Abstract
Background
The aberrant cell-cycle progression of HIV-1-infected kidney cells plays a major role in the pathogenesis of HIV-associated nephropathy, however the mechanisms whereby HIV-1 induces infected glomerular podocytes or infected tubular epithelium to exit quiescence are largely unknown. Here, we ask whether the expression of HIV-1 genes in infected podocytes induces cyclin D1 and phospho-pRb (Ser780) expression, hallmarks of cyclin D1-mediated G1 → S phase progression.
Results
We assessed cyclin D1 and phospho-pRb (Ser780) expression in two well-characterized models of HIV-associated nephropathy pathogenesis: HIV-1 infection of cultured podocytes and HIV-1 transgenic mice (Tg26). Compared to controls, cultured podocytes expressing HIV-1 genes, and podocytes and tubular epithelium from hyperplastic nephrons in Tg26 kidneys, had increased levels of phospho-pRb (Ser780), a target of active cyclin D1/cyclin-dependent kinase-4/6 known to promote G1 → S phase progression. HIV-1-infected podocytes showed markedly elevated cyclin D1 mRNA and cyclin D1 protein, the latter of which did not down-regulate during cell-cell contact or differentiation, suggesting post-transcriptional stabilization of cyclin D1 protein levels by HIV-1. The selective suppression of HIV-1 transcription by the cyclin-dependent kinase inhibitor, flavopiridol, abrogated cyclin D1 expression, underlying the requirement for HIV-1 encoded products to induce cyclin D1. Indeed, HIV-1 virus deleted of nef failed to induce cyclin D1 mRNA to the level of other single gene mutant viruses.
Conclusions
HIV-1 expression induces cyclin D1 and phospho-pRb (Ser780) expression in infected podocytes, suggesting that HIV-1 activates cyclin D1-dependent cell-cycle mechanisms to promote proliferation of infected renal epithelium.
Background
Proliferation and dedifferentiation of HIV-1 infected renal epithelium is singularly unique compared to the effects of HIV-1 infection in other non-lymphoid tissues. Indeed, unlike the atrophy and cell-death of infected central nervous system [1], cardiac [2], or gastro-intestinal [3] parenchyma, aberrant cell-cycle progression of infected renal epithelium is a defining feature of HIV-associated nephropathy (HIVAN) [4,5]. Yet, the mechanisms are largely unknown whereby HIV-1 subverts the cell-cycle controls that normally maintain renal epithelial in G0. Nonetheless, several observations from in vitro and in vivo models of HIVAN pathogenesis suggest that HIV-1 expression itself may be a key pathogenic factor. Infection of cultured podocytes with replication incompetent HIV-1 demonstrated that HIV-1 gene expression leads to the proliferation and dedifferentiation of the infected podocytes [6-8], recapitulating the phenotypes observed in HIVAN specimens [9,10]. Similarly, renal expression of replication incompetent HIV-1 proviral transgenes in mice and rats produces a proliferative renal lesion that is indistiguisable both histopathologically and clinically from human HIVAN [11-14]. These observations raise the possibility that HIV-1 gene products may interact with mitogenic pathways in infected renal epithelium to cause aberrant cell-cycle engagement.
In comparison to the two general alternative mechanisms utilized by known transforming viruses to promote cell-cycle progression, namely, by activating or bypassing endogenous D-type cyclins (herein, referred to as "cyclin D"), it has not been established whether HIV-1 gene products trigger either cyclin D-dependent or cyclin D-independent proliferation in non-lymphoid tissues [15]. In infected renal epithelium, HIV-1 could disrupt the inhibitory binding of pRb to E2F independent of endogenous cyclin D, analogous to transforming viral mechanisms that bypass and down-regulate endogenous cyclin D, such as SV-40 T antigen binding to pRb [16], HHV-8 v-cyclin binding to and activating cyclin-dependent kinase-6 (CDK) [17], and activation of cyclin E by EBV-induced c-myc [18]. Alternatively, HIV-1 could activate cyclin D-dependent proliferation, as exemplified by RSV v-src-mediated signaling [19]. In most tissues, the selective activation of these cell-cycle pathways is readily discernable. Indeed, if cells depend on cyclin D-mediated G1 → S transition, passage from G0 through the restriction point in G1 is dependent on the de novo expression of cyclin D transcript, accumulation of nuclear cyclin D protein, and cyclin D/CDK-4/6 phosphorylation of pRb [20]. Then, although nuclear-localized cyclin D decreases in S phase in individual cells [21], total cyclin D in asynchronously proliferating cell populations remain elevated compared to quiescent cells [20]. In renal epithelial and mesenchymal cells, cyclin D1 is the dominant D-type cyclin activated during cell-cycle progression, both during nephrogenesis [22] and in non-viral-mediated kidney cell proliferation [23].
