CME, Part 2 of 3
Target audience: All physicians
Learning objectives:
Identify prophylaxis and treatment options for anthrax, botulism, plague, and smallpox.
Discuss how an institution and an emergency department should respond to a bioterrorism incident.
Understand psychological aspects of a bioterrorism incident.
Faculty credentials/disclosure:
Captain Malone is an infectious disease physician; fellow and member of the Emerging Infection Committee of the Infectious Diseases Society of America; member of the Bioterrorism Task Force of the Association for Professionals in Infection Control; associate professor in the Uniformed Services University of the Health Sciences; member of the Executive Steering Committee and goal champion of the Environment of Care Strategic Planning Board, Naval Medical Center San Diego; past consultant of the Chemical Biological Incident Response Force, US Marine Corps; and past chairman, Nuclear Biological Chemical Response Planning Group, National Naval Medical Center. Medication recommendations do not represent uses currently approved by the US Food and Drug Administration or an official position on the part of the US Navy or Department of Defense. Captain Malone has no significant relationships with pharmaceutical or medical device companies.
Before beginning this activity, please read the instructions for CME on p. 333. This page also provides important information on method of physician participation, estimated time to complete the educational activity, medium used for instruction, and date of release and expiration. The quiz, evaluation form, and certification appear on pp. 333–335.
Dallas, Texas, and the surrounding community is certainly at risk for a bioterrorist attack. Dallas is the eighth largest city in the USA with the third largest international airport and is host to major events such as the Cotton Bowl or possibly the Olympic Games. Terrorists target critical infrastructure or major events attended by thousands of people. Other events placing cities at risk include meetings of major political leaders or parties and the presence of federal or state government buildings or military bases.
An appropriate planned response to a bioterrorist attack will minimize morbidity and mortality. The response centers on a public health approach, as in any other infectious disease outbreak. The basic steps remain the same: identify the organism, form a case definition, identify cases, plot cases daily on a curve, and develop effective prevention strategies, including vaccinations, antimicrobial medications, and case cohorting and isolation. Each year, the same techniques are used to respond to the influenza epidemic that infects millions, with an annual mortality that exceeds 20,000 people.
The risk of bioterrorism comes from several countries, as well as from misguided Americans. According to a 1993 report (Office of Technology Assessment, US Congress), Iran, Iraq, Israel, North Korea, China, Libya, Syria, and Taiwan all have access to biological agents. Fortunately, efforts at developing these agents into large-scale weapons are complicated by the need for an effective delivery system. A simple delivery method is use of a commercial agricultural sprayer in a small airplane or vehicle to create a line source aerosol dispersion upwind from a large population center. Dissemination of agents from a point source explosion such as bombs or missiles is not as effective in infecting a large number of people. For example, an aircraft carrying an advertising banner could follow the parade route of the Cotton Bowl and disperse 100 kg of anthrax spores. On a clear, sunny day, such a release may cause over 100,000 casualties; on an overcast day or calm night, up to 1 million casualties. Anthrax is the most lethal of all potential biological weapons (Table1). In contrast, aerial release of 1000 kg of sarin gas would cause several hundred casualties.
Table 1.
Lethality of possible bioterrorism agents based on airplane dissemination of 50 kg of the agent along a 2-km line upwind for a population center of 500,000*
| Agent | Downward reach (km) | Dead | Incapacitated |
| Anthrax | ≫≫20 | 95,000 | 125,000 |
| Tularemia | ≫20 | 30,000 | 125,000 |
| Q fever | >20 | 150 | 125,000 |
| Brucellosis | 10 | 500 | 100,000 |
| Typhus | 5 | 19,000 | 85,000 |
*Modified from World Health Organization. Health Aspects of Chemical and Biological Weapons, 1970.
The focus of this article is on treatment and prophylaxis of 4 agents that are of particular concern: anthrax, botulinum toxin, plague, and smallpox. Some specific guidelines for institutional preparedness and prevention of psychological sequelae are also addressed.
ANTHRAX
Bacillus anthracis forms spores. The spores exist in a viable state almost indefinitely, since they require neither a stable temperature nor growth medium. Being 3 to 5 microns in size, the spores can lodge in the terminal alveoli. Larger particles tend to get trapped in the upper airways, and smaller particles move in and out of the airways without being deposited. When the spores germinate, they produce viable bacilli, and the bacilli produce toxins such as edema factor and lethal factor. The lethal dose for 50% of individuals is estimated at 4000 spores, an amount inhaled in one breath. These characteristics make anthrax an effective agent for bioterrorism.
Spores deposited in the alveoli are transported to the mediastinum by macrophages. Upon germination, the toxins produce edema, hemorrhagic necrosis, and mediastinitis. Inhalational anthrax can be recognized radiographically by a wide mediastinum and clear lung fields. Small pleural effusions may be present. Meningitis through bloodstream transmission is also possible.
Anthrax may first be suspected in the laboratory by a positive blood culture with anaerobic gram-positive rods. The organism should not be dismissed as diphtheroids and skin contaminants, since this is a key identifier of initial patients. When anthrax was accidentally released from a bioweapons production facility in Sverdlovsk, Russia, deaths occurred primarily in older men, many of whom were smokers and welders. This may be a clue about those who are most at risk.
