Abstract
Human T-cell leukemia virus type 1 (HTLV-1) is an oncogenic retrovirus associated primarily with adult T-cell leukemia and neurological disease. HTLV-1 encodes the positive trans-regulatory proteins Tax and Rex, both of which are essential for viral replication. Tax activates transcription initiation from the viral long terminal repeat and modulates the transcription or activity of a number of cellular genes. Rex regulates gene expression posttranscriptionally by facilitating the cytoplasmic expression of incompletely spliced viral mRNAs. Tax and Rex mutants have been identified that have defective activities or impaired biochemical properties associated with their function. To ultimately determine the contribution of specific protein activities on viral replication and cellular transformation of primary T cells, mutants need to be characterized in the context of an infectious molecular clone. Since the tax and rex genes are in partially overlapping reading frames, mutation in one gene frequently disrupts the other, confounding interpretation of mutational analyses in the context of the virus. Here we generated and characterized a unique proviral clone (H1IT) in which the tax and rex genes were separated by expressing Tax from an internal ribosome entry site. We showed that H1IT expresses both functional Tax and Rex. In short- and long-term coculture assays, H1IT was competent to infect and immortalize primary human T cells similar to wild-type HTLV-1. In contrast, H1IT failed to efficiently replicate and persist in inoculated rabbits, thus emphasizing the importance of temporal and quantitative regulation of specific mRNA for viral survival in vivo.
Human T-cell leukemia virus type 1 (HTLV-1) is a complex retrovirus that is associated with adult T-cell leukemia as well as a neurological disorder termed HTLV-associated myelopathy/tropical spastic paraparesis (16, 17, 34, 54). The genome organization of HTLV-1 resembles that of other complex retroviruses that contain overlapping regulatory and accessory genes in addition to structural and enzymatic genes (9, 12, 18). Tax and Rex are two trans-acting regulatory gene products that are essential for viral replication (9, 46). Upon HTLV-1 infection of a susceptible host cell, the randomly integrated provirus is expressed at low levels. Tax, being the major transcriptional activator of the viral long terminal repeat (LTR), mediates high-level viral gene expression necessary for efficient viral protein production. Tax-mediated activation of HTLV-1 LTR is dependent on three imperfect 21-bp repeats, termed the Tax response elements (6, 7, 14). Tax also has been shown to disrupt cellular gene expression by recruiting or interacting with cellular transcription coactivators, by indirectly activating the NF-κB activation pathway, or by modulating the activity of cellular proteins (27, 32, 42, 58). Many of the cellular genes modulated by Tax are involved in growth, differentiation, apoptosis, or cell cycle control (2, 5, 44, 52, 53). Thus, a strong body of evidence suggests that the effects of Tax on cellular processes are required for the transforming or oncogenic capacity of HTLV (13, 21, 48, 49). Indeed, mutational analysis in the context of a replicating virus directly demonstrated that Tax of both HTLV-1 and HTLV-2 is essential for virus-mediated cellular transformation of primary human T cells and that Tax activation of NF-κB and CREB/ATF signaling plays a key role in the malignant process (45, 48, 49).
Rex is a posttranscriptional regulator that is essential for efficient cytoplasmic expression of incompletely spliced viral mRNA (59). The HTLV structural and enzymatic proteins are expressed from intron-containing mRNAs that would be targeted for splicing or degradation in the nucleus in the absence of Rex (20, 29, 31). Rex binds to its viral RNA responsive element (RxRE), stabilizes the mRNA, and facilitates its export from the nucleus. Based on the critical role of Rex in the expression of structural and enzymatic gene products, it is considered to positively regulate the switch between the early, latent phase and the late, productive phase of the HTLV life cycle (59). Although Rex is not absolutely required for cellular transformation in vitro, it is essential for efficient viral infectivity, spread, and survival in vivo (57).
Extensive mutational analyses of both Tax and Rex have mapped specific activities and biochemical properties to distinct regions or domains of the protein (56, 59). However, the majority of these mutations, particularly those that maintain replication competence, have been studied in cDNA expression systems in nonlymphoid cells, and their overall contribution to the virus life cycle or HTLV-mediated transformation has yet to be tested. Since Tax and Rex are encoded on the same viral mRNA in partially overlapping reading frames, introduction of specific mutations in one gene in many cases disrupts the integrity of the other. Therefore, studying such mutants precludes a clear interpretation of structure and function analyses in the context of the virus.
The goal of this study was to generate and characterize an infectious proviral clone in which the tax and rex genes were separated by expressing Tax from an internal ribosome entry site (IRES) (37). In this uniquely modified provirus, HTLV-1-IRES-Tax (H1IT), Rex is expressed from the natural doubly spliced mRNA, and Tax is initiated 3′ of Rex as a result of the inserted IRES. Since the IRES-Tax was introduced at the 3′ end of the provirus, Tax now has the potential to be expressed from all viral messages. Transient-transfection studies in 293T cells indicated that H1IT expresses both functional Tax and Rex and is able to produce p19 Gag. Furthermore, we showed that 729-H1IT stable transfectants produced replication-competent virus with the capacity to immortalize primary human T cells. Interestingly, H1IT was not able to persist in a rabbit model of infection, consistent with the conclusion that temporal and quantitative regulation of specific mRNAs is key to viral survival in vivo. This new reagent will be particularly valuable for future studies designed to dissect the contribution of specific activities of either Tax or Rex on HTLV-1 pathobiology, such as viral transformation of primary T lymphocytes.
MATERIALS AND METHODS
Cells.
293T cells and 729 cells were maintained in Dulbecco's modified Eagle's medium and Iscove's modified Eagle's medium, respectively. The media were supplemented to contain 10% fetal bovine serum, 2 mM glutamine, penicillin (100 U/ml), and streptomycin (100 μg/ml). Human peripheral blood mononuclear cells (PBMCs) were isolated from freshly drawn human blood as previously described (57). Briefly, blood was diluted 1:1 with phosphate-buffered saline and layered over 12 ml of Ficoll-Hypaque (Amersham, Piscataway, NJ) and then centrifuged for 20 min at 900 × g. The Ficoll-plasma interphase (buffy coat layer) was gently removed, transferred to a new tube, and washed once with RPMI. The cell pellets were resuspended in RPMI supplemented with 20% fetal bovine serum, antibiotics, and 10 U/ml of human interleukin-2 (IL-2; Roche, Indianapolis, IN).
Plasmids.
The plasmid containing the wild-type (wt) HTLV-1 proviral clone, pACHneo, was used in this study (4, 57). HTLV-1 F4Term is a Tax knockout proviral clone. It was generated by site-directed mutagenesis (QuikChange; Stratagene, La Jolla, CA) of pACHneo introducing TC-to-AG point mutations (nucleotides 7307 and 7308) that resulted in a stop codon at amino acid 4 of Tax while maintaining the Rex reading frame. HTLV-1 F4Term was used as a template to generate the H1IT proviral clone. An EcoR1 restriction enzyme site was introduced in sequences just downstream of the Rex stop codon and was used to replace the remaining 3′ HTLV-1 proviral sequences with the encephalomyocarditis virus IRES, the complete tax gene, and the 3′ LTR (Fig. 1). The full-length H1IT construct was confirmed by restriction enzyme diagnostics and sequencing. The LTR-1-Luc Tax reporter plasmid has been described previously (60). The human immunodeficiency virus type 1 (HIV-1) Tat expression vector, pctat, contains HIV-1 tat cDNA cloned downstream of the cytomegalovirus (CMV) promoter. The pCgagRxRE-I reporter contains the HIV-1 LTR promoter and gag gene linked to a fragment of HTLV-1, spanning the RxRE (R-U5 region of the LTR) (10). CMV-βgal or CMV-Luc plasmids were used to control for transfection efficiency in each experiment.
