Abstract
We identified a novel mouse gene, mRTVP-1, as a p53 target gene using differential display PCR and extensive promoter analysis. The mRTVP-1 protein has 255 amino acids and differs from the human RTVP-1 (hRTVP-1) protein by two short in-frame deletions of two and nine amino acids. RTVP-1 mRNA was induced in multiple cancer cell lines by adenovirus-mediated delivery of p53 and by gamma irradiation or doxorubicin both in the presence and in the absence of endogenous p53. Analysis of RTVP-1 expression in nontransformed and transformed cells further supported p53-independent gene regulation. Using luciferase reporter and electrophoretic mobility shift assays we identified a p53 binding site within intron 1 of the mRTVP-1 gene. Overexpression of mRTVP-1 or hRTVP-1 induced apoptosis in multiple cancer cell lines including prostate cancer cell lines 148-1PA, 178-2BMA, PC-3, TSU-Pr1, and LNCaP, a human lung cancer cell line, H1299, and two isogenic human colon cancer cell lines, HCT116 p53+/+ and HCT116 p53−/−, as demonstrated by annexin V positivity, phase-contrast microscopy, and in selected cases 4′,6′-diamidino-2-phenylindole staining and DNA fragmentation. Deletion of the signal peptide from the N terminus of RTVP-1 reduced its apoptotic activities, suggesting that a secreted and soluble form of RTVP-1 may mediate, in part, its proapoptotic activities.
The p53 tumor suppressor protein is induced by specific stress-related cell stimuli and in turn mediates important regulatory pathways involving cell cycle progression, apoptosis, senescence, DNA repair, cell differentiation, and angiogenesis (reviewed in references 5, 9, 11, 13, 16, and 27). Mutations in the p53 gene can lead to critical cellular malfunctions that contribute to malignant growth. Many human cancers, including colon, lung, breast, and prostate cancers have been shown to harbor p53 mutations (reviewed in reference 42). Numerous studies also support a specific metastasis suppressor role for p53 in prostate cancer (reviewed in reference 39). Initial experimental results using an in vivo mouse model of prostate cancer metastasis demonstrated that loss of p53 function can lead to the development of metastases that seed from relatively small numbers of cells within the primary tumor (38). Subsequent studies demonstrated that although p53 mutations in human primary prostate cancer tissues are heterogeneous and relatively infrequent, they occur at significant levels in metastatic disease, with mutation frequencies ranging from 21 to 30% in lymph node metastasis to >90% in androgen-insensitive disseminated disease (7, 19, 23, 28, 35, 43). This pattern of mutations suggests that only a few cells harboring p53 mutations in the primary tumor can seed metastases that clonally expand at distant sites. Indeed, studies have demonstrated that specific p53 mutations in primary tumors are clonally expanded in metastatic prostate cancer (35) and that a pattern of aberrant p53 expression in primary tumors termed clustered p53 staining has significant prognostic value in predicting recurrence following radical prostatectomy (28, 36, 43). An important question regarding the role of p53 metastasis suppression is the nature of the functional alterations that promote metastasis in prostate cancer cells containing p53 mutations. Since metastatic cells have increased capacity to survive under adverse conditions, these alterations most likely result, in part, from aberrant regulation of genes under the transcriptional control of p53 that have been shown to mediate apoptosis (9, 13, 20, 25, 34, 46).
Because of the established association between loss of p53 function and prostate cancer metastasis, we have pursued the identification, characterization, and functional analysis of p53 target genes in prostate cancer. It was also anticipated that some p53 target genes would be relevant for other malignancies within the context of either tumorigenesis or metastasis. We identified the mouse RTVP-1 (mRTVP-1; mnemonic for related to testes-specific, vespid, and pathogenesis proteins) mRNA as being upregulated by p53 in mouse prostate cancer cells. Although the human RTVP-1 (hRTVP-1) DNA was previously identified in specific brain tumors (21, 32), to our knowledge there are no published reports that have defined the functional significance of RTVP-1 expression. We demonstrate here that p53 upregulates mRTVP-1 by binding to a site located in intron 1 of mRTVP-1 and overexpression of mRTVP-1 results in apoptosis in multiple cancer cell lines, suggesting that mRTVP may play a role in inhibition of malignant growth and progression through its proapoptotic activities. Furthermore, our results indicate that the signal peptide in the N terminus of RTVP-1 is important for its proapoptotic activities.
MATERIALS AND METHODS
Cell lines and cell culture.