Here, we ask whether infected podocytes expressing HIV-1 genes show the hallmarks of cyclin D1-dependent proliferation, specifically the up-regulation of cyclin D1 transcript and protein expression and the subsequent phosphorylation of pRb on serine 780, a target site of active cyclin D1/CDK-4/6 that is not shared with cyclin E/CDK-2 complexes [24]. Utilizing two well-characterized models of HIVAN pathogenesis, HIV-1 transgenic mice [11-13] and HIV-1 infection of cultured podocytes [6-8] we find not only these hallmarks of cyclin D1-dependent proliferation, but also dysregulation of cyclin D1 expression that normally occurs with cell-cell contact and differentiation [25].
Results
Phospho-pRb (Ser780) expression in vivo
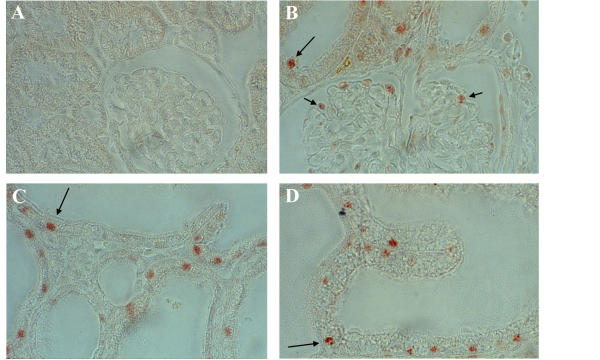
Cyclin D1 is characterized as the major D-type cyclin controlling G1-phase progression in kidney cells, both during normal and abnormal proliferation [22,23]. To determine whether cyclin D1 may be active in promoting G1 → S progression in kidneys expressing HIV-1 genes, we utilized a well-characterized HIV-1 transgenic mouse model (Tg26) of HIVAN [11-13]. Secondary to renal expression of the HIV-1 NL4-3 Δgag-pol proviral transgene, adolescent Tg26 transgenics develop a progressive, proliferative renal disease marked by podocyte and tubular epithelial hyperplasia that is indistinguishable both clinically and histopathologically from human HIVAN. In order to determine if cyclin D1 plays a role in this hyperplasia, kidneys from three proteinuric 40-day old transgenic siblings and three non-transgenic littermates were stained by immunohistochemistry with antibody to phospho-pRb (Ser780), a site of cyclin D1/CDK-4/6 phosphorylation not targeted by cyclin E/CDK-2 complexes that disrupts pRb/E2F binding for G1 → S progression [24]. Figure 1 depicts the intense, heterogeneous nuclear staining of glomerular podocytes and tubular epithelium from hyperplastic nephrons in the diseased transgenics versus the absent staining of nephrons in the non-transgenics consistent with the very low mitotic index in normal kidneys of adult animals [26]. This suggests that cyclin D1 is promoting cell-cycle progression during the proliferation of epithelium in diseased kidneys.
Figure 1.
In situ immunodetection of phosphorylated serine 780 on pRb in Tg26 kidneys. Phospho-pRb (Ser780) is not detected in the nuclei of epithelium of normal kidneys (A), but is readily detected in podocytes (short arrows) and tubular epithelial cells (long arrows) in hyperplastic nephrons of diseased transgenic kidneys at cortical (B), juxta-medullary (C), and medullary (D) depths. The prominent staining in many, but not all nuclei, is characteristic of asynchronously proliferating cells. (magnification = 40×)
Cyclin D1 and phospho-pRb (Ser780) expression in vitro
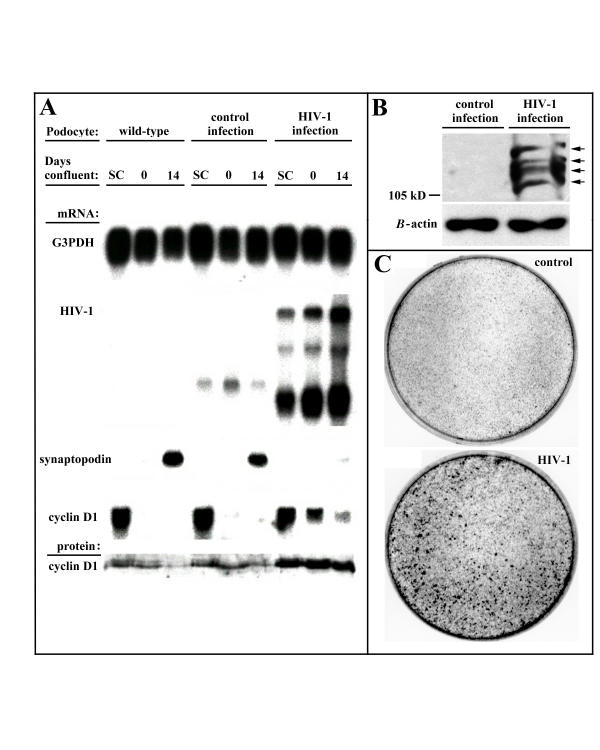
HIV-1 expression in cultured podocytes recapitulates the phenotypic abnormalities induced by HIV-1 expression in infected podocytes in vivo, specifically proliferation, loss of contact-mediated growth control, and dedifferentiation [6-8]. To determine if these phenotypes were marked by enhanced cyclin D1 and phospho-pRb (Ser780) expression, we utilized the podocyte cell-culture protocol developed by Rieser et al. [27,28] to study both sub-confluent podocyte growth and podocyte growth during the formation of mature cell-cell contacts at confluence. In this protocol, normal podocytes are growth-arrested 4–5 days after initial confluence and are fully differentiated with mature cell-cell contacts approximately 10 days later. Figure 2A shows that cyclin D1 message levels are nearly equivalent between mock ("wild-type")-, control virus-, and HIV-1-infected subconfluent podocytes, although only the HIV-1-infected