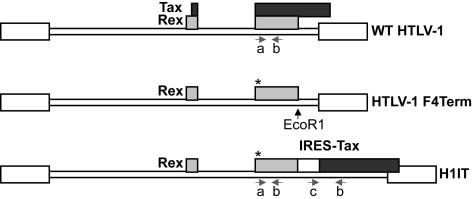
FIG. 1.
Schematic diagram of the generation of the H1IT proviral clone. The wt HTLV-1 was expressed from pACHneo. Only Tax and Rex ORFs are represented here, in black and gray, respectively. HTLV-1 F4Term is a Tax knockout proviral clone that was generated from pACHneo and contains a stop codon at amino acid 4 of Tax (asterisk) while maintaining the Rex reading frame. HTLV-1 F4Term was used as a template to generate H1IT. An EcoR1 restriction enzyme site was introduced in sequences downstream of the Rex stop codon and subsequently was used to replace the remaining 3′ HTLV-1 proviral sequences with an IRES from encephalomyocarditis virus (white box), the complete tax gene, and the 3′ LTR. Arrows indicate primer pairs used for diagnostic PCR. Primer pair a and b amplifies sequences in both wt HTLV-1 and H1IT, whereas c and d amplify sequences from H1IT only.
Transfection, luciferase reporter assay, and Gag ELISA.
To measure Tax CREB/ATF activating function (viral LTR), 2 × 105 293T cells were transfected using Lipofectamine (Invitrogen, Carlsbad, CA) according to the manufacturer's recommendation. The total amount of DNA was kept constant and was composed of 0.2 μg LTR-1-Luc, 0.1 μg CMV-βgal, and 1 μg wt HTLV-1 or H1IT proviral clone. After 48 h of growth, cells were pelleted and 450 μl of cell supernatants was used for p19 Gag enzyme-linked immunosorbent assay (ELISA) according to manufacturer's recommendations (Zeptometrix, Buffalo, NY). The cell pellets were lysed in passive lysis buffer (Promega, Madison, WI), and Tax activity was measured as luciferase light units. All experiments were performed independently three times in triplicate, and results were normalized for transfection efficiency using β-galactosidase. The Rex functional assay was performed as described elsewhere (38). Briefly, 0.4 μg of control plasmid, HTLV-1, or H1IT proviral clone was cotransfected with 0.1 μg CMV-Luc, 0.1 μg pctat, and 0.3 μg of pCgagRxRE-I reporter. Cell lysates were prepared in passive lysis buffer 48 h posttransfection, and luciferase activity was determined to control for transfection efficiency. HIV-1 p24 Gag levels in cell lysate were measured using p24 Gag ELISA (Beckman-Coulter, Fullerton, CA).
Generation and characterization of a stable 729-H1IT cell line.
To generate stable transfectants, proviral plasmid clones containing the Neor gene were introduced into cells by electroporation as described previously (8, 19). Stable transfectants were isolated following incubation in 24-well culture plates in medium containing 1 mg/ml of Geneticin. After 4 to 5 weeks of selection, viable cells were single cell cloned, expanded, and maintained in culture for further analysis. The clones were screened for p19 Gag expression in the cell supernatant using an ELISA. 729ACHneo, a previously generated and characterized stable wt HTLV-1 producer cell line (4), was used as a positive control.
DNA preparation, PCR, and Western blotting.
Genomic DNA was isolated from permanently transfected cell clones or from immortalized PBMCs using the PUREGENE DNA purification system (Gentra, Minneapolis, MN). A primer set that amplifies a 270-bp fragment from both HTLV-1 and H1IT is 670, 7335CGGATACCCAGTCTACGTGT7354, and ACH3AS, 7605GGGTGGAATGTTGGGGG7589, whereas the primer set LTax, 5′CGATGATAATATGGCCCACTTCCCAGGGTTTG, and ACH3AS amplifies a 318-bp fragment from H1IT only. PCR products were run on an agarose gel and visualized by ethidium bromide staining.
To detect viral proteins, 729 stable transfectants were lysed with modified RIPA buffer (50 mM Tris-Cl [pH 8.0], 150 mM NaCl, 1% Nonidet P-40, 0.5% desoxycholate, 0.1% sodium dodecyl sulfate [SDS], 2.0 mM phenylmethanesulfonyl fluoride, 20 μg/ml aprotinin, 1.0 mM Na3VO4, and 1 mM NaF) on ice for 30 min. After centrifugation, the cell lysates were subjected to 4-to-12% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose (Amersham, Piscataway, NJ). Rabbit anti-Tax-1, anti-HBZ, or anti-HTLV-1 patient serum was used to detect different viral proteins. Viral proteins were visualized using the ECL Western blotting analysis system (Santa Cruz Biotechnology, Santa Cruz, CA).
Short-term coculture microtiter proliferation and long-term immortalization assays.
Short-term microtiter proliferation assays were performed as described previously with slight modifications (43). Briefly, freshly isolated human PBMCs were prestimulated with 2 μg/ml phytohemagglutinin and 10U/ml IL-2 for 2 days. 729 HTLV producer cells (200 cells) were gamma irradiated with 10,000 rads and cocultured with 104 prestimulated PBMCs in the presence of IL-2 in 96-well round-bottom plates. Wells were enumerated for growth and split 1:4 at weekly intervals. This procedure ensured that only dividing cells growing fast enough to show visible pellets at the bottom of the wells were maintained, whereas in the wells with nondividing cells or limited proliferation, pellets became smaller each week and usually disappeared by 3 to 4 weeks. Each week, eight random wells were selected for quantitative measurement of cell proliferation and apoptosis using the MTS (Promega, Madison WI) and cell death detection ELISA (Roche, Indianapolis, IN), respectively, as recommended by the manufacturers.
For the long-term immortalization assays, 106 irradiated 729 producer cells were cocultivated with 2 × 106 freshly isolated PBMCs with 10 U/ml IL-2 in 24-well culture plates (19). The presence of HTLV expression was confirmed by detection of p19 Gag protein in the culture supernatant using an ELISA at weekly intervals. Viable cells were counted weekly by trypan blue exclusion. Cells inoculated with wt HTLV-1 or H1IT that continued to produce p19 Gag antigen and proliferate 8 weeks postcoculture in the presence of exogenous IL-2 were identified as HTLV immortalized.
