Abstract
Genetic and cytologic data from Saccharomyces cerevisiae and mammals implicate the Mre11 complex, consisting of Mre11, Rad50, and Nbs1, as a sensor of DNA damage, and indicate that the complex influences the activity of ataxia-telangiectasia mutated (ATM) in the DNA damage response. Rad50S/S mice exhibit precipitous apoptotic attrition of hematopoietic cells. We generated ATM- and Chk2-deficient Rad50S/S mice and found that Rad50S/S cellular attrition was strongly ATM and Chk2 dependent. The hypomorphic Mre11ATLD1 and Nbs1ΔB alleles conferred similar rescue of Rad50S/S-dependent hematopoietic failure. These data indicate that the Mre11 complex activates an ATM–Chk2-dependent apoptotic pathway. We find that apoptosis and cell cycle checkpoint activation are parallel outcomes of the Mre11 complex–ATM pathway. Conversely, the Rad50S mutation mitigated several phenotypic features of ATM deficiency. We propose that the Rad50S allele is hypermorphic for DNA damage signaling, and that the resulting constitutive low-level activation of the DNA damage response accounts for the partial suppression of ATM deficiency in Rad50S/S Atm-/- mice.
Keywords: Checkpoints, DNA damage signaling, apoptosis
The cellular response to DNA damage includes activation of DNA damage-dependent cell cycle checkpoints, DNA repair, and in some contexts, apoptosis. The Mre11 complex, consisting of the highly conserved proteins Mre11, Rad50, and Nbs1, has been implicated in DNA damage recognition and the subsequent activation of cell cycle checkpoints, as well as in DNA repair. Human and mouse hypomorphic Nbs1 and Mre11 mutations (Carney et al. 1998; Stewart et al. 1999; Kang et al. 2002; Williams et al. 2002; Theunissen et al. 2003; Difilippantonio et al. 2005) in Nijmegen breakage syndrome (NBS) and the ataxia-telangiectasia-like disorder (A-TLD), respectively, are associated with ionizing radiation (IR) sensitivity, checkpoint deficiency, and chromosome instability. Despite these defects in the DNA damage response pathway, IR-dependent apoptotic induction does not appear to be compromised in Nbs1 or Mre11 mutant mice (Theunissen et al. 2003).
Several lines of evidence demonstrate that the Mre11 complex functions with the ataxia-telangiectasia mutated (ATM) kinase to affect aspects of the response to DNA double-strand breaks (DSBs). This conclusion is supported by molecular observations in addition to the phenotypic similarities between NBS, A-TLD, and ataxia-telangiectasia (A-T) (Petrini 2000). Nbs1 is phosphorylated by ATM in response to DNA damage, and this event is required for activation of the intra-S-phase checkpoint (Gatei et al. 2000; Lim et al. 2000; Wu et al. 2000; Zhao et al. 2000). The complex also functions upstream of ATM and possibly the ataxia-telangiectasia and Rad3-related (ATR) transducing kinase by recognizing DNA damage and potentiating their activation and activity. The complex associates with DNA damage independently of ATM or ATR, or any modifications that depend on those kinases (Petrini and Stracker 2003), and appears to recruit ATM to break sites via a conserved domain at the Nbs1 C terminus (Kitagawa et al. 2004; Falck et al. 2005; You et al. 2005). This behavior is markedly diminished in cells established from A-TLD patients (Stewart et al. 1999), and IR-dependent ATM activation is impaired as a result (Uziel et al. 2003). A similar impairment is seen in cells from NBS patients (Mochan et al. 2003; Horejsi et al. 2004). The degradation of the Mre11 complex in adenovirus-infected cells attenuates ATM activation (Stracker et al. 2002; Carson et al. 2003). Finally, Mre11 complex-dependent ATM activation has been demonstrated in vitro with purified components and in Xenopus extracts (Costanzo et al. 2004; Lee and Paull 2005). DNA-PKcs and SMG1(ATX) also appear to be activated by DNA damage, although the significance of the Mre11 complex in these events has not been addressed (Abraham 2004; Brumbaugh et al. 2004).
In contrast to Nbs1 and Mre11 mutations, the murine Rad50S allele is not associated with checkpoint deficiency, nor does it impair DSB recognition by the Mre11 complex (Bender et al. 2002). Cells from Rad50S/S mice exhibit indices of chronic genotoxic stress, the in vivo consequence of which is the rapid apoptotic attrition of hematopoietic and spermatogenic cells. Accordingly, p53 deficiency rescues the Rad50S/S phenotype. Bone-marrow depletion in Rad50S/S mice is primarily attributable to stem-cell failure, since Rad50S/S stem cells are unable to reconstitute the hematopoietic compartment in lethally irradiated wild-type mice. Because genotoxic stress in Rad50S/S mice is not associated with an overt DSB repair defect, and the impact of the Rad50S allele on chromosome stability is extremely mild (Bender et al. 2002; de Jager and Kanaar 2002), we have proposed that the murine Rad50S allele is an “up mutant” for activation of the DNA damage response (Stracker et al. 2004). This proposal was initially based on the hypermorphic DNA damage signaling behavior of the Saccharomyces cerevisiae rad50S mutant (Usui et al. 2001) and is supported by data presented herein.
In this study, we exploit the Rad50S allele to examine the Mre11 complex's role in ATM-dependent apoptosis. We show that Rad50S/S apoptotic attrition is mitigated in genetic contexts that impair ATM activation. Crosses between Rad50S/S and mutants in the ATM targets Chk2 and SMC1 revealed a branch point in the ATM-dependent apoptotic pathway. Hematopoietic attrition was rescued in Rad50S/S Chk2-/- double mutants, whereas crosses with mice expressing a Smc1 mutant in which the two ATM phosphorylation sites Ser957 and Ser966 have been mutated had no effect (Kitagawa et al. 2004).
In ATM-deficient mice, the hypermorphic behavior of Rad50S resulted in the suppression of Atm-/--associated lymphomagenesis, senescence, and IR sensitivity. This suppression appeared to depend on the activation of other PI3K-like kinase family members as indicated by an increase in phoshorylated SQ/TQ motifs and γ-H2AX in Rad50S/S Atm-/-. Collectively, these data demonstrate that the Mre11 complex promotes apoptotic induction in an ATM–Chk2–p53-dependent manner, and that activation of an alternative DNA damage-transducing kinase can mitigate the pathologic outcomes associated with ATM deficiency.
Results
Genetic determinants of Rad50S/S pathology
In order to determine the mechanism of Rad50S-dependent cellular attrition, we defined the genetic dependencies of this Rad50S/S phenotypic outcome. First, to determine whether Rad50S is a hypermorphic allele, we crossed Rad50S/S with Rad50+/Δ mice, reasoning that reduced Rad50S dosage would increase phenotypic severity if it were hypomorphic, or decrease severity if it were hypermorphic (Luo et al. 1999). Although most Rad50Δ/S mice died from hematopoietic failure, the mean survival was significantly higher than that of Rad50S/S: 58% of Rad50Δ/S mice survived to 5 mo (Fig. 1A). The increased survival of Rad50Δ/S was associated with malignancy as three out of 29 mice died with lymphoma (Fig. 1A). Hence, reduced Rad50S dosage in Rad50Δ/S mice enhanced survival, indicating that Rad50S is a hypermorphic allele.
Figure 1.
