Abstract
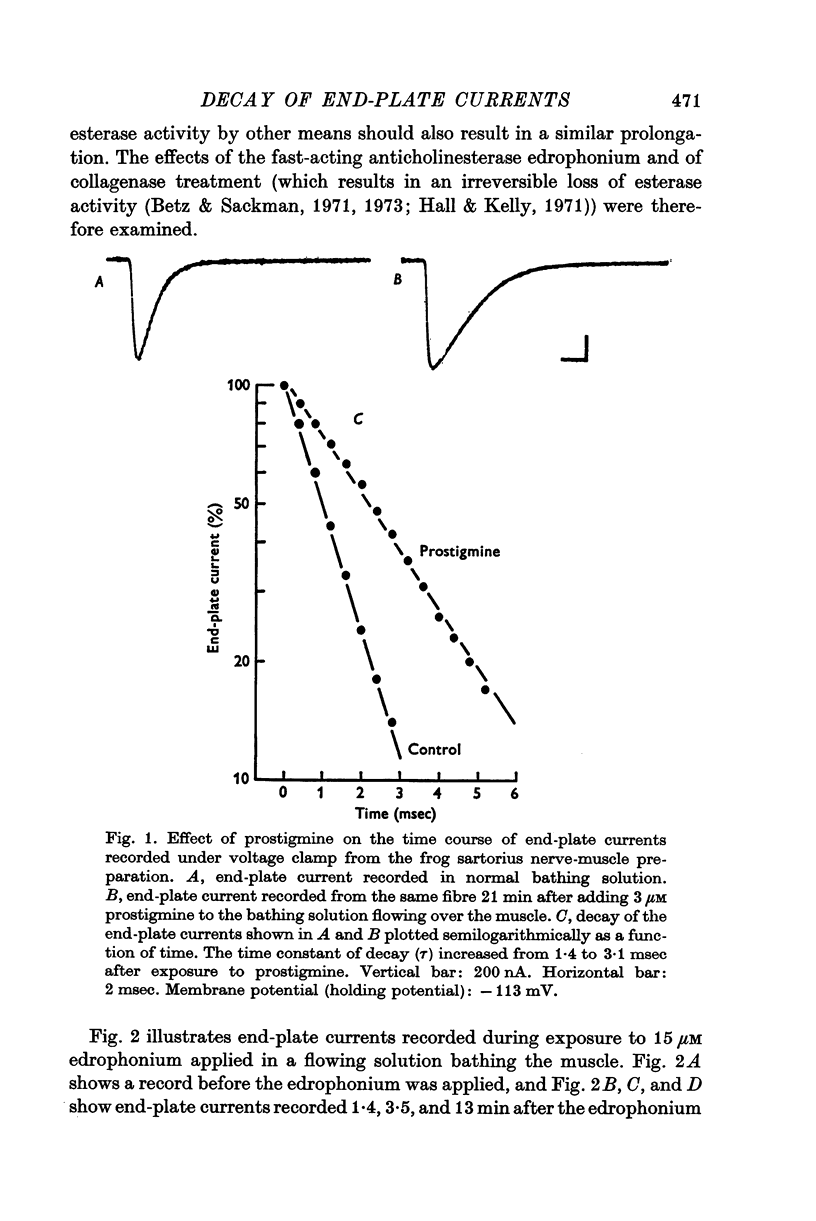
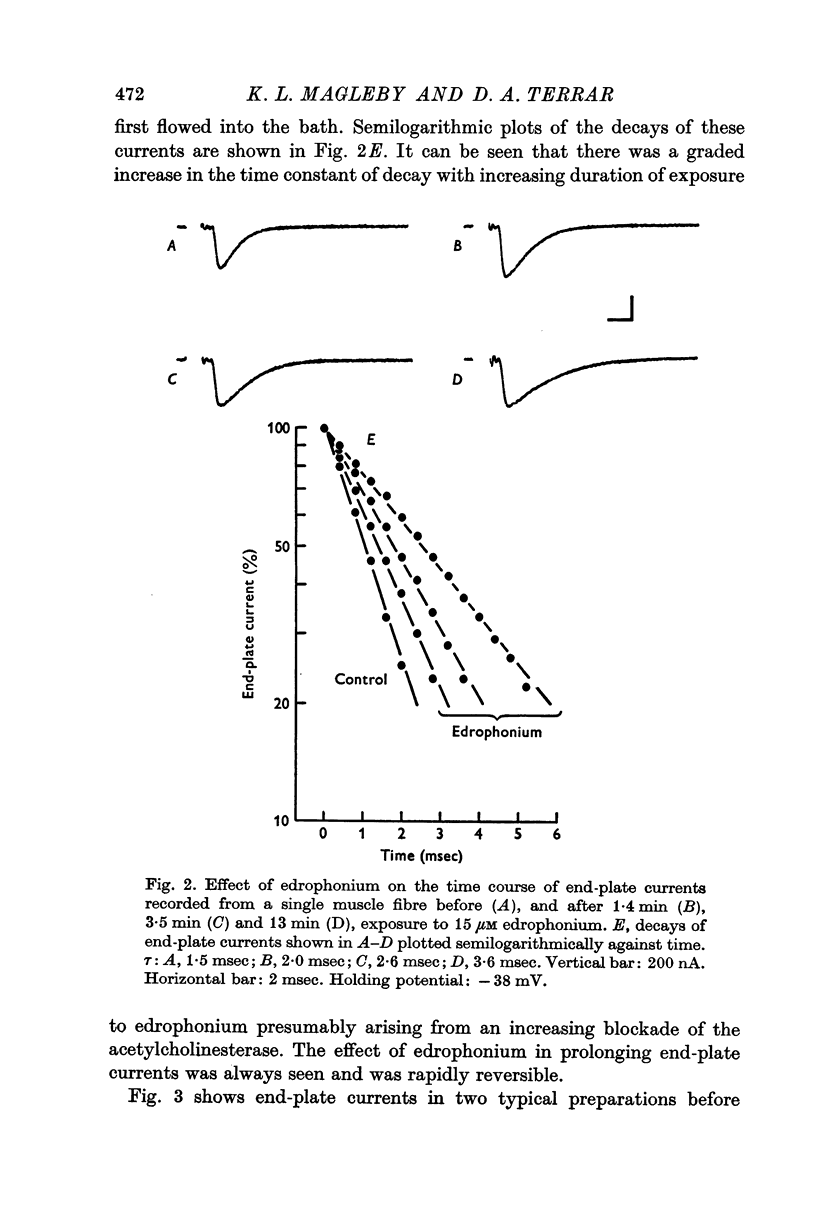
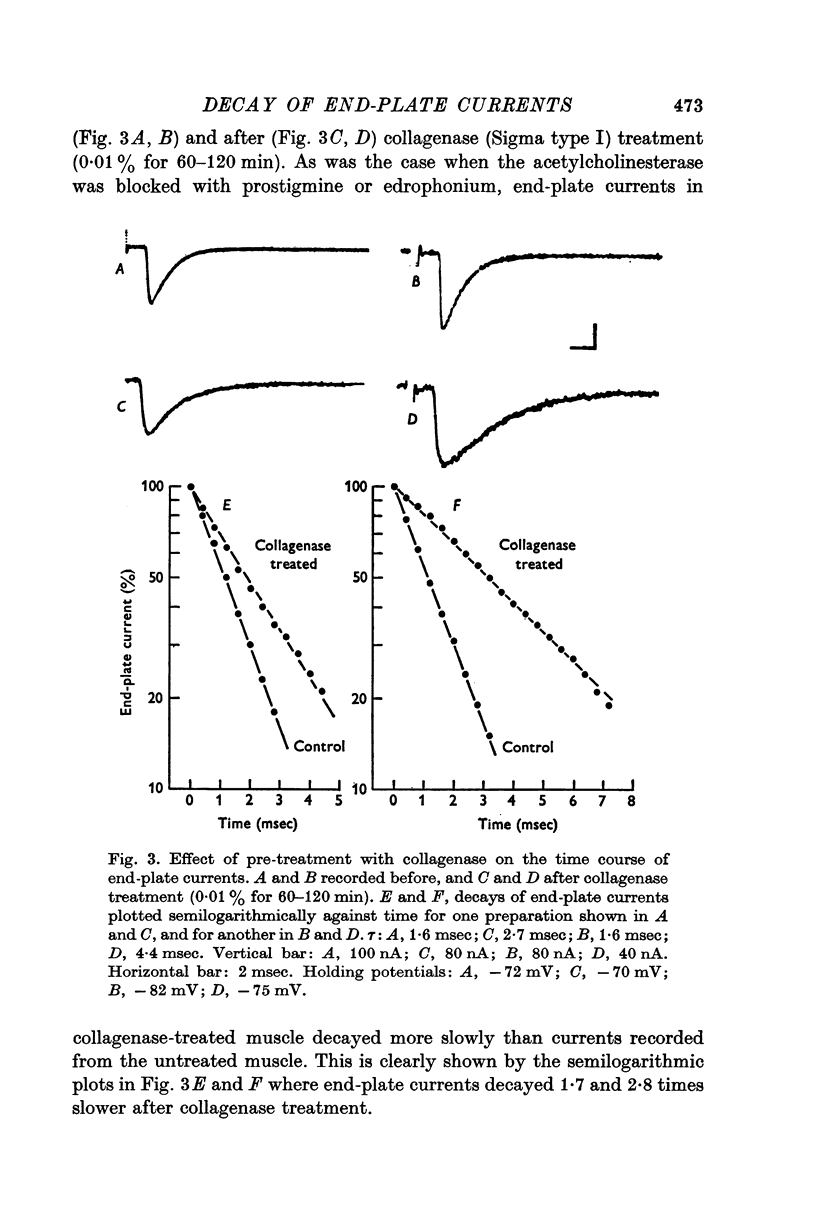
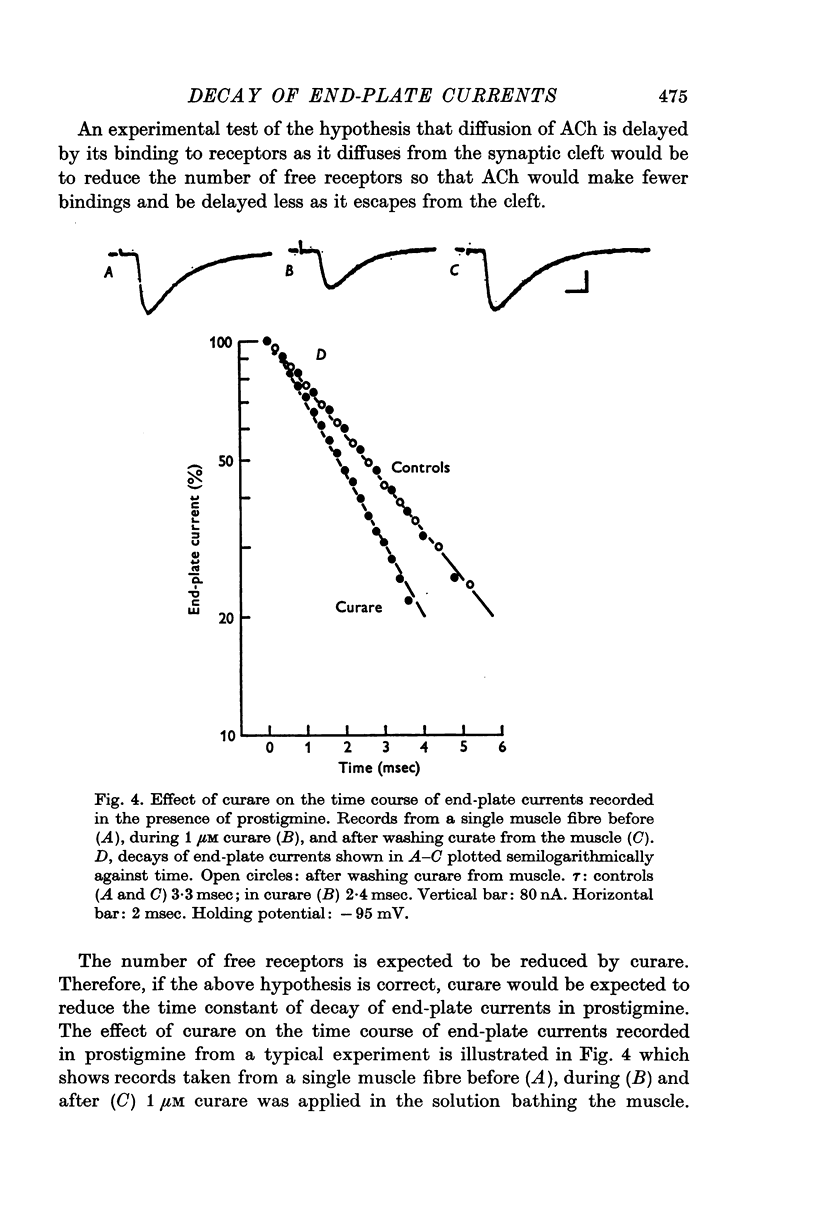
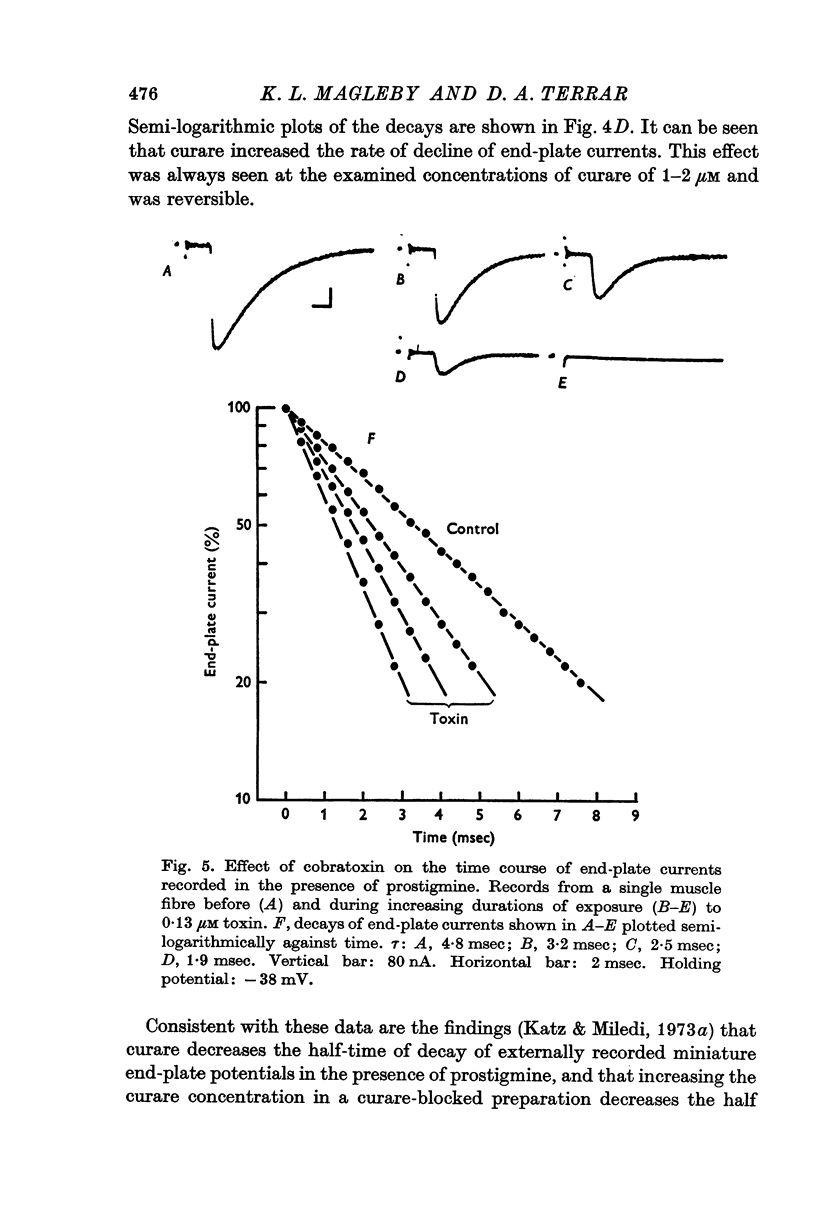
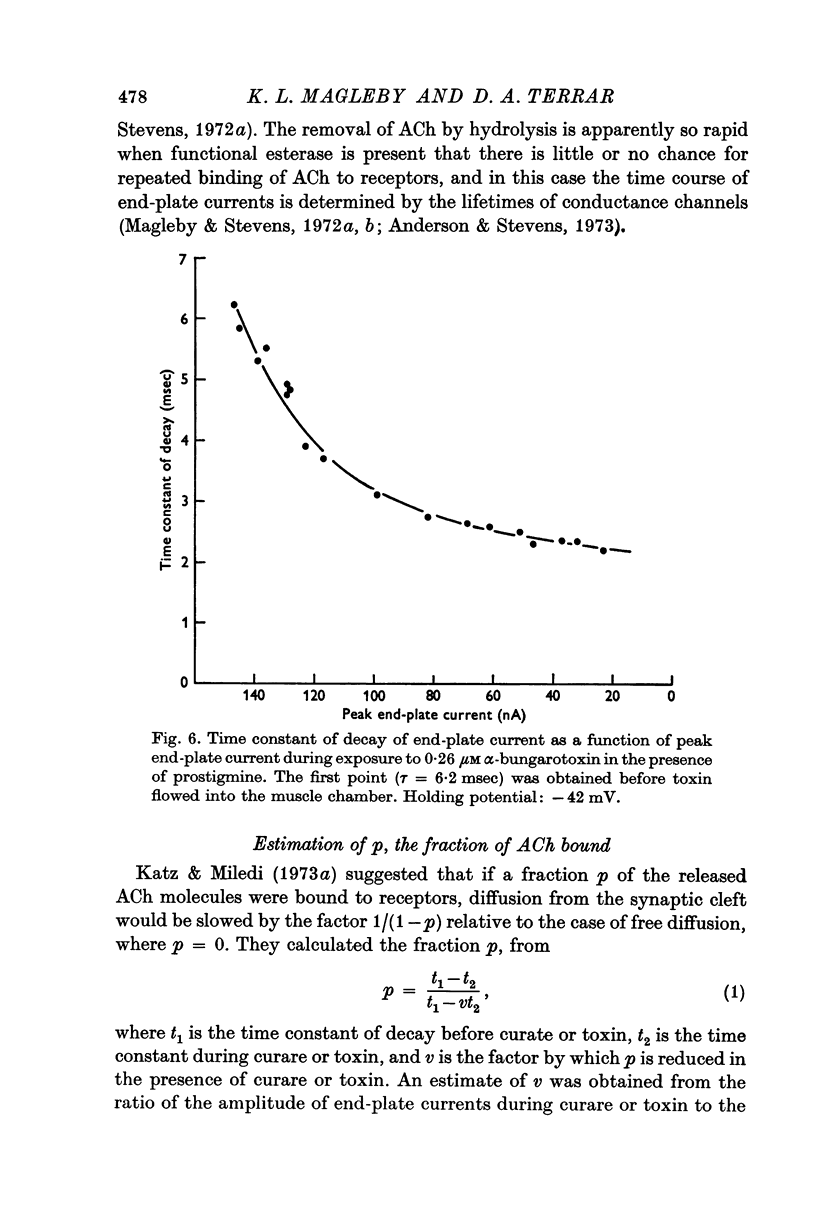
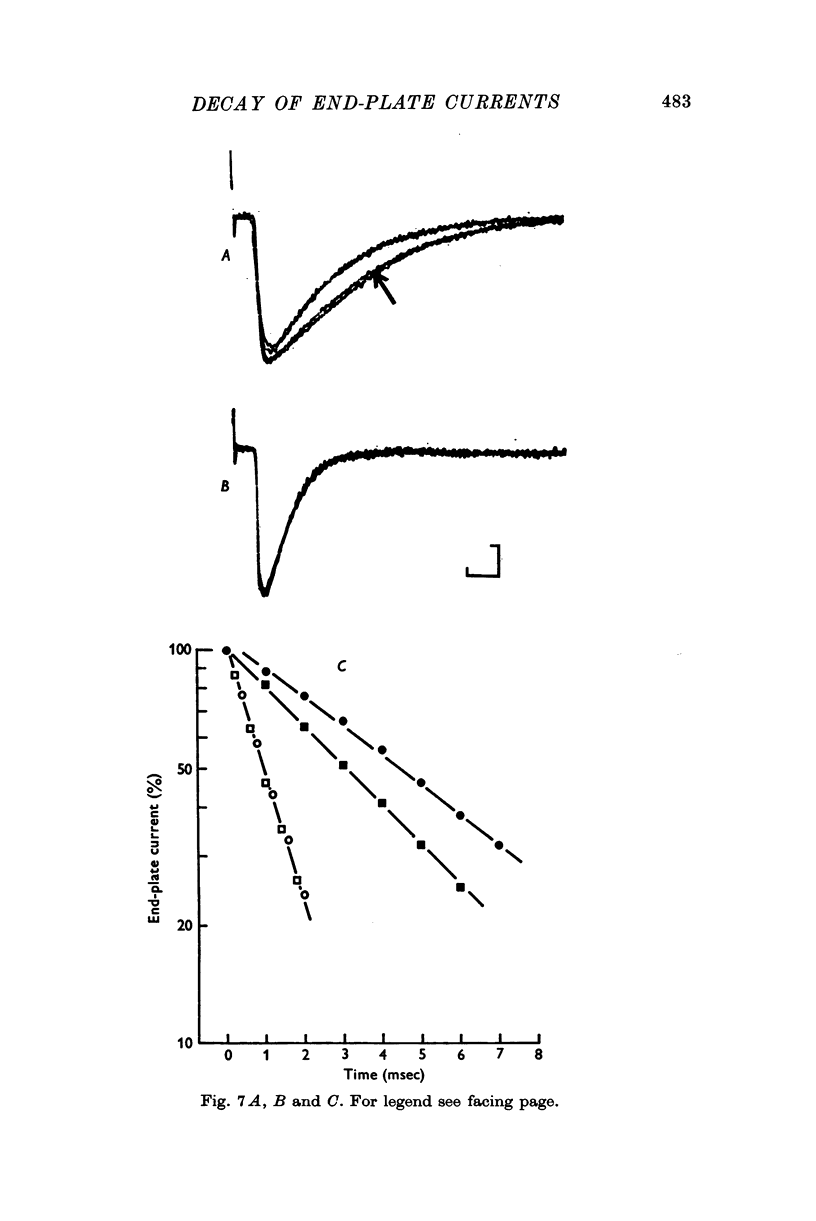
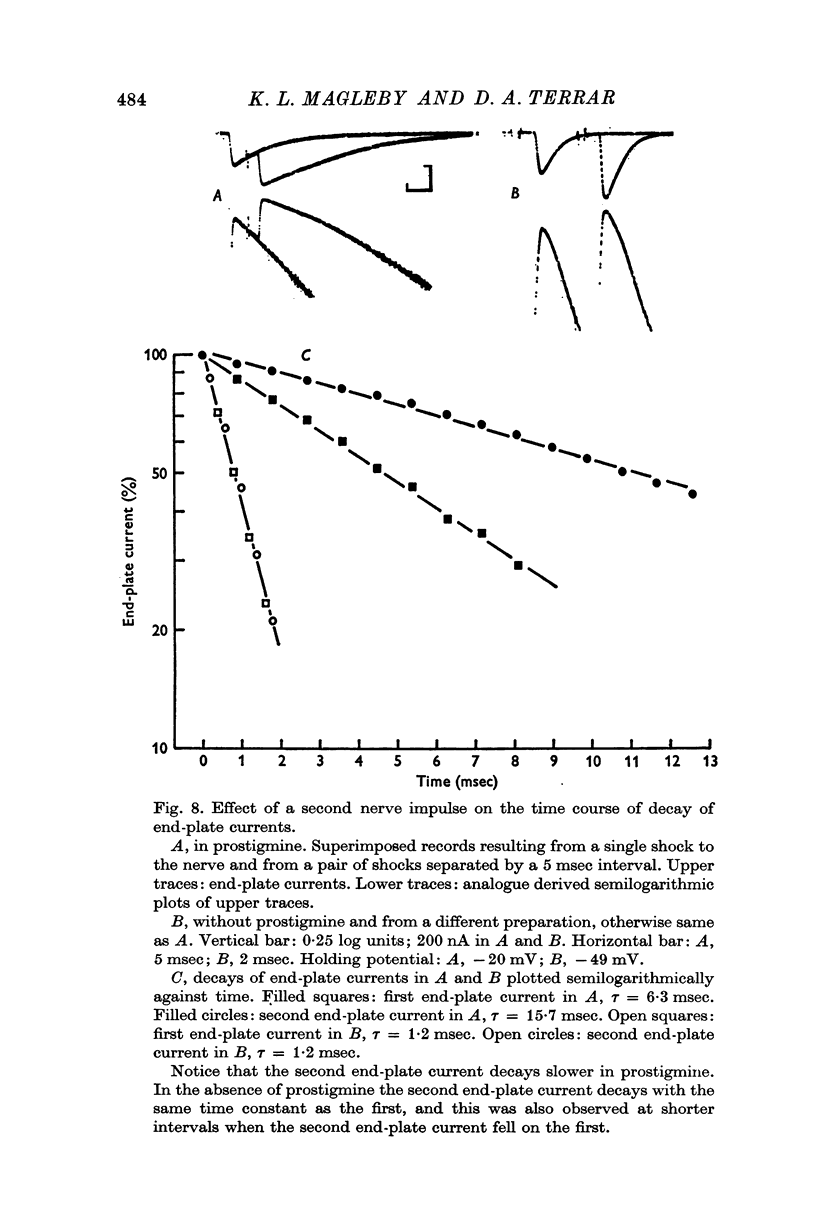
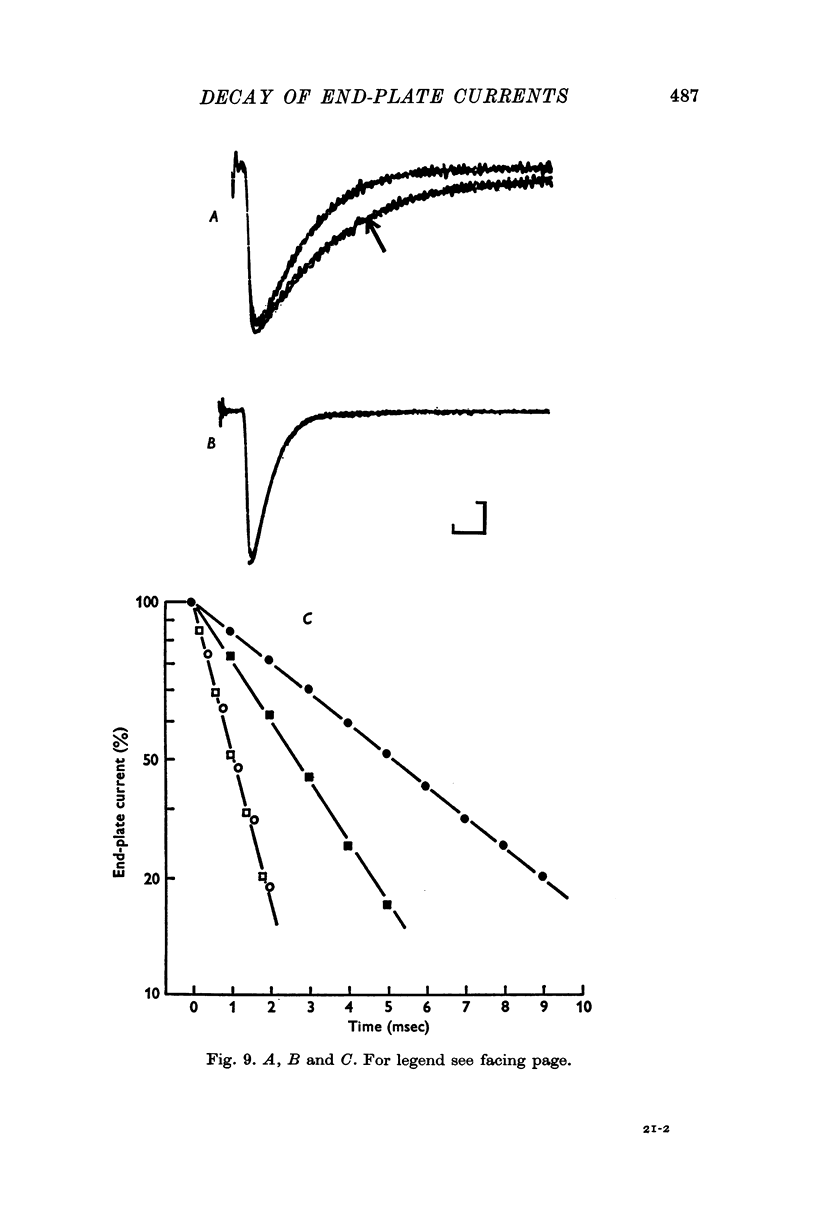
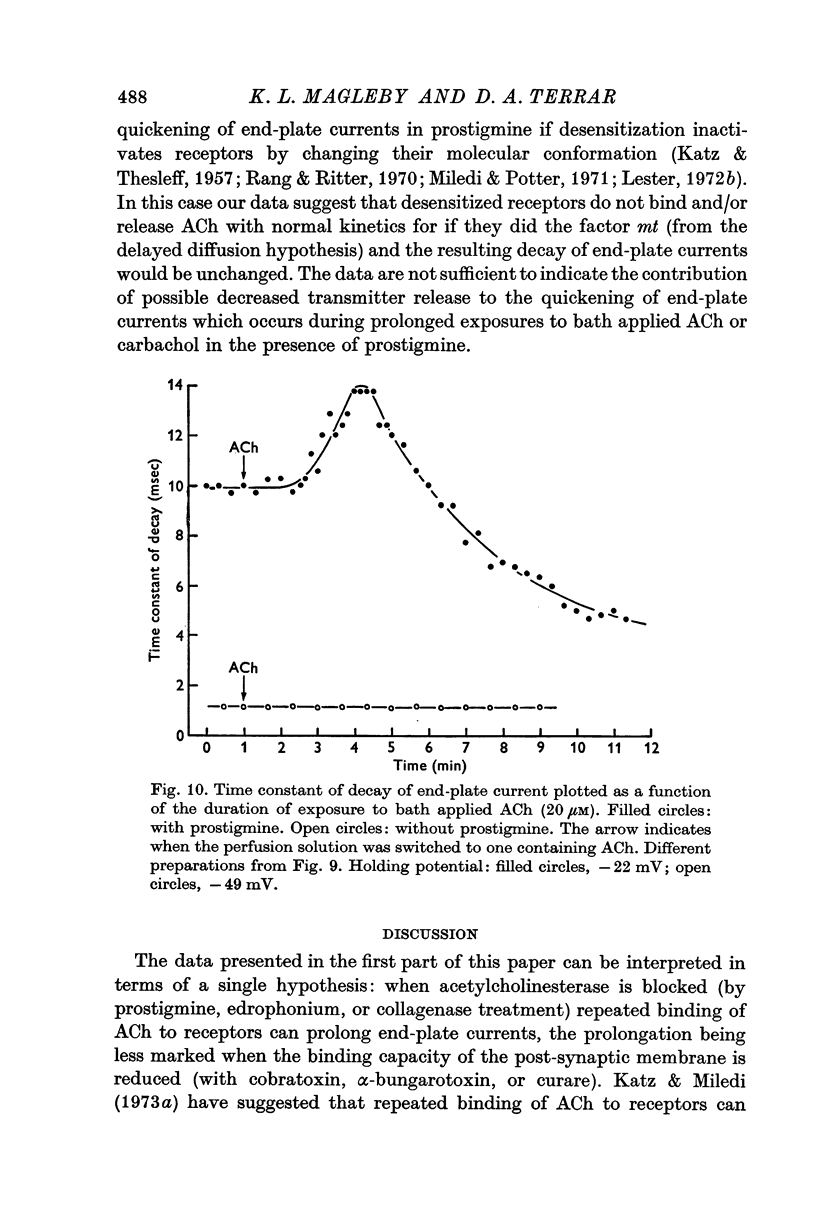
1. End-plate currents have been studied in gylcerol-treated frog sartorius nerve-muscle preparations with the voltage-clamp technique. 2. Adding the anticholinesterase prostigmine (3 muM) to the solution bathing the muscle caused a 2-7 (mean 3-3) times increase in the time constant of decay of end-plate currents. The anticholinesterase edrophonium (15 muM) also prolonged the time course of end-plate currents. 3. Pre-treatment of the preparation with collagenase, which leads to the removal of acetylcholinesterase in the synaptic cleft, prolongs the time course of end-plate currents. 4. Curare (1-2 muM), cobratoxin (0-13 muM), or alpha-bungarotoxin (0-13-0-26 muM) decreased the time constant of decay of end-plate currents in the presence of prostigmine. 5. These observations are consistant with the suggestion that repeated binding of acetylcholine (ACh) molecules to receptors as the ACh escapes from the synaptic cleft can contribute to the prolongation of end-plate currents which occurrs when acetylcholinesterase activity is eliminated. 6. Increasing the amount of transmitter released from the presynaptic nerve terminal leads to a prolongation of end-plate currents in the presence of prostigmine. 7. In the presence of prostigmine, the second of two end-plate currents (interval 2-10 msec) decays more slowly than the first. 8. ACh (1-40 muM) or carbachol (40 muM) applied in the solution bathing the muscle prolongs end-plate currents in the presence of prostigmine. 