Abstract
It has long been known that leaf senescence can be induced in many plant species by detaching leaves and placing them in the darkness. It recently has been shown that entire Arabidopsis plants placed in the darkness are not induced to senesce, as judged by visible yellowing and certain molecular markers. Here, we show that when individual Arabidopsis leaves are darkened, but not when entire plants are darkened, senescence is induced in the covered leaves. This induction of senescence is highly localized. The phenomenon is leaf age dependent in that it occurs more rapidly and strongly in older leaves than in younger ones, as is the case with many forms of induced senescence. Whole adult plants placed in darkness, in contrast, show delayed senescence, although seedlings lacking primary leaves do not. These observations imply that the light status of the entire plant affects the senescence of individual leaves. A model summarizing the results is presented.
Leaf senescence is an active process regulated by exogenous and endogenous factors. An important exogenous factor is light. The interplay between light and senescence is complex, and many reports have been published describing both its senescence-inhibiting and -promoting qualities. The latter mostly have been reported in the context of relatively high light levels, and interpreted as the reactions of photosynthesis metabolically “aging” a leaf, and indirectly inducing its senescence (Biswal and Biswal, 1984; Noodén et al., 1996). Darkness is more commonly considered to be an inducer of senescence. Most of the work on darkness-induced senescence has been done on either detached adult leaves or the attached cotyledons or primary leaves of seedlings, however, which raises the question of the relationship of these results to the arguably more ecologically relevant system of shaded attached adult leaves. In fact, it has been shown in Arabidopsis that, by certain parameters, adult attached leaves are not induced to senesce when whole plants are placed in the darkness, although detached leaves are (Weaver et al., 1998).
Here, we show that in Arabidopsis leaf senescence is not induced but is in fact inhibited when whole plants are placed in the darkness, whereas in contrast it is strongly accelerated when individual leaves are darkened while the rest of the plant remains in the light. These results demonstrate that the light status of the rest of the plant influences the senescence progression of the individual leaf.
RESULTS
Senescence Is Induced When Individual Leaves Are Darkened, But Not When Whole Plants Are Darkened
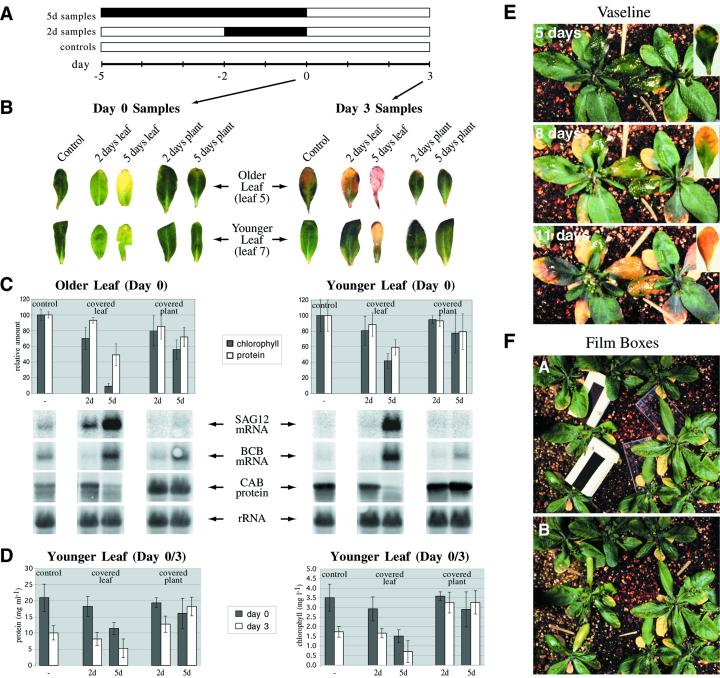
Whole Arabidopsis plants in their pots were placed in the darkness, or individual leaves were covered with cloth “mittens” (see “Materials and Methods”). Treatments were continued for either 2 or 5 d (control plants remained in continuous light) and all plants were harvested on the same day (d 0 in Fig. 1A). Consistent with previous results (Weaver et al., 1998), placing whole plants in the darkness did not induce senescence by most parameters measured: No visible yellowing occurred (Fig. 1B, d 0 samples; although treated leaves appeared pale relative to controls), the chlorophyll a/b-binding proteins (CAB) were not degraded (Fig. 1C), and SAG12 transcript, which is believed to be a good marker of senescence (Lohman et al., 1994; Weaver et al., 1998), was not induced (Fig. 1C). Total chlorophyll and protein levels did decline as a result of the treatment (although in younger leaves declines were slight at 2 d), but not as strongly as when individual leaves were covered (Fig. 1C). The mRNA levels of BCB, a senescence-associated gene also known to be induced by darkness (Van Gysel et al., 1993; Lohman et al., 1994; Weaver et al., 1998) and used here as a positive control for the dark treatment, did increase (Fig. 1C). The plants in general appeared as if they had largely ceased development in the darkness.
Figure 1.