subconfluent podocytes had elevated levels of cyclin D1 protein. In contrast, cell-cell contact in wild-type and control-infected, but not HIV-1-infected podocytes, caused a severe down-regulation of cyclin D1 transcript and protein concomitant with the up-regulation of synaptopodin, a podocyte differentiation marker [7,28], by the end of the 14-day differentiation period. Although HIV-1 infected podocytes showed some decrease in cyclin D1 message by the end of the differentiation period, cyclin D1 protein levels remained elevated, suggesting activation by HIV-1 of mechanisms that stabilize cyclin D1 protein levels. Moreover, HIV-1-infected podocytes, but not control-infected podocytes, had readily detectable phospho-pRb (Ser780) at the end of the differentiation period, suggesting that the stably up-regulated cyclin D1 protein remained active in promoting cell-cycle progression (Figure 2B). The ongoing proliferation of HIV-1-infected podocytes following cell-cell contact led to the formation of multiple foci (Figure 2C) with intense, heterogenous nuclear cyclin D1 immunofluorescence consistent with ongoing asynchronous proliferation in this population of cells (Figure 3). These data indicate that HIV-1 expression in infected podocytes induces cyclin D1 and phospho-pRb (Ser780) expression, hallmarks of cyclin D1-mediated G1 → S progression. The data, however, do not prove that cyclin D1 expression alone is sufficient to cause cell-cycle progression.
Figure 2.
Expression of cyclin D1 and phospho-pRb (Ser780) in HIV-1-infected podocytes. (A) Northern and western blot detection of cyclin D1 expression in wild-type, control-infected, and HIV-1-infected podocytes during subconfluent (SC) growth at 33°C and during 14 days of differentiation of confluent podocytes at 37°C shows that neither the cyclin D1 transcript nor the elevated cyclin D1 protein in HIV-1-infected podocytes (full-length, singly-spliced, and multiply spliced HIV-1 transcripts) are significantly down-regulated by podocyte cell-cell contact or by podocyte differentiation (synaptopodin transcript) when compared to wild-type or control-infected (one transcript from LTR sequences) podocytes. (B) Immunodetection of phospho-pRb (Ser780) on several phosphorylated forms (arrows) of pRb in the asynchronously proliferating, HIV-1-infected podocytes that is absent in the quiescent, control-infected podocytes after 14 days of differentiation at 37°C. (C) Abundant foci formation in confluent HIV-1-infected podocytes but not in confluent control-infected podocytes due to the ongoing proliferation of HIV-1-infected podocytes after cell-cell contact. Blots are representative of two independent experiments.
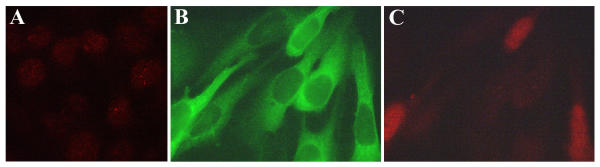
Figure 3.
Heterogenous nuclear localization of cyclin D1 in HIV-1-infected podocytes. Immunofluorescent detection of nuclear cyclin D1 in quiescent, wild-type podocytes after 14 days of differentiation at 37°C shows light speckled staining in all nuclei (A). This differs markedly from the intense nuclear staining of cyclin D1 in some, but not all nuclei, of the asynchronously proliferating, HIV-1-infected podocytes (GFP positive podocytes) (B and C). (magnification = 100×)
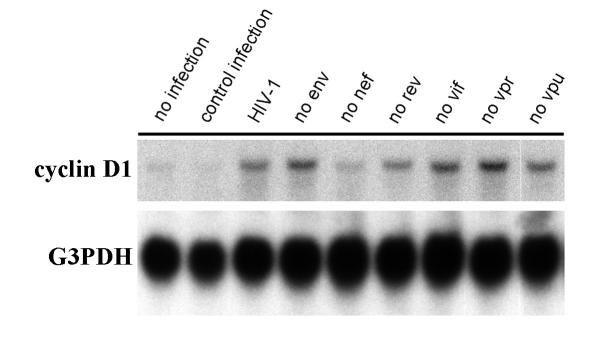
Requirement of HIV-1 expression for induction of cyclin D1
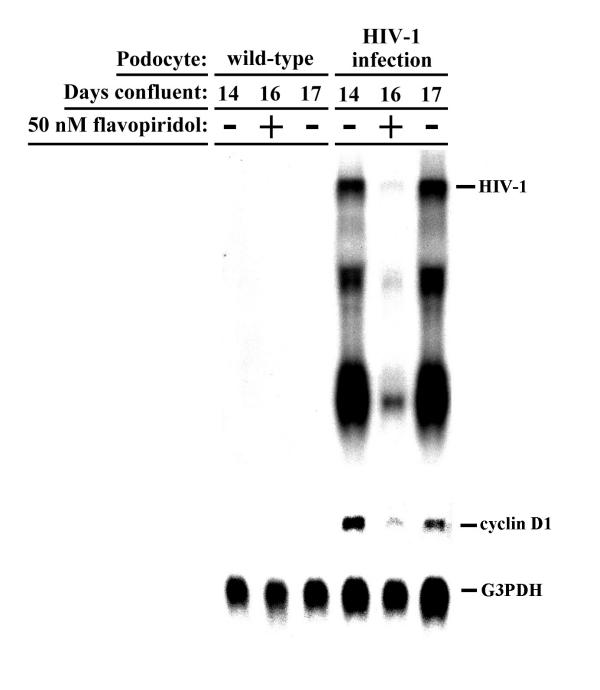