Rescue of Rad50S/S survival by Rad50-null, Atm-null, Mre11ATLD1, and Nbs1ΔB alleles. (A) Kaplan-Meier survival curves of Rad50S/S and Rad50Δ/S mice. (B) Kaplan-Meier survival curves of Rad50S/S, Atm-/-, Rad50S/S Atm+/-, and Rad50S/S Atm-/- mice. (C) Kaplan-Meier survival curves of Rad50S/S, Rad50S/S Mre11+/ATLD1, Rad50S/S Mre11ATLD/ATLD1, Rad50S/S Nbs1+/ΔB, and Rad50S/S Nbs1ΔB/ΔB mice. (A–C) In each table below the survival curves, for each cohort, the number of mice that were found dead without overt signs of anemia and malignancy is equal to the number of mice minus the number of alive mice, the number of dead mice with anemia, and the number of dead mice with lymphoma. (D) Hematoxylin and eosin staining (upper pictures) or cleaved Caspase-3 detected by immunohistochemistry (lower pictures) in bone marrow sections from 4-wk-old Rad50S/S, Rad50S/S Atm-/-, Rad50S/S Mre11+/ATLD1, and Rad50S/S Mre11ATLD/ATLD1 mice. Magnification, 100×. Bar, 100 μM.
Since the Mre11 complex and ATM function in the same DNA damage response pathway (D'Amours and Jackson 2002; Petrini and Stracker 2003), we established Rad50S/S Atm-/- double-mutant mice to determine whether the hypermorphic character of the Rad50S allele was ATM dependent. Whereas Rad50S/S mice are recovered at 50% of the expected Mendelian frequency (Bender et al. 2002), Rad50S/S Atm-/- mice were recovered at normal Mendelian ratios.
Phenotypic rescue was also observed in mature animals. Peripheral blood cell numbers in Rad50S/S Atm-/- mice were comparable to wild-type levels (red blood cell percentages: wild type, 41%; Rad50S/S, 32%; Rad50S/S Atm-/-, 39%; platelet numbers: wild type, 575 × 103 cells/mL; Rad50S/S, 252 × 103 cells/mL; Rad50S/S Atm-/-, 500 × 103 cells/mL). Flow cytometry on 4-wk-old mice indicated that hematopoietic cells in both Rad50S/S Atm+/- and Rad50S/S Atm-/- mice were comparable to those in Atm-/- mice (Fig. 2; Supplementary Table 1). The rescue of Rad50S/S hematopoietic attrition was associated with significantly increased survival of Rad50S/S Atm+/- and Rad50S/S Atm-/- mice compared with Rad50S/S (Fig. 1B). Whereas most (>90%) Rad50S/S mice died during the first 5 mo of age, 85% of Rad50S/S Atm+/- mice and 42% of Rad50S/S Atm-/- mice lived longer than 5 mo. The increase in survival relative to Rad50S/S alone was significant at 5, 10, and 15 mo of age (P values < 0.001). Rad50S/S Atm-/- died of malignancy and Rad50S/S Atm+/- predominantly succumbed to anemia, albeit significantly later than most Rad50S/S mice. The data indicated that the attrition of hematopoietic cells in Rad50S/S mice was dependent on ATM.
Figure 2.
Rescue of Rad50S/S hematopoietic attrition by Atm-null, Mre11ATLD1, and Nbs1ΔB alleles, and by Chk2-/-. Flow cytometric analysis of hematopoietic tissues from 4–9-wk-old mice. The numbers of double-negative T cells in the thymus (DN T cells; A), and macrophages (B) and pro-B cells in the bone marrow (C), normalized to wild-type data, are depicted for the indicated genotypes.
Genetic interactions with Mre11 complex mutants
As with cells established from A-TLD patients, cells from mice expressing the Mre11ATLD1 allele (Mre11ATLD1/ATLD1 mice) exhibit indices of attenuated ATM activity (Theunissen et al. 2003). We established Rad50S/S Mre11ATLD1/ATLD1 double mutants to determine whether reduced ATM activation by the Mre11 complex would also rescue Rad50S/S cellular attrition. As with the Atm-null allele, Mre11ATLD1 increased Rad50S/S survival in a dose-dependent manner (Fig. 1C). Cohorts of 29 Rad50S/S Mre11ATLD1/ATLD1 and 49 Rad50S/S Mre11+/ATLD1 mice were aged and monitored for indications of Rad50S/S-dependent pathology. Twenty-four out of 29 Rad50S/S Mre11ATLD1/ATLD1 mice were alive and older than 5 mo with no overt signs of anemia at the time of this writing, and four double mutants developed lymphoma.
A similar, but less pronounced rescue was observed in Rad50S/S Mre11+/ATLD1 mice. Seventeen Rad50S/S Mre11+/ATLD1 mice were alive and older than 5 mo. Out of the 32 Rad50S/S Mre11+/ATLD1 mice that died, 19 exhibited hematopoietic attrition, 10 succumbed to lymphoma, and three died without signs of anemia and malignancy (Fig. 1C). Flow-cytometric analysis of bone marrow from 6-wk-old animals confirmed that increased survival in Rad50S/S Mre11ATLD1/ATLD1 and Rad50S/S Mre11+/ATLD1 was associated with reduced hematopoietic attrition. The numbers of double-negative (DN) T cells, pro-B cells, and macrophages in both Rad50S/S Mre11ATLD1/ATLD1 and Rad50S/S Mre11+/ATLD1 were within 2.5-fold of wild-type levels, showing that Rad50S/S hematopoietic attrition was affected by a single copy of Mre11ATLD1 (Fig. 2; Supplementary Table 1).
Nbs1ΔB/ΔB mice also exhibit reduced indices of ATM activity (Williams et al. 2002). Similar to the hematopoietic rescue of Rad50S/S by Mre11ATLD1, the numbers of DN T cells, pro-B cells, and macrophages in both Rad50S/S Nbs1ΔB/ΔB and Rad50S/S Nbs1+/ΔB were within fivefold of wild-type levels (Fig. 2; Supplementary Table 1). As with ATM and MRE11, the increase in Rad50S/S survival was dependent on NBS1 gene dosage (Fig. 1C).
Rad50S/S cellular attrition is due to apoptosis
Attrition of Rad50S/S hematopoietic cells was attributable to apoptosis. Immunohistochemical analysis of Rad50S/S bone marrow revealed markedly increased levels of cleaved Caspase-3, whereas wild type did not show appreciable staining (Fig. 1D). The staining of cleaved Caspase-3 was reduced to wild-type levels in Rad50S/S Atm-/-, Rad50S/S Mre11ATLD1/ATLD1, and Rad50S/S Mre11+/ATLD1 bone marrow (Fig. 1D). Similar results were obtained in thymus and testes sections (data not shown). This interpretation was further supported by genetic evidence. We crossed Rad50S/S with Eμ-Bcl2 mice, which carry a transgene of the anti-apoptotic BCL2 gene (Strasser et al. 1990). This transgene increases resistance to apoptosis in B and T lymphocytes (Strasser et al. 1991a,b). The depletion of hematopoietic stem cells in Rad50S/S mice appears to be partially attributable to the attrition of committed precursors (Bender et al. 2002). Therefore, we reasoned that reduction in the loss of B and T cells in Rad50S/S Eμ-Bcl2 would confer increased survival. This was indeed the case, as Rad50S/S Eμ-Bcl2 mice succumbed to anemia significantly later than Rad50S/S (Fig. 3A). These observations confirm that apoptosis underlies the precipitous Rad50S/S hematopoietic failure, consistent with the previously established p53 dependency for Rad50S/S cellular attrition (Bender et al. 2002). Collectively, the experiments in which reduced ATM activity rescues Rad50S/S hematopoiesis demonstrate that Rad50S/S, and by extension, the Mre11 complex, promotes ATM-dependent apoptosis.
Figure 3.
