Abstract
We have measured the rates of insertion into, desorption from, and spontaneous interlayer translocation (flip-flop) in liquid-disordered and liquid-ordered phase lipid bilayer membranes, of the fluorescent phospholipid derivative NBD-dimyristoylphosphatidyl ethanolamine. This study made use of a recently described method that exploits a detailed knowledge of the binding kinetics of an amphiphile to bovine serum albumin, to recover the insertion and desorption rate constants when the albumin-bound amphiphile is transferred through the aqueous phase to the membrane and vice versa. The lipid bilayers, studied as large unilamellar vesicles, were prepared from pure 1-palmitoyl-2-oleoylphosphatidylcholine in the liquid-disordered phase; and from two cholesterol-containing binary lipid mixtures, 1-palmitoyl-2-oleoylphosphatidylcholine and cholesterol (molar ratio of 1:1), and egg sphingomyelin and cholesterol (molar ratio of 6:4), both in the liquid-ordered phase. Insertion, desorption, and translocation rate constants and equilibrium constants for association of the amphiphile monomer with the lipid bilayers were directly measured between 15° and 35°C, and the standard free energies, enthalpies, and entropies, as well as the activation energies for these processes, were derived from this data. The equilibrium partition coefficients for partitioning of the amphiphile between the aqueous phase and the different membrane phases were also derived, and permitted the estimation of hypothetical partition coefficients and the respective energetic parameters for partitioning between the different lipid phases if these were to coexist in the same membrane.
INTRODUCTION
A detailed knowledge of the kinetics of insertion of amphiphiles into membranes, and desorption from them, besides providing a baseline for the understanding of several biological phenomena, also elucidates some of the physical-chemical properties of membranes. In recent work (Pokorny et al., 2000, 2001; Abreu et al., 2003) we have shown that it is possible to obtain equilibrium partition coefficients for amphiphile partitioning between the aqueous phase and membrane phases as well as between coexisting membrane phases from insertion/desorption kinetics. Information concerning amphiphile partitioning between coexisting membrane phases is of particular relevance to biological systems in which phase coexistence (i.e., rafts) may play an important role in cell physiology (Vaz and Almeida, 1993; Simons and Ikonen, 1997; Brown and London, 1998; Simons and Vaz, 2004). As discussed elsewhere (Vaz and Melo, 2001) there are only a limited number of methods that permit obtaining reliable values of amphiphile partition coefficients for partitioning between the aqueous phase and the membrane phase, and particularly between coexisting membrane phases. Many amphiphiles of biological interest have very low critical aggregation concentrations (CAC) in the aqueous phase and determinations of the partition coefficients are complicated by the coexistence of the complex amphiphile aggregation equilibria.
There is a very large body of literature, going back over some decades, concerning the kinetics of spontaneous desorption of phospholipids and phospholipid derivatives from lipid bilayers, biological membranes, and other organized lipid structures such as lipoproteins (Silvius and Leventis, 1993; Brown, 1992; Jones and Thompson, 1989, 1990; Wimley and Thompson, 1990; Homan and Pownall, 1988; Ferrell et al., 1985; Nichols, 1985; Nichols and Pagano, 1981, 1982; McLean and Phillips, 1981, 1984; Massey et al., 1982; and literature cited by these authors). However, direct measurement of the kinetics of insertion of these lipids into bilayers seems to have eluded investigation. Measurement of the equilibrium partition coefficient, KP(L/W), for partitioning of these amphiphiles between the membrane and aqueous phases, together with measurement of the desorption rate constant would, of course, permit a reliable estimate of the insertion rate constant (Nichols, 1985). However, reliable measurement of KP(L/W) is hindered by the fact that these amphiphiles usually have critical aggregation concentrations, CAC, well below 10−8 M. This makes most optical techniques unreliable and more sensitive methods, such as radioactive tracer techniques, are often not amenable to rapid kinetic studies.
We have recently described a method that partly overcomes this difficulty (Abreu et al., 2003). Some amphiphiles bind rather strongly to bovine serum albumin (BSA) (KB ≈ 106 M−1 or higher) so that in the presence of this protein the concentration of the free amphiphile in the aqueous solution can be significantly reduced. If a reduction of the amphiphile concentration in the aqueous phase to a value below the CAC can be achieved, the addition of lipid bilayer membrane vesicles to a solution of the amphiphile and albumin results in a transfer of the amphiphile to the membrane via monomers in the aqueous phase. The monomer in aqueous solution is replenished because it is in equilibrium with amphiphile bound to the protein. It had been previously shown (Abreu et al., 2003) that transfer of the amphiphile between albumin and the membranes does not occur via a direct encounter between the protein and the lipid bilayers. The three species of amphiphile in the system are, therefore, the monomer in the aqueous solution, the amphiphile bound to the albumin and the amphiphile associated with the membranes. The relative amount of each species is determined by the association constants of the amphiphile with the albumin and the lipid bilayer membranes and the respective total concentrations. If there is a difference in the measured signal originating from each of the amphiphile species in the system (for example, fluorescence in the case of fluorescent phospholipid derivatives, as is the case in this work), the net resultant signal allows us to follow the path to equilibrium in time and, therefore, to directly measure the reaction rate constants. Independent knowledge of the rate constants for amphiphile interaction with the protein allows us to fit the experimental kinetic curves with only two unknown variables: the rate constants for interaction with the membrane. If spontaneous transbilayer translocation (flip-flop) occurs, this process must be independently measured. The ratio of the rate constants for insertion into/desorption from the lipid bilayer membranes provides the equilibrium association constant of the amphiphile with the bilayer membrane vesicles, KL, and from this value the equilibrium partition coefficient for partitioning between the lipid and the aqueous phases, KP(L/W), can be derived as previously described (Abreu et al., 2003).
In this work we report direct measurements of the insertion and desorption rate constants for association of the fluorescent phospholipid derivative (labeled on the polar headgroup), NBD-DMPE, with LUVs prepared by extrusion through porous membranes with a 0.1-μm pore diameter. We also report the independently measured spontaneous translocation rate for NBD-DMPE in these membranes. The LUVs were prepared from pure POPC, and from two binary mixtures: a mixture of POPC and cholesterol (at a molar ratio of 1:1) and a mixture of SpM and cholesterol (at a molar ratio of 6:4). All measurements were done as a function of temperature between 15° and 35°C. In this temperature range the membranes prepared from pure POPC were a liquid-disordered phase (Koynova and Caffrey, 1998) whereas the other two were liquid-ordered phases (Sankaram and Thompson, 1990; Mateo et al., 1993). The temperature-dependence of the rate constants for insertion into and desorption from the membrane phases from/to the aqueous phase and the derived KL and KP(L/W) enabled us to derive the standard free energy, enthalpy, and entropy changes associated with transfer of NBD-DMPE to the membrane phase from the aqueous phase as well as the Arrhenius activation energies for the insertion and desorption processes. Finally, from the values of KP(L/W) we derived hypothetical KP(L1/L2) values for partitioning of the amphiphile between any two of the membrane phases examined if they were to coexist in the same membrane. Since this information is also available as a function of temperature, we are able to report the values of the thermodynamic parameters (standard free energy, enthalpy, and entropy changes) for the partitioning of NBD-DMPE between lipid bilayer phases.
MATERIALS AND METHODS
NBD-DMPE (purity >99%) and POPC were from Avanti Polar Lipids (Alabaster, AL); Egg Yolk SpM and BSA, essentially free of fatty acids (∼0.005%), were from Sigma-Aldrich Química (Sintra, Portugal); and cholesterol was from Serva/Boehringer Ingelheim (Heidelberg, Germany). All reagents were of the highest commercially available purity. Solvents of analytical reagent grade were from Merck Portuguesa (Lisbon, Portugal).