9. It is suggested on the basis of the observations described in paragraphs 6 to 8 that the time constant of decay of end-plate currents in the presence of prostigmine increases with increasing concentrations of ACh in the synaptic cleft. In the absence of prostigmine, increasing the concentration of ACh in the synaptic cleft did not change the time constant for decay of end-plate currents. 10. We interpret these results to suggest that ACh can have a cooperative action on receptors such that the association of ACh with one receptor (defined as binding a single ACh molecule) favours the binding or retention of ACh at other receptors. This implies that receptors can interact.
Full text
PDF


















Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albuquerque E. X., Sokoll M. D., Sonesson B., Thesleff S. Studies on the nature of the cholinergic receptor. Eur J Pharmacol. 1968 Aug;4(1):40–46. doi: 10.1016/0014-2999(68)90007-1. [DOI] [PubMed] [Google Scholar]
- Anderson C. R., Stevens C. F. Voltage clamp analysis of acetylcholine produced end-plate current fluctuations at frog neuromuscular junction. J Physiol. 1973 Dec;235(3):655–691. doi: 10.1113/jphysiol.1973.sp010410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beránek R., Vyskocil F. The effect of atropine on the frog sartorius neuromuscular junction. J Physiol. 1968 Mar;195(2):493–503. doi: 10.1113/jphysiol.1968.sp008470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Betz W., Sakmann B. "Disjunction" of frog neuromuscular synapses by treatment with proteolytic enzymes. Nat New Biol. 1971 Jul 21;232(29):94–95. doi: 10.1038/newbio232094a0. [DOI] [PubMed] [Google Scholar]
- Betz W., Sakmann B. Effects of proteolytic enzymes on function and structure of frog neuromuscular junctions. J Physiol. 1973 May;230(3):673–688. doi: 10.1113/jphysiol.1973.sp010211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowen J. M. Estimation of the dissociation constant of d-tubocurarine and the receptor for endogenous acetylcholine. J Pharmacol Exp Ther. 1972 Nov;183(2):333–340. [PubMed] [Google Scholar]