Rescue of Rad50S/S survival by Chk2-null allele and the Eμ-Bcl2 transgene. (A) Kaplan-Meier survival curves of Rad50S/S and Rad50S/S Eμ-Bcl2 mice. (B) Kaplan-Meier survival curves of Rad50S/S, Rad50S/S Chk2+/-, Rad50S/S Chk2-/-, and Rad50S/S Smc12SA/2SA mice. Each table below the survival curves is as in Figure 1.
Genetic interactions with downstream ATM targets
To define the ATM targets that are activated in this context, we crossed Rad50S/S with Chk2-/- and Smc1S957AS966A/S957AS966A (Smc12SA/2SA) mutant mice (Hirao et al. 2002; Kitagawa et al. 2004). Rad50S/S Chk2-/- hematopoietic cell numbers were within two-fold of wild type (Fig. 2; Supplementary Table 1). In our cohort of Rad50S/S Chk2-/- mice, 52 out of 65 were alive and older than 5 mo at the time of this writing (Fig. 3B). Although less pronounced than with ATM, MRE11, or NBS1, the rescue of Rad50S/S survival by Chk2 deficiency was partially dependent on gene dosage, with 10 of 41 Rad50S/S Chk2+/- mice alive at 5 mo-of-age (Fig. 3B).
In contrast, Rad50S/S Smc12SA/2SA mice were indistinguishable from Rad50S/S with respect to hematopoietic cell numbers and survival (Figs. 2, 3B). These data demonstrate that Rad50S/S activates Mre11 complex ATM-dependent apoptosis through the Chk2 kinase. Given that phosphorylation of SMC1 by ATM is necessary for activation of the intra-S-phase checkpoint (Kim et al. 2002; Yazdi et al. 2002; Kitagawa et al. 2004), the data further indicate that apoptotic induction and intra-S-phase checkpoint activation are parallel endpoints of the Mre11 complex–ATM pathway.
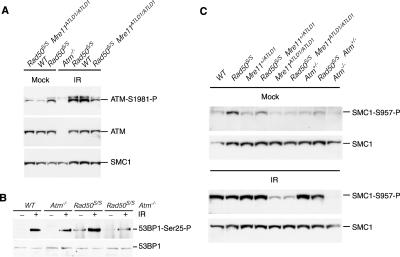
ATM-dependent signaling was examined at the molecular level to identify the targets responsible for the Rad50S/S apoptotic phenotype. Irradiation induces autophosphorylation of ATM Ser1981, and this phosphorylation event correlates with ATM activation (Bakkenist and Kastan 2003). In asynchronous primary ear fibroblast cultures, phosphorylation of ATM Ser1981 was increased in unirradiated Rad50S/S cells compared with wild type (Fig. 4A). This outcome was associated with phosphorylation in untreated cells of the ATM targets H2AX (Fig. 5D), 53BP1 (Fig. 4B), and SMC1 (Fig. 4C) on Ser139, Ser25, and Ser957, respectively. Constitutive ATM autophosphorylation (Fig. 4A) and SMC1 phosphorylation were reduced in the genetic contexts that led to enhanced Rad50S/S survival, (i.e., Rad50S/S Mre11ATLD1/ATLD1, Rad50S/S Atm+/-, and moderate reduction in Rad50S/S Mre11+/ATLD1) (Fig. 4C), consistent with the interpretation that ATM-dependent signaling accounts for cellular attrition in Rad50S/S.
Figure 4.
Constitutive ATM activity in Rad50S/S is rescued by ATM deficiency and Mre11ATLD1. (A) Analysis of ATM Ser1981 phosphorylation. Extracts from wild-type (WT), Rad50S/S, Rad50S/S Mre11ATLD1/ATLD1, and Atm-/- p4 ear fibroblasts were prepared after mock treatment and 1 h after 10 Gy of IR. For the ATM Ser1981 phosphorylation Western blot (ATM-Ser1981-P), the lower reactive species represents ATM, as indicated by the ATM Western blot (ATM), and the upper band is nonspecific. SMC1 is included as a loading control. (B) Analysis of 53BP1 phosphorylation in primary ear fibroblasts. Ser25-phosphorylated 53BP1 was immunoprecipitated from untreated or irradiated (10 min after 4 Gy) primary ear fibroblast extracts, and detected with a 53BP1 antibody. A 53BP1 Western blot of the same extracts is shown as a loading control. (C) Analysis of SMC1 Ser 957 phosphorylation in the indicated mutants. Extracts from p4 ear fibroblasts were prepared after mock treatment and 1 h after 10 Gy of IR. The extracts were sequentially immunoblotted with SMC1-Ser957-P and SMC1 (loading control) antisera. (WT) Mock-treated wild-type lane underloaded for SMC1. As untreated Mre11+/ATLD1 cells were equivalent to wild type for the levels of SMC1 phosphorylation (data not shown), compare mutants with that lane.
Figure 5.
DNA damage signaling is enhanced in Rad50S/S Atm-/- cells compared with Atm-/- cells. (A) SQ/TQ foci formation in wild-type (WT), Atm-/-, Rad50S/S, and Rad50S/S Atm-/- primary ear fibroblasts was assessed by immunofluorescence. Primary ear fibroblasts were harvested 30 min after mock treatment (white bars) or 4 Gy of IR (gray bars). Two-hundred-fifty cells were counted for presence or absence of SQ/TQ foci. A cell is considered positive when it shows ≥10 foci. Data are from three experiments. (B) SQ/TQ foci from mock-treated or 4 Gy-irradiated primary ear fibroblasts. (C) Primary ear fibroblasts were pretreated for 1 h with caffeine (20 mM) or vehicle and harvested 30 min after treatment with 4 Gy IR as indicated. For each treatment, 250 cells were counted for presence or absence of SQ/TQ foci. Data are from two experiments run in duplicate. (D) Extracts from primary MEF cultures prepared after mock treatment (-); 1 h after 1 μM CPT, 2 mM HU, or 20 J/m2 UV; or 10 min after 5 Gy of IR treatment were sequentially immunoblotted with γ-H2AX and Actin (loading control) antisera. “Short” and “Long” represent short and long exposure, respectively.
Rad50S/S suppresses ATM deficiency
Remarkably, the phenotypic rescue in Rad50S/S Atm-/- was bidirectional. Whereas the data clearly demonstrated that the Mre11 complex activates ATM-dependent apoptotic pathways, Rad50S/S Atm-/- exhibited an additional striking feature. The latency of lymphomagenesis associated with ATM deficiency was profoundly increased in Rad50S/S Atm-/- double mutants. The majority of Atm-/- mice in our colony died with thymic lymphomas by 5 mo of age, 2.9% lived to 10 mo, and none lived beyond 15 mo (Fig. 1B). In contrast, Rad50S/S Atm-/- mice showed increased survival compared with Atm-/- mice: 20.5% of Rad50S/S Atm-/- mice survived to 10 mo (P < 0.005) and 18% survived to 15 mo (P < 0.001). It is conceivable that the increased latency of lymphomagenesis is due to a qualitative difference between the lymphomas arising in Atm-/- versus Rad50S/S Atm-/- mice. Unlike Atm-/- mice, T-cell development in Rad50S/S Atm-/- mice was essentially the same as in wild-type mice (data not shown).
