Abstract
Cis elements that mediate transcription factor binding are abundant within genomes, but the rules governing occupancy of such motifs in chromatin are not understood. The transcription factor GATA-1 that regulates red blood cell development binds with high affinity to GATA motifs, and initial studies suggest that these motifs are often unavailable for occupancy in chromatin. Whereas GATA-2 regulates the differentiation of all blood cell lineages via GATA motif binding, the specificity of GATA-2 chromatin occupancy has not been studied. We found that conditionally active GATA-1 (ER-GATA-1) and GATA-2 occupy only a small subset of the conserved GATA motifs within the murine β-globin locus. Kinetic analyses in GATA-1-null cells indicated that ER-GATA-1 preferentially occupied GATA motifs at the locus control region (LCR), in which chromatin accessibility is largely GATA-1-independent. Subsequently, ER-GATA-1 increased promoter accessibility and occupied the βmajor promoter. ER-GATA-1 increased erythroid Krüppel-like factor and SWI/SNF chromatin remodeling complex occupancy at restricted LCR sites. These studies revealed three phases of β-globin locus activation: GATA-1-independent establishment of specific chromatin structure features, GATA-1-dependent LCR complex assembly, and GATA-1-dependent promoter complex assembly. The differential utilization of dispersed GATA motifs therefore establishes spatial/temporal regulation and underlies the multistep activation mechanism.
Keywords: erythropoiesis, globin, histone, epigenetic, erythoid Krüppel-like factor
The transcription factor GATA-1 drives the differentiation of red blood cells from hematopoietic precursor cells (1–6). Whereas multiple GATA factor family members regulate development, GATA-1 has nonredundant functions to induce erythropoiesis and the maturation of platelets and mast cells (7). Major progress has been made in defining biological activities elicited by GATA-1 and other GATA factors, but many questions remain unanswered regarding their biochemical mechanisms (8).
By contrast to zinc fingers that solely mediate DNA binding, the GATA-1 zinc fingers are multifunctional. The GATA-1 C-terminal finger binds GATA motifs [(A/T)GATA(A/G)] (9, 10), and the N-terminal finger binds the nine zinc finger-containing protein Friend of GATA-1 (FOG-1) (11, 12). The N finger also stabilizes DNA binding at certain GATA motifs (13–15). GATA-1-mediated activation and repression can require FOG-1, although GATA-1 target genes can also be FOG-1 independent (12). FOG-1 increases GATA-1 chromatin occupancy (16, 17) and is required for GATA switches in which GATA-1 displaces chromatin-bound GATA-2 (16). Despite the multiple zinc fingers, intrinsic FOG-1 DNA binding activity has not been demonstrated. GATA-1 also binds the histone acetyltransferases CREB binding protein/p300 (18), which interact with a plethora of activators (19). Elucidating mechanisms underlying GATA-1-mediated transcriptional regulation and identifying GATA-1 target genes should provide key insights into how GATA-1 regulates cell differentiation and proliferation.
Major progress has been made in defining the GATA-1-instigated genetic network. Studies of protein–DNA interactions at the β-globin locus led to the identification of the first GATA-1 target genes, the β-globin genes (20, 21). Chromatin immunoprecipitation (ChIP) analyses showed that GATA-1 occupies β-globin (22) and α-globin loci (23) regulatory regions. Gene profiling studies in GATA-1-null cells (G1E), with or without a conditionally active estrogen receptor ligand binding domain fusion to GATA-1 (ER-GATA-1), revealed many positively and negatively regulated genes (24). Translating microarray data into direct targets can be complex, because ER-GATA-1 activation rapidly blocks proliferation (25), which can indirectly affect transcription. This problem is further complicated by the abundance of GATA motifs in the genome.
Genomic DNA regions occupied by factors in cells can be identified by ChIP coupled with microarray chip (26). However, given the novelty of this technology, little is known about false-positive and false-negative rates. Bioinformatics analysis of cis-element conservation can predict functional cis elements in certain contexts (27). ChIP, without microarray, analysis requires knowledge of a prospective DNA binding region (28, 29). Because bioinformatics predictions yield prospective binding regions, a coupled ChIP-bioinformatics approach allows one to definitively assess factor occupancy. Herein, we used this approach to analyze ER-GATA-1 and GATA-2 chromatin occupancy at the murine β-globin locus.
The murine β-globin locus contains Ey and βH1 genes, active during embryogenesis, and the adult βmajor and βminor genes (30). Upstream of Ey resides the LCR (31, 32), consisting of four erythroid-specific DNaseI hypersensitive sites (HSs) (HS1–4) (33, 34). GATA-1 occupies HS1–4 and the βmajor promoter in adult erythroid cells (22), but occupancy has not been tested at many conserved GATA motifs of the locus. βmajor transcriptional activation is associated with higher-order chromatin rearrangements within the LCR (35) and looping to bring the LCR in proximity of βmajor (36–38). Looping requires GATA-1 (38) and the erythroid Krüppel-like factor (EKLF) (37) that binds certain CACCC motifs (39–41). Although EKLF occupancy in cells has not been described, studies with an EKLF-null cell line (42, 43), altered specificity EKLF mutants (40), and targeted deletions (44–46) indicate that EKLF regulates β-globin transcription. EKLF binds CREB binding protein/p300 (47) and the Brahma-related gene 1 (BRG1) component of the SWI/SNF chromatin remodeling complex (43, 48, 49) and appears to function via establishing active chromatin. Targeted deletion of EKLF abrogates DNaseI hypersensitivity at HS3 and the β-globin promoter (40), and EKLF mediates chromatin remodeling in vitro (48).
