Abstract
The main objective of this work was to improve the early serologic diagnosis of toxoplasmosis in children at risk of congenital infection by using recombinant antigens. Serum samples from 104 infants born to mothers with primary Toxoplasma gondii infection acquired during pregnancy, of which 35 were congenitally infected and 22 had clinical silent toxoplasmosis at birth, were included. Immunoglobulin M (IgM), IgG, and IgG subtype antibodies against epitopes carried by fragments of T. gondii MIC2, MIC3, MIC4, M2AP, AMA1, and SAG1 gene products were measured by performing parallel enzyme immunoassays (Rec-ELISAs). Recombinant antigens preferentially reacted with IgG antibodies from infected infants compared to uninfected subjects (P < 0.0001), indicating that sera from infected children recognized a more diverse repertoire of antigens than sera transferred over the placenta from the mothers. Using two serial samples collected within 3 months of life, it was possible to demonstrate a neosynthesis of specific anti-MIC2 and anti-SAG1 immunoglobulin G, mainly of the IgG2 subtype, in 13 out of 20 infants with congenital toxoplasmosis. IgM antibodies in 97% of infected infants reacted with at least one of the recombinant antigens, confirming the diagnosis of congenital infection as soon as 2 months after birth (P < 0.0001). The use of recombinant antigens is effective in distinguishing T. gondii-infected from uninfected infants and shows that assays based on recombinant antigens improve the diagnosis of newborns with congenital toxoplasmosis.
Primary infection with Toxoplasma gondii acquired during pregnancy can be transmitted to the fetus and may cause miscarriage, neonatal malformations, or reduced eyesight (15, 37, 43). Toxoplasmosis in gestation represents a challenge for the clinician due to its subclinical course in the majority of pregnant women and to the unpredictable long-term outcome of congenital infection. Transplacental transmission occurs in 10 to 80% of maternal infections, depending on gestational age of the fetus (10, 11). The clinical severity for the fetus decreases and the transmission rate increases as gestational age at the time of maternal infection progresses (10, 21).
At birth, up to 90% of congenitally infected infants are asymptomatic but are at risk of developing retinochoroiditis during the first year of life or in early adulthood. Evidence from cohort studies shows that 15% to 80% of children with prenatal toxoplasmosis develop ocular disease (19, 25-27). Treatment should be started soon after birth, which requires rapid diagnosis (11, 19, 32, 42).
Detection of fetal T. gondii infection before birth can be established using PCR assays or isolating parasites by mouse inoculation from amniotic liquid samples. However, diagnosis of congenital toxoplasmosis during pregnancy by PCR and/or mouse inoculation usually identifies no more than 60 to 70% of the infected fetuses (13, 24, 38, 39, 41). Thus, exploring the antibody response to T. gondii in the newborn child seems an obvious approach to improve the neonatal diagnosis of congenital toxoplasmosis.
Detection of Toxoplasma-specific immunoglobulin M (IgM) and IgA antibodies after birth confirms the diagnosis of congenital toxoplasmosis. However, in 30% to 60% of infected children the Toxoplasma-specific IgM and/or IgA antibody responses are absent or undetectable using standard serological assays, using lysed whole-cell T. gondii antigen (6, 7, 12, 37). Only the persistence or increase of IgG antibodies within the first 12 months of life can confirm congenital infection in the absence of clinical signs. To overcome this extended time lag between diagnosis and initiation of therapy, several additional tests based on comparison of the mother's immunological profile to that of her child have been developed (8, 17, 30, 34-36, 38).
The aim of this study was to improve the early serologic diagnosis of toxoplasmosis in children at risk of congenital infection by using recombinant Toxoplasma antigens. Using sera from infants born to mothers with primary toxoplasmosis acquired during pregnancy, we found that recombinant antigens containing regions of the T. gondii MIC2, MIC3, MIC4, AMA1, M2AP, and SAG1 gene products in enzyme immunoassays improve early diagnosis of congenital toxoplasmosis in newborns.
MATERIALS AND METHODS
Patients.
One hundred four infants born to mothers with primary toxoplasmosis in pregnancy and referred for postnatal follow-up at the Center for Perinatal Infection of Campania Region, Italy, were included in the study. Maternal diagnosis of primary T. gondii infection was based on seroconversion during gestation. All of the women were offered tests for the antenatal diagnosis of congenital toxoplasmosis. When informed consent was granted, amniotic fluid was drawn and then analyzed by PCR for the presence of T. gondii-specific nucleic acids (18). Prenatal diagnosis on amniotic fluid was attempted for 24 of the 104 women. The remaining 100 women did not consent or seroconverted late in gestation.
Treatment. (i) Mothers.
We treated 74 women with spiramycin (3 g/day) alone up to delivery. After 16 weeks of gestation, a 4-week regimen alternating the combination of pyrimethamine (50 mg/day for the first 3 days and then 25 mg/day) plus sulfadiazine (3 g/day) and folinic acid supplementation (P/S therapy) with spiramycin (3 g/day) was instituted in 19 women and continued up to delivery as follows: (i) first maternal IgM-positive test after 20 weeks of gestation (10, 15, 16); (ii) positive PCR result on amniotic fluid (22); and (iii) ultrasonographic anomalies (14). In 11 women treatment was lacking since the diagnosis was made close to delivery or because of the patient's refusal.
(ii) Infants.
Treatment of infants was started when specific anti-Toxoplasma IgM and/or IgA was detected in the infant serum or a lack of IgG decay was demonstrated in two consecutive samples taken more than 2 weeks apart during the diagnostic follow-up. The disease onset was considered severe, benign, or subclinical according to the criteria of Hohlfeld et al. (21). Treatment of cases of severe-onset disease consisted of 6 months of P/S combination (pyrimethamine, 2 mg/kg of body weight per day in the first 3 days, then 1 mg/kg alternating days; sulfadiazine, 100 mg/day) and folinic acid supplementation (5 mg/day, alternating days) followed by 6 months of a regimen alternating 4 weeks of the P/S combination (same dosage as above) with 4 weeks of spiramycin (125 mg/day). Cases of benign or subclinical disease were treated for 12 months with the regimen alternating 4 weeks of P/S combination with 4 weeks of spiramycin.
Diagnosis of congenital toxoplasmosis in newborns.
Postnatal diagnosis in infants was carried out by determining specific anti-Toxoplasma IgM and IgA in the first 3 months of age and IgG at birth and at 1, 2, 3, 6, 9, and 12 months of age. Congenital toxoplasmosis was proven on the basis of persistence of Toxoplasma-specific IgG beyond 12 months of age and was excluded by their disappearance. Brain ultrasonography, cranial computed axial tomography, and indirect fundoscopy established the severity of clinical onset (21).
Analysis of Toxoplasma-specific IgG, IgM, and IgA antibodies and measurement of anti-Toxoplasma IgG titers (given as international units/ml) in infant sera was done by the whole-cell, lysed Toxoplasma antigen assays ELFA-IgG, ELFA-IgM, and ISAGA-IgM (bioMérieux, Marcy-l'Etoile, France) and enzyme-linked immunosorbent assay (ELISA)-IgA (Sanofi-Pasteur, Marnes La Coquette, France). Sera taken from 30 Toxoplasma IgG-negative women referred to the center for other infectious diseases (cytomegalovirus, hepatitis C virus, or rubella virus) were used as controls. All serum samples were analyzed in a blinded fashion.
Recombinant antigens.
