Abstract
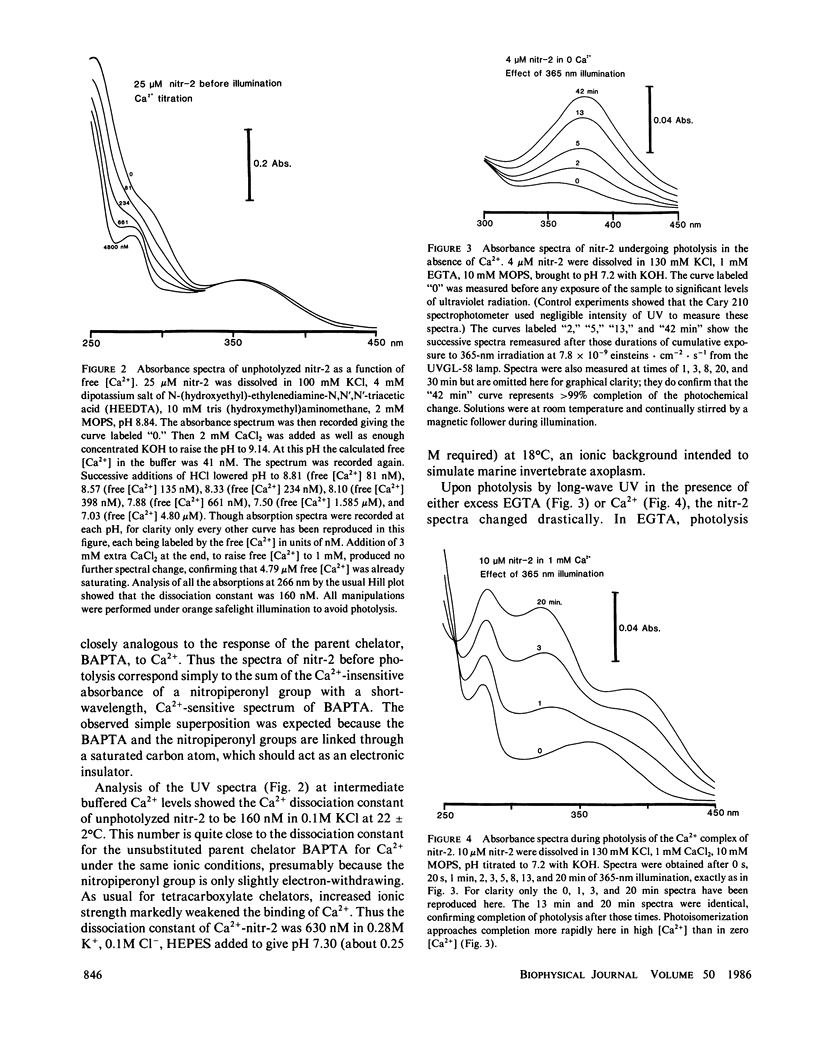
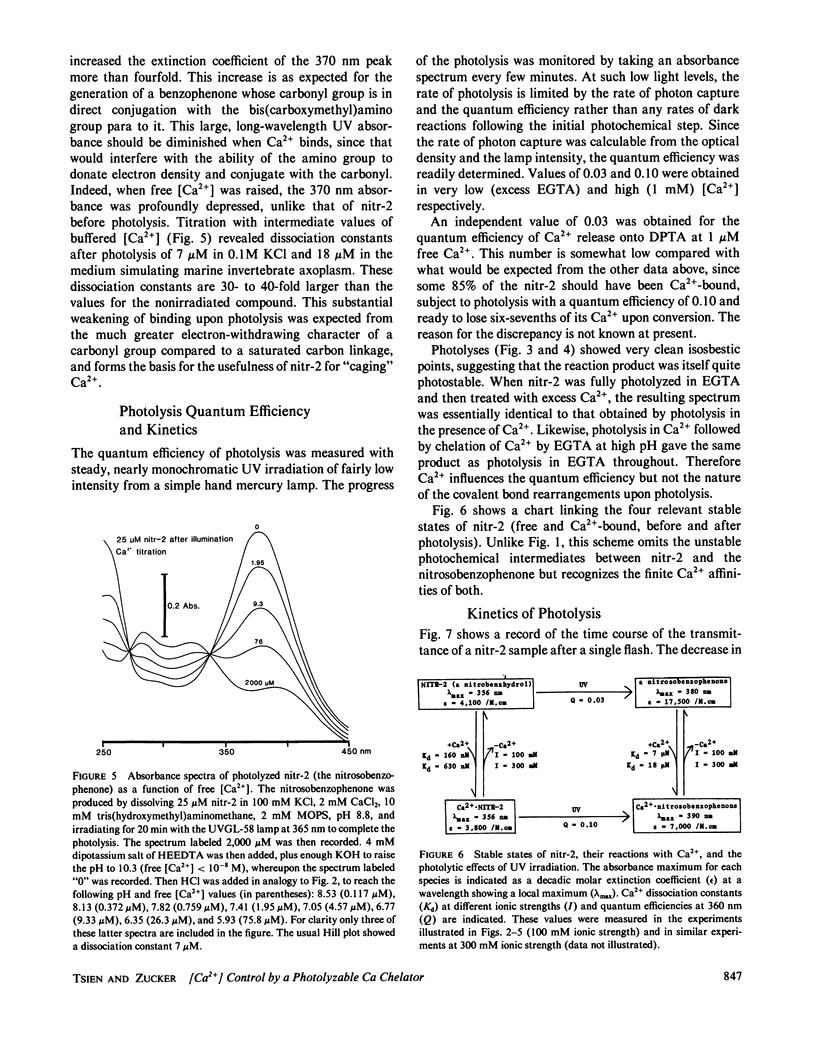
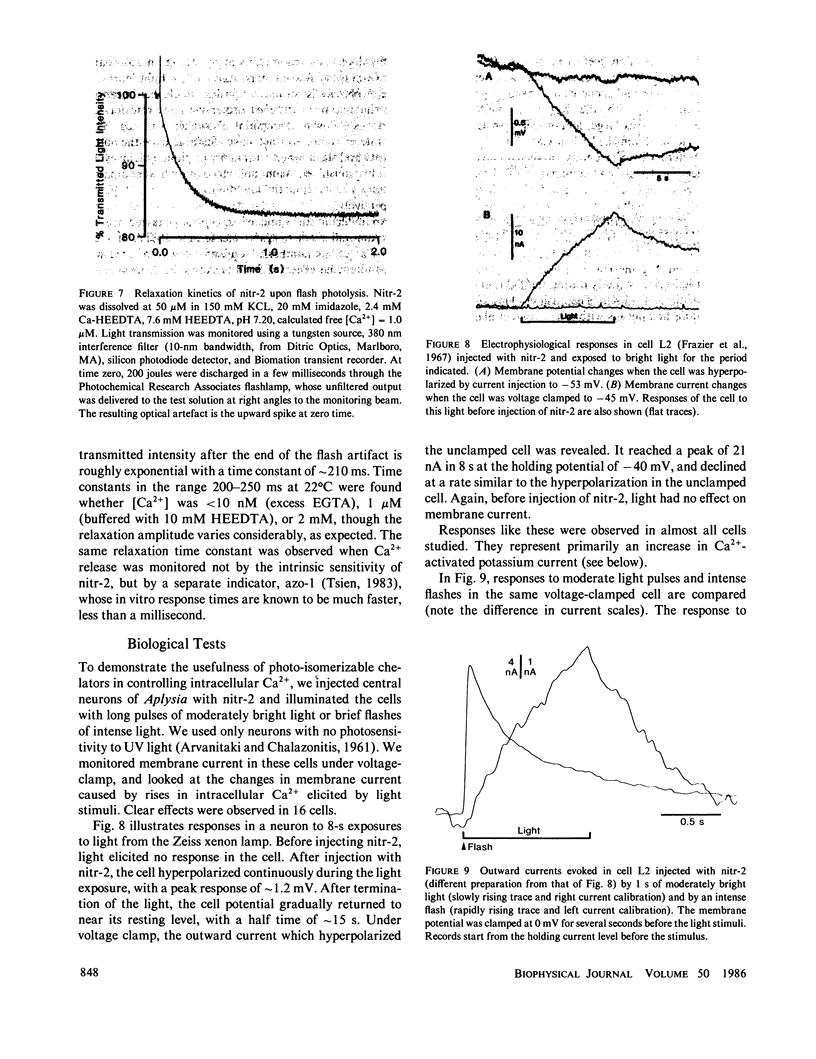
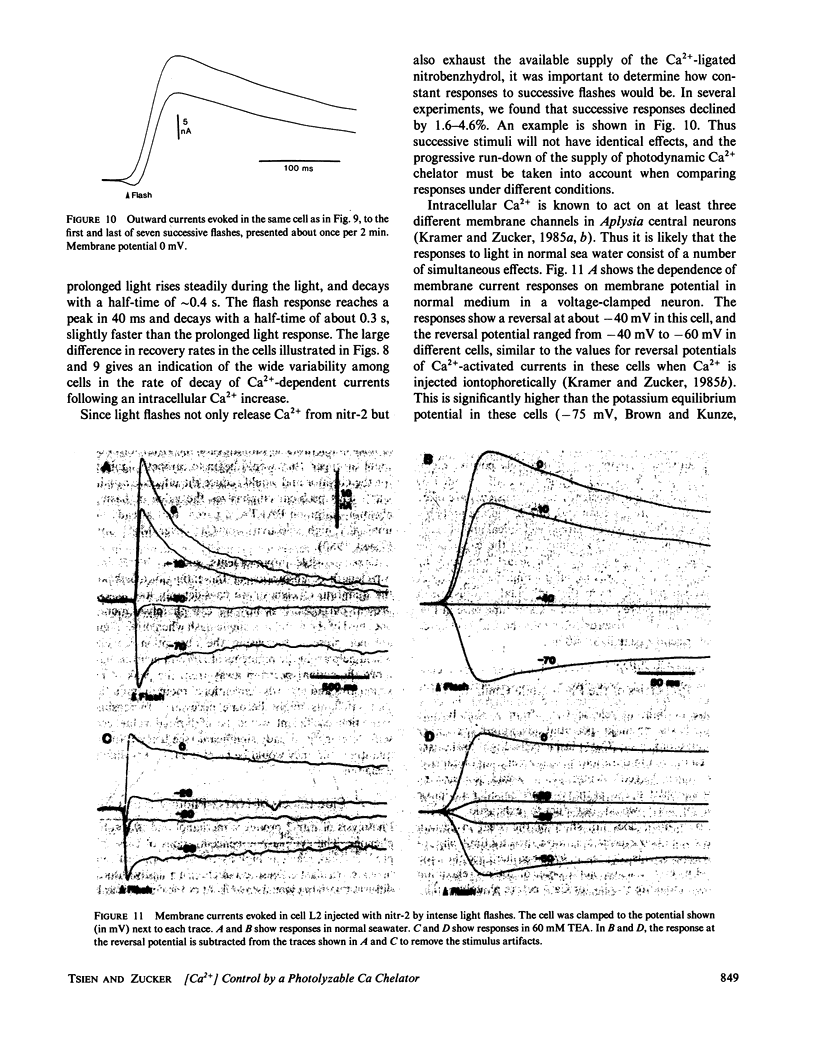
This paper introduces nitr-2, a new Ca2+ chelator designed to release Ca2+ upon illumination with near UV (300-400 nm) light. Before illumination nitr-2 has Ca2+ dissociation constants of 160 and 630 nM in 0.1 and 0.3 M ionic strength respectively; after photoconversion to a nitrosobenzophenone the values shift to 7 and 18 microM, high enough to liberate substantial amounts of Ca2+ under intracellular conditions. The speed of release is limited by a dark reaction with rate constant 5 s-1. Aplysia central neurons injected with nitr-2 and exposed to UV light exhibit two separate Ca2+-dependent membrane currents: one carried by potassium ions and one a nonspecific cation current. A quantitative estimate of the spatial distribution of intracellular [Ca2+] changes in large cells filled with a high concentration of nitr-2 and exposed to an intense UV flash is offered.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ARMSTRONG C. M., BINSTOCK L. THE EFFECTS OF SEVERAL ALCOHOLS ON THE PROPERTIES OF THE SQUID GIANT AXON. J Gen Physiol. 1964 Nov;48:265–277. doi: 10.1085/jgp.48.2.265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alemà S., Calissano P., Rusca G., Giuditta A. Identification of a calcium-binding, brain specific protein in the axoplasm of squid giant axons. J Neurochem. 1973 Mar;20(3):681–689. doi: 10.1111/j.1471-4159.1973.tb00028.x. [DOI] [PubMed] [Google Scholar]