A, Time course of the whole-plant verses individual leaf darkness experiments. White bars indicate time in light and dark bars time in darkness. A synchronously growing population of Arabidopsis was established. Five days before the first harvest (day = −5), 5-d dark-treated plants were placed in the darkness or leaves were covered, whereas 2 d before harvest (day = −2), 2-d dark-treated plants were placed in the darkness or leaves were covered. Controls were harvested at the same time and were the same age as the dark treated plants. A second harvest was performed 3 d after the dark-treated plants/leaves had been returned to the light (d 3), to assay recovery. B, Photographs of representative leaves at the time of harvest from the whole-plant verse individual leaf darkness experiment diagrammed in A. C, Analysis of total chlorophyll and protein levels and gene expression (RNA or protein blots) in individually darkened leaves versus darkened plants, from the experiment diagrammed in A (d-0 samples). SAG12 is a known specific senescence-associated gene. BCB is a gene known to be induced by both senescence and darkness. Chlorophyll a/b-binding protein (CAB) is known to be associated with photosynthesis. 18S ribosomal RNA (rRNA) is used as a loading control for the RNA samples. “Older” leaves are leaf 5. “Younger” leaves are leaf 7. Chlorophyll and protein measurements were normalized separately for older and younger leaves such that the control value (undarkened leaves) was always 100. D, Analysis of total chlorophyll and protein levels in darkened leaves versus darkened plants before (d 0) and after (d 3) a return to the light, from the experiment diagrammed in A. Individual leaves or entire plants were darkened for the indicated times (treatment ended at d 0; controls were never darkened), and then returned to the light for 3 d (d 3), and total chlorophyll and protein levels determined as described in “Materials and Methods.” Results shown are for leaf 7. E, Petroleum jelly-covered leaves were not induced to senesce. Leaf 5 was covered with petroleum jelly, which induced no visible senescence after 5 d (untreated control shown inset; petroleum jelly-covered leaves are indicated by toothpicks). Three and 6 d later (8 d and 11 d covered), both the petroleum jelly-covered and control leaves went on to senesce at a similar rate. F, Leaves covered by black boxes, but not clear boxes, are induced to senesce. Leaf 5 was covered with shallow boxes made of black or clear x-ray film. Leaves covered with the black boxes were induced to yellow, whereas those covered with clear boxes were not.
In contrast to the whole-plant dark treatment, senescence was in fact induced by all measured parameters when individual leaves were darkened. After 2 d, visible yellowing was observed (Fig. 1B, d 0 samples), total chlorophyll and protein levels had declined significantly (Fig. 1C), and SAG12 expression was induced (Fig. 1C). These effects were observed in both older and younger leaves. They were more pronounced in the older ones, however, indicating that, as with other inducers of senescence (e.g. ethylene; Grbic and Bleecker, 1995), endogenous factors (i.e. age) remain important in mediating the competence of the leaf to respond.
To address whether the senescence was in fact induced by the darkness itself, and not by some artifact of the experimental design, three controls were done. First, individual leaves were covered with mittens and then the whole plants were placed in the darkness. The covered leaves in this case behaved as uncovered leaves in darkness; i.e. the covered leaves were not induced to senesce (data not shown). This indicates that the mittens themselves do not induce senescence. A second possibility is that covered leaves have reduced stomatal conductance, and that it is reduced transpiration rather than darkness that is actually inducing senescence. To address this issue, leaves were covered with petroleum jelly on both adaxial and abaxial surfaces, a treatment that presumably eliminates transpiration. Figure 1E shows that 5 d after petroleum jelly covering, no visible yellowing was induced relative to controls (inset), and that, despite the covering, yellowing ultimately did occur at a rate similar to controls (8- and 11-d time points). This suggests that reduced transpiration does not induce senescence in this experimental system. Finally, leaves were covered not with cloth mittens but with small boxes made of either black or clear x-ray film. Leaves covered with black boxes were induced to yellow, whereas leaves covered with clear boxes were not (Fig. 1F). Thus, the only difference between the two treatments is likely to be the light received, which indicates that it is in fact darkness causing the senescence.
Darkness-Induced Senescence of Individual Leaves Is Not Reversed by a Return to Light, and Whole Dark-Treated Plants Show Delayed Senescence Relative to Non-Treated Controls
After the dark treatments (d 0 in Fig. 1A), plants/leaves that were not harvested were returned to the light, and harvested 3 d later to assay recovery (d 3 in Fig. 1A). Figure 1B (d 3 samples) shows representative leaves at this second harvest. Individually covered leaves did not recover when returned to the light, and in fact senesced further. Leaves from darkened plants, in contrast, appeared less senescent than individually darkened leaves and even less senescent than control leaves that had never seen darkness. This indicates that, rather than inducing senescence, whole-plant darkness actually delays it. Figure 1D compares total chlorophyll and protein levels at d 0 and 3 in leaf 7 (most formerly covered leaves 5 were dead, dry, and not harvested). It indicates that total chlorophyll and protein levels are higher in leaves of darkened plants returned to the light than in either individually darkened leaves returned to the light or controls left in continuous light, and are highest in the plants that were darkened for longest. In the case of the 2-d time point, chlorophyll and protein continued to decline in the formerly darkened plants in an age-dependent fashion after return to the light, but more slowly than in non-darkened controls. In the case of the 5-d time point, chlorophyll and protein levels increased slightly after return to the light. Chlorophyll and protein levels in darkened leaves, in contrast, declined after return to the light at a rate greater than controls, with the greatest decline in the leaves darkened for the longest. The data are consistent with the notion that whole plants placed in the darkness show inhibited development, including progression to senescence, with the result that leaves from darkened plants are effectively “younger” than control leaves. Thus, the “youngest” leaves of all are those from plants in the dark the longest (recall that the plants put in the dark the longest were put in the dark when they were youngest because all were removed on the same date and at the same age; see Fig. 1A). When individual leaves are darkened, in contrast, senescence is induced, and this induced senescence is not reversed after return to the light.
