Abstract
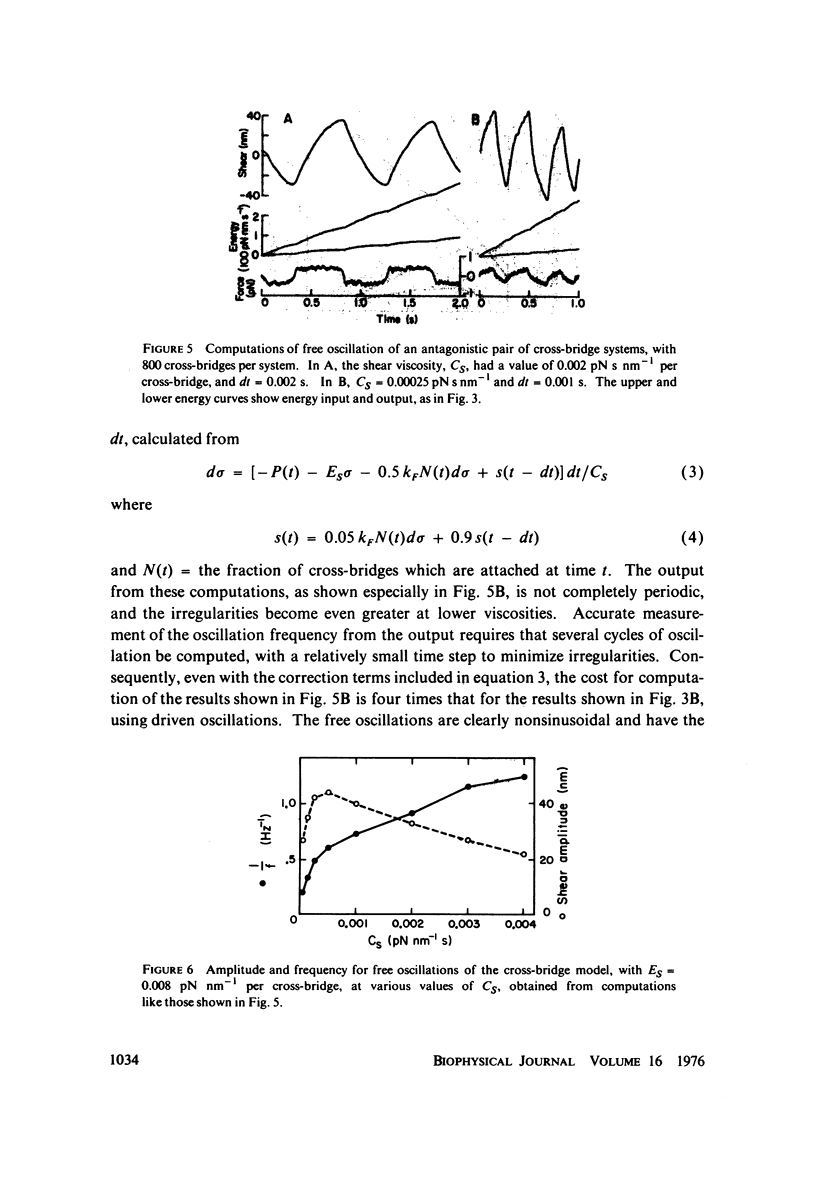
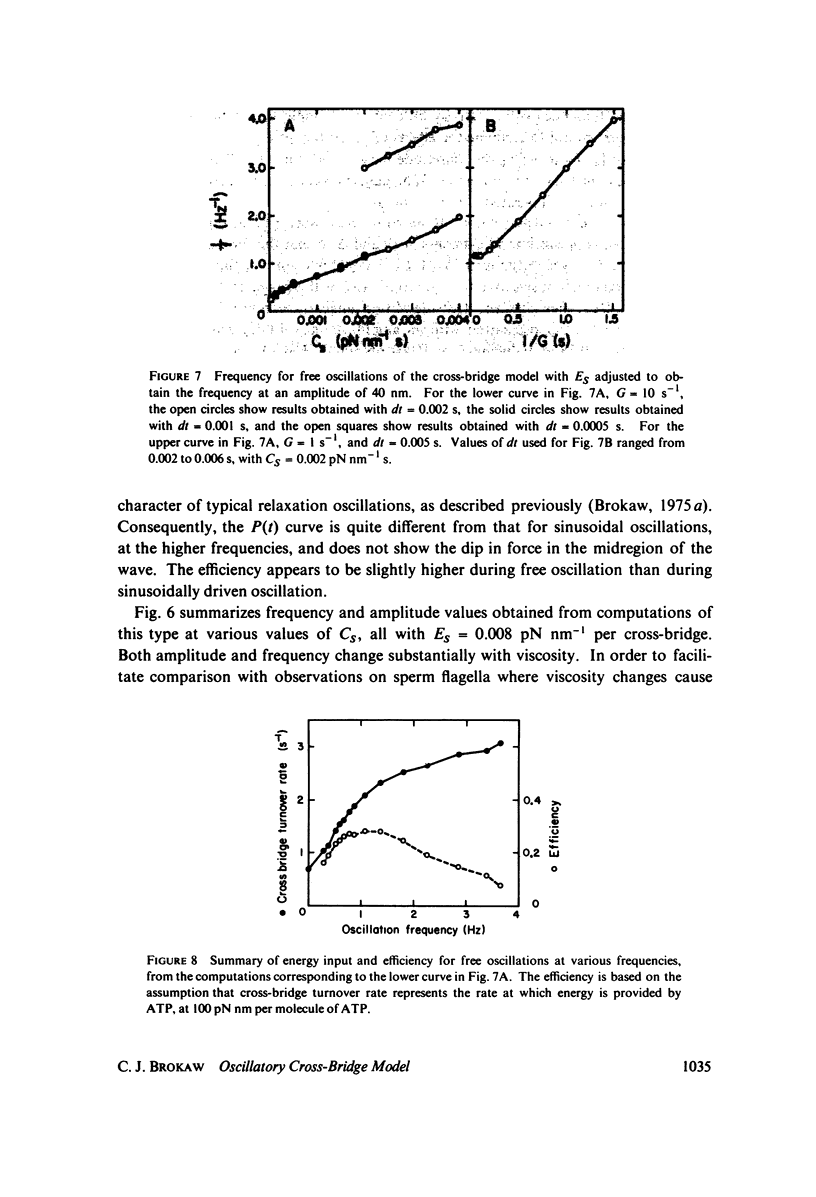
A stochastic computational method is used to examine the properties of a simple two-state cross-bridge model, of a type which has been shown previously to self-oscillate without requiring any feedback control of the active process. The force transients obtained with this model show the major features observed with oscillatory insect fibrillar flight muscle. The effects of viscosity and cross-bridge detachment rate on the frequency of oscillation of the model resemble the effects of viscosity and ATP concentration on flagellar oscillation, and the relationship between turnover rate and frequency of oscillation is also consistent with observations on flagella. However, the amplitude of oscillation of the model does not show the degree of frequency-independence which is typical of flagella.