Rabbit inoculation procedures. Twelve-week-old specific-pathogen-free New Zealand White rabbits (Hazelton, Kalamazoo, MI) were inoculated via the lateral ear vein with approximately 1 × 107 gamma-irradiated (7,500 rad) 729-HTLV-1 (six rabbits), 729-H1IT (two rabbits), or 729 uninfected control cells (two rabbits). Cell inocula were equilibrated based on their p19 Gag production. At weeks 0, 2, 4, 6, and 8 after inoculation, 10 ml of blood was drawn from the central ear artery of each animal. Serum reactivity to specific viral antigenic determinants was detected using a commercial HTLV-1 Western blot assay (Zeptometrix, Buffalo, NY) adapted for rabbit plasma by use of avidin-conjugated goat anti-rabbit immunoglobulin G (1:200 dilution; Sigma, St. Louis, MO) (11). Serum showing reactivity to Gag (p24 or p19) and Env (gp21 or gp46) antigens was classified as positive for HTLV-1 seroreactivity. To detect integrated proviruses, genomic DNA was harvested using the PUREGENE DNA purification system (Gentra Systems, Minneapolis, MN), and 0.5 μg of DNA was subjected to 40-cycle PCR using primer pair 670 and 671 to amplify a 159-bp fragment specific for the HTLV-1 tax/rex region (31), and rabbit glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was detected using primer pair rGAPDH-S, 5′GATGCTGGTGCCGAGTACGTGG, and rGAPDH-AS, 5′GTGGTGCAGGATGCGTTGCTGA. PCR products were run on an agarose gel and visualized by ethidium bromide staining.
RESULTS
Construction and characterization of an HTLV-1 proviral clone with nonoverlapping Tax and Rex sequences.
In order to separate Tax and Rex open reading frames (ORFs) in an HTLV-1 proviral clone, we designed a strategy that utilizes an IRES from the encephalomyocarditis virus. The insertion of the IRES between two ORFs in a bicistronic mRNA allows the translation of the downstream ORF in a cap-independent manner. The strategy for the generation of an HTLV-1 proviral clone with nonoverlapping Tax and Rex (H1IT) is highlighted in Fig. 1. Although the addition of the IRES would result in a severely truncated (173-amino-acid) nonfunctional Tax (51), the original Tax ORF was truncated further by mutating the fourth amino acid codon of Tax to introduce a termination codon. In this construct (HTLV-1 F4Term), the proviral clone is completely deficient for Tax activity while Rex is still functional (data not shown). It should be noted that the F4Term mutation introduces a single amino acid substitution of proline to alanine in the p30/p13 reading frame, which did not disrupt the repressive function of p30 on tax/rex mRNA (41, 60) (data not shown). Using an EcoR1 site that was introduced immediately downstream of the Rex stop codon, a cassette that contains IRES-Tax cDNA in addition to the rest of the 3′-LTR was introduced. The new proviral clone termed H1IT is ∼1 kb larger than wt HTLV-1.
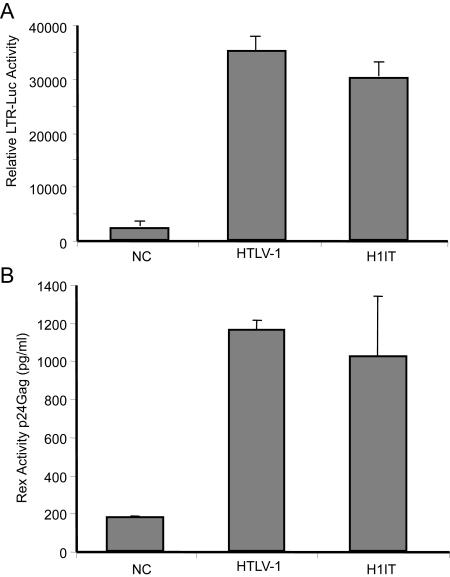
Since efficient replication and virion production are highly dependent on functional Tax and Rex, we tested the activities of these two proteins expressed by H1IT compared to the wt parental clone. Cotransfection of 293T cells with the HTLV-1 or H1IT proviral clone and the Tax reporter LTR-1-Luc resulted in a 20- to 30-fold increase in Tax-dependent gene expression from both clones (Fig. 2A). Similarly, when these clones were cotransfected with a Rex reporter that expresses HIV-1 p24 Gag under the control of the Rex response element, there was approximately a sixfold induction in Rex functional activity (Fig. 2B). Our data indicate that Tax or Rex activity expressed from H1IT was not significantly different from wt HTLV-1. We were surprised at this result, since it has been reported that IRES-dependent expression is less efficient than cap-dependent expression (36). We speculate that the recovery of full Tax activity and, indirectly, Rex activity in H1IT is due to the fact that in H1IT, the primary viral transcriptional regulator, Tax, is potentially expressed from all viral transcripts, compared to a single doubly spliced transcript in wt HTLV-1.
FIG. 2.
Functional activities of Tax and Rex expressed from H1IT. (A) Transiently transfected 293T cells were assayed for Tax activity by cotransfection of wt HTLV-1, H1IT, or an empty vector along with the Tax reporter LTR-1-luciferase. CMV-βgal was used to control for transfection efficiency. The data shown is representative of three independent experiments. There was no significant difference in Tax activity (20- to 30-fold induction over background) when it was expressed from either proviral clone. (B) Transiently transfected 293T cells were assayed for Rex activity by cotransfection of wt HTLV-1, H1IT, or an empty vector along with pcTat and pCgag-RxRE reporter. CMV-Luc was used to control for transfection efficiency, and data shown are representative of three independent experiments. Rex activity measured as a function of p24 Gag production using ELISA was not significantly different between the two proviral clones.
Generation and characterization of H1IT viral producer cell lines.
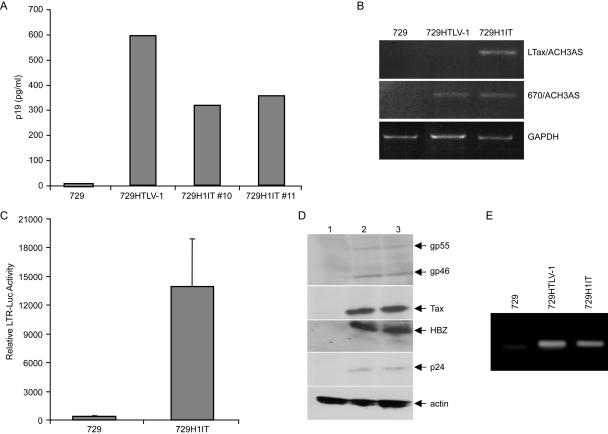
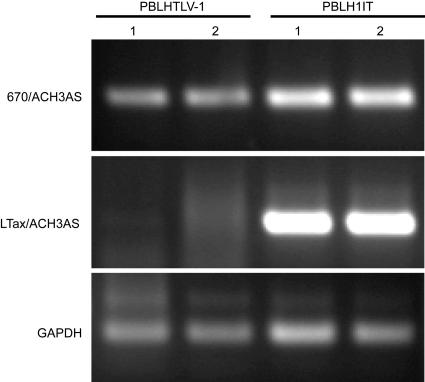
To assess the ability of the H1IT proviral clone to produce viral proteins, direct viral replication, and induce cellular immortalization, stable H1IT producer cells were generated. 729 B cells were electroporated with the H1ITneo plasmid, and 3 to 4 weeks after transfection, wells containing visible clumps of Geneticin-resistant cells were single cell cloned and further characterized. Several cell clones were screened using ELISA for the production of p19 Gag released into culture supernatants. At least two cell clones showed robust p19 Gag production (Fig. 3A), and the H1IT clone 11 was used for the rest of this study. The integration of intact proviral sequences was determined using diagnostic PCR analysis of genomic DNA isolated from the stable producer cells. Using a primer that is specific for the IRES-Tax junction (Fig. 1), it was possible to differentiate between wt HTLV-1 and H1IT proviruses. As shown in Fig. 3B, only 729-H1IT cells showed a product when using the LTax/ACH3AS primer pair, whereas a PCR product was visible in both 729-H1IT and 729-HTLV-1 cells when using the 670/ACH3AS primer pair. Further characterization was conducted to confirm that the IRES is still functional in the stable producer cells. Since Tax translation in this context is tightly dependent on a functional IRES, we confirmed the expression of a functional Tax in 729-H1IT following transfection of the Tax reporter LTR-1-Luc (Fig. 3C).
