Abstract
We investigated the mechanism of imipenem resistance in Enterobacter aerogenes strain 810, a clinical isolate from the United States for which the imipenem MIC was 16 μg/ml and the meropenem MIC was 8 μg/ml. An imipenem-susceptible revertant, strain 810-REV, was obtained after multiple passages of the strain on nonselective media. For the revertant, the imipenem MIC was ≤1 μg/ml and the meropenem MIC was ≤0.25 μg/ml. Cefepime MICs also decreased from 8 to 1 μg/ml; however, the MICs of ceftazidime (≥128 μg/ml), cefoxitin (≥32 μg/ml), and cefotaxime (≥64 μg/ml) remained the same. The β-lactamase and porin profiles of the parent, the revertant, and carbapenem-susceptible type strain E. aerogenes ATCC 13048 were determined. Strains 810 and 810-REV each produced two β-lactamases with pIs of 8.2 and 5.4. The β-lactamase activities of the parent and revertant were similar, even after induction with subinhibitory concentrations of imipenem. While 810-REV produced two major outer membrane proteins of 42 and 39 kDa that corresponded to Escherichia coli porins OmpC and OmpF, respectively, the parent strain appeared to produce similar quantities of the 39-kDa protein (OmpF) but decreased amounts of the 42-kDa protein (OmpC). When the parent strain was grown in the presence of imipenem, the 42-kDa protein was not detectable by gel electrophoresis. However, Western blot analysis of the outer membrane proteins of the parent and revertant with polyclonal antisera raised to the OmpC and OmpF analogs of Klebsiella pneumoniae (anti-OmpK36 and anti-OmpK35, respectively) showed that strain 810 expressed only the 42-kDa OmpC analog in the absence of imipenem (the 39-kDa protein was not recognized by the anti-OmpF antisera) and neither the OmpC nor the OmpF analog in the presence of imipenem. The OmpC analog is apparently down-regulated in the presence of imipenem; however, 810-REV expressed both OmpC and OmpF analogs. These data suggest that imipenem resistance in E. aerogenes 810 is primarily associated with the lack of expression of the analogs of the OmpC (42-kDa) and OmpF (39-kDa) outer membrane proteins, which also results in decreased susceptibility to meropenem and cefepime.
Enterobacter aerogenes is a major cause of health care-associated infections throughout the world (9, 15, 17, 27, 29). E. aerogenes strains isolated from hospitalized patients generally exhibit resistance to a variety of broad-spectrum antimicrobial agents, including β-lactams (1, 2, 25, 27). Resistance to β-lactams is usually a result of β-lactamase production or alteration of porins (5, 6, 8), but it may be due to modification of the target site (the penicillin binding proteins) or drug efflux (19). Carbapenems often have been used to treat infections caused by multidrug-resistant E. aerogenes strains; however, resistance to carbapenems is starting to emerge (11, 13). The mechanism of carbapenem resistance is often a combination of specific carbapenemase production and alteration of porins (3, 31). Carbapenem resistance due to lipopolysaccharide alterations has also been reported in E. aerogenes (18). Recently, it has been shown that the mar operon of E. aerogenes is involved in the multidrug resistance phenotype (7).
The goal of this study was to characterize the mechanism of carbapenem resistance in E. aerogenes strain 810. In addition, we sought to explain why the outer membrane protein (OMP) profiles differed when the strain was grown in the presence and absence of imipenem. Our results suggest that analysis of OMPs exclusively by gel electrophoresis can provide misleading results and that Western blotting with anti-OmpF and anti-OmpC antisera is critical to understanding the role of OmpF and OmpC analogs in carbapenem resistance.
MATERIALS AND METHODS
Bacterial strains.
Carbapenem-resistant E. aerogenes strain 810 was obtained from a laboratory in Michigan participating in Project ICARE (Intensive Care Antimicrobial Resistance Epidemiology) (14). E. aerogenes ATCC 13048 (type strain), a carbapenem-susceptible isolate, was obtained from the American Type Culture Collection (Manassas, Va.) and was used as a control for porin profiles.
Antimicrobial susceptibility testing.
Organisms were tested by broth microdilution with Mueller-Hinton broth (BD Biosciences, Sparks, Md.) as described in NCCLS document M7-A5 (23) and by disk diffusion with Mueller-Hinton agar (Difco Laboratories, Detroit, Mich.) as described in NCCLS document M2-A7 (22). Escherichia coli ATCC 25922, Enterococcus faecalis ATCC 29212, Klebsiella pneumoniae ATCC 700603, and Pseudomonas aeruginosa ATCC 27853 were used for quality control.