Whole-Plant Darkness Inhibits, But Does Not Block, Senescence in an Age-Dependent Manner
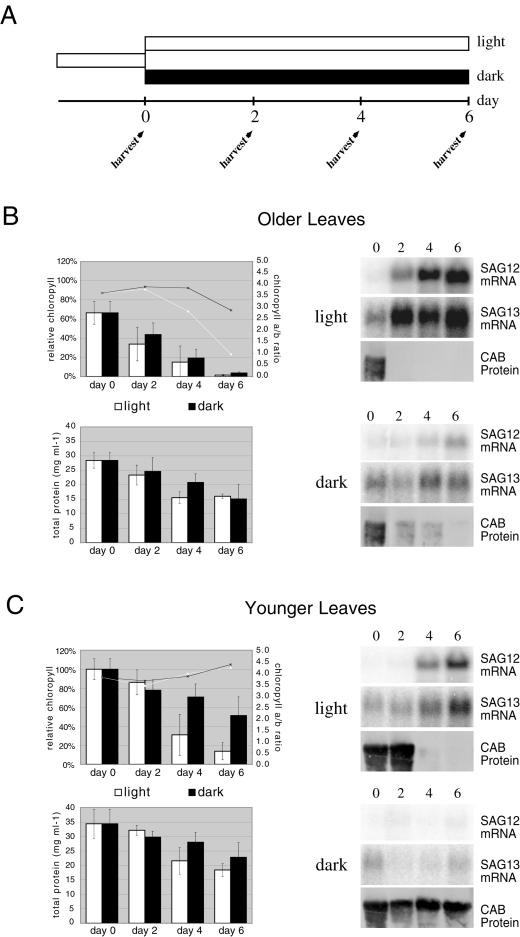
In the experiment depicted in Figure 1, A through D, plants were relatively young when they were first placed in the darkness (they had bolted, but had few or no siliques). To determine if older leaves would also show whole-plant, darkness-inhibited senescence, or perhaps even show a reversal of senescence already initiated, an experiment was done in which older plants (leaf 5 already showing early visible senescence at the onset of the experiment, and leaf 7 fully expanded but not visibly senescent) were transferred to the darkness at the same time and removed and harvested over a period of days (Fig. 2A). Control plants left in the light senesced as expected in an age-dependent fashion by all measured parameters: total protein and chlorophyll loss, loss of CAB protein, expression of SAG12 and SAG13 mRNAs, declines of chlorophyll a/b ratios in older leaves (Fig. 2, B and C), and leaf yellowing (data not shown). SAG13 is another molecular marker of senescence, known to be induced earlier in senescence than SAG12 (Lohman et al., 1994; Weaver et al., 1998). In plants that were transferred to darkness, all or most of those symptoms of senescence also occurred, but more slowly. In older darkened plants, SAG12 mRNA was only expressed at significant levels by d 6, whereas in younger darkened plants it was still not evident by d 6 (Fig. 2). In contrast, SAG12 expression became evident in older controls by d 2 and in younger controls by d 4. CAB protein behaved in an analogous, although opposite, manner: In older, darkened leaves, it remained present for 4 d longer than in non-darkened controls, whereas in younger darkened leaves it continued to be present throughout the course of the experiment. These results are consistent with the notion that whole-plant darkness delays, but does not prevent, senescence. However, it should be noted that in the course of the experiment SAG12 and SAG13 expression in the older darkened leaves never attained the high levels present in the older non-darkened controls (Fig. 2B), despite the fact that on the basis of appearance, total chlorophyll and protein levels, and CAB protein levels both were fully senescent by d 6. This might indicate that darkness inhibits certain aspects of senescence (such as SAG12 expression) more than others.
Figure 2.
Whole-plant darkness delays, but does not prevent, senescence. A, Time course of the experiment. White bars indicate time in light and dark bars time in darkness. A synchronously growing population of Arabidopsis was established, and when leaf 5 had begun to senesce but leaf 7 was still completely green, plants were transferred to the darkness (controls were left in the light). Plants were then harvested from both groups over a period of 6 d. B, Analysis of various parameters of senescence in “older leaves” (leaf 5). The upper graph shows both total chlorophyll levels (expressed as a percent of the level in the younger leaves at d 0; shown using bars), and chlorophyll a/b ratios (shown using lines). The lower graph shows total protein (also expressed as a percent of the level in the younger leaves at d 0). Blots shown are RNA or protein blots. SAG12 and SAG13 are both known senescence-associated genes, and SAG13 is known to be up-regulated during senescence earlier than SAG12. CAB is known to be associated with photosynthesis. C, As in B, but data shown are for “younger” leaves (leaf 7).
One difference between older and younger Arabidopsis plants is that older ones typically have many developing siliques, which act as sinks and in many species can induce leaf senescence. To investigate if the developing seed are responsible for the senescence that does continue to occur in older plants placed in the darkness, a similar experiment was conducted, in which flowering stalks (bolts) were removed before transfer to the darkness. The degree of inhibition of senescence appeared similar in those plants to controls in which the bolts were not removed, suggesting that sink effects do not play a large role in this phenomenon (data not shown). The removal of bolts may trigger other sinks, however (for instance, the release of lateral meristems), and so sink effects cannot be ruled out.
The “Individual Leaf” Darkness Response Is Localized
The above show that darkness delays senescence when delivered at the level of the whole plant, but promotes it when delivered at the level of the leaf. To examine if in the latter case the darkness is sensed at the level of the leaf or at a lower level (i.e. tissue or cellular), leaves were partially covered (with mittens with a hole punched in them). Yellowing occurred in all covered portions of the leaf but not beneath the hole (Fig. 3), indicating that the senescence response is highly localized, and possibly cell autonomous. These results are consistent with the observation that senescence is sometimes visibly induced when a portion of a leaf is covered by another leaf (which is what suggested these experiments to begin with). These results are also consistent with those of Rousseaux et al., in which far-red light-induced chlorophyll loss is also shown to be very localized (Rousseaux et al., 1997).
Figure 3.
Darkness-induced promotion of senescence is very localized. Leaves were covered with mittens in which a hole had been punched (A). After 5 d, the darkened areas of the leaf had gone on to senesce, but the areas that received light did not (B).
