Abstract
Antigen receptor gene assembly is regulated by transcriptional promoters and enhancers, which control the accessibility of gene segments to a lymphocyte-specific V(D)J recombinase. However, it remained unclear whether accessibility depends on the process of transcription itself or chromatin modifications that accompany transcription. By using T cell receptor β substrates that integrate stably into nuclear chromatin, we show that promoter location, rather than germ-line transcription or histone acetylation, is a primary determinant of recombination efficiency. These spatial constraints on promoter positioning may reflect an RNA polymerase-independent mechanism to target adjacent gene segments for chromatin remodeling events that facilitate rearrangement.
Antigen receptor genes are assembled in developing lymphocytes by a series of recombination events that fuse variable (V), diversity (D), and joining (J) gene segments. Two lymphocyte-specific proteins, termed RAG-1 and RAG-2, are essential components of the V(D)J recombinase complex (1, 2). These proteins introduce double-strand DNA breaks at selected recombination signal sequences (RSSs), which flank all Ig and T cell receptor (TCR) gene segments. The RSSs contain conserved heptamer and nonamer sequences separated by a nonconserved spacer (12 or 23 bp). Under physiological conditions, RAG-mediated cleavage requires the synapsis of a 12-bp and a 23-bp RSS (3, 4). Ubiquitous DNA repair enzymes then fuse participating gene segments and RSSs to generate a chromosomal coding join and a signal join, respectively (5). Although most RSSs are interchangeable, V(D)J recombinase is targeted to specific Ig and TCR loci during lymphocyte development. For example, assembly of TCR genes is restricted to thymocytes, initiating with rearrangement of Dβ and Jβ segments, followed by Vβ to DβJβ and ultimately Vα to Jα recombination (6).
Prevailing models for the developmental control of V(D)J recombination invoke changes in the accessibility of specific gene segments to the RAG-1/2 complex (6–8). Extensive correlations exist between transcription of unrearranged gene segments and their recombination potential (9, 10), suggesting that germ-line transcription and RAG accessibility share key regulatory components. Indeed, deletion of transcriptional enhancers from most Ig and TCR loci dramatically impairs the assembly of linked gene segments (3, 6). We and others have shown that the TCRβ enhancer (Eβ) mediates efficient Dβ1Jβ rearrangement via activation of a promoter located directly upstream of the Dβ1 gene segment (PDβ; refs. 11–13). Because prior studies have shown that chromatin impairs the access of RAG proteins to RSSs (14), promoter activation likely regulates recombination by initiating a cascade of chromatin modifications that culminate in gene transcription. In this regard, the acetylation of histones H3 and H4 correlates tightly with the transcription and recombination of antigen receptor loci (7, 15, 16). Despite these correlations, it remains unknown whether promoter-directed transcription is a prerequisite for recombination or is simply coincident with chromatin modifications that produce a RAG-accessible configuration.
To dissect the molecular determinants of accessibility, we have engineered recombinase-inducible lymphocytes containing TCRβ miniloci integrated stably into nuclear chromatin (17). Importantly, substrate rearrangement in these cells recapitulates all aspects of enhancer- and promoter-dependent recombination observed for endogenous TCRβ loci in vivo (9, 18). Here, we test the relative contributions of germ-line transcription and promoter-dependent changes in chromatin to recombinase accessibility. We find that both orientations of PDβ mediate efficient rearrangement of chromosomal substrates, whereas promoter orientation dramatically affects levels of germ-line transcription through the DβJβ gene segments. The stimulatory effect of PDβ on substrate rearrangement is position-dependent, requiring placement of the promoter proximal to the Dβ gene segment. Unexpectedly, inaccessible substrates lacking PDβ are associated with hyperacetylated histones, a chromatin feature that normally correlates with recombination. Based on these findings, we conclude that full accessibility to recombinase requires a promoter-directed remodeling of chromatin that is highly localized and mechanistically distinct from histone acetylation and germ-line transcription.
Materials and Methods
TCRβ Miniloci and Transfectants.
Stable transfection of M12 B cells was used to generate 5B3 (17), which contains tetracycline-inducible vectors encoding RAG-1 (19) and a green fluorescent protein fusion at the N terminus of RAG-2. Stable transfection of TCRβ miniloci into 5B3 was performed as described (17). Assays for germ-line transcription, signal ends (SEs), and DβJβ rearrangement were performed with independent pools of stable transfectants (≥20 independent integrations), as well as with multiple subclones for each construct. Results obtained with pooled transfectants were highly consistent with data obtained from subclones harboring a range of substrate copy numbers. Assays for restriction enzyme (RE) sensitivity were performed with at least two subclones for each construct.
The P+E−, P−E+, P+E+, and mP+E+ substrates have been described (12). All other miniloci contain an AccI/BglII fragment spanning Dβ1, which harbors an ATA to CGT substitution in the TATA box of the Dβ1 5′ RSS (DβΔT). The TATA mutation in Dβ1 limits transcriptional initiation to the minimal PDβ cassette inserted at other sites in the substrate. Control experiments demonstrated that mP+E+ containing this TATA box mutation was rearranged and expressed at wild-type levels. Minimal promoter/Dβ combinations were inserted into the NotI site of D−/E+. To avoid recognition of PDβ by RAG proteins, the Dβ 5′RSS nonamer (CTTTTTTGT) was converted to CTTTCGCGT in the promoter-repositioned substrates.