The studies described herein investigate the specificities of ER-GATA-1 and GATA-2 interactions with GATA motifs, the relationship between ER-GATA-1 concentration/activity and chromatin occupancy, and how ER-GATA-1 influences specific steps in β-globin locus activation. ER-GATA-1 and GATA-2 occupied only a small subset of conserved GATA motifs, and EKLF occupancy was unpredictable from the distribution of conserved CACCC motifs. We established the spatial/temporal regulation of ER-GATA-1-instigated molecular events, which revealed ER-GATA-1-independent and -dependent phases of β-globin locus activation.
Methods
Cell Culture. G1E cells stably expressing ER-GATA-1 (50), mouse erythroleukemia, and FOG-1-null cells were maintained as described in Supporting Methods, which is published as supporting information on the PNAS web site.
Quantitative ChIP Assay. Quantitative ChIP analysis was performed as described in ref. 29 and in Supporting Methods.
Quantitative RT-PCR Assay. cDNA was prepared from 1 μg of purified total RNA. RT-PCRs (20 μl) contained 2 μl of cDNA solution with the appropriate primers. Product was measured by SYBR green fluorescence. Relative expression levels were determined from a standard curve of serial dilutions of cDNA samples.
Restriction Endonuclease Accessibility Assay. Assays were performed as described in ref. 52.
Protein Analysis. Protein analysis was conducted as described in Supporting Methods.
Primers and Antibodies. Primers and antibodies are described in Supporting Methods (see also Tables 1 and 2, which are published as supporting information on the PNAS web site).
Results and Discussion
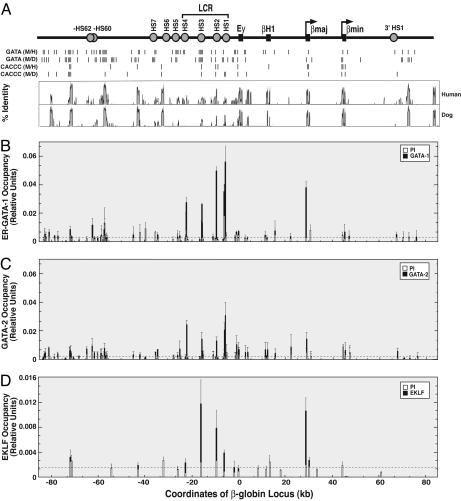
ER-GATA-1, GATA-2, and EKLF Occupy Only a Small Subset of Conserved DNA Motifs in Chromatin. The canonical GATA motif WGATAR, and WGATA and GATAR motifs that bind GATA factors with high-affinity in vitro (9, 10), are abundant in chromatin. As the rules of GATA factor chromatin occupancy are not understood (8), we used highly specific anti-GATA-1 and -GATA-2 antibodies (Fig. 8, which is published as supporting information on the PNAS web site) to analyze ER-GATA-1 and GATA-2 occupancy at 72 conserved (mouse vs. human) WGATAR, WGATA, and GATAR motifs (within 64 amplicons) of the β-globin locus in tamoxifen-treated G1E-ER-GATA-1 cells (Fig. 1A). ER-GATA-1 and GATA-1 occupy chromatin similarly (22). The conservation of murine β-globin locus sequence, relative to that of human and dog, and GATA motifs are shown in Fig. 1A. ER-GATA-1 occupancy was detected at nine GATA motif-containing amplicons (Fig. 1B). Analysis of the Ey, βH1, βmajor, and βminor promoters lacking conserved GATA motifs revealed occupancy only at βmajor, which contains one nonconserved WGATAR motif. ER-GATA-1 occupancy was not detected at HS5, which contains two conserved WGATA sequences. Similar results were obtained by using different G1E-ER-GATA-1 cell clonal lines expressing ER-GATA-1 at a level equivalent to or lower than mouse erythroleukemia cell GATA-1 (data not shown). Thus, ER-GATA-1 occupies a small subset of the conserved GATA motifs, and motif conservation and hypersensitivity are insufficient to predict occupancy.
Fig. 1.
ER-GATA-1, GATA-2, and EKLF occupy a small subset of binding motifs within the β-globin locus. (A) Organization of the murine β-globin locus. DNaseI hypersensitive sites (HSs) are depicted as filled circles, and embryonic (Ey and βH1) and adult (βmaj and βmin) globin genes are depicted as boxes. Vertical bars depict conserved motifs between mouse and human (M/H) or mouse and dog (M/D), and a vista plot of sequence identity between mouse and human or dog is plotted as a function of genomic coordinates (kb) by using the mouse sequence as a reference. The value 1 was assigned to the translation start site of Ey. (B–D) Quantitative ChIP analysis of ER-GATA-1 (B), GATA-2 (C), and EKLF (D) occupancy at conserved WGATAR, WGATA, GATAR (B and C) and CACCC (D) motifs at the murine β-globin locus in untreated (C) or tamoxifen-treated (B) (48 h) and (D) (20 h) G1E-ER-GATA-1 cells (mean ± SE, two to five independent experiments). Solid bars, signals obtained with anti-GATA-1, -GATA-2, and -EKLF antibodies (B, C, and D, respectively); open bars, preimmune signals; dotted line, averaged preimmune signals.
ER-GATA-1 expression in G1E cells represses GATA-2 transcription (50, 53). GATA-1 and GATA-2 bind similar GATA motifs in vitro, although GATA-2 binds AGATCTTA with higher affinity than GATA-1 (10). To compare how ER-GATA-1 and GATA-2 discriminate among GATA motifs, GATA-2 occupancy was measured in untreated G1E-ER-GATA-1 cells (Fig. 1C). No qualitative differences between GATA-2 and ER-GATA-1 occupancy were detected, but signals at HS2, relative to HS1, HS3, and HS4, were ≈2-fold lower for GATA-2 vs. ER-GATA-1.