- CHANG C. C., LEE C. Y. ISOLATION OF NEUROTOXINS FROM THE VENOM OF BUNGARUS MULTICINCTUS AND THEIR MODES OF NEUROMUSCULAR BLOCKING ACTION. Arch Int Pharmacodyn Ther. 1963 Jul 1;144:241–257. [PubMed] [Google Scholar]
- CIANI S., EDWARDS C. THE EFFECT OF ACETYLCHOLINE ON NEUROMUSCULAR TRANSMISSION IN THE FROG. J Pharmacol Exp Ther. 1963 Oct;142:21–23. [PubMed] [Google Scholar]
- Chang C. C., Lee C. Y. Electrophysiological study of neuromuscular blocking action of cobra neurotoxin. Br J Pharmacol Chemother. 1966 Nov;28(2):172–181. doi: 10.1111/j.1476-5381.1966.tb01883.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Changeux J. P., Podleski T. R. On the excitability and cooperativity of the electroplax membrane. Proc Natl Acad Sci U S A. 1968 Mar;59(3):944–950. doi: 10.1073/pnas.59.3.944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connor J. A., Stevens C. F. Inward and delayed outward membrane currents in isolated neural somata under voltage clamp. J Physiol. 1971 Feb;213(1):1–19. doi: 10.1113/jphysiol.1971.sp009364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DEL CASTILLO J., ENGBAEK L. The nature of the neuromuscular block produced by magnesium. J Physiol. 1954 May 28;124(2):370–384. doi: 10.1113/jphysiol.1954.sp005114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DEL CASTILLO J., KATZ B. A comparison of acetylcholine and stable depolarizing agents. Proc R Soc Lond B Biol Sci. 1957 May 7;146(924):362–368. doi: 10.1098/rspb.1957.0017. [DOI] [PubMed] [Google Scholar]