To analyze the mechanism(s) by which the Rad50S allele suppressed ATM deficiency, we assessed DNA damage signaling in primary Rad50S/S Atm-/- cells. Substrates of the three ATM-related kinases, ATM, DNA-PKcs, and ATR have SQ/TQ motifs at the site of phosphorylation (Kim et al. 1999). We reasoned that a general assessment of DNA damage signaling in Rad50S/S and Rad50S/S Atm-/- cells could be obtained by immunofluorescence experiments using phospho-SQ/TQ antiserum. This approach has recently been applied to the analysis of Mlh1-dependent checkpoint signaling (Stojic et al. 2004). Consistent with the previous data indicating constitutive DNA damage signaling, SQ/TQ foci were observed in unirradiated Rad50S/S cells at a significantly higher frequency than in wild-type cells (Fig. 5A,B). After γ-irradiation, >90% of wild-type and Rad50S/S cells formed foci, whereas only 16% of Atm-/- fibroblasts had foci. Rad50S/S Atm-/- fibroblasts exhibited SQ/TQ foci in 36% of the cells (Fig. 5A). Focus formation was reduced in caffeine (20 mM)-treated Rad50S/S Atm-/- cells (Fig. 5C), and partial colocalization with 53BP1 foci was observed in all genotypes studied (data not shown). Although we cannot rule out the possibility that Rad50S/S cells exhibit a small amount of DNA damage, these data suggest that Rad50S activates a compensating DNA damage response pathway that leads to the rescue of ATM deficiency.
H2AX phosphorylation to create γ-H2AX is another general indicator of DNA damage signaling. This event can be effected by ATM, ATR, or DNA-PKcs according to the experimental setting (Burma et al. 2001; Ward and Chen 2001). Consistent with previous data (Bender et al. 2002), γ-H2AX was present in untreated Rad50S/S mouse embryonic fibroblasts (MEFs) (Fig. 5D). γ-H2AX formation was observed in response to camptothecin (CPT), hydroxyurea (HU), ultraviolet light (UV), and IR treatment in wild-type and Rad50S/S MEFs. Atm-/- cells exhibited weak γ-H2AX signal following camptothecin, HU, and UV, whereas signal was absent following IR. In contrast, substantial γ-H2AX formation was seen in Rad50S/S Atm-/- cells following all treatments, including IR (Fig. 5D).
These molecular indices were associated with the mitigation of cellular phenotypic features in Rad50S/S Atm-/- cells. Cultured Atm-/- MEFs exhibit senescence-like proliferative failure (Barlow et al. 1996; Xu and Baltimore 1996). In contrast, the growth of Rad50S/S Atm-/- MEFs was comparable to wild type and Rad50S/S (Fig. 6A). Rad50S/S Atm-/- cells were also significantly more radioresistant than Atm-/- cells, with the double mutants approximating wild-type and Rad50S/S survival at low IR doses (Fig. 6B). The enhanced radioresistance, as well as the increase in SQ/TQ foci formation, is not an effect of cell cycle distribution (data not shown). Rad50S/S did not reduce Mre11ATLD1/ATLD1 IR sensitivity, as both Mre11ATLD1/ATLD1 and Rad50S/S Mre11ATLD1/ATLD1 MEFs exhibited the same degree of IR sensitivity (Fig. 6C).
Figure 6.
Rad50S rescues the growth defects and IR-sensitivity of Atm-/- cells. (A) Cumulative growth curve. Passage 2 MEFs from two cultures were seeded in triplicate onto 6-well plates, counted every 3 d, and replated for four passages. Genotypes as indicated. (B,C) IR sensitivity determined by colony forming assay. (B) The surviving fractions of wild-type (WT), Atm-/-, Rad50S/S, and Rad50S/S Atm-/- SV40-transformed MEFs after various doses of IR are shown. Note that the wild-type and Rad50S/S curves overlap. Graph contains data from three independent experiments in which cells were plated in triplicate at each dose. Similar results were obtained with SV40-transformed ear fibroblasts. (C) The surviving fractions of wild-type (WT), Mre11ATLD/ATLD1, Rad50S/S, Rad50S/S Mre11+/ATLD1, and Rad50S/S Mre11ATLD/ATLD1 SV40-transformed MEFs after various doses of IR are shown.
In addition, Rad50S/S Atm-/- cells had a slight reduction in chromosome instability relative to Atm-/-; however, Rad50S/S had no effect on instability in Mre11ATLD1/ATLD1 (Supplementary Tables 2, 3).
Although certain aspects of ATM deficiency were substantially mitigated in Rad50S/S Atm-/- mice, the effect was relatively circumscribed. Rad50S/S did not rescue the IR-induced G1/S, intra-S-phase, and G2/M checkpoint defects in Atm-/- or Mre11ATLD1/ATLD1 (Supplementary Fig. 1A–C). Nor did Rad50S/S rescue the IR-dependent Atm-/- phosphorylation defects on 53BP1 Ser25 (Fig. 4B), SMC1 Ser957, Chk2, and p53 Ser18 (Fig. 4C; Supplementary Fig. 1D), or the IR-dependent Mre11ATLD1/ATLD1 and Nbs1ΔB/ΔB phosphorylation defects on SMC1 Ser957 and Chk2 (Supplementary Fig. 1D). This suggests that the alternative pathway activated by Rad50S has a relatively limited overlap with the canonical ATM pathway.
Discussion
In this study, we exploit the behavior of the Rad50S allele to examine the Mre11 complex's role in promoting apoptosis. We show that the precipitous apoptotic attrition of hematopoietic cells in Rad50S/S mice is abrogated by ATM deficiency. Consistent with the ATM dependence of this rescue, hypomorphic mutations in the Mre11 complex (Mre11ATLD1 and Nbs1ΔB alleles) that are associated with reduced ATM activity also abrogate Rad50S/S cellular attrition. Similar rescue is effected by Chk2 and p53 deficiency. These data indicate that the Mre11 complex governs an ATM–Chk2–p53-dependent apoptotic pathway. Our results further indicate that in addition to the aforementioned ATM-dependent apoptotic pathway, Rad50S also partially suppresses the lymphomagenesis, senescence, and radiosensitivity associated with ATM deficiency by activating a compensating pathway.
Parallel endpoints of the Mre11 complex–ATM DNA damage response pathway
Whereas it is well-established that the Mre11 complex and ATM collaborate in the activation of cell cycle checkpoints, (D'Amours and Jackson 2002; Stracker et al. 2004), these data provide the first indication that the Mre11 complex has an effect on ATM-dependent apoptosis. This finding was somewhat unexpected as neither Nbs1ΔB/ΔB nor Mre11ATLD1/ATLD1 mice exhibit defects in IR-induced thymocyte apoptosis (Theunissen et al. 2003). Hematopoietic attrition in Rad50S/S mice is rescued by reduced ATM dosage, and by two Mre11 complex mutations that attenuate ATM activity, the Mre11ATLD1 allele (Theunissen et al. 2003) and the Nbs1ΔB allele (Williams et al. 2002). Accordingly, levels of cleaved Caspase-3 are reduced in the thymus and bone marrow of these mice. Rad50S/S Eμ-bcl-2 mice also exhibit significantly increased survival compared with Rad50S/S, confirming that the Rad50S allele activates apoptosis. Chk2 and p53 are the downstream regulators of the apoptotic pathway governed by the Mre11 complex, since deficiency of those proteins abrogates the rapid apoptotic attrition seen in Rad50S/S mice (Bender et al. 2002; this study).
The Mre11 complex's influence on apoptosis was separable from its influence on checkpoint activation. SMC1 is phosphorylated by ATM following DNA damage. This event is dependent upon the Mre11 complex and is required for activation of the intra-S-phase checkpoint (Kim et al. 2002; Yazdi et al. 2002; Kitagawa et al. 2004). SMC1 is constitutively phosphorylated in Rad50S/S mice. This outcome is abrogated by ATM deficiency and Mre11ATLD1. We reasoned that if apoptosis were a downstream consequence of SMC1 phosphorylation, Smc12SA/2SA would rescue Rad50S/S cellular attrition. However, Smc12SA/2SA had no effect on the Rad50S/S phenotype, indicating that SMC1 is not in the apoptotic pathway governed by the Mre11 complex. Thus, SMC1 defines a branch in the Mre11 complex–ATM pathway, and apoptosis and checkpoint activation are parallel outcomes of the pathway. Collectively, these data reveal that the Mre11 complex is required for ATM to initiate parallel DNA damage responses, leading to the regulation of chromosome stability and cell cycle checkpoints on one hand, and to the induction of apoptosis on the other.