Seven Toxoplasma gondii antigens were used: MIC2a, MIC2b, MIC4, M2AP, AMA 1 (5), MIC3, and SAG1 (3). Recombinant antigens were expressed in bacterial cells as fusion proteins with glutathione S-transferase (GST) and purified by affinity chromatography as previously described (3). Briefly, recombinant Escherichia coli cells were induced with IPTG (isopropylthiogalactopyranoside), centrifuged and suspended in STE buffer (10 mM Tris-HCl [pH 8], 150 mM NaCl) containing 100 μg/ml of lysozyme and protease inhibitor cocktail (Boehringer, Germany). The mixture was sonicated at 4°C and Triton-X 100 was added to a final concentration of 1%. After centrifugation at 10,000 × g for 30 min at 4°C, the supernatant was recovered and incubated with glutathione-Sepharose (Amersham Pharmacia Biotech, Sweden). Fusion proteins were eluted by following the manufacturer's instructions and stored at −20°C.
Recombinant protein enzyme immunoassays. (i) IgG Rec-ELISA.
Maxisorb plates (Nunc, Denmark) were coated overnight with recombinant proteins at a concentration of 5 μg/ml in coating buffer (50 mM NaHCO3, pH 9.6). Plates were blocked for 1 h at 37°C with blocking buffer (5% nonfat dry milk, 0.05% Tween 20 in phosphate-buffered saline [PBS]) and then incubated for 1 h at 37°C with serum samples (diluted 1:200). After being washed with 0.05% Tween 20 in PBS, the plates were incubated with alkaline phosphatase-conjugated anti-human IgG antibodies (Sigma-Aldrich,) diluted 1:7,500 in blocking buffer. After 30 min at 37°C the plates were washed and incubated with the chromogenic substrate p-nitrophenylphosphate (pNPP; Sigma-Aldrich) in developing solution (10% diethanolamine [pH 9.8], 0.5 mM MgCl2, 0.05% NaN3). Results were recorded as the difference between the optical density (OD) at 405 and 620 nm using an automated ELISA reader (Multiskan Labsystems, Finland). For each serum, the assay was done in triplicate and the values given represent the mean of triplicate determinations. For every GST fusion product the cutoff value was determined as the mean plus two times the standard deviation (SD) of the absorbency readings obtained from the Toxoplasma-negative sera.
(ii) IgM Rec-ELISA.
Maxisorb plates were coated with anti-human IgM antibodies (Sigma-Aldrich) at a concentration of 10 μg/ml in coating buffer. Plates were blocked as above and subsequently incubated for 1 h at 37°C with serum samples (diluted 1:50). Plates were washed and then incubated for 3 h at room temperature with the GST fusion proteins at a concentration of 50 μg/ml. After being washed, the plates were incubated for 1 h at room temperature with anti-GST horseradish peroxidase-conjugated antibody (Sigma-Aldrich), diluted 1:5,000 in blocking solution. Finally, the enzymatic activity was revealed using the substrate tetramethylbenzidine (Sigma-Aldrich). Results were recorded as the difference between the absorbance at 450 and 620 nm. For each sample the assay was done in triplicate, and for every GST protein the cutoff value was determined as the mean plus 2 SD of the absorbency readings obtained with sera from uninfected infants.
(iii) IgG subtype Rec-ELISA.
Maxisorb plates were coated with the recombinant proteins as described above and then blocked for 1 h at 37°C with 0.2% gelatin and 0.05% Tween 20 in PBS. Plates were incubated for 1 h at 37°C with sera (diluted 1:100). After being washed the plates were incubated for 1 h at 37°C with biotin-conjugated antibodies specific for human IgG1, IgG2, IgG3, and IgG4 (Sigma-Aldrich) diluted in blocking solution at 1:1,000 for IgG1 and IgG3 and at 1:5,000 for IgG2 and IgG4. Plates were washed and then incubated for 30 min at 37°C with avidin-alkaline phosphatase (Sigma Aldrich) diluted 1:10,000 in blocking solution. Finally, the enzymatic activity was revealed using the substrate pNPP as described above. For each serum sample, the assay was done in triplicate and average values were calculated. For each assay, the cutoff value was determined as the mean plus 2 SD of the absorbency readings obtained from the Toxoplasma-negative sera.
Statistical analysis.
Proportions were compared by chi-square test with P < 0.05 considered statistically significant. The receiver operating characteristic (ROC) curves were calculated using the Excel-based statistical package Analyse-it (http://www.analyse-it.com/, accessed 25 May 2005).
RESULTS
In a retrospective study, sera from 104 infants of mothers with primary Toxoplasma gondii infection during pregnancy were analyzed. Thirty-five infants had congenital toxoplasmosis and 69 were uninfected, as demonstrated by the persistence or disappearance, respectively, of Toxoplasma-specific IgG antibodies after 12 months of age. Table 1 shows the characteristics of the infected-patient cohort. The gestational age at the time of maternal infection was the second trimester in 15 mothers and the third trimester in 20 mothers. Detection of fetal T. gondii infection by PCR from amniotic fluid was performed for only four women (mothers of patients T6, T18, T19, and T25) because of the patient's refusal or primary infection acquired late in gestation, giving a positive result in only one case (patient T6). All but six of the mothers were treated, and 17 women received the regimen alternating P/S therapy with spiramycin after 16 weeks of gestation up to delivery. Infant sera were tested before starting postnatal treatment in all but five of the patients. According to the criteria of Hohlfeld et al. (21), disease onset was considered severe in five infants, benign in eight, and subclinical in 22.
TABLE 1.
Characteristics of the infected infants
| Patient no. | Maternal infectiona | Prenatal treatmentb | Onsetc | Clinical manifestationsd
|
Postnatal treatmente | ||
|---|---|---|---|---|---|---|---|
| RC | EC | HC | |||||
| T1 | II | Spir | B | − | + | − | − |
| T2 | II | Spir | Sub | − | − | − | − |
| T3 | III | Spir | Sub | − | − | − | − |
| T4 | III | Spir | Sub | − | − | − | − |
| T5 | III | P/S | B | + | − | − | − |
| T6 | II | P/S | B | − | + | − | − |
| T7 | II | P/S | Sub | − | − | − | − |
| T8 | III | P/S | B | − | + | − | − |
| T9 | III | P/S | Sub | − | − | − | − |
| T10 | III | P/S | B | + | + | + | + |
| T11 | III | Sub | − | − | − | − | |
| T12 | II | Spir | Sub | − | − | − | − |
| T13 | III | P/S | Sub | − | − | − | − |
| T14 | III | P/S | Sub | − | − | − | − |
| T15 | III | B | − | + | − | − | |
| T16 | III | Sub | − | − | − | − | |
| T17 | III | Spir | Sub | − | − | − | − |
| T18 | II | Spir | B | − | + | − | − |
| T19 | II | P/S | Sub | − | − | − | − |
| T20 | III | Sub | − | − | − | − | |
| T21 | III | P/S | S | + | + | − | − |
| T22 | III | P/S | Sub | − | − | − | − |
| T23 | II | Spir | Sub | − | − | − | − |
| T24 | II | P/S | B | − | + | − | − |
| T25 | II | P/S | S | + | + | + | + |
| T26 | II | Spir | S | + | + | + | + |
| T27 | III | Spir | Sub | − | − | − | − |
| T28 | II | Spir | S | + | + | − | + |
| T29 | III | Sub | − | − | − | − | |
| T30 | III | Spir | S | + | + | − | − |
| T31 | III | Sub | − | − | − | − | |
| T32 | III | P/S | Sub | − | − | − | − |
| T33 | III | P/S | Sub | − | − | − | − |
| T34 | III | P/S | Sub | − | − | − | + |
| T35 | III | P/S | Sub | − | − | − | − |
Trimester of gestation.
Spir, spiramycin; P/S, pyrimethamine-sulfadiazine in combination with spiramycin.
Severity of clinical onset: S, severe; B, benign; Sub, subclinical.
RC, retinochoroiditis; EC, endocranial calcifications; HC, hydrocephalus.