- DEL CASTILLO J., KATZ B. Localization of active spots within the neuromuscular junction of the frog. J Physiol. 1956 Jun 28;132(3):630–649. doi: 10.1113/jphysiol.1956.sp005554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DEL CASTILLO J., KATZ B. Quantal components of the end-plate potential. J Physiol. 1954 Jun 28;124(3):560–573. doi: 10.1113/jphysiol.1954.sp005129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DEL CASTILLO J., KATZ B. Statistical factors involved in neuromuscular facilitation and depression. J Physiol. 1954 Jun 28;124(3):574–585. doi: 10.1113/jphysiol.1954.sp005130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ECCLES J. C., JAEGER J. C. The relationship between the mode of operation and the dimensions of the junctional regions at synapses and motor end-organs. Proc R Soc Lond B Biol Sci. 1958 Jan 1;148(930):38–56. doi: 10.1098/rspb.1958.0003. [DOI] [PubMed] [Google Scholar]
- ECCLES J. C., MacFARLANE W. V. Actions of anti-cholinesterases on endplate potential of frog muscle. J Neurophysiol. 1949 Jan;12(1):59–80. doi: 10.1152/jn.1949.12.1.59. [DOI] [PubMed] [Google Scholar]
- Eldefrawi M. E., Eldefrawi A. T. Cooperativities in the binding of acetylcholine to its receptor. Biochem Pharmacol. 1973 Dec 1;22(23):3145–3150. doi: 10.1016/0006-2952(73)90209-8. [DOI] [PubMed] [Google Scholar]
- FATT P., KATZ B. An analysis of the end-plate potential recorded with an intracellular electrode. J Physiol. 1951 Nov 28;115(3):320–370. doi: 10.1113/jphysiol.1951.sp004675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FATT P. The electromotive action of acetylcholine at the motor end-plate. J Physiol. 1950 Oct 16;111(3-4):408–422. doi: 10.1113/jphysiol.1950.sp004492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gage P. W., Eisenberg R. S. Action potentials without contraction in frog skeletal muscle fibers with disrupted transverse tubules. Science. 1967 Dec 29;158(3809):1702–1703. doi: 10.1126/science.158.3809.1702. [DOI] [PubMed] [Google Scholar]