We sought to further confirm that HIV-1 expression, and not some idiosyncratic effect of infection, was directly inducing cyclin D1 expression in infected podocytes. First, we utilized the small molecule cyclin-dependent kinase inhibitor, flavopiridol, which at low concentrations (e.g., less than 100 nM) can selectively suppress HIV-1 transcript elongation without altering cellular transcripts in cell types of various lineages, including podocytes [7,29,30]. Cyclin D1 transcript levels were followed before, during, and after treatment of confluent wild-type or HIV-1 infected podocytes with 50 nM flavopiridol, a dose that suppresses HIV-1 transcription by 70% [7]. Analysis of HIV-1, cyclin D1, and glyceraldehyde-3-phosphate dehydrogenase (G3PDH) transcript levels in podocytes treated with flavopiridol for 2 days, followed by drug washout for 1 day, showed that cyclin D1 expression paralleled HIV-1 expression in infected podocytes before, during and after suppression of HIV-1 genes (Figure 4). Secondly, we utilized single gene mutants of HIV-1 [8] to determine which HIV-1 gene(s) may be involved with up-regulating cyclin D1 expression. Analysis of cyclin D1 transcript levels in podocytes infected with HIV-1 singly mutated with premature stop codons in the genes encoding the various structural and regulatory proteins of HIV-1 (except tat, which is present in every virus) showed that nef-deficient virus did not significantly up-regulate cyclin D1 expression when compared to the other mutant viruses (Figure 5). Taken together, these data indicate that induction of cyclin D1 expression requires HIV-1 gene expression and that HIV-1 nef is important for this induction.
Figure 4.
Parallel expression of HIV-1 and cyclin D1. Northern blot de tection of HIV-1, cyclin D1, and G3PDH transcripts before (day 14), during (two days with drug, day 16), and after (one day after drug washout, day 17) treatment of wild-type and HIV-1-infected podocytes with 50 nM flavopiridol, a small molecule CDK inhibitor of HIV-1 transcription, showing that cyclin D1 expression parallels HIV-1 expression. Blot is representative of two independent experiments.
Figure 5.
Mapping of HIV-1 genes that induce cyclin D1 expression. Northern blot detection of cyclin D1 and G3PDH transcripts from wild-type, control-infected, and HIV-1-infected podocytes infected with single gene mutants of HIV-1 (tat present in every virus). Similar to wild-type and control-infected podocytes, podocytes infected with nef-deficient HIV-1 do not significantly up-regulated cyclin D1 expression compared to podocytes infected with other single gene mutant viruses.
Discussion
The mechanisms whereby HIV-1 subverts cell-cycle controls in infected renal epithelium have been unclear. Here, we demonstrate that podocytes expressing HIV-1 genes display the hallmarks of cyclin D1-dependent cell-cycle progression, specifically, that cyclin D1 transcript and protein expression are markedly up-regulated, and that levels of phospho-pRb (Ser780), a target of cyclin D1/CDK-4/6 but not cyclin E/CDK-2 complexes [24], are induced in vitro and in vivo. In addition, preliminary micro-array gene profiling of HIV-1-infected tubular epithelium showed that cyclin D1 expression is up-regulated several fold when compared to uninfected tubular epithelium (Michael Ross, personal communication). This suggests that the HIV-1-induced proliferation of infected renal epithelium may be caused by a cyclin D1-dependent mechanism, unlike other viruses that bypass a requirement for G1-phase cyclins to trigger aberrant cell-cycle progression. Moreover, normal cellular controls on cell-cycle progression appears to be dysregulated by HIV-1 expression because cell-cell contact and differentiation, events that normally lead to the down-regulation of cyclin D1 expression [25], did not significantly alter cyclin D1 protein levels in HIV-1 infected podocytes. This suggests a novel stabilization of cyclin D1 protein in podocytes by HIV-1. We are exploring whether this occurs from enhanced translation of cyclin D1 message and/or decreased degradation of cyclin D1 protein and whether other D-type cyclins may be involved in phosphorylating pRb in HIVAN.