Activation of the β-globin genes requires multiple factors including GATA-1, p45 subunit of nuclear factor erythroid-2 (p45/NF-E2), and EKLF. Because EKLF binds certain CACCC motifs in vitro (41), EKLF occupancy was measured at all 17 β-globin locus regions containing conserved CACCC motifs (Fig. 1A) and five functionally important regions lacking conserved CACCC motifs. Occupancy was detected at HS2, HS3, and the βmajor promoter and weakly at HS1 (Fig. 1D). No occupancy was detected at 18 regions, indicating that EKLF occupies a small subset of the motifs. Because GATA-1 and EKLF interact (54, 55), these factors might cooccupy or bind sites independently. The ER-GATA-1 and EKLF occupancy patterns differ in that little or no EKLF occupancy occurred at HS4 (Fig. 1D); ER-GATA-1 occupancy at HS4 was high. EKLF occupancy at HS1 was less than HS2 and HS3, whereas ER-GATA-1 occupancy at HS1 was equivalent to or higher than at other HSs. These results indicate that ER-GATA-1 and EKLF cooccupy HS2, HS3, and the promoter, but not all sites.
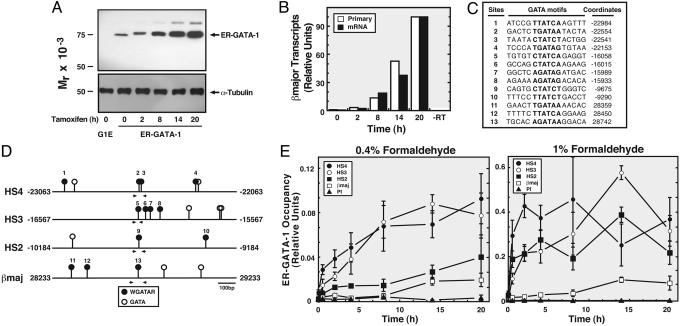
Although ER-GATA-1 Occupies the LCR and the βmajor Promoter in Definitive Erythroid Cells, ER-GATA-1 Preferentially Occupies the LCR When ER-GATA-1 Is Limiting. All WGATAR motifs and many derivatives thereof bind GATA factors with high-affinity in vitro (9, 10). Thus, ER-GATA-1 and GATA-2 occupancy of a small subset of conserved GATA motifs is unrelated to naked DNA binding affinities. To determine whether sites occupied by GATA factors in cells are differentially accessible to ER-GATA-1, we asked whether ER-GATA-1 occupies different sites with identical or distinct kinetics. Tamoxifen treatment of G1E-ER-GATA-1 cells for up to 20 h increased ER-GATA-1 levels/activity (Fig. 2 A and B). Low-level βmajor mRNA and primary transcripts were detected by 8 h after tamoxifen treatment. The transcripts were half-maximal by 14 h and further increased by 20 h (Fig. 2B). ER-GATA-1 occupancy at HS2, HS3, and HS4 was rapid, whereas promoter occupancy required 14 h (Fig. 2 C–E) by using either 0.4% or 1% formaldehyde (Fig. 2E). G1E-ER-GATA-1 cells were also treated with increasing concentrations of tamoxifen for 12 h. Although ER-GATA-1 occupied HS4 and HS3, almost no promoter occupancy was detected (Fig. 9, which is published as supporting information on the PNAS web site). Analysis of GATA-1 binding to oligonucleotides containing GATA motifs from HS2 or the βmajor promoter by EMSA, by using a range of protein concentrations, revealed indistinguishable high-affinity binding in both cases, confirming that the βmajor promoter GATA motif mediates high-affinity binding (data not shown). The finding that ER-GATA-1 occupancy of the LCR vs. the promoter can be segregated provides a strategy to elucidate spatial/temporal relationships among reactions that activate the β-globin locus.
Fig. 2.
ER-GATA-1 preferentially occupies the LCR vs. the βmajor promoter. (A) ER-GATA-1 Western blot analysis in G1E and G1E-ER-GATA-1 cells. (B) Real-time RT-PCR analysis of βmajor primary transcripts and mRNA normalized by GAPDH in G1E-ER-GATA-1 cells after treatment with 1 μM tamoxifen (mean values from two independent experiments). -RT, no reverse transcriptase. (C) Sequences of WGATAR motifs at the LCR and promoter regions. (D) WGATAR and GATA motifs at sites examined on the murine β-globin locus. WGATAR motifs, filled circles (1–13); GATA motifs, open circles. Primers used for ChIP analysis are indicated by arrowheads. (E) ChIP analysis of ER-GATA-1 occupancy at HS4, HS3, HS2, and βmajor promoter with varying time (mean ± SE, four to five independent experiments). PI, preimmune sera. βmaj, βmajor promoter.
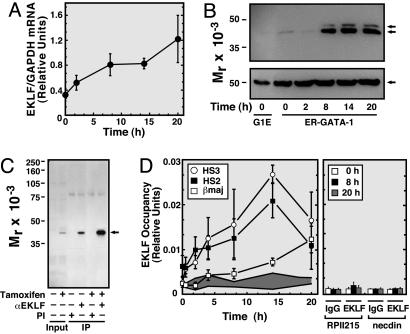
GATA-1 Mediates Spatially Restricted EKLF and BRG1 Recruitment. Because ER-GATA-1 elevates FOG-1 and EKLF mRNA (24), delayed ER-GATA-1 occupancy of the promoter might require the induction of these components. Cycloheximide cannot be used to test whether ER-GATA-1 occupancy at the promoter requires protein synthesis, because promoter occupancy requires ≈14 h, cycloheximide elicited toxicity after several hours, and factors such as GATA-2 with short half-lives do not persist upon cycloheximide treatment (data not shown). RT-PCR analysis was conducted to determine whether FOG-1 and EKLF mRNA increases under conditions in which ER-GATA-1 differentially occupies the LCR and promoter. FOG-1 mRNA was constant (data not shown), but EKLF mRNA (Fig. 3A) and protein (Fig. 3 B and C) increased by 8 h after tamoxifen treatment. ER-GATA-1 is not absolutely required for EKLF synthesis, because cells lacking ER-GATA-1 activity express EKLF (Fig. 3 B and C).