In keeping with the Greek name anthracis, meaning black, cutaneous anthrax manifests with a black eschar and a surrounding area of edema from toxin production. This localized cutaneous infection is not life threatening.
Anthrax spores wash off with soap and water. Anthrax pulmonary infection and cutaneous lesions are not contagious to health care workers; however, pathologists conducting an autopsy require protective respiratory equipment to prevent spore exposure when opening the chest. Any health care worker who receives a needle stick from a potentially bacteremic anthrax infected patient should receive prophylactic antibiotics.
Prophylaxis for aerosol exposure consists of antibiotic medication for 30 days and anthrax toxoid vaccination immediately and 2 and 4 weeks afterwards. If the toxoid vaccination is not available, the exposed person requires antibiotic prophylaxis for 8 weeks. This recommendation comes from the results of a study in rhesus monkeys. After inhalational exposure to 11 times the lethal dose of anthrax spores, the mortality rate was significantly lower (1/9) in monkeys that received ciprofloxacin as post- exposure prophylaxis within 24 hours than in monkeys that received placebo. The one ciprofloxacin-treated monkey that died of anthrax expired after 30 days of antibiotic administration. The toxin, especially the component labeled decades ago as “protective antigen,” is responsible for developing immunity. The antibiotics may not be bactericidal to the intracellular encapsulated spores; they only prevent the spores from becoming viable bacilli and producing toxin with protective immunity. The anthrax toxoid vaccination initiates the immune response.
The suggested antibiotics for prophylaxis are listed in Table 2. Treatment consists of the same antibiotics (Table 3). If the strain of anthrax is susceptible, penicillin is the most effective drug, with a reported minimum inhibitory concentration (MIC) of <0.03. Ciprofloxacin and ofloxacin have also shown good activity in vitro, with MICs of 0.03–0.06. All fluoroquinolones, including levofloxacin and the more recent arrivals such as gatifloxacin and moxifloxacin, will probably be effective. The only fluoroquinolone for which animal efficacy data are available is ciprofloxacin. Ciprofloxacin was recently approved by the Food and Drug Administration (FDA) for anthrax postexposure prophylaxis and treatment. Ciprofloxacin is not FDA approved for children but may be used for postexposure inhalational anthrax in time of crisis until penicillin sensitivity can be confirmed. Over 120 articles have documented ciprofloxacin safety in children with minimal effects on cartilage. Third-generation cephalosporins perform poorly in vitro against anthrax.
Table 2.
Antibiotics recommended for prophylaxis of anthrax in adults and children
| Oral antibiotic | Adults | Chlidren |
| Ciprofloxacin | 500 mg twice a day | 20–30 mg/kg/day divided into 2 doses* |
| Levofloxacin | 500 mg a day | Not recommended |
| Ofloxacin | 400 mg twice a day | Not recommended |
| Doxycycline | 100 mg twice a day | 5 mg/kg/day divided into 2 doses* |
| Amoxicillin | 875 mg twice a day | 40 mg/kg/day every 8 h (max 500 mg every 8 h) |
*Pediatric use of fluoroquinolones and tetracyclines is associated with adverse effects that must be weighed against the risk of a lethal disease. Penicillin is the drug of choice for susceptible strains. Newer fluoroquinolone agents such as gatifloxacin 400 mg once daily or moxifloxacin 400 mg once daily are also likely to be effective.
Table 3.
Antibiotics recommended for treatment of anthrax in adults and children
| Antibiotic* | Adults | Children |
| Ciprofloxacin | 500 mg twice a day | 20–30 mg/kg/day divided into 2 doses (maximum dose, 1000 mg/day)† |
| IV: 400 mg every 12 hours | IV: 10 mg/kg (max 400 mg) every 12 hours | |
| Levofloxacin | 500 mg a day | Not recommended |
| Ofloxacin | 400 mg twice a day | Not recommended |
| Doxycycline | 200 mg, then 100 mg twice a day | 2.5 mg/kg every 12 hours; daypatients ≫45 kg should receive adult dose† |
| Penicillin G | 4 million U every 4 hours | 400,000 U/kg/day divided into 4 doses‡ |
| Amoxicillin | 3 g 3 times a day | 2 g in a 24-hour period (<3 months: maximum 30 mg/kg/day) |
*Quinolones and doxycycline can be delivered orally or intravenously.
†Pediatric use of fluoroquinolones is associated with adverse effects that must be weighed against the risk of a lethal disease. Penicillin is the drug of choice for susceptible strains.
‡Alternative pediatric dose for penicillin G: age <12 years: 50,000 U/kg every 6 hours; age ≫12 years: 4 million U every 4 hours.
Pets can develop anthrax as well; they usually develop the cutaneous form rather than the inhalational form. For most pet exposures, a bath with soap and water is sufficient to remove the spores. In cases of heavy contamination, the owners may wish to bathe animals in a 0.5% hypochlorite solution (household bleach diluted 1 to 10) or Exspor solution (Alcide Corp, Conn). Veterinarians are an important resource in a bioterrorism incident, since a majority of the possible bioterrorism agents originated in early domesticated livestock and then spread to the human population.