Phospholipid concentrations were determined using a modified version of the Bartlett phosphate assay (Bartlett, 1959) and cholesterol concentrations were determined by the Lieberman-Burchard method as described by Taylor et al. (1978). BSA concentrations were determined using the method of Lowry et al. (1951) or by their absorbance at 278 nm using an extinction of 0.66 mg−1 mL cm−1 (Peters, 1997), and NBD-DMPE concentration was determined by spectrophotometry using a molar extinction coefficient at 463 nm of 21,000 M−1 cm−1 in methanol. Absorption spectra were recorded on a Unicam UV530 UV/Vis spectrophotometer and fluorescence measurements were performed on a Cary Eclipse fluorescence spectrophotometer equipped with a thermostated multicell holder accessory (Varian, Cary, NC). The samples were stirred continuously during measurements. Stopped-flow measurements were performed as previously described (Abreu et al., 2003) on a thermostated stopped-flow fluorimeter (Hi-Tech model SF-61, Hi-Tech Scientific, Salisbury, UK). Data was analyzed using Excel and Solver (Microsoft, Seattle, WA).
The critical aggregation concentration of NBD-DMPE was determined by measuring the fluorescence emission intensity of this amphiphile, in buffered aqueous solution, at 540 nm (excitation at 480 nm) as a function of concentration between 1 × 10−9 M and 1 × 10−6 M. A plot of the concentration-normalized fluorescence intensity versus the logarithm of the amphiphile concentration (see Fig. 1) showed a change in slope with the inflexion point at ∼5 × 10−9 M. This was considered to be the CAC.
FIGURE 1.
Determination of the critical aggregation concentration of NBD-DMPE in aqueous solution. The change in slope of a plot of the concentration-normalized fluorescence intensity of NBD-DMPE in buffered aqueous solution versus log([NBD-DMPE]) was considered to be the critical aggregation concentration for the amphiphile.
In this work it was necessary to preassociate NBD-DMPE with BSA before addition of LUVs to the reaction mixture. Preassociation of NBD-DMPE with BSA was done by squirting a methanol solution of the phospholipid derivative into an aqueous buffered (0.01 M sodium phosphate, 0.02% sodium azide, 0.15 M sodium chloride, pH 8) solution of BSA at the desired concentration while vortexing the latter. The solution was then allowed to attain equilibrium over a period of 18–24 h at 23°C. The final concentration of methanol was always <1% (v/v).
Aqueous suspensions of lipids were prepared by evaporating a solution of the desired lipid or premixed lipid mixture in chloroform/methanol (1/1, v/v) solution by blowing dry nitrogen over the heated (blowing hot air over the external surface of the tube) solution and then leaving the residue in a vacuum desiccator for at least 8 h at 23°C. The solvent-free residue, heated in a water bath at 60°C, was then hydrated with 0.15 M sodium chloride, 0.02% sodium azide, adjusted to pH 8, which had been previously heated to the same temperature and the mixture was left to hydrate for ∼10 min at 60°C. The amount of hydrating medium added was calculated to result in a final lipid concentration of ∼10−3 M. The hydrated lipid was vigorously vortexed at room temperature to produce a suspension of multilamellar vesicles that was then extruded, using a minimum of 10 passes, through two stacked polycarbonate filters (Nucleopore) with a pore diameter of 0.1 μm (Hope et al., 1985). During extrusion, the water-jacketed extruder (Lipex Biomembranes, Vancouver, BC, Canada) was maintained at a temperature of 65°C (for membranes prepared from mixtures of SpM and cholesterol) or 23°C (for other membranes). The LUV suspensions obtained after extrusion were diluted in buffer to obtain the desired lipid concentration for fluorimetric experiments.
The kinetics of association of NBD-DMPE with LUVs were studied in two modes:
The process was initiated by addition of the desired amount of LUVs to a solution of NBD-DMPE which had been equilibrated with BSA over a period of 18–24 h. The amount of BSA used was such that the fraction of unbound NBD-DMPE in the system was <0.2% of the total. Fluorescence emission intensity changes were followed in the fluorimeter either during the initial stage of the process of transfer of NBD-DMPE from BSA to the LUV or until the process had attained a state close to equilibrium. As discussed in Results, the time course of the process at very short times after initiation of the reaction is particularly sensitive to the insertion rate constant, k+. Analysis of the entire process permits obtaining both the insertion and desorption rate constants, of course, but is not always advisable, especially when very long times are necessary to attain equilibrium.
NBD-DMPE was incorporated into the LUV through premixing with the desired lipid composition in organic solvent. The desired amount of BSA was then added to the NBD-DMPE-containing LUVs and fluorescence emission intensity changes were recorded for short times after initiation of the reaction. As discussed in Results, the time course is, in this case, particularly sensitive to the rate constant for desorption, k−. The effective total concentration of NBD-DMPE in these experiments is only half the real total concentration since only the probe that is in the outer leaflet may equilibrate with the BSA (there is no BSA inside the LUV). The fraction of probe that leaves the bilayer during the experiment is very small (≅5%), thus making the process insensitive to translocation between the two leaflets.
The rate of spontaneous translocation of NBD-DMPE between the monolayers of a LUV was measured by studying the time course of reduction of the NBD groups located in the outer monolayer of LUVs using sodium dithionite (McIntyre and Sleight, 1991). Briefly, 20 μL of a freshly prepared 1 M sodium dithionite solution in 1 M Tris-HCl, pH 10, was added to a suspension of symmetrically labeled LUVs (1 × 10−3 M in total lipid) containing 0.1 mol % NBD-DMPE, in 0.01 M HEPES, pH 7.4. The NBD groups in the outer monolayer of the LUVs were rapidly (within 1 min) reduced, which resulted in a reduction of the fluorescence intensity to a value very close to 50% of the original value. Spontaneous translocation of the probe from the inner to the outer monolayer was a distinctly slower process (if it occurred at all) and could be followed continuously in time to yield the translocation rate constant either from an analysis of the entire bleaching curve or from the initial velocity.
RESULTS
Binding of NBD-DMPE to bovine serum albumin
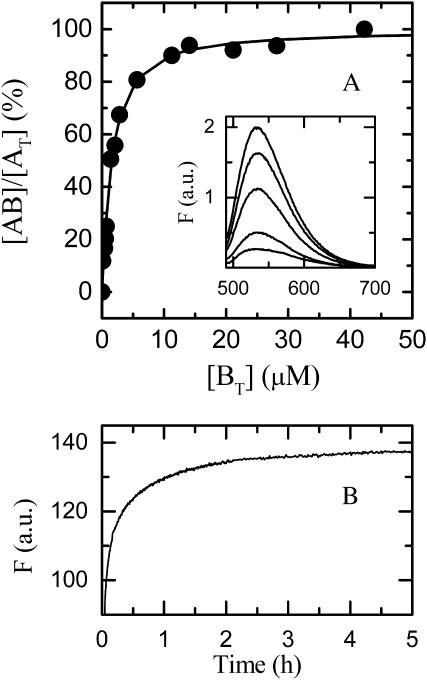
When a solution of NBD-DMPE in methanol is squirted into a gently vortexed buffered solution of BSA, the relative fluorescence emission quantum yield of the solution excited at 450 nm increases with increasing BSA concentration as shown in the insert to Fig. 2 A. This increase, a consequence of the binding of NBD-DMPE to the protein, was used to obtain the titration curve shown in Fig. 2 A. The binding of NBD-DMPE, at concentrations above its CAC to BSA, is a slow process, as seen in Fig. 2 B. The time course of this process reflects both the kinetics of association of the lipid with its binding site(s) on BSA as well as the kinetics of disaggregation of NBD-DMPE aggregates that form in aqueous solution and was not studied further. An estimate of the value of KB can be obtained from fitting a theoretical curve to the experimental data in Fig. 2 A and serves as the basis for an exact determination of this parameter. The respective rate constants may be obtained from stopped-flow experiments as described elsewhere (Abreu et al., 2003). If NBD-DMPE binding to BSA may be described in terms of binding to two independent binding sites on the protein, the best fits to the association kinetic curve is obtained with equilibrium association constants, KB1 = 2.9 × 106 M−1 and KB2 = 2.8 × 106 M−1, and characteristic times for equilibration ≤ 0.1 s. It is not clear whether the binding stoichiometry is 2:1 or whether the protein exists in two conformational states in solution (Peters, 1997) under the conditions of our experiment, each with a binding stoichiometry of 1:1. We are currently investigating this question. However, as will be evident below, it is not relevant to the objective and conclusions of this work.