Fig. 3.
EKLF and ER-GATA-1 occupancy share common temporal regulation and have overlapping, but distinct, spatial regulation. (A) Real-time RT-PCR analysis of EKLF mRNA in tamoxifen-treated G1E-ER-GATA-1 cells (mean ± SE, four independent experiments). (B) Western blot analysis of EKLF (Upper, double arrow) and α-tubulin (Lower, arrow) in tamoxifen-treated G1E-ER-GATA-1 cells. (C) Western blot analysis of immunoprecipitated EKLF (arrow) in tamoxifen-treated G1E-ER-GATA-1 cells. (D) Quantitative ChIP analysis of EKLF occupancy at HS3 and HS2, and the βmajor, RPII215, and necdin promoters (mean ± SE, three to four independent experiments). (Left) Gray zone (1% formaldehyde), preimmune value range.
Because ER-GATA-1 elevates EKLF before ER-GATA-1 occupancy at the βmajor promoter (Figs. 2E and 3B), and EKLF occupies the promoter upon ER-GATA-1 activation for 20 h (Fig. 1D), EKLF might be required for ER-GATA-1 to occupy the promoter. Alternatively, EKLF might occupy the promoter after ER-GATA-1. EKLF resembled ER-GATA-1 in rapidly occupying HS2 and HS3 and subsequently occupying the promoter (Fig. 3D); no EKLF occupancy was detected at the active RPII215 promoter nor the inactive necdin promoter. The ER-GATA-1 and EKLF occupancy levels correlated with R2 values of 0.96, 0.74, and 0.71 for HS3, HS2, and the promoter, respectively (Fig. 10, which is published as supporting information on the PNAS web site).
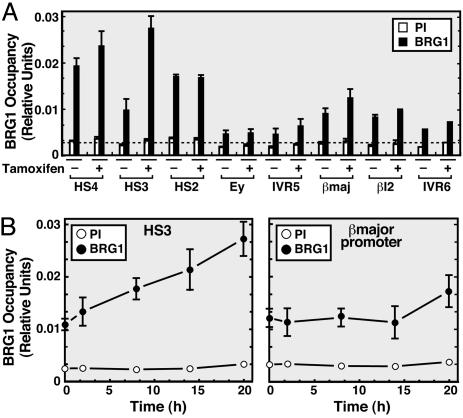
The SWI/SNF chromatin remodeling complex is required for EKLF-mediated activation of a β-globin promoter reconstituted into chromatin in vitro (48, 56). Because EKLF binds the BRG1 component of the complex (56), EKLF might recruit SWI/SNF complexes (57, 58) to chromatin. We tested whether BRG1 occupies the β-globin locus, whether BRG1 and EKLF are distributed similarly, and whether ER-GATA-1 activation affects the BRG1 distribution. In untreated G1E-ER-GATA-1 cells, BRG1 occupancy was detected at HS4, HS3, HS2, βmajor promoter, and βmajor intron 2 (βI2) (Fig. 4A). Thus, ER-GATA-1 activity and maximal EKLF levels are not required to recruit BRG1 to the locus. However, ER-GATA-1 activation for 20 h increased BRG1 occupancy at HS3 (Fig. 4A), which progressively increased upon ER-GATA-1 activation (Fig. 4B Left). BRG1 occupancy at the βmajor promoter slightly increased at 20 h (Fig. 4B Right).
Fig. 4.
ER-GATA-1 activation increases BRG1 recruitment at HS3. (A) Quantitative ChIP analysis of BRG1 occupancy. Dotted line, mean preimmune values. BRG1 occupancy profile (A) and kinetics (B) at the β-globin locus in tamoxifen-treated G1E-ER-GATA-1 cells (mean ± SE, four to five independent experiments). PI, preimmune sera; βmaj, βmajor promoter; βI2, βmajor intron 2.
The hematopoietic factor NF-E2 also occupies the LCR and, to a lesser extent, the βmajor promoter (51, 59–61). p45/NF-E2 occupies HS2 in an ER-GATA-1-independent manner, whereas occupancy at HS1 and HS3 is ER-GATA-1-dependent (22). We asked whether ER-GATA-1-dependent p45/NF-E2 occupancy occurs concomitant with ER-GATA-1 occupancy at the LCR, at the promoter, or during the intervening time. p45/NF-E2 levels and occupancy at HS2 were constant during the tamoxifen treatment (Fig. 11 A and B, which is published as supporting information on the PNAS web site). Occupancy increased more rapidly at HS3 vs. the promoter (Fig. 11B). Because ER-GATA-1 enhances Pol II recruitment to the LCR and promoter (22, 62) and p45/NF-E2 enhances Pol II recruitment to the promoter (51), we tested whether these spatially distinct events have shared or distinct kinetics. EKLF, p45/NF-E2, and Pol II occupied the LCR before the promoter, similar to ER-GATA-1 (Fig. 9C).