Whole-Plant, Dark Inhibition of Senescence Is Not Observed in Cotyledons, Which Do Senesce in the Darkness, But Is Observed in Primary Leaves
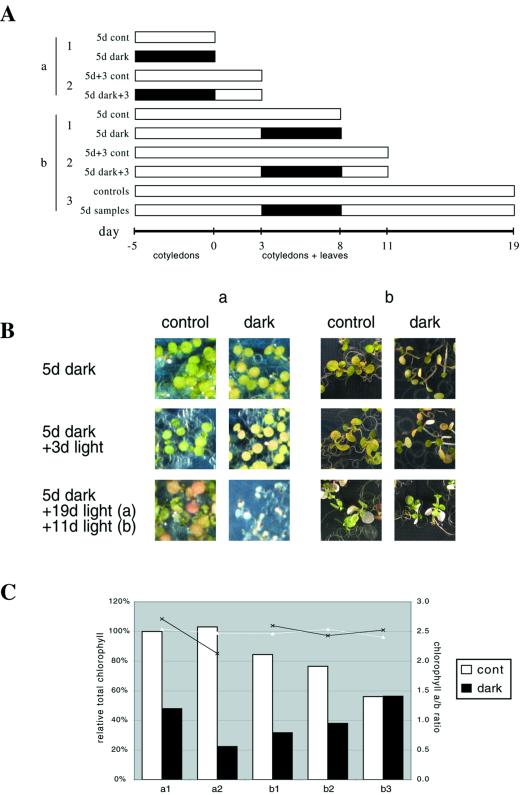
Much work has been done in other species indicating that cotyledon senescence is induced, not repressed, when whole seedlings are placed in the darkness (e.g. Biswal and Biswal, 1984). Because those results would appear to contradict ours, we also assayed the effects of darkness on the cotyledons of intact Arabidopsis seedlings placed in the darkness. An experiment was done in which individuals from a synchronously growing population of culture-grown seedlings were placed in the darkness at two separate times. At the first time (day −5 in Fig. 4A; experiment “a”) seedlings were at a stage when only cotyledons were present (i.e. visible primary leaves had yet to appear in most individuals). At the second time (d 3 in Fig. 4A; experiment “b”), both cotyledons and the first two primary leaves were evident.
Figure 4.
Darkened seedlings. A, Time course of the experiments. Seedlings were grown on plates, and transferred to darkness and returned to the light as indicated. White segments indicate time in light and dark segments indicate time in darkness. Each bar represents a sample that was harvested at the indicated time. The first group of seedlings to be placed in the dark (a) had only cotyledons. The second group (b) had both cotyledons and primary leaves. B, Photographs of representative seedlings. The bottom row were all taken on d 19, when the “a” samples had been returned to light for 19 d and the “b” samples had been returned to light for 11 d. C, Total chlorophyll (expressed as a percent of d-0 controls) and chlorophyll a/b ratios of the seedlings.
In the first experiment, in accord with what has been reported in other species, we observed senescence (assayed at the level of visible yellowing and chlorophyll loss) to be induced in cotyledons when whole seedlings were placed in the darkness (Fig. 4, B a and C a1). This senescence was not reversed by a return to the light, but rather accelerated. By 3 d after return to the light, cotyledons were very yellow and both total chlorophyll and the chlorophyll a/b ratio had declined (Fig. 4, B a and C a2), and within a few more days almost all the seedlings had died (Fig. 4B). These results were similar to those obtained when individual adult leaves were darkened and then returned to the light (see Fig. 1, B and D).
When older seedlings, comprising both cotyledons and primary leaves, were treated similarly, cotyledons appeared to respond similarly, yellowing as a result of the darkness and then further yellowing and ultimately dying upon return to the light (Fig. 4B b). Primary leaves, however, behaved differently. The primary leaves, and the rest of the seedling, appeared etiolated upon removal from the darkness, but not yellow, and both resumed growth and development after return to the light. Figure 4C (b1) indicates that chlorophyll levels did drop considerably following the dark treatment (presumably at least in part because cotyledons were senescing) but then increased upon return to the light (Fig. 4C, b2 and b3). Chlorophyll a/b ratios remained essentially constant throughout (Fig. 4C, b1–b3). By the final time point, chlorophyll levels in both dark-treated plants and controls (neither of which any longer had cotyledons) were similar. The chlorophyll data for this experiment are similar to that obtained when whole plants were darkened and returned to the light (see Fig. 1D), and are consistent with the notion that primary leaves, unlike cotyledons, are not induced to senesce by whole-plant darkness.
Covered Leaf Senescence Occurs in Various Photoreceptor Mutants
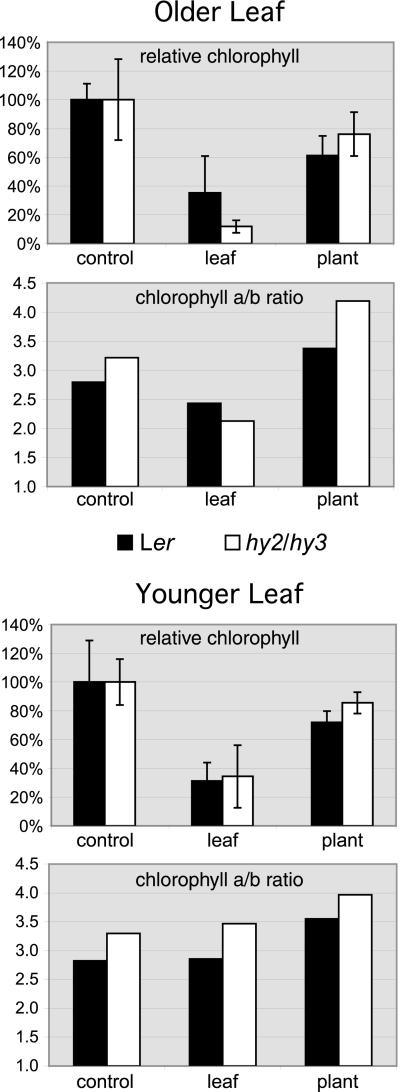
Previous work has appeared to implicate both phytochrome and/or blue light in darkness-induced senescence (Biswal and Biswal, 1984; Biswal and Choudhury, 1986; Guiamet et al., 1989; Rousseaux et al., 1996), and for this reason covered-leaf senescence was examined in several light-sensing mutants, in an attempt to explore the genetic basis of the phenomenon. hy2 is a phytochrome chromophore mutant in which all phytochrome responses are severely attenuated (Koornneef et al., 1980; Parks and Quail, 1991), and hy3 is a phytochrome B mutant (Koornneef et al., 1980; Somers et al., 1991). An hy2/hy3 double mutant line (which has no phytochrome B and low levels of all other phytochromes) was examined after 5 d of individual leaf or whole-plant darkness, at the levels of visible yellowing, total chlorophyll, and chlorophyll a/b ratios. It was observed to behave much like the wild-type control (Fig. 5, and data not shown; leaves 4 and 5 were used as “older” and “younger” leaves, respectively, rather than leaves 5 and 7 as in the previous experiments, because the hy2/hy3 mutants flowered earlier and most plants developed only five leaves). In both mutant and wild-type plants, covering individual leaves resulted in a sharp drop in chlorophyll levels and visible yellowing, whereas covering whole plants produced a more mild drop in chlorophyll levels and no visible yellowing. Several other light-sensing mutants were also examined at the level of visible yellowing when individual leaves were covered for 5 d, including the phytochrome chromophore mutant hy1 (Koornneef et al., 1980; Davis et al., 1999), the blue-light receptor mutant hy4 (cry1; Koornneef et al., 1980), the light signal transduction mutant hy5 (Koornneef et al., 1980), the blue-light receptor mutant fha (cry2; Koornneef et al., 1991), and the double and triple mutants cry1/cry2, phya/phyb, and phya/phyb/phyd. In all cases, leaves that were covered visibly senesced as in wild type (data not shown).