FIG. 3.
Characterization of a 729H1IT stable producer cell line. (A) Supernatants from individual cell clones were tested for the expression of p19 Gag by ELISA. At least two cell clones showed robust p19 Gag production, and the H1IT clone 11 was used for the rest of this study. (B) The stable integration of intact proviral sequences into 729 B cells was determined using diagnostic PCR analysis of genomic DNA. 729-H1IT cells showed a product unique to H1IT when the LTax/ACH3AS primer pair was used (see primers c and d in Fig. 1 for location), whereas a PCR product was visible in both 729-H1IT and 729-HTLV-1 cells when the 670/ACH3AS primer pair was used (see primers a and b in Fig. 1). (C) Since Tax expression is tightly dependent on IRES in H1IT, Tax functional activity was measured in order to confirm that the IRES is still functional in the stable producer cells following transfection of the Tax reporter LTR-1-Luc. (D) Viral protein production in 729-H1IT compared to 729-HTLV-1 was determined using Western blot analysis. Total proteins from each cell line were extracted and separated by SDS-PAGE. Using HTLV-1-specific antibodies and HTLV-1 patient antisera, comparable levels of p24 Gag, Tax, gp46 Env, gp55, and HBZ from 729-H1IT as well as 729-HTLV-1, but not 729 control cells, were detected. (E) Reverse transcription-PCR analysis on viral RNA extracted from virions present in supernatants of individual cell clones shows no pronounced reduction of genomic viral RNA, indicating that packaging efficiency of H1IT is not significantly reduced.
We next monitored viral protein production in 729-H1IT compared to 729-HTLV-1. Total proteins from each cell line were extracted and separated by SDS-PAGE. Using HTLV-1-specific antibodies and HTLV-1 patient antisera, we were able to detect comparable levels of p24Gag, Tax, gp46Env, gp55, and HBZ from 729-H1IT as well as 729-HTLV-1, but not 729 control cells (Fig. 3D). Taken together, these results indicate that viral gene expression in producer cells was not significantly disrupted either by the separation of Tax and Rex or by IRES-dependent Tax expression.
Since the H1IT genome is approximately 1 kb larger than HTLV-1, we next tested whether this could have an effect on packaging efficiency of the virus. Cell-free virions were isolated from an equivalent number of 729 producer cells cultured for 3 days. Virion RNA was extracted and subjected to reverse transcription-PCR using a gag/pol-specific primer pair. Our data indicate that the H1IT genome is packaged with an efficiency similar to wild-type HTLV-1 (Fig. 3E).
H1IT immortalizes PBMCs.
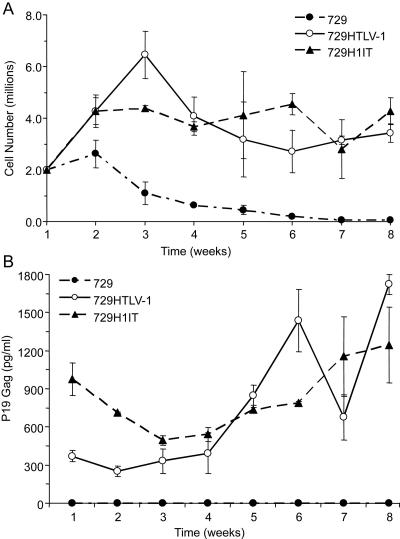
We next assessed the capacity of the H1IT viruses to immortalize human PBMCs. 729, 729-HTLV-1, and 729-H1IT cells were lethally irradiated and cocultured with freshly isolated PBMCs in the presence of 10 U/ml of human IL-2 in 24-well plates. Cell number and viability were monitored at approximately weekly intervals to follow the immortalization process and the characteristic expansion of cells from the PBMC mixed cell population. It is important to note that the irradiated cells die off within 3 weeks of the coculture; hence, detection of HTLV-1 p19 Gag beyond that point would be attributed to the infected and proliferating PBMCs. A growth curve of a representative assay indicated a progressive loss of viable cells over time in cocultures containing irradiated uninfected 729 cells and PBMCs (Fig. 4A). In contrast, PBMCs cocultured with either 729-HTLV-1 or 729-H1IT showed very similar progressive growth patterns consistent with immortalization.
FIG. 4.
H1IT immortalizes human PBMCs. A total of 1 × 106 729, 729-HTLV-1, and 729-H1IT producer cells were lethally irradiated and cocultured with 2 × 106 freshly isolated PBMCs in 24-well plates. (A) To follow the immortalization process, growth curves were determined by monitoring cell number and viability by trypan blue exclusion at weekly intervals. Cells were fed once per week with RPMI 1640 supplemented with 20% FBS and IL-2. The mean and standard deviation of each time point were determined from three independent samples. (B) The presence of HTLV gene expression was confirmed by detection of structural Gag protein in the culture supernatant by p19 Gag ELISA. The mean and standard deviation of each time point were determined from three independent samples.
Since viral p19 Gag production is a good indicator of viral replication, supernatants from the immortalized cells also were monitored at weekly intervals for the release of p19 Gag (Fig. 4B). The first few weeks in coculture reflected residual p19 Gag from the producer cell lines, while starting at week 4 there was a consistent and continuous accumulation of p19 Gag in the culture supernatant, indicating viral replication and virion production. Our data indicated that H1IT, like wt HTLV-1, had the capacity to productively infect PBMCs and induced sustained proliferation or immortalization of T lymphocytes in vitro.
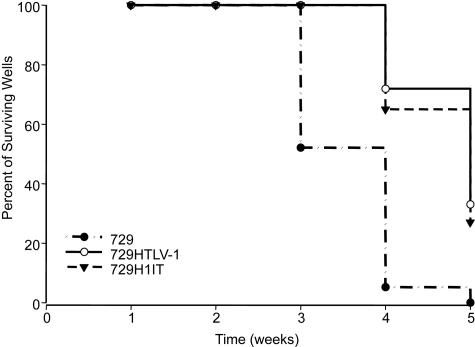
In order to get a more quantitative measure of the ability of the H1IT virus to infect and transform PBMCs, a short-term limiting dilution infectivity assay was employed. In this assay, a fixed number of PBMCs is cocultured with different dilutions of virus producing cells in a 96-well plate. Each week, the cells are enumerated, resuspended, and split at a ratio of 1:4. Since three-fourths of the cells in each well are discarded, only wells containing actively dividing cells will be able to replenish the cell number and appear as a white cell pellet at the bottom of the well. Within 3 weeks, irradiated producer cells as well as cell debris were not visible and cell pellets reflected growing cells (Fig. 5). Since this assay is very stringent, slowly growing or nondividing cells are eliminated very quickly and the percentage of surviving wells is an accurate measure of the immortalization efficiency of viruses. A Kaplan-Meier plot of HTLV-1-induced T-cell proliferation indicated the percentage of wells containing proliferating lymphocytes was not significantly different between wt HTLV-1 and H1IT (Fig. 5). Data presented in Fig. 5 are based on the assessment of whether cells per well increase in number without presenting the differences in proliferation and/or apoptosis per well. Therefore, eight wells per line were selected weekly for more quantitative measurement of proliferation and apoptosis using MTS and cell death detection ELISA, respectively. No significant differences in either growth or induction of apoptosis were observed between HTLV-1- or H1IT-infected cells at any time point during coculture (data not shown). Our results are consistent with the conclusion that H1IT and wt HTLV-1 have equivalent immortalization efficiencies.