FIGURE 2.
(A) Titration curve used to determine KB for the binding of NBD-DMPE to BSA at 35°C. The insert compares the fluorescence spectra of NBD-DMPE in aqueous solution and bound to BSA. (B) Time course of the binding of NBD-DMPE, dispersed in aqueous solution at a concentration above its CAC, to BSA at 25°C.
Transfer of NBD-DMPE between BSA and lipid bilayer vesicles via the aqueous phase
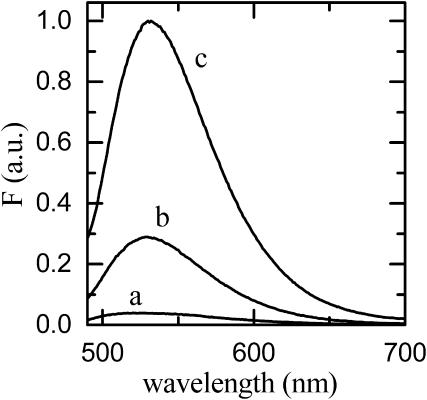
As seen above, binding of NBD-DMPE to BSA results in an enhancement of its fluorescence quantum yield. This is also the case when NBD-DMPE is transferred from the aqueous phase to a membrane phase. The relative fluorescence quantum yield of NBD-DMPE is even higher in the membrane phase than it is when bound to BSA (see Fig. 3). Thus, by monitoring the fluorescence intensity we can follow the transfer of NBD-DMPE from the aqueous phase or from BSA (via the aqueous phase) to the membranes. We have previously shown that the transfer of amphiphiles between BSA and membranes occurs via aqueous phase monomers and not via a second order collisional interaction between the protein and the membrane (Abreu et al., 2003). When lipid vesicles are added to a mixture of albumin and NBD-DMPE, the following kinetic scheme has to be considered:
 |
(1) |
where A is the amphiphile (NBD-DMPE), B1 and B2 are the two independent binding sites (or two conformational states of BSA, with one binding site each) that bind the probe, LV are the lipid vesicles, and  is A located in the outer (inner) leaflet of the bilayer, kf being the rate constant for the spontaneous translocation (flip-flop) between the two leaflets. There will, of course, be an additional equilibrium between the probe in the inner leaflet,
is A located in the outer (inner) leaflet of the bilayer, kf being the rate constant for the spontaneous translocation (flip-flop) between the two leaflets. There will, of course, be an additional equilibrium between the probe in the inner leaflet,  and the aqueous phase trapped within the vesicles. Considering the fact that this trapped volume is very small, that the expected value of KL (or KP(L/W)) is very large, and that there is no BSA in the trapped volume, we have opted to ignore the contribution of this equilibrium.
and the aqueous phase trapped within the vesicles. Considering the fact that this trapped volume is very small, that the expected value of KL (or KP(L/W)) is very large, and that there is no BSA in the trapped volume, we have opted to ignore the contribution of this equilibrium.
FIGURE 3.
Comparison of the fluorescence emission spectra of NBD-DMPE in aqueous solution (a), bound to BSA (b), and associated with LUVs prepared from POPC (c).
In a typical experiment, a solution of 0.9 × 10−6 M NBD-DMPE was first equilibrated at room temperature with 2 × 10−4 M BSA (both referred to the total volume) overnight. Under these conditions, with KB = 2.8 × 106 M−1, the concentration of free NBD-DMPE in the aqueous phase is 1.6 × 10−9 M, which is below the CAC for this amphiphile. Thus the free NBD-DMPE in the aqueous phase is expected to be monomeric. To this solution LUVs were added to give a final lipid concentration of between 0.9 and 1.5 × 10−3 M (0.9 and 1.5 × 10−8 M in LUVs, assuming 105 lipid molecules per LUV) and the fluorescence of the mixture was followed in time. Typical traces are shown in Fig. 4. It is evident that the process of insertion into/desorption from the lipid bilayers is extremely slow (characteristic times of ∼2 h for POPC and POPC/Chol and ∼10 h for SpM-Chol, see Fig. 4, curves) compared to the characteristic times (≤0.1 s) for association with the protein binding site(s). The kinetics of interaction of NBD-DMPE with the lipid bilayers, therefore, dominates the time course of the curve. This introduces an important simplification on the kinetic description of the overall process because it may be assumed that the interaction between the probe and the protein is in rapid equilibrium at all times. When the total concentration of protein is much higher than the total concentration of the probe, a further simplification may be made: [B] ≅ [B]T, where [B]T is the total concentration of B. The kinetics of the process may then be described by
 |
(2) |
When the characteristic time for spontaneous interlayer translocation, τf = 1/kf, is longer by at least two orders of magnitude than the characteristic time for interaction between aqueous amphiphile and the outer leaflet of the bilayer, 1/(k−+k+[LV]/(1+KB[B]T)), the kinetics of the interaction with the bilayer and translocation are uncoupled. In this case it may be considered that kf ≅ 0 in Eq. 2 and the integration of the differential equations is straightforward, resulting in
 |
(3) |
If kf is not negligible, the integration of the differential expressions in Eq. 2 leads to a rather complicated result.
FIGURE 4.
(A) Experimentally observed kinetics, at 35°C, of the transfer of NBD-DMPE between BSA and LUVs in the ld phase prepared from POPC, ♦, and in the lo phase prepared from an equimolar mixture of POPC and cholesterol, ▪, and from a 6:4 molar mixture of SpM and cholesterol, •. Each curve was fitted by numerical integration of Eq. 2. The respective residuals, as Δ[ALv]/[A]T (%), are shown in B–D for POPC, POPC-Chol, and SpM-Chol, respectively. The recovered rate constants are presented in Table 1.
For very small reaction times, (t ≅ 0), the total amphiphile concentration in the bilayer, [ALv], is not dependent on kf, but varies linearly in time, and is given by
 |
(4) |
Following the entire process until equilibrium is attained may take several days and, therefore, raises questions concerning sample and equipment stability. Also, there is no advantage in following the entire process to obtain the values of k+ and k−. We have used complete analysis of association kinetics for those cases in which the characteristic time for the process was <12 h. In all other cases (and this applies obligatorily to experiments with SpM-Chol at T ≤ 25°C), k+ and k− were obtained from initial rates as described above.
The fluorescence at any given time, F(t), is given by
 |
(5) |
where ϕi is a factor of proportionality between the relative emission of the ith species and its concentration, [i]. Considering that the contribution F[A](t) is negligibly small at all times in the course of the experiment both in terms of the concentration of this species under the experimental conditions and in terms of its relative quantum yield (Fig. 3), it may be neglected in Eq. 5 above. Under these conditions, the time dependence of the fluorescence emission, for t ≅ 0, is given by
 |
(6) |
If the probe is first equilibrated with the protein and the reaction is initiated by the addition of a LUV suspension, the initial conditions are given by
 |
(7) |
and the linear region of the time-dependence of F(t) is described by
 |
(8) |
This permits a reliable estimate of k+ by following the reaction for a very short time after its initiation.