Individual minimal promoter/DβΔT combinations were prepared by inserting a 380-bp PCR fragment from p387/3′ (11) into the EcoRV (P- and P′-DJ) or SmaI site (D-P/P′-J) of pDβΔT. In each case, P denotes the 3′-5′, and P′ denotes the 5′-3′ orientation of PDβ compared with the endogenous locus. The inclusion of palindromic polylinker sequence at the 3′ end of the PDβ cassette inhibited transcriptional read through. The bidirectional nature of PDβ sustained transcription when placed in the 3′-5′ orientation. Thus, for simplicity, the P and P′ designations were based on transcriptional activity of P-DJ (transcriptionally active) and P′-DJ constructs (silent). A single enhancer/promoter cassette was used to construct DJ-P and DJ-P′. This cassette was isolated as a nonamer mutated PCR fragment from the iEκ/PDβ substrate, in which the iEκ element was positioned immediately 5′ of the minimal PDβ/Dβ sequence (12). PCR primers used for iEκ/PDβ nonamer mutagenesis were 5′iEκ (12) and 3′DβΔ9mer (ACAGCTTTATACGCGAAAGGACCC). The promoter/enhancer was inserted into the XhoI site of D−/E−.
DNA and Reverse Transcription–PCR Analyses.
Total RNA was isolated by using Trizol (Life Technologies, Grand Island, NY) and treated with RNase-free DNase I (38°C, 30 min) in 10 mM Tris⋅Cl (pH 8.3)/50 mM KCl/2 mM MgCl2. Treated RNAs (3 μg) were reverse transcribed by using random hexanucleotides. Germ-line JβCμ and β-actin transcripts were measured by PCR as described (12).
Genomic DNA extracts were prepared at final concentrations of 1 × 103 cells per μl (20). Relative levels of DβJβ and VλJλ rearrangements, as well as total DNA content (Cλ), were assessed by PCR assays as described (12).
Enzyme Cleavage Assays.
For SE analysis, genomic DNA was harvested from 5 × 106 cells 24 h after RAG induction by using 300 μl of DNA extraction buffer. Genomic DNA was ligated to BW-1/BW-2 linkers in a final volume of 40 μl (21). For analysis of RE cleavage products, 5B3 transfectants were resuspended at 2.5 × 107 cells per ml in ice-cold buffer containing 10 mM Tris·Cl (pH 7.4), 10 mM NaCl, 5 mM MgCl2, 1 mM EGTA, and 0.5 mM PMSF. Cells were lysed by addition of 3 volumes resuspension buffer supplemented with 0.1% Nonidet P-40. Nuclei were isolated by centrifugation and resuspended at 105 cells per μl buffer. For RE cleavage, 5 × 105 nuclei were incubated with either XmnI or EcoRV in 1 × RE buffer (30 μl) for 1.5 h at either room temperature (XmnI) or 0°C (EcoRV). RE cleavage reactions were halted with DNA extraction buffer (150 μl). Genomic DNA was ligated to BW-1/BW-2 linkers in a final volume of 40 μl (21).
For SE and RE assays, ligated DNA (5 μl) was amplified in a 50-μl reaction containing the appropriate primers (see below) for 12 cycles (SE assays) or 15 cycles (RE assays) after hot-start addition of Taq. The cycling profile for Jλ2/3 SEs has been described (22). Dβ SE and RE cycling profiles were 94°C, 30 s; 62°C, 30 s; and 72°C, 30 s. A 2-μl sample of PCR1 was used as template in 27-cycle (SE assays) or 30-cycle (RE assays) PCRs with nested primers. Loading control (Cλ) and copy number (Jβ1) PCRs were performed on 3 μl of ligated DNA.
Quantitation of RE sensitivity assays was accomplished by PhosphorImager analysis (Fuji). LM-PCR signals for XmnI and EcoRV digestion were normalized for DNA content (Cλ), substrate copy number (Jβ1), and digestion efficiency at XmnI or EcoRV sites in the accessible Cκ (XmnI) and c-myc (EcoRV) loci (see below).
Primers and Probes.
Primers used in assays for 5′Jλ2/3 SEs (22), VλJλ coding joins, JβCμ transcripts, Cλ, and β-actin have been described (12). Additional primers and probes are as follows. DβJβ joins: 5′-DβTATA-2 (AAGCTGTAACATTGTGGGGACAGG), 3′-Jβ2-2 (ATGTAGGTCCCAGACATGAGAGAGC), probe-PFJβ2 (AAAGCCTGGTCCCTGAGCCGA); 3′ Dβ SE and XmnI cleavage PCR1: 5′-BW-1 (21), 3′-Dβ13-D (24); 3′ Dβ SE PCR2: 5′-BW-1H, 3′-3′Dβ13-C (TGCATCCTTTGCTGCTAGGGCC); XmnI PCR2: 5′-BW-1X (CCGGGAGATCTGAATTCCTTTC); 3′-Dβ13-C: probe-3′Dβ-3 (AGTGGCCCTAGCAGCAAAGG); EcoRV cleavage PCR1: 5′-5′Jβ-1 (CAGACCACCATCAGTGGATAGGTG), 3′-BW-1; EcoRV PCR2: 5′-5′Jβ-2 (CAGCTCTTGATGAATATCATCATAGG), 3′-BW-1E (CCGGGAGATCTGAATTCATC), probe-3′Dβ2 (AGTAATCGCTTTGTG); Cκ XmnI control PCR1: 5′-Cκ-X1 (GCTGCTCATGCTGTAGGTGCTGTC), 3′-BW-1; Cκ XmnI PCR2: 5′-Cκ-X2 (CTGTCTTTGCTGTCCTGATCAGTC), 3′-BW-1Xκ (CCGGGAGATCTGAATTCACTTC), probe-Cκ-XP (GATTGATGGCAGTGAACG); c-myc EcoRV control PCR1: 5′-mycRV-1 (CACCATGTCTCCTCCAAGTAACTC), 3′-BW-1; c-myc EcoRV PCR2: 5′-mycRV-2 (CGGTCATCATCTGCAGCTGATCGG), 3′-BW-1E, probe-mycRV-P (GAGCCGCCGCTCCGGGCTCT); copy number (Jβ1): 5′-5′Jβ-2, 3′-PFJβ1, probe-3′Dβ2.