FIG. 5.
Representative Kaplan-Meier plot for T-lymphocyte proliferation in short-term microtiter assay. Prestimulated PBMCs (104) were cocultured with 200 irradiated 729 stable producer cells in 96-well plates. The percentages of proliferating wells were plotted as a function of time (weeks). Kaplan-Meier plots for wt HTLV-1 and H1IT and uninfected 729 control cells are shown. Results indicated that the percentage of wells containing proliferating lymphocytes was not significantly different between wt HTLV-1 and H1IT.
Lastly, we confirmed by PCR that the immortalized PBMCs harbor viral sequences in their genome. Our data showed that PBMC-H1IT cells harbor the unique sequences of IRES-Tax, suggesting that viral transmission was responsible for the immortalization of PBMCs (Fig. 6).
FIG. 6.
Immortalized human PBMCs harbor integrated viral sequences in their genome. Diagnostic PCR was performed on genomic DNA extracted from immortalized cells 8 weeks postcoculture. Using primers that differentiate between wt HTLV-1 and H1IT proviruses, it was confirmed that immortalized peripheral blood lymphocytes (PBL) contained the expected proviral sequences.
H1IT fails to persist in vivo.
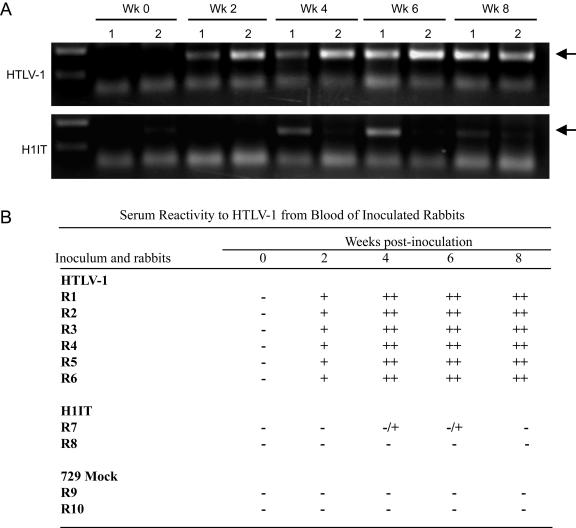
Our data show that H1IT expresses similar levels of Tax as the wt HTLV-1 clone, despite the fact that IRES-dependent expression is less efficient than cap-dependent expression. This likely can be attributed to introducing the Tax cassette at the 3′ end of the provirus, where it becomes a component of all viral transcripts. While this expression pattern of Tax did not affect in vitro immortalization of primary T lymphocytes, we next tested if this genome modification had an overall affect on viral survival in vivo. It has been hypothesized that temporal and quantitative expression of different transcripts in HTLV-1 is tightly balanced and critical for virus survival and pathogenesis. To evaluate the importance of this tight regulation in vivo, we compared the abilities of 729, 729-HTLV-1, and 729-H1IT cell lines to establish infection and persistence in our rabbit model. Rabbits were inoculated with lethally irradiated cell lines, and blood was drawn at weeks 0, 2, 4, 6, and 8 after inoculation. Rabbit PBMCs were isolated from blood to determine viral DNA integration by PCR, and rabbit serum was assessed for anti-HTLV-1 antibody response by Western blotting. wt HTLV-1 sequences were detectable at all time points postinoculation, indicating that the virus was able to infect and persist in rabbits (Fig. 7A). Conversely, the H1IT virus failed to productively infect and persist in rabbit PBMCs. Only one of the rabbits inoculated with 729-H1IT showed a transient signal by PCR, which was lost by the end of the 8-week study. We also used sera from these rabbits to test the immune response to the different viruses. HTLV-1 induced a potent antibody response starting week 2, whereas the response to H1IT was low and transient (Fig. 7B). These data suggest that unregulated Tax expression is not advantageous for the virus that needs to have strict temporal control over its specific transcripts for persistence in vivo.
FIG. 7.
H1IT virus fails to persist in vivo. (A) DNA isolated from PBMCs of two rabbits from each group inoculated with lethally irradiated cell lines was tested for viral DNA integration by PCR at weeks 0, 2, 4, 6, and 8 after inoculation. In contrast to wt HTLV-1, whose sequences were detectable at all time points postinoculation, H1IT failed to productively infect and persist in rabbit PBMCs. Only one of the rabbits inoculated with 729-H1IT showed a transient signal. (B) Rabbit sera were assessed for anti-HTLV-1 antibody response by Western blotting. The representative seroconversion patterns from each of the inoculated groups are summarized. HTLV-1 induced a potent antibody response starting at week 2, whereas the H1IT response was low and transient.
DISCUSSION
Tax and Rex are two regulatory proteins that are essential for HTLV replication (18). Abundant information has been accumulated about the biochemical properties of both proteins. Mutational analyses have identified Tax or Rex mutants with defective activities or impaired biochemical properties that are associated with protein function. For example, Tax mutants that specifically diminish the protein's ability to activate viral transcription without affecting its ability to activate the NF-κB pathway or vice versa have been identified (6, 15, 33, 47, 50). Conversely, certain Tax mutants are deficient for interacting with cell cycle components but still are able to induce NF-κB activity as well as transcription from the LTR (2, 3, 22, 23, 39). Such mutants are of special importance, because they allow for the dissection of the several Tax activities and their specific role in HTLV-1-mediated transformation of primary human T lymphocytes. In addition, several Rex mutants that either inhibit Rex activity or render the protein constitutively active have been described (1, 24, 26, 59). Thus, it is worthwhile to study the effect of loss of Rex regulation on the viral replication cycle. Although some of these mutants have been known for several years, our ability to study their effects directly in the context of a full-length replication-competent provirus is hindered by the fact that a significant portion of the tax and rex ORFs is overlapping. Mutations in one gene would inevitably alter the other, making interpretation of viral phenotypes very difficult.
In this study we developed a novel HTLV-1 molecular clone (H1IT) in which tax and rex ORFs are separated. We replaced the original tax gene by introducing an IRES followed by the entire Tax cDNA immediately downstream of the Rex termination codon. In doing so, both Tax and Rex still are expressed from one bicistronic mRNA, but they are on consecutive rather than overlapping ORFs. Our results demonstrated that the rearrangement of tax and rex ORFs did not affect their expression or function in transient assays. Stable cell lines harboring the H1IT provirus had significant expression of matrix p19 Gag protein as measured by ELISA as well as Env glycoprotein, Tax, and capsid p24 Gag detected by Western blotting. Based on previous reports that suggest that an IRES is less efficient in expressing a downstream ORF to the same level of cap-dependent expression (36, 40, 55), we did not expect the expression or activity of Tax produced by H1IT to be indistinguishable from that of wt HTLV-1. We explain the efficient expression of Tax from H1IT by introducing the IRES-Tax cassette at the 3′ end of the provirus. Since all viral transcripts share the same 3′ end, it is inevitable that the IRES-Tax cassette is part of all these transcripts. Therefore, the low efficiency of IRES-mediated expression is compensated for by multiple copies of the IRES-Tax cassette. A myriad of functions is attributed to Tax, ranging from expression of viral genes, dysregulation of cell cycle, and interference with DNA repair to HTLV-induced transformation of T cells. Thus, it is critical that Tax be expressed at efficient levels to study these functions in vitro.
