Abstract
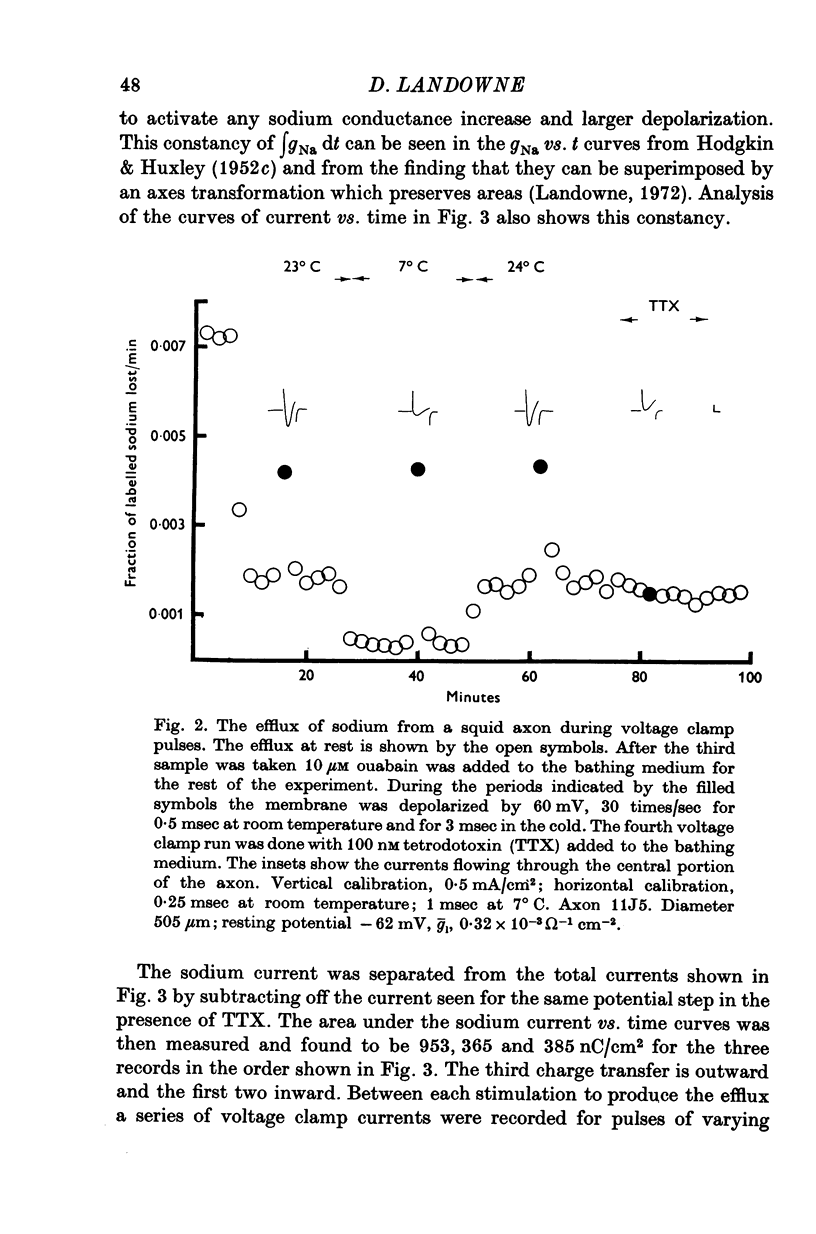
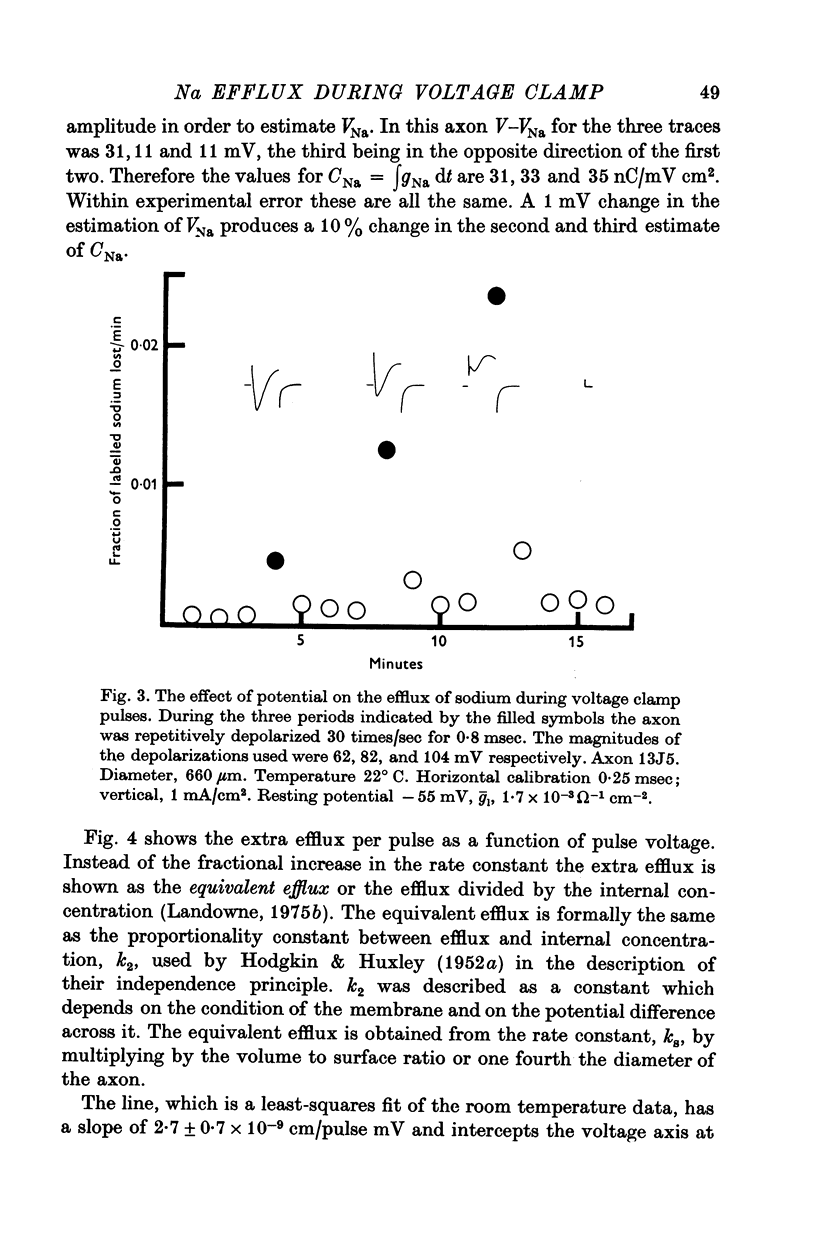
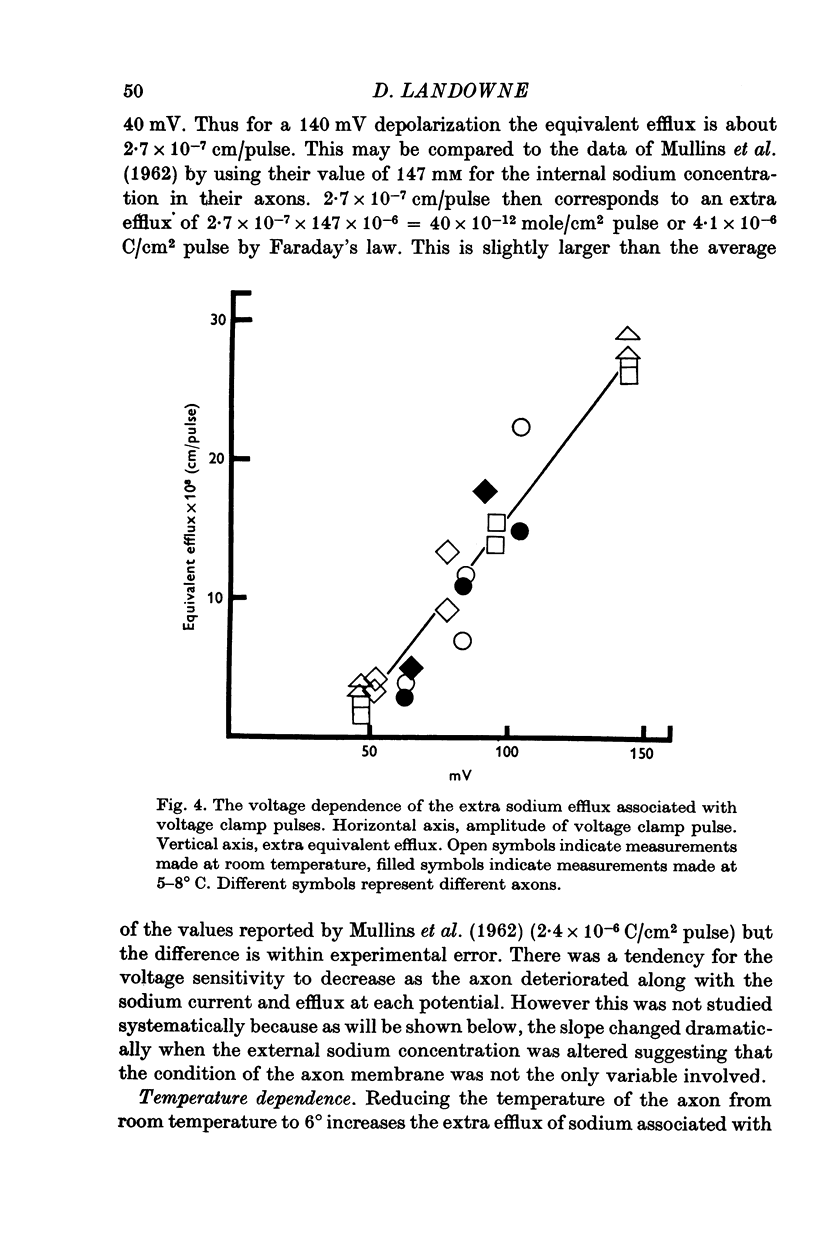
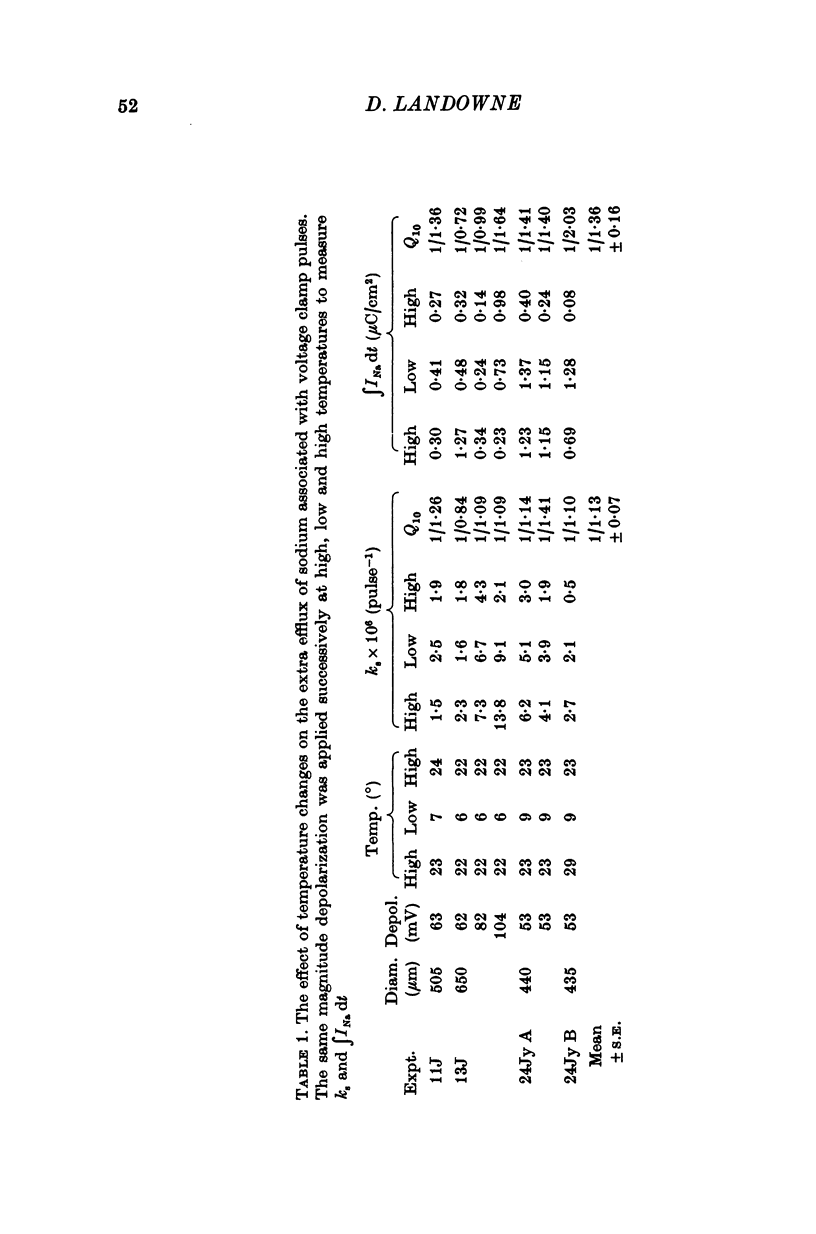
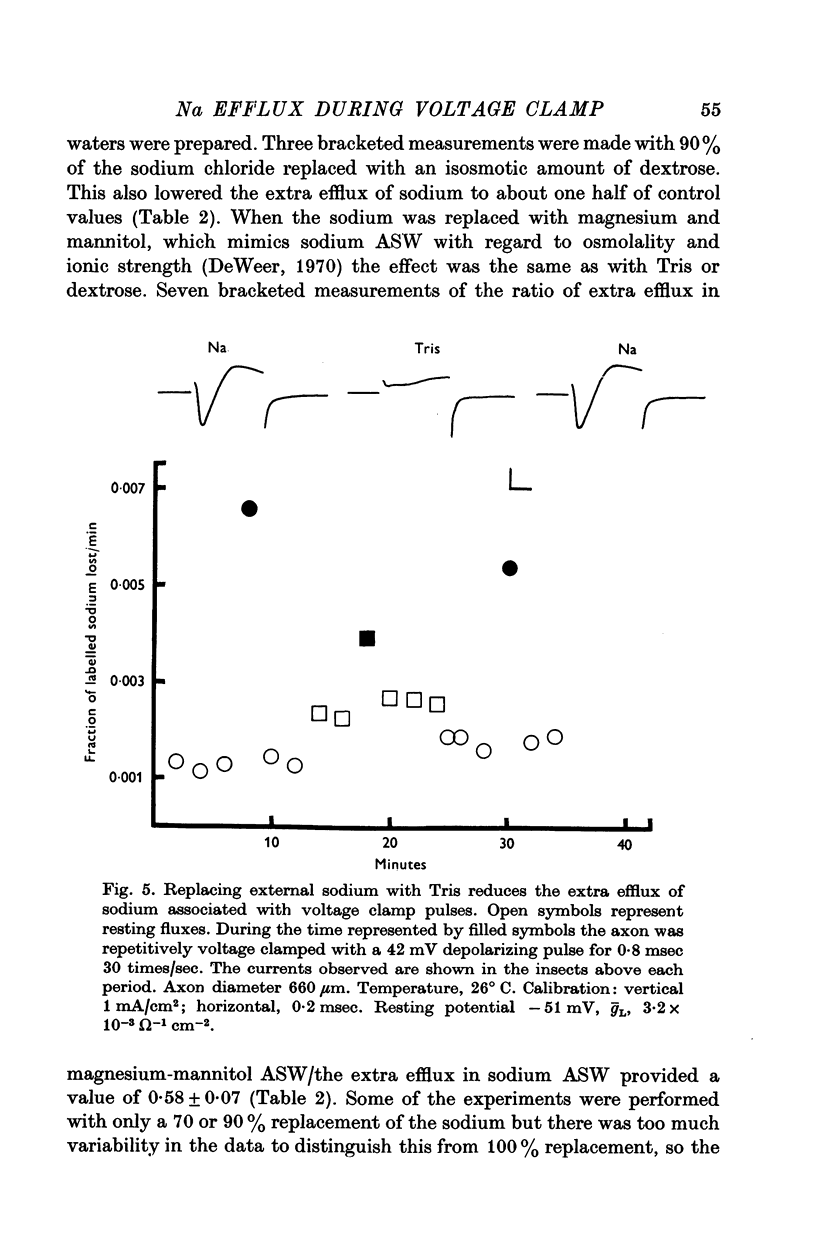
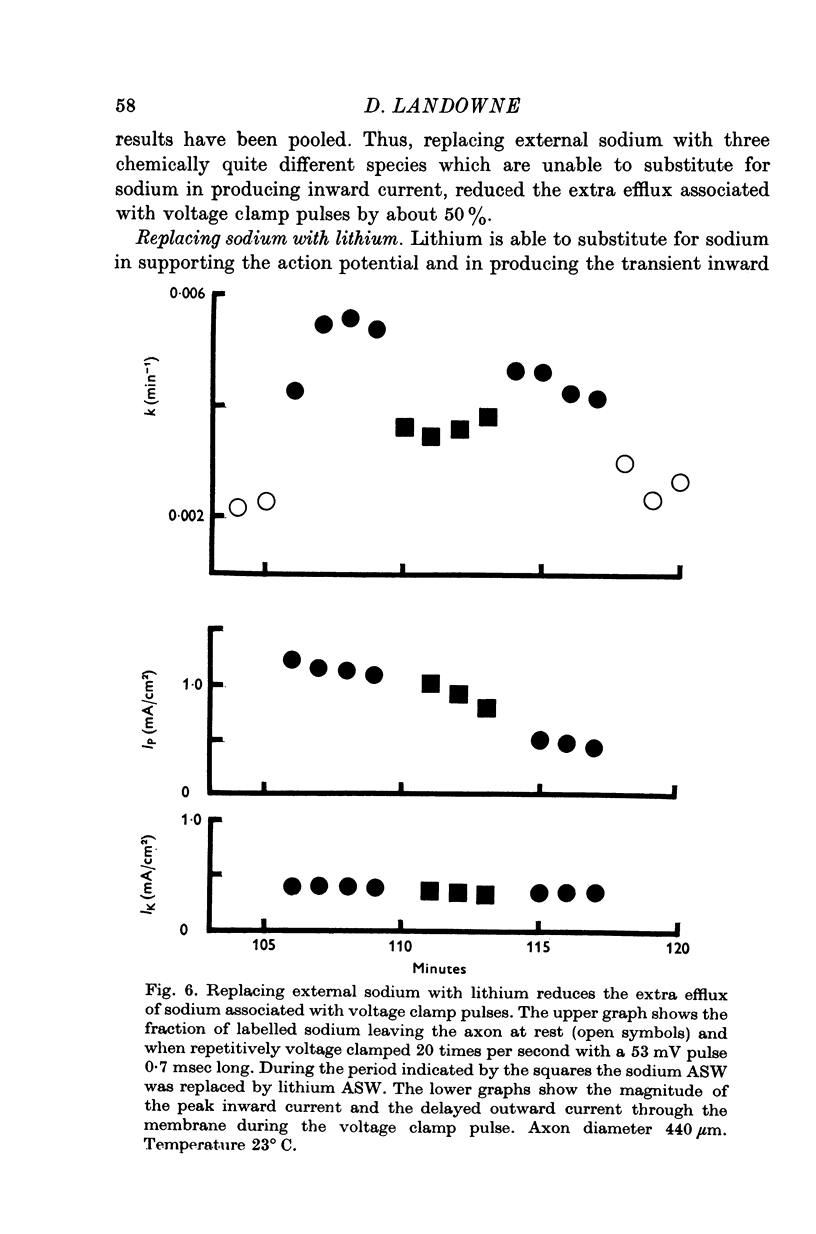
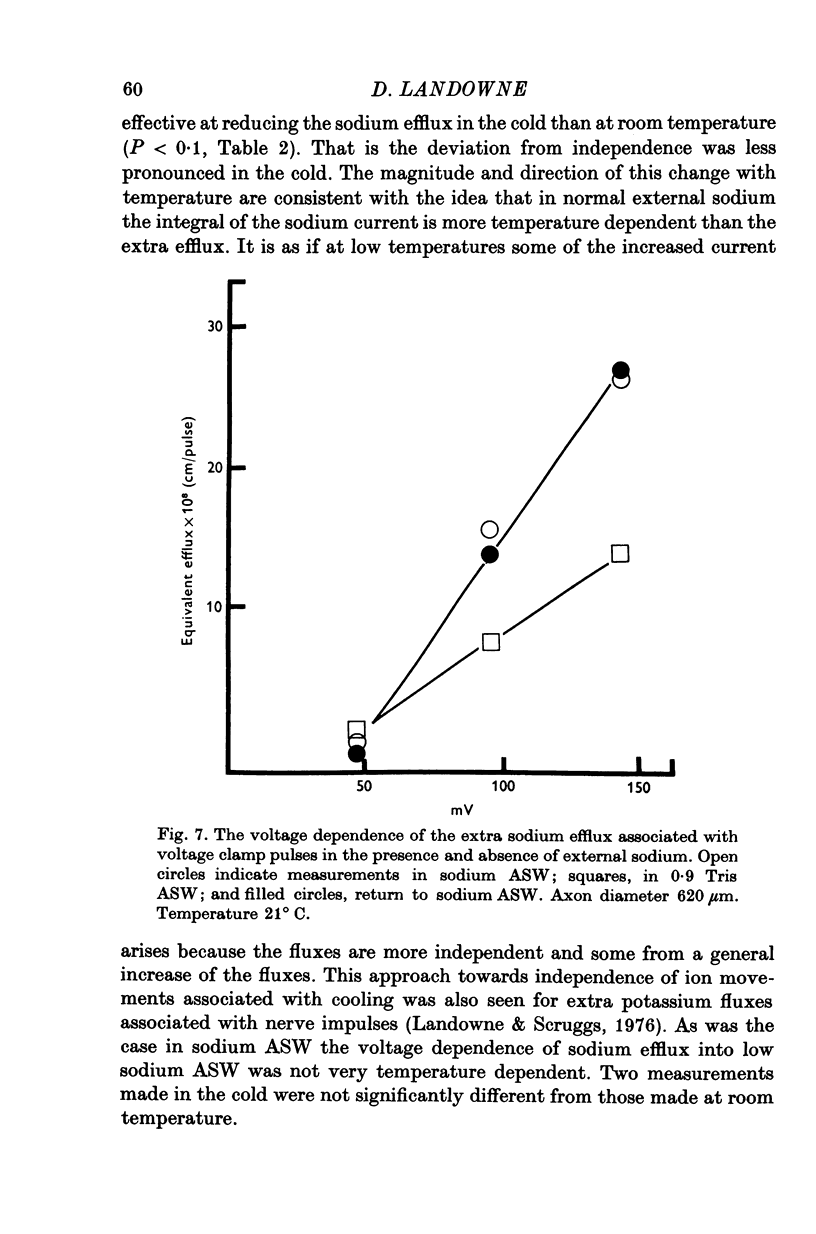
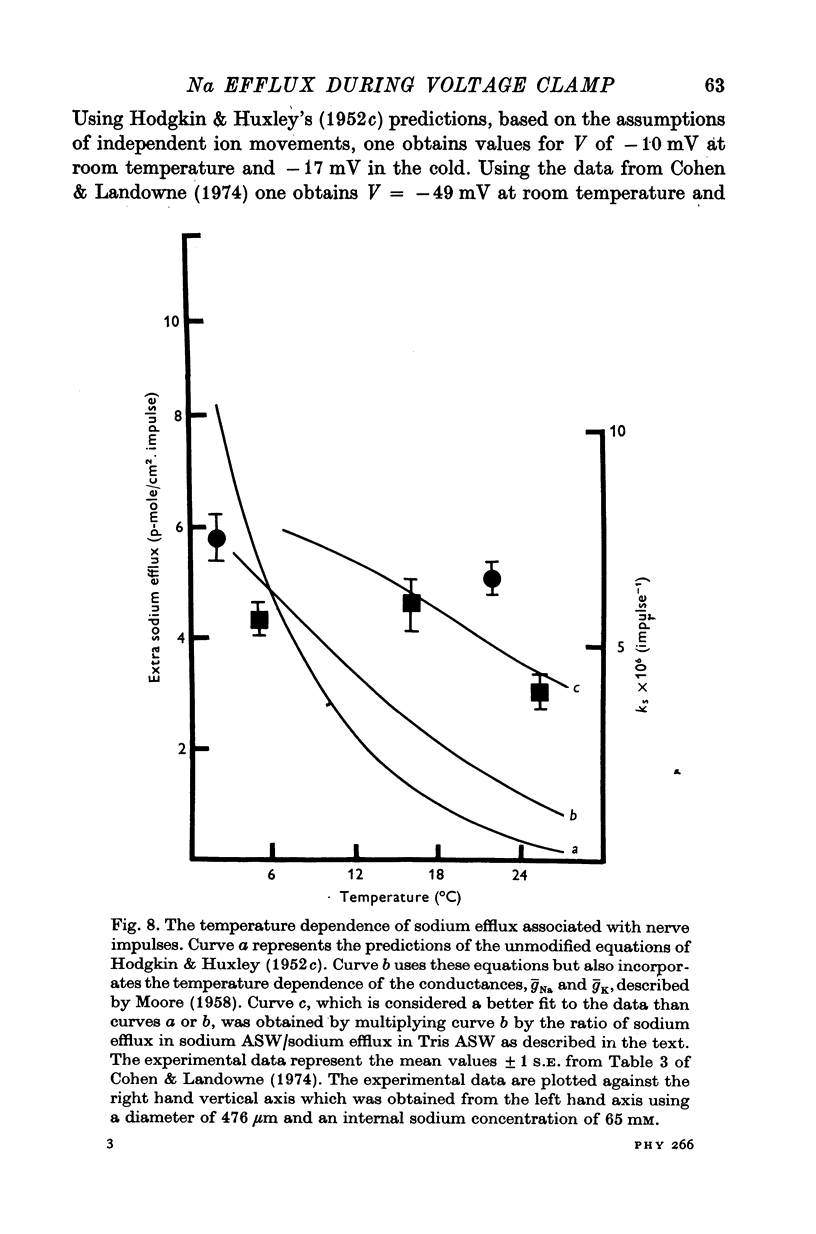