Figure 5.
hy2/hy3 double mutants and wild-type (Ler) plants respond similarly to both individual leaf and whole-plant darkness. Experiments were performed similarly to those in Figure 1 (d 0 samples), except that in this experiment the “older leaf” was leaf 4 and the “younger leaf” was leaf 5 (because in most instances the hy2/hy3 plants flowered after forming only five leaves). Whole plants or individual leaves were darkened for 5 d and then leaf 4 or 5 was excised from the plant for analysis. Controls were leaves 4 or 5 excised from age-matched, light-grown plants. Total chlorophyll levels were measured and normalized separately for the older and younger leaves and for each line such that the value of the control leaf was always 100%. Absolute chlorophyll levels of the hy2/hy3 plants were 80% of those of the Ler plants in the younger control leaf, and 63% of the Ler plants in the older control leaf. Relative total chlorophyll levels and chlorophyll a/b ratios are shown.
DISCUSSION
That darkness can induce senescence has been known for many years, and many studies of this phenomenon have been published. In almost all instances, however, the studies were done in either detached leaves (e.g. Thimann and Satler, 1979; Biswal et al., 1983; Veirerskov, 1987) or intact seedlings (e.g. Wittenbach, 1977; Biswal et al., 1982; Blank and McKeon, 1991b; for review, see Biswal and Biswal, 1984). It has been shown previously that by several parameters senescence is also induced in detached Arabidopsis leaves in response to darkness (Oh et al., 1997; Weaver et al., 1998). Little work, however, has been done in attached adult leaves, in Arabidopsis or other species, and the few related reports have been somewhat contradictory, at least in their conclusions. All reports with which we are familiar agree that the transfer of whole plants to darkness induces chlorophyll and/or protein loss in true leaves (Blank and McKeon, 1991a, 1991b; Oh et al., 1996; Kleber-Janke and Krupinska, 1997; Weaver et al., 1998), and all those in which reversibility was assayed agree that chlorophyll and protein losses could be reversed by returning plants to the light, at least within several days of the dark treatment (Blank and McKeon, 1991a, 1991b; Kleber-Janke and Krupinska, 1997). When the expression of various senescence- and photosynthesis-associated genes was examined in response to both whole-plant darkness and natural senescence, however, the results were more varied, with some genes responding similarly to both treatments and others responding very differently (Blank and Mckeon, 1991b; Oh et al., 1996; Kleber-Janke and Krupinska, 1997; Weaver et al., 1998). Kleber-Janke and Krupinska (working in the primary leaves of barley seedlings) have interpreted this to mean that when whole plants are darkened a portion of the senescence program is induced, perhaps one corresponding to an early stage of senescence. We interpreted our results, in which we observed (in adult Arabidopsis leaves) that of 10 genes known to be induced during age-mediated senescence, only one-half were induced, and two were repressed, by whole-plant darkness, to mean that senescence is not induced during whole-plant darkness, and that genes that are induced during both may be primarily stress responsive (Weaver et al., 1998).
We first became interested in the possibility that darkening individual leaves might induce senescence when we observed that in densely growing Arabidopsis populations areas of leaves covered by other leaves are occasionally induced to yellow. Covering individual Arabidopsis leaves with cloth “mittens” revealed that by the criteria of appearance, total chlorophyll and protein levels, decline of CAB protein, and increase of SAG12 mRNA, senescence was induced in the covered leaves. Senescence was induced in both older and younger leaves by this treatment, although more quickly and strongly in the older ones, indicating an age-mediated component to the response. When whole plants were darkened, in contrast, by all criteria except loss of total protein and chlorophyll (which still occurred less quickly and less strongly than with individual leaf darkness), senescence was not induced. Thus, the changes brought about when whole plants are darkened are qualitatively different from those induced by age alone, whereas those induced when individual leaves are darkened appear similar to those induced by age.
When the reversibility of the changes induced by the dark treatments was examined, it was observed that 3 d after a return to the light, leaves from darkened plants were less senescent than controls, and that plants darkened for the longest were least senescent. Individually darkened leaves, in contrast, were more senescent after return to the light than controls. It was also observed that light-grown plants transferred to darkness senesced less quickly than those that remained in the light. We conclude from these experiments that darkening entire plants does not simply fail to promote senescence, but contrary to previously published reports delays it. To our knowledge, this is the first report of darkness delaying senescence. One should note that it remains unclear whether this is a delay of senescence per se or a more general developmental arrest.