Treatment of the infected infant at the time of serum collection.
Seven antigenic regions of the T. gondii polypeptides MIC2a, MIC2b, MIC3, MIC4, M2AP, AMA1, and SAG1, which had been shown previously to contain B-cell epitopes (3, 5), were expressed as fusion products with the bacterial protein GST. Immunoreactivity of GST fusion proteins with IgG antibodies in sera from infected and uninfected infants was determined. Each of the antigens was coated separately onto individual microtiter wells and tested in the IgG Rec-ELISAs with sera from the 104 newborns. Thirty Toxoplasma-negative sera were also assayed as controls. Specific levels of anti-Toxoplasma IgG, determined by the whole-cell Toxoplasma immunoassay (VIDAS system from Biomerieux) (31), ranged from 26 to 2,549 IU/ml for sera from infected infants, from 9 to 427 IU/ml for sera from uninfected subjects, and from 0 to 6 IU/ml for the negative controls.
As shown in Table 2, with the exception of MIC4 and AMA1, all of the fusion proteins reacted with more than 75% of the sera from the congenitally infected infants. One of the 30 Toxoplasma-negative sera did react with the GST-AMA1 antigen and none recognized the GST-MIC2a, GST-MIC2b, GST-MIC3, GST-MIC4, GST-M2AP, and GST-SAG1 antigens. Interestingly, the number of reactive sera from infected versus uninfected infants, reflecting maternal antibodies, was significantly different (P < 0.0001). In particular, the recombinant GST-MIC3 reacted with IgG antibodies in 94% of sera from infected infants, while it failed to react with 71% of sera from uninfected subjects, indicating that sera from infected children recognized a more complex repertoire of antigens than sera from the mothers. Overall, IgG antibodies from 100% of the congenitally infected patients reacted with at least one recombinant antigen, while sera from 23 out of 69 uninfected subjects did not react with any recombinant product (data not shown).
TABLE 2.
Toxoplasma-specific IgG reactivity of 104 serum samples from 104 infants of mothers with primary T. gondii infection in pregnancy
| Groupa | No. of samples | Mean IgG levelb (IU/ml) | No. (%) [P]c of samples reactive in IgG Rec-ELISA against:
|
||||||
|---|---|---|---|---|---|---|---|---|---|
| MIC2a | MIC2b | MIC3 | MIC4 | M2AP | AMA1 | SAG1 | |||
| Infected | 35 | 338 | 34 (97) | 29 (83) | 33 (94) | 17 (49) | 31 (89) | 20 (57) | 24d (75) |
| Uninfected | 69 | 61 | 34 (49) [<0.0001] | 22 (32) [<0.0001] | 20 (29) [<0.0001] | 2 (3) [<0.0001] | 30 (43) [<0.0001] | 10 (15) [<0.0001] | 23 (33) [<0.0001] |
| Controls | 30 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | |
Serum samples collected from: A, newborns with congenital toxoplasmosis; B, uninfected infants; C, Toxoplasma-seronegative controls.
Toxoplasma-specific IgG levels, expressed as International Units (IU).
Chi-square test comparing uninfected and infected newborns.
Only 32 samples tested against SAG1.
Next, we examined the immunoreactivity of recombinant antigens with immunoglobulin M of infant sera. To this aim, a double-sandwich immunoassay was employed (IgM Rec-ELISA). The GST-MIC2a, GST-MIC2b, GST-MIC3, GST-M2AP, and GST-SAG1 fusion proteins were assayed with the 104 serum samples from infected and uninfected babies, and the results obtained with commercial assays employing either lysed whole-cell T. gondii antigen or fixed parasites (ELFA-IgM and ISAGA-IgM, respectively) were compared. As a control, the IgM reactivity against wild-type GST protein was assessed for each serum. The diagnostic criterion used to assign a positive IgM reactivity against single antigens was an ODGST-antigen greater than the cutoff and an ODGST-antigen greater than the ODGST.
As shown in Table 3, the number of IgM-reactive sera ranged from 66% to 31% using the GST-SAG1 and GST-MIC3 antigens, respectively. Among uninfected infants, IgM antibodies in 1.4% of newborns reacted with the GST-MIC2b and GST-M2AP antigens, those in 4.3% of infants reacted with GST-SAG1, and none recognized the GST-MIC2a and GST-MIC3 antigens, indicating the occurrence of few unspecific reactions. In Table 4 are summarized the results of the IgM Rec-ELISAs with individual sera from infected infants. Overall, IgM antibodies in 34 out of 35 sera from the congenitally infected patients reacted with at least one recombinant antigen. Notably, all sera from infected infants with silent toxoplasmosis (subclinical onset, see Table 1) were reactive using the IgM Rec-ELISAs. In contrast, only four of them (patients T3, T11, T29, and T32) had positive results when the whole-cell assays were employed.
TABLE 3.
Toxoplasma-specific IgM reactivity of 104 serum samples from 104 infants of mothers with primary T. gondii infection in pregnancya
| Group | No. | No. (%) of reactive sera [P]
|
||||||
|---|---|---|---|---|---|---|---|---|
| ELFA IgM | ISAGA IgM | IgM Rec-ELISA
|
||||||
| MIC2a | MIC2b | MIC3 | M2AP | SAG1 | ||||
| Infected | 35 | 9 (26) | 9 (26) | 21 (60) | 18 (51) | 11 (31) | 17 (49) | 23 (66) |
| Uninfected | 69 | 0 | 0 | 0 [<0.0001] | 1 (1.4) [<0.0001] | 0 [<0.0001] | 1 (1.4) [<0.0001] | 3 (4.3) [<0.0001] |
Values indicate the number of reactive sera using commercial assays (ELFA and ISAGA) or recombinant T. gondii antigens.
TABLE 4.