IEF of β-lactamases.
Crude cell lysates were prepared by a previously described freeze-thaw procedure (30). Isoelectric focusing (IEF) was performed as described by Matthew and Harris (20). Cell extracts were loaded onto commercially prepared PAG plates (pH 3.5 to 9.5; Pharmacia LKB, Piscataway, N.J.) and electrophoresed to equilibrium with an LKB Multifor II apparatus (Pharmacia LKB). β-Lactamases were visualized by staining the IEF gel with a 0.05% (0.96 mM) solution of nitrocefin (BD Biosciences). The isoelectric points were calculated by comparison to those of TEM-12 (pI 5.25), TEM-10 (pI 5.6), SHV-3 (pI 7.0), SHV-18 (pI 7.8), SHV-5 (pI 8.2), and MIR-1 (pI 8.6). Parallel gels were overlaid with a solution containing 100 μg of cloxacillin (Sigma-Aldrich) per ml prior to staining with nitrocefin to block the actions of AmpC β-lactamases (28). Inhibition assays were also performed in microtiter plates by adding cloxacillin to cell lysates prior to the addition of nitrocefin.
Examination of porin genes and porin expression.
OMPs were isolated as described by Rasheed et al. (26), except that 2% Triton X-100 was used for extraction of OMPs. Protein concentrations were determined with the bicinchoninic acid (BCA) protein assay kit (Pierce, Rockford, Ill.) as described by the manufacturer. The OMPs were examined with several gel electrophoresis systems in an attempt to optimize differentiation of the OmpC and OmpF analogs. The systems included two types of Tris-glycine-sodium dodecyl sulfate (SDS) linear gradient gels (Invitrogen, Carlsbad, Calif.), 4 to 16% and 4 to 12%, as described by the manufacturer, and 10% and 4 to 12% NuPAGE gels with morpholinepropanesulfonic acid buffer, as described by the manufacturer (Invitrogen). The gels were visualized by staining with Coomassie blue R250.
Western blotting of porins was performed as follows: proteins from 4 to 12% Tris-glycine-SDS gels or 10% NuPAGE gels were transferred to Immobilon-P filters (Millipore) (12, 16). The filters were blocked with 1% bovine serum albumin in phosphate-buffered saline (PBS). After they were washed, the filters were incubated with a 1:100 dilution of polyclonal anti-OmpK36 (analog of OmpC) or polyclonal anti-OmpK35 (analog of OmpF) antibody (12, 16) (the antibodies were the generous gift of S. Alberti, Madrid, Spain) and then with a 1:5,000 dilution of alkaline phosphatase-labeled goat anti-rabbit immunoglobulin G (Sigma). The filters were developed as described by the manufacturer. All incubations were carried out at room temperature for 1 h in 1% bovine serum albumin-0.05% Tween 20-PBS, and after incubation with the antiserum, the filters were washed with 0.05% Tween 20-PBS.
β-Lactamase activity determinations and β-lactamase inactivation assays.
β-Lactamase activity was determined with extracts obtained from cultures of the parent and revertant strains that were grown in the absence and presence of a subinhibitory concentration of imipenem (i.e., one-fourth the MIC of imipenem for the parent and the revertant) and lysed by a freeze-thaw procedure (4, 30). Hydrolysis of 50 μM nitrocefin was measured spectrophotometrically (SpectraMax Plus microplate spectrophotometer; Molecular Devices, Sunnyvale, Calif.) at 25°C in 0.1 M phosphate buffer (pH 7.0) at 482 nm for 10 min. The total protein concentration was determined by using the BCA protein assay kit (Pierce), and activity was reported in nanomoles of nitrocefin hydrolyzed per minute per milligram of protein (32).
In order to determine whether resistance to imipenem, ceftazidime, cefepime, cefotaxime, ceftriaxone, and piperacillin was due to production of a β-lactamase, a disk diffusion bioassay was performed. A suspension of E. coli DH5α equivalent to a 1.0 McFarland standard was inoculated onto a Mueller-Hinton agar plate as for disk diffusion (22). Then, antimicrobial agent disks were applied and were evenly spaced on the plate. Ten microliters of crude IEF lysate was used to make a 15-mm streak on each side of one imipenem, ceftazidime, cefepime, cefotaxime, ceftriaxone, and piperacillin disk (the lysate for the piperacillin disk was inoculated at a right angle) on the periphery of the plate. The plates were incubated at 37°C for 18 to 20 h. Alterations in the shapes of the zones of inhibition around the disks indicated β-lactamase or carbapenemase activity. Negative controls for carbapenemase production included E. aerogenes ATCC 13048.