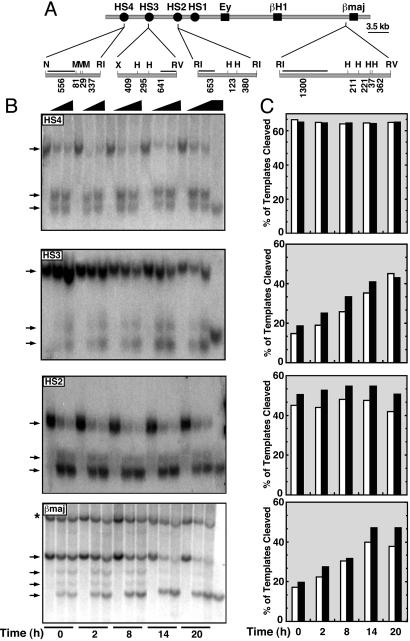
GATA-1-Dependent and -Independent Components of β-Globin Locus Chromatin Architecture. ER-GATA-1 increases histone acetylation at the LCR and the βmajor promoter (63, 64), but β-globin locus chromatin structure in cells, with or without GATA-1, has not been studied. Differences in GATA motif accessibility in chromatin might underlie the early and late phases of LCR and promoter occupancy, respectively. In this regard, GATA motifs are required for human LCR fragments to form HSs in transgenic mice (65). We used a restriction endonuclease accessibility assay (52, 66) to determine whether LCR and βmajor promoter accessibility is ER-GATA-1 dependent. Accessibility was measured in nuclei from G1E-ER-GATA-1 cells treated with tamoxifen for increasing times (Fig. 5A). MboI and HaeIII sites at HS4 and HS2, respectively, were cleaved on a high percentage of templates equivalently in nuclei from control and tamoxifen-treated cells (Fig. 5 B and C). By contrast, HaeIII cleavage at HS3 and the βmajor promoter was increased by ER-GATA-1 (Fig. 5 B and C). These results suggest that a promoter chromatin impediment underlies the delayed ER-GATA-1, EKLF, p45/NF-E2, and Pol II occupancy kinetics at the promoter, and this impediment must be overcome to assemble the promoter complex. However, ER-GATA-1 rapidly occupies HS3, despite the fact that HS3 accessibility is enhanced by ER-GATA-1. The HS3 scenario might be analogous to GATA-4 occupancy of a reconstituted, condensed chromatin template, which induced chromatin unfolding (67).
Fig. 5.
Chromatin accessibility at βmajor promoter and HS3, but not HS4 and HS2, is GATA-1 dependent. Nuclei from G1E-ER-GATA-1 cells treated for increasing times with 1 μM tamoxifen were incubated with 0, 50, or 100 units of HaeIII or MboI for 45 min at 37°C, and cleavage was measured by Southern blotting. (A) Southern blotting strategy. β-globin genes, HSs, and probes are depicted as boxes, circles, and black bars, respectively. N, NheI; M, MboI; RI, EcoRI; H, HaeIII; X, XmnI; RV, EcoRV. (B) Representative Southern blot. Parental fragments and cleavage products are indicated by arrows. The last lane of each blot shows results obtained with purified genomic DNA digested to completion with NheI/MboI/EcoRI, XmnI/HaeIII/EcoRV, EcoRI/HaeIII, and EcoRI/HaeIII/EcoRV, for analysis of HS4, HS3, HS2, and βmajor promoter, respectively. The asterisk indicates a polymorphic restriction fragment. (C) PhosphorImager analysis (mean values from two independent experiments). Open bars, 50 units; filled bars, 100 units.
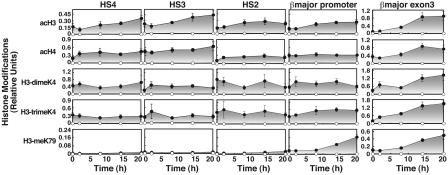
Based on the role of histone acetylation in increasing chromatin accessibility (68), insufficient acetylation might cause the factor access blockade, or other epigenetic marks mediating active chromatin formation (70) might be limiting. ER-GATA-1 increases acH3 and acH4 (acH, acetylated histone) at the promoter (63, 64) without increasing histone H3 dimethylated at lysine 4 (H3-dimeK4) (64). ER-GATA-1 modestly increased acH3 and acH4 at the LCR and acH3 at the promoter and the ORF (Fig. 6). H3-di/trimeK4 increased at the ORF. Histone H3 methylated at lysine 79 (H3-meK79), which has been implicated in counteracting Silent Information Regulator-mediated repression (69), is enriched at the βmajor promoter vs. the LCR (70). ER-GATA-1 activation increased H3-meK79 at the promoter and the ORF. The impediment to factor occupancy at the promoter therefore correlates with reduced HaeIII accessibility, acH3, and H3-meK79.
Fig. 6.
GATA-1-dependent and -independent epigenetic marks at the murine β-globin locus. ChIP analysis was used to measure the relative enrichments of histone modifications at HS4, HS3, HS2, βmajor promoter, and βmajor exon3 in tamoxifen-treated G1E-ER-GATA-1 cells (mean ± SE, four to six independent experiments). Filled and open circles, specific and preimmune signals, respectively. Relative enrichments of acH3, acH4, histone H3 dimethylated at lysine 4 (H3-dimeK4), histone H3 trimethylated at lysine 4 (H3-trimeK4), and H3-meK79 were measured by using specific antibodies. The preimmune data represent the highest averaged value.
Multistep Mechanism of β-Globin Locus Transcriptional Activation. The preferential occupancy at the LCR and the differential kinetics of LCR vs. promoter occupancy have important implications for β-globin locus activation. It is attractive to propose that a GATA-1 level sufficient to occupy the LCR is achieved during hematopoiesis, before occupancy of both the LCR and the promoter. Hypersensitivity and elevated histone acetylation at HS2 also precede β-globin locus activation (71, 72). The establishment of the active β-globin chromatin domain therefore occurs in a stepwise fashion, and our results segregate steps in relation to spatial/temporal regulation.