- HIGMAN H. B., PODLESKI T. R., BARTELS E. APPARENT DISSOCIATION CONSTANTS BETWEEN CARBAMYLCHOLINE, DELTA-TUBOCURARINE AND THE RECEPTOR. Biochim Biophys Acta. 1963 Sep 24;75:187–193. doi: 10.1016/0006-3002(63)90597-3. [DOI] [PubMed] [Google Scholar]
- HUBBARD J. I. REPETITIVE STIMULATION AT THE MAMMALIAN NEUROMUSCULAR JUNCTION, AND THE MOBILIZATION OF TRANSMITTER. J Physiol. 1963 Dec;169:641–662. doi: 10.1113/jphysiol.1963.sp007286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall Z. W., Kelly R. B. Enzymatic detachment of endplate acetylcholinesterase from muscle. Nat New Biol. 1971 Jul 14;232(28):62–63. doi: 10.1038/newbio232062a0. [DOI] [PubMed] [Google Scholar]
- Howell J. N. A lesion of the transverse tubules of skeletal muscle. J Physiol. 1969 May;201(3):515–533. doi: 10.1113/jphysiol.1969.sp008770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JENKINSON D. H. The antagonism between tubocurarine and substances which depolarize the motor end-plate. J Physiol. 1960 Jul;152:309–324. doi: 10.1113/jphysiol.1960.sp006489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JENKINSON D. H. The nature of the antagonism between calcium and magnesium ions at the neuromuscular junction. J Physiol. 1957 Oct 30;138(3):434–444. doi: 10.1113/jphysiol.1957.sp005860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson D. H., Terrar D. A. Influence of chloride ions on changes in membrane potential during prolonged application of carbachol to frog skeletal muscle. Br J Pharmacol. 1973 Feb;47(2):363–376. doi: 10.1111/j.1476-5381.1973.tb08334.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KATZ B., THESLEFF S. A study of the desensitization produced by acetylcholine at the motor end-plate. J Physiol. 1957 Aug 29;138(1):63–80. doi: 10.1113/jphysiol.1957.sp005838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlin A. On the application of "a plausible model" of allosteric proteins to the receptor for acetylcholine. J Theor Biol. 1967 Aug;16(2):306–320. doi: 10.1016/0022-5193(67)90011-2. [DOI] [PubMed] [Google Scholar]