If NBD-DMPE is first incorporated into the membranes and BSA is then added to the suspension of NBD-PE-containing LUVs, the protein rapidly binds the NBD-DMPE in aqueous solution followed by the NBD-DMPE that subsequently desorbs from the lipid bilayers until equilibrium is reached. The time dependence of the fluorescence emission at t ≅ 0 is obtained by substituting the initial conditions, after the instantaneous equilibration of the aqueous amphiphile with the protein,
 |
(9) |
into Eq. 6, and is given by
 |
(10) |
allowing a reliable estimate of k−. Equation 10 was derived with the assumption that the fraction of amphiphile in the aqueous media, [A](t)/[A]T, is negligible at all times, which is equivalent to KB[B]T;KL[LV]≫1.
Spontaneous translocation of NBD-DMPE between the monolayers of LUVs
Analysis of the complete (or near-complete) processes of transfer of NBD-DMPE between bovine serum albumin and LUV membranes were performed by numerical integration of Eq. 2. Since kf is a parameter that is amenable to independent evaluation, we decided to determine its value independently to reduce the number of unknown variables in the analysis. Spontaneous translocation of amphiphiles that contain the NBD fluorophore between the monolayers of a membrane is conveniently measured by the rapid reduction of NBD to a nonfluorescent group using sodium dithionite as a reducing agent (McIntyre and Sleight, 1991). The characteristic times for spontaneous transverse translocation of NBD-PE have been reported to be on the order of ∼30 min in ld phase lipid bilayers (Moss and Bhattacharya, 1995) thereby possibly invalidating the condition that led to Eq. 3 above. Fig. 5 shows the time course of a typical bleaching experiment using sodium dithionite. A rapid bleaching of the NBD-DMPE in the outer monolayer of the LUV reduces the initial fluorescence intensity to approximately one-half of its initial value. Since dithionite does not cross the lipid bilayer, the slower, simple exponential process that follows this first rapid bleach can be ascribed to the bleaching of NBD-DMPE that spontaneously translocates from the inner to the outer monolayer of the LUV where it is rapidly bleached by dithionite. The rate constant for translocation, kf, may be obtained from this second process. Fig. 6 shows the temperature-dependence of kf between 15° and 35°C for the lipid bilayers studied in this work and the corresponding activation energy is listed in Table 1.
FIGURE 5.
Time course of the bleaching of NBD-DMPE associated with LUV membranes by sodium dithionite at 35°C. The curves are for LUVs prepared from ♦, POPC (ld phase); ▪ an equimolar mixture of POPC and cholesterol (lo phase); and •, a 6:4 (molar ratio) mixture of SpM and cholesterol (lo phase). The rates of translocation were obtained from the initial rates (also shown). The recovered rate constants are presented in Table 1.
FIGURE 6.
Arrhenius plots for the translocation of NBD-DMPE between the monolayers of LUVs prepared from ♦, POPC (ld phase); ▪, an equimolar mixture of POPC and cholesterol (lo phase); and •, a 6:4 (molar ratio) mixture of SpM and cholesterol (lo phase). The lines are the linear fits to the two independent experiments shown and the recovered parameters are presented in Table 1.
TABLE 1.
Kinetic and thermodynamic constants for insertion and desorption of NBD-DMPE into/from lipid bilayer membranes at 308 K
| POPC | POPC-Cholesterol (1:1) | SpM-Cholesterol (6:4) | |
|---|---|---|---|
| k+ (M−1 s−1) | (2.3 ± 0.1) × 106 | (2.6 ± 0.2) × 106 | (1.8 ± 0.1) × 105 |
| k− (s−1) | (2.8 ± 1.2) × 10−5 | (4.6 ± 1.0) × 10−5 | (1.4 ± 0.2) × 10−5 |
| kf (s−1) | 4.0 × 10−4 | 1.4 × 10−4 | 3.4 × 10−5 |
| KL (M−1) | 8.2 × 1010 | 5.6 × 1010 | 1.3 × 1010 |
| ΔGo (kJ mol−1) | −64 | −63 | −59 |
| ΔHo (kJ mol−1) | −31 | −30 | 7 |
| TΔSo (kJ mol−1) | 33 | 34 | 69 |
| Eact (desorb) (kJ mol−1) | 121 | 106 | 90 |
| Eact (insert) (kJ mol−1) | 87 | 74 | 99 |
 (kJ mol−1) (kJ mol−1) |
102 | 101 | 104 |
 (kJ mol−1) (kJ mol−1) |
119 | 104 | 87 |
 (kJ mol−1) (kJ mol−1) |
16 | 3 | −17 |
 (kJ mol−1) (kJ mol−1) |
38 | 38 | 45 |
 (kJ mol−1) (kJ mol−1) |
85 | 71 | 97 |
 (kJ mol−1) (kJ mol−1) |
46 | 33 | 52 |
| Eact (transloc) (kJ mol−1) | 85 | 87 | 53 |
| KP(L/W) | 1.0 × 106 | 7.1 × 105 | 1.7 × 105 |
Insertion and desorption rate constants and their temperature dependence
Insertion and desorption rate constants were measured in three different liquid phase lipid membrane systems between 15° and 35°C. The lipid membranes were either in an ld phase prepared from pure POPC, or in one of two lo phases prepared from a binary mixture of POPC and cholesterol (1:1 molar ratio) or from a binary mixture of SpM and cholesterol (6:4 molar ratio). The latter lo phase is similar to the lipid raft phase in biological membranes (Simons and Vaz, 2004). Arrhenius plots for the insertion and desorption steps are shown in Fig. 7 for the three types of lipid bilayers examined. Table 1 lists the kinetic rate constants at 308 K and the derived equilibrium constants and thermodynamic parameters. The activation free energies, enthalpies, and entropies, calculated from the Arrhenius plots and using transition state theory (Steinfeld et al., 1999), are also listed in Table 1.
FIGURE 7.
Arrhenius plots for (A) insertion, and (B) desorption of NBD-DMPE into/from lipid bilayers. Data are the average and standard deviation of five independent experiments for ♦, POPC (ld phase); ▪, an equimolar mixture of POPC and cholesterol (lo phase); and •, a 6:4 (molar ratio) mixture of SpM and cholesterol (lo phase), in each panel. The lines are weighted linear fits to the data and the recovered parameters are presented in Table 1.
Equilibrium association constants and membrane/water equilibrium partition coefficients
From the measured rate constants we derived values of the equilibrium association constants, KL, for the association of aqueous phase monomeric NBD-DMPE with ld phase LUVs prepared from POPC and lo phase LUVs prepared from a binary POPC-cholesterol (1:1 molar ratio) mixture and a binary SpM-cholesterol (6:4 molar ratio) mixture. Fig. 8 shows van't Hoff plots for the membrane-association equilibria and the respective thermodynamic parameters are listed in Table 1.
FIGURE 8.
The van't Hoff plots for the association of NBD-DMPE monomers with lipid bilayer membranes. Data are for ♦, POPC (ld phase); ▪, an equimolar mixture of POPC and cholesterol (lo phase); and •, a 6:4 (molar ratio) mixture of SpM and cholesterol (lo phase). The lines are weighted linear fits to the data and the recovered parameters are presented in Table 1.
Since the KL are for association/dissociation equilibria of NBD-DMPE monomers in the aqueous phase with the LUVs, they are related to the equilibrium partition coefficient, KP(L/W), for partitioning of this amphiphile between the lipid phase and the aqueous phase by the expression
 |
where  is the molar volume of the lipid, and it is assumed that there are 105 lipid molecules per LUV. We have assumed a value of 0.795 L mol−1 (Wiener and White, 1992) for
is the molar volume of the lipid, and it is assumed that there are 105 lipid molecules per LUV. We have assumed a value of 0.795 L mol−1 (Wiener and White, 1992) for  for all the lipid bilayers used in this work. This assumption is clearly not strictly applicable to all the systems used here, but will induce only an insignificant error in the calculated values of KP(L/W) reported in Table 1.
for all the lipid bilayers used in this work. This assumption is clearly not strictly applicable to all the systems used here, but will induce only an insignificant error in the calculated values of KP(L/W) reported in Table 1.