DNA sequence analysis.
Amplification and sequence analysis of blaTEM-1 DNA were performed as previously described by Rasheed et al. (26).
RESULTS
Antimicrobial susceptibility testing.
The MICs of a variety of antimicrobial agents for E. aerogenes strain 810, the strain 810 revertant (strain 810-REV), and E. aerogenes ATCC 13048 are shown in Table 1. By using the interpretive criteria of NCCLS (23), strain 810 was resistant to imipenem and intermediate to meropenem; the MICs were 16 and 8 μg/ml, respectively. The isolate was also resistant to cefpodoxime, ceftazidime, cefotaxime, and ceftriaxone and showed borderline susceptibility to cefepime and aztreonam (MICs, 8 μg/ml for both drugs). The MICs of ceftazidime, cefotaxime, ceftriaxone, cefpodoxime, and cefepime did not decrease when they were tested in combination with clavulanic acid, indicating that 810 probably did not contain an extended-spectrum β-lactamase. The MICs of cefepime (0.5 μg/ml), meropenem (≤0.25 μg/ml), and imipenem (1 μg/ml) were lower for the revertant 810-REV (the revertant was isolated after five passages on nonselective medium) and approximated those for carbapenem-susceptible E. aerogenes type strain ATCC 13048.
TABLE 1.
Antimicrobial susceptibility profiles of E. aerogenes strains 810, 810-REV, and ATCC 13048
| Antimicrobial agent | MIC (μg/ml)
|
||
|---|---|---|---|
| 810 | 810-REV | ATCC 13048 | |
| Imipenem | 16 | 1 | 2 |
| Meropenem | 8 | ≤0.25 | NDb |
| Cefpodoxime-clavulanatea | >64 | >64 | 2 |
| Cefpodoxime | >64 | >64 | 2 |
| Ampicillin | >64 | >64 | ND |
| Amoxicillin-clavulanatec | >32 | >32 | ND |
| Piperacillin | >128 | >128 | 4 |
| Piperacillin-tazobactamd | >128 | >128 | 4 |
| Ticarcillin-clavulanatea | >64 | >64 | 2 |
| Ceftazidime | >128 | >128 | 0.5 |
| Ceftazidime-clavulanatea | >128 | >128 | 0.5 |
| Cefotaxime | 64 | 32 | 0.125 |
| Cefotaxime-clavulanatea | 64 | 32 | 0.125 |
| Ceftriaxone | 64 | 32 | 0.125 |
| Ceftriaxone-clavulanatea | 64 | 32 | 0.125 |
| Cefepime | 8 | 1 | <0.03 |
| Cefepime-clavulanatea | 4 | 1 | <0.03 |
| Cefoxitin | >32 | >32 | ND |
| Cefazolin | >32 | >32 | ND |
| Aztreonam | 8 | 4 | ND |
| Amikacin | 2 | 1 | 2 |
| Ciprofloxcacin | 0.5 | 0.5 | 0.06 |
Clavulanate was tested at a fixed concentration of 4 μg/ml.
ND, not determined.
Amoxicillin-clavulanate was tested at a fixed ratio of 2:1.
Tazobactam was tested at a fixed concentration of 4 μg/ml.
Carbapenem resistance is not associated with production of β-lactamase.
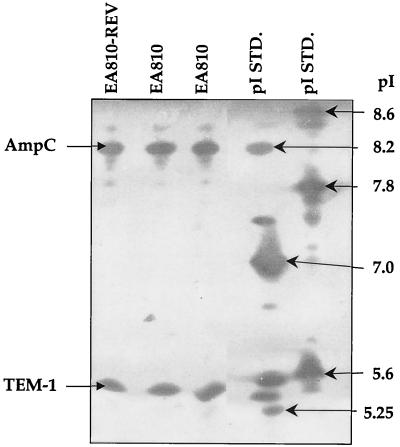
IEF of E. aerogenes parent strain 810 revealed two β-lactamases with pIs of 8.2 and 5.4 (Fig. 1). Imipenem-susceptible revertant 810-REV also produced two β-lactamases with pIs of 8.2 and 5.4 (Fig. 1). Overlaying of the IEF gel with 100 μg of cloxacillin per ml blocked the hydrolysis of nitrocefin by the β-lactamase of pI 8.2 but not the activity of the β-lactamase of pI 5.4, indicating that the pI 8.2 enzyme is likely an AmpC enzyme (data not shown). Amplification of DNA from strain 810 with blaTEM-specific PCR primers, followed by DNA sequence analysis, confirmed that the β-lactamase with the pI of 5.4 was TEM-1.