Immunoreactivity of recombinant antigens with IgM antibodies in serum samples from infants with congenital toxoplasmosisa
| Patient no. | Time after birth (wk) | IgG level (IU/ml) | ELISA IgA result | ISAGA IgM result | ELFA IgM cutoffb | Rec-ELISA IgM cutoffc
|
||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC2a | MIC2b | MIC3 | M2AP | SAG1 | ||||||
| T1 | 1 | 1,192 | Pos. | Pos. | 1.22 | 1.13 | 0.78 | 0.69 | 1.03 | 0.88 |
| T2 | 1 | 300 | Neg. | Neg. | 0.09 | 0.59 | 0.74 | 0.73 | 0.73 | 1.46 |
| T3 | 1 | 217 | Pos. | Pos. | 7.00 | 1.13 | 0.95 | 1.29 | 0.80 | 0.91 |
| T4 | 1 | 153 | Neg. | Neg. | 0.08 | 1.42 | 1.28 | 1.21 | 1.69 | 1.60 |
| T5 | 1 | 988 | Neg. | Neg. | 0.03 | 0.86 | 0.67 | 0.95 | 0.45 | 1.44 |
| T6 | 2 | 39 | Neg. | Neg. | 0.06 | 0.82 | 0.76 | 0.69 | 0.50 | 1.17 |
| T7 | 2 | 673 | Neg. | Neg. | 0.11 | 1.05 | 1.30 | 1.01 | 1.45 | 1.54 |
| T8 | 2 | 206 | Neg. | Neg. | 0.05 | 0.25 | 0.61 | 0.92 | 0.73 | 0.94 |
| T9 | 2 | 124 | Neg. | Neg. | 0.13 | 1.62 | 1.57 | 1.67 | 1.76 | 1.78 |
| T10 | 2 | 226 | Pos. | Pos. | 0.91 | 1.39 | 1.01 | 0.76 | 0.65 | 0.87 |
| T11 | 2 | 157 | Pos. | Pos. | 7.50 | 0.61 | 0.63 | 0.49 | 0.85 | 1.31 |
| T12 | 3 | 148 | Neg. | Neg. | 0.07 | 1.21 | 1.12 | 1.04 | 0.79 | 1.57 |
| T13 | 3 | 57 | Neg. | Neg. | 0.05 | 0.88 | 0.69 | 0.91 | 0.47 | 1.54 |
| T14 | 3 | 73 | Neg. | Neg. | 0.08 | 1.18 | 1.08 | 0.97 | 1.22 | 1.39 |
| T15 | 3 | 161 | Pos. | Pos. | 4.02 | 1.18 | 1.44 | 1.22 | 0.62 | 1.31 |
| T16 | 3 | 191 | Neg. | Neg. | 0.18 | 1.00 | 1.92 | 0.77 | 1.17 | 1.00 |
| T17 | 3 | 96 | Neg. | Neg. | 0.08 | 0.96 | 0.77 | 0.71 | 0.70 | 1.32 |
| T18 | 4 | 195 | Neg. | Neg. | 0.11 | 0.63 | 1.01 | 1.38 | 0.77 | 0.97 |
| T19 | 4 | 103 | Neg. | Neg. | 0.13 | 1.35 | 1.17 | 1.18 | 1.09 | 1.52 |
| T20 | 4 | 153 | Neg. | Neg. | 0.08 | 1.32 | 1.22 | 1.04 | 1.67 | 1.52 |
| T21 | 4 | 578 | Pos. | Pos. | 5.95 | 0.96 | 0.88 | 0.93 | 0.95 | 1.45 |
| T22 | 4 | 218 | Neg. | Neg. | 0.04 | 1.28 | 0.84 | 1.29 | 1.26 | 0.84 |
| T23 | 5 | 51 | Neg. | Neg. | 0.09 | 0.69 | 1.09 | 0.71 | 1.11 | 0.98 |
| T24 | 5 | 47 | Neg. | Neg. | 0.11 | 0.62 | 0.80 | 0.73 | 0.76 | 1.18 |
| T25 | 5 | 1,147 | Neg. | Neg. | 0.07 | 0.78 | 0.96 | 1.67 | 0.59 | 1.26 |
| T26 | 5 | 816 | Pos. | Neg. | 0.04 | 1.21 | 1.22 | 0.75 | 1.05 | 1.00 |
| T27 | 5 | 26 | Neg. | Neg. | 0.11 | 0.98 | 0.89 | 0.77 | 1.13 | 0.94 |
| T28 | 6 | 2,549 | Neg. | Pos. | 0.88 | 1.63 | 1.09 | 1.91 | 0.89 | 0.83 |
| T29 | 6 | 292 | Pos. | Pos. | 5.77 | 1.59 | 1.41 | 1.94 | 1.42 | 1.52 |
| T30 | 6 | 28 | Neg. | Neg. | 0.11 | 1.38 | 0.62 | 2.75 | 0.58 | 1.78 |
| T31 | 7 | 90 | Neg. | Neg. | 0.04 | 1.25 | 1.01 | 1.00 | 1.86 | 1.31 |
| T32 | 7 | 260 | Neg. | Pos. | 2.23 | 1.57 | 1.18 | 0.87 | 1.14 | 1.23 |
| T33 | 8 | 145 | Neg. | Neg. | 0.31 | 1.18 | 1.04 | 0.80 | 0.88 | 1.31 |
| T34 | 8 | 59 | Neg. | Neg. | 0.07 | 1.56 | 0.93 | 1.24 | 1.18 | 0.99 |
| T35 | 8 | 67 | Neg. | Neg. | 0.12 | 1.30 | 1.11 | 1.28 | 1.73 | 1.63 |
Serum samples (T1 to T35) were analyzed using IgM Rec-ELISAs with individual GST fusion products or commercial assays employing whole-cell Toxoplasma antigen (ELISA IgA, ELFA IgM, and ISAGA IgM).
Cutoff value for the commercial ELFA-IgM assay was 0.65, as indicated by the manufacturer (bioMerieux, France). Bold type, values > cutoff.
The numeric values were calculated as the ratio of the test OD to the cutoff of the corresponding antigen. Cutoff values for GST-MIC2a, GST-MIC2b, GST-MIC3, GST-M2AP, and GST-SAG1 were 0.40, 0.45, 0.35, 0.64, and 0.90, respectively. Bold type, ODGST-antigen > cutoff and ODGST-antigen > ODGST.
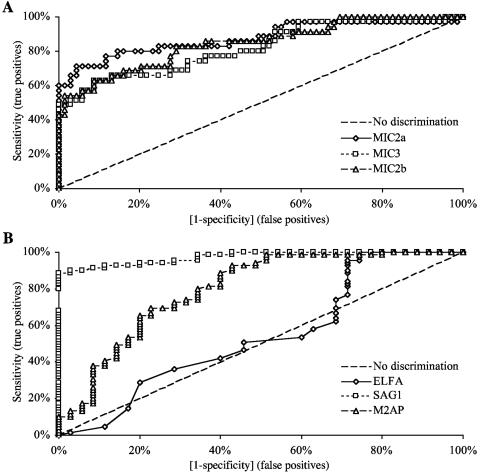
The clinical usefulness of recombinant antigens in the IgM-capture assay was investigated further by analyzing in detail the diagnostic performance of single recombinant antigen assays and combinations of assays. The ROC curves for the individual antigens clearly show that the IgM Rec-ELISA using the recombinant SAG1 antigen has the best performance, followed by the assays employing MIC2a, MIC2b, and MIC3 antigens, whereas the whole lysed Toxoplasma antigen (ELFA assay) displays the worst performance (Fig. 1). Table 5 shows the statistics for the single IgM Rec-ELISAs compared to the results obtained with the most relevant combinations of recombinant antigen immunoassays. The best outcome, in terms of sensitivity, agreement, and negative predictive value, was found when results from the MIC2a, MIC2b, and SAG1 IgM Rec-ELISAs were combined. With regard to specificity and positive predictive value, the best outcome was found when the results from the MIC2a and MIC3 assays were combined.
FIG. 1.
Statistical analysis of the IgM capture immunoassays. ROC curves for the IgM capture immunoassays using the five recombinant antigens MIC2a, MIC2b, and MIC3 (panel A) and M2AP, SAG1, and the whole lysed Toxoplasma antigen (ELFA) (panel B).
TABLE 5.
Diagnostic performance of the IgM-capture assays based on recombinant antigensa
| IgM Rec-ELISA antigen(s) | Sensitivity (%) | Specificity (%) | Agreement (%) | PPV (%) | NPV (%) |
|---|---|---|---|---|---|
| MIC2a | 60 | 100 | 86.5 | 100 | 83.1 |
| MIC2b | 51.4 | 98.6 | 82.7 | 94.7 | 80 |
| MIC3 | 31.4 | 100 | 76.9 | 100 | 74.2 |
| M2AP | 48.6 | 98.6 | 81.7 | 94.4 | 79.1 |
| SAG1 | 65.7 | 95.8 | 85.6 | 88.5 | 84.6 |
| MIC2a + SAG1 | 85.7 | 95.8 | 92.3 | 90.9 | 93 |
| MIC2a + MIC3 | 65.7 | 100 | 88.5 | 100 | 85.2 |
| MIC2a + MIC2b + SAG1 | 94.3 | 94.5 | 94.2 | 89.2 | 97 |
| MIC2a + MIC3 + SAG1 | 88.6 | 95.8 | 93.3 | 91.2 | 94.3 |
| MIC2a + MIC2b + MIC3 | 71.4 | 98.6 | 89.4 | 96.2 | 87.2 |
PPV, positive predictive value; NPV, negative predictive value.