BOTULINUM TOXIN
The 7 botulinum toxins produced by Clostridium botulinum are neurotoxins that act presynaptically to prevent the release of acetylcholine. People exposed to this toxin would present with a variety of signs and symptoms, such as dilated pupils, ptosis, diplopia due to extraocular palsies, dysphonia, dysarthria, hypotension, respiratory failure, and paralysis. They would be afebrile. Those who receive a sublethal dose may have delayed symptoms. Mental status changes, such as cognition problems or depression weeks to months later, have been reported. Toxin release should be considered a chemical exposure, since no viable organisms are involved. Antibiotics are not indicated for bioterrorism botulism; ventilatory support may be required. There is no nosocomial transmission, and routine standard precautions for infection control apply.
A vaccine is available for prophylaxis and treatment. An effective pentavalent toxoid vaccine for prophylaxis was developed by the US Army and used under an investigational new drug status. This formalin-fixed culture supernatant, administered subcutaneously immediately, 2 and 12 weeks afterwards, and then yearly, develops antibody titers at 14 weeks with an 80% effectiveness rate. Passive protection can be afforded with a heptavalent antibotulinum toxin immunoglobulin for the 7 serotypes (A through G). Since this immunoglobulin was developed from horse serum, 20% of recipients have a serum sickness reaction, and a skin test for hypersensitivity is recommended. This vaccine is held at the US Army Medical Research Institute of Infectious Diseases (USAMRIID), Fort Detrick, Maryland. In addition, the Centers for Disease Control and Prevention (CDC) has a trivalent equine antitoxin for serotypes A, B, and E, which has been approved by the FDA.
PLAGUE
In a bioterrorism incident, viable Yersinia pestis bacilli would be transmitted through aerosolization, resulting in pneumonic plague, an overwhelming, rapidly advancing, severe gram-negative coccobacillary pneumonia, which may be confused with Haemophilus influenzae on Gram stain. This pneumonic process differs from bubonic plague, which is characterized by regional adenopathy (buboes) draining the site of initial infection after inoculation from an infected flea that obtained a blood meal from a bacteremic animal (i.e., rat, vole, prairie dog). Occasional cases of bubonic plague are reported from the southwestern USA in summer months.
Nosocomial transmission of pneumonic plague is possible until the patient has completed 72 hours of antimicrobial therapy. Until the patient has received effective therapy for at least that period, health care personnel should use droplet precautions, wearing a surgical-type mask when they are within 3 feet of the patient or upon entering the patient's room.
Prophylactic therapy for pneumonic plague exposure involves 7 days of therapy with doxycycline (100 mg twice a day), tetracycline (500 mg 4 times a day), a quinolone, or a combination of trimethoprim and sulfamethoxazole. Historically, therapy for those with active disease has consisted of one of the following: streptomycin (30 mg/kg/day divided into 2 doses intramuscularly for 10 days), gentamicin (3 mg/kg/day), doxycycline (200 mg initially, then 100 mg daily for 10 to 14 days), or chloramphenicol (1 g every 6 hours for 10 to 14 days). The plague vaccine does not protect against aerosolized transmission in animal studies.
SMALLPOX
Smallpox (variola virus) presents as vesicles starting peripherally on the extremities (palms and soles) and face and spreading centrally to the trunk. The lesions are in the same stage at the same time (synchronous). In contrast, varicella (chickenpox) vesicles begin centrally on the trunk and appear in multiple stages. The severity of smallpox varies from patient to patient: some are mildly affected while others have a very serious case. The death rate from smallpox is 30%.
A smallpox patient is infectious from the onset of rash until the scabs separate, a period of about 3 weeks. Airborne precautions (respirators) and contact precautions (gowns, gloves, and antimicrobial handwash) are necessary to minimize transmission.
The only effective prophylaxis available is the vaccinia vaccine, licensed in the USA under the trade name Dryvax (manufactured by Wyeth, Philadelphia, Pa). Vaccination within 3 days of exposure will prevent the disease, and vaccination within 5 days is life saving. Currently, the CDC has 12 to 14 million doses of smallpox vaccine, and the World Health Organization has about 20 million doses. The US government recently set aside $28 million to develop an oral form of smallpox vaccine by the year 2004.
The vaccinia virus in Dryvax has an obscure origin; it is based primarily upon the cowpox virus and may represent a reassortment of smallpox and cowpox viruses. Antigenically similar to smallpox, this reconstituted live infectious virus delivered intradermally through multiple skin passages with a bifurcated needle (scarification process) results in a localized vesicular skin eruption and production of T cells and B-cell antibodies. The current vaccine was created in a unique process. The shaved skin of a calf was scraped and inoculated with vaccinia virus. The external lymphatic drainage from the cutaneous infection was collected, purified, and lyophilized. Because it is a live virus vaccination, complications can occur with dissemination in immunocompromised persons or those with severe eczema (eczema vaccinatum). Ocular autoinoculation is also a risk.
Treatment options for confirmed cases of smallpox are limited. Cidofovir has shown some effectiveness in vitro. The recommended dosage is 5 mg/kg intravenously per week for 2 weeks and then every other week. Prehydration is required. Adefovir dipivoxil is probably not effective, although theoretically it can inhibit replication of variola virus DNA. Vaccinia immune globulin (VIG) (0.6 mL/kg intramuscularly within 3 days of exposure) is another possibility for treatment and may also be useful for complications of smallpox infection. Unfortunately, a majority of the available VIG is in very poor condition.