The mouse embryo fibroblast cell lines MEF p53+/+ and MEF p53−/−, mouse prostate cancer cell lines 148-1 PA (22, 38), RM-9 (3), 178-2 PA, 178-2 BMA (33, 37), and human prostate cancer cell line ND1 were grown in Dulbecco's modified Eagle's medium with 10% fetal bovine serum (FBS). Nontransformed mouse urogenital epithelial cell lines CUGE (3) and NUGE (38) were grown in serum-free Ham's F12 supplemented as previously described (3). The human colon cancer cell lines HCT116 p53+/+, HCT116 p53−/− (4) were grown in McCoy's 5A with 10% FBS. The human prostate cancer cell lines LNCaP, PC-3 M, TSU-Pr1, and PPC1 were grown in RPMI 1640 with 10% FBS; PC-3 cell lines were grown in Kaighn's modified Ham's F12 with 10% FBS; and DU145 cell lines were grown in modified Eagle medium (MEM) with 10% FBS. Human skin fibroblast cell line BUD8 was grown in MEM with 2 mM l-glutamine, Earle's balanced salt solution, 0.1 mM nonessential amino acid, 1.0 mM sodium pyruvate, and 10% FBS; human lung diploid cell line CCD11 in MEM, Earle's balanced salt solution, 0.1 mM nonessential amino acids, and 10% FBS; human osteosarcoma cancer cell line SAOS2 in McCoy's 5A with 15% FBS; human lung carcinoma cell line H1299 in RPMI 1640 with 5% FBS; and human normal umbilical vein endothelial cell line (HUVEC) in Kaighn's modified Ham's F12, 0.1 mg of heparin per ml, 0.03 to 0.05 mg of endothelial cell growth supplement per ml, and 10% FBS.
Gamma irradiation and Dox treatment.
Subconfluent cells were trypsinized, and a single cell suspension was prepared and subjected to gamma irradiation at a dose of 10 Gy. The irradiated cells were reseeded onto petri dishes and grown under regular conditions. RNA was prepared at 0, 6, 12, 24, and 48 h after gamma irradiation. For doxorubicin (Dox) treatment, subconfluent cells were treated with 1 μM Dox in regular medium and RNA was prepared at 0, 6, 12, 24, and 48 h after the treatment.
RNA isolation, Northern blotting, differential display PCR, and RT-PCR.
Total RNA was isolated from cell lines by using Ultraspec RNA kit (Biotecx). mRNA was directly purified from cultured cells using the PolyA tract mRNA Isolation System (Promega). Northern blot analysis was performed as described previously (31). Differential display PCR was performed essentially as described previously (31, 44) with RNA isolated from Adp53- or Adβgal-infected 148-1 PA cells. Briefly, one of a set of unique 10-mer deoxyoligonucleotide primers with an arbitrarily chosen sequence (CTGCTTGATG) was used for reverse transcription (RT) and as a both 5′ and 3′ primer for amplification by PCR. RT of mRNA was performed using the Perkin-Elmer/Cetus GeneAmp RNA PCR kit. Samples were separated on a nondenaturing 5% polyacrylamide gel (29:1) with 5% glycerol at 9 W for 18 h. The differentially displayed bands were excised from the gel, reamplified by PCR, and used to make a 32P-labeled probe for Northern blot analysis or cloned into TA cloning vector (pCR 2.0 vector; Invitrogen) for sequencing analysis. For RT-PCR analysis of RTVP-1 expression, RNA was isolated from gamma-irradiated or Dox-treated mouse and human cell lines at indicated times. For each RT-PCR, 200 ng of total RNA was reverse transcribed using oligo(dT)16 and Moloney murine leukemia virus reverse transcriptase (Perkin-Elmer). The cDNA was then amplified by PCR using HotStar Taq polymerase (QIAGEN) with the following primer pairs: 5′GTTGTTTGGGCAGATAGTTA3′ and 5′GGTTGGTTTTTGGGTTTT3′ for hRTVP-1; 5′TTCTTTGCTGACCTGCTGGATT3′ and 5′GCTTGCGACCTTGACCATCTTT3′ for hHPRT; 5′GTCTGCCCAAACAACCTGA3′ and 5′TGCCAGATATAACAAACGAGGAC3′ for mRTVP-1; and 5′CTGTCCCTGTATGCCTCTG3′ and 5′TGATGTCACGCACGATTT3′ for mouse β-actin. PCR conditions were 95°C for 15 min to activate HotStar Taq polymerase, followed by cycling at 94°C for 45 s, 57°C for 45 s, and 72°C for 45 s. The numbers of cycles were 32 for hRTVP-1, 25 for hHPRT, 37 for mRTVP-1, and 20 for mouse β-actin.
mRTVP-1 genomic clones and reporter constructs.