FIG. 1.
IEF patterns of E. aerogenes 810 and imipenem-susceptible revertant E. aerogenes 810-REV. The gel was stained with nitrocefin. The pIs of the β-lactamases were calculated by using the known pIs of TEM-12 (pI 5.25), TEM-10 (pI 5.6), SHV-3 (pI 7.0), SHV-18 (pI 7.8), SHV-5 (pI 8.2), and MIR-1 (pI 8.6). STD., standard.
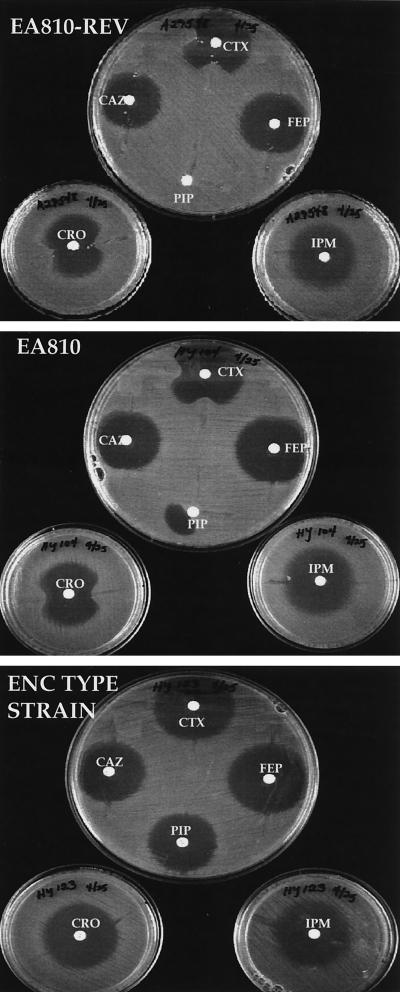
In order to determine whether carbapenem resistance could be attributed to production of one of these β-lactamases, a disk diffusion inactivation assay was performed (Fig. 2). Alterations of the zones of inhibition around cefotaxime, ceftriaxone, and piperacillin disks were observed for strains 810 and 810-REV; however, the zones of inhibition around the imipenem, cefepime, and ceftazidime disks were not affected. This indicates that β-lactamase production was not responsible for imipenem resistance in 810. It is unclear why ceftazidime was not more clearly inactivated.
FIG. 2.
Assay for antimicrobial agent inactivation in which cell extracts containing potential β-lactamases were streaked outward from disks containing various antimicrobial agents. E. aerogenes 810 and 810-REV, an imipenem-susceptible revertant, produced β-lactamases capable of hydrolyzing piperacillin (PIP), cefotaxime (CTX), and ceftriaxone (CRO) but not imipenem (IPM), ceftazidime (CAZ), or cefepime (FEP). The lysate was applied at a right angle for the piperacillin disk.
β-Lactamase activity was measured in extracts of the parent and revertant strains grown in the presence and absence of imipenem. Activity in the parent strain varied from an average of 155 nmol of nitrocefin hydrolyzed/min/mg of protein in uninduced extracts to an average of 284 nmol of nitrocefin hydrolyzed/min/mg of protein in induced extracts, a 1.8-fold increase. After induction there was a corresponding increase in the imipenem MIC for the parent strain from 8 to 16 μg/ml. The β-lactamase activity of revertant strain 810-REV ranged from an average of 236 nmol of nitrocefin hydrolyzed/min/mg of protein in the uninduced extracts to 365 nmol of nitrocefin hydrolyzed/min/mg of protein in the induced extracts (a 1.6-fold increase); no change in the imipenem MICs for the strain was observed. These data suggest that the contribution of the AmpC β-lactamase to the carbapenem resistance phenotype was minimal.
Analysis of OMPs.