Hypothetical partitioning between coexisting lipid phases
From the derived values of KP(L/W), we calculated the hypothetical equilibrium partition coefficients, KP(L1/L2), for partitioning of NBD-DMPE between any two of the lipid phases examined if these phases were to coexist in the same lipid bilayer using the relationship (Abreu et al, 2003) of
 |
The results, as a function of temperature between 15° and 35°C, are reported in Fig. 8 as van't Hoff plots from which we derived the corresponding transfer enthalpies and entropies. The values of the standard free energy for each hypothetical transfer process at 308 K are listed in Table 2. Clearly, the ld and lo phases used here are not compositionally identical with those found in biological membranes but if the compositions of the latter are known, the method described here provides an unambiguous and simple way to characterize partition equilibria of fluorescent lipid amphiphiles in cell membranes.
TABLE 2.
Hypothetical equilibrium partition coefficients and thermodynamic parameters for partitioning of NBD-DMPE between coexisting lipid phases at 299 K
| Hypothetical system | Type of phase coexistence | KP(L1/L2) |
 (kJ mol−1) (kJ mol−1) |
|---|---|---|---|
| POPC/POPC-Cholesterol (1:1) | ld/lo | 1.4 | −1 |
| POPC/SpM-Cholesterol (6:4) | ld/lo | 5.9 | −5 |
| POPC-Cholesterol (1:1)/SpM-Cholesterol (6:4) | lo/lo | 4.2 | −4 |
DISCUSSION
Understanding how amphiphilic molecules interact with lipid bilayers is important from a biological perspective because it provides a baseline for understanding the exchange of chemical components between the membranes of a cell, between cell membranes and organized lipid aggregates such as lipoproteins, and between cell membranes and proteins. From the pharmacological perspective it provides an important model for understanding the processes underlying the interaction of amphiphilic drugs with cells and tissues. From a physicochemical perspective it is an important tool in describing the physical properties of lipid bilayers that constitute various types of cellular membranes or coexist within the same membrane. In particular, systematic studies of amphiphile interaction with lipid bilayers from the kinetic and thermodynamic perspectives have an important predictive value. It is, therefore, not surprising that this field has been actively investigated within the past few decades.
A complete description of the association of amphiphiles with lipid bilayer membranes requires that the association and dissociation rate constants, the equilibrium constants, and the thermodynamic properties of the interaction be quantitatively defined. Such a definition requires that the temperature-dependence of at least two of the three parameters, namely, forward and reverse reaction rate constants and equilibrium association constant, be directly measured. Amphiphile aggregation in the aqueous phase complicates these measurements due to the complex kinetics of monomer-aggregate equilibria. Thus, experimental conditions have to be found in which the only free amphiphile species in aqueous solution that can interact with the membrane in the reaction mixture is the monomer. This is particularly a problem for amphiphiles with a very low CAC (≤10−8 M) such as phospholipids and their derivatives as well as cholesterol. Consequently, the number of reports in which the required two out of three variables have been directly measured for the interaction of phospholipids and their derivatives with membranes is very limited (Nichols, 1985).
The use of simultaneous equilibria of an amphiphile in the aqueous phase between a membrane and a protein that does not have a catalytic amphiphile-exchange function is one way to circumvent the problems involved (Abreu et al., 2003). If the binding and dissociation rate constants and the equilibrium association constant(s) for amphiphile interaction with the protein are independently known, the protein concentration can be adjusted so that the only free amphiphile species in aqueous solution is the monomer. The direct measurement of the rate constants for transfer of the amphiphile to membranes added to the mixture is then rather straightforward. BSA binds the phospholipid derivative, NBD-DMPE, rather strongly (KB ≈ 106 M−1) so that 2 × 10−4 M BSA (∼13 mg/mL) in the solution can reduce the free amphiphile concentration to <0.2% of its total concentration in the system. This reduces the NBD-DMPE concentration in the aqueous phase under our experimental conditions to <2 × 10−9 M, which is well below its CAC of ∼5 × 10−9 M. In this work we have used NBD-DMPE concentrations in the 1 × 10−6 M range to maximize the signal/noise ratio since the primary aim in this work is to validate the method. However, under our experimental conditions, it is possible to reduce the amphiphile concentration by up to a factor of 15-fold, making it viable to use similar amphiphiles with a CAC in the range of 1 × 10−10 M. In previous work we had shown that the transfer of BSA-bound amphiphiles to membranes occurs via monomers in the aqueous phase and not through a collisional interaction between the amphiphile-loaded protein and the membrane.
We have measured the reaction rate constants for insertion of NBD-DMPE from the aqueous phase into lipid bilayers in the liquid-disordered (pure POPC bilayers) and liquid-ordered (POPC-cholesterol (1:1), and SpM-cholesterol (6:4) bilayers) phases between 15° and 35°C. The equilibrium association constants and the equilibrium partition coefficients, for partitioning of NBD-DMPE between the membrane phases and the aqueous phase, as well as the thermodynamic parameters for passage through the activated state during insertion and desorption, were calculated from these rate constants (Table 1).
The second-order rate constants for insertion of NBD-DMPE into the lipid bilayers examined by us are in the range of 104 M−1 s−1 to 106 M−1 s−1, with the values for ld phase POPC membranes and lo phase POPC-Chol membranes being very similar to each other and those for lo phase SpM-Chol membranes notably lower. Under our experimental conditions the calculated diffusion-controlled second-order rate constant for encounter between the NBD-DMPE molecule and the LUV is ∼2 × 1011 M−1 s−1. The difference between the two values reflects the fact that insertion is not synonymous with encounter. Instead, upon encounter, the probe probably executes a random walk on the LUV surface until it encounters an adequate free volume that allows its insertion. Not encountering such a free volume it would diffuse back into the aqueous phase. Its residence time on the LUV surface would depend upon the enthalpy of interaction with this surface. Although our experimental conditions did not permit an exact estimate, we were able to conclude that 
The rate constants measured by us for desorption of NBD-DMPE from the lipid bilayers examined are in the range of 10−6 to 5 × 10−5 s−1. These values are lower than the values measured by other workers. At 35°C, McLean and Phillips (1984) have reported k− ≈ 8 × 10−5 s−1 for desorption of DMPC from small unilamellar vesicles of DMPC, Wimley and Thompson (1990) reported k− ≈ 6 × 10−5 s−1 for desorption of DMPC from DMPC LUVs, and Nichols (1985) reported k− ≈ 0.35 s−1 at 25°C for the desorption of (1-myristoyl-2-[6-[(7-nitro-2,1,3-benzoxadiazol-4-yl)amino]caproyl]phosphatidylcholine), an alkyl chain-labeled NBD-PC, from DOPC vesicles. Our results for the desorption rate constant are similar to those reported by McLean and Phillips (1984) and those of Wimley and Thompson (1990). The far more rapid desorption of NBD-PC from lipid bilayers (Nichols, 1985) probably results from packing difficulties of the bulky NBD group in the hydrophobic portion of this probe. The use of NBD-PC as a model for phospholipid behavior must, therefore, be treated with caution.