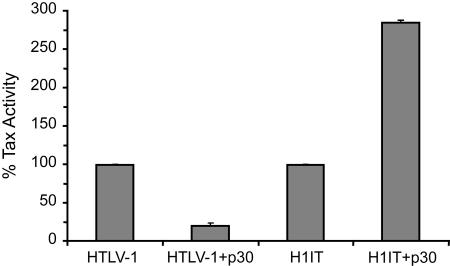
While our H1IT provirus was able to express adequate levels of Tax to induce sufficient viral replication and, more importantly, HTLV-induced immortalization of primary human T lymphocytes, we also had a unique opportunity to evaluate the dysregulation of Tax expression on viral persistence in vivo. Although not studied in detail, it has been suggested that there is strict temporal and quantitative control on expression of specific viral transcripts during the course of viral infection, replication, latency, and persistence in vivo. For example, tax/rex mRNA is one of the initial transcripts to be made upon viral integration (18). The products of this transcript are essential for regulating the expression of other mRNAs. Rex, for instance, is needed for full expression of incompletely spliced gag, pol, and env mRNA, leading to the assumption that these mRNAs are late products of infection (59). On the other hand, in late-stage HTLV-1-associated leukemias, Tax is rarely detected, which suggests that this protein is down-regulated and not required to maintain the tumorigenic phenotype (25, 28, 30, 35). Collectively, these observations lead to the conclusion that Tax expression is temporally regulated during the life cycle of HTLV-1 infection and disease induction. In our H1IT provirus, in addition to being expressed from its original tax/rex transcript, Tax has the potential to be expressed from all viral mRNA species, which causes a disruption of its temporal regulation. For example, data from our lab and others showed that p30 of HTLV-1 and p28 of HTLV-2 play a role in specifically down-regulating tax/rex mRNA expression at a posttranscriptional level (41, 60-62). This negative regulation of Tax and Rex was proposed to be important for the virus to evade immune recognition in vivo. In H1IT, p30 repression of Tax would be disrupted (Fig. 8). We speculate that at a certain point during infection, while Tax expressed from the original tax/rex mRNA would still be inhibited by p30, Tax would be expressed from other transcripts that are unaffected by p30, such as gag/pol mRNA. Our in vivo data are in agreement with the hypothesis that temporal regulation of Tax is important for viral persistence in an immune-competent animal model. Alternative explanations for the lack of persistence of H1IT are also possible. We ruled out that inefficient packaging of a larger genome plays a role, since our data show that similar amounts of viral RNA were packaged into HTLV-1 or H1IT virions. Although the presence of the IRES could also interfere with reverse transcriptase, leading to less efficient infection, this seems unlikely given that the virus has a high capacity to infect and immortalize primary T lymphocytes in vitro. A disruption in the expression profile of HTLV-1 accessory proteins could also lead to an in vivo phenotype similar to that of H1IT. However, since the IRES-Tax cassette was introduced well beyond all splice donor or acceptor sites on the plus strand and downstream of any of the accessory protein ORFs, including a fully functional Rex, it is unlikely that any of these proteins have been disrupted in H1IT. It does remain a possibility that the IRES could alter the translation of the accessory proteins. Unfortunately, this cannot be tested, since the accessory proteins do not appear to be expressed at detectable levels from the provirus. The only ORF whose splicing might be affected by the IRES insertion is that of HBZ on the minus strand. The IRES-Tax cassette lies in the intron of HBZ RNA and, hence, might affect splicing. We rule out such a mechanism because our Western blot data show that HBZ is efficiently expressed in H1IT. Lastly, we cannot eliminate the possibility that an IRES-specific immune response could lead to clearance of IRES-containing infected cells.
FIG. 8.
H1IT Tax activity is not repressed by p30. 293T cells were transfected with either wild-type HTLV-1 or H1IT proviral DNA along with the Tax reporter LTR-1-luciferase in the presence or absence of a p30 expression plasmid. CMV-βgal was used to control for transfection efficiency. Cell lysates were assayed 48 h posttransfection for Tax activity. The data shown are representative of three independent experiments. As previously reported, p30 is able to inhibit Tax activity in HTLV-1, whereas H1IT had an opposite effect, with Tax activity showing a two- to threefold induction in the presence of p30.
In summary, our novel H1IT provirus provides a valuable reagent to study the effects of specific Tax or Rex mutants on viral replication and viral-induced transformation in vitro. Furthermore, it also gives us insight into the paramount importance of strict regulation of viral transcript expression in a temporal and quantitative manner in vivo.
Acknowledgments
We thank Tim Vojt for preparation of the figures and Kate Hayes for editorial comments.
This work is supported by a grant from the National Institutes of Health (CA77556).
REFERENCES
- 1.Adachi, Y., T. D. Copeland, C. Takahashi, T. Nosaka, A. Ahmed, S. Oroszlan, and M. Hatanaka. 1992. Phosphorylation of the Rex protein of human T-cell leukemia virus type I. J. Biol. Chem. 267:21977-21981. [PubMed] [Google Scholar]
- 2.Akagi, T., H. Ono, and K. Shimotohno. 1996. Expression of cell-cycle regulatory genes in HTLV-I infected T-cell lines: possible involvement of Tax1 in the altered expression of cyclin D2, p18Ink4, and p21Waf1/Cip1/Sdi1. Oncogene 12:1645-1652. [PubMed] [Google Scholar]
- 3.Akita, K., S. Kawata, and K. Shimotohno. 2005. p21WAF1 modulates NF-κB signaling and induces anti-apoptotic protein Bcl-2 in Tax-expressing rat fibroblast. Virology 332:249-257. [DOI] [PubMed] [Google Scholar]
- 4.Anderson, M. D., J. Ye, L. Xie, and P. L. Green. 2004. Transformation studies with a human T-cell leukemia virus type 1 molecular clone. J. Virol. Methods 116:195-202. [DOI] [PubMed] [Google Scholar]
- 5.Armstrong, A. P., A. A. Franklin, M. N. Henbogaard, H. A. Giebler, and J. K. Nyborg. 1993. Pleiotropic effect of the human T-cell leukemia virus Tax protein on the DNA binding activity of eukaryotic transcription factors. Proc. Natl. Acad. Sci. USA 90:7303-7307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bex, F., M. J. Yin, A. Burny, and R. B. Gaynor. 1998. Differential transcriptional activation by human T-cell leukemia virus type 1 Tax mutants is mediated by distinct interactions with CREB binding protein and p300. Cell 18:2392-2405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brady, J., K. T. Jeang, J. Durall, and G. Khoury. 1987. Identification of the p40x-responsive regulatory sequences within the human T-cell leukemia virus type I long terminal repeat. J. Virol. 61:2175-2181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cann, A. J., Y. Koyanagi, and I. S. Y. Chen. 1988. High efficiency transfection of primary human lymphocytes and studies of gene expression. Oncogene 3:123-128. [Google Scholar]