We also examined the effects of darkness on whole seedlings. We observed that cotyledons, in contrast to true leaves, do appear to senesce when whole seedlings are placed in the darkness, whereas primary leaves do not. This would imply that in cotyledons (in which much of the work on dark-induced senescence has been done), senescence is regulated differently than in true leaves. This differential effect may be explained by the fact that cotyledons serve as storage organs.
Darkness induces individually covered leaves to senesce, which means that light inhibits that senescence. Previous work has suggested directly or indirectly that phytochrome might be involved in this light-mediated inhibition of senescence. At least two groups have indicated that both continuous white light and pulses of red light are able to inhibit senescence in detached leaves, and that the red light effect is reversible by far-red light (Tucker, 1981; Biswal and Biswal, 1984; Biswal and Choudhury, 1986). It was shown more recently that leaves of soybean (Guiamet et al., 1989) and sunflower (Rousseaux et al., 1996) senesced more quickly when the red:far-red ratio of the light they received was decreased, and that far-red light induces chlorophyll loss in tobacco leaves (Rousseaux et al., 1997). It also has been shown that tobacco and overexpressing oat phytochrome A display both delayed leaf senescence (Cherry et al., 1991) and an inhibited response to the senescence-promoting effects of far-red light (Rousseaux et al., 1997). We examined a hy2/hy3 phytochrome double mutant line, in which there is no phytochrome B and low levels of all other phytochromes (Parks and Quail, 1991; Somers et al., 1991), and observed it to behave much like wild-type controls, after both whole-plant and individual leaf dark treatments. We also examined several other photosensing mutants, including those deficient in both the phytochrome and cryptochrome pathways, to determine if, at the level of visible yellowing at least, covering individual leaves induced senescence as in wild-type plants. In all the mutants examined that was the case. One should note, however, that if phytochrome is involved in inhibiting senescence it might not be surprising that covered leaf senescence would continue to occur in phytochrome mutants, and in some instances might occur more strongly. Our experiments were designed primarily to detect a loss of the phenomenon, however, and would not necessarily have detected a promotion of it, particularly if it were subtle.
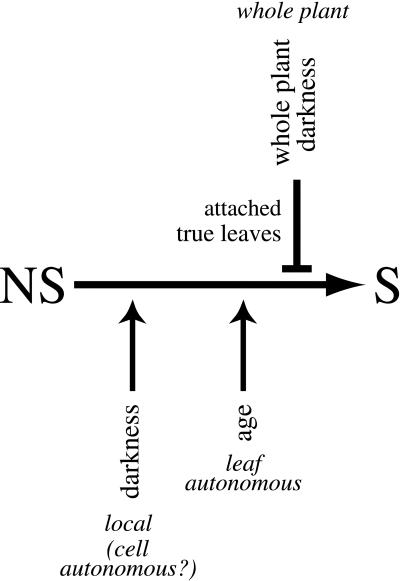
Taken together, these results imply that darkness (or light) mediates two separate senescence-related responses, depending on the level at which it is perceived. A model summarizing this is presented in Figure 6. In what might be regarded as the “default” response to darkness, senescence is induced in a localized, and possibly cell-autonomous, fashion. Age-mediated factors remain important, however, because older leaves respond more quickly and more strongly to darkness than younger ones, and dark and age thus can be viewed as separate, additive promoters of senescence. Superimposed over this default response is the ability of darkness, when experienced at the level of the entire plant, to delay not only the darkness-mediated senescence that would otherwise occur but also age-mediated (i.e. “natural”) senescence. This extra layer of control is not observed, however, in detached leaves (in which there is no whole plant) or in cotyledons.
Figure 6.
A model diagramming the promoting/delaying effects of darkness on senescence. The “default” effect of darkness (sensed locally, possibly at the level of the cell) appears to be to promote senescence. Age is a separate promoter of senescence, which is additive with darkness (and sensed at the level of the leaf). Superimposed over this, and epistatic to it, is the ability of darkness, when given at the level of the whole plant, to repress both age-mediated and individual leaf-mediated senescence (possibly due to a general inhibition of development). The inhibitory pathway is not present in detached leaves or cotyledons, however. NS, Non-senescent leaves; S, senescent leaves.
This work has not addressed the mechanisms of the promotion or inhibition of senescence. The fact that leaves senesce when individually darkened but not when the whole plant is darkened demonstrates that senescence of an individual leaf is controlled by the light status of the rest of the plant. This implies communication, which could be mediated either directly via light (e.g. photoreceptors directly initiating signal transduction pathways) or indirectly via photosynthesis and source-sink effects. Regardless of the mechanism, however, it seems clear that a leaf is able to senesce both with and without any contribution from the rest of plant (both when attached and detached), and in both the light and the darkness. Thus, whether or not leaves senesce varies with the level at which the darkness is perceived, and with the organs that perceive it.
MATERIALS AND METHODS
Plant Materials and Treatments
Seeds of Arabidopsis ecotype Landsberg erecta were originally obtained from the Arabidopsis Stock Center at Ohio State University (Columbus). Plants were grown on Fafard germination mix (Conrad Fafard Inc., Agawam, MA) under continuous cool-white fluorescent light (120 μmol m2 s−1) in growth chambers at 22°C. Under these conditions, plants flowered after forming approximately eight rosette leaves (cotyledons were not counted). Because the age of a leaf can affect its response to factors that influence senescence, plants were taken from synchronously growing populations and, with the exception of the seedling experiments, only identically aged leaves were pooled (e.g. the fifth and seventh true leaves were pooled separately). In most experiments, leaf 5 was used as an “older” leaf (beginning to visibly yellow in controls at the time of harvest) and leaf 7 was used as a “younger” leaf (fully expanded but showing no visible signs of senescence in controls; see Fig. 1B). Plants at this stage had flowered and contained developing siliques. This stage corresponds to d 0 in both Figure 1A (in which the experiment was then nearly finished) and Figure 2A (in which the experiment was then just beginning). At d −5 in Figure 1A, plants had bolted but had few or no siliques.
