Abstract
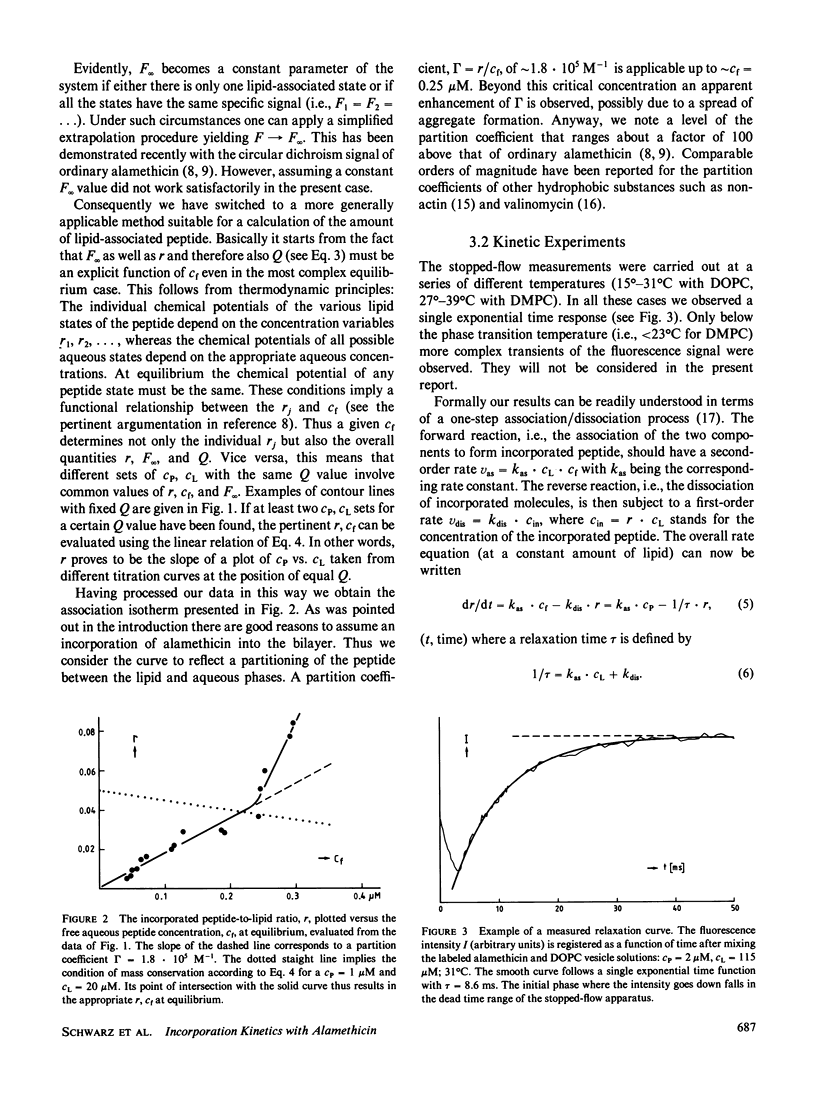
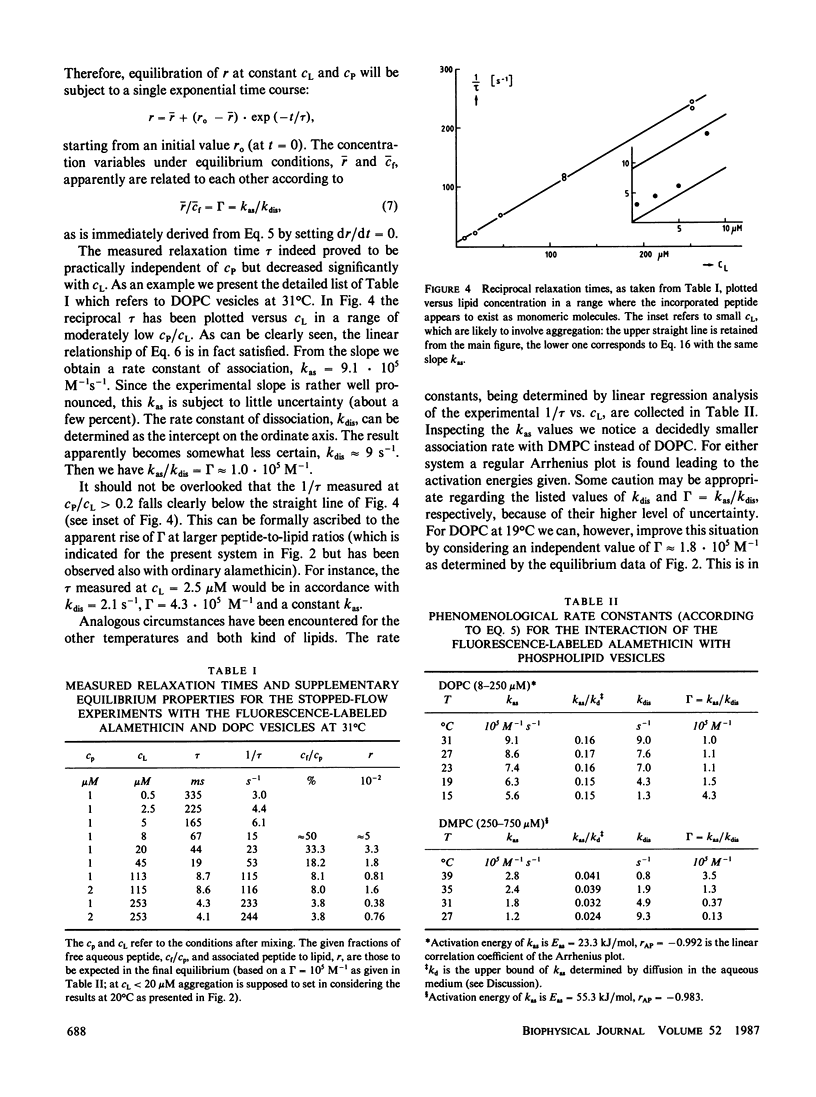
The reaction of fluorescence-labeled alamethicin with unilamellar phospholipid vesicles (DOPC and DMPC) has been investigated in a stopped-flow apparatus. Clearly single exponential time functions have been observed at temperatures above the phase transition of the bilayer. This can be interpreted in terms of an essentially one-step incorporation process. The pseudo first-order forward rate is found to be quite fast, falling in a range somewhat below the diffusion controlled upper bound. The data are quantitatively very well described on the basis of a simple mechanism. This comprises diffusion of peptide into the bilayer accompanied by a more or less slower change of the secondary structure. Aggregation of the incorporated molecules at higher concentrations is indicated to be comparatively rapid.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boheim G. Statistical analysis of alamethicin channels in black lipid membranes. J Membr Biol. 1974;19(3):277–303. doi: 10.1007/BF01869983. [DOI] [PubMed] [Google Scholar]
- Fringeli U. P., Fringeli M. Pore formation in lipid membranes by alamethicin. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3852–3856. doi: 10.1073/pnas.76.8.3852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall J. E., Vodyanoy I., Balasubramanian T. M., Marshall G. R. Alamethicin. A rich model for channel behavior. Biophys J. 1984 Jan;45(1):233–247. doi: 10.1016/S0006-3495(84)84151-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knoll W. Raman-spectroscopic evidence for the incorporation of alamethicin into dimyristoylphosphatidylcholine bilayers. Biochim Biophys Acta. 1986 Dec 16;863(2):329–331. doi: 10.1016/0005-2736(86)90276-2. [DOI] [PubMed] [Google Scholar]
- Latorre R., Miller C. G., Quay S. Voltage-dependent conductance induced by alamethicin-phospholipid conjugates in lipid bilayers. Biophys J. 1981 Dec;36(3):803–809. doi: 10.1016/S0006-3495(81)84767-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMullen A. I., Stirrup J. A. The aggregation of alamethicin. Biochim Biophys Acta. 1971 Sep 14;241(3):807–814. doi: 10.1016/0005-2736(71)90008-3. [DOI] [PubMed] [Google Scholar]
- Paul C., Kirschner K., Haenisch G. Calibration of stopped-flow spectrophotometers using a two-step disulfide exchange reaction. Anal Biochem. 1980 Jan 15;101(2):442–448. doi: 10.1016/0003-2697(80)90211-0. [DOI] [PubMed] [Google Scholar]
