Abstract
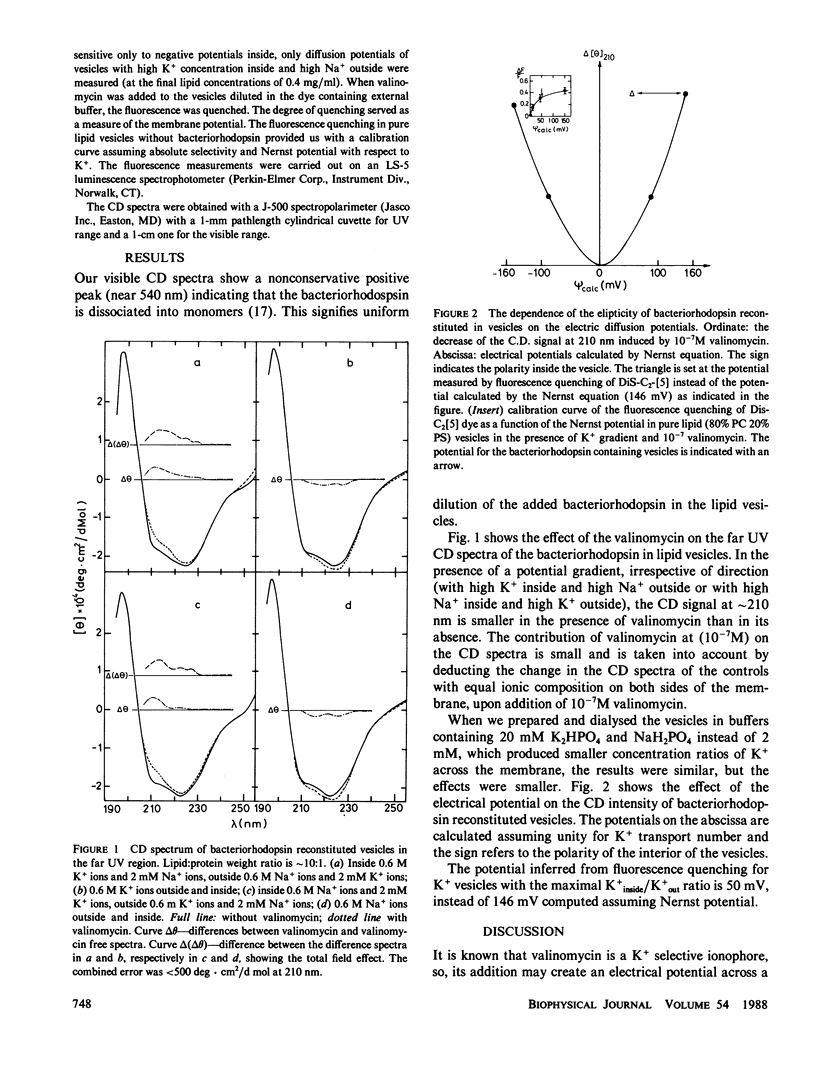
The effect of applied diffusion potential on circular dichroism (CD) of bacteriorhodopsin, reconstituted in lipid vesicles, was measured. The change in CD indicates that the applied electrical field, irrespective of its direction, decreases the α-helical fraction and increases the random fraction of the protein. The results are interpreted by unfolding of edges of the helices, upon their submerging into polar environment when the lipid bilayer is electrostricted or (and) the helices are stretched by the electrical field across the membrane.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Becher B. M., Cassim J. Y. Improved isolation procedures for the purple membrane of Halobacterium halobium. Prep Biochem. 1975;5(2):161–178. doi: 10.1080/00327487508061568. [DOI] [PubMed] [Google Scholar]
- Becher B., Cassim J. Y. Effects of light adaptation on the purple membrane structure of Halobacterium halobium. Biophys J. 1976 Oct;16(10):1183–1200. doi: 10.1016/S0006-3495(76)85767-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang C. T., Wu C. S., Yang J. T. Circular dichroic analysis of protein conformation: inclusion of the beta-turns. Anal Biochem. 1978 Nov;91(1):13–31. doi: 10.1016/0003-2697(78)90812-6. [DOI] [PubMed] [Google Scholar]
- Chen Y. H., Yang J. T., Chau K. H. Determination of the helix and beta form of proteins in aqueous solution by circular dichroism. Biochemistry. 1974 Jul 30;13(16):3350–3359. doi: 10.1021/bi00713a027. [DOI] [PubMed] [Google Scholar]
- Corda D., Pasternak C., Shinitzky M. Increase in lipid microviscosity of unilamellar vesicles upon the creation of transmembrane potential. J Membr Biol. 1982;65(3):235–242. doi: 10.1007/BF01869967. [DOI] [PubMed] [Google Scholar]
- Dencher N. A., Heyn M. P. Formation and properties of bacteriorhodopsin monomers in the non-ionic detergents octyl-beta-D-glucoside and Triton X-100. FEBS Lett. 1978 Dec 15;96(2):322–326. doi: 10.1016/0014-5793(78)80427-x. [DOI] [PubMed] [Google Scholar]
- Eisenman G., Krasne S., Ciani S. The kinetic and equilibrium components of selective ionic permeability mediated by nactin- and valinomycin-type carriers having systematically varied degrees of methylation. Ann N Y Acad Sci. 1975 Dec 30;264:34–60. doi: 10.1111/j.1749-6632.1975.tb31474.x. [DOI] [PubMed] [Google Scholar]
- Hwang S. B., Stoeckenius W. Purple membrane vesicles: morphology and proton translocation. J Membr Biol. 1977 May 12;33(3-4):325–350. doi: 10.1007/BF01869523. [DOI] [PubMed] [Google Scholar]