- 9.Chen, I. S. Y., D. W. Golde, D. J. Slamon, and W. Wachsman. 1985. Comparative studies of HTLV-I and HTLV-II, p. 137-149. In R. P. Gale and D. W. Golde (ed.), Leukemia 1985: UCLA symposia, vol. 28. Alan R. Liss, New York, N.Y. [Google Scholar]
- 10.Ciminale, V., L. Zotti, D. M. D'agostino, and L. Chieco-Bianchi. 1997. Inhibition of human T-cell leukemia virus type 2 Rex function by truncated forms of Rex encoded in alternately spliced mRNAs. J. Virol. 71:2810-2818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Collins, N. D., G. C. Newbound, B. Albrecht, J. Beard, L. Ratner, and M. D. Lairmore. 1998. Selective ablation of human T-cell lymphotropic virus type 1 p12I reduces viral infectivity in vivo. Blood 91:4701-4707. [PubMed] [Google Scholar]
- 12.Cullen, B. R. 1992. Mechanism of action of regulatory proteins encoded by complex retroviruses. Microbiol. Rev. 56:375-394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Endo, K., A. Hirata, K. Iwai, M. Sakurai, M. Fukushi, M. Oie, M. Higuchi, W. W. Hall, F. Gejyo, and M. Fujii. 2002. Human T-cell leukemia virus type 2 (HTLV-2) Tax protein transforms a rat fibroblast cell line but less efficiently than HTLV-1 Tax. J. Virol. 76:2648-2653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fujisawa, J., M. Seiki, M. Sata, and M. Yoshida. 1986. A transcriptional enhancer sequence of HTLV-I is responsible for trans-activation mediated by p40xI of HTLV-I. EMBO J. 5:713-718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goren, I., O. J. Semmes, K. T. Jeang, and K. Moelling. 1995. The amino terminus of Tax is required for interaction with the cyclic AMP response element binding protein. J. Virol. 69:5806-5811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Grant, C., K. Barmak, T. Alefantis, J. Yao, S. Jacobson, and B. Wigdahl. 2002. Human T cell leukemia virus type I and neurologic disease: events in bone marrow, peripheral blood, and central nervous system during normal immune surveillance and neuroinflammation. J. Cell Physiol. 190:133-159. [DOI] [PubMed] [Google Scholar]
- 17.Green, P. L., and I. S. Y. Chen. 1994. Molecular features of the human T-cell leukemia virus: mechanisms of transformation and leukemogenicity, p. 227-311. In J. A. Levy (ed.), The Retroviridae, vol. 3. Plenum Press, New York, N.Y. [Google Scholar]
- 18.Green, P. L., and I. S. Y. Chen. 2001. Human T-cell leukemia virus types 1 and 2, p. 1941-1969. In D. M. Knipe, P. Howley, D. Griffin, R. Lamb, M. Martin, and S. Straus (ed.), Virology, 4th ed. Lippincott Williams & Wilkins, Philidelphia, Pa.
- 19.Green, P. L., T. M. Ross, I. S. Y. Chen, and S. Pettiford. 1995. Human T-cell leukemia virus type II nucleotide sequences between env and the last exon of tax/rex are not required for viral replication or cellular transformation. J. Virol. 69:387-394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gröne, M., C. Koch, and R. Grassmann. 1996. The HTLV-1 Rex protein induces nuclear accumulation of unspliced viral RNA by avoiding intron excision and degradation. Virology 218:316-325. [DOI] [PubMed] [Google Scholar]
- 21.Grossman, W. J., J. T. Kimata, F. H. Wong, M. Zutter, T. J. Ley, and L. Ratner. 1995. Development of leukemia in mice transgenic for the tax gene of human T-cell leukemia virus type I. Proc. Natl. Acad. Sci. USA 92:1057-1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Haller, K., T. Ruckes, I. Schmitt, D. Saul, E. Derow, and R. Grassmann. 2000. Tax-dependent stimulation of G1 phase-specific cyclin-dependent kinases and increased expression of signal transduction genes characterize HTLV type 1-transformed T cells. AIDS Res. Hum. Retrovir. 16:1683-1688. [DOI] [PubMed] [Google Scholar]
- 23.Haller, K., Y. Wu, E. Derow, I. Schmitt, K. T. Jeang, and R. Grassmann. 2002. Physical interaction of human T-cell leukemia virus type 1 tax with cyclin-dependent kinase 4 stimulates the phosphorylation of retinoblastoma protein. Mol. Cell. Biol. 22:3327-3338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Heger, P., O. Rosorius, C. Koch, G. Casari, R. Grassmann, and J. Hauber. 1998. Multimer formation is not essential for nuclear export of human T-cell leukemia virus type 1 rex trans-activator protein. J. Virol. 72:8659-8668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hishizawa, M., K. Imada, T. Ishikawa, and T. Uchiyama. 2004. Kinetics of proviral DNA load, soluble interleukin-2 receptor level and tax expression in patients with adult T-cell leukemia receiving allogeneic stem cell transplantation. Leukemia 18:167-169. [DOI] [PubMed] [Google Scholar]
- 26.Hofer, L., I. Weichselbraun, S. Quick, G. K. Farrington, E. Bohnlein, and J. Hauber. 1991. Mutational analysis of the human T-cell leukemia virus type I trans-acting rex gene product. J. Virol. 65:3379-3383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jin, D.-Y., V. Giordano, K. V. Kilber, H. Nakano, and K.-T. Jeang. 1999. Role of adapter function in oncoprotein-mediated activation of NFkB: HTLV-1 Tax interacts directly with IkB kinase g. J. Biol. Chem. 274:17402-17405. [DOI] [PubMed] [Google Scholar]
- 28.Kato, T., H. Asahara, M. S. Kurokawa, K. Fujisawa, T. Hasunuma, H. Inoue, M. Tsuda, S. Takahashi, S. Motokawa, T. Sumida, and K. Nishioka. 2004. HTLV-I env protein acts as a major antigen in patients with HTLV-I-associated arthropathy. Clin. Rheumatol. 23:400-409. [DOI] [PubMed] [Google Scholar]
- 29.Kim, J. H., P. A. Kaufman, S. M. Hanly, L. T. Rimsky, and W. C. Greene. 1991. Rex transregulation of human T-cell leukemia virus type II gene expression. J. Virol. 65:405-414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kurihara, K., N. Harashima, S. Hanabuchi, M. Masuda, A. Utsunomiya, R. Tanosaki, M. Tomonaga, T. Ohashi, A. Hasegawa, T. Masuda, J. Okamura, Y. Tanaka, and M. Kannagi. 2005. Potential immunogenicity of adult T cell leukemia cells in vivo. Int. J. Cancer 114:257-267. [DOI] [PubMed] [Google Scholar]
- 31.Kusuhara, K., M. Anderson, S. M. Pettiford, and P. L. Green. 1999. Human T-cell leukemia virus type 2 Rex protein increases stability and promotes nuclear to cytoplasmic transport of gag/pol and env RNAs. J. Virol. 73:8112-8119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li, X. H., and R. B. Gaynor. 2000. Mechanisms of NF-κB activation by the HTLV type 1 tax protein. AIDS Res. Hum. Retrovir. 16:1583-1590. [DOI] [PubMed] [Google Scholar]
- 33.Li, X. H., K. M. Murphy, K. T. Palka, R. M. Surabhi, and R. B. Gaynor. 1999. The human T-cell leukemia virus type-1 Tax protein regulates the activity of the IκB kinase complex. J. Biol. Chem. 274:34417-34424. [DOI] [PubMed] [Google Scholar]