Chromatin Immunoprecipitation Assays.
Preparation of mononucleosomal DNA and chromatin immunoprecipitations using antisera for diacetylated histone H3 and tetraacetylated histone H4 (Upstate Biotechnology, Lake Placid, NY) were performed as described (7). Analysis of immunoprecipitated chromatin was performed by real-time PCR using a Roche LightCycler and a FastStart DNA Master Syber Green I kit (Roche Diagnostics). Primers used were GATCCAGAATGCTTTCACG and CTGCATCCTTTGCTGCTA for Dβ1, TCTTCACAAAAGGGATGTAAG and AGGACCATAGGAGGAGTAA for Jβ1, and TAGTTGCCGCTGCCAAACAC and GGGTCAGCTCAGTCAAAGCACA for glucose-6-phosphate dehydrogenase. T early α (TEA) primers have been described (7). The PCR program consisted of denaturation for 4 min at 95°C followed by 20 s at 95°C, 20 s at annealing temperature, and 20 s at 72°C for 50 cycles. Annealing temperatures were 65°C for Dβ1, 60°C for Jβ1 and TEA, and 68°C for glucose-6-phosphate dehydrogenase. Acetylation values were expressed as (bound/total for experimental)/(bound/total for glucose-6-phosphate dehydrogenase). The mean and SE of triplicate determinations are reported.
Results
Transcriptional Control Elements Regulate RAG Accessibility in TCRβ Miniloci.
We have developed recombinase-inducible lymphocytes (5B3) to dissect the function of promoters and enhancers in the regulation of gene segment accessibility. In this B cell system, tetracycline withdrawal induces expression of RAG-1 and a RAG-2/green fluorescent protein hybrid. The inducible nature of 5B3 permits integration of unrearranged TCRβ miniloci (Fig. 1A, ref. 25) into nuclear chromatin before assessment of their recombination efficiencies. Germ-line transcription of DβJβ gene segments is directed by the Igκ enhancer (iEκ) positioned downstream of the Dβ and Jβ elements, which activates the upstream Dβ1 promoter (PDβ).
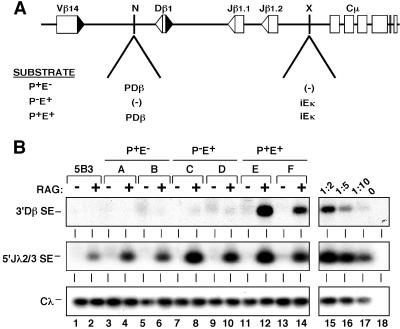
Fig 1.
Promoters and enhancers confer RAG accessibility to RSSs. (A) Schematic depiction of TCRβ miniloci. NotI (N) and XhoI (X) sites used to introduce PDβ and iEκ are shown. (B) Generation of SEs from chromosomal TCRβ miniloci. Genomic DNA was harvested from independent pools of 5B3 transfectants (letters above each lane) containing the specified miniloci (lanes 3–14) or untransfected 5B3 (lanes 1 and 2). All pooled transfectants harbored an average of 5–6 substrate copies and are derived from ≥20 clones. Cells were cultured in the absence or presence of tetracycline (24 h) and subjected to LM-PCR analysis (13) to measure induced cleavage of the 3′Dβ RSS. Control assays for recombinase activity (endogenous Jλ2/3-SEs) and DNA content (Cλ) are shown in Middleand Bottom, respectively. The linearity of each assay was confirmed by serial dilution (lanes 15–18) of the RAG-induced P+E+ sample shown in lane 12. Similar results were obtained with multiple independent subclones for each construct (data not shown).
Analogous to endogenous TCRβ loci, assembly of DβJβ coding joins in the minilocus critically depends on the presence of cis-acting enhancers and promoters (12). However, it remained unclear whether these elements directly regulate the chromatin accessibility of gene segments or facilitate DβJβ joining following RAG-mediated cleavage of RSSs (26). To address this question, we measured levels of Dβ1 SEs, which are the primary products of RAG-mediated cleavage, immediately following induction of the recombinase complex. For this purpose, genomic DNA from induced and uninduced 5B3 cells harboring wild-type (Fig. 1A, P+E+), enhancerless (P+E−), or promoterless (P−E+) miniloci were assessed for cleavage of the 3′Dβ RSSs by using a LM-PCR assay (13). Significant levels of Dβ1 SEs were detected in multiple, independent P+E+ transfectants contingent upon RAG induction (Fig. 1B, lanes 11–14, and data not shown). The Dβ1 SEs were not produced from endogenous TCRβ loci, which are inaccessible in 5B3 (lanes 1 and 2). However, SEs corresponding to the Jλ2 and Jλ3 gene segments in the accessible Igλ locus were readily detected in all transfectants, indicating comparable levels of recombinase activity. Importantly, 3′Dβ RSS cleavage was blocked in miniloci lacking either the enhancer (lanes 3–6) or PDβ promoter (lanes 7–10). These data indicate that enhancers and promoters in TCRβ miniloci directly regulate the accessibility of DβJβ chromatin to the RAG protein complex (24, 27).