Table 6 shows the performance characteristics of the commercial assays (ELFA-IgM, ELISA-IgA, and ISAGA-IgM), employed as individual tests or in combination, compared to the results obtained with the array of recombinant antigen IgM assays (five-antigen Rec-ELISAs). The best outcome, in terms of sensitivity, agreement, and negative predictive value, was found with results from the recombinant antigen IgM tests compared to the results obtained with commercial assays used alone or in any combination. With regard to specificity and positive predictive value, the best performance was found when the results from the commercial assays were combined.
TABLE 6.
Diagnostic performance of combinations of antibody-based immunoassaysa
| Diagnostic test(s) | Sensitivity (%) | Specificity (%) | Agreement (%) | PPV (%) | NPV (%) |
|---|---|---|---|---|---|
| ELFA-IgM | 25.7 | 100 | 75 | 100 | 72.6 |
| ISAGA-IgM | 25.7 | 100 | 75 | 100 | 72.6 |
| ELISA-IgA | 22.9 | 100 | 74 | 100 | 71.9 |
| Rec-ELISA-IgM (5 antigens) | 97.1 | 93.2 | 94.2 | 87.2 | 98.5 |
| ELFA-IgM + ISAGA-IgM | 25.7 | 100 | 75 | 100 | 72.6 |
| ELFA-IgM + ELISA-IgA | 28.6 | 100 | 76 | 100 | 73.4 |
| ISAGA-IgM + ELISA-IgA | 28.6 | 100 | 76 | 100 | 73.4 |
| ELFA-IgM + Rec-ELISA-IgM | 97.1 | 93.2 | 94.2 | 87.2 | 98.5 |
| ISAGA-IgM + Rec-ELISA-IgM | 97.1 | 93.2 | 94.2 | 87.2 | 98.5 |
| ELISA-IgA + Rec-ELISA-IgM | 97.1 | 93.2 | 94.2 | 87.2 | 98.5 |
PPV, positive predictive value; NPV, negative predictive value.
We finally investigated the presence in the infant sera of specific anti-Toxoplasma IgG subtypes, which were unequivocally synthesized by newborns with congenital infection. The immunoreactivity of the GST-MIC2a and GST-SAG1 fusion proteins against IgG1, IgG2, IgG3, and IgG4 subtypes was assessed with two consecutive samples collected from each subject (the interval between sample collection was 2 to 12 weeks). We analyzed 40 serum samples from 20 infected infants, and 20 Toxoplasma-negative sera were also assayed as a control.
Table 7 shows the results obtained with the IgG subtype analysis. Overall, 13 out of 20 infected infants displayed an increase of specific IgG subtypes reactivity against the recombinant antigens between the first and 13th weeks after birth. Among these subjects, 12 (92%), 7 (54%), and 5 (38%) infected infants displayed an increase in specific IgG2, IgG3, and IgG1 reactivity, respectively, and only one (8%) displayed an increase in IgG4 reactivity against the recombinant T. gondii antigens. None of the IgG subclasses in sera from the negative controls reacted with the GST-MIC2a and GST-SAG1 antigens (data not shown).
TABLE 7.
Immunoreactivity of recombinant antigens with IgG subtype antibodies in serum samples from infants with congenital T. gondii infectiona
| Patient no. | IgG level (IU/ml) | Time after birth (wk) | MIC2a Rec-ELISA cutoff
|
SAG1 Rec-ELISA cutoff
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| IgG1 | IgG2 | IgG3 | IgG4 | IgG1 | IgG2 | IgG3 | IgG4 | |||
| T1 | 905 | 7 | 23.5 | 1.0 | 2.5 | 1.0 | 7.7 | 1.2 | 0.5 | 0.7 |
| 220 | 13 | 10.4 | 1.0 | 1.3 | 0.8 | 4.9 | 1.5 | 0.5 | 0.7 | |
| T2 | 300 | 1 | 4.3 | 1.2 | 1.1 | 0.8 | 2.9 | 4.3 | 1.6 | 0.9 |
| 275 | 4 | 2.9 | 0.7 | 0.7 | 0.7 | 1.8 | 1.5 | 0.9 | 0.6 | |
| T4 | 153 | 1 | 3.6 | 2.3 | 1.5 | 1.0 | 1.1 | 0.7 | 0.4 | 0.5 |
| 87 | 5 | 2.1 | 2.6 | 1.4 | 1.0 | 1.3 | 2.4 | 1.2 | 0.8 | |
| T5 | 277 | 6 | 2.3 | 0.8 | 0.7 | 0.8 | 1.0 | 1.3 | 0.7 | 0.7 |
| 146 | 10 | 1.5 | 0.8 | 0.6 | 0.9 | 0.9 | 1.5 | 0.6 | 0.6 | |
| T7 | 240 | 7 | 2.6 | 2.0 | 0.8 | 0.9 | 1.4 | 0.7 | 0.8 | 0.6 |
| 190 | 11 | 2.6 | 1.5 | 1.2 | 0.8 | 2.0 | 1.3 | 1.0 | 0.5 | |
| T8 | 206 | 2 | 2.3 | 0.9 | 0.8 | 0.8 | 2.0 | 1.6 | 0.6 | 0.9 |
| 64 | 8 | 1.4 | 0.8 | 0.7 | 0.9 | 1.2 | 2.8 | 0.6 | 1.0 | |
| T9 | 124 | 2 | 4.0 | 2.5 | 1.3 | 1.0 | 2.7 | 2.6 | 1.8 | 1.5 |
| 73 | 7 | 2.8 | 4.1 | 1.7 | 1.2 | 1.4 | 4.4 | 1.3 | 1.0 | |
| T10 | 226 | 2 | 3.9 | 1.2 | 1.1 | 1.0 | 2.4 | 1.1 | 0.9 | 0.5 |
| 216 | 4 | 2.3 | 1.7 | 1.8 | 0.7 | 1.9 | 0.7 | 0.8 | 0.5 | |
| T11 | 157 | 2 | 0.9 | 0.8 | 1.2 | 0.7 | 0.9 | 1.1 | 1.0 | 0.8 |
| 140 | 7 | 3.0 | 0.9 | 0.9 | 0.7 | 1.2 | 1.3 | 0.7 | 0.6 | |
| T12 | 148 | 3 | 2.7 | 1.3 | 1.1 | 0.7 | 1.1 | 1.5 | 1.2 | 0.6 |
| 136 | 6 | 2.0 | 1.9 | 1.2 | 0.9 | 1.1 | 2.0 | 1.5 | 0.6 | |
| T13 | 57 | 3 | 0.9 | 0.9 | 1.2 | 0.8 | 0.8 | 1.3 | 1.1 | 0.6 |
| 39 | 8 | 0.8 | 1.2 | 0.8 | 0.8 | 0.7 | 1.2 | 0.6 | 0.6 | |
| T14 | 73 | 3 | 1.3 | 1.0 | 0.9 | 0.7 | 0.7 | 0.6 | 0.3 | 0.6 |
| 43 | 9 | 0.9 | 0.9 | 0.8 | 0.7 | 0.7 | 0.5 | 0.4 | 0.5 | |
| T20 | 153 | 4 | 2.6 | 0.8 | 0.8 | 0.5 | 1.1 | 0.7 | 0.6 | 0.5 |
| 132 | 8 | 1.9 | 0.9 | 0.8 | 0.6 | 0.9 | 0.7 | 0.5 | 0.6 | |
| T22 | 218 | 4 | 2.0 | 0.9 | 0.8 | 0.7 | 1.9 | 1.3 | 0.9 | 0.8 |
| 221 | 6 | 1.6 | 0.9 | 0.7 | 0.7 | 1.3 | 1.1 | 0.8 | 0.8 | |
| T24 | 47 | 5 | 0.9 | 1.1 | 0.6 | 0.8 | 1.3 | 2.0 | 0.5 | 0.7 |
| 30 | 9 | 0.9 | 0.9 | 0.6 | 0.8 | 0.8 | 2.3 | 0.5 | 0.6 | |
| T25 | 1,147 | 5 | 11.8 | 1.2 | 2.7 | 0.6 | 2.3 | 1.2 | 0.7 | 0.7 |
| 17 | 1.2 | 0.9 | 1.0 | 0.8 | 0.8 | 0.9 | 0.7 | 0.5 | ||
| T29 | 292 | 6 | 25.2 | 1.3 | 5.8 | 0.7 | 2.9 | 0.8 | 1.1 | 0.6 |
| 300 | 10 | 14.6 | 1.1 | 3.8 | 0.5 | 2.8 | 0.7 | 0.8 | 0.5 | |
| T30 | 39 | 3 | 2.2 | 0.9 | 1.3 | 0.7 | 0.8 | 1.0 | 0.4 | 0.5 |
| 28 | 6 | 9.7 | 1.0 | 8.3 | 0.7 | 0.8 | 0.9 | 0.3 | 0.5 | |
| T34 | 59 | 8 | 3.3 | 0.7 | 0.9 | 0.9 | 1.1 | 1.1 | 0.4 | 0.8 |
| 176 | 13 | 6.9 | 0.9 | 3.1 | 1.0 | 1.0 | 1.6 | 0.6 | 0.6 | |
| T35 | 91 | 4 | 1.3 | 0.9 | 0.8 | 0.5 | 1.3 | 1.7 | 1.0 | 0.4 |
| 67 | 8 | 0.9 | 0.8 | 0.4 | 0.4 | 1.0 | 0.9 | 0.5 | 0.5 | |
Serial serum samples from infants with congenital toxoplasmosis were analyzed using IgG subtype Rec-ELISAs with GST fusion products. The values were calculated as the ratio of the test OD to the cutoff of the corresponding antigen. Italic type, OD > cutoff; bold type, serum sample pairs showing increasing IgG subtype reactivity with time.