- Barrett J. N., Magleby K. L., Pallotta B. S. Properties of single calcium-activated potassium channels in cultured rat muscle. J Physiol. 1982 Oct;331:211–230. doi: 10.1113/jphysiol.1982.sp014370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown A. M., Kunze D. L. Ionic activities in identifiable Aplysia neurons. Adv Exp Med Biol. 1974;50(0):57–73. doi: 10.1007/978-1-4615-9023-1_6. [DOI] [PubMed] [Google Scholar]
- Corson D. W., Fein A. Quantitative pressure injection of picoliter volumes into Limulus ventral photoreceptors. Biophys J. 1983 Dec;44(3):299–304. doi: 10.1016/S0006-3495(83)84303-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiPolo R., Rojas H., Vergara J., Lopez R., Caputo C. Measurements of intracellular ionized calcium in squid giant axons using calcium-selective electrodes. Biochim Biophys Acta. 1983 Mar 9;728(3):311–318. doi: 10.1016/0005-2736(83)90500-x. [DOI] [PubMed] [Google Scholar]
- Eckert R., Tillotson D. L. Calcium-mediated inactivation of the calcium conductance in caesium-loaded giant neurones of Aplysia californica. J Physiol. 1981 May;314:265–280. doi: 10.1113/jphysiol.1981.sp013706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorman A. L., Levy S., Nasi E., Tillotson D. Intracellular calcium measured with calcium-sensitive micro-electrodes and Arsenazo III in voltage-clamped Aplysia neurones. J Physiol. 1984 Aug;353:127–142. doi: 10.1113/jphysiol.1984.sp015327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grynkiewicz G., Poenie M., Tsien R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985 Mar 25;260(6):3440–3450. [PubMed] [Google Scholar]
- Hagiwara S., Byerly L. Calcium channel. Annu Rev Neurosci. 1981;4:69–125. doi: 10.1146/annurev.ne.04.030181.000441. [DOI] [PubMed] [Google Scholar]
- Hermann A., Gorman A. L. External and internal effects of tetraethylammonium on voltage-dependent and Ca-dependent K+ currents components in molluscan pacemaker neurons. Neurosci Lett. 1979 Apr;12(1):87–92. doi: 10.1016/0304-3940(79)91485-x. [DOI] [PubMed] [Google Scholar]