RESPONSE TO A BIOTERRORISM INCIDENT
Treatment of bioterrorism exposure is multifactorial, multidisciplinary, and longitudinal—similar to the response for multiple trauma victims. Psychological aspects must be addressed.
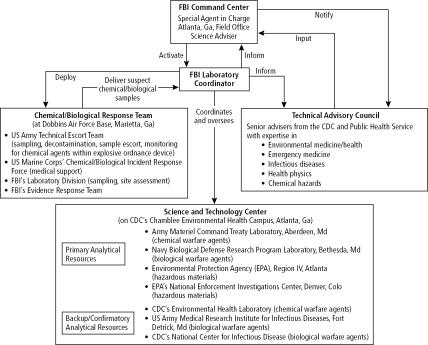
Overall, the Federal Bureau of Investigation (FBI) is in charge of the domestic terrorism response at the crime scene and has access to many other governmental resources (Figure). Throughout the complex “task force” response effort, the FBI will depend upon the local public health department and local health care personnel. A military-style command, control, communications, and intelligence model has limitations in this situation. Intense cooperation, communication, and trust between differing levels of civilian and military authorities are necessary to maximize an effective and timely response. For example, the governor has control of the National Guard and the state health department; the mayor has control of the local police department, fire department, and public health department; and the hospitals and local physicians oversee their patients and personnel. Quarantine authority resides with the local public health officer.
Figure.
Federal analytical capabilities deployed at the 1996 Summer Olympic Games in Atlanta, Georgia. Reprinted with permission from Ember LR. FBI takes lead in developing counterterrorism effort. Chem Eng News 1996(Nov 4):10–16. Copyright 1996 American Chemical Society.
In a bioterrorism incident, health care institutions would be expected to secure and control all access and egress by locking doors and posting guards. Tunnels, walkways, and roof access require special attention. The goal of such security is to prevent hospital contamination and protect the health of medical personnel so that they may continue to provide service. By preventing exit in the setting of communicable diseases such as smallpox or plague (pending vaccinations or antibiotic prophylaxis), transmission of disease from potentially contagious health care workers to their families and the community is minimized.
Institutions should also maintain order with the media. Communication with the public is critical; “not what you say but how you say it” is important. Institutional leaders should consider risk communication training and ensure strong working relationships between the chief executive officer, public affairs spokesperson, legal counsel, and clinical care directors. Risk communication is best done as a dialogue and is more effective when trusted community leaders are involved. Leaders may also want to make use of the Internet to share key messages.
Emergency department personnel are the first line of defense for hospitals in the response to bioterrorism. During influenza outbreaks, they treat hundreds of patients, recording basic demographic data, diagnosing pneumonia, delivering antibiotics, and minimizing morbidity and mortality.
Institutions should ensure that their emergency personnel are prepared for decontamination and triage. Regarding decontamination, the clothing of those who have been exposed should be removed and placed in a plastic bag. Generally, a shower (with warm water to prevent hypothermia and minimize additional distress) with soap is all that is required; bleach is needed only for chemical decontamination.
Somatization disorders following an attack can quickly over-whelm an emergency department. For example, in the 1995 Tokyo sarin attack, 5000 people sought medical treatment: 12 died, 17 were critically injured, and 1300 were affected mildly or moderately. The vast majority of patients had no injuries yet quickly and severely congested the health care delivery system. In such settings, rapid and accurate triage to provide effective treatment, offer a supportive environment, and convey a return to normal activities is most important. Sensitive risk communication will support the efforts. Health care providers need to be protected from extreme stress by work/rest cycles and debriefing.
An uninhabitable condition of the emergency department due to secondary aerosols from a bacterial-contaminated patient is unlikely. For example, an Aberdeen Proving Ground study during the Persian Gulf War estimated that only 120 spores remained on an average-sized adult after heavy exposure to a large anthrax cloud. Spores reside primarily in hair, including men's beards.
If the specific agent released during a bioterrorist attack—and its antibiotic sensitivity—have not been identified, public health departments and health care workers can administer fluoroquinolone antibiotic prophylaxis. Fluoroquinolones are active against anthrax, plague, cholera, tularemia, brucellosis, Q fever, and community-acquired pneumonia. Engineering resistance to quinolones is difficult since they act as DNA gyrase inhibitors. Fluoroquinolones with once-daily dosing would allow daily observed therapy and permit a large number of people to be initially treated with limited regional supplies. The CDC recently spent $52 million stockpiling pharmaceuticals and equipment in 12 different US locations to increase immediate access.
PSYCHOLOGICAL ASPECTS
Mental health resources—social workers, psychologists, psychiatrists, and clergy—will be essential during a bioterrorism incident. Clergy members are especially important because they enjoy a very high level of trust in the community.
The invisible agents of bioterrorism are very frightening to people, and some may have strong responses. Administering antibiotics and immunizations for prophylaxis is also stressful. Some people can demonstrate acute autonomic arousal: muscle tension, tachycardia, hyperventilation, sweating, tremor, and a sense of foreboding. They may attribute these symptoms to infection and be concerned or upset if a health care worker dismisses their complaints. Again, accurate triage is important. Fever is a definitive sign.