Recombinational Accessibility Is Independent of DβJβ Expression Levels.
The activation of enhancers and promoters is accompanied by modifications of surrounding chromatin, including H3 and H4 acetylation (28). Coactivation of transcription and histone acetylation in rearranging loci (7, 15, 16, 29) suggests three mechanisms by which cis-acting elements regulate accessibility to V(D)J recombinase. First, germ-line transcription of gene segments may be necessary and sufficient for RAG accessibility. Second, enhancer-directed modifications of chromatin may be sufficient to generate a RAG-accessible configuration, and transcription arises as a byproduct of chromatin relaxation. Third, RAG targeting specificity may require both enhancer-dependent alterations of chromatin as well as transcription of the relevant gene segments.
To address whether the level of DβJβ germ-line transcription is a primary determinant of minilocus accessibility, we placed a minimal PDβ cassette containing its own TATA element in both promoter orientations upstream of Dβ1 (P and P′, Fig. 2A). Each substrate (P-DJ and P′-DJ) harbored a mutation destroying the TATA element in the 5′Dβ RSS and included the iEκ enhancer downstream of the Jβ gene segments. Analogous to the endogenous TCRβ locus, germ-line transcription in miniloci initiates at multiple sites downstream of the TATA box located in PDβ (11). The region of transcriptional initiation includes the 3′ Dβ RSS, which is targeted by recombinase during DβJβ rearrangement. Primary germ-line transcripts are spliced from Jβ1 segments to downstream constant region exons (Fig. 2A). As shown in Fig. 2B, levels of JβCμ transcripts in P-DJ transfectants were comparable to those observed for wild-type miniloci containing a germ-line configuration of the minimal PDβ/Dβ element (mP+E+; lanes 1, 2, 6, and 7). In contrast, expression of transcripts through the Dβ and Jβ RSSs was dramatically attenuated (greater than 10-fold) in substrates harboring the P′ orientation of PDβ (lanes 4 and 5). The reduced levels of transcription through target RSSs in P′-DJ transfectants were comparable to those observed for promoterless miniloci (lane 3). Similar results were obtained with multiple, independent clones for each construct and with an assay that detects transcripts originating within the Dβ RSS (data not shown). All enhancer-containing substrates expressed spliced transcripts in the neighboring Cμ region (Iμ transcripts, data not shown). Because Iμ transcripts are expressed in PDβ-deficient substrates, which are impaired for recombination (Fig. 2C, lanes 5 and 6), we conclude that nearby transcription is insufficient to generate accessibility at the DβJβ cluster.
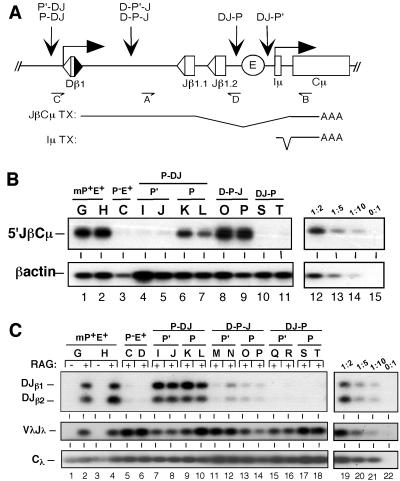
Fig 2.
Promoter positioning affects substrate rearrangement independent of germ-line transcription. (A) Diagram of iEκ-containing miniloci harboring repositioned promoters. A PDβ cassette was introduced into the TCRβ minilocus in both orientations (P and P′) at the indicated sites. Structures of JβCμ and Iμ transcripts, as well as the reverse transcription–PCR primers used to detect JβCμ germ-line transcripts (primers A and B) and DβJβ rearrangement (primers C and D) are shown. (B) Germ-line transcription in miniloci with repositioned promoters. Total RNA from independent pools of 5B3 transfectants (letters above each lane) containing the specified miniloci were subjected to a reverse transcription–PCR assay specific for JβCμ transcripts (A). Total cDNA levels were measured by using a PCR assay for β-actin transcripts (Lower). The linearity of each assay was confirmed by serial dilution of the P-DJ sample shown in lane 6 (lanes 12–15). Similar results were obtained with multiple independent subclones for each construct and with an assay that measures spliced transcripts originating within the Dβ RSS (data not shown). (C) DβJβ rearrangement in TCRβ miniloci. Genomic DNA was harvested from independent pools of 5B3 transfectants for each substrate (letters above each lane). Pooled cells were cultured in either the absence or presence of RAG expression (48 h). PCR assays (A) yielded amplification products corresponding to Dβ1Jβ1.1 (DJβ1) or Dβ1Jβ1.2 (DJβ2) rearrangements (Top). Control assays for recombinase activity (endogenous VλJλ rearrangement) and total DNA content (Cλ) are shown in Middle and Bottom, respectively. The linearity of each assay was confirmed by serial dilution of the RAG-induced P-DJ sample shown in lane 10 (lanes 19–22). Similar results were obtained with multiple independent subclones for each construct (data not shown).