For the whole-plant dark treatment, whole, soil-grown plants in their pots were placed in dark boxes in the same chambers in which they had initially been grown. For the individual leaf dark treatments, leaves were covered with cloth “mittens” as shown in Figure 3 (in most experiments the mittens did not have holes in them, however).
In the seedling experiments, plants were grown in culture on agar-solidified medium containing 0.65 g L−1 Peters Excel 15-5-15 fertilizer (Grace Sierra, Milpitas, CA).
RNA Extraction and Blotting
Total RNA was extracted using RNA Isolator (Genosys Biotechnologies, The Woodlands, TX). RNA was size fractionated by electrophoresis on 1% (v/v) formaldehyde-agarose gels and transferred onto nylon membranes by capillary blotting. Fifteen micrograms of total RNA was loaded in each lane. Probes were 32P-labeled by random priming (Prime-a-Gene kit, Promega Corporation, Madison, WI). Hybridization was done at 65°C overnight in 0.25 m NaH2PO4 (pH 7.4), 7% (w/v) SDS, 1% (w/v) casein, and 1 mm EDTA, and membranes were washed two times for 45 min each in 0.04 m NaH2PO4 (pH 7.2), 1% (w/v) SDS, and 1 mm EDTA. Probe hybridization was visualized with a phosphorimager using ImageQuant software (Molecular Dynamics, Sunnyvale, CA).
Immunoblotting
Leaf extracts were prepared by grinding tissue under liquid N2 and adding equal volumes of lysis buffer (50 mm Tris-HCl, pH 7.5, 1 mm EDTA, 100 mm NaCl, 1% [v/v] NP40, 0.1% [w/v] SDS, 0.1% [v/v] Triton X-100, 0.7% [v/v] 2-mercaptoethanol, and 1 mm phenylmethylsulfonyl fluoride). Samples were vortexed, centrifuged, and the supernatant added to an equal volume of 2× load buffer (125 mm Tris-HCl, pH 7.5, 1% [v/v] 2-mercaptoethanol, 4% [w/v] SDS, 20% [v/v] glycerol, and 0.01% [w/v] bromphenol blue). Equal volumes of each sample (representing protein derived from equal volumes of leaf tissue) were electrophoresed on SDS-PAGE gels and electroblotted onto polyvinylidene difluoride membranes (Bio-Rad, Hercules, CA). Immunodetection was performed as by Shanklin et al. (1987). The CAB antibody is described by Sigrist and Staehelin (1992), and recognizes the LHC2b family of proteins.
Total Protein and Chlorophyll Extraction and Quantitation
Single Arabidopsis leaf discs were frozen in liquid nitrogen, ground in 2.2 mL of 96% (v/v) ethanol, incubated at room temperature in the dark for 30 min, pelleted in a microcentrifuge, and the chlorophyll content of the supernatant quantified spectrophotometrically using the method of Wintermans and DeMots (1965). The pellet was then rinsed once with 96% (v/v) ethanol, allowed to air dry, and resuspended in 60 μL of 1% (w/v) SDS, 1% (v/v) NP40, and 25 mm Tris, pH 7.5, by vortexing and heating for 30′ at 70°C. Protein was then quantified using the Bio-Rad DC Protein Assay kit according to the manufacturer's instructions (Bio-Rad Laboratories; protein for immunoblots was extracted differently, however, as described above). For each experimental condition, protein and chlorophyll concentrations were determined for three to eight independent leaf discs (from three–eight separate leaves) and the results averaged. Normalization was performed as described in the figure legends. Error bars show 1 sd. In the seedling experiments, seedlings were pooled and chlorophyll concentrations determined from an aliquot of the pooled tissue. Three replicate measurements were made for each pool and the results averaged.
ACKNOWLEDGMENTS
We thank Janet Meehl and Andrew Staehelin for generously providing the CAB antibodies. We also thank Scott Michaels for growing his Arabidopsis at such high density, and for noticing (and more than once pointing out) what sometimes happens when one leaf covers another. We also thank Carlos Ballaré for many helpful editorial comments.
Footnotes
This work was supported by the Consortium for Plant Biotechnology Research (grant no. DE–FG02–97ER20280), by U.S.–Israel Binational Agricultural Research and Development Fund (grant no. IS–2894–97), and by the Graduate School of the University of Wisconsin. L.M.W. was partially supported by the National Institutes of Health (training grant no. T32 GM07215).
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.010312.