Those with somatic symptoms need further care as well. Posttraumatic stress disorder is a possibility, as are depression, panic attack, alcohol/drug abuse, or antisocial behavior. Patients who have had major depression in the past are at risk for a relapse. The criteria for posttraumatic stress disorder include the following: a traumatic stressor exposure, intrusive reexperience (memories, nightmares, reminders), avoidance and numbing (interest loss, detachment, affect blunting), and hyperarousal (insomnia, anger, difficulty concentrating). Sertraline and fluoxetine are effective adjuncts.
CONCLUSION
The major enemies during a bioterrorism incident are fear, panic, stress, and misinformation. Four agents of particular concern are anthrax, botulinum toxin, plague, and smallpox. Institutions require security and decontamination plans. The major defense is the trust developed among colleagues, employees, supervisors, and institutional authorities. We can handle a bioterrorism incident. The better prepared we are, the less likely the event will occur.
Anthrax
- 1.Abramova FA, Grinberg LM, Yampolskaya OV, Walker DH. Pathology of inhalational anthrax in 42 cases from the Sverdlovsk outbreak of 1979. Proc Natl Acad Sci U S A. 1993;90:2291–2294. doi: 10.1073/pnas.90.6.2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bartlett JG, Dowell SF, Mandell LA, File TM, Jr, Musher DM, Fine MJ. Practice guidelines for the management of community-acquired pneumonia in adults. Infectious Diseases Society of America. Clin Infect Dis. 2000;31:347–382. doi: 10.1086/313954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.[Pneumonia in the context of bioterrorism] Centers for Disease Control and Prevention Bioterrorism alleging use of anthrax and interim guidelines for management—United States, 1998. MMWR Morb Mortal Wkly Rep. 1999;48:69–74. [PubMed] [Google Scholar]
- 4.English JF, Cundiff MY, Malone JD, Pfeiffer JA, Bell M, Steele L, Miller M. Bioterrorism readiness plan: a template for healthcare facilities. APIC Bioterrorism Task Force/CDC Hospital Infections Program Bioterrorism Working Group. Available at http://www.apic.org. Accessed April 13, 1999.
- 5.Friedlander A. Anthrax. In: Zajtchuk R, Bellamy RF, editors. Textbook of Military Medicine: Medical Aspects of Chemical and Biological Warfare. Washington, DC: Office of the Surgeon General, US Department of the Army; 1997. pp. 467–478. [Google Scholar]
- 6.Gold H. Anthrax: report of 117 cases. Arch Intern Med. 1955;96:387–396. doi: 10.1001/archinte.1955.00250140109012. [DOI] [PubMed] [Google Scholar]
- 7.Inglesby TV, Henderson DA, Bartlett JG, Ascher MS, Eitzen E, Friedlander AM, Hauer J, McDade J, Osterholm MT, O'Toole T, Parker G, Perm TM, Russell PK, Tonat K. Anthrax as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA. 1999;281:1737–1745. doi: 10.1001/jama.281.18.1735. [DOI] [PubMed] [Google Scholar]
- 8.Keim M, Kaufmann AF. Principles for emergency response to bioterrorism. Ann Emerg Med. 1999;34:177–182. doi: 10.1016/s0196-0644(99)70227-1. [DOI] [PubMed] [Google Scholar]
- 9.Knudson GB. Treatment of anthrax in man: history and current concepts. Mil Med. 1986;181:71–71. [PubMed] [Google Scholar]
- 10.Levy LM, Baker N, Meyer MP, Crosland P, Hampton J. Anthrax meningitis in Zimbabwe. Cent Afr J Med. 1981;27:101–104. [PubMed] [Google Scholar]
- 11.Lightfoot NF, Scott RJD, Turnbull BCB. Antimicrobial susceptibility of Bacillus anthracis. Salisbury Med Bull Suppl. 1990;68:95–98. [Google Scholar]
- 12.Meselson M, Guillemin J, Hugh-Jones M, Langmuir A, Popova I, Shelokov A, Yampolskaya O. The Sverdlovsk anthrax outbreak of 1979. Science. 1994;266:1202–1208. doi: 10.1126/science.7973702. [DOI] [PubMed] [Google Scholar]
- 13.Pile J, Malone JD, Eitzen EM, Friedlander AM. Anthrax as a potential biological warfare agent. Arch Intern Med. 1998;158:429–434. doi: 10.1001/archinte.158.5.429. [DOI] [PubMed] [Google Scholar]
- 14.Richards CF, Burstein JL, Waeckerle JF, Hutson HR. Emergency physicians and biological terrorism. Ann Emerg Med. 1999;34:183–190. doi: 10.1016/s0196-0644(99)70228-3. [DOI] [PubMed] [Google Scholar]
- 15.Walker DH, Yampolska O, Grinberg LM. Death at Sverdlovsk: what have we learned? Am J Pathol. 1994;144:1135–1141. [PMC free article] [PubMed] [Google Scholar]
Anthrax vaccine
- 16.Anthrax vaccine adsorbed [package insert]. Lansing, Mich: Michigan Department of Public Health, 1987.