- 34.Mahieux, R., and A. Gessain. 2003. HTLV-1 and associated adult T-cell leukemia/lymphoma. Rev. Clin. Exp. Hematol. 7:336-361. [PubMed] [Google Scholar]
- 35.Mitre, E., R. W. Thompson, E. M. Carvalho, T. B. Nutman, and F. A. Neva. 2003. Majority of interferon-gamma-producing CD4+ cells in patients infected with human T cell lymphotrophic virus do not express tax protein. J. Infect. Dis. 188:428-432. [DOI] [PubMed] [Google Scholar]
- 36.Mizuguchi, H., Z. Xu, A. Ishii-Watabe, E. Uchida, and T. Hayakawa. 2000. IRES-dependent second gene expression is significantly lower than cap-dependent first gene expression in a bicistronic vector. Mol. Ther. 1:376-382. [DOI] [PubMed] [Google Scholar]
- 37.Morgan, R. A., L. Couture, O. Elroy-Stein, J. Ragheb, B. Moss, and W. F. Anderson. 1992. Retroviral vectors containing putative internal ribosome entry sites: development of a polycistronic gene transfer system and applications to human gene therapy. Nucleic Acids Res. 20:1293-1299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Narayan, M., K. Kusuhara, and P. L. Green. 2001. Phosphorylation of two serine residues regulates human T-cell leukemia virus type 2 Rex function. J. Virol. 75:8440-8448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Neuveut, C., and K. T. Jeang. 2002. Cell cycle dysregulation by HTLV-I: role of the tax oncoprotein. Front. Biosci. 7:D157-D163. [DOI] [PubMed] [Google Scholar]
- 40.Ngoi, S. M., A. C. Chien, and C. G. Lee. 2004. Exploiting internal ribosome entry sites in gene therapy vector design. Curr. Gene Ther. 4:15-31. [DOI] [PubMed] [Google Scholar]
- 41.Nicot, C., J. M. Dundr, J. R. Johnson, J. R. Fullen, N. Alonzo, R. Fukumoto, G. L. Princler, D. Derse, T. Misteli, and G. Franchini. 2004. HTLV-1-encoded p30II is a post-transcriptional negative regulator of viral replication. Nat. Med. 10:197-201. [DOI] [PubMed] [Google Scholar]
- 42.Nicot, C., F. Tie, and C. Z. Giam. 1998. Cytoplasmic forms of human T-cell leukemia virus type 1 Tax induce NF-κB activation. J. Virol. 72:6777-6784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Persaud, D., J. L. Munoz, S. L. Tarsis, E. S. Parks, and W. P. Parks. 1995. Time course and cytokine dependence of human T-cell lymphotropic virus type 1 T-lymphocyte transformation as revealed by a microtiter infectivity assay. J. Virol. 69:6297-6303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ressler, S., G. F. Morris, and S. J. Marriott. 1997. Human T-cell leukemia virus type 1 Tax transactivates the human proliferating cell nuclear antigen promoter. J. Virol. 71:1181-1190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Robek, M. D., and L. Ratner. 1999. Immortalization of CD4+ and CD8+ T-lymphocytes by human T-cell leukemia virus type 1 Tax mutants expressed in a functional molecular clone. J. Virol. 73:4856-4865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rosenblatt, J. D., A. J. Cann, D. J. Slamon, I. S. Smalberg, N. P. Shah, J. Fujii, W. Wachsman, and I. S. Y. Chen. 1988. HTLV-II trans-activation is regulated by two overlapping nonstructural genes. Science 240:916-919. [DOI] [PubMed] [Google Scholar]
- 47.Rosin, R., C. Koch, I. Schmitt, O. J. Semmes, K.-T. Jeang, and R. Grassmann. 1998. A human T-cell leukemia virus Tax variant incapable of activating NF-κB retains its immortalizing potential for primary T-lymphocytes. J. Biol. Chem. 273:6698-6703. [DOI] [PubMed] [Google Scholar]
- 48.Ross, T. M., M. Narayan, Z. Y. Fang, A. C. Minella, and P. L. Green. 2000. Tax transactivation of both NF-κB and CREB/ATF is essential for human T-cell leukemia virus type 2-mediated transformation of primary human T cells. J. Virol. 74:2655-2662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ross, T. M., S. M. Pettiford, and P. L. Green. 1996. The tax gene of human T-cell leukemia virus type 2 is essential for transformation of human T lymphocytes. J. Virol. 70:5194-5202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Semmes, O. J., and K. T. Jeang. 1995. Definition of a minimal activation domain in human T-cell leukemia virus type I Tax. J. Virol. 69:1827-1833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Semmes, O. J., and K. T. Jeang. 1992. Mutational analysis of human T-cell leukemia virus type I Tax: regions necessary for function determined with 47 mutant proteins. J. Virol. 66:7183-7192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Trejo, S. R., W. E. Fah, and L. Ratner. 1996. c-sis/PDGF-B promoter transactivator by the Tax protein of the human T-cell leukemia virus type 1. J. Biol. Chem. 271:14584-14590. [DOI] [PubMed] [Google Scholar]
- 53.Wano, Y., M. Feinberg, J. B. Hosking, H. Bogerd, and W. C. Greene. 1988. Stable expression of the HTLV-I Tax protein in human T-cells activates specific cellular genes involved in growth. Proc. Natl. Acad. Sci. USA 85:9733-9737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Watanabe, T. 1997. HTLV-1-associated diseases. Int. J. Hematol. 66:257-278. [DOI] [PubMed] [Google Scholar]
- 55.Wong, E. T., S. M. Ngoi, and C. G. Lee. 2002. Improved co-expression of multiple genes in vectors containing internal ribosome entry sites (IRESes) from human genes. Gene Ther. 9:337-344. [DOI] [PubMed] [Google Scholar]
- 56.Wycuff, D. R., and S. J. Marriott. 2005. The HTLV-1 Tax oncoprotein: hypertasking at the molecular level. Front. Biosci. 10:620-642. [DOI] [PubMed] [Google Scholar]
- 57.Ye, J., L. Sileverman, M. D. Lairmore, and P. L. Green. 2003. HTLV-1 Rex is required for viral spread and persistence in vivo but is dispensable for cellular immortalization in vitro. Blood 102:3963-3969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yin, M.-J., L. B. Christerson, Y. Yamamoto, Y.-T. Kwak, S. Xu, F. Mercurio, M. Barbosa, M. H. Cobb, and R. B. Gaynor. 1998. HTLV-1 Tax protein binds to MEKK1 to stimulate IκB kinase activity and NF-κB activation. Cell 93:875-884. [DOI] [PubMed] [Google Scholar]
- 59.Younis, I., and P. L. Green. 2005. The human T-cell leukemia virus Rex protein. Front. Biosci. 10:431-445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Younis, I., L. Khair, M. Dundr, M. D. Lairmore, G. Franchini, and P. L. Green. 2004. Repression of human T-cell leukemia virus type 1 and 2 replication by a viral mRNA-encoded posttranscriptional regulator. J. Virol. 78:11077-11083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang, W., J. W. Nisbet, B. Albrecht, W. Ding, F. Kashanchi, J. T. Bartoe, and M. D. Lairmore. 2001. Human T-lymphotropic virus type 1 p30II regulates gene transcription by binding CREB binding protein/p300. J. Virol. 75:9885-9895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhang, W., J. W. Nisbet, J. T. Bartoe, W. Ding, and M. D. Lairmore. 2000. Human T-lymphotropic virus type 1 p30II functions as a transcription factor and differentially modulates CREB-responsive promoters. J. Virol. 74:11270-11277. [DOI] [PMC free article] [PubMed] [Google Scholar]