LITERATURE CITED
- Biswal UC, Biswal B. Photocontrol of leaf senescence. Photochem Photobiol. 1984;39:875–879. [Google Scholar]
- Biswal B, Choudhury NK. Photocontrol of chlorophyll loss in papaya leaf discs. Plant Cell Physiol. 1986;27:1439–1444. [Google Scholar]
- Biswal B, Choudhury NK, Sahu P, Biswal UC. Senescence of detached fern leaves. Plant Cell Physiol. 1983;24:1203–1208. [Google Scholar]
- Biswal UC, Kasemir H, Mohr H. Phytochrome control of degreening of attached cotyledons and primary leaves of mustard (Sinapis alba L.) seedlings. Photochem Photobiol. 1982;35:237–241. [Google Scholar]
- Blank A, McKeon T. Three RNases in senescent and nonsenescent wheat leaves: characterization by activity staining in sodium dodecyl sulfate polyacrylamide gels. Plant Physiol. 1991a;97:1402–1408. doi: 10.1104/pp.97.4.1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blank A, McKeon TA. Expression of three RNase activities during natural and dark-induced senescence of wheat leaves. Plant Physiol. 1991b;97:1409–1413. doi: 10.1104/pp.97.4.1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherry JR, Hershey HP, Vierstra RD. Characterization of tobacco expressing functional oat phytochrome: domains responsible for the rapid degradation of Pfr are conserved between monocots and dicots. Plant Physiol. 1991;96:775–785. doi: 10.1104/pp.96.3.775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis SJ, Kurepa J, Vierstra RD. The Arabidopsis thaliana HY1 locus, required for phytochrome-chromophore biosynthesis, encodes a protein related to heme oxygenases. Proc Natl Acad Sci USA. 1999;96:6541–6546. doi: 10.1073/pnas.96.11.6541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grbic V, Bleecker AB. Ethylene regulates the timing of leaf senescence in Arabidopsis. Plant J. 1995;8:595–602. [Google Scholar]
- Guiamet JJ, Willemoes JG, Montaldi ER. Modulation of progressive leaf senescence by the red far-red ratio of incident light. Bot Gaz. 1989;150:148–151. [Google Scholar]
- Kleber-Janke T, Krupinska K. Isolation of cDNA clones for genes showing enhanced expression in barley leaves during dark-induced senescence as well as during senescence under field conditions. Planta. 1997;203:332–340. doi: 10.1007/s004250050199. [DOI] [PubMed] [Google Scholar]
- Koornneef M, Hanhart CJ, van der Veen JH. A genetic and physiological analysis of late flowering mutants in Arabidopsis thaliana. Mol Gen Genet. 1991;229:57–66. doi: 10.1007/BF00264213. [DOI] [PubMed] [Google Scholar]
- Koornneef M, Rolff E, Spruit CJP. Genetic control of light inhibited hypocotyl elongation in Arabidopsis thaliana. Pflanzenphysiologie. 1980;100:147–160. [Google Scholar]
- Lohman K, Gan S, John M, Amasino RM. Molecular analysis of natural leaf senescence in Arabidopsis thaliana. Physiol Plant. 1994;92:322–328. [Google Scholar]
- Noodén LD, Hillsberg JW, Schneider MJ. Induction of leaf senescence in Arabidopsis thaliana by long days through a light-dosage effect. Physiol Plant. 1996;96:491–495. [Google Scholar]
- Oh SA, Lee SY, Chung IK, Lee CH, Nam HG. A senescence-associated gene of Arabidopsis thaliana is distinctively regulated during natural and artificially induced leaf senescence. Plant Mol Biol. 1996;30:739–754. doi: 10.1007/BF00019008. [DOI] [PubMed] [Google Scholar]
- Oh SA, Park JH, Lee GI, Paek KH, Park SK, Nam HG. Identification of three genetic loci controlling leaf senescence in Arabidopsis thaliana. Plant J. 1997;12:527–535. doi: 10.1046/j.1365-313x.1997.00527.x. [DOI] [PubMed] [Google Scholar]
- Parks BM, Quail PH. Phytochrome-deficient hy1 and hy2 long hypocotyl mutants of Arabidopsis are defective in phytochrome chromophore biosynthesis. Plant Cell. 1991;3:1177–1186. doi: 10.1105/tpc.3.11.1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rousseaux MC, Ballare CL, Jordan ET, Vierstra RD. Directed overexpression of PHYA locally suppresses stem elongation and leaf senescence responses to far-red radiation. Plant Cell Environ. 1997;20:1551–1558. [Google Scholar]
- Rousseaux MC, Hall AJ, Sanchez RA. Far-red enrichment and photosynthetically active radiation level influence leaf senescence in field-grown sunflower. Physiol Plant. 1996;96:217–224. [Google Scholar]
- Shanklin J, Jabben M, Vierstra RD. Red light-induced formation of ubiquitin-phytochrome conjugates: identification of possible intermediates of phytochrome degradation. Proc Natl Acad Sci USA. 1987;84:359–363. doi: 10.1073/pnas.84.2.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigrist M, Staehelin LA. Identification of type 1 and type 2 light-harvesting chlorophyll a/b-binding proteins using monospecific antibodies. Biochim Biophys Acta. 1992;1098:191–200. doi: 10.1016/s0005-2728(05)80336-6. [DOI] [PubMed] [Google Scholar]
- Somers DE, Sharrock RA, Tepperman JM, Quail PH. The hy3 long hypocotyl mutant of Arabidopsis is deficient in phytochrome B. Plant Cell. 1991;3:1263–1274. doi: 10.1105/tpc.3.12.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thimann KV, Satler S. Relation between senescence and stomatal opening: senescence in darkness. Proc Natl Acad Sci USA. 1979;76:2770–2773. doi: 10.1073/pnas.76.6.2770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tucker DJ. Phytochrome regulation of leaf senescence in cucumber and tomato. Plant Sci Lett. 1981;23:103–108. [Google Scholar]
- Van Gysel A, Van Montagu M, Inze D. A negatively light-regulated gene from Arabidopsis thaliana encodes a protein showing high similarity to blue copper-binding proteins. Gene. 1993;136:79–85. doi: 10.1016/0378-1119(93)90450-h. [DOI] [PubMed] [Google Scholar]
- Veirerskov B. Irradiance-dependent senescence of isolated leaves. Physiol Plant. 1987;71:316–320. [Google Scholar]
- Weaver LM, Gan S, Quirino B, Amasino RM. A comparison of the expression patterns of several senescence-associated genes in response to stress and hormone treatments. Plant Mol Biol. 1998;37:455–469. doi: 10.1023/a:1005934428906. [DOI] [PubMed] [Google Scholar]
- Wintermans JFGM, DeMots A. Spectrophotometric characteristics of chlorophylls a and b and their pheophytins in ethanol. Biochim Biophys Acta. 1965;109:448–453. doi: 10.1016/0926-6585(65)90170-6. [DOI] [PubMed] [Google Scholar]
- Wittenbach VA. Induced senescence of intact wheat seedlings and its reversibility. Plant Physiol. 1977;59:1039–1042. doi: 10.1104/pp.59.6.1039. [DOI] [PMC free article] [PubMed] [Google Scholar]