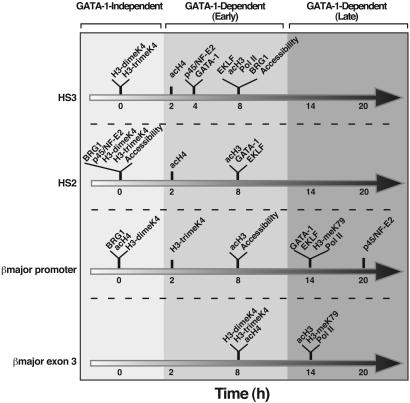
Because certain features of the erythroid-specific β-globin locus chromatin structure are preestablished in G1E cells lacking ER-GATA-1 activity, ER-GATA-1-independent molecular events constitute an initial phase of β-globin locus activation (Fig. 7). ER-GATA-1 occupancy at the LCR before the promoter instigates multiple events, including increased acH3, acH4, EKLF, p45/NF-E2, and Pol II occupancy at the LCR, representing the second phase of activation. Late in the second phase, β-globin transcripts become detectable. ER-GATA-1 occupancy at the promoter occurs concomitant with EKLF occupancy and increased acH3 at the promoter. These events, followed by increased p45/NF-E2 at the promoter, increased H3-meK79 at the promoter and the ORF, and increased H3-di/trimeK4, acH3, and acH4 at the ORF, represent the third phase of activation. Because targeted deletion of the LCR does not abrogate p45/NF-E2 (60) and GATA-1 (38) occupancy at the promoter, phase 2 is not required for phase 3. This multiphasic mechanism exemplifies the complexities of how cell type-specific activators instigate diverse reactions with stringent spatial/temporal control, thereby activating a chromatin domain. Further dissection of the intraphase reactions and the interphase relationships, in the context of the three-dimensional nuclear milieu, is expected will yield a comprehensive understanding of how cellular factors function combinatorially to regulate hemoglobin synthesis.
Fig. 7.
Multistep mechanism of ER-GATA-1-mediated activation of the βmajor gene. The time assigned for each event, derived from Figs. 2, 3, 4, 5, 6 and 9, represents at least a 50 ± 20% of the maximal change posttamoxifen treatment. HS2 and HS4 accessibility, and BRG1 occupancy at HS2, HS4, and the promoter are ER-GATA-1-independent (HS4 summary not shown). AcH4 at the promoter and H3-di/trimeK4 at the LCR and promoter are also ER-GATA-1 independent. acH3 rapidly increases at the LCR and the promoter upon ER-GATA-1 activation. BRG1 occupancy at HS3, and EKLF occupancy at HS3, HS2, and the promoter coincides with ER-GATA-1 occupancy at these sites. H3-meK79 increases at the promoter and the ORF late in activation.
Supplementary Material
Acknowledgments
We thank Soumen Pal for critical comments. We acknowledge support from National Institutes of Health (NIH) Grants DK50107 (to E.H.B.), DK55700 (to E.H.B.), and DK46865 (to J.J.B.). H.I. is a Predoctoral Fellow of the American Heart Assocation. K.D.J. acknowledges prior support by NIH Training Grant/NIH Grant DKK0169488 and current support from NIH Grant T32NL07899.
Author contributions: H.I., J.A.G., and E.H.B. designed research; H.I., J.A.G., K.D.J., S.-I.K., and M.E.B. performed research; A.N.I. and J.J.B. contributed new reagents/analytic tools; H.I., J.A.G., K.D.J., and E.H.B. analyzed data; and H.I. and E.H.B. wrote the paper.
Conflict of interest statement: No conflicts declared.
This paper was submitted directly (Track II) to the PNAS office.
Abbreviations: acH, acetylated histone; ChIP, chromatin immunoprecipitation; EKLF, erythroid Krüppel-like factor; ER-GATA-1, estrogen receptor hormone binding domain fusion to GATA-1; FOG-1, Friend of GATA-1; G1E, GATA-1 null; H3-meK79, histone H3 methylated at lysine 79; HS, DNaseI hypersensitive site; LCR, locus control region; p45/NF-E2, p45 subunit of nuclear factor erythroid-2.
References
- 1.Tsai, S. F., Martin, D. I., Zon, L. I., D'Andrea, A. D., Wong, G. G. & Orkin, S. H. (1989) Nature 339, 446-451. [DOI] [PubMed] [Google Scholar]
- 2.Evans, T. & Felsenfeld, G. (1989) Cell 58, 877-885. [DOI] [PubMed] [Google Scholar]
- 3.Pevny, L., Simon, M. C., Robertson, E., Klein, W. H., Tsai, S. F., D'Agati, V., Orkin, S. H. & Costantini, F. (1991) Nature 349, 257-260. [DOI] [PubMed] [Google Scholar]
- 4.Simon, M. C., Pevny, L., Wiles, M. V., Keller, G., Costantini, F. & Orkin, S. H. (1992) Nat. Genet. 1, 92-98. [DOI] [PubMed] [Google Scholar]
- 5.Pevny, L., Lin, C. S., D'Agati, V., Simon, M. C., Orkin, S. H. & Costantini, F. (1995) Development (Cambridge, U.K.) 121, 163-172. [DOI] [PubMed] [Google Scholar]
- 6.Fujiwara, Y., Browne, C. P., Cunniff, K., Goff, S. C. & Orkin, S. H. (1996) Proc. Natl. Acad. Sci. USA 93, 12355-12358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Crispino, J. D. (2005) Semin. Cell Dev. Biol. 16, 137-147. [DOI] [PubMed] [Google Scholar]
- 8.Bresnick, E. H., Martowicz, M. L., Pal, S. & Johnson, K. D. (2005) J. Cell. Physiol. 205, 1-9. [DOI] [PubMed] [Google Scholar]