To examine the effects of promoter inversion on recombinase accessibility, we used a PCR assay that measures DβJβ coding joins following RAG induction in pools of stable 5B3 transfectants (17). Levels of DβJβ coding joins were indistinguishable in the wild-type (mP+E+) and P-DJ transfectants (Fig. 2C, lanes 2, 4, 9, and 10). Importantly, substrates harboring the inverted promoter were equally accessible to recombinase (lanes 7 and 8), despite levels of germ-line transcription that were comparable to the promoterless minilocus. Thus, enhanced levels of germ-line transcription do not augment the rearrangement potential of chromosomal substrates. We obtained similar results with six independent 5B3 clones harboring each of the TCRβ substrates (data not shown). We conclude that PDβ confers accessibility to V(D)J recombinase independent of its effect on the levels of germ-line transcription through target gene segments.
Recombination Depends on Promoter Positioning.
Both promoters and enhancers mediate efficient DβJβ rearrangement in an orientation-independent fashion (Fig. 2, refs. 12, 30). We have shown previously that enhancers also facilitate DβJβ rearrangement irrespective of their position within TCRβ miniloci (12). As such, we tested whether promoter positioning relative to Dβ or Jβ directly influences rearrangement efficiency. For this purpose, PDβ was positioned between Dβ1 and Jβ1.1 (D-P-J) or downstream of Jβ1.2 (DJ-P) in the TCRβ substrate (Fig. 2A). As shown in Fig. 2B, relocation of the promoter between the gene segments supported levels of JβCμ transcription that were comparable to the wild-type substrate (lanes 8 and 9). As expected, JβCμ transcripts were absent when the promoter was positioned downstream of the DβJβ cassette (lanes 10 and 11). However, Iμ transcripts initiating at the PDβ/Iμ promoter position were readily detected in these substrates (data not shown).
Although the D-P-J substrate retained high levels of Jβ transcription, DβJβ rearrangement was significantly attenuated (≈10% of P-DJ; Fig. 2C, lanes 13 and 14). This inhibitory effect on recombination was not observed for substrates containing a nonsense sequence, equal in length to PDβ, between Dβ1 and Jβ (data not shown). When PDβ was inserted further downstream of Dβ (DJ-P), substrate rearrangement was completely blocked (lanes 17 and 18). Identical results were obtained with substrates harboring the P′ orientation at either downstream position (lanes 11, 12, 15, and 16), and with multiple independent clones for each construct (data not shown). Thus, we observed a gradient effect on recombinase accessibility that was related to the distance of PDβ downstream from its native site 5′ of Dβ1. Coupled with our finding that PDβ functions in either orientation in its native location (lanes 7–10), we conclude that the positioning of PDβ relative to Dβ1 is an important determinant of minilocus accessibility to the RAG complex.
Restriction Endonuclease Sensitivity Is Distinct from Accessibility to Recombinase.
Under physiologic conditions, RAG-mediated cleavage requires the recognition of two compatible RSSs (31, 32). As such, coding join assays cannot be used to distinguish whether deletion of a single regulatory element impairs chromatin accessibility locally (at only the most proximal RSS) or impedes access to the entire Dβ/Jβ region. To explore the independent roles of the promoter and enhancer in controlling chromatin accessibility, we isolated genomic DNA from 5B3 transfectants following incubation of nuclei with escalating amounts of the REs XmnI and EcoRV. These enzymes cleave at sites proximal to the PDβ/Dβ region and between Jβ1.1 and Jβ1.2, respectively. Restriction enzyme cleavage was assayed by ligation-mediated (LM)-PCR (Fig. 3A).
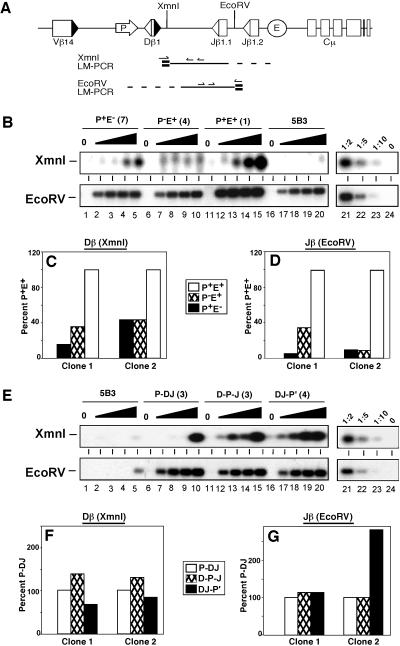
Fig 3.
Restriction endonuclease sensitivity is independent of promoter location. (A) Diagram of LM-PCR strategies for XmnI or EcoRV cleavage products. BW-1/2 linkers are shown in bold, and TCRβ-specific primers are indicated by arrows. (B and E) Levels of RE cleavage products within modified substrates. Nuclei from 5B3 clones containing the indicated TCRβ substrate or untransfected 5B3 cells were incubated with XmnI (0, 0.5, 1, 5, and 10 units) or EcoRV (at 0, 0.1, 0.5, 1, and 5 units). Genomic DNA from treated nuclei was subjected to nested LM-PCR for cleavage products (A). The linearity of each assay was confirmed by serial dilutions (lanes 21–24) of the maximally digested P+E+ sample (lane 15). (C, D, F, and G) Quantification of RE sensitivity in TCRβ miniloci. LM-PCR signals for XmnI (5 units) or EcoRV (5 units) treatment were quantified by PhosphorImager analysis. Values were normalized to signals obtained with PCR assays for DNA content (Cλ), substrate copy number (Jβ1.1), and cleavage of accessible loci (Cκ for XmnI or c-myc for EcoRV). Normalized values are shown relative to data for P+E+ (C and E) or P-DJ (D and F).