The free energy of association of NBD-DMPE with LUVs of POPC (ld phase) and POPC-Chol (lo phase) has approximately equal enthalpic and entropic contributions whereas that for LUVs of SpM-Chol (lo phase) is dominated by entropy (Table 1). A large part of the entropic contribution may be understood to be a result of the hydrophobic effect (Tanford, 1991). However, the entropic contribution due to the hydrophobic effect alone would not be expected to be significantly dependent upon the type of membrane into which the NBD-DMPE was being inserted. We understand the high entropy of association in the case of lo phase SpM-Chol membranes to imply a decrease in order of this phase upon insertion of the probe and a consequent increase in the entropy of the membrane which adds on to the entropic contribution from the hydrophobic effect. The small positive enthalpy of association in this case would appear to confirm the proposed decrease in order. In the case of ld phase POPC membranes and lo phase POPC-Chol membranes, insertion of NBD-DMPE would appear to increase the order of these systems as evidenced by the appreciable, although not dominant, enthalpic contribution to the association free energy. It is of particular interest to note the similarity of ld phase POPC membranes and lo phase POPC-Chol membranes in what concerns the thermodynamics of NBD-DMPE association with them. The discrepancy between the values of the thermodynamic parameters reported here by us for NBD-DMPE association with POPC membranes (Table 1) and the considerably lower values for the same parameters reported by Nichols (1985) for NBD-PC association with DOPC membranes is understood on the basis of the probe structure. The NBD-labeled chain in NBD-PC is probably not very soluble in the apolar membrane interior so that this probe acts more like a lysophosphatidylcholine than like a diacyl phospholipid (Huster et al., 2001). Recent results from our laboratory (J. Sampaio, unpublished results) on the association of NBD-lysoPE with DOPC membranes lend support to this conclusion.
Table 1 lists the thermodynamic parameters of activation for the processes of desorption and insertion. For the desorption step  is ∼100 kJ mol−1. The process is dominated in all cases by the enthalpic contribution,
is ∼100 kJ mol−1. The process is dominated in all cases by the enthalpic contribution,  which is highest in the case of the ld phase POPC bilayer and lowest in the case of the lo phase SpM-cholesterol bilayer. ΔS‡o, in the process of desorption of the probe from the lipid bilayer to the transition state, involves a small increase in the case of the ld phase POPC bilayer; is almost zero for the lo phase POPC-Chol bilayer; and involves a small decrease in
which is highest in the case of the ld phase POPC bilayer and lowest in the case of the lo phase SpM-cholesterol bilayer. ΔS‡o, in the process of desorption of the probe from the lipid bilayer to the transition state, involves a small increase in the case of the ld phase POPC bilayer; is almost zero for the lo phase POPC-Chol bilayer; and involves a small decrease in  for the SpM-Chol lo phase bilayer. The values are comparable to those reported by Wimley and Thompson (1990) for desorption of DMPC from ld phase DMPC LUVs.
for the SpM-Chol lo phase bilayer. The values are comparable to those reported by Wimley and Thompson (1990) for desorption of DMPC from ld phase DMPC LUVs.
ΔG‡o for the insertion step is on the order of 40 kJ mol−1, being comparable for the ld phase POPC bilayer and the lo phase POPC-cholesterol bilayer and slightly higher for the most ordered lo phase SpM-cholesterol bilayer. The formation of the activated state is dominated by an enthalpic contribution. However, the increase in entropy associated with formation of the activated state, roughly half of the enthalpic contribution in all cases, is also significant.
We understand these results on the basis of a model for the transition state that has been widely utilized in the literature (Aniansson et al., 1976; Nichols, 1985; Jones and Thompson 1990), described pictorially in Fig. 9, to explain the activation energetics of amphiphile desorption from lipid micelles and bilayers. If we consider first the desorption process, the transition state is assumed to be one in which the molecule exiting the lipid bilayer is almost totally out of the bilayer. The consequence is that the transition state requires the simultaneous existence of two cavities, one in the lipid bilayer caused by removal of the test particle and the other, in the aqueous phase immediately above, into which the test particle is now inserted. The creation of these two cavities results in an increase in the enthalpy. Displacement of a few layers of ordered water molecules from the bilayer surface to the bulk aqueous phase in creating the aqueous phase cavity also results in an increase in the entropy of the system. On the other hand, the water around the cavity in which the exiting molecule in the transition state encounters itself is more ordered than the bulk water, so that there is a compensatory reduction in the entropy of the system. There is also an increase in the entropy as a consequence of the increased liberty of the lipid molecules around the cavity in the bilayer. Everything else being equal, the increase in entropy in forming the transition state is expected to be largest for the least ordered lipid bilayer which is, in fact, seen to be the case (Table 1).
FIGURE 9.
Model for the passage through the transition state during the transfer of NBD-DMPE between the aqueous and membrane phases, and vice versa.
If we assume the transition state to be the same in desorption and insertion processes, the insertion starts with an amphiphile molecule in the aqueous phase surrounded by an ordered layer of water molecules of unknown thickness around it. The transition state has two cavities, one in the aqueous phase and the other in the lipid bilayer, the creation of which increases the enthalpy of the system. At the bilayer surface, a fraction of the ordered water at the membrane-water interface, as well as a part of the ordered water shell surrounding the amphiphile in the aqueous phase, will be dislocated to the bulk aqueous phase, resulting in an increase in the entropy of the system. To this will be added the increase in entropy resulting from the increased liberty of the lipid molecules around the cavity in the lipid bilayer. There is no compensatory decrease in entropy in this case. The increase in entropy in going to the transition state thus makes a very significant contribution to 
Partition equilibria of membrane components between lo and ld phases are of particular interest since their role in membrane physiology involving “raft” domains may be important (Simons and Vaz, 2004). These equilibria are difficult to quantify by most routinely used methods due to low signal/noise ratios and/or complicated and often poorly understood analytical requirements (Vaz and Melo, 2001). The use of large molar fractions of quenchers (usually spin-labeled or brominated lipid derivatives) which dominate the composition of an ld phase in biphasic (lo−ld or so−ld phase-coexistence) lipid bilayers has been used to obtain partition coefficients for the partition behavior of fluorescent lipid derivatives between ordered and disordered lipid membrane phases (London and Feigenson, 1981; Florine and Feigenson, 1987; Yeager and Feigenson, 1990; Silvius, 1990; 1992; Wang and Silvius, 2001). This method, although it provides important insights into the partitioning equilibrium in biphasic lipid bilayer systems, suffers from the drawback that the lipid composition of the systems studied is, by definition, unnatural and may not be quantitatively representative of what could happen in a biological membrane. The effective dipolar potential of a membrane phase may, for example, have very important effects upon the partitioning behavior in a multiphase membrane (Estronca et al., 2002). An alternative is the determination of partition coefficients through fluorescence resonance energy transfer between donor and acceptor fluorophores in a resonance-energy-transfer pair (see, for example, Feigenson, 1997). In this case, unless the partition coefficient of one of the fluorophores (donor or acceptor) is independently known, only a relative partition coefficient (that compares several donors or acceptors to each other) can be determined. Our determination of KP(L1/L2) from a measured KP(L/W) also suffers from the drawback (common to all methods that use probes) that we are measuring the partition of a fluorescent derivative and not a biologically relevant structure. However, the membranes used can be biological or any convenient model that adequately approximates to them. Besides, this approach gives well-defined quantitative information in what concerns the individual KP(L/W) values determined.
In this work we have examined a hypothetical partitioning of NBD-DMPE between ld and lo phases or between two lo phases that do not necessarily coexist in the same membrane. With reference to the recently published phase diagram of lipid bilayer membranes formed from a ternary mixture of SpM, POPC, and Chol (de Almeida et al, 2003), the chemical compositions of the membranes used in this work are the three limiting compositions of liquid phases at temperatures below 37°C. These particular liquids would not coexist in the same membrane if it were constituted from these three chemical constituents. Any determination of KP for membranes with phase coexistence prepared from a given ternary mixture of SpM, POPC, and Chol would have to examine membranes with the corresponding ld and lo phase compositions (read off from the phase diagram) independently, and then confirm that the membranes with phase coexistence showed association kinetics that were a simple weighted average of the kinetics for the two coexisting phases. The physical nature and chemical composition of the phase boundaries relative to those of the coexisting phases could result in significant deviations from a simple weighted average (Pokorny et al, 2000).