- 9.Merika, M. & Orkin, S. H. (1993) Mol. Cell. Biol. 13, 3999-4010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ko, L. J. & Engel, J. D. (1993) Mol. Cell. Biol. 13, 4011-4022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tsang, A. P., Visvader, J. E., Turner, C. A., Fujuwara, Y., Yu, C., Weiss, M. J., Crossley, M. & Orkin, S. H. (1997) Cell 90, 109-119. [DOI] [PubMed] [Google Scholar]
- 12.Crispino, J. D., Lodish, M. B., MacKay, J. P. & Orkin, S. H. (1999) Mol. Cell 3, 219-228. [DOI] [PubMed] [Google Scholar]
- 13.Martin, D. I. & Orkin, S. H. (1990) Genes Dev. 4, 1886-1898. [DOI] [PubMed] [Google Scholar]
- 14.Trainor, C. D., Omichinski, J. G., Vandergon, T. L., Gronenborn, A. M., Clore, G. M. & Felsenfeld, G. (1996) Mol. Cell. Biol. 16, 2238-2247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Newton, A., MacKay, J. P. & Crossley, M. (2001) J. Biol. Chem. 276, 35794-35801. [DOI] [PubMed] [Google Scholar]
- 16.Pal, S., Cantor, A. B., Johnson, K. D., Moran, T. B., Boyer, M. E., Orkin, S. H. & Bresnick, E. H. (2004) Proc. Natl. Acad. Sci. USA 101, 980-985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Letting, D. L., Chen, Y.-Y., Rakowski, C., Reedy, S. & Blobel, G. A. (2004) Proc. Natl. Acad. Sci. USA 101, 476-481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blobel, G. A., Nakajima, T., Eckner, R., Montminy, M. & Orkin, S. H. (1998) Proc. Natl. Acad. Sci. USA 95, 2061-2066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vo, N. & Goodman, R. H. (2001) J. Biol. Chem. 276, 13505-13508. [DOI] [PubMed] [Google Scholar]
- 20.Evans, T., Reitman, M. & Felsenfeld, G. (1988) Proc. Natl. Acad. Sci. USA 85, 5976-5980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martin, D. I., Tsai, S. F. & Orkin, S. H. (1989) Nature 338, 435-438. [DOI] [PubMed] [Google Scholar]
- 22.Johnson, K. D., Grass, J. A., Boyer, M. E., Kiekhaefer, C. M., Blobel, G. A., Weiss, M. J. & Bresnick, E. H. (2002) Proc. Natl. Acad. Sci. USA 99, 11760-11765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Anguita, E., Hughes, J., Heyworth, C., Blobel, G. A., Wood, W. G. & Higgs, D. R. (2004) EMBO J. 23, 2841-2852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Welch, J. J., Watts, J. A., Vakoc, C. R., Yao, Y., Wang, H., Hardison, R. C., Blobel, G. A., Chodosh, L. A. & Weiss, M. J. (2004) Blood 104, 3136-3147. [DOI] [PubMed] [Google Scholar]
- 25.Rylski, M., Welch, J. J., Chen, Y. Y., Letting, D. L., Diehl, J. A., Chodosh, L. A., Blobel, G. A. & Weiss, M. J. (2003) Mol. Cell. Biol. 23, 5031-5042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kirmizis, A. & Farnham, P. J. (2004) Exp. Biol. Med. 229, 705-721. [DOI] [PubMed] [Google Scholar]
- 27.Conkright, M. D., Guzman, E., Flechner, L., Su, A. I., Hogenesch, J. B. & Montminy, M. (2003) Mol. Cell 11, 1101-1108. [DOI] [PubMed] [Google Scholar]
- 28.Johnson, K. D. & Bresnick, E. H. (2002) Methods 26, 27-36. [DOI] [PubMed] [Google Scholar]
- 29.Im, H., Grass, J. A., Johnson, K. D., Boyer, M. E., Wu, J. & Bresnick, E. H. (2004) Methods Mol. Biol. 284, 129-146. [DOI] [PubMed] [Google Scholar]
- 30.Trimborn, T., Gribnau, J., Grosveld, F. & Fraser, P. (1999) Genes Dev. 13, 112-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Forrester, W. C., Takegawa, S., Papayannopoulou, T., Stamatoyannopoulos, G. & Groudine, M. (1987) Nucleic Acids Res. 15, 10159-10177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Grosveld, F., van Assendelft, G. B., Greaves, D. R. & Kollias, G. (1987) Cell 51, 975-985. [DOI] [PubMed] [Google Scholar]
- 33.Tuan, D. & London, I. M. (1984) Proc. Natl. Acad. Sci. USA 81, 2718-2722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Forrester, W. C., Thompson, C., Elder, J. T. & Groudine, M. (1986) Proc. Natl. Acad. Sci. USA 83, 1359-1363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tolhuis, B., Palstra, R. J., Splinter, E., Grosveld, F. & de Laat, W. (2002) Mol. Cell 10, 1453-1475. [DOI] [PubMed] [Google Scholar]
- 36.Carter, D., Chakalova, L., Osborne, C. S., Dai, Y. F. & Fraser, P. (2002) Nat. Genet. 32, 623-626. [DOI] [PubMed] [Google Scholar]
- 37.Drissen, R., Palstra, R. J., Gillemans, N., Splinter, E., Grosveld, F., Philipsen, S. & de Laat, W. (2004) Genes Dev. 18, 2485-2490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Vakoc, C. R., Letting, D. L., Gheldof, N., Sawado, T., Bender, M. A., Groudine, M., Weiss, M. J., Dekker, J. & Blobel, G. A. (2005) Mol. Cell 17, 453-462. [DOI] [PubMed] [Google Scholar]
- 39.Miller, I. J. & Bieker, J. J. (1993) Mol. Cell. Biol. 13, 2776-2786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gillemans, N., Tewari, R., Lindeboom, F., Rottier, R., de Wit, T., Wijgerde, M., Grosveld, F. & Philipsen, S. (1998) Genes Dev. 12, 2863-2873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bieker, J. J. (2001) J. Biol. Chem. 276, 34355-34358. [DOI] [PubMed] [Google Scholar]