Representative data for RE sensitivity in the 5B3 nuclei are shown in Fig. 3B. Both sites were cleaved at low enzyme doses in the P+E+ substrate (lanes 11–15), whereas cleavage at analogous sites within the endogenous locus was impaired (5B3, lanes 16–20). Consistent with their reduced accessibility to RAG proteins, digestion at both sites in P+E− (lanes 1–5) and P−E+ (lanes 6–10) was reduced relative to P+E+. LM-PCR signals were then normalized for DNA content, substrate copy number, and enzymatic cleavage efficiency in each sample (see Materials and Methods). Normalized data for cleavage near Dβ1 (XmnI) or between the Jβ segments (EcoRV) are shown in Fig. 3 C–F. Substrates lacking either the promoter or enhancer exhibited decreased chromatin accessibility at both the Dβ1 and Jβ proximal sites (Fig. 3 C and D). In contrast, relocation of PDβ downstream of the Dβ segment had no significant effect on chromatin accessibility to restriction endonucleases (Fig. 3 E–G), despite a dramatic attenuation of DβJβ recombination. Together, these data indicate that the acquisition of RE sensitivity is mechanistically distinct from accessibility to recombinase, which requires promoter proximity to the Dβ gene segment.
Histone Acetylation Is Insufficient for Recombinational Accessibility.
Transcriptional activation is initiated by the recruitment of a multisubunit preinitiation complex to promoter regions and the concomitant reorganization of chromatin surrounding the target gene (33). Many promoter-bound preinitiation complexes contain histone acetyltransferases, which modify local chromatin by loosening histone/DNA association within nucleosomes (34). Recent reports suggest that V(D)J recombination and histone acetylation at target gene segments are inextricably linked (7, 15, 16, 29). However, it remained entirely unclear whether histone acetylation is sufficient to confer recombinational accessibility. To test this hypothesis, we used chromatin immunoprecipitation assays to measure the acetylation of histones associated with Dβ1 and Jβ1.1 in the 5B3 transfectants.
Mononucleosomal DNA was immunoprecipitated with anti-diacetylated H3 serum or anti-tetraacetylated H4 serum, and the relative abundance of Dβ1- and Jβ1-associated sequences in the antibody-bound fractions was determined by real time PCR (Fig. 4). Control analyses of parental 5B3 cells confirmed that the signals detected in this assay derive from the multicopy TCRβ miniloci rather than from the endogenous TCRβ locus (data not shown). As a control for hypoacetylated chromatin, we also included a real-time PCR assay specific for the TEA promoter, which is situated in the TCRα/δ locus (35) and is activated in a T cell-specific fashion (7).
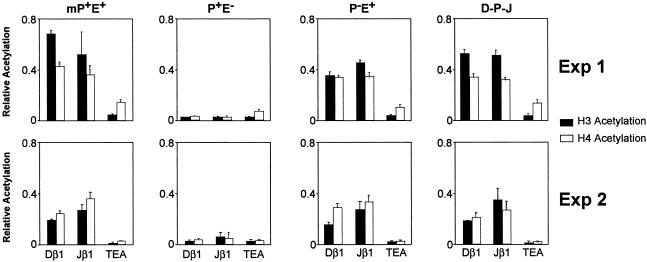
Fig 4.
Histone hyperacetylation is insufficient for recombinational accessibility. Acetylation levels of histones H3 and H4 associated with the Dβ1 and Jβ1.1 gene segments are shown for independent pools of 5B3 transfectants (Exp. 1 and Exp. 2). Chromatin immunoprecipitation assays were performed on mononucleosome preparations with antibodies specific for acetylated lysines on H3 (black bars) or H4 (white bars). Acetylation of histones at the endogenous TEA element, which is inactive in 5B3 cells, is shown as a negative control. Acetylation values were calculated as described in Materials and Methods.
Analysis of transfectants containing PDβ in its native position (mP+E+) revealed elevated acetylation of H3 and H4 at both gene segments in two independent transfectant pools (Fig. 4, Exp. 1 and Exp. 2). Hyperacetylation at Dβ and Jβ1.1 was critically dependent on inclusion of an enhancer because acetylation was reduced to the level of TEA in the P+E− transfectant. Surprisingly, elimination of PDβ (P−E+) had little (Exp. 1) or no (Exp. 2) effect on chromatin hyperacetylation within the DβJβ region, despite the low recombination efficiency of P−E+. Likewise, repositioning PDβ downstream (D-P-J) had no significant impact on Dβ1/Jβ1.1 hyperacetylation. Taken together, our findings clearly demonstrate that acetylation of associated histones is insufficient to confer recombinational accessibility upon chromosomal gene segments.
Discussion
Differential targeting of antigen receptor gene segments by V(D)J recombinase is modulated by cis-acting promoters and enhancers. These transcriptional control elements regulate the chromatin accessibility of RSSs (Fig. 1). Prior studies have correlated V(D)J rearrangement efficiencies with levels of germ-line transcription and histone acetylation at target gene segments (9). In this study, we have used chromosomal TCRβ miniloci to dissect these three processes. Our data indicate that (i) promoters augment rearrangement of linked gene segments independent of their effect on levels of germ-line transcription; (ii) promoter positioning relative to Dβ1 is critical for efficient rearrangement but not for general chromatin accessibility of the DβJβ gene segments; and (iii) histone hyperacetylation is insufficient to confer accessibility to V(D)J recombinase.