DISCUSSION
To provide proper clinical care for newborns, it is important to have a sensible and accurate method for diagnosis to distinguish, early after birth, congenitally infected infants from uninfected babies born to mothers who acquired primary Toxoplasma gondii infection during gestation.
Because Toxoplasma infection crosses the placenta in only ∼20% of maternal infections, screening out uninfected babies is an important goal to avoid unnecessary treatment. Also, identification of infected babies, whos infections are often subclinical at birth, is crucial to help avoid or minimize delayed sequelae, such as impaired mental development or ocular complications seen later in childhood or adolescence. As the wide spectrum of clinical manifestations in congenital toxoplasmosis overlaps those of other vertically transmittable diseases (e.g., cytomegalovirus, herpes simplex virus, rubella, and syphilis) (37) and the clinical expression of toxoplasmosis is absent in up to 85% of infected newborns (6, 15, 16, 22, 27), the diagnosis of congenital infection early after birth can only be made either indirectly by serological methods or directly by PCR from blood, other fluids, or tissue samples (for a review, see reference 29). Serological methods are generally preferred since the sensitivity of PCR results can be affected by the appropriateness of sample handling, shipping and storage conditions, and the technique used for PCR amplification and detection. Furthermore, parasitemia may be of short duration, further limiting the value of PCR of peripheral blood.
Serological diagnosis of congenital toxoplasmosis in the newborn is most commonly made by detecting Toxoplasma-specific IgM and/or IgA antibodies in infant serum. However, Toxoplasma-specific IgM and IgA antibodies are usually found in only up to 75% of infected babies (6, 7, 15, 16, 22, 27).
Most commercial assays for detecting specific anti-Toxoplasma immunoglobulins use the whole T. gondii soluble extract as the antigen. Consequently, the immunoassay will take into account the relative concentrations of the specific anti-Toxoplasma antibodies in serum and the corresponding antigens in the parasite extract. Our hypothesis was that using a combination of recombinant antigens individually challenged with antibodies from infected individuals might enhance the sensitivity of an antibody-based immunoassay. Moreover, a major advantage of using recombinant antigens is the higher reproducibility of assays, which is the first step toward a real standardization of toxoplasmosis serodiagnosis.
Several previous studies have found that recombinant antigens improve the serological diagnosis of T. gondii infections in adults with acquired infection (1, 4, 20, 23, 28, 33, 40). However, no studies so far have described the use of recombinant antigens for the diagnosis of congenital toxoplasmosis in newborns.
Immunodominant regions of T. gondii proteins have previously been identified (2-5, 9). The immunoreactivity of seven distinct fragments of T. gondii, the MIC2, MIC3, MIC4, M2AP, AMA1, and SAG1 proteins, expressed as GST fusion products, was assessed with sera from infants with congenital T. gondii infection. IgG antibodies from all infected infants reacted with at least one of the antigen fragments, emphasizing the immunogenicity of these proteins in humans. The recombinant MIC3 and MIC4 reacted prevalently with IgG antibodies from the congenitally infected infants versus uninfected babies, even though all of the newborn sera contained maternal IgG antibodies. This suggests that specific anti-MIC3 and anti-MIC4 IgG antibodies may indicate actively replicating T. gondii, whereas they disappear in subjects with latent T. gondii infection.
To discriminate between IgG antibodies transferred during gestation from the mother to her fetus from those newly synthesized by infants after birth, we analyzed the immunoreactivity of recombinant antigens against the distinct subtypes of immunoglobulins G in sera from newborns with congenital toxoplasmosis. In 13 of 20 infected newborns it was possible to highlight the presence of infant-specific IgG antibodies. The synthesis of Toxoplasma-specific IgG by infants appears to involve mainly the IgG2 and IgG3 subtypes, while the level of IgG1 subtype generally decreases soon after birth. This suggests that IgG1 is prevalently transferred from the mother during gestation, while synthesis of the IgG2 and IgG3 subtypes is mainly directed by infants in response to congenital T. gondii infection.
The recombinant antigens were assayed with IgM antibodies in sera from the patient cohorts by using a double-sandwich immunoassay, and the results of the IgM Rec-ELISAs were compared with those obtained with whole-cell Toxoplasma antigen. Overall, 97% of infants (34/35) with congenital toxoplasmosis were found to be positive using the IgM Rec-ELISA analysis, whereas only 29% of infected individuals (10/35) were detected by the whole-cell immunoassays (combination of ISAGA-IgM or ELFA-IgM plus ELISA-IgA; see Table 6).
The IgM assays with recombinant antigens generally displayed a good performance in terms of sensitivity and specificity. Only with the SAG1 antigen were a few false-positive results (3 out of 69 negative controls) observed, probably due to unspecific reactions of IgM antibodies from the sera of the uninfected infants. However, the false-positive results could be avoided by increasing the cutoff value of the SAG1 assay (e.g., using 3 SDs), although this resulted in missing two cases of infected infants (T6 and T24) (data not shown). Therefore, a lower cutoff value was employed despite reduced assay specificity.
It is noteworthy that 22 of the infected infants had a subclinical form of congenital toxoplasmosis, thus representing suspected cases of T. gondii infection, which were clinically undiagnosed at birth. Our results indicate that by employing an IgM immunoassay based on recombinant antigens, it was possible to correctly confirm the congenital infection in all of these subjects as soon as 8 weeks after birth, thus potentially allowing prompt treatment for patients with asymptomatic toxoplasmosis in early infancy.
In conclusion, this study demonstrates that the use of recombinant antigens is effective in distinguishing T. gondii-infected from uninfected infants and could provide the basis of valuable serologic tests for improving early diagnosis of newborns with congenital toxoplasmosis.