Possible mechanisms of Rad50S-dependent pathology
The molecular basis of chronic DNA damage signaling in Rad50S/S is not clear. We have proposed that in S. cerevisiae and mammals, the Rad50S-containing Mre11 complex is an up-mutant for DNA damage signaling, and in this regard, may analogize a receptor mutant in which a signaling takes place in the absence of ligand—in this speculative scenario, the ligand in question would be DNA damage (Petrini and Stracker 2003). This hypothesis is based in part on the observation that, despite indices of chronic genotoxic stress, no evidence for the accumulation of DNA damage in either S. cerevisiae or murine Rad50S mutants has been obtained, and neither organism exhibits overt DNA repair defects.
Several lines of evidence from this study support this hypothesis. Reducing Rad50S dosage (in Rad50Δ/S mice) enhances survival. If Rad50S were a hypomorphic allele that affected Mre11 complex-mediated DNA repair, Rad50Δ/S would likely exhibit a more severe phenotype than Rad50S/S. The fact that neither Nbs1ΔB/ΔB nor Mre11ATLD1/ATLD1, both of which are bona fide hypomorphic mutants, enhance the severity of Rad50S/S is also inconsistent with the idea that impaired DNA damage metabolism underlies the Rad50S/S phenotype. The Mre11 complex's cytologic behavior is normal in Rad50S/S cells (Bender et al. 2002), and we have not observed a significant increase in Rad51 spontaneous foci (data not shown). These observations, together with the fact that Rad50S/S cells do not exhibit sensitivity to a broad range of clastogens (Bender et al. 2002), support the interpretation that the enhanced signaling in Rad50S/S is not due to a defect in DSB repair. Finally, the Rad50S allele's extremely mild effect on chromosome stability would appear to be insufficient to account for the severity of its effect on apoptosis (Bender et al. 2002; de Jager and Kanaar 2002). For comparison, nothing resembling the Rad50S/S phenotype is seen in murine mutants associated with chromosome instability or DNA repair defects. Some examples include Mre11ATLD1/ATLD1 mice (Theunissen et al. 2003), Ku (Difilippantonio et al. 2000), scid (Bosma et al. 1983), Rad54 (Essers et al. 1997), Mus81 (McPherson et al. 2004), Brca1 (Ludwig et al. 2001), and H2AX (Bassing et al. 2003; Celeste et al. 2003). Notably, the level of chromosome breakage, both spontaneous and induced, appears to be substantially higher in Mre11ATLD1/ATLD1 mice than in the strains noted above, yet bears no phenotypic resemblance to Rad50S/S.
An alternative, nonexclusive hypothesis is also supported by the data. The promotion of apoptosis by the Rad50S allele may reflect a gain of function that leads to the induction or stabilization of a unique lesion. This lesion would necessarily be one that is not a major product of the clastogenic agents we have used (IR, mitomycin C, or HU), as no sensitivity of Rad50S/S cells to those agents is evident (Bender et al. 2002; data not shown). Supporting this possibility is the fact that Rad50S/S double mutants in which cellular attrition is attenuated are frequently associated with lymphomagenesis. Rad50S/S Chk2-/- is particularly affected; 10 of the 12 mice died with lymphoma (Fig. 3B). It is important to consider that the lesion hypothesis does not exclude the possibility that Rad50S/S is hypermorphic for signaling, and that the Rad50S/S phenotype may be a composite outcome of the two.
An intriguing possibility for the Rad50S/S-dependent lesion is a covalent DNA–protein complex. The Mre11 complex is implicated in the nucleolytic removal of covalently attached topoisomerase II or topoisomerase II-like proteins in bacteriophage T4 and S. cerevisiae (Keeney and Kleckner 1995; Nairz and Klein 1997; Tsubouchi and Ogawa 1998; Stohr and Kreuzer 2001). For example, the removal of Spo11 from meiotic DSBs is completely blocked in S. cerevisiae rad50S mutants (Keeney and Kleckner 1995). Recent data demonstrate that Spo11 is removed from DSB ends by endonucleolytic cleavage adjacent to the bound protein in both S. cerevisiae and mice, and further, that this mode of repair may be generally relevant to the removal of covalent topoisomerase II cleavage complexes (Neale et al. 2005). The involvement of the Mre11 complex in this repair process remains to be addressed.
Activation of an alternative DNA damage response pathway in Rad50S/S mice
An equally striking aspect of Rad50S/S is its effect on ATM deficiency. Rad50S/S rescues the lymphomagenesis, senescence, and radiosensitivity of ATM-deficient mice and cells. This rescue is associated with enhancement of several generic readouts of ATM/ATR signaling in Rad50S/S Atm-/- cells. For example, SQ/TQ foci form in response to IR treatment at a higher frequency in Rad50S/S Atm-/- compared with Atm-/- fibroblasts. Since this outcome is blocked by caffeine treatment, the data suggest that Rad50S/S activates a PI3K-like kinase with overlapping specificity to that of ATM. In addition, γ-H2AX formation after treatment with several DNA damaging agents is enhanced in Rad50S/S Atm-/- MEFs. These results support the hypothesis that activation of a compensating DNA damage response pathway in Rad50S/S is the underlying basis for suppression of ATM deficiency in Rad50S/S Atm-/- mice. This situation is reminiscent of the observation that a S. cerevisiae rad50S mutation is associated with partial suppression of the checkpoint defects in Mec1-deficient cells. These outcomes are dependent upon the Mec1 paralog, Tel1, and the Mre11 complex (Usui et al. 2001).
Rad50S/S did not mitigate phenotypic outcomes in Mre11ATLD1/ATLD1. This may reflect that Mre11ATLD1/ATLD1 impairs the activity of both the alternative and the ATM-dependent pathway. Supporting this interpretation, Mre11ATLD1/ATLD1 is synthetically lethal with Atm-/- (Table 1), as is Nbs1ΔB/ΔB (Williams et al. 2002).
Table 1.
Synthetic lethality of Atm-/- Mre11ATLD1/ATLD1
| Genotype father | Genotype mother | Total no. of pups | Expected no. of double mutantsa | Observed no. of double mutants | P-valueb |
|---|---|---|---|---|---|
| Atm+/- Mre11+/ATLD1 | Atm+/- Mre11+/ATLD1 | 245 | 4.8 | 0 | 7.8 × 10-3 |
| Atm+/- Mre1ATLD1/ATLD1 | Atm+/- Mre11+/ATLD1 | 112 | 7.8 | 0 | 3.0 × 10-4 |
The number of expected double mutants is calculated based on a physical distance of 28 centimorgans (cM) between the ATM and MRE11 loci. On the Mouse Genome Informatics Web site (http://www.informatics.jax.org), the ATM locus maps to 30 cM on chromosome 9 and GPR83 (a marker 25,820 bp from the MRE11 Ensembl Transcript Report [http://www.ensemble.org]) maps to 2 cM on chromosome 9.
P-value based on binomial distribution.
Since neither DNA-PKcs nor SMG1 have been implicated in the suppression of lymphomas, the alternative pathway is most likely to be ATR dependent. Consistent with this view, recent data suggest that Nbs1 influences the activity of ATR on certain targets (Stiff et al. 2004). However, the data in hand do not rigorously exclude a role for DNA-PKcs or SMG1 in the Rad50S/S-dependent suppression of ATM deficiency. Irrespective of the kinase involved, the data clearly support the view that the influence of the Mre11 complex on the DNA damage response is not limited to ATM-dependent functions.