1. The efflux of radioactive sodium was measured from squid axons during simultaneous voltage clamp experiments such that it was possible to determine the efflux of sodium associated with a measured voltage clamp current. 2. The extra efflux of sodium associated with voltage clamp pulses increased linearly with the magnitude of the depolarization above 40 mV. A 100 mV pulse of sufficient duration to produce all of the sodium current increased the rate constant of efflux by about 10(-6). 3. Application of 100 nM tetrodotoxin eliminated the sodium current and the extra efflux of radioactive sodium. 4. Cooling the axon increased the extra efflux/voltage clamp pulse slightly with a Q10 of 1/1-1. On the same axons cooling increased the integral of the sodium current with a Q10 of 1/1-4. 5. Replacing external sodium with Tris, dextrose or Mg-mannitol reduced the extra efflux of sodium by about 50%. The inward sodium current was replaced with an outward current as expected. 6. Replacing external sodium with lithium also reduced the extra efflux by about 50% but the currents seen in lithium were slightly larger than those in sodium. 7. The effect of replacing external sodium was not voltage dependent. Cooling reduced the effect so that there was less reduction of efflux on switching to Tris ASW in the cold than in the warm. 8. The extra efflux of sodium into sodium-free ASW is approximately the same as the integral of the sodium current. Adding external sodium produces a deviation from the independence principle such that there is more exchange of sodium than predicted. Such a deviation from prediction was noted by Hodgkin & Huxley (1952c). 9. Using the equations of Hodgkin & Huxley (1952c) modified to include the deviation from independence reported in this paper and its temperature dependence, one can predict the temperature dependence of the sodium efflux associated with action potentials and obtain much better agreement than is possibly without these phenomena. 10. This deviation from independence in the sodium fluxes is the type expected from some kind of mixing and binding of sodium within the membrane phase.