Acknowledgments
We are grateful to Franco Felici and Paola Del Porto for very helpful suggestions and advice during this work. We acknowledge Luca Bruno for skillful technical assistance and Annalisa DeVita and Concetta Papallo for secretarial assistance.
REFERENCES
- 1.Aubert, D., G. T. Maine, I. Villena, J. C. Hunt, L. Howard, M. Sheu, S. Brojanac, L. E. Chovan, S. F. Nowlan, and J. M. Pinon. 2000. Recombinant antigens to detect Toxoplasma gondii-specific immunoglobulin G and immunoglobulin M in human sera by enzyme immunoassay. J. Clin. Microbiol. 38:1144-1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beghetto, E., A. Pucci, O. Minenkova, A. Spadoni, L. Bruno, W. Buffolano, D. Soldati, F. Felici, and N. Gargano. 2001. Identification of a human immunodominant B-cell epitope within the GRA1 antigen of Toxoplasma gondii by phage display of cDNA libraries. Int. J. Parasitol. 31:1659-1668. [DOI] [PubMed] [Google Scholar]
- 3.Beghetto, E., A. Spadoni, W. Buffolano, M. Del Pezzo, O. Minenkova, E. Pavoni, A. Pucci, R. Cortese, F. Felici, and N. Gargano. 2003. Molecular dissection of the human B-cell response against Toxoplasma gondii infection by lambda display of cDNA libraries. Int. J. Parasitol. 33:163-173. [DOI] [PubMed] [Google Scholar]
- 4.Beghetto, E., W. Buffolano, A. Spadoni, M. Del Pezzo, M. Di Cristina, O. Minenkova, E. Petersen, F. Felici, and N. Gargano. 2003. Use of an immunoglobulin G avidity assay based on recombinant antigens for diagnosis of primary Toxoplasma gondii infection during pregnancy. J. Clin. Microbiol. 41:5414-5418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beghetto, E., H. V. Nielsen, P. Del Porto, W. Buffolano, S. Guglietta, F. Felici, E. Petersen, and N. Gargano. 2005. A combination of antigenic regions of Toxoplasma gondii microneme proteins induce protective immunity against oral infection with parasite cysts. J. Infect. Dis. 191:637-645. [DOI] [PubMed] [Google Scholar]
- 6.Buffolano, W., M. Lappalainen, L. Hedman, F. Ciccimarra, M. Del Pezzo, R. Rescaldani, N. Gargano, and K. Hedman. 2004. Delayed maturation of IgG avidity in congenital toxoplasmosis. Eur. J. Clin. Microbiol. Infect. Dis. 23:825-830. [DOI] [PubMed] [Google Scholar]
- 7.Candolfi, E., M. H. Bessieres, P. Mart, B. Cimon, F. Gandilhon, H. Pelloux, and P. Thulliez. 1993. Determination of a new cut-off value for the diagnosis of congenital toxoplasmosis by detection of specific IgM in an enzyme immunoassay. Eur. J. Clin. Microbiol. Infect. Dis. 12:396-398. [DOI] [PubMed] [Google Scholar]
- 8.Chumpitazi, B. F., A. Boussaid, H. Pelloux, C. Racinet, M. Bost, and A. Goullier-Fleuret. 1995. Diagnosis of congenital toxoplasmosis by immunoblotting and relationship with other methods. J. Clin. Microbiol. 33:1479-1485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Di Cristina, M., P. Del Porto, W. Buffolano, E. Beghetto, A. Spadoni, S. Guglietta, E. Piccolella, F. Felici, and N. Gargano. 2004. The Toxoplasma gondii bradyzoite antigens BAG1 and MAG1 induce early humoral and cell- mediated immune responses upon human infection. Microb. Infect. 6:164-171. [DOI] [PubMed] [Google Scholar]
- 10.Dunn, D., M. Wallon, F. Peyron, E. Petersen, C. S. Peckham, and R. Gilbert. 1999. Mother to child transmission of toxoplasmosis: risk estimates for clinical counseling. Lancet 353:1829-1833. [DOI] [PubMed] [Google Scholar]
- 11.Foulon, W., I. Villena, B. Stray-Pedersen, A. Decoster, M. Lappalainen, J. M. Pinon, P. A. Jenum, K. Hedman, and A. Naessens. 1999. Treatment of toxoplasmosis during pregnancy: a multicenter study of impact on fetal transmission and children's sequelae at age 1 year. Am. J. Obstet. Gynaecol. 180:410-415. [DOI] [PubMed] [Google Scholar]
- 12.Fricker-Hidalgo, H., H. Pelloux, C. Racinet, M. Bost, A. Goullier-Fleuret, and P. Ambroise-Thomas. 1996. Congenital toxoplasmosis: specific IgM in fetal blood, cord blood and in the newborn. Ann. Biol. Clin. 54:165-168. [PubMed] [Google Scholar]
- 13.Fricker-Hidalgo H., H. Pelloux, C. Racinet, I. Grefenstette, C. Bost-Bru, A. Goullier-Fleuret, and P. Ambroise-Thomas. 1998. Detection of Toxoplasma gondii in 94 placentae from infected women by polymerase chain reaction, in vivo, and in vitro cultures. Placenta 19:545-549. [DOI] [PubMed] [Google Scholar]
- 14.Gay-Andrieu F., P. Marty, J. Pialat, G. Sournies, T. Drier de Laforte, and F. Peyron. 2003. Fetal toxoplasmosis and negative amniocentesis: necessity of an ultrasound follow-up. Prenat. Diagn. 23:558-560. [DOI] [PubMed] [Google Scholar]
- 15.Gilbert, R., D. Dunn, M. Wallon, M. Hayde, A. Prusa, M. Lebech, T. Kortbeek, F. Peyron, A. Pollak, and E. Petersen. 2001. Ecological comparison of the risks of mother-to-child transmission and clinical manifestations of congenital toxoplasmosis according to prenatal treatment protocol. Epidemiol. Infect. 127:113-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gilbert, R., L. Grass, and the European Multicentre Study on Congenital Toxoplasmosis. 2003. Effect of timing and type of treatment on the risk of mother to child transmission of Toxoplasma gondii. Br. J. Obstet. Gynaecol. 110:112-120. [DOI] [PubMed] [Google Scholar]
- 17.Gross, U., C. G. Luder, V. Hendgen, C. Heeg, I. Sauer, A. Weidner, D. Krczal, and G. Enders. 2000. Comparative immunoglobulin G antibody profiles between mother and child (CGMC test) for early diagnosis of congenital toxoplasmosis. J. Clin. Microbiol. 38:3619-3622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grover, C. M., P. Thulliez, J. S. Remington, and J. D. Boothroyd. 1990. Rapid prenatal diagnosis of congenital Toxoplasma infection by using polymerase chain reaction and amniotic fluid. J. Clin. Microbiol. 28:2297-2301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guerina, N. G., H. W. Hsu, H. C. Meissner, J. H. Maguire, R. Lynfield, B. Stechenberg, I. Abroms, M. S. Pasternack, R. Hoff, R. B. Eaton, et al. 1994. Neonatal serologic screening and early treatment for congenital Toxoplasma gondii infection. The New England Regional Toxoplasma Working Group. N. Engl. J. Med. 330:1858-1863. [DOI] [PubMed] [Google Scholar]
- 20.Harning, D., J. Spenter, A. Metsis, J. Vuust, and E. Petersen. 1996. Recombinant Toxoplasma gondii surface antigen 1 (P30) expressed in Escherichia coli is recognized by human Toxoplasma-specific immunoglobulin M (IgM) and IgG antibodies. Clin. Diagn. Lab. Immunol. 3:355-357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hohlfeld, P., F. Daffos, P. H. Thulliez, C. Aufrant, J. Couvreur, J. MacAleese, D. Descombey, and F. Forestier. 1989. Fetal toxoplasmosis: outcome of pregnancy and infant follow-up after in utero treatment. J. Pediatr. 115:765-769. [DOI] [PubMed] [Google Scholar]