Finally, the suggestion that Rad50S/S activates both ATM-dependent and ATM-independent responses may have therapeutic implications. On one hand, the fact that a pathway that mitigates ATM deficiency exists and can be activated may provide a basis for the development of treatments for ataxia-telangiectasia. On the other hand, the potent pro-apoptotic behavior of Rad50S raises the possiblity that compounds mimicking this activity could be developed as anti-tumor agents that act with reduced mutagenic effects.
Materials and methods
Mice derivation and genotyping
Rad50S/S, Rad50+/Δ, Atm-/-, Mre11ATLD1/ATLD1, Nbs1ΔB/ΔB, Chk2-/-, Smc12SA/2SA (= Smc1S957AS966A/S957AS966A), and Eμ-Bcl2 mice, survival, and genotyping have been described (Strasser et al. 1990; Barlow et al. 1996; Luo et al. 1999; Bender et al. 2002; Hirao et al. 2002; Williams et al. 2002; Theunissen et al. 2003; Kitagawa et al. 2004). All mice maintained on mixed 129/SvEv and C57BL6 background. Rad50S/S, Atm-/-, Rad50S/S Atm-/-, and Rad50S/S Atm+/- mice were derived at the University of Wisconsin Medical School and at Memorial Sloan-Kettering Cancer Center, and Figure 1B is a compilation of mice in both colonies. All other survival curves include mice born only at Memorial Sloan Kettering Cancer Center.
Cell derivation and culture
Murine embryonic fibroblasts (MEFs) and ear fibroblasts were generated and cultured as described (Bender et al. 2002). Immortalization was achieved by pX-8 transfection, an SV40 plasmid containing an inactivated origin of replication (Fromm and Berg 1982). For cumulative growth, the fold increase in cell number (R) was calculated from passage number 2 to passage number 6. Between each passage number, 100,000 cells were plated on six-well plates, and R = X/100,000 with X = cell number after 3 d. The cumulative cell number Y at each passage number (p) was obtained by solving Y(p) = Y(p - 1) × R with Y(2) = 100,000 and p = 3, 4, 5, and 6.
Survival analyses
Statistical significance determined by Wilcoxon Rank Sum Test or Fisher's exact test using Mstat software (Norman Drink-water, McArdle Laboratory for Cancer Research). Kaplan Meier survival curves made with Prism 4 (GraphPad Software).
Hematopoietic cell preparation and analysis
Single-cell suspensions from the thymus and bone marrow prepared and analyzed as described (Bender et al. 2002). Briefly, the number of DN T cells was equal to the number of CD4 CD8 double-negative cells in the thymus, the number of macrophages was equal to the number of the B220-negative and CD43-positive bone marrow cells, and the number of pro-B cells was equal to the number of B220 CD43 double-positive bone marrow cells.
Cellular assays
Sensitivity to IR, intra-S-phase, and G2/M checkpoint functions assessed as described (Williams et al. 2002). Cells were irradiated with a Mark I 137Cs source at 222 cGy/min. G1/S checkpoint assays carried out as described (Xu et al. 1996). Chromosomal analyses were prepared as described previously (Bender et al. 2002) and carried out on blinded samples. Splenocytes were stimulated with lipopolysaccharide for 48 h prior to addition of colcemid for 3 h. Ear fibroblasts were incubated for 4 h with colcemid.
Immunohistochemistry
Four percent paraformaldehyde-fixed tissues were processed at the Research Animal Resource Center of the Cornell University Medical College. Paraffin-embedded 5–10-μm sections were stained with hematoxylin and eosin, and cleaved Caspase-3 (Cell Signalling; #9661) immunohistochemistry was performed at the Molecular Cytology Core Facility of Memorial Sloan-Kettering Cancer Center. Color images were captured using a Nikon digital camera and processed using Adobe Photoshop.
Immunoprecipitations and Western blotting
For 53BP1 IP, whole-cell extracts of primary ear fibroblasts were obtained in lysis buffer (300 mM NaCl, 20 mM Hepes at pH 7.9 and 0.5% NP-40 with protease and phosphatase inhibitors). One microgram of 53BP1-Ser25-P antibody (gift from P. Carpenter, Harvard Medical School, Boston, MA) was added to the extracts for 30 min, and proteins were immunoprecipitated for 1 h with protein A/G agarose. IPs were washed twice with wash buffer (150 mM NaCl, 20 mM Hepes at pH 7.9 and 0.5% NP-40 with protease and phosphatase inhibitors) and loaded on a gel. Lysates for Western blot were obtained in lysis buffer (200–400 mM NaCl, 50 mM Tris-HCl at pH 7.5, 1% NP-40 with protease and phosphatase inhibitors). To prepare extracts for γ-H2AX detection, the chromatin pellet from regular lysates was further incubated for 30 min in 0.1 M HCl, and the resulting supernatant was used for immunoblotting. Chk2 (Upstate Biotechnology; 05-649) antiserum used at 1:400 dilution in 2% BSA/PBS. 53BP1 (gift from P. Carpenter) and γ-H2AX (Cell Signaling) antisera used at a 1:500 dilution in 5% Milk/0.1% Tween/PBS (MTP). p53 (Novacastra; NCL-p53-CM5p), p53-Ser15-P (Cell Signaling; #9284), SMC1-Ser957-P (Cell Signaling; #4805), and ATM-Ser1981-P (Cell Signaling; #4526) antisera were used at 1:1000 dilution in MTP. Actin (Sigma; A4700), PCNA (Calbiochem; NA03), SMC1 (Novus Biologicals; ab9262), and ATM (MAT3; gift from Y. Shiloh, Sackler School of Medicine, Tel Aviv University, Tel Aviv, Israel) antisera were used at 1:2000 dilution in MTP. For ATM detection, IPs and lysates were loaded on 3%–8% Tris-Acetate gels (NuPAGE, Invitrogen).
Immunofluorescence assays
Immunofluorescence experiments were done as previously described (Maser et al. 1997). SQ/TQ antibody (Cell Signaling; #2851) was used at a 1:500 dilution in 1% BSA/PBS.
Supplementary Material
Acknowledgments
We are grateful to Fred Alt and David Ferguson for SKY analyses for Atm-/- and Rad50S/S Atm-/- splenocytes, and Tak Mak and Howard Petrie for mouse strains. We thank Doug Bishop and members of the Petrini laboratory for insights, and Andy Koff and Scott Keeney for critical reading of the manuscript. This work was supported by NIH grants GM56888, CA03632, and CA21765, and the Joel and Joan Smilow Initiative.
Supplemental material is available at http://www.genesdev.org.
Article and publication are at http://www.genesdev.org/cgi/doi/10.1101/gad.1373705.
