Abstract
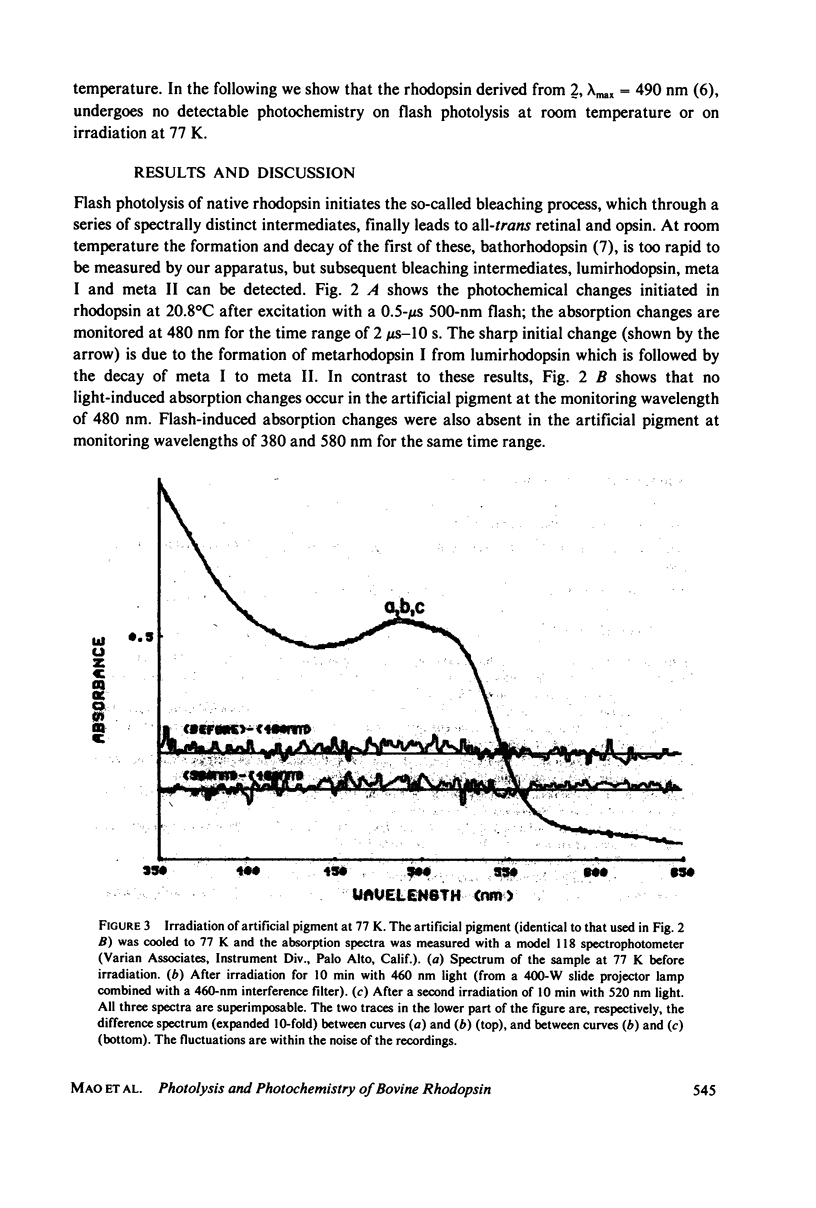
Nonbleachable rhodopsins containing retinal moieties with fixed 11-ene structures have been prepared. When the nonbleachable rhodopsin analogue corresponding to the natural pigment was flash-photolysed at 20.8 degrees C, no absorption changes occurred at the monitoring wavelengths of 380, 480, and 580 nm for the time range of 2 microseconds--10 s. This observation is in contrast to that of natural rhodopsin which showed the formation of metarhodopsin I and its decay to meta II. Irradiation of the artificial rhodopsin, 77 K, with light of 460 and 540 nm, also gave no spectral changes; in the case of natural rhodopsin, however, the irradiation leads to formation of the red-shifted intermediate bathorhodopsin. The absence of photochemistry in the artificial pigment shows that an 11-cis to trans photoisomerization of the retinal moiety is a crucial step in inducing the chain of events in te photolysis of rhodopsin.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Callender R. H., Doukas A., Crouch R., Nakanishi K. Molecular flow resonance Raman effect from retinal and rhodopsin. Biochemistry. 1976 Apr 20;15(8):1621–1629. doi: 10.1021/bi00653a005. [DOI] [PubMed] [Google Scholar]
- Chan W. K., Nakanishi K., Ebrey T. G., Honig B. Letter: Properties of 14-methylretinal, 13-desmethyl-14-methylretinal, and visual pigments formed therefrom. J Am Chem Soc. 1974 May 29;96(11):3642–3644. doi: 10.1021/ja00818a045. [DOI] [PubMed] [Google Scholar]
- Honig B., Ebrey T., Callender R. H., Dinur U., Ottolenghi M. Photoisomerization, energy storage, and charge separation: a model for light energy transduction in visual pigments and bacteriorhodopsin. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2503–2507. doi: 10.1073/pnas.76.6.2503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubbard R., Kropf A. THE ACTION OF LIGHT ON RHODOPSIN. Proc Natl Acad Sci U S A. 1958 Feb;44(2):130–139. doi: 10.1073/pnas.44.2.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huppert D., Rentzepis P. M., Kliger D. S. Picosecond and nanosecond isomerization kinetics of protonated 11-cis retinylidene Schiff bases. Photochem Photobiol. 1977 Feb;25(2):193–197. doi: 10.1111/j.1751-1097.1977.tb06897.x. [DOI] [PubMed] [Google Scholar]
- Peters K., Applebury M. L., Rentzepis P. M. Primary photochemical event in vision: proton translocation. Proc Natl Acad Sci U S A. 1977 Aug;74(8):3119–3123. doi: 10.1073/pnas.74.8.3119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warshel A. Charge stabilization mechanism in the visual and purple membrane pigments. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2558–2562. doi: 10.1073/pnas.75.6.2558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Meer K., Mulder J. J., Lugtenburg J. A new facet in rhodopsin photochemistry. Photochem Photobiol. 1976 Oct;24(4):363–367. doi: 10.1111/j.1751-1097.1976.tb06837.x. [DOI] [PubMed] [Google Scholar]