- 17.Brachman PS, Friedlander AM. Anthrax. In: Plotkin SA, Mortimer EA, editors. Vaccines. 2nd ed. Philadelphia: WB Saunders Co; 1994. pp. 729–739. [Google Scholar]
- 18.Brachman PS, Gold H, Plotkin SA, Fekety FR, Werrin M, Ingraham NR. Field evaluation of human anthrax vaccine. Am J Public Health. 1962;52:632–645. doi: 10.2105/ajph.52.4.632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Broster MG, Hibbs SE. Protective efficacy of anthrax vaccines against aerosol challenge. Salisbury Med Bull Suppl. 1990;68:91–92. [Google Scholar]
- 20.Friedlander AM, Welkos SL, Pitt ML, Ezzell JW, Worsham PL, Rose KJ, Ivins BE, Lowe RJ, Howe GB, Mikesell P, et al. Postexposure prophylaxis against experimental inhalation anthrax. J Infect Dis. 1993;167:1239–1243. doi: 10.1093/infdis/167.5.1239. [DOI] [PubMed] [Google Scholar]
- 21.Ivins BE, Fellows PF, Pitt MLM, Estep JE, Welkos SL, Worsham PL, Friedlander AM. Efficacy of standard human anthrax vaccine against Bacillus anthracis aerosol spore challenge in rhesus monkeys. Salisbury Med Bull Suppl. 1996;87:125–125. [Google Scholar]
- 22.Pitt MLM, Ivins BE, Estep JE, Frachus J, Friedlander AM. Comparison of the efficacy of purified protection antigen and MDPH to protect non-human primates from inhalational anthrax. Salisbury Med Bull Suppl. 1996;87:130. [Google Scholar]
- 23.Turnbull PCB. Anthrax vaccines: past, present and future. Vaccine. 1991;9:533–533. doi: 10.1016/0264-410x(91)90237-z. [DOI] [PubMed] [Google Scholar]
Anthrax pathogenesis
- 24.Fritz DL, Jaax NKI, Lawrence WB, Davis KJ, Pitt ML, Ezzell JW, Friedlander AM. Pathology of experimental inhalation anthrax in the rhesus monkey. Lab Invest. 1995;73:691–691. [PubMed] [Google Scholar]
- 25.Hanna PC, Acosta D, Collier RJ. On the role of macrophages in anthrax. Proc Natl Acad Sci U S A. 1993;90:10198–10198. doi: 10.1073/pnas.90.21.10198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leppla SH. Anthrax toxin edema factor: a bacterial adenylate cyclase that increases cyclic AMP concentrations of eukaryotic cells. Proc Natl Acad Sci U S A. 1982;79:3162–3162. doi: 10.1073/pnas.79.10.3162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lincoln RE, Fish DC. Anthrax toxin. In: Montie TC, Kadis S, Aji SJ, editors. Microbial Toxins, vol. 3, Bacterial Protein Toxins. New York: Academic Press; 1970. pp. 316–414. [Google Scholar]
- 28.Ross JM. The pathogenesis of anthrax following the administration of spores by the respiratory route. J Pathol Bact. 1957;73:485–485. [Google Scholar]
- 29.Smith H, Keppie J. Observations on experimental anthrax: demonstration of a specific factor produced in vivo by Bacillus anthracis. Nature. 1954;173:869–869. doi: 10.1038/173869a0. [DOI] [PubMed] [Google Scholar]
Anthrax epidemiology
- 30.Carr EA, Rew R. Recovery of Bacillus anthracis from the nose and throat of apparently healthy workers. J Infect Dis. 1957:169–171. doi: 10.1093/infdis/100.2.169. [DOI] [PubMed] [Google Scholar]
- 31.Dahlgren C, Buchanan LM, Decker HM, Freed SW, Philips CR, Brachman PS. Bacillus anthracis aerosols in goat hair processing mills. Am J Hyg. 1960;72:24–24. doi: 10.1093/oxfordjournals.aje.a120131. [DOI] [PubMed] [Google Scholar]
- 32.Watson A, Keir D. Information on which to base assessments of risk from environments contaminated with anthrax spores. Epidemiol Infect. 1994;133:479–479. doi: 10.1017/s0950268800068497. [DOI] [PMC free article] [PubMed] [Google Scholar]
Botulism
- 33.Franz D. Defense against toxin weapons. In: Zajtchuk R, Bellamy RF, editors. Textbook of Military Medicine: Medical Aspects of Chemical and Biological Warfare. Washington, DC: Office of the Surgeon General, US Department of Army; 1997. pp. 603–619. [Google Scholar]
- 34.Shapiro RL, Hatheway C, Becher J, Swerdlow DL. Botulism surveillance and emergency response. A public health strategy for a global challenge. JAMA. 1997;278:433–433. [PubMed] [Google Scholar]
- 35.Medical Defense Against Biological Warfare Agents [course text]. Fort Detrick, Md: USAMRIID, 1997.
Plague
- 36.Franz DR, Jahrling PB, Friedlander AM, McClain DJ, Hoover DL, Bryne WR, Pavlin JA, Christopher GW, Eitzen EM., Jr Clinical recognition and management of patients exposed to biological warfare agents. JAMA. 1997;278:399–411. doi: 10.1001/jama.278.5.399. [DOI] [PubMed] [Google Scholar]
- 37.McGovern TW, Friedlander AM. Plague. In: Zajtchuk R, Bellamy RF, editors. Textbook of Military Medicine: Medical Aspects of Chemical and Biological Warfare. Washington, DC: Office of the Surgeon General, US Department of the Army; 1997. pp. 479–502. [Google Scholar]
Smallpox
- 38.Board WJ, Miller J. Smallpox stocks hidden, report says, Iraq, N. Korea, Russia cited by intelligence assessment. New York Times, reprinted in Cleveland Plain Dealer, June 13, 1999.
- 39.DeClercq E, Sakuma T, Baba M, Pauwels R, Balzarini J, Rosenberg I, Holy A. Antiviral activity of phosphonylmethoxyalkyl derivatives of purines and pyrimidines. Antiviral Res. 1987;8:261–272. doi: 10.1016/s0166-3542(87)80004-9. [DOI] [PubMed] [Google Scholar]
- 40.Fenner F. Poxviruses. In: Richman DD, Whitley RJ, Hayden FG, editors. Clinical Virology. New York: Churchill Livingstone; 1997. pp. 357–367. [Google Scholar]
- 41.Henderson DA, Inglesby TV, Bartlett JG, Ascher MS, Eitzen E, Jahrling PB, Hauer J, Layton M, McDade J, Osterholm MT, O'Toole T, Parker G, Perl T, Russell PK, Tonat K. Smallpox as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA. 1999;281:2127–2137. doi: 10.1001/jama.281.22.2127. [DOI] [PubMed] [Google Scholar]
- 42.Huggins JW, Robertson M, Kefauver D, Laughlin C. Potential antiviral therapeutics for smallpox and other pox virus infections. Presented at the 11th International Meeting on Poxviruses and Iridovirus, May 4–9,1996, Toledo, Spain.
- 43.Huggins JW, Smee D, Martinez MJ. Cidofovir, HPMPC treatment of monkeypox. Presented at the 11th Conference on Antiviral Research, April 5–10, 1998, San Diego, Calif.
- 44.McClain DJ. Smallpox. In: Zajtchuk R, Bellamy RF, editors. Textbook of Military Medicine: Medical Aspects of Chemical and Biological Warfare. Washington, DC: 1997. –Office of the Surgeon General, US Department of the Army.pp. 539–559. [Google Scholar]
- 45.McEvoy GK. AHFS Drug Information 2001. Bethesda, Md: American Society of Health-Systems Pharmacists; 2001. Smallpox vaccine. [Google Scholar]
- 46.Naesens L, Snoeck R, Andrei G, et al. HPMPC (cidofovir), PMEA (adefovir) and related acyclic-nucleoside phosphonate analogues: a review of their pharmacology and clinical potential in the treatment of viral infections. Antiviral Chem Chemother. 1997;8:1–23. [Google Scholar]
Quinolone utilization
- 47.Burkhardt JE, Walterspiel JN, Schaad UB. Quinolone arthropathy in animals versus children. Clin Infect Dis. 1997;25:1196–1204. doi: 10.1086/516119. [DOI] [PubMed] [Google Scholar]
- 48.Doganay M, Aydin N. Antimicrobial susceptibility of Bacillus anthracis. Scand J Infect Dis. 1991;23:333–335. doi: 10.3109/00365549109024319. [DOI] [PubMed] [Google Scholar]
- 49.Hampel B, Hullmann R, Schmidt H. Ciprofloxacin in pediatrics: worldwide clinical experience based on compassionate use—safety report. Pediatr Infect Dis J. 1997;16:127–129. doi: 10.1097/00006454-199701000-00036. [DOI] [PubMed] [Google Scholar]
- 50.Hooper DC. Mechanisms of action and resistance of older and newer fluoroquinolones. Clin Infect Dis. 2000;31(Suppl 2):S24–S28. doi: 10.1086/314056. [DOI] [PubMed] [Google Scholar]
- 51.Kohlhepp SJ, Grunkemeir G, Leggett JE, Dworkin RJ, Slaughter SE, Gilbert DN. Phenotypic resistance of penicillin-susceptible and penicillin-resistant Streptococcus pneumoniae after single and multiple in vitro exposures to ciprofloxacin, gatifloxacin, levofloxacin, moxifloxacin and trovafloxacin [abstract 97]. 38th Annual Meeting, Infectious Diseases Society of America, New Orleans, La, 2000.
- 52.Millchap JJ, Pestova EV, Noskin GA, Peterson LR. Evidence for the presence of non-PMRA multidrug transport proteins in Streptococcus pneumoniae [abstract 99]. 38th Annual Meeting, Infectious Diseases Society of America, New Orleans, La, 2000.
- 53.Plouffe JF. Importance of atypical pathogens of community-acquired pneumonia. Clin Infect Dis. 2000;31(Suppl 2):S35–S39. doi: 10.1086/314058. [DOI] [PubMed] [Google Scholar]
- 54.Sabella C, Goldfarb J. Fluoroquinolone therapy in pediatrics: where we stand. Clin Pediatr (Phila) 1997;336:445–448. doi: 10.1177/000992289703600802. [DOI] [PubMed] [Google Scholar]
- 55.Warren R. Rheumatologic aspects of pediatric cystic fibrosis patients treated with fluoroquinolones. Pediatr Infect Dis J. 1997;16:118–122. doi: 10.1097/00006454-199701000-00034. [DOI] [PubMed] [Google Scholar]
Psychological response
- 56.Burgess JL, Kirk M, Borron SW, Cisek J. Emergency department hazardous materials protocol for contaminated patients. Ann Emerg Med. 1999;34:205–212. doi: 10.1016/s0196-0644(99)70230-1. [DOI] [PubMed] [Google Scholar]
- 57.DiGiovanni C., Jr Domestic terrorism with chemical or biological agents: psychiatric aspects. Am J Psychiatry. 1999;156:1500–1505. doi: 10.1176/ajp.156.10.1500. [DOI] [PubMed] [Google Scholar]
- 58.Fullerton CS, Ursano RJ. Behavioral and psychological responses to chemical and biological warfare. Mil Med. 1990;155:54–59. [PubMed] [Google Scholar]
- 59.Holloway HC, Norwood AE, Fullerton CS, Engel CC, Jr, Ursano RJ. The threat of biological weapons. Prophylaxis and mitigation of psychological and social consequences. JAMA. 1997;278:425–427. [PubMed] [Google Scholar]
- 60.Kline NA, Dow BM, Brown SA, Matloff JL. Sertraline efficacy in depressed combat veterans with posttraumatic stress disorder. Am J Psychiatry. 1994;151:621. doi: 10.1176/ajp.151.4.621a. [DOI] [PubMed] [Google Scholar]
- 61.Klonoff DC. Chronic fatigue syndrome. Clin Infect Dis. 1992;15:812–823. doi: 10.1093/clind/15.5.812. [DOI] [PubMed] [Google Scholar]
- 62.Malone JD, Paige-Dobson B, Ohl C, DiGiovanni C, Cunnion S, Roy MJ. Possibilities for unexplained chronic illnesses among reserve units deployed in Operation Desert Shield/Desert Storm. South Med J. 1996;89:1147–1155. doi: 10.1097/00007611-199612000-00003. [DOI] [PubMed] [Google Scholar]
- 63.Michels RM, Marzuk PM. Progress in psychiatry. Part 2. Mood disorder. N Engl J Med. 1993;329:628–638. doi: 10.1056/NEJM199308263290908. [DOI] [PubMed] [Google Scholar]
- 64.North CS, Nixon SJ, Shariat S, Mallonee S, McMillin JC, Spitznagel EL, Smith EM. Psychiatric disorders among survivors of the Oklahoma City bombing. JAMA. 1999;282:755–762. doi: 10.1001/jama.282.8.755. [DOI] [PubMed] [Google Scholar]
- 65.van der Kolk BA, Dreyfuss D, Michaels M, Shera D, Berkowitz R, Fisler R, Saxe G. Fluoxetine in posttraumatic stress disorder. J Clin Psychiatry. 1994;55:517–522. [PubMed] [Google Scholar]
Emergency response
- 66.Cieslak TJ, Christopher GW, Kortepeter MG, Rowe JR, Pavlin JA, Culpepper RC, Eitzen EM., Jr Immunization against potential biological warfare agents. Clin Infect Dis. 2000;30:843–850. doi: 10.1086/313812. [DOI] [PubMed] [Google Scholar]
- 67.Crosby AW. America's Forgotten Pandemic. Cambridge, UK: Cambridge University Press; 1998. [Google Scholar]
- 68.Diamond J. Guns, Germs, and Steel, the Fates of Human Societies. New York: WW Norton; 1999. [Google Scholar]
- 69.Henderson DA. Weapons for the future. Lancet. 1999;354(Suppl S4):64. doi: 10.1016/s0140-6736(99)90407-5. [DOI] [PubMed] [Google Scholar]
- 70.Higgins JA, Ibrahim MS, Knauert FK, Ludwig GV, Kijek TM, Ezzell JW, Courtney BC, Henchal EA. Sensitive and rapid identification of biological threat agents. Ann N Y Acad Sci. 1999;894:130–148. doi: 10.1111/j.1749-6632.1999.tb08056.x. [DOI] [PubMed] [Google Scholar]
- 71.Hood E. Chemical and biological weapons: new questions, new answers. Environ Health Perspect. 1999;107:931–931. doi: 10.1289/ehp.99107931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Inglesby T, Grossman R, O'Toole T. A plague on your city: observations from TOPOFF. Clin Infect Dis. 2001;32:436–445. doi: 10.1086/318513. [DOI] [PubMed] [Google Scholar]
- 73.Macintyre AG, Christopher GW, Eitzen E, Jr, Gum R, Weir S, DeAtley C, Tonat K, Barbera JA. Weapons of mass destruction events with contaminated casualties: effective planning for health care facilities. JAMA. 2000;283:242–249. doi: 10.1001/jama.283.2.242. [DOI] [PubMed] [Google Scholar]
- 74.Olson KB. Aum Shinrikyo: once and future threat? Emerg Infect Dis. 1999;5:513–516. doi: 10.3201/eid0504.990409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Steffan R, Melling J, Woodall JP, Rollin RH, Lang RH, Luthy R, Waldvogel A. Preparation for emergency relief after biological warfare. J Infect. 1997;34:127–132. doi: 10.1016/s0163-4453(97)92433-6. [DOI] [PubMed] [Google Scholar]
- 76.Waeckerle JF. Domestic preparedness for events involving weapons of mass destruction. JAMA. 2000;283:252–254. doi: 10.1001/jama.283.2.252. [DOI] [PubMed] [Google Scholar]