- Karlsson E., Arnberg H., Eaker D. Isolation of the principal neurotoxins of two Naja naja subspecies. Eur J Biochem. 1971 Jul 15;21(1):1–16. doi: 10.1111/j.1432-1033.1971.tb01433.x. [DOI] [PubMed] [Google Scholar]
- Katz B., Miledi R. Further observations on acetylcholine noise. Nat New Biol. 1971 Jul 28;232(30):124–126. doi: 10.1038/newbio232124b0. [DOI] [PubMed] [Google Scholar]
- Katz B., Miledi R. The binding of acetylcholine to receptors and its removal from the synaptic cleft. J Physiol. 1973 Jun;231(3):549–574. doi: 10.1113/jphysiol.1973.sp010248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz B., Miledi R. The effect of alpha-bungarotoxin on acetylcholine receptors. Br J Pharmacol. 1973 Sep;49(1):138–139. doi: 10.1111/j.1476-5381.1973.tb08278.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz B., Miledi R. The effect of atropine on acetylcholine action at the neuromuscular junction. Proc R Soc Lond B Biol Sci. 1973 Nov 27;184(1075):221–226. doi: 10.1098/rspb.1973.0046. [DOI] [PubMed] [Google Scholar]
- Katz B., Miledi R. The statistical nature of the acetycholine potential and its molecular components. J Physiol. 1972 Aug;224(3):665–699. doi: 10.1113/jphysiol.1972.sp009918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuba K., Albuquerque E. X., Barnard E. A. Diisopropylfluorophosphate: suppression of ionic conductance of the cholinergic receptor. Science. 1973 Aug 31;181(4102):853–856. doi: 10.1126/science.181.4102.853. [DOI] [PubMed] [Google Scholar]
- Kuba K., Tomita T. Effect of prostigmine on the time course of the end-plate potential in the rat diaphragm. J Physiol. 1971 Mar;213(3):533–544. doi: 10.1113/jphysiol.1971.sp009398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEVIN A. P., JANDORF B. J. Inactivation of cholinesterase by compounds related to neostigmine. J Pharmacol Exp Ther. 1955 Feb;113(2):206–211. [PubMed] [Google Scholar]