Prior studies in HIV-1 transgenic animals support the model that HIV-1 gene products interact with endogenous mitogenic pathways in infected renal epithelium. [11-14]. Kidney transplants between normal and transgenic Tg26 siblings suggested that specific HIV-1 proteins within the kidney, and not circulating factors, transform infected renal epithelium [13]. Indeed, HIV-1 transgenic mice generated with an HIV-1 proviral construct mutated in the Nef "PXXP" SH3-binding motif for Src-family kinases failed to develop nephropathy [31], and a recent in vitro analysis of podocytes infected with single gene mutants of HIV-1 further suggest that Nef plays a central role in causing the proliferative phenotype in HIVAN [8]. Utilizing these same single gene mutant viruses, we showed here that Nef appears to be play a role in the up-regulation of cyclin D1 by HIV-1 in vitro. Importantly, these single gene mutants do not exclude the possibility that other HIV-1 gene(s) may cooperate with nef to induce cyclin D1, particularly tat, which is present in every virus. Cumulatively, however, these observations do suggest a mechanism whereby HIV-1 expression in renal epithelium may induce cyclin D1: activation of Src-family kinases by Nef would be expected to up-regulate cyclin D1 gene expression and promote the synthesis and stabilization of cyclin D1 protein via Src-Ras-MAPK and Src-PI3K-mTOR signal transduction pathways, respectively [32-34]. Interestingly, our unpublished in vitro observations and published data showing marked expression of basic fibroblast growth factor in the interstitium of HIVAN kidneys [35] suggest that soluble mitogens may be important cofactors with HIV-1 expression to induce cyclin D1. Thus, interrupting HIV-1 gene expression should down-regulate cyclin D1 and correct the phenotypic abnormalities in infected podocytes. In support of this notion, we show here that cyclin D1 expression parallels HIV-1 expression before, during, and after suppression of HIV-1 transcription with flavopiridol, and recently, we showed that longer periods of treatment with flavopiridol ameliorate the phenotypic abnormalities of infected podocytes [7].
Conclusions
The following results of this study suggest that HIV-1 expression leads to cyclin D1-mediated G1 → S progression in infected podocytes: cyclin D1 transcript and protein levels are markedly up-regulated, phospho-Rb (Ser780) levels are increased, cyclin D1 protein and phospho-Rb (Ser780) levels do not decrease with podocyte cell-cell contact and differentiation, and the up-regulation of cyclin D1 requires the expression of HIV-1 genes, particularly HIV-1 nef. In sum, these results suggest HIV-induced cell-cycle mechanisms may contribute to aberrant epithelial proliferation in HIVAN.
Methods
Podocyte cell culture and infection
Murine podocytes immortalized with interferon-inducible temperature-sensitive SV-40 T antigen (ts-SV-40 T) were isolated and infected with replication incompetent GFP-expressing HIV-1 virus, replication incompetent GFP-expressing single-gene mutants of HIV-1, or replication incompetent GFP-expressing control lentivirus containing GFP but no HIV-1 genes as previously described [7,8]. In all cases, 70% to 80% of podocytes showed GFP expression 4 days post infection (data not shown). Podocytes were used before passage twenty and maintained in RPMI 1640 (Life Technologies) supplemented with 10% fetal calf serum, 1 × PenStrep (Life Technologies), and 20 U/ml recombinant interferon-gamma (Life Technologies) at 33°C. Unless otherwise indicated, all studies were conducted at cell confluency and 14 days after inactivation of ts-SV-40 T at the non-permissive temperature (37°C) when wild-type podocytes are fully differentiated [27,28].
Immunohistochemistry
Kidneys from three 40 day-old Tg26 heterozygous transgenic siblings demonstrating 2+ proteinuria (Chemstrip 2GP, Roche) and three of their non-transgenics littermates were collected in accordance with Animal Use and Care Committee guidelines, fixed (10% buffered formalin), then paraffin-embedded (Department of Pathology, Mount Sinai School of Medicine). Four micron full coronal kidney sections from each animal were adhered to lysine-coated slides and used for immunodetection of phospho-Rb (Ser780) as follows: sections were deparaffinized and hydrated by placing in xylene for 8 minutes, followed by 3 minutes each of 100%, 96%, 80%, 70%, 50%, 30% ethanol, ending with 10 minutes in water; sections then underwent antigen retrieval (Glyca Reagent, Biogenex) followed by immunodetection (Super Sensitive Detection Kit, Biogenex) of phospho-Rb (Ser780) after a 12 hour incubation with a 1:1000 dilution of rabbit anti-phospho-Rb (Ser780) antibody (Cell Signaling) or control rabbit anti-sera (Biogenex) at 4°C.
Immunofluorescence
Following the 14 day podocyte differentiation protocol on coverslips to establish mature podocytes, wild-type and HIV-1 infected podocytes underwent immunofluorescent detection of cyclin D1 as follows: coverslips were washed three times with 1 × PBS, fixed in 60% acetone/3.7% formaldehyde at -20°C for 20 minutes, then washed twice with 1 × PBS; the coverslips where then incubated with a 1:200 dilution of rabbit anti-cyclin D1 antibody (Santa Cruz Biotechnology) or control rabbit sera (Biogenex) for 2 hours at room temperature followed by three washes with 1 × PBS; the coverslips were then incubated with a 1:250 dilution of rhodamine-labeled donkey-anti-rabbit anti-sera (Chemicon) for 45 minutes to detect the primary antibody; following three final washes with 1 × PBS, the coverslips were mounted on glass slides using ProLong Antifade (Molecular Probes) for fluorescent detection of GFP and cyclin D1 on an Olympus IX70 microscope at 100 × magnification.