Full text
PDF
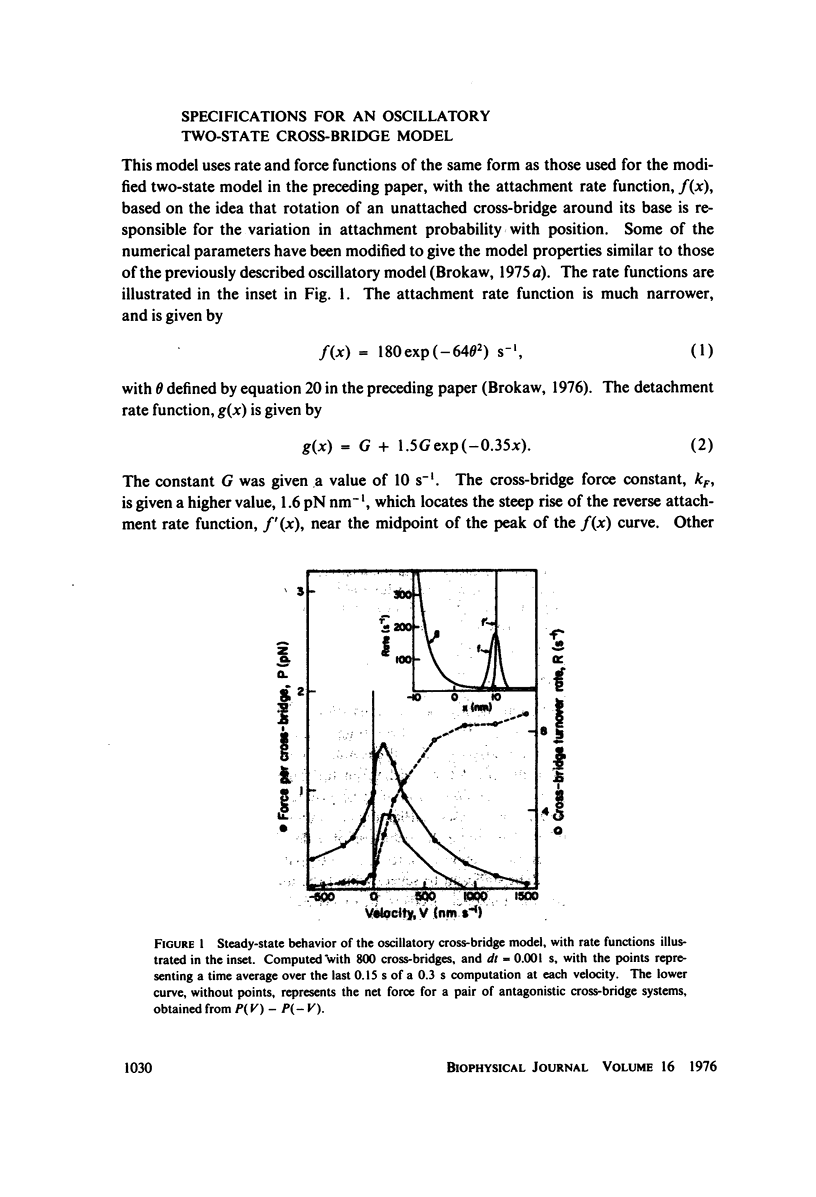
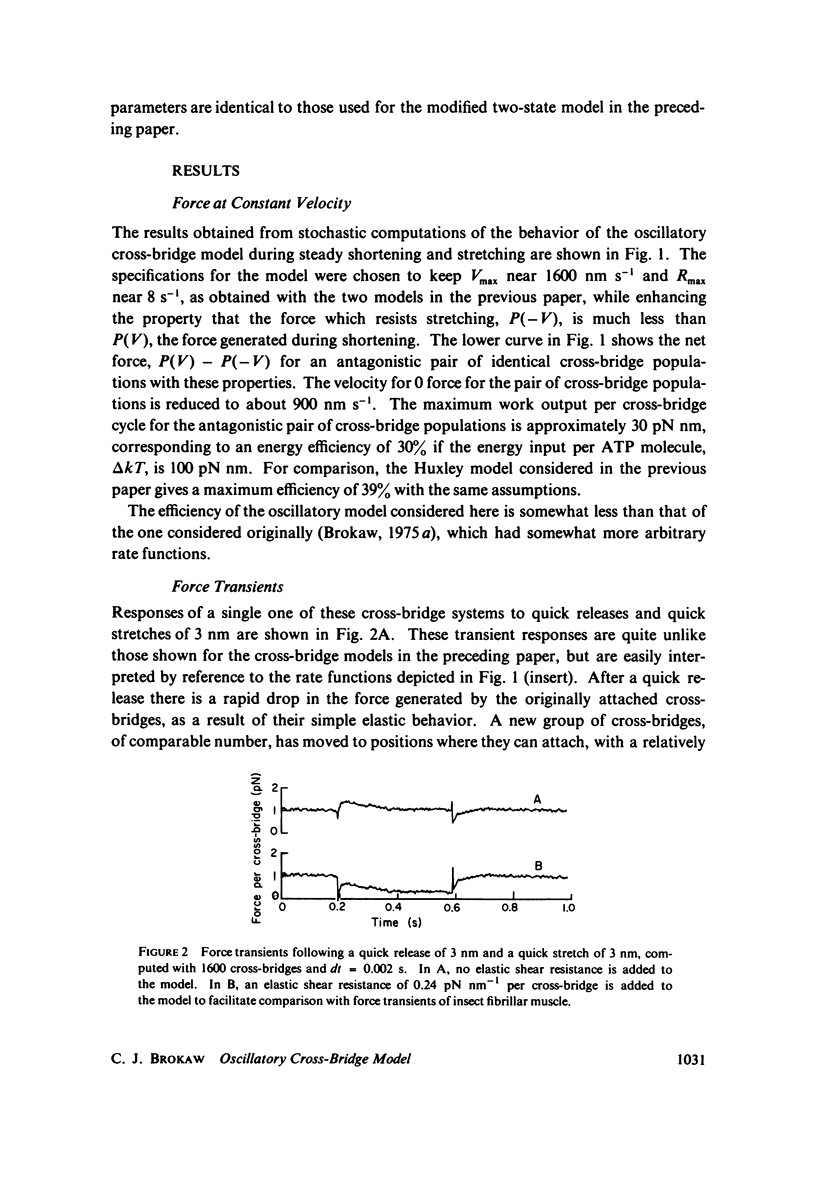
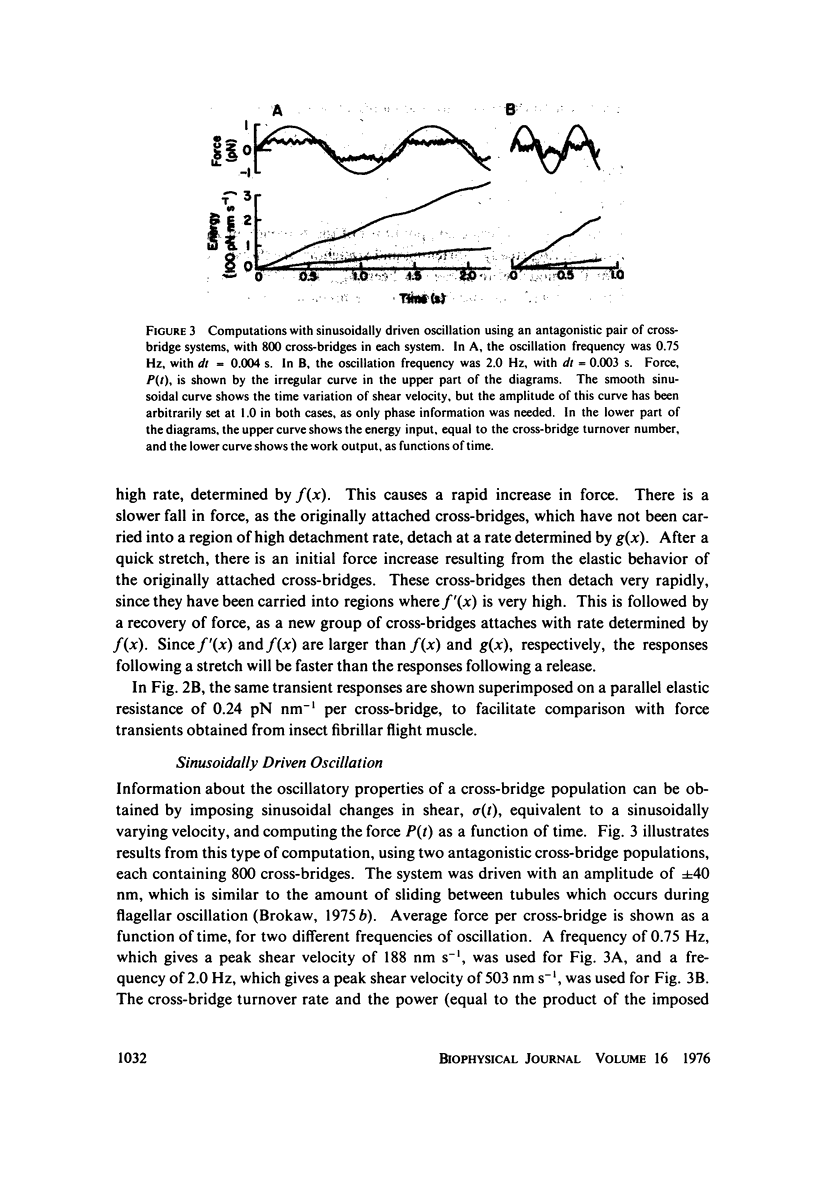
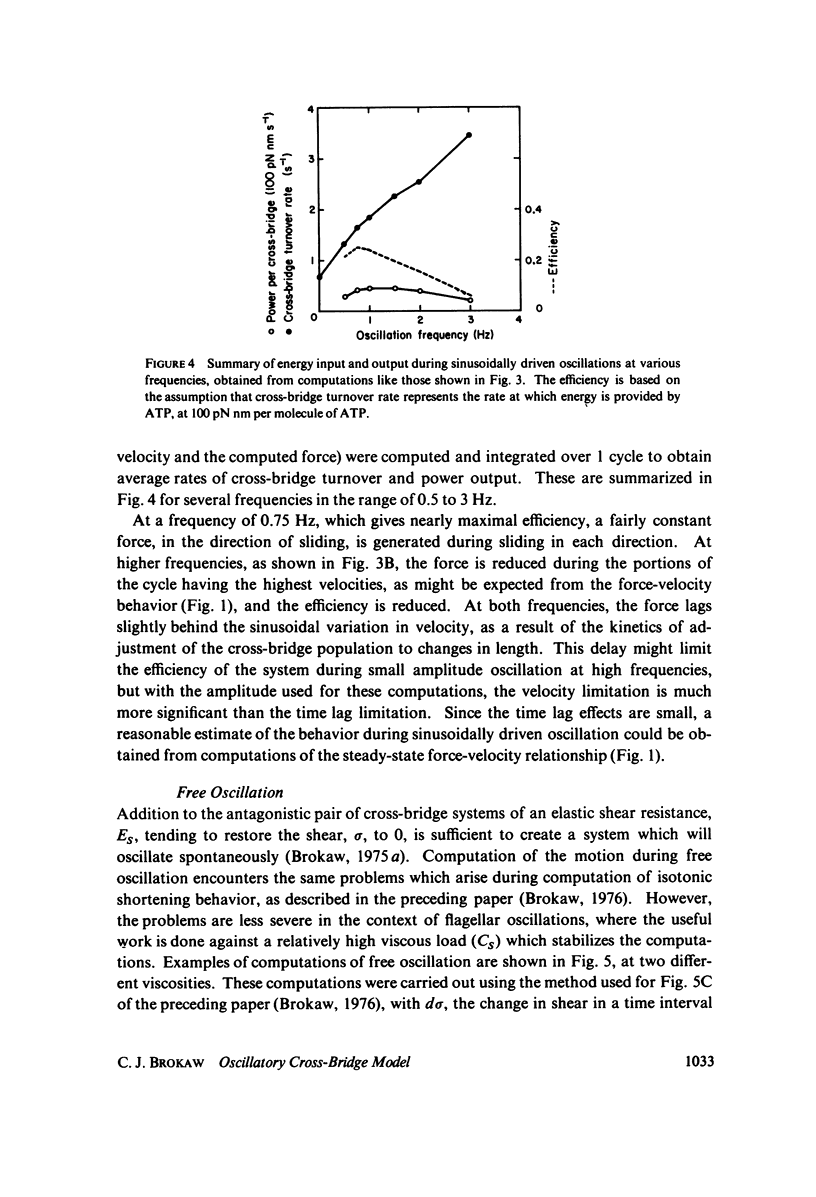






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amos L., Klug A. Arrangement of subunits in flagellar microtubules. J Cell Sci. 1974 May;14(3):523–549. doi: 10.1242/jcs.14.3.523. [DOI] [PubMed] [Google Scholar]
- Brokaw C. J. Bend propagation by a sliding filament model for flagella. J Exp Biol. 1971 Oct;55(2):289–304. doi: 10.1242/jeb.55.2.289. [DOI] [PubMed] [Google Scholar]
- Brokaw C. J., Benedict B. Mechanochemical coupling in flagella. I. Movement-dependent dephosphorylation of ATP by glycerinated spermatozoa. Arch Biochem Biophys. 1968 Jun;125(3):770–778. doi: 10.1016/0003-9861(68)90513-4. [DOI] [PubMed] [Google Scholar]
- Brokaw C. J., Benedict B. Mechanochemical coupling in flagella. II. Effects of viscosity and thiourea on metabolism and motility of Ciona spermatozoa. J Gen Physiol. 