- Kaplan J. H., Forbush B., 3rd, Hoffman J. F. Rapid photolytic release of adenosine 5'-triphosphate from a protected analogue: utilization by the Na:K pump of human red blood cell ghosts. Biochemistry. 1978 May 16;17(10):1929–1935. doi: 10.1021/bi00603a020. [DOI] [PubMed] [Google Scholar]
- Kramer R. H., Zucker R. S. Calcium-dependent inward current in Aplysia bursting pace-maker neurones. J Physiol. 1985 May;362:107–130. doi: 10.1113/jphysiol.1985.sp015666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramer R. H., Zucker R. S. Calcium-induced inactivation of calcium current causes the inter-burst hyperpolarization of Aplysia bursting neurones. J Physiol. 1985 May;362:131–160. doi: 10.1113/jphysiol.1985.sp015667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lester H. A., Nerbonne J. M. Physiological and pharmacological manipulations with light flashes. Annu Rev Biophys Bioeng. 1982;11:151–175. doi: 10.1146/annurev.bb.11.060182.001055. [DOI] [PubMed] [Google Scholar]
- Liu C., Hermann T. E. Characterization of ionomycin as a calcium ionophore. J Biol Chem. 1978 Sep 10;253(17):5892–5894. [PubMed] [Google Scholar]
- MOORE J. W., ULBRICHT W., TAKATA M. EFFECT OF ETHANOL ON THE SODIUM AND POTASSIUM CONDUCTANCES OF THE SQUID AXON MEMBRANE. J Gen Physiol. 1964 Nov;48:279–295. doi: 10.1085/jgp.48.2.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meech R. W. Calcium-dependent potassium activation in nervous tissues. Annu Rev Biophys Bioeng. 1978;7:1–18. doi: 10.1146/annurev.bb.07.060178.000245. [DOI] [PubMed] [Google Scholar]