Since the MICs of imipenem, meropenem, and cefepime were much lower for the imipenem-susceptible revertant than for parent strain 810 and resistance did not appear to be β-lactamase mediated, we examined strains 810 and 810-REV for alterations in OMP expression and compared their OMP expression to the OMP expression of E. aerogenes type strain ATCC 13048 (Fig. 3 and 4).
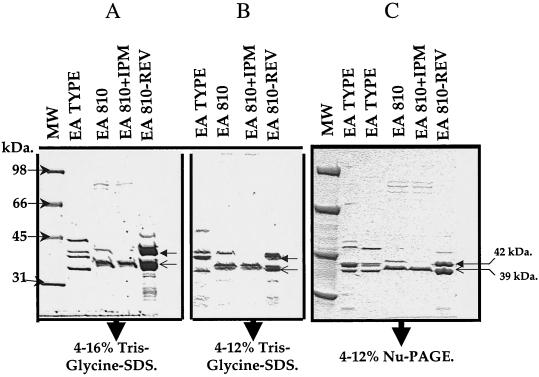
FIG. 3.
Resolution of OMPs from E. aerogenes type strain ATCC 13048, E. aerogenes 810, and E. aerogenes 810-REV on 4 to 16% (A) and 4 to 12% (B) Tris-glycine-SDS gels and 4 to 12% NuPAGE gels (C). Strain 810 was grown in the presence and absence of imipenem. The closed arrowheads indicate the 42-kDa porin (presumed OmpC analog), and the open arrowheads indicate the 39-kDa porin (presumed OmpF analog).
FIG. 4.
SDS-polyacrylamide gel electrophoresis and Western blot analysis of OMPs from E. aerogenes 810, carbapenem-susceptible revertant 810-REV, and E. aerogenes ATCC 13048 (control strain). (A) Analysis of OMPs on a 4 to 12% Tris-glycine-SDS gel; (B) Western blot analysis of OMPs with polyclonal anti-OmpK35 antiserum (the OmpK35 antibody cross-reacts with OmpK36 [16]); (C) Western blot analysis of OMPs with polyclonal anti-OmpK36 antiserum. The arrows with the white heads indicate the OmpC (OmpK36) analogs; the arrows with black heads indicate the OmpF (OmpK35) analogs.
The OMPs were analyzed on three different commercially available gel systems to determine which gel system produced the optimal resolution of the protein patterns. The results obtained with the gel systems used are shown in Fig. 3.
Imipenem-susceptible revertant 810-REV demonstrated two major OMPs of 42 and 39 kDa, which were presumed to be the OmpC and OmpF analogs, respectively (Fig. 3). Strain 810 grown in the absence of imipenem produced two possible OMPs of 42 and 39 kDa, although the level of production of the 42-kDa protein was apparently lower than that in 810-REV. However, when strain 810 was grown in the presence of imipenem, it apparently produced only a 39-kDa OMP; the 42-kDa protein was not visible on the gel (Fig. 3 and 4). By contrast, ATCC 13048 produced four possible OMPs of 45, 42, 41, and 38 kDa (Fig. 3).
To determine whether the 42- and 39-kDa proteins of 810-REV were OmpC and OmpF analogs, we prepared Western blots of the OMPs using two different gel systems and analyzed them using polyclonal anti-OmpK36 (OmpC analog) and polyclonal anti-OmpK35 (OmpF analog) antisera raised against K. pneumoniae OMPs. The anti-OmpK35 (OmpF) antiserum reacts with OmpK35 and cross-reacts with OmpK36 (OmpC) (16), while the anti-OmpK36 antiserum reacts only with OmpC analogs. Two bands of 38 and 41 kDa, corresponding to OmpF (OmpK35) and OmpC (OmpK36), respectively, were identified in ATCC 13048 (Fig. 4A). Western blot analysis of the OMPs of the parent and revertant with anti-OmpK36 and anti-OmpK35 antisera showed that 810 expressed only the 42-kDa OmpC analog in the absence of imipenem (the 39-kDa protein was not recognized by either the anti-OmpF or anti-OmpC antisera) and neither the OmpC nor the OmpF analog in the presence of imipenem (Fig. 4B and C). The apparent 39-kDa protein observed by polyacrylamide gel electrophoresis may be a derivative of the OmpF analog that is no longer functional as a porin (Fig. 4B). The OmpC analog is apparently down-regulated in the presence of imipenem. Strain 810-REV expressed both OmpF and OmpC analogs (Fig. 4). These data suggest that imipenem resistance in 810 is primarily associated with the lack of expression of the analogs of OmpC (42 kDa) and OmpF (39 kDa), which also results in decreased susceptibilities to meropenem and cefepime.