Podocyte cell-cell contact
Total RNA and protein were extracted from wild-type, control-infected, and HIV-1 infected podocytes during the Reiser, et al. protocol to study subconfluent podocyte growth and podocyte growth during establishment of mature cell-cell contacts between differentiated podocytes [27,28]. Briefly, RNA and protein were extracted from parallel plates of podocytes using TRIZOL Reagent or RIPA buffer, respectively, under three conditions: 1) during sub-confluent growth at 33°C; 2) immediately upon reaching confluency (day zero); and 3) on day 14 after switching confluent cultures to 37°C. At the end of the differentiation protocol (day 14), additional plates were stained to detect foci formation as follows: cells were washed with 1 × PBS, fixed in methanol for 1 minute, stained with bromophenol blue for 1 minute, then washed again with 1 × PBS. In experiments utilizing the single gene mutant of HIV-1, total RNA was extract from infected podocytes 14 days after switching confluent cultures to 37°C. All experiments were performed twice.
Flavopiridol treatment
After the 14-day podocyte differentiation protocol described above, total RNA was extracted from wild-type and HIV-1-infected podocytes before treatment with flavopiridol (Developmental Therapeutics Program, National Cancer Institute) (day 14), two days after suppression of HIV-1 transcription with media containing 50 nM flavopiridol (day 16), and one day after drug washout using media without flavopiridol (day 17). The experiment was performed twice.
Cyclin D1 transcript and protein
RNA and protein samples were analyzed as follows: Podocyte RNA (10 μg) was analyzed by northern blot using probes for HIV-1 nef-LTR[7], mouse cyclin D1 (cDNA generated by RT-PCR from mouse podocytes and verified by sequence analysis), synaptopodin [7], and G3PDH. Following 7.5% SDS/PAGE, podocyte protein (20 μg) was analyzed by western blot for cyclin D1 (ECL detection of a 1:1000 dilution of rabbit anti-cyclin D1 antibody, Santa Cruz BioTechnology). Protein (250 μg) from the control-infected or HIV-1-infected podocytes on day 14 of the differentiation protocol was also analyzed by western blot for total pRb (ECL detection of a 1:1000 dilution of rabbit anti-Rb, Santa Cruz Biotechnology) and phospho-Rb (Ser780) (ECL detection of a 1:500 dilution of rabbit anti-phospho-Rb (Ser780) antibody binding, Cell Signaling). Protein concentrations were determined using the Bio-Rad Protein Assay (Bio-Rad Laboratories), and blotting of equivalent amounts of protein was confirmed by transient staining of membranes with Ponceau S solution (0.5% Ponceau S/1% acetic acid) and by western blot for β-actin (Chemicon). Signal intensity was calculated using the UN-SCAN-IT gel v4.3 (Silk Scientific).
Authors' contributions
P.J.N. conceived and designed the study; performed the immunohistochemistry, immunofluorescence, and western blotting; and drafted the manuscript. M.S. cultured the podocytes and performed the northern blotting. M.H. designed and prepared the single gene mutants of HIV-1. I.H.G. mentored the authors and participated in the study's design and coordination.
Acknowledgments
Acknowledgements
We thank Paul Klotman for the use of podocyte cell-lines, HIV-1 constructs, and Tg26 mice. This work was supported by a National Kidney Foundation Research Award (P.J.N) and by the NIH grant DK 56492–02.
Contributor Information
Peter J Nelson, Email: peter.nelson@mssm.edu.
Masaaki Sunamoto, Email: masaaki.sunamoto@mssm.edu.
Mohammad Husain, Email: mohammad.husain@mssm.edu.
Irwin H Gelman, Email: irwin.gelman@mssm.edu.
