL are controlled by quorum sensing and by RpoS. J. Bacteriol. 182:6401-6411. [DOI] [PMC free article] [PubMed] [Google Scholar]