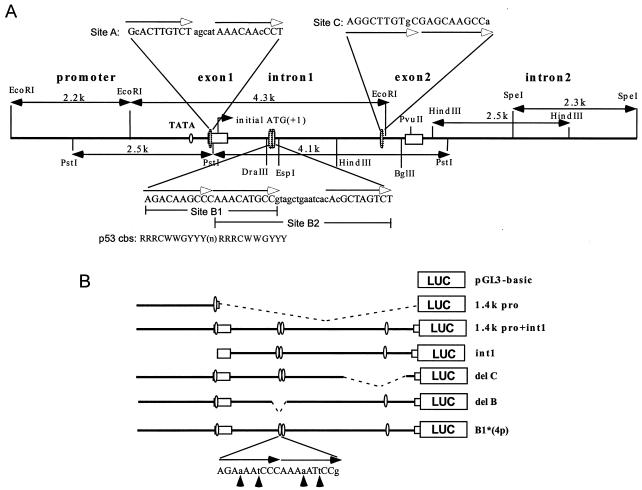
A 500-nucleotide (nt) probe from mRTVP-1 cDNA (5′ end to the AccI site) was used for PAC library screening. One positive clone (163K10) was restriction mapped, digested, and subcloned into pBluescript II. DNA sequencing was performed using an ABI prism 310 automatic sequencer. An ∼11-kb genomic sequence, obtained by aligning six overlapping subclones, included 3.6 kb upstream of the initial ATG (+1), exon1 (325 nt), intron 1 (3.2 kb), exon 2 (242 nt), and intron 2 (4.6 kb) (see schematic in Fig. 4A). The transcription initiation site was determined as the first nucleotide in cDNA clones from a 148-1 PA library.
FIG. 4.
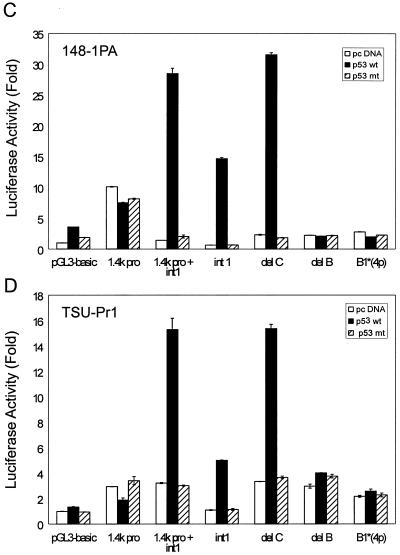
Schematic of genomic organization of mRTVP-1 and identification of p53 binding sites. (A) mRTVP-1 genomic sequence identified in clone 163K10 of the mouse RPCI-21 PAC library (26). The four potential p53 binding sites are located in exon 1 (site A) and intron 1 (sites B1, B2, and C). The p53 consensus binding site (cbs) (8) is shown under the B1 site sequence. (B) Schematic of luciferase reporter constructs as detailed in Materials and Methods. In B1*(4p), the C and G in the core sequence of both half-sites were converted to a and t as indicated by arrows. Luciferase activities with 148-1 PA (C) and TSU-Pr1 (D) were determined 24 h after transfection of mRTVP-1-luciferase reporter constructs along with a plasmid expressing wild-type p53 (2) (solid boxes), mutant p53 (2) (hatched boxes), or a control plasmid (pcDNA) (open boxes), as well as pCMVβ-gal for standardization. Luciferase activities were expressed as fold of the activity in the cells transfected by pGL3-basic and pcDNA. Error bars represent standard deviations.
An EcoRI-PstI fragment containing a 1.4-kb promoter and 79-nt exon 1 was ligated into the luciferase reporter vector pGL3-basic (Promega) to generate 1.4k pro-Luc. A PstI-PvuII fragment with 246 nt of exon1, 3.2-kb intron 1, and 72-nt exon 2 was fused in frame to the luciferase gene to produce int1-Luc. The 1.4-kb (EcoRI-PstI) promoter fragment was inserted into int1-Luc to generate 1.4k pro+int1-Luc. Luciferase reporters with deletions of the p53 binding sites B1+B2 or C were generated from 1.4k pro+int1-Luc by restriction enzyme digestion. Four point mutations were introduced into both half-sites of the B1 p53 binding core sequence by changing C to A and G to T by PCR using four primer pairs: primer 1, 5′aATtCCGTAGCTGAATCACACGCTAGTCT3′; primer 2, 5′ATAGCTCAACATCCTTAGCC3′; primer 3, 5′ATTCAGCTACGGaATtTTTGGGaTTtTCT3′; primer 4, 5′CCTGTTAATATTTGTGAGGTTTCTT3′ (lowercase letters indicate mismatched nucleotides). Two PCR fragments were amplified with primers 1 + 2 or primers 3 + 4. The two overlapped PCR fragments were then annealed and extended, and they served as a template for another round of PCR using primer 2 and primer 4. The resulting 700-nt PCR product was used to produce a 191-nt DraIII-EspI restriction fragment containing a mutated B1 site that replaced the wild-type sequence in 1.4k pro+int1-Luc to generate B1*(4p)-Luc.