- 42.Coghill, E., Eccleston, S., Fox, V., Cerruti, L., Brown, C., Cunningham, J., Jane, S. & Perkins, A. (2001) Blood 97, 1861-1868. [DOI] [PubMed] [Google Scholar]
- 43.Brown, R. C., Pattison, S., van Ree, J., Coghill, E., Perkins, A., Jane, S. M. & Cunningham, J. M. (2002) Mol. Cell. Biol. 22, 161-170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Perkins, A. C., Sharpe, A. H. & Orkin, S. H. (1995) Nature 375, 318-322. [DOI] [PubMed] [Google Scholar]
- 45.Nuez, B., Michalovich, D., Bygrave, A., Ploemacher, R. & Grosveld, F. (1995) Nature 375, 316-318. [DOI] [PubMed] [Google Scholar]
- 46.Wijgerde, M., Gribnau, J., Trimborn, T., Nuez, B., Philipsen, S., Grosveld, F. & Fraser, P. (1996) Genes Dev. 10, 2894-2902. [DOI] [PubMed] [Google Scholar]
- 47.Zhang, W. & Bieker, J. J. (1998) Proc. Natl. Acad. Sci. USA 95, 9855-9860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Armstrong, J. A., Bieker, J. J. & Emerson, B. M. (1998) Cell 95, 93-104. [DOI] [PubMed] [Google Scholar]
- 49.Zhang, W., Kadam, S., Emerson, B. M. & Bieker, J. J. (2001) Mol. Cell. Biol. 21, 2413-2422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Weiss, M. J., Yu, C. & Orkin, S. H. (1997) Mol. Cell. Biol. 17, 1642-1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Johnson, K. D., Christensen, H. M., Zhao, B. & Bresnick, E. H. (2001) Mol. Cell 8, 465-471. [DOI] [PubMed] [Google Scholar]
- 52.Im, H., Grass, J. A., Christensen, H. M., Perkins, A. & Bresnick, E. H. (2002) Biochemistry 41, 15152-15160. [DOI] [PubMed] [Google Scholar]
- 53.Grass, J. A., Boyer, M. E., Paul, S., Wu, J., Weiss, M. J. & Bresnick, E. H. (2003) Proc. Natl. Acad. Sci. USA 100, 8811-8816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gregory, R. C., Taxman, D. J., Seshasayee, D., Kensinger, M. H., Bieker, J. J. & Wojchowski, D. M. (1996) Blood 87, 1793-1801. [PubMed] [Google Scholar]
- 55.Merika, M. & Orkin, S. H. (1995) Mol. Cell. Biol. 15, 2437-2447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kadam, S., McAlpine, G. S., Phelan, M. L., Kingston, R. E., Jone, K. A. & Emerson, B. M. (2000) Genes Dev. 14, 2441-2451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Imbalzano, A. N. & Xiao, H. (2004) Adv. Protein Chem. 67, 157-179. [DOI] [PubMed] [Google Scholar]
- 58.Wang, W. (2003) Curr. Top. Microbiol. Immunol. 274, 143-169. [DOI] [PubMed] [Google Scholar]
- 59.Forsberg, E. C., Downs, K. M. & Bresnick, E. H. (2000) Blood 96, 334-339. [PubMed] [Google Scholar]
- 60.Sawado, T., Igarashi, K. & Groudine, M. (2001) Proc. Natl. Acad. Sci. USA 98, 10226-10231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Brand, M., Ranish, J. A., Kummer, N. T., Hamilton, J., Igarashi, K., Francastel, C., Chi, T. H., Crabtree, G. R., Aebersold, R. & Groudine, M. (2003) Nat. Struct. Mol. Biol. 11, 73-80. [DOI] [PubMed] [Google Scholar]
- 62.Johnson, K. D., Grass, J. A., Im, H., Park, C., Choi, K. & Bresnick, E. H. (2003) Mol. Cell. Biol. 23, 6484-6493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Letting, D. L., Rakowski, C., Weiss, M. J. & Blobel, G. A. (2003) Mol. Cell. Biol. 23, 1334-1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kiekhaefer, C. M., Grass, J. A., Johnson, K. D., Boyer, M. E. & Bresnick, E. H. (2002) Proc. Natl. Acad. Sci. USA 99, 14309-14314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Stamatoyannopoulos, J. A., Goodwin, A., Joyce, T. & Lowrey, C. H. (1995) EMBO J. 14, 106-116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bresnick, E. H., Bustin, M., Marsaud, V., Richard-Foy, H. & Hager, G. L. (1992) Nucleic Acids Res. 20, 273-278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Cirillo, L., Lin, F. R., Cuesta, I., Friedman, D., Jarnik, M. & Zaret, K. S. (2002) Mol. Cell 9, 279-289. [DOI] [PubMed] [Google Scholar]
- 68.Lee, D. Y., Hayes, J. J., Pruss, D. & Wolffe, A. P. (1993) Cell 72, 73-84. [DOI] [PubMed] [Google Scholar]
- 69.van Leeuwen, F., Gafken, P. R. & Gottschling, D. E. (2002) Cell 109, 745-756. [DOI] [PubMed] [Google Scholar]
- 70.Im, H., Park, C., Feng, Q., Johnson, K. D., Kiekhaefer, C. M., Choi, K., Zhang, Y. & Bresnick, E. H. (2003) J. Biol. Chem. 278, 18346-18352. [DOI] [PubMed] [Google Scholar]
- 71.Jimenez, G., Griffiths, S. D., Ford, A. M., Greaves, M. F. & Enver, T. (1992) Proc. Natl. Acad. Sci. USA 89, 10618-10622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Forsberg, E. C., Downs, K. M., Christensen, H. M., Im, H., Nuzzi, P. A. & Bresnick, E. H. (2000) Proc. Natl. Acad. Sci. USA 97, 14494-14499. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.