Full text
PDF

















Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armstrong C. M., Bezanilla F. Charge movement associated with the opening and closing of the activation gates of the Na channels. J Gen Physiol. 1974 May;63(5):533–552. doi: 10.1085/jgp.63.5.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atwater I., Bezanilla F., Rojas E. Time course of the sodium permeability change during a single membrane action potential. J Physiol. 1970 Dec;211(3):753–765. doi: 10.1113/jphysiol.1970.sp009302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker P. F., Blaustein M. P., Hodgkin A. L., Steinhardt R. A. The influence of calcium on sodium efflux in squid axons. J Physiol. 1969 Feb;200(2):431–458. doi: 10.1113/jphysiol.1969.sp008702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezanilla F., Rojas E., Taylor R. E. Time course of the sodium influx in squid giant axon during a single voltage clamp pulse. J Physiol. 1970 Mar;207(1):151–164. doi: 10.1113/jphysiol.1970.sp009054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandler W. K., Meves H. Voltage clamp experiments on internally perfused giant axons. J Physiol. 1965 Oct;180(4):788–820. doi: 10.1113/jphysiol.1965.sp007732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen L. B., Landowne D. The temperature dependence of the movement of sodium ions associated with nerve impulses. J Physiol. 1974 Jan;236(1):95–111. doi: 10.1113/jphysiol.1974.sp010424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Weer P. Effects of intracellular adenosine-5'-diphosphate and orthophosphate on the sensitivity of sodium efflux from squid axon to external sodium and potassium. J Gen Physiol. 1970 Nov;56(5):583–620. doi: 10.1085/jgp.56.5.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952 Aug;117(4):500–544. doi: 10.1113/jphysiol.1952.sp004764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. Currents carried by sodium and potassium ions through the membrane of the giant axon of Loligo. J Physiol. 1952 Apr;116(4):449–472. doi: 10.1113/jphysiol.1952.sp004717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F., KATZ B. Measurement of current-voltage relations in the membrane of the giant axon of Loligo. J Physiol. 1952 Apr;116(4):424–448. doi: 10.1113/jphysiol.1952.sp004716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., HUXLEY A. F. The dual effect of membrane potential on sodium conductance in the giant axon of Loligo. J Physiol. 1952 Apr;116(4):497–506. doi: 10.1113/jphysiol.1952.sp004719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN A. L., KEYNES R. D. The potassium permeability of a giant nerve fibre. J Physiol. 1955 Apr 28;128(1):61–88. doi: 10.1113/jphysiol.1955.sp005291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hille B. Ionic selectivity, saturation, and block in sodium channels. A four-barrier model. J Gen Physiol. 1975 Nov;66(5):535–560. doi: 10.1085/jgp.66.5.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hille B. The permeability of the sodium channel to metal cations in myelinated nerve. J Gen Physiol. 1972 Jun;59(6):637–658. doi: 10.1085/jgp.59.6.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KEYNES R. D., SWAN R. C. The effect of external sodium concentration on the sodium fluxes in frog skeletal muscle. J Physiol. 1959 Oct;147:591–625. doi: 10.1113/jphysiol.1959.sp006264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KEYNES R. D. The ionic movements during nervous activity. J Physiol. 1951 Jun;114(1-2):119–150. doi: 10.1113/jphysiol.1951.sp004608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keynes R. D., Rojas E. Kinetics and steady-state properties of the charged system controlling sodium conductance in the squid giant axon. J Physiol. 1974 Jun;239(2):393–434. doi: 10.1113/jphysiol.1974.sp010575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landowne D. A comparison of radioactive thallium and potassium fluxes in the giant axon of the squid. J Physiol. 1975 Oct;252(1):79–96. doi: 10.1113/jphysiol.1975.sp011135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landowne D. A new explanation of the ionic currents which flow during the nerve impulse. J Physiol. 1972 Apr;222(1):46P–47P. [PubMed] [Google Scholar]
- Landowne D., Scruggs V. The temperature dependence of the movement of potassium and chloride ions associated with nerve impulses. J Physiol. 1976 Jul;259(1):145–158. doi: 10.1113/jphysiol.1976.sp011458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MULLINS L. J., ADELMAN W. J., Jr, SJODIN R. A. Sodium and potassium ion effluxes from squid axons under voltage clamp conditions. Biophys J. 1962 May;2:257–274. doi: 10.1016/s0006-3495(62)86854-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rojas E., Canessa-Fischer M. Sodium movements in perfused squid giant axons. Passive fluxes. J Gen Physiol. 1968 Aug;52(2):240–257. doi: 10.1085/jgp.52.2.240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- USSING H. H. Transport of ions across cellular membranes. Physiol Rev. 1949 Apr;29(2):127–155. doi: 10.1152/physrev.1949.29.2.127. [DOI] [PubMed] [Google Scholar]