DISCUSSION
E. aerogenes is a common gram-negative pathogen which causes health care-associated infections worldwide (15, 24, 29). Over the last 5 years, multidrug-resistant strains of E. aerogenes have caused outbreaks in a number of intensive care units around the world (1, 9, 25). Carbapenems, such as imipenem and meropenem, often are used to treat multidrug-resistant isolates of Enterobacter, especially those producing extended-spectrum β-lactamases (11, 25). However, a significant increase in the frequency of carbapenem resistance for E. aerogenes has been observed (13, 31), complicating therapy of these infections.
Our data suggest that alteration of both the OmpF and OmpC porin analogs is primarily responsible for the imipenem-resistant phenotype of E. aerogenes 810. Similar observations regarding the role of porins in imipenem resistance in E. aerogenes were reported by Bornet et al. (3). We were able to obtain an imipenem-susceptible revertant of 810 by subculturing the organism five times on nonselective medium. Examination of the porin profiles on gels suggested that the resistant phenotype was due to the change of a single porin, OmpC. However, Western blotting with antisera that recognize OmpC and OmpF indicated that both porin analogs are involved. The OMP presumed to be OmpF in strain 810 did not react with anti-OmpK35 antisera. Thus, this 39-kDa protein may or may not have been related to the original OmpF analog, but if it was, it clearly lost not only its antigenicity but also its functionality. Thus, confirmation of the presence of the specific OMP analogs by Western blotting was critical to deciphering the roles of the OMPs in the phenotypes of the parent and the revertant.
The parent and revertant isolates were also highly resistant to extended-spectrum cephalosporins, presumably due to production of the chromosomal AmpC β-lactamase. The cefepime and meropenem MICs were also elevated for E. aerogenes 810, but this was likely due to the change in porin expression rather than β-lactamase production. The role of the AmpC β-lactamase in increased imipenem resistance in the revertant isolate, based on the results of β-lactamase assays, appeared to be minimal. Similarly, although we have no direct data, the role of efflux systems, such as mar (7) or the AcrAB system of E. coli (21), which have been shown to play a role in β-lactam resistance, also is likely to be minimal in this isolate, since the isolate remained susceptible to tetracycline and fluoroquinolones, drugs known to be extruded by efflux systems (7, 21).
The differences in the porin profiles of E. aerogenes 810 when grown in the presence and absence of imipenem were striking and suggest that the regulation of porin expression could make detection of imipenem or meropenem resistance difficult in the clinical laboratory. Our data and the data published by Bornet et al. (3) and Chevalier et al. (6) suggest that the incidence of carbapenem resistance in E. aerogenes may well increase as the use of carbapenems increases (10) and that a constellation of mechanisms, including carbapenemases, efflux systems, and enhanced AmpC production rather than a single mechanism will be responsible.
Acknowledgments
This work was supported by an American Society for Microbiology-National Center for Infectious Diseases postdoctoral fellowship to H.Y. and by Bristol-Myers Squibb.
We thank Project ICARE for providing E. aerogenes strain 810. We thank R. Colonno, J. Fung-Tomc, and D. P. Bonner for support of the study. We also thank S. Alberti for providing anti-OmpK35- and anti-OmpK36-specific antibodies.
Use of trade names is for identification purposes only and does not constitute endorsement by the Public Health Service or the U.S. Department of Health and Human Services.