References
- Gray F, Adle-Biassette H, Chretien F, Lorin de la Grandmaison G, Force G, Keohane C. Neuropathology and neurodegeneration in human immunodeficiency virus infection. Pathogenesis of HIV-induced lesions of the brain, correlations with HIV-associated disorders and modifications according to treatments. Clin Neuropathol. 2001;20:146–55. [PubMed] [Google Scholar]
- Barbaro G, Di Lorenzo G, Grisorio B, Barbarini G. Incidence of dilated cardiomyopathy and detection of HIV in myocardial cells of HIV-positive patients. Gruppo Italiano per lo Studio Cardiologico dei Pazienti Affetti da AIDS. N Engl J Med. 1998;339:1093–9. doi: 10.1056/NEJM199810153391601. [DOI] [PubMed] [Google Scholar]
- Janoff EN, Smith PD. Emerging concepts in gastrointestinal aspects of HIV-1 pathogenesis and management. Gastroenterology. 2001;120:607–21. doi: 10.1053/gast.2001.22427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shankland SJ, Eitner F, Hudkins KL, Goodpaster T, D'Agati V, Alpers CE. Differential expression of cyclin-dependent kinase inhibitors in human glomerular disease: role in podocyte proliferation and maturation. Kidney Int. 2000;58:674–83. doi: 10.1046/j.1523-1755.2000.00213.x. [DOI] [PubMed] [Google Scholar]
- Barisoni L, Mokrzycki M, Sablay L, Nagata M, Yamase H, Mundel P. Podocyte cell cycle regulation and proliferation in collapsing glomerulopathies. Kidney Int. 2000;58:137–43. doi: 10.1046/j.1523-1755.2000.00149.x. [DOI] [PubMed] [Google Scholar]
- Schwartz EJ, Cara A, Snoeck H, Ross MD, Sunamoto M, Reiser J, et al. Human immunodeficiency virus-1 induces loss of contact inhibition in podocytes. J Am Soc Nephrol. 2001;12:1677–84. doi: 10.1681/ASN.V1281677. [DOI] [PubMed] [Google Scholar]
- Nelson PJ, Gelman IH, Klotman PE. Suppression of HIV-1 expression by inhibitors of cyclin-dependent kinases promotes differentiation of infected podocytes. J Am Soc Nephrol. 2001;12:2827–31. doi: 10.1681/ASN.V12122827. [DOI] [PubMed] [Google Scholar]
- Husain M, Gusella GL, Klotman ME, Gelman IH, Ross MD, Schwartz EJ, et al. HIV-1 Nef Induces Proliferation and Anchorage-Independent Growth in Podocytes. J Am Soc Nephrol. 2002;13:1806–15. doi: 10.1097/01.asn.0000019642.55998.69. [DOI] [PubMed] [Google Scholar]
- Barisoni L, Kriz W, Mundel P, D'Agati V. The dysregulated podocyte phenotype: a novel concept in the pathogenesis of collapsing idiopathic focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol. 1999;10:51–61. doi: 10.1159/000017097. [DOI] [PubMed] [Google Scholar]
- Barisoni L, Bruggeman LA, Mundel P, D'Agati VD, Klotman PE. HIV-1 induces renal epithelial dedifferentiation in a transgenic model of HIV-associated nephropathy. Kidney Int. 2000;58:173–81. doi: 10.1046/j.1523-1755.2000.00152.x. [DOI] [PubMed] [Google Scholar]
- Dickie P, Felser J, Eckhaus M, Bryant J, Silver J, Marinos N, et al. HIV-associated nephropathy in transgenic mice expressing HIV-1 genes. Virology. 1991;185:109–19. doi: 10.1016/0042-6822(91)90759-5. [DOI] [PubMed] [Google Scholar]
- Kopp JB, Klotman ME, Adler SH, Bruggeman LA, Dickie P, Marinos NJ, et al. Progressive glomerulosclerosis and enhanced renal accumulation of basement membrane components in mice transgenic for human immunodeficiency virus type 1 genes. Proc Natl Acad Sci U S A. 1992;89:1577–81. doi: 10.1073/pnas.89.5.1577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruggeman LA, Dikman S, Meng C, Quaggin SE, Coffman TM, Klotman PE. Nephropathy in human immunodeficiency virus-1 transgenic mice is due to renal transgene expression. J Clin Invest. 1997;100:84–92. doi: 10.1172/JCI119525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid W, Sadowska M, Denaro F, Rao S, Foulke J, Jr, Hayes N, et al. An HIV-1 transgenic rat that develops HIV-related pathology and immunologic dysfunction. Proc Natl Acad Sci U S A. 2001;98:9271–6. doi: 10.1073/pnas.161290298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frankel AD, Young JA. HIV-1: fifteen proteins and an RNA. Annu Rev Biochem. 1998;67:1–25. doi: 10.1146/annurev.biochem.67.1.1. [DOI] [PubMed] [Google Scholar]
- Lukas J, Muller H, Bartkova J, Spitkovsky D, Kjerulff AA, Jansen-Durr P, et al. DNA tumor virus oncoproteins and retinoblastoma gene mutations share the ability to relieve the cell's requirement for cyclin D1 function in G1. J Cell Biol. 1994;125:625–38. doi: 10.1083/jcb.125.3.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojala PM, Yamamoto K, Castanos-Velez E, Biberfeld P, Korsmeyer SJ, Makela TP. The apoptotic v-cyclin-CDK6 complex phosphorylates and inactivates Bcl-2. Nat Cell Biol. 2000;2:819–25. doi: 10.1038/35041064. [DOI] [PubMed] [Google Scholar]