- Jap B. K., Maestre M. F., Hayward S. B., Glaeser R. M. Peptide-chain secondary structure of bacteriorhodopsin. Biophys J. 1983 Jul;43(1):81–89. doi: 10.1016/S0006-3495(83)84326-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keszthelyi L. Orientation of membrane fragments by electric field. Biochim Biophys Acta. 1980 Jun 6;598(3):429–436. doi: 10.1016/0005-2736(80)90023-1. [DOI] [PubMed] [Google Scholar]
- Lee D. C., Chapman D. Infrared spectroscopic studies of biomembranes and model membranes. Biosci Rep. 1986 Mar;6(3):235–256. doi: 10.1007/BF01115153. [DOI] [PubMed] [Google Scholar]
- Lee D. C., Hayward J. A., Restall C. J., Chapman D. Second-derivative infrared spectroscopic studies of the secondary structures of bacteriorhodopsin and Ca2+-ATPase. Biochemistry. 1985 Jul 30;24(16):4364–4373. doi: 10.1021/bi00337a018. [DOI] [PubMed] [Google Scholar]
- Loew L. M., Rosenberg I., Bridge M., Gitler C. Diffusion potential cascade. Convenient detection of transferable membrane pores. Biochemistry. 1983 Feb 15;22(4):837–844. doi: 10.1021/bi00273a020. [DOI] [PubMed] [Google Scholar]
- Mao D., Wallace B. A. Differential light scattering and absorption flattening optical effects are minimal in the circular dichroism spectra of small unilamellar vesicles. Biochemistry. 1984 Jun 5;23(12):2667–2673. doi: 10.1021/bi00307a020. [DOI] [PubMed] [Google Scholar]
- Nabedryk E., Bardin A. M., Breton J. Further characterization of protein secondary structures in purple membrane by circular dichroism and polarized infrared spectroscopies. Biophys J. 1985 Dec;48(6):873–876. doi: 10.1016/S0006-3495(85)83848-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papp E., Fricsovszky G., Meszéna G. Electrodichroism of purple membrane: ionic strength dependence. Biophys J. 1986 May;49(5):1089–1100. doi: 10.1016/S0006-3495(86)83737-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehorek M., Heyn M. P. Binding of all-trans-retinal to the purple membrane. Evidence for cooperativity and determination of the extinction coefficient. Biochemistry. 1979 Oct 30;18(22):4977–4983. doi: 10.1021/bi00589a027. [DOI] [PubMed] [Google Scholar]
- Reyenolds J. A., Stoeckenius W. Molecular weight of bacteriorhodopsin solubilized in Triton X-100. Proc Natl Acad Sci U S A. 1977 Jul;74(7):2803–2804. doi: 10.1073/pnas.74.7.2803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuji K., Neumann E. Conformational flexibility of membrane proteins in electric fields. I. Ultraviolet absorbance and light scattering of bacteriorhodopsin in purple membranes. Biophys Chem. 1983 Mar;17(2):153–163. doi: 10.1016/0301-4622(83)80009-x. [DOI] [PubMed] [Google Scholar]
- Wallace B. A., Teeters C. L. Differential absorption flattening optical effects are significant in the circular dichroism spectra of large membrane fragments. Biochemistry. 1987 Jan 13;26(1):65–70. doi: 10.1021/bi00375a010. [DOI] [PubMed] [Google Scholar]