REFERENCES
- 1.Allerberger, F., T. Koeuth, C. Lass-Flor, M. P. Dierch, C. Putensen, E. Schmutzhard, I. Mohsenipuor, H. Grundmann, D. Hartung, A. Bauernfeind, E. Eberlein, and J. R. Lupski. 1996. Epidemiology of infection due to multiresistant Enterobacter aerogenes in a university hospital. Eur. J. Clin. Microbiol. Infect. Dis. 15:517-520. [DOI] [PubMed] [Google Scholar]
- 2.Arpin, C., C. Coze, A. M. Rogues, J. P. Gachie, C. Bebear, and C. Quentin. 1996. Epidemiological study of an outbreak due to multidrug-resistant Enterobacter aerogenes in a medical intensive care unit. J. Clin. Microbiol. 34:2163-2169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bornet, C., A. Davin-Regli, C. Bosi, J. M. Pages, and C. Bollet. 2000. Imipenem resistance of Enterobacter aerogenes mediated by outer membrane permeability. J. Clin. Microbiol. 38:1048-1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bush, K., and S. B. Singer. 1989. Effective cooling allows sonication to be used for liberation of β-lactamase from gram-negative bacteria. J. Antimicrob. Chemother. 24:82-84. [DOI] [PubMed] [Google Scholar]
- 5.Charrel, R. N., J. M. Pages, P. D. Micco, and M. Mallea. 1996. Prevalence of outer membrane porin alteration in beta-lactam antibiotic-resistant Enterobacter aerogenes. Antimicrob. Agents Chemother. 40:2854-2858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chevalier, J., J. M. Pages, and M. Mallea. 1999. In vitro modification of porin activity conferring antibiotic resistance to Enterobacter aerogenes. Biochem. Biophys. Res. Commun. 266:248-251. [DOI] [PubMed] [Google Scholar]
- 7.Chollet, R., C. Bollet, J. Chevalier, M. Mallea, J. M. Pages, and A. Davin-Regli. 2002. mar operon involved in multidrug resistance of Enterobacter aerogenes. Antimicrob. Agents Chemother. 46:1093-1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cornaglia, G., K. Russell, G. Satta, and R. Fontana. 1995. Relative importance of outer membrane permeability and group 1 β-lactamase as determinants of meropenem and imipenem activities against Enterobacter cloacae. Antimicrob. Agents Chemother. 39:350-355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davin-Regli, A., D. Monnet, P. Saux, C. Bosi, R. Charrel, A. Barthelemy, and C. Bollet. 1996. Molecular epidemiology of Enterobacter aerogenes acquisition: one-year prospective study in two intensive care units. J. Clin. Microbiol. 34:1474-1480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De, E., A. Basle, M. Jaquinod, N. Saint, M. Mallea, G. Molle, and J. M. Pages. 2001. A new mechanism of antibiotic resistance in Enterobacteriaceae induced by a structural modification of the major porin. Mol. Microbiol. 41:189-198. [DOI] [PubMed] [Google Scholar]
- 11.De Gheldre, Y., N. Maes, F. Rost, R. De Ryck, P. Clevenbergh, J. L. Vincent, and M. J. Struelens. 1997. Molecular epidemiology of an outbreak of multidrug-resistant Enterobacter aerogenes infections and in vivo emergence of imipenem resistance. J. Clin. Microbiol. 35:152-160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Domenech-Sanchez, A., S. Hernandez-Alles, L. Martinez-Martinez, V. J. Benedi, and S. Alberti. 1999. Identification and characterization of a new porin gene of Klebsiella pneumoniae: its role in β-lactam antibiotic resistance. J. Bacteriol. 181:2726-2732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ehrhardt, A. F., C. C. Sanders, K. S. Thomson, C. Watanakunakorn, and I. Trulillano-Martin. 1993. Emergence of resistance to imipenem in Enterobacter strains masquerading as Klebsiella pneumoniae during therapy with imipenem/cilastatin. Clin. Infect. Dis. 17:120-122. [DOI] [PubMed] [Google Scholar]
- 14.Fridkin, S. K., C. D. Steward, J. R. Edwards, E. R. Pryor, J. E. McGowan, Jr., L. K. Archibald, R. P. Gaynes, and F. C. Tenover. 1999. Surveillance of antimicrobial use and antimicrobial resistance in United States hospitals: Project ICARE phase 2. Project Intensive Care Antimicrobial Resistance Epidemiology (ICARE) hospitals. Clin. Infect. Dis. 29:245-252. [DOI] [PubMed] [Google Scholar]
- 15.Georghiou, P. R., R. J. Hamill, C. E. Wright, J. Versalovic, T. Koeuth, D. A. Watson, and J. Lupski. 1995. Molecular epidemiology of infections due to Enterobacter aerogenes: identification of hospital outbreak-associated strains by molecular techniques. Clin. Infect. Dis. 20:84-94. [DOI] [PubMed] [Google Scholar]