- Santoni-Rugiu E, Falck J, Mailand N, Bartek J, Lukas J. Involvement of Myc activity in a G(1)/S-promoting mechanism parallel to the pRb/E2F pathway. Mol Cell Biol. 2000;20:3497–509. doi: 10.1128/MCB.20.10.3497-3509.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frame MC, Fincham VJ, Carragher NO, Wyke JA. v-Src's hold over actin and cell adhesions. Nat Rev Mol Cell Biol. 2002;3:233–45. doi: 10.1038/nrm779. [DOI] [PubMed] [Google Scholar]
- Ekholm SV, Reed SI. Regulation of G(1) cyclin-dependent kinases in the mammalian cell cycle. Curr Opin Cell Biol. 2000;12:676–84. doi: 10.1016/S0955-0674(00)00151-4. [DOI] [PubMed] [Google Scholar]
- Baldin V, Lukas J, Marcote MJ, Pagano M, Draetta G. Cyclin D1 is a nuclear protein required for cell cycle progression in G1. Genes Dev. 1993;7:812–21. doi: 10.1101/gad.7.5.812. [DOI] [PubMed] [Google Scholar]
- Park SK, Kim W, Lee H, Lee DY, Koh GY. Differential changes of cell cycle regulators and activities in kidneys during pre- and postnatal development. Nephron. 1999;81:334–41. doi: 10.1159/000045302. [DOI] [PubMed] [Google Scholar]
- Shankland SJ, Wolf G. Cell cycle regulatory proteins in renal disease: role in hypertrophy, proliferation, and apoptosis. Am J Physiol Renal Physiol. 2000;278:F515–29. doi: 10.1152/ajprenal.2000.278.4.F515. [DOI] [PubMed] [Google Scholar]
- Kitagawa M, Higashi H, Jung HK, Suzuki-Takahashi I, Ikeda M, Tamai K, et al. The consensus motif for phosphorylation by cyclin D1-Cdk4 is different from that for phosphorylation by cyclin A/E-Cdk2. Embo J. 1996;15:7060–9. [PMC free article] [PubMed] [Google Scholar]
- Nelson PJ, Daniel TO. Emerging targets: Molecular mechanisms of cell contact-mediated growth control. Kidney Int. 2002;61:99–105. doi: 10.1046/j.1523-1755.2002.0610s1099.x. [DOI] [PubMed] [Google Scholar]
- Pabst R, Sterzel RB. Cell renewal of glomerular cell types in normal rats. An autoradiographic analysis. Kidney Int. 1983;24:626–31. doi: 10.1038/ki.1983.203. [DOI] [PubMed] [Google Scholar]
- Reiser J, Kriz W, Kretzler M, Mundel P. The glomerular slit diaphragm is a modified adherens junction. J Am Soc Nephrol. 2000;11:1–8. doi: 10.1681/ASN.V1111. [DOI] [PubMed] [Google Scholar]
- Mundel P, Reiser J, Zuniga Mejia Borja A, Pavenstadt H, Davidson GR, Kriz W, et al. Rearrangements of the cytoskeleton and cell contacts induce process formation during differentiation of conditionally immortalized mouse podocyte cell lines. Exp Cell Res. 1997;236:248–58. doi: 10.1006/excr.1997.3739. [DOI] [PubMed] [Google Scholar]
- Chao SH, Fujinaga K, Marion JE, Taube R, Sausville EA, Senderowicz AM, et al. Flavopiridol inhibits P-TEFb and blocks HIV-1 replication. J Biol Chem. 2000;275:28345–8. doi: 10.1074/jbc.C000446200. [DOI] [PubMed] [Google Scholar]
- Chao SH, Price DH. Flavopiridol inactivates P-TEFb and blocks most RNA polymerase II transcription in vivo. J Biol Chem. 2001;276:31793–9. doi: 10.1074/jbc.M102306200. [DOI] [PubMed] [Google Scholar]
- Hanna Z, Weng X, Kay DG, Poudrier J, Lowell C, Jolicoeur P. The pathogenicity of human immunodeficiency virus (hiv) type 1 nef in cd4c/hiv transgenic mice is abolished by mutation of its sh3-binding domain, and disease development is delayed in the absence of hck. J Virol. 2001;75:9378–92. doi: 10.1128/JVI.75.19.9378-9392.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penuel E, Martin GS. Transformation by v-Src: Ras-MAPK and PI3K-mTOR mediate parallel pathways. Mol Biol Cell. 1999;10:1693–703. doi: 10.1091/mbc.10.6.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muise-Helmericks RC, Grimes HL, Bellacosa A, Malstrom SE, Tsichlis PN, Rosen N. Cyclin D expression is controlled post-transcriptionally via a phosphatidylinositol 3-kinase/Akt-dependent pathway. J Biol Chem. 1998;273:29864–72. doi: 10.1074/jbc.273.45.29864. [DOI] [PubMed] [Google Scholar]
- Hashemolhosseini S, Nagamine Y, Morley SJ, Desrivieres S, Mercep L, Ferrari S. Rapamycin inhibition of the G1 to S transition is mediated by effects on cyclin D1 mRNA and protein stability. J Biol Chem. 1998;273:14424–9. doi: 10.1074/jbc.273.23.14424. [DOI] [PubMed] [Google Scholar]
- Ray PE, Bruggeman LA, Weeks BS, Kopp JB, Bryant JL, Owens JW, Notkins AL, Klotman PE. bFGF and its low affinity receptors in the pathogenesis of HIV-associated nephropathy in transgenic mice. Kidney Int. 1994;46:759–72. doi: 10.1038/ki.1994.331. [DOI] [PubMed] [Google Scholar]