- Nargeot J., Nerbonne J. M., Engels J., Lester H. A. Time course of the increase in the myocardial slow inward current after a photochemically generated concentration jump of intracellular cAMP. Proc Natl Acad Sci U S A. 1983 Apr;80(8):2395–2399. doi: 10.1073/pnas.80.8.2395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed P. W., Lardy H. A. A23187: a divalent cation ionophore. J Biol Chem. 1972 Nov 10;247(21):6970–6977. [PubMed] [Google Scholar]
- Smith S. J., Zucker R. S. Aequorin response facilitation and intracellular calcium accumulation in molluscan neurones. J Physiol. 1980 Mar;300:167–196. doi: 10.1113/jphysiol.1980.sp013157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsien R. Y. Intracellular measurements of ion activities. Annu Rev Biophys Bioeng. 1983;12:91–116. doi: 10.1146/annurev.bb.12.060183.000515. [DOI] [PubMed] [Google Scholar]
- Tsien R. Y. New calcium indicators and buffers with high selectivity against magnesium and protons: design, synthesis, and properties of prototype structures. Biochemistry. 1980 May 27;19(11):2396–2404. doi: 10.1021/bi00552a018. [DOI] [PubMed] [Google Scholar]
- Wong B. S., Lecar H., Adler M. Single calcium-dependent potassium channels in clonal anterior pituitary cells. Biophys J. 1982 Sep;39(3):313–317. doi: 10.1016/S0006-3495(82)84522-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zucker R. S., Steinhardt R. A. Prevention of the cortical reaction in fertilized sea urchin eggs by injection of calcium-chelating ligands. Biochim Biophys Acta. 1978 Jul 17;541(4):459–466. doi: 10.1016/0304-4165(78)90155-1. [DOI] [PubMed] [Google Scholar]
- Zucker R. S. Tetraethylammonium contains an impurity which alkalizes cytoplasm and reduce calcium buffering in neurons. Brain Res. 1981 Mar 16;208(2):473–478. doi: 10.1016/0006-8993(81)90580-1. [DOI] [PubMed] [Google Scholar]