- 16.Hernandez-Alles, S., S. Alberti, D. Alvarez, A. Domenech-Sanchez, L. Martinez-Martinez, J. Gil, J. M. Tomas, and V. J. Benedi. 1999. Porin expression in clinical isolates of Klebsiella pneumoniae. Microbiology 145:673-679. [DOI] [PubMed] [Google Scholar]
- 17.Jalaluddin, S., J. M. Devaster, R. Scheen, M. Gerard, and J. P. Butzler. 1998. Molecular epidemiological study of nosocomial Enterobacter aerogenes isolates in a Belgian hospital. J. Clin. Microbiol. 36:1846-1852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Leying, H., W. Cullmann, and W. Dick. 1991. Carbapenem resistance in Enterobacter aerogenes is due to lipopolysaccharide alterations. Chemotherapy 37:106-113. [DOI] [PubMed] [Google Scholar]
- 19.Mallea, M., J. Chevalier, C. Bornet, A. Eyraud, A. Davin-Regli, C. Bollet, and J. M. Pages. 1998. Porin alteration and active efflux: two in vivo drug resistance strategies used by Enterobacter aerogenes. Microbiology 144:3003-3009. [DOI] [PubMed] [Google Scholar]
- 20.Matthew, M., and A. M. Harris. 1976. Identification of β-lactamases by analytical isoelectric focusing: correlation with bacterial taxonomy. J. Gen. Microbiol. 94:55-67. [DOI] [PubMed] [Google Scholar]
- 21.Mazzariol, A., G. Cornaglia, and H. Nikaido. 2000. Contributions of the AmpC β-lactamase and the AcrAB multidrug efflux system in intrinsic resistance of Escherichia coli K-12 to β-lactams. Antimicrob. Agents Chemother. 44:1387-1390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.National Committee for Clinical Laboratory Standards. 2000. Performance standards for antimicrobial disk susceptibility tests. Approved standard M2-A7. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 23.National Committee for Clinical Laboratory Standards. 2000. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard M7-A5. National Committee for Clinical Laboratory Standards, Wayne, Pa.
- 24.National Nosocomial Infections Surveillance System. 1996. A report from the National Nosocomial Infections Surveillance (NNIS) System. Am. J. Infect. Control 24:380-388. [PubMed] [Google Scholar]
- 25.Neuwirth, C., E. Siebor, J. Lopez, A. Pechinot, and A. Kazmierzak. 1996. Outbreak of TEM-24-producing Enterobacter aerogenes in an intensive care unit and dissemination of the extended-spectrum beta-lactamase to other members of the family Enterobacteriaceae. J. Clin. Microbiol. 34:76-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rasheed, J. K., C. Jay, B. Metchock, F. Berkowitz, L. Weigel, J. Crellin, C. Steward, B. Hill, A. A. Medeiros, and F. C. Tenover. 1997. Evolution of extended-spectrum β-lactam resistance (SHV-8) in a strain of Escherichia coli during multiple episodes of bacteremia. Antimicrob. Agents Chemother. 41:647-653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ronveaux, O., Y. De Gheldre, Y. Glupczynski, M. J. Struelens, and P. De Mol. 1999. Emergence of Enterobacter aerogenes as a major antibiotic-resistant nosocomial pathogen in Belgian hospitals. Clin. Microbiol. Infect. 5:622-627. [DOI] [PubMed] [Google Scholar]
- 28.Sanders, C. C., W. E. Sanders, Jr., and E. S. Moland. 1986. Characterization of β-lactamases in situ on polyacrylamide gels. Antimicrob. Agents Chemother. 30:951-952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sanders, W. E., and C. C. Sanders. 1997. Enterobacter spp.: pathogens poised to flourish at the turn of the century. Clin. Microbiol. Rev. 10:220-241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sykes, R. B., D. P. Bonner, K. Bush, and N. H. Georgopapadakou. 1982. Aztreonam (SQ 26,776), a synthetic monobactam specifically active against aerobic gram-negative bacteria. Antimicrob. Agents Chemother. 21:85-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tzouvelekis, L. S., E. Tzelepi, A. F. Mentis, A. C. Vatopoulos, and A. Tsakris. 1992. Imipenem resistance in Enterobacter aerogenes is associated with derepression of chromosomal cephalosporinases and impaired permeability. FEMS Microbiol. Lett. 74:195-199. [DOI] [PubMed] [Google Scholar]
- 32.Wu, P.-J., K. Shannon, and I. Phillips. 1994. Effect of hyperproduction of TEM-1 β-lactamase on in vitro susceptibility of Escherichia coli to β-lactam antibiotics. Antimicrob. Agents Chemother. 38:494-498. [DOI] [PMC free article] [PubMed] [Google Scholar]