1968 Aug;52(2):283–299. doi: 10.1085/jgp.52.2.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brokaw C. J., Benedict B. Mechanochemical coupling in flagella. IV. Absence of coupling between metabolism and motility in Chaetopterus spermatozoa. J Mechanochem Cell Motil. 1976 Mar;3(3):201–205. [PubMed] [Google Scholar]
- Brokaw C. J. Computer simulation of flagellar movement. I. Demonstration of stable bend propagation and bend initiation by the sliding filament model. Biophys J. 1972 May;12(5):564–586. doi: 10.1016/S0006-3495(72)86104-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brokaw C. J. Computer simulation of movement-generating cross-bridges. Biophys J. 1976 Sep;16(9):1013–1027. doi: 10.1016/S0006-3495(76)85752-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brokaw C. J. Effects of increased viscosity on the movements of some invertebrate spermatozoa. J Exp Biol. 1966 Aug;45(1):113–139. doi: 10.1242/jeb.45.1.113. [DOI] [PubMed] [Google Scholar]
- Brokaw C. J. Effects of viscosity and ATP concentration on the movement of reactivated sea-urchin sperm flagella. J Exp Biol. 1975 Jun;62(3):701–719. doi: 10.1242/jeb.62.3.701. [DOI] [PubMed] [Google Scholar]
- Brokaw C. J. Molecular mechanism for oscillation in flagella and muscle. Proc Natl Acad Sci U S A. 1975 Aug;72(8):3102–3106. doi: 10.1073/pnas.72.8.3102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brokaw C. J., Rintala D. R. Computer simulation of flagellar movement. III. Models incorporating cross-bridge kinetics. J Mechanochem Cell Motil. 1975;3(2):77–86. [PubMed] [Google Scholar]
- Gibbons B. H., Gibbons I. R. Flagellar movement and adenosine triphosphatase activity in sea urchin sperm extracted with triton X-100. J Cell Biol. 1972 Jul;54(1):75–97. doi: 10.1083/jcb.54.1.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbons B. H., Gibbons I. R. Properties of flagellar "rigor waves" formed by abrupt removal of adenosine triphosphate from actively swimming sea urchin sperm. J Cell Biol. 1974 Dec;63(3):970–985. doi: 10.1083/jcb.63.3.970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein S. F. Form of developing bends in reactivated sperm flagella. J Exp Biol. 1976 Feb;64(1):173–184. doi: 10.1242/jeb.64.1.173. [DOI] [PubMed] [Google Scholar]
- Julian F. J. Activation in a skeletal muscle contraction model with a modification for insect fibrillar muscle. Biophys J. 1969 Apr;9(4):547–570. doi: 10.1016/S0006-3495(69)86403-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pringle J. W. The contractile mechanism of insect fibrillar muscle. Prog Biophys Mol Biol. 1967;17:1–60. doi: 10.1016/0079-6107(67)90003-x. [DOI] [PubMed] [Google Scholar]
- Thorson J., White D. C. Distributed representations for actin-myosin interaction in the oscillatory contraction of muscle. Biophys J. 1969 Mar;9(3):360–390. doi: 10.1016/S0006-3495(69)86392-7. [DOI] [PMC free article] [PubMed] [Google Scholar]