References
- Abraham R.T. 2004. PI 3-kinase related kinases: `Big' players in stress-induced signaling pathways. DNA Repair 3: 883-887. [DOI] [PubMed] [Google Scholar]
- Bakkenist C.J. and Kastan, M.B. 2003. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 421: 499-506. [DOI] [PubMed] [Google Scholar]
- Barlow C., Hirotsune, S., Paylor, R., Liyanage, M., Eckhaus, M., Collins, F., Shiloh, Y., Crawley, J.N., Ried, T., Tagle, D., et al. 1996. Atm-deficient mice: A paradigm of ataxia telangiectasia. Cell 86: 159-171. [DOI] [PubMed] [Google Scholar]
- Bassing C.H., Suh, H., Ferguson, D.O., Chua, K.F., Manis, J., Eckersdorff, M., Gleason, M., Bronson, R., Lee, C., and Alt, F.W. 2003. Histone H2AX: A dosage-dependent suppressor of oncogenic translocations and tumors. Cell 114: 359-370. [DOI] [PubMed] [Google Scholar]
- Bender C.F., Sikes, M.L., Sullivan, R., Huye, L.E., Le Beau, M.M., Roth, D.B., Mirzoeva, O.K., Oltz, E.M., and Petrini, J.H. 2002. Cancer predisposition and hematopoietic failure in Rad50(S/S) mice. Genes & Dev. 16: 2237-2251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosma G., Custer, R., and Bosma, M. 1983. A severe combined immunodeficiency mutation in mice. Nature 301: 527-530. [DOI] [PubMed] [Google Scholar]
- Brumbaugh K.M., Otterness, D.M., Geisen, C., Oliveira, V., Brognard, J., Li, X., Lejeune, F., Tibbetts, R.S., Maquat, L.E., and Abraham, R.T. 2004. The mRNA surveillance protein hSMG-1 functions in genotoxic stress response pathways in mammalian cells. Mol. Cell 14: 585-598. [DOI] [PubMed] [Google Scholar]
- Burma S., Chen, B.P., Murphy, M., Kurimasa, A., and Chen, D.J. 2001. ATM phosphorylates histone H2AX in response to DNA double-strand breaks. J. Biol. Chem. 276: 42462-42467. [DOI] [PubMed] [Google Scholar]
- Carney J.P., Maser, R.S., Olivares, H., Davis, E.M., Le Beau, M., Yates III, J.R., Hays, L., Morgan, W.F., and Petrini, J.H. 1998. The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: Linkage of double-strand break repair to the cellular DNA damage response. Cell 93: 477-486. [DOI] [PubMed] [Google Scholar]
- Carson C.T., Schwartz, R.A., Stracker, T.H., Lilley, C.E., Lee, D.V., and Weitzman, M.D. 2003. The Mre11 complex is required for ATM activation and the G(2)/M checkpoint. EMBO J. 22: 6610-6620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Celeste A., Difilippantonio, S., Difilippantonio, M.J., Fernandez-Capetillo, O., Pilch, D.R., Sedelnikova, O.A., Eckhaus, M., Ried, T., Bonner, W.M., and Nussenzweig, A. 2003. H2AX haploinsufficiency modifies genomic stability and tumor susceptibility. Cell 114: 371-383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costanzo V., Paull, T., Gottesman, M., and Gautier, J. 2004. Mre11 assembles linear DNA fragments into DNA damage signaling complexes. PLoS Biol. 2: E110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Amours D. and Jackson, S.P. 2002. The Mre11 complex: At the crossroads of DNA repair and checkpoint signalling. Nat. Rev. Mol. Cell. Biol. 3: 317-327. [DOI] [PubMed] [Google Scholar]
- de Jager M. and Kanaar, R. 2002. Genome instability and Rad50(S): Subtle yet severe. Genes & Dev. 16: 2173-2178. [DOI] [PubMed] [Google Scholar]
- Difilippantonio M.J., Zhu, J., Chen, H.T., Meffre, E., Nussenzweig, M.C., Max, E.E., Ried, T., and Nussenzweig, A. 2000. DNA repair protein Ku80 suppresses chromosomal aberrations and malignant transformation. Nature 404: 510-514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Difilippantonio S., Celeste, A., Fernandez-Capetillo, O., Chen, H.T., Reina San Martin, B., Van Laethem, F., Yang, Y.P., Petukhova, G.V., Eckhaus, M., Feigenbaum, L., et al. 2005. Role of Nbs1 in the activation of the Atm kinase revealed in humanized mouse models. Nat. Cell. Biol. 7: 675-685. [DOI] [PubMed] [Google Scholar]
- Essers J., Hendriks, R.W., Swagemakers, S.M., Troelstra, C., de Wit, J., Bootsma, D., Hoeijmakers, J.H., and Kanaar, R. 1997. Disruption of mouse RAD54 reduces ionizing radiation resistance and homologous recombination. Cell 89: 195-204. [DOI] [PubMed] [Google Scholar]
- Falck J., Coates, J., and Jackson, S.P. 2005. Conserved modes of recruitment of ATM, ATR and DNA-PKcs to sites of DNA damage. Nature 434: 605-611. [DOI] [PubMed] [Google Scholar]
- Fromm M. and Berg, P. 1982. Deletion mapping of DNA regions required for SV40 early region promoter function in vivo. J. Mol. Appl. Genet. 1: 457-481. [PubMed] [Google Scholar]
- Gatei M., Young, D., Cerosaletti, K.M., Desai-Mehta, A., Spring, K., Kozlov, S., Lavin, M.F., Gatti, R.A., Concannon, P., and Khanna, K. 2000. ATM-dependent phosphorylation of nibrin in response to radiation exposure. Nat. Genet. 25: 115-119. [DOI] [PubMed] [Google Scholar]
- Hirao A., Cheung, A., Duncan, G., Girard, P.M., Elia, A.J., Wakeham, A., Okada, H., Sarkissian, T., Wong, J.A., Sakai, T., et al. 2002. Chk2 is a tumor suppressor that regulates apoptosis in both an ataxia telangiectasia mutated (ATM)-dependent and an ATM-independent manner. Mol. Cell. Biol. 22: 6521-6532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horejsi Z., Falck, J., Bakkenist, C.J., Kastan, M.B., Lukas, J., and Bartek, J. 2004. Distinct functional domains of Nbs1 modulate the timing and magnitude of ATM activation after low doses of ionizing radiation. Oncogene 23: 3122-3127. [DOI] [PubMed] [Google Scholar]
- Kang J., Bronson, R., and Xu, Y. 2002. Targeted disruption of NBS1 reveals its roles in mouse development and DNA repair. EMBO J. 21: 1447-1455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keeney S. and Kleckner, N. 1995. Covalent protein–DNA complexes at the 5′ strand termini of meiosis-specific double-strand breaks in yeast. Proc. Natl. Acad. Sci. 92: 11274-11278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S.T., Lim, D.S., Canman, C.E., and Kastan, M.B. 1999. Substrate specificities and identification of putative substrates of ATM kinase family members. J. Biol. Chem. 274: 37538-37543. [DOI] [PubMed] [Google Scholar]
- Kim S.-T., Xu, B., and Kastan, M.B. 2002. Involvement of the cohesin protein Smc1, in Atm-dependent and independent responses to DNA damage. Genes & Dev. 16: 560-570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitagawa R., Bakkenist, C.J., McKinnon, P.J., and Kastan, M.B. 2004. Phosphorylation of SMC1 is a critical downstream event in the ATM–NBS1–BRCA1 pathway. Genes & Dev. 18: 1423-1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J.H. and Paull, T.T. 2005. ATM activation by DNA double-strand breaks through the Mre11–Rad50–Nbs1 complex. Science 308: 551-554. [DOI] [PubMed] [Google Scholar]
- Lim D.S., Kim, S.T., Xu, B., Maser, R.S., Lin, J., Petrini, J.H., and Kastan, M.B. 2000. ATM phosphorylates p95/nbs1 in an S-phase checkpoint pathway. Nature 404: 613-617. [DOI] [PubMed] [Google Scholar]