In Table 2 we have listed KP and ΔGo for partitioning of NBD-DMPE between the membrane phases studied in this work. We note, again, that the present results point to a considerable similarity between the ld phase membranes of POPC and the lo phase membranes of POPC-Chol, both in the kinetics of NBD-DMPE association with both bilayer systems and in the thermodynamics of transfer of this probe from the aqueous phase to the membranes and to its transfer between these lipid bilayers.
In this work we have also reported the rate constants, kf, and the activation energies for spontaneous translocation of NBD-DMPE between the monolayers of lipid bilayer membranes in ld and lo phases. The rates measured by us are compatible with results previously reported in the literature (Wimley and Thompson, 1990; Moss and Bhattacharya, 1995; John et al., 2002). For all the membranes studied, the translocation of NBD-DMPE was found to occur on the same timescale as the equilibration between the water and the membrane. Thus, the translocation may be expected to affect the kinetics and the equilibrium and Eq. 3 is not valid. The efficient translocation affects the equilibrium value of ALV because only the probe in the outer half of the bilayer may equilibrate with the protein. At equilibrium the total concentration of probe in the membrane is given by [ALV](∞)=2KL[LV][A]T/1+2KL[LV]+KB[B] in contrast with Eq. 3. The kinetics of the insertion will, under those conditions, be biexponential. However, the kinetic profiles for the interaction of NBD-DMPE with all the membranes studied do not deviate significantly from a monoexponential. This is due to the high values of KL and reflects the fact that the additional amount of probe that inserts into the bilayer, due to the translocation into the inner leaflet, is negligible under the experimental conditions. Therefore, if Eq. 3 were used to fit the data, the fits would be reasonable but the values recovered for k− would be approximately one-half the true values for all the cases studied.
Although it is tempting to discuss the activation energetics of the translocation process, and the presented data would allow it for the case of NBD-DMPE, we shall not do so here, but instead reserve this discussion for a more detailed study in which apolar structure will be systematically varied.
In previous work (Mesquita et al., 2000) we examined the partitioning of a homologous series of NBD-PE, with different acyl chain lengths and saturation, between so and ld phase membranes to evaluate how these parameters affected partitioning. The extension of that work to ld−lo phase partitioning was attempted and judged to lack sufficient accuracy to have any useful predictive value. The method described in this work clearly does not have that deficiency and is currently being used by us to quantitatively define the dependence of partitioning (between coexisting ld and lo phases), of lipid or lipid-attached particles, upon the acyl chain length and saturation. In other recent work we showed that the partitioning of amphiphiles between ld and lo phases was dependent upon the magnitude of the dipole moment of the amphiphile and its orientation relative to the dipolar potential of the host lipid phase (Estronca et al., 2002). We are presently also systematically studying this aspect.
Acknowledgments
This work was supported through research grants from the POCTI program of the Fundacao para a Ciencia e a Tecnologia (FCT) of the Portuguese Ministry for Higher Education and Scientific Research. M.S.C.A. was supported by a stipend for initiation into scientific research (BIC) through projects POCTI/38861/BCI/2001 funded by the FCT.
Winchil Vaz dedicates this article to Thomas E. Thompson, a friend and mentor, who was among those that pioneered the area of research that the work reported here represents.
Abbreviations used: BSA, bovine serum albumin; CAC, critical aggregation concentration; DMPC, 1,2-dimyristoylphosphatidylcholine; DOPC, 1,2-dioleoyl-phosphatidylcholine; KB, equilibrium association constant for the association of an amphiphile with bovine serum albumin; KL, equilibrium association constant for the association of an amphiphile with large unilamellar lipid vesicles; KP, equilibrium partition coefficient; KP(L/W), equilibrium partition coefficient for partitioning between a lipid membrane phase and the aqueous phase; KP(L1/L2), equilibrium partition coefficient for partitioning between two coexisting lipid membrane phases; LUVs, large unilamellar vesicles with an average diameter of 0.1 μm prepared by an extrusion process; ld, liquid-disordered; lo, liquid-ordered; NBD-DMPE, n-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)-amino-1,2-dimyristoylphosphatidyl-ethanolamine; POPC, 1-palmitoyl-2-oleoyl phosphatidylcholine; so, solid-ordered phase; SpM, egg sphingomyelin.
References
- Abreu, M. S. C., L. M. B. B. Estronca, M. J. Moreno, and W. L. C. Vaz. 2003. Binding of a fluorescent lipid amphiphile to albumin and its transfer to lipid bilayer membranes. Biophys. J. 84:386–399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aniansson, E. A. G., S. N. Wall, M. Almgren, H. Hoffmann, I. Klelmann, W. Ulbricht, R. Zana, J. Lang, and C. Tondre. 1976. Theory of the kinetics of micellar equilibria and quantitative interpretation of chemical relaxation studies of micellar solutions of ionic surfactants. J. Phys. Chem. 80:905–922. [Google Scholar]
- Bartlett, G. R. 1959. Phosphorous assay in column chromatography. J. Biol. Chem. 234:466–468. [PubMed] [Google Scholar]
- Brown, D. A., and E. London. 1998. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 14:111–136. [DOI] [PubMed] [Google Scholar]
- Brown, R. E. 1992. Spontaneous lipid transfer between organized lipid assemblies. Biochim. Biophys. Acta. 1113:375–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Almeida, R. F. M., A. Fedorov, and M. Prieto. 2003. Sphingomyelin/phosphatidylcholine/cholesterol phase diagram: boundaries and composition of lipid rafts. Biophys. J. 85:2406–2416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estronca, L. M. B. B., M. J. Moreno, M. S. C. Abreu, E. Melo, and W. L. C. Vaz. 2002. Solubility of amphiphiles in membranes: influence of phase properties and amphiphile head group. Biochem. Biophys. Res. Commun. 296:596–603. [DOI] [PubMed] [Google Scholar]
- Feigenson, G. W. 1997. Partitioning of a fluorescent phospholipid between fluid bilayers: dependence on host lipid acyl chains. Biophys. J. 73:3112–3121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrell, J. E., K. J. Lee, and W. H. Huestis. 1985. Lipid transfer between phosphatidylcholine vesicles and human erythrocytes: exponential decrease in rate with increasing acyl chain length. Biochemistry. 24:2857–2864. [DOI] [PubMed] [Google Scholar]
- Florine, K. I., and G. W. Feigenson. 1987. Protein redistribution in model membranes: clearing of M13 coat protein from calcium-induced gel-phase regions in phosphatidylserine/phosphatidylcholine multilamellar vesicles. Biochemistry. 26:2978–2983. [DOI] [PubMed] [Google Scholar]
- Homan, R., and H. J. Pownall. 1988. Transbilayer diffusion of phospholipids: dependence on headgroup structure and acyl chain length. Biochim. Biophys. Acta. 938:155–166. [DOI] [PubMed] [Google Scholar]