Germ-line transcription of Ig and TCR gene segments has frequently been used as a readout for recombinational accessibility (9, 23, 36). Indeed, TCRβ loci lacking the PDβ region are significantly attenuated for both transcription and rearrangement (12, 13). However, our findings clearly demonstrate that substrates containing opposite orientations of PDβ are equally accessible to RAG proteins but differ dramatically in their expression of transcripts through the target RSS elements (Fig. 2). Thus, levels of germ-line transcription do not necessarily reflect a chromatin configuration that is either accessible or inaccessible to recombinase (37, 38). Others have used chromatinized episomes to show that low levels of recombination can be detected in the absence of RSS transcription (39). These studies were unable to address whether transcription is required to amplify low levels of substrate accessibility. Importantly, we show that restoration of DβJβ rearrangement in promoterless constructs is largely unaffected by levels of germ-line transcription but is only contingent upon insertion of PDβ immediately upstream of the Dβ1 gene segment (Fig. 2). Although we cannot exclude the possibility that low levels of transcription through the target RSS elements are required for recombination, these unexpected observations strongly suggest that promoter-activated accessibility to RAG proteins is mechanistically distinct from gene segment transcription.
In contrast to germ-line transcription, promoter positioning relative to the Dβ gene segment contributes significantly to the rearrangement efficiency of TCRβ miniloci. Indeed, levels of DβJβ recombination exhibited an inverse relationship with the distance between Dβ1 and a PDβ element placed downstream (Fig. 2), suggesting that the proximity of PDβ to the Dβ1 gene segment or its positioning upstream of Dβ1 is essential for RAG accessibility. These data cannot be attributed to changes in overall chromatin accessibility at the gene segments because RE sensitivity was unchanged (Fig. 3). Moreover, PDβ retained the capacity to direct transcription when repositioned distal to the Dβ element (Fig. 2B). These data provide the first genetic demonstration that nuclear chromatin may be permissive to nucleases and transcription but refractory to cleavage by V(D)J recombinase. In TCRβ miniloci, a RAG-accessible configuration uniquely requires colocalization or 5′ positioning of the promoter relative to Dβ1 and its associated RSS.
Recent studies have shown that assembly of RSSs into mononucleosomes dramatically inhibits RAG-1/2 cleavage (14, 15). These suppressive effects are partly relieved by assembly with tailless or acetylated nucleosomes (14). Independent studies have correlated the rearrangement of antigen receptor loci with changes in histone acetylation (7, 15, 16, 29). However, a causal relationship between histone acetylation and recombinase accessibility had not been established. We now demonstrate that miniloci lacking a germ-line promoter are refractory to recombinase but remain hyperacetylated in nuclear chromatin (Fig. 4). Thus, enhancer-mediated acetylation of TCRβ chromatin is mechanistically distinct from accessibility to recombinase, which requires both a promoter and an enhancer. Moreover, our analyses of acetylation and RE sensitivity demonstrate that chromatin reorganization occurs throughout the DβJβ region in recombinationally impaired substrates containing repositioned promoters (Figs. 3 and 4). Thus, histone acetylation and chromatin relaxation may be necessary components of recombinase accessibility, but promoters must also direct a highly localized remodeling of Dβ chromatin. Our findings are fully consistent with recent results obtained by Emerson and colleagues, who show that optimal accessibility of recombination substrates in vitro requires extensive remodeling of RSS chromatin by the SWI/SNF complex, regardless of acetylation or transcription (B. Emerson and M. Oettinger, unpublished results).
Our data suggest a model that provides a compelling explanation for the conservation of germ-line promoters and their positioning directly upstream from certain gene segments (3, 6, 9). In the TCRβ locus, enhancer-mediated reorganization of chromatin may lead to modest levels of recombinase accessibility within the DβJβ clusters. However, promoter activation adjacent to target Dβ segments may be required to recruit additional chromatin modifiers, such as SWI/SNF. Localized alterations of nucleosomes then unmask the Dβ-RSS and permit RAG-mediated cleavage of synapsed Dβ/Jβ substrates. Alternatively, promoters and enhancers may serve to directly recruit RAG proteins to linked RSS elements. Additional studies will be necessary to delineate the specific hierarchy of factors recruited by transcriptional promoters to direct DβJβ recombination in vivo.
Acknowledgments
We thank Dean Ballard for many helpful comments. This work was supported by National Institutes of Health Grants AI37944 and AI01412 (to E.M.O.) and GM41052 and AI49934 (to M.S.K.).