- Lass Y., Halevi Y., Landau E. M., Gitter S. A new model for transmitter mobilization in the frog neuromuscular junction. Pflugers Arch. 1973 Oct 17;343(2):157–163. doi: 10.1007/BF00585711. [DOI] [PubMed] [Google Scholar]
- Lester H. A. Blockade of acetylcholine receptors by cobra toxin: electrophysiological studies. Mol Pharmacol. 1972 Nov;8(6):623–631. [PubMed] [Google Scholar]
- Lester H. A. Vulnerability of desensitized or curare-treated acetylcholine receptors to irreversible blockade by cobra toxin. Mol Pharmacol. 1972 Nov;8(6):632–644. [PubMed] [Google Scholar]
- Magleby K. L., Stevens C. F. A quantitative description of end-plate currents. J Physiol. 1972 May;223(1):173–197. doi: 10.1113/jphysiol.1972.sp009840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magleby K. L., Stevens C. F. The effect of voltage on the time course of end-plate currents. J Physiol. 1972 May;223(1):151–171. doi: 10.1113/jphysiol.1972.sp009839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magleby K. L. The effect of tetanic and post-tetanic potentiation on facilitation of transmitter release at the frog neuromuscular junction. J Physiol. 1973 Oct;234(2):353–371. doi: 10.1113/jphysiol.1973.sp010349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallart A., Martin A. R. An analysis of facilitation of transmitter release at the neuromuscular junction of the frog. J Physiol. 1967 Dec;193(3):679–694. doi: 10.1113/jphysiol.1967.sp008388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miledi R., Potter L. T. Acetylcholine receptors in muscle fibres. Nature. 1971 Oct 29;233(5322):599–603. doi: 10.1038/233599a0. [DOI] [PubMed] [Google Scholar]