- Hope, M. J., M. B. Bally, G. Webb, and P. R. Cullis. 1985. Production of large unilamellar vesicles by a rapid extrusion procedure—characterization of size distribution, trapped volume and ability to maintain a membrane potential. Biochim. Biophys. Acta. 812:55–65. [DOI] [PubMed] [Google Scholar]
- Huster, D., P. Muller, K. Arnold, and A. Herrmann. 2001. Dynamics of membrane penetration of the fluorescent 7-nitro-2-oxa-1,3-diazol-4-yl (NBD) group attached to an acyl chain of phosphatidylcholine. Biophys. J. 80:822–831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- John, K., S. Schreiber, J. Kubelt, A. Hermann, and P. Müller. 2002. Transbilayer movement of phospholipids at the main phase transition of lipid membranes: implications for rapid flip-flop in biological membranes. Biophys. J. 83:3315–3323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones, J. D., and T. E. Thompson. 1989. Spontaneous phosphatidylcholine transfer by collision between vesicles at high lipid concentration. Biochemistry. 28:129–134. [DOI] [PubMed] [Google Scholar]
- Jones, J. D., and T. E. Thompson. 1990. Mechanism of spontaneous, concentration-dependent phospholipid transfer between bilayers. Biochemistry. 29:1593–1600. [DOI] [PubMed] [Google Scholar]
- Koynova, R., and M. Caffrey. 1998. Phases and phase transitions of the phosphatidylcholines. Biochim. Biophys. Acta. 1376:91–145. [DOI] [PubMed] [Google Scholar]
- London, E., and G. W. Feigenson. 1981. Fluorescence quenching in model membranes. 1. Characterization of quenching caused by a spin-labeled phospholipid. Biochemistry. 20:1932–1938. [DOI] [PubMed] [Google Scholar]
- Lowry, O. H., N. J. Rosebrough, A. L. Farr, and R. J. Randall. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–275. [PubMed] [Google Scholar]
- Massey, J. B., A. M. Gotto, and H. J. Pownall. 1982. Kinetics and mechanism of the spontaneous transfer of fluorescent phospholipids between apolipoprotein-phospholipid recombinants: effect of the polar headgroup. J. Biol. Chem. 257:444–448. [PubMed] [Google Scholar]
- Mateo, C. R., J. C. Brochon, M. P. Lillo, and A. U. Acuna. 1993. Liquid crystalline phases of cholesterol/lipid bilayers as revealed by the fluorescence of trans-parinaric acid. Biophys. J. 65:2237–2247.8298047 [Google Scholar]
- McLean, L., and M. C. Phillips. 1981. Mechanism of cholesterol and phosphatidylcholine exchange or transfer between unilamellar vesicles. Biochemistry. 20:2893–2900. [DOI] [PubMed] [Google Scholar]
- McIntyre, J. C., and R. G. Sleight. 1991. Fluorescence assay for phospholipid membrane asymmetry. Biochemistry. 30:11819–11827. [DOI] [PubMed] [Google Scholar]
- McLean, L. R., and M. C. Phillips. 1984. Kinetics of phosphatidylcholine and lyso-phosphatidylcholine exchange between unilamellar vesicles. Biochemistry. 23:4624–4630. [DOI] [PubMed] [Google Scholar]
- Mesquita, R. M. R. S., E. Melo, T. E. Thompson, and W. L. C. Vaz. 2000. Partitioning of amphiphiles between coexisting ordered and disordered phases in two-phase lipid bilayer membranes. Biophys. J. 78:3019–3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss, R. A., and S. Bhattacharya. 1995. Transverse membrane asymmetry in model phospholipid bilayers: NBD-phosphatidylethanolamine and the separation of flip from flop. J. Am. Chem. Soc. 117:8688–8689. [Google Scholar]
- Nichols, J. W. 1985. Thermodynamics and kinetics of phospholipid monomer-vesicle interaction. Biochemistry. 24:6390–6398. [DOI] [PubMed] [Google Scholar]
- Nichols, J. W., and R. E. Pagano. 1981. Kinetics of soluble lipid monomer diffusion between vesicles. Biochemistry. 20:2783–2789. [DOI] [PubMed] [Google Scholar]
- Nichols, J. W., and R. E. Pagano. 1982. Use of resonance energy transfer to study the kinetics of amphiphile transfer between vesicles. Biochemistry. 21:1720–1726. [DOI] [PubMed] [Google Scholar]
- Peters, T., Jr. 1997. All About Albumin: Biochemistry, Genetics, and Medical Applications. Academic Press, San Diego, CA.
- Pokorny, A., P. F. F. Almeida, E. C. C. Melo, and W. L. C. Vaz. 2000. Kinetics of amphiphile association with two-phase lipid bilayer vesicles. Biophys. J. 78:267–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pokorny, A., P. F. F. Almeida, and W. L. C. Vaz. 2001. Association of a fluorescent amphiphile with lipid bilayer vesicles in regions of solid-liquid-ordered phase coexistence. Biophys. J. 80:1384–1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sankaram, M. B., and T. E. Thompson. 1990. Interaction of cholesterol with various glycerophospholipids and sphingomyelin. Biochemistry. 29:10670–10675. [DOI] [PubMed] [Google Scholar]
- Silvius, J. R. 1990. Calcium-induced phase separations and interactions of phosphatidylcholine/anionic phospholipid vesicles. Fluorescence studies using carbazole-labeled and brominated phospholipids. Biochemistry. 29:2930–2938. [DOI] [PubMed] [Google Scholar]
- Silvius, J. R. 1992. Cholesterol modulation of lipid intermixing in phospholipid and glycosphingolipid mixtures. Evaluation using fluorescent lipid probes and brominated lipid quenchers. Biochemistry. 31:3398–3408. [DOI] [PubMed] [Google Scholar]
- Silvius, J. R., and R. Leventis. 1993. Spontaneous interbilayer transfer of phospholipids: dependence on acyl chain composition. Biochemistry. 32:13318–13326. [DOI] [PubMed] [Google Scholar]
- Simons, K., and E. Ikonen. 1997. Functional rafts in cell membranes. Nature. 387:569–572. [DOI] [PubMed] [Google Scholar]
- Simons, K., and W. L. C. Vaz. 2004. Model systems, lipid rafts and cell membranes. Annu. Rev. Biophys. Biomol. Struct. 33:269–295. [DOI] [PubMed] [Google Scholar]
- Steinfeld, J. I., J. S. Francisco, and W. L. Hase. 1999. Chemical Kinetics and Dynamics, 2nd Ed. Prentice Hall, Upper Saddle River, NJ. 300–302.
- Tanford, C. 1991. The Hydrophobic Effect: Formation of Micelles and Biological Membranes, 2nd Ed. Krieger Publishing, Malabar, FL.
- Taylor, R. P., A. V. Broccoli, and C. M. Grisham. 1978. Enzymatic and colorimetric determination of total serum cholesterol. J. Chem. Educ. 55:63–64. [DOI] [PubMed] [Google Scholar]
- Vaz, W. L. C., and P. F. F. Almeida. 1993. Phase topology and percolation in multi-phase lipid bilayers. Is the biological membrane a domain mosaic? Curr. Opin. Struct. Biol. 3:482–488. [Google Scholar]
- Vaz, W. L. C., and E. Melo. 2001. Fluorescence studies on phase heterogeneity in lipid bilayer membranes. J. Fluorescence. 11:255–271. [Google Scholar]
- Wang, T. Y., and J. R. Silvius. 2001. Partitioning of lapidated peptide sequences into liquid-ordered lipid domains in model and biological membranes. Biochemistry. 40:13031–13040. [DOI] [PubMed] [Google Scholar]
- Wiener, M. C., and S. H. White. 1992. Structure of a fluid dioleoylphosphatidylcholine bilayer determined by joint refinement of x-ray and neutron diffraction data. Distribution and packing of terminal methyl groups. Biophys. J. 61:428–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wimley, W. C., and T. E. Thompson. 1990. Exchange and flip-flop of dimyristoyl phosphatidylcholine in liquid-crystalline, gel, and two-component, two-phase large unilamellar vesicles. Biochemistry. 29:1296–1303. [DOI] [PubMed] [Google Scholar]
- Yeager, M. D., and G. W. Feigenson. 1990. Fluorescence quenching in model membranes: phospholipid acyl chain distributions around small fluorophores. Biochemistry. 29:4380–4392. [DOI] [PubMed] [Google Scholar]