- Ludwig T., Fisher, P., Ganesan, S., and Efstratiadis, A. 2001. Tumorigenesis in mice carrying a truncating Brca1 mutation. Genes & Dev. 15: 1188-1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo G., Yao, M.S., Bender, C.F., Mills, M., Bladl, A.R., Bradley, A., and Petrini, J.H. 1999. Disruption of mRad50 causes embryonic stem cell lethality, abnormal embryonic development, and sensitivity to ionizing radiation. Proc. Natl. Acad. Sci. 96: 7376-7381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maser R.S., Monsen, K.J., Nelms, B.E., and Petrini, J.H. 1997. hMre11 and hRad50 nuclear foci are induced during the normal cellular response to DNA double-strand breaks. Mol. Cell. Biol. 17: 6087-6096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McPherson J.P., Lemmers, B., Chahwan, R., Pamidi, A., Migon, E., Matysiak-Zablocki, E., Moynahan, M.E., Essers, J., Hanada, K., Poonepalli, A., et al. 2004. Involvement of mammalian Mus81 in genome integrity and tumor suppression. Science 304: 1822-1826. [DOI] [PubMed] [Google Scholar]
- Mochan T.A., Venere, M., DiTullio Jr., R.A., and Halazonetis, T.D. 2003. 53BP1 and NFBD1/MDC1-Nbs1 function in parallel interacting pathways activating ataxia-telangiectasia mutated (ATM) in response to DNA damage. Cancer Res. 63: 8586-8591. [PubMed] [Google Scholar]
- Nairz K. and Klein, F. 1997. mre11S—A yeast mutation that blocks double-strand-break processing and permits nonhomologous synapsis in meiosis. Genes & Dev. 11: 2272-2290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neale M.J., Pan, J., and Keeney, S. 2005. Endonucleolytic processing of covalent protein-linked DNA double-strand breaks. Nature 436: 1053-1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrini J.H. 2000. The Mre11 complex and ATM: Collaborating to navigate S phase. Curr. Opin. Cell. Biol. 12: 293-296. [DOI] [PubMed] [Google Scholar]
- Petrini J.H. and Stracker, T.H. 2003. The cellular response to DNA double-strand breaks: Defining the sensors and mediators. Trends Cell. Biol. 13: 458-462. [DOI] [PubMed] [Google Scholar]
- Stewart G.S., Maser, R.S., Stankovic, T., Bressan, D.A., Kaplan, M.I., Jaspers, N.G., Raams, A., Byrd, P.J., Petrini, J.H., and Taylor, A.M. 1999. The DNA double-strand break repair gene hMRE11 is mutated in individuals with an ataxia-telangiectasia-like disorder. Cell 99: 577-587. [DOI] [PubMed] [Google Scholar]
- Stiff T., Reis, C., Alderton, G.K., Woodbine, L., O'Driscoll, M., and Jeggo, P.A. 2004. Nbs1 is required for ATR-dependent phosphorylation events. EMBO J 24: 199-208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stohr B.A. and Kreuzer, K.N. 2001. Repair of topoisomerase-mediated DNA damage in bacteriophage t4. Genetics 158: 19-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stojic L., Mojas, N., Cejka, P., Di Pietro, M., Ferrari, S., Marra, G., and Jiricny, J. 2004. Mismatch repair-dependent G2 checkpoint induced by low doses of SN1 type methylating agents requires the ATR kinase. Genes & Dev. 18: 1331-1344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stracker T.H., Carson, C.T., and Weitzman, M.D. 2002. Adenovirus oncoproteins inactivate the Mre11–Rad50–NBS1 DNA repair complex. Nature 418: 348-352. [DOI] [PubMed] [Google Scholar]
- Stracker T.H., Theunissen, J.W., Morales, M., and Petrini, J.H. 2004. The Mre11 complex and the metabolism of chromosome breaks: The importance of communicating and holding things together. DNA Repair 3: 845-854. [DOI] [PubMed] [Google Scholar]
- Strasser A., Harris, A.W., Vaux, D.L., Webb, E., Bath, M.L., Adams, J.M., and Cory, S. 1990. Abnormalities of the immune system induced by dysregulated bcl-2 expression in transgenic mice. Curr. Top. Microbiol. Immunol. 166: 175-181. [DOI] [PubMed] [Google Scholar]
- Strasser A., Harris, A.W., and Cory, S. 1991a. bcl-2 transgene inhibits T cell death and perturbs thymic self-censorship. Cell 67: 889-899. [DOI] [PubMed] [Google Scholar]
- Strasser A., Whittingham, S., Vaux, D.L., Bath, M.L., Adams, J.M., Cory, S., and Harris, A.W. 1991b. Enforced BCL2 expression in B-lymphoid cells prolongs antibody responses and elicits autoimmune disease. Proc. Natl. Acad. Sci. 88: 8661-8665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theunissen J.W., Kaplan, M.I., Hunt, P.A., Williams, B.R., Ferguson, D.O., Alt, F.W., and Petrini, J.H. 2003. Checkpoint failure and chromosomal instability without lymphomagenesis in Mre11(ATLD1/ATLD1) mice. Mol. Cell 12: 1511-1523. [DOI] [PubMed] [Google Scholar]
- Tsubouchi H. and Ogawa, H. 1998. A novel mre11 mutation impairs processing of double-strand breaks of DNA during both mitosis and meiosis. Mol. Cell. Biol. 18: 260-268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Usui T., Ogawa, H., and Petrini, J.H. 2001. A DNA damage response pathway controlled by Tel1 and the Mre11 complex. Mol. Cell 7: 1255-1266. [DOI] [PubMed] [Google Scholar]
- Uziel T., Lerenthal, Y., Moyal, L., Andegeko, Y., Mittelman, L., and Shiloh, Y. 2003. Requirement of the MRN complex for ATM activation by DNA damage. EMBO J. 22: 5612-5621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward I.M. and Chen, J. 2001. Histone H2AX is phosphorylated in an ATR-dependent manner in response to replicational stress. J. Biol. Chem. 276: 47759-47762. [DOI] [PubMed] [Google Scholar]
- Williams B.R., Mirzoeva, O.K., Morgan, W.F., Lin, J., Dunnick, W., and Petrini, J.H. 2002. A murine model of nijmegen breakage syndrome. Curr. Biol. 12: 648-653. [DOI] [PubMed] [Google Scholar]
- Wu X., Ranganathan, V., Weisman, D.S., Heine, W.F., Ciccone, D.N., O'Neill, T.B., Crick, K.E., Pierce, K.A., Lane, W.S., Rathbun, G., et al. 2000. ATM phosphorylation of Nijmegen breakage syndrome protein is required in a DNA damage response. Nature 405: 477-482. [DOI] [PubMed] [Google Scholar]
- Xu Y. and Baltimore, D. 1996. Dual roles of ATM in the cellular response to radiation and in cell growth control. Genes & Dev. 10: 2401-2410. [DOI] [PubMed] [Google Scholar]
- Xu Y., Ashley, T., Brainerd, E.E., Bronson, R.T., Meyn, M.S., and Baltimore, D. 1996. Targeted disruption of ATM leads to growth retardation, chromosomal fragmentation during meiosis, immune defects, and thymic lymphoma. Genes & Dev. 10: 2411-2422. [DOI] [PubMed] [Google Scholar]
- Yazdi P.T., Wang, Y., Zhao, S., Patel, N., Lee, E.Y., and Qin, J. 2002. SMC1 is a downstream effector in the ATM/NBS1 branch of the human S-phase checkpoint. Genes & Dev. 16: 571-582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- You Z., Chahwan, C., Bailis, J., Hunter, T., and Russell, P. 2005. ATM activation and its recruitment to damaged DNA require binding to the C terminus of Nbs1. Mol. Cell. Biol. 25: 5363-5379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao S., Weng, Y.C., Yuan, S.S., Lin, Y.T., Hsu, H.C., Lin, S.C., Gerbino, E., Song, M.H., Zdzienicka, M.Z., Gatti, R.A., et al. 2000. Functional link between ataxia-telangiectasia and Nijmegen breakage syndrome gene products. Nature 405: 473-477. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.