- Reiss-Husson F. Structure des phases liquide-cristallines de différents phospholipides, monoglycérides, sphingolipides, anhydres ou en présence d'eau. J Mol Biol. 1967 May 14;25(3):363–382. doi: 10.1016/0022-2836(67)90192-1. [DOI] [PubMed] [Google Scholar]
- Rinehart K. L., Jr, Cook J. C., Jr, Meng H., Olson K. L., Pandey R. C. Mass spectrometric determination of molecular formulas for membrane-modifying antibiotics. Nature. 1977 Oct 27;269(5631):832–833. doi: 10.1038/269832a0. [DOI] [PubMed] [Google Scholar]
- Rizzo V., Stankowski S., Schwarz G. Alamethicin incorporation in lipid bilayers: a thermodynamic study. Biochemistry. 1987 May 19;26(10):2751–2759. doi: 10.1021/bi00384a015. [DOI] [PubMed] [Google Scholar]
- Schwarz G. Basic kinetics of binding and incorporation with supramolecular aggregates. Biophys Chem. 1987 May 9;26(2-3):163–169. doi: 10.1016/0301-4622(87)80019-4. [DOI] [PubMed] [Google Scholar]
- Schwarz G., Savko P. Structural and dipolar properties of the voltage-dependent pore former alamethicin in octanol/dioxane. Biophys J. 1982 Aug;39(2):211–219. doi: 10.1016/S0006-3495(82)84510-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz G., Stankowski S., Rizzo V. Thermodynamic analysis of incorporation and aggregation in a membrane: application to the pore-forming peptide alamethicin. Biochim Biophys Acta. 1986 Sep 25;861(1):141–151. doi: 10.1016/0005-2736(86)90573-0. [DOI] [PubMed] [Google Scholar]
- Tank D. W., Wu E. S., Meers P. R., Webb W. W. Lateral diffusion of gramicidin C in phospholipid multibilayers. Effects of cholesterol and high gramicidin concentration. Biophys J. 1982 Nov;40(2):129–135. doi: 10.1016/S0006-3495(82)84467-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watts A., Marsh D., Knowles P. F. Characterization of dimyristoylphosphatidylcholine vesicles and their dimensional changes through the phase transition: molecular control of membrane morphology. Biochemistry. 1978 May 2;17(9):1792–1801. doi: 10.1021/bi00602a034. [DOI] [PubMed] [Google Scholar]
- Yantorno R., Takashima S., Mueller P. Dipole moment of alamethicin as related to voltage-dependent conductance in lipid bilayers. Biophys J. 1982 May;38(2):105–110. doi: 10.1016/S0006-3495(82)84536-0. [DOI] [PMC free article] [PubMed] [Google Scholar]