To test the B1 and B2 sites independently for their p53 binding abilities, oligonucleotide pairs 5′AGACAAGCCCAAACATGCCg3′ and 5′cGGCATGTTTGGGCTTGTCT3′ for B1 and 5′AAACATGCCgtagctgaatcacAcGCTAGTCT3′ and 5′AGACTAGCgTgtgattcagctacGGCATGTTT3′ for B2 were annealed and then subcloned into the SmaI site of a luciferase reporter vector with a minimal simian virus 40 promoter (pGL3-promoter; Promega). The clones containing a single copy or concatemerized dual copies of the binding site were selected and designated B1(1x)-pro-Luc, B1(2x)-pro-Luc, B2(1x)-pro-Luc, and B2(2x)-pro-Luc. An oligonucleotide pair with the p53 consensus binding site (5′cgagAGACATGCCCAGGCATGCCT3′ and 5′AGGCATGCCTGGGCATGTCTctcg3′) was also annealed and inserted into pGL3 promoter luciferase, and it was termed p53cbs. All constructs were confirmed by DNA sequencing.
Luciferase assay.
One microgram of luciferase reporter construct, 0.1 μg of wild-type or mutant pCMVp53-expressing vectors (2) or control empty vector pcDNA3.1(+) and 0.2 μg of pCMVβ-gal were cotransfected into 148-1 PA or TSU-Pr1 cells using Lipofectamine (Life Technologies). Luciferase activity assays were performed 24 h after transfection and standardized to the cotransfected β-galactosidase as previously described (17, 40). Luciferase activity was expressed as fold of control (control is pcDNA with pGL3basic- or pGL3promoter-Luciferase).
Electrophoretic mobility shift assay (EMSA).
148-1 PA cells (p53 null) were transfected with pCMVp53 (wild type) or pCMVβgal. Nuclear extracts were prepared as described by Furukawa et al. (10). Briefly, 24 h after transfection, cells were collected, resuspended in an ice-cold solution containing 10 mM HEPES-NaOH (pH 7.9), 10 mM KCl, 0.75 mM spermidine, 0.15 mM spermine, 0.2 mM EDTA, 0.2 mM EGTA, 0.5 mM dithiothreitol, and 0.5 mM phenylmethylsulfonyl fluoride, and incubated on ice for 15 min. The cells were then mechanically broken by passing 10 times through a 25-gauge needle, and the lysate was centrifuged. The resulting nuclear pellet was resuspended in an ice-cold solution containing 20 mM HEPES-NaOH (pH 7.9), 0.4 M NaCl, 0.75 mM spermidine, 0.15 mM spermine, 0.2 mM EDTA, 0.2 mM EGTA, 0.5 mM dithiothreitol, 0.5 mM phenylmethylsulfonyl fluoride, and 25% (vol/vol) glycerol, incubated for 30 min on ice with frequent gentle mixing, and then centrifuged for 20 min at 4°C to remove insoluble material. The resulting nuclear extract was stored at −80°C. Ten micrograms of nuclear extract protein was incubated for 30 min at 37°C in a binding buffer containing 10 mM Tris-HCl (pH 7.5), 50 mM NaCl, 1 mM MgCl2, 1 mM EDTA, 8% glycerol, 1 mM dithiothreitol, 0.1 μg of poly(dI-dC), and ∼10 pM γ-32P-labeled oligonucleotide probe with a final volume of 20 μl in the absence or presence of cold probe competitors at a 1,000-fold excess (1,000×). The oligonucleotide probes were prepared from the following pairs of oligonucleotides: 5′tagAGACAAGCCCAAACATGCCGtagctgaatcacacgct3′ and 5′agcgtgtgattcagctaCGGCATGTTTGGGCTTGTCTