Abstract
Resistance to and control of Cryptosporidium parvum infection in mice in the absence of adaptive immunity appears to be gamma interferon (IFN-γ) dependent. Using an IFN-γ-neutralizing antibody in a murine model, we demonstrated increased susceptibility to infection within 24 h. We correlated this early resistance and control with increased mucosal expression of IFN-γ and demonstrate that CD8+ T-cell receptor αβ intestinal intraepithelial lymphocytes express and secrete this cytokine shortly after infection. The rapid kinetics of IFN-γ expression and secretion by naive CD8+ T cells in response to a protozoan pathogen have not previously been demonstrated.
Cryptosporidium parvum is an enteric, intracellular, zoonotic parasite that causes significant morbidity, particularly among people suffering from AIDS and children in developing countries. With the exception of highly active antiretroviral therapy, there remains no effective therapy for AIDS-related cryptosporidiosis. Our understanding of the immune response to C. parvum is not complete. From studies of human and animal cryptosporidiosis, it is apparent that adaptive immunity, in the form of CD4+ T cells and the cytokine gamma interferon (IFN-γ), is important for resistance to and clearance of the infection (24). However, there is convincing evidence from studies of C. parvum infection in scid mice of an IFN-γ-dependent innate resistance to infection with this parasite (7). The mechanism of this innate immune response as well as the source of IFN-γ in this setting, however, remains undefined. Since C. parvum infects primarily the gastrointestinal epithelium, we hypothesized that immune cells within the mucosa contribute to this innate IFN-γ-dependent immunity.
The purpose of this study was to demonstrate the presence of and characterize the IFN-γ-dependent innate immune response to C. parvum infection in mice. We hypothesized that intraepithelial lymphocytes (iIEL) are mediators of this IFN-γ-dependent innate immunity. Using a neutralizing anti-IFN-γ antibody, we show increased susceptibility to C. parvum infection in C57BL/6 wild-type mice within 24 h after infection. We also demonstrate increased IFN-γ expression within the terminal ileum in mice 24 h after infection with C. parvum and identify the specific cellular compartments that contribute to this early cytokine expression.
Early resistance to C. parvum infection is IFN-γ dependent.
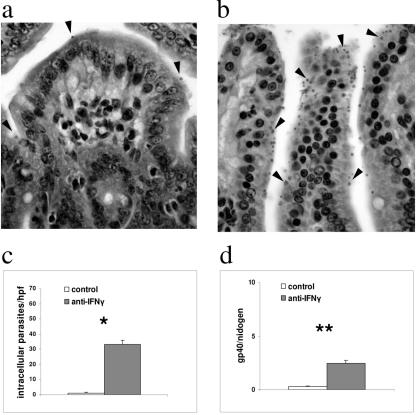
There is indirect evidence that mice have an IFN-γ-dependent resistance to C. parvum in the absence of adaptive immunity (7). However, in these studies, the degree of C. parvum infection was not assessed until 3 weeks after infection. Others have shown that IFN-γ-deficient mice begin to shed oocysts 3 days after infection (16). Since we hypothesized that an innate immune response to C. parvum infection would be necessarily rapid, we sought to determine the importance of IFN-γ within the first 24 h after infection. We treated 3-week-old wild-type C57BL/6 mice (Charles River Laboratories, Wilmington, MA) with 1 mg of IFN-γ-neutralizing antibody (XMG 1.2, a kind gift of Saul Tzipori, originally provided by Robert Coffman, DNAX Institute) intraperitoneally 24 h prior to infection. These mice and the untreated controls received 107 C. parvum oocysts of the IOWA isolate (Pleasant Valley and Bunch Grass Farms, Idaho) in phosphate-buffered saline by oral gavage. The mice were then euthanized after 24 h, and infection was quantified in sections of the terminal ileum (Fig. 1). For histological analysis, the number of parasites per sample was expressed as the mean number of intracellular forms of the parasite within a 10 by 10 grid counted in five separate high-powered fields (10). Using this method of quantification, infection was detected in untreated mice at 24 h but was significantly more severe in the mice that received the XMG 1.2 antibody (P = 0.0001 at 24 h) (Fig. 1b and c). A t test with a two-tailed test of significance was used for this and all subsequent statistical analyses, except where noted. C. parvum infection was also quantified by real-time PCR using DNA (extracted using a GNOME DNA kit; Qbiogene, Irvine, CA) from the terminal ilea of C. parvum-infected mice. We designed C. parvum-specific primers for quantitative PCR to amplify a conserved region of the Cpgp40/15 gene (5). The sequences of these primers were as follows: forward (5′-TCA TTT GTA ATG TGG TTC GGA GAA-3′) and reverse (5′-AGG GTA AAG GCA AAC AAA TCG A-3′). Primers for the murine housekeeping gene nidogen were used as an endogenous control as previously described (19). Quantitative PCR was performed on an ABI Prism 7700 (Applied Biosystems, Foster City, CA) machine using a Quantitect SYBR green PCR kit (QIAGEN, Valencia, CA) at 95°C for 15 min, followed by 40 cycles of 94°C for 30 seconds, 60°C for 1 min, and 72°C for 30 seconds. Using this technique, the mean number of parasites was also significantly greater in mice that had received the neutralizing antibody at 24 h (P = 0. 001) (Fig. 1d). Therefore, the technique of quantitative PCR to measure infection in vivo at 24 h correlated with standard histological analysis. In summary, these data are consistent with an early IFN-γ-dependent resistance to and control of C. parvum infection, which is present before the onset of the adaptive immune response.
FIG. 1.
IFN-γ neutralization increases the susceptibility of wild-type mice within 24 h after C. parvum infection. (a and b) Representative hematoxylin- and eosin-stained sections of terminal ileum taken 24 h after mice were infected with C. parvum. In panel b, the mouse received an intraperitoneal injection of anti-IFN-γ antibodies (XMG 1.2) 24 h prior to infection, and panel a shows the results with an untreated control. Black arrowheads indicate parasites. (c) Histogram depicting the mean number (+standard deviation) of parasites counted in histological sections from five mice per group (*, P = 0.0001). (d) Histogram showing samples from the same experiment quantified by real-time PCR. Each bar represents the mean number of copies of Cpgp40/15 normalized to the number of copies of nidogen (**, P = 0.001). Data are representative of two independent experiments.
IFN-γ expression is increased in the small intestine within 24 h after C. parvum infection.
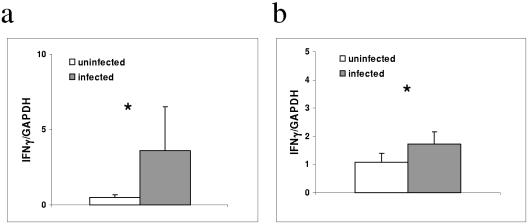
It had previously been shown that IFN-γ expression was increased in the terminal ileum as early as 3 days after C. parvum infection (14). Earlier expression of IFN-γ, well prior to the onset of adaptive immunity, has not been reported. We sought to determine whether the expression of IFN-γ was increased in the small intestines of mice within 24 h after C. parvum infection. In this experiment, RNA was purified from the terminal ilea of C. parvum-infected and uninfected mice 24 h after C. parvum infection using a kit (RNeasy; QIAGEN, Valencia, CA). After DNase treatment (Ambion, Austin, TX), cDNA was prepared from RNA using oligo(dT) (Invitrogen, Carlsbad, CA), followed by reverse transcription with Stratascript (Stratagene, La Jolla, CA). Quantitative RT-PCR was performed using fluorescently labeled probes for IFN-γ and GAPDH (glyceraldehyde-3-phosphate dehydrogenase) (QIAGEN, Valencia, CA). The expression of IFN-γ was significantly greater in the terminal ilea of C. parvum infected mice at 24 h than in uninfected controls (P = 0.0004) (Fig. 2a). This rapid mucosal expression of IFN-γ in response to C. parvum infection is consistent with the IFN-γ-dependent resistance and control that we had shown using the neutralizing antibody. Furthermore, these data suggest that IFN-γ is rapidly expressed within the mucosa in response to C. parvum infection.
FIG. 2.
Increased IFN-γ expression in the small intestines of mice 24 h after C. parvum infection. (a) Quantitative PCR demonstrating increased IFN-γ in the terminal ilea of C. parvum-infected mice compared with levels in sham-infected mice. Each bar represents mean copies of IFN-γ normalized to mean copies of GAPDH by quantitative PCR for five mice per group (*, P < 0.05). Data are representative of two independent experiments. (b) Increased IFN-γ expression in iIEL from infected compared with its expression in uninfected mice. Each bar represents IFN-γ normalized to GAPDH by quantitative PCR from pooled samples of two mice (*, P = 0.01). Data are the means from two independent experiments.
IFN-γ expression is increased among small intestine intraepithelial lymphocytes 24 h after C. parvum infection.
We hypothesized that iIEL were a likely source of IFN-γ within the intestinal mucosa. Therefore, we isolated small intestinal iIEL using a standard protocol (16a) from C. parvum-infected mice and sham-infected controls (phosphate-buffered saline alone) at 24 h and then quantified the expression of IFN-γ. We used a MoFlo cell sorter (Dako Cytomation, Fort Collins, CO) to enrich for lymphoid cells by the removal of larger and more-granulated epithelial cells according to forward- and side-scatter characteristics (19). After reverse transcription of RNA extracted from these purified cells, we used quantitative PCR to measure IFN-γ expression. We found significantly increased IFN-γ expression in the iIEL 24 h after infection compared with the expression in sham-infected controls (P = 0.01) (Fig. 2b).
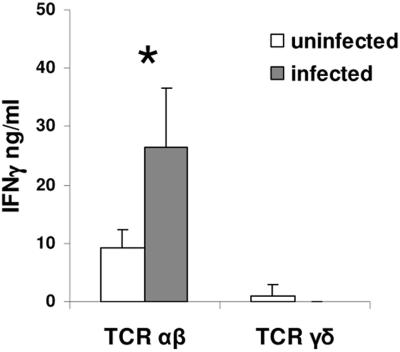
Since the majority of iIEL are CD8+ in mice (13) and there is in vitro evidence that CD8+ iIEL can rapidly be activated to secrete IFN-γ (25), we reasoned that these might be potential sources of IFN-γ. We measured IFN-γ secretion ex vivo by enzyme-linked immunosorbent assay (ELISA) in supernatants of purified iIEL from infected and uninfected mice. In these experiments, iIEL were isolated from four mice 24 h after C. parvum infection and sorted using a MoFLo cell sorter into CD8α+ T-cell receptor αβ+ (TCRαβ+) and CD8α+ TCRγδ+ cells. Equal numbers of both populations of cells from infected and uninfected mice were cultured in the presence of anti-CD3 and anti-CD28 (Pharmingen) for 48 h, after which time the supernatants were harvested and IFN-γ was measured by ELISA (Pharmingen). We found significantly greater IFN-γ secretion in CD8α+ TCRαβ+ iIEL from infected mice than in iIEL from uninfected mice (P = 0.028). Interestingly, we found comparably little IFN-γ produced by CD8α+ TCRγδ+ iIEL from either infected or uninfected mice (Fig. 3). These data suggest that iIEL, particularly CD8α TCRαβ+ cells, are a source of the rapidly induced expression of IFN-γ seen in the terminal ilea of C. parvum-infected mice.
FIG. 3.
ELISA measuring IFN-γ secretion from ex vivo-sorted TCRαβ+ and TCRγδ+ CD8α+ iIEL from C. parvum-infected and sham-infected mice (*, P = 0.028). Data are representative of three independent experiments.
While rapid IFN-γ secretion from CD8+ T cells has been well described in the context of memory responses to intracellular pathogens, there is increasing evidence that naive CD8+ effector T cells can rapidly be activated in a pathogen-specific and non-pathogen-specific manner (27). Rapid effector CD8+ T-cell responses in mice have been demonstrated in the primary immune response to Listeria monocytogenes by antigen-dependent (15) and antigen-independent (3) mechanisms. There are also reports of rapid IFN-γ secretion by naïve CD8 T cells in a murine model of meliodosis (3, 17). In the case of protozoan infections, effector CD8+ T-cell responses have been demonstrated primarily in immunized hosts (4, 21), although in vitro activation of naive CD8+ T cells by antigens derived from Toxoplasma gondii has been reported (9). It is also possible that the rapid activation of CD8+ T cells that we have described is not pathogen specific. This is consistent with reports of clearance of C. parvum from chronically infected scid mice using adoptively transferred splenocytes from DO11.10 RAG−/− mice (18).
There is substantial prior evidence that mucosal lymphocytes are necessary for an effective adaptive immune response to C. parvum. Increased numbers of small intestinal iIEL, particularly CD8+ cells, and lamina propria lymphocytes have been reported as early as 3 days after C. parvum infection in several different mammalian species (1, 2, 6, 8, 11, 12, 20, 28, 29). More definitively, other studies have used adoptive transfer to show that iIEL promote the resolution of infection in chronically infected scid mice (22). In contrast to what is known for animals, our understanding of the mucosal immune response in human cryptosporidiosis is more limited. Increases in colonic lamina propria CD8+ T cells were observed in AIDS-related cryptosporidiosis (23), and increased colonic mucosal CD4+ T cells were associated with the resolution of AIDS-related cryptosporidiosis (26). In a cohort of humans experimentally infected with Cryptosporidium, mucosal IFN-γ expression correlated with serologic evidence of prior infection and was observed within the lamina propria of the jejunum. However, IFN-γ could not be detected in biopsies of nonimmune humans (26). The lack of “innate” mucosal IFN-γ expression noted in humans may be one explanation for the differences between species in susceptibility to Cryptosporidium. Despite these differences, further exploration of innate immune responses in animal models of cryptosporidiosis can be used as a tool to discover immunogenic components of the parasite in the hopes of developing strategies aimed at improving resistance to infection in humans and animals.
Acknowledgments
We thank Carla Cugini, Barbara Woodman, and Leon Lai for assistance.
This work was supported by NIAID grant K08 AI5158 (B.A.L.); the Center for Gastroenterology Research on Absorptive and Secretory Processes (GRASP); the NIDDK (grant P30 DK-34928-19); the Crohn's and Colitis Foundation of America (M.Y.); NIDDK grants DK44319, DK51362, and DK53056 (R.S.B.); and NIAID grant AI52786 (H.W.).
Editor: W. A. Petri, Jr.
REFERENCES
- 1.Abrahamsen, M. S., C. A. Lancto, B. Walcheck, W. Layton, and M. A. Jutila. 1997. Localization of alpha/beta and gamma/delta T lymphocytes in Cryptosporidium parvum-infected tissues in naive and immune calves. Infect. Immun. 65:2428-2433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Adjei, A. A., A. K. Shrestha, M. Castro, and F. J. Enriquez. 2000. Adoptive transfer of immunity with intraepithelial lymphocytes in Cryptosporidium parvum-infected severe combined immunodeficient mice. Am. J. Med. Sci. 320:304-309. [DOI] [PubMed] [Google Scholar]
- 3.Berg, R. E., C. J. Cordes, and J. Forman. 2002. Contribution of CD8+ T cells to innate immunity: IFN-gamma secretion induced by IL-12 and IL-18. Eur. J. Immunol. 32:2807-2816. [DOI] [PubMed] [Google Scholar]
- 4.Buzoni-Gatel, D., H. Debbabi, M. Moretto, I. H. Dimier-Poisson, A. C. Lepage, D. T. Bout, and L. H. Kasper. 1999. Intraepithelial lymphocytes traffic to the intestine and enhance resistance to Toxoplasma gondii oral infection. J. Immunol. 162:5846-5852. [PubMed] [Google Scholar]
- 5.Cevallos, A. M., X. Zhang, M. K. Waldor, S. Jaison, X. Zhou, S. Tzipori, M. R. Neutra, and H. D. Ward. 2000. Molecular cloning and expression of a gene encoding Cryptosporidium parvum glycoproteins gp40 and gp15. Infect. Immun. 68:4108-4116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chai, J. Y., S. M. Guk, H. K. Han, and C. K. Yun. 1999. Role of intraepithelial lymphocytes in mucosal immune responses of mice experimentally infected with Cryptosporidium parvum. J. Parasitol. 85:234-239. [PubMed] [Google Scholar]
- 7.Chen, W., J. A. Harp, A. G. Harmsen, and E. A. Havell. 1993. Gamma interferon functions in resistance to Cryptosporidium parvum infection in severe combined immunodeficient mice. Infect. Immun. 61:3548-3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Culshaw, R. J., G. J. Bancroft, and V. McDonald. 1997. Gut intraepithelial lymphocytes induce immunity against Cryptosporidium infection through a mechanism involving gamma interferon production. Infect. Immun. 65:3074-3079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Denkers, E. Y., P. Caspar, and A. Sher. 1994. Toxoplasma gondii possesses a superantigen activity that selectively expands murine T cell receptor V beta 5-bearing CD8+ lymphocytes. J. Exp. Med. 180:985-994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Enriquez, F. J., and C. R. Sterling. 1991. Cryptosporidium infections in inbred strains of mice. J. Protozool. 38:100S-102S. [PubMed] [Google Scholar]
- 11.Fayer, R., L. Gasbarre, P. Pasquali, A. Canals, S. Almeria, and D. Zarlenga. 1998. Cryptosporidium parvum infection in bovine neonates: dynamic clinical, parasitic and immunologic patterns. Int. J. Parasitol. 28:49-56. [DOI] [PubMed] [Google Scholar]
- 12.Guk, S. M., T. S. Yong, and J. Y. Chai. 2003. Role of murine intestinal intraepithelial lymphocytes and lamina propria lymphocytes against primary and challenge infections with Cryptosporidium parvum. J. Parasitol. 89:270-275. [DOI] [PubMed] [Google Scholar]
- 13.Hayday, A., E. Theodoridis, E. Ramsburg, and J. Shires. 2001. Intraepithelial lymphocytes: exploring the Third Way in immunology. Nat. Immunol. 2:997-1003. [DOI] [PubMed] [Google Scholar]
- 14.Kapel, N., Y. Benhamou, M. Buraud, D. Magne, P. Opolon, and J. G. Gobert. 1996. Kinetics of mucosal ileal gamma-interferon response during cryptosporidiosis in immunocompetent neonatal mice. Parasitol. Res. 82:664-667. [DOI] [PubMed] [Google Scholar]
- 15.Kerksiek, K. M., D. H. Busch, I. M. Pilip, S. E. Allen, and E. G. Pamer. 1999. H2-M3-restricted T cells in bacterial infection: rapid primary but diminished memory responses. J. Exp. Med. 190:195-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lacroix, S., R. Mancassola, M. Naciri, and F. Laurent. 2001. Cryptosporidium parvum-specific mucosal immune response in C57BL/6 neonatal and gamma interferon-deficient mice: role of tumor necrosis factor alpha in protection. Infect. Immun. 69:1635-1642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16a.Lefrancois, L., and N. Lycke. 1991. Isolation of mouse small intestinal intraepithelial lymphocytes, peyer’s patch and lamina propria cells. In J. Coligan, A. Kruisbeek, D. Margulies, E. Shevach, and W. Strober (ed.), Current protocols in immunology, vol. 3. John Wiley & Sons, Inc., Newark, N.J. [DOI] [PubMed]
- 17.Lertmemongkolchai, G., G. Cai, C. A. Hunter, and G. J. Bancroft. 2001. Bystander activation of CD8+ T cells contributes to the rapid production of IFN-gamma in response to bacterial pathogens. J. Immunol. 166:1097-1105. [DOI] [PubMed] [Google Scholar]
- 18.Lukin, K., M. Cosyns, T. Mitchell, M. Saffry, and A. Hayward. 2000. Eradication of Cryptosporidium parvum infection by mice with ovalbumin-specific T cells. Infect. Immun. 68:2663-2670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Reference deleted.
- 20.McDonald, V., H. A. Robinson, J. P. Kelly, and G. J. Bancroft. 1996. Immunity to Cryptosporidium muris infection in mice is expressed through gut CD4+ intraepithelial lymphocytes. Infect. Immun. 64:2556-2562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moretto, M., L. M. Weiss, and I. A. Khan. 2004. Induction of a rapid and strong antigen-specific intraepithelial lymphocyte response during oral encephalitozoon cuniculi infection. J. Immunol. 172:4402-4409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reijasse, D., N. Patey-Mariaud de Serre, D. Canioni, M. Huerre, E. Haddad, M. Leborgne, S. Blanche, and N. Brousse. 2001. Cytotoxic T cells in AIDS colonic cryptosporidiosis. J. Clin. Pathol. 54:298-303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schmidt, W., U. Wahnschaffe, M. Schafer, T. Zippel, M. Arvand, A. Meyerhans, E. O. Riecken, and R. Ullrich. 2001. Rapid increase of mucosal CD4 T cells followed by clearance of intestinal cryptosporidiosis in an AIDS patient receiving highly active antiretroviral therapy. Gastroenterology 120:984-987. [DOI] [PubMed] [Google Scholar]
- 24.Theodos, C. M. 1998. Innate and cell-mediated immune responses to Cryptosporidium parvum. Adv. Parasitol. 40:87-119. [DOI] [PubMed] [Google Scholar]
- 25.Wang, H. C., Q. Zhou, J. Dragoo, and J. R. Klein. 2002. Most murine CD8+ intestinal intraepithelial lymphocytes are partially but not fully activated T cells. J. Immunol. 169:4717-4722. [DOI] [PubMed] [Google Scholar]
- 26.White, A. C., P. Robinson, P. C. Okhuysen, D. E. Lewis, I. Shahab, S. Lahoti, H. L. DuPont, and C. L. Chappell. 2000. Interferon-gamma expression in jejunal biopsies in experimental human cryptosporidiosis correlates with prior sensitization and control of oocyst excretion. J. Infect. Dis. 181:701-709. [DOI] [PubMed] [Google Scholar]
- 27.Wong, P., and E. G. Pamer. 2003. CD8 T cell responses to infectious pathogens. Annu. Rev. Immunol. 21:29-70. [DOI] [PubMed] [Google Scholar]
- 28.Wyatt, C. R., E. J. Brackett, L. E. Perryman, A. C. Rice-Ficht, W. C. Brown, and K. I. O'Rourke. 1997. Activation of intestinal intraepithelial T lymphocytes in calves infected with Cryptosporidium parvum. Infect. Immun. 65:185-190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wyatt, C. R., E. J. Brackett, and J. Savidge. 2001. Evidence for the emergence of a type-1-like immune response in intestinal mucosa of calves recovering from cryptosporidiosis. J. Parasitol. 87:90-95. [DOI] [PubMed] [Google Scholar]