- 22.Hohlfeld, P., F. Daffos, J. M. Costa, P. H. Thuillez, F. Forestier, and M. Vidaud. 1994. Prenatal diagnosis of congenital toxoplasmosis with a polymerase-chain reaction test on amniotic fluid. New Engl. J. Med. 331:695-699. [DOI] [PubMed] [Google Scholar]
- 23.Jacobs, D., M. Vercammen, and E. Saman. 1999. Evaluation of recombinant dense granule antigen 7 (GRA7) of Toxoplasma gondii for detection of immunoglobulin G antibodies and analysis of a major antigenic domain. Clin. Diagn. Lab. Immunol. 6:24-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jenum, P. A., M. Holberg-Petersen, K. K. Melby, and B. Stray-Pedersen. 1998. Diagnosis of congenital Toxoplasma gondii infection by polymerase chain reaction (PCR) on amniotic fluid samples. The Norwegian experience. APMIS 106:680-686. [PubMed] [Google Scholar]
- 25.Koppe, J. G., D. H. Loewer-Sieger, and H. De Roever-Bonnet. 1986. Results of 20 year follow-up of congenital toxoplasmosis. Lancet 101:254-257. [DOI] [PubMed] [Google Scholar]
- 26.Lappalainen, M., M. Koskiniemi, V. Hilesmaa, P. Ammala, K. Teramo, P. Koskela, M. Lebech, K. O. Raivio, and K. Hedman. 1995. Outcome of children after maternal primary infection during pregnancy with emphasis on avidity of specific IgG. Pediatr. Infect. Dis. J. 14:354-361. [DOI] [PubMed] [Google Scholar]
- 27.Lebech, M., O. Andersen, N. C. Christensen, J. Hertel, H. E. Nielsen, B. Peitersen, C. Rechnitzer, S. O. Larsen, B. Norgaard-Pedersen, and E. Petersen. 1999. Feasibility of neonatal screening for Toxoplasma infection in the absence of prenatal treatment. Lancet 353:1834-7. [DOI] [PubMed] [Google Scholar]
- 28.Li, S., G. Galvan, F. G. Araujo, Y. Suzuki, J. S. Remington, and S. Parmley. 2000. Serodiagnosis of recently acquired Toxoplasma gondii infection using an enzyme-linked immunosorbent assay with a combination of recombinant antigens. Clin. Diagn. Lab. Immunol. 7:781-787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Montoya, J. G., and O. Liesenfeld. 2004. Toxoplasmosis. Lancet 363:1975-1976. [DOI] [PubMed] [Google Scholar]
- 30.Nielsen, H. V., D. R. Schmidt, and E. Petersen. 2005. Diagnosis of congenital toxoplasmosis by two-dimensional immunoblot differentiation of mother and child immunoglobulin g profiles. J. Clin. Microbiol. 43:711-715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pelloux, H., P. Ciapa, A. Goullier-Fleuret, and P. Ambroise-Thomas. 1993. Evaluation du systeme VIDAS pour le diagnostic serologique de la toxoplasmose. Ann. Biol. Clin. 50:875-878. [PubMed] [Google Scholar]
- 32.Petersen, E., A. Pollak, and I. Reiter-Owona. 2001. Recent trends in research on congenital toxoplasmosis. Int. J. Parasitol. 31:115-144. [DOI] [PubMed] [Google Scholar]
- 33.Pietkiewicz, H., E. Hiszyska-Sawicka, J. Kur, E. Petersen, H. V. Nielsen, M. Stankiewicz, I. Andrzejewska, and P. Myjak. 2004. Usefulness of Toxoplasma gondii recombinant antigens in serodiagnosis of human toxoplasmosis. J. Clin. Microbiol. 42:1779-1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pinon, J. M., C. Chemla, I. Villena, F. Foudrinier, D. Aubert, D. Puygauthier-Toubas, B. Leroux, D. Dupouy, C. Quereux, M. Talmud, T. Trenque, G. Potron, M. Pluot, G. Remy, and A. Bonhomme. 1996. Early neonatal diagnosis of congenital toxoplasmosis: value of comparative enzyme- linked immunofiltration assay immunological profiles and anti-Toxoplasma gondii immunoglobulin M (IgM) or IgA immunocapture and implications for postnatal therapeutic strategies. J. Clin. Microbiol. 34:579-583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pinon, J.M., H. Dumon, C. Chemla, J. Franck, E. Petersen, M. Lebech, J. Zufferey, M.H. Bessieres, P. Marty, R. Holliman, J. Johnson, V. Luyasu, B. Lecolier, E. Guy, D.H. Joynson, A. Decoster, G. Enders, H. Pelloux, and E. Candolfi. 2001. Strategy for diagnosis of congenital toxoplasmosis: evaluation of methods comparing mothers and newborns and standard methods for postnatal detection of immunoglobulin G, M, and A antibodies. J. Clin. Microbiol. 39:2267-2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Remington, J.S., F.G. Araujo, and G. Desmonts. 1985. Recognition of different Toxoplasma antigens by IgM and IgG antibodies in mothers and their congenitally infected newborns. J. Infect. Dis. 152:1020-1024. [DOI] [PubMed] [Google Scholar]
- 37.Remington, J. S., R. McLeod, P. Thuillez, and G. Desmonts. 2001. Toxoplasmosis, p. 205-346. In J. S. Remington and J. Klein (ed.), Infectious diseases of the foetus and newborn infant, 5th ed. W.B. Saunders, Philadelphia, Pa.
- 38.Robert-Gangneux, F., M.F. Gavinet, T. Ancelle, J. Raymond, C. Tourte- Schaefer, and J. Dupouy-Camet. 1999. Value of prenatal diagnosis and early postnatal diagnosis of congenital toxoplasmosis: retrospective study of 110 cases. J. Clin. Microbiol. 37:2893-2898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Romand S., M. Wallon, J. Franck, P. Thulliez, F. Peyron, and H. Dumon. 2001. Prenatal diagnosis using polymerase chain reaction on amniotic fluid for congenital toxoplasmosis. Obstet. Gynecol. 97:296-300. [DOI] [PubMed] [Google Scholar]
- 40.Suzuki, Y., R. Ramirez, C. Press, S. Li, S. Parmley, P. Thulliez, and J.S. Remington. 2000. Detection of immunoglobulin M antibodies to P35 antigen of Toxoplasma gondii for serodiagnosis of recently acquired infection in pregnant women. J. Clin. Microbiol. 38:3967-3970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Thalib L., L. Gras, S. Romand, A. Prusa, M.H. Bessieres, E. Petersen, and R. E. Gilbert. 2005. Prediction of congenital toxoplasmosis by polymerase chain reaction analysis of amniotic fluid. Br. J. Obstet. Gynaecol. 112:567-574. [DOI] [PubMed] [Google Scholar]
- 42.Villena, I., D. Aubert, B. Leroux, D. Dupouy, M. Talmud, C. Chemla, T. Trenque, G. Schmit, C. Quereux, M. Guenounon, M. Pluot, A. Bonhomme, and J. M. Pinon. 1998. Pyrimethamine-sulfadoxine treatment of congenital toxoplasmosis: follow-up of 78 cases between 1980 and 1997. Scand. J. Infect. Dis. 30:295-300. [DOI] [PubMed] [Google Scholar]
- 43.Wallon, M., L. Kodjikian, C. Binquet, J. Garweg, J. Fleury, C. Quantin, and F. Peyron. 2004. Long-term ocular prognosis in 327 children with congenital toxoplasmosis. Pediatrics 113:1567-1572. [DOI] [PubMed] [Google Scholar]