- Moody T., Schmidt J., Raftery M. A. Binding of acetylcholine and related compounds to purified acetylcholine receptor from Torpedo Californica electroplax. Biochem Biophys Res Commun. 1973 Aug 6;53(3):761–772. doi: 10.1016/0006-291x(73)90158-7. [DOI] [PubMed] [Google Scholar]
- OTSUKA M., ENDO M., NONOMURA Y. Presynaptic nature of neuromuscular depression. Jpn J Physiol. 1962 Dec 15;12:573–584. doi: 10.2170/jjphysiol.12.573. [DOI] [PubMed] [Google Scholar]
- Rang H. P. Drug receptors and their function. Nature. 1971 May 14;231(5298):91–96. doi: 10.1038/231091a0. [DOI] [PubMed] [Google Scholar]
- Rang H. P., Ritter J. M. On the mechanism of desensitization at cholinergic receptors. Mol Pharmacol. 1970 Jul;6(4):357–382. [PubMed] [Google Scholar]
- Steinbach A. B. A kinetic model for the action of xylocaine on receptors for acetylcholine. J Gen Physiol. 1968 Jul;52(1):162–180. doi: 10.1085/jgp.52.1.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinbach A. B. Alteration by xylocaine (lidocaine) and its derivatives of the time course of the end plate potential. J Gen Physiol. 1968 Jul;52(1):144–161. doi: 10.1085/jgp.52.1.144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TAKEUCHI A., TAKEUCHI N. Active phase of frog's end-plate potential. J Neurophysiol. 1959 Jul;22(4):395–411. doi: 10.1152/jn.1959.22.4.395. [DOI] [PubMed] [Google Scholar]
- THESLEFF S. Motor end-plate 'desensitization' by repetitive nerve stimuli. J Physiol. 1959 Oct;148:659–664. doi: 10.1113/jphysiol.1959.sp006314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WILSON I. B. The interaction of tensilon and neostigmine with acetylcholinesterase. Arch Int Pharmacodyn Ther. 1955 Dec 1;104(2):204–213. [PubMed] [Google Scholar]
- Weber M., Changeux J. P. Binding of Naja nigricollis (3H)alpha-toxin to membrane fragments from Electrophorus and Torpedo electric organs. II. Effect of cholinergic agonists and antagonists on the binding of the tritiated alpha-neurotoxin. Mol Pharmacol. 1974 Jan;10(1):15–34. [PubMed] [Google Scholar]