Abbreviations
RSS, recombination signal sequence
TCR, T cell receptor
RE, restriction enzyme
SE, signal end
LM, ligation-mediated
TEA, T early α
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Oettinger M. A., Schatz, D. G., Gorka, C. & Baltimore, D. (1990) Science 248, 1517-1523. [DOI] [PubMed] [Google Scholar]
- 2.Schatz D. G., Oettinger, M. A. & Baltimore, D. (1989) Cell 59, 1036-1048. [DOI] [PubMed] [Google Scholar]
- 3.Hesslein D. G. & Schatz, D. G. (2001) Adv. Immunol. 78, 169-232. [DOI] [PubMed] [Google Scholar]
- 4.Oettinger M. A. (1999) Curr. Opin. Cell Biol. 11, 325-329. [DOI] [PubMed] [Google Scholar]
- 5.Roth D. B. & Roth, S. Y. (2000) Cell 103, 699-702. [DOI] [PubMed] [Google Scholar]
- 6.Sleckman B. P., Gorman, J. R. & Alt, F. W. (1996) Annu. Rev. Immunol. 14, 459-481. [DOI] [PubMed] [Google Scholar]
- 7.McMurry M. T. & Krangel, M. S. (2000) Science 287, 495-498.10642553 [Google Scholar]
- 8.Stanhope-Baker P., Hudson, K. M., Shaffer, A. L., Constantinescu, A. & Schlissel, M. S. (1996) Cell 85, 887-897. [DOI] [PubMed] [Google Scholar]
- 9.Oltz E. M. (2001) Immunol. Res. 23, 121-133. [DOI] [PubMed] [Google Scholar]
- 10.Yancopoulos G. D. & Alt, F. W. (1985) Cell 40, 271-281.2578321 [Google Scholar]
- 11.Sikes M. L., Gomez, R. J., Song, J. & Oltz, E. M. (1998) J. Immunol. 161, 1399-1405. [PubMed] [Google Scholar]
- 12.Sikes M. L., Suarez, C. C. & Oltz, E. M. (1999) Mol. Cell. Biol. 19, 2773-2781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Whitehurst C. E., Schlissel, M. S. & Chen, J. (2000) Immunity 13, 703-714. [DOI] [PubMed] [Google Scholar]
- 14.Kwon J., Morshead, K. B., Guyon, J. R., Kingston, R. E. & Oettinger, M. A. (2000) Mol. Cell 6, 1038-1048. [DOI] [PubMed] [Google Scholar]
- 15.McBlane F. & Boyes, J. (2000) Curr. Biol. 10, 483-486. [DOI] [PubMed] [Google Scholar]
- 16.Chowdhury D. & Sen, R. (2001) EMBO J. 20, 6394-6403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sikes M. L. & Oltz, E. M. (1999) J. Immunol. Methods 224, 25-29. [DOI] [PubMed] [Google Scholar]
- 18.Sleckman B. P., Bassing, C. H., Hughes, M. M., Okada, A., D'Auteuil, M., Wehrly, T. D., Woodman, B. B., Davidson, L., Chen, J. & Alt, F. W. (2000) Proc. Natl. Acad. Sci. USA 97, 7975-7980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shockett P., Difilippantonio, M., Hellman, N. & Schatz, D. G. (1995) Proc. Natl. Acad. Sci. USA 92, 6522-6526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schlissel M. S., Corcoran, L. M. & Baltimore, D. (1991) J. Exp. Med. 173, 711-720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schlissel M. S. (1998) Mol. Cell. Biol. 18, 2029-2038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bendall H. H., Sikes, M. L. & Oltz, E. M. (2001) J. Immunol. 167, 264-269. [DOI] [PubMed] [Google Scholar]
- 23.Bouvier G., Watrin, F., Naspetti, M., Verthuy, C., Naquet, P. & Ferrier, P. (1996) Proc. Natl. Acad. Sci. USA 93, 7877-7881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Whitehurst C. E., Chattopadhyay, S. & Chen, J. (1999) Immunity 10, 313-321. [DOI] [PubMed] [Google Scholar]
- 25.Ferrier P., Krippl, B., Blackwell, T. K., Furley, A. J., Suh, H., Winoto, A., Cook, W. D., Hood, L., Costantini, F. & Alt, F. W. (1990) EMBO J. 9, 117-125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hempel W. M., Stanhope-Baker, P., Mathieu, N., Huang, F., Schlissel, M. S. & Ferrier, P. (1998) Genes Dev. 12, 2305-2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mathieu N., Hempel, W. M., Spicuglia, S., Verthuy, C. & Ferrier, P. (2000) J. Exp. Med. 192, 625-637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Struhl K. (1998) Genes Dev. 12, 599-606. [DOI] [PubMed] [Google Scholar]
- 29.Tripathi R., Jackson, A. & Krangel, M. S. (2002) J. Immunol. 168, 2316-2324. [DOI] [PubMed] [Google Scholar]
- 30.Oltz E. M., Alt, F. W., Lin, W. C., Chen, J., Taccioli, G., Desiderio, S. & Rathbun, G. (1993) Mol. Cell. Biol. 13, 6223-6230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Eastman Q. M., Leu, T. M. & Schatz, D. G. (1996) Nature (London) 390, 85-88. [DOI] [PubMed] [Google Scholar]
- 32.van Gent D. C., Ramsden, D. A. & Gellert, M. (1996) Cell 85, 107-113. [DOI] [PubMed] [Google Scholar]
- 33.Workman J. L. & Kingston, R. E. (1998) Annu. Rev. Biochem. 67, 545-579. [DOI] [PubMed] [Google Scholar]
- 34.Kadonaga J. T. (1998) Cell 92, 307-313. [DOI] [PubMed] [Google Scholar]
- 35.Villey I., Quartier, P., Selz, F. & de Villartay, J. P. (1997) Eur. J. Immunol. 27, 1619-1625. [DOI] [PubMed] [Google Scholar]
- 36.Bories J. C., Demengeot, J., Davidson, L. & Alt, F. W. (1996) Proc. Natl. Acad. Sci. USA 93, 7871-7876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Goebel P., Janney, N., Valenzuela, J. R., Romanow, W. J., Murre, C. & Feeney, A. J. (2001) J. Exp. Med. 194, 645-656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Angelin-Duclos C. & Calame, K. (1998) Mol. Cell. Biol. 18, 6253-6254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cherry S. R. & Baltimore, D. (1999) Proc. Natl. Acad. Sci. USA 96, 10788-10793. [DOI] [PMC free article] [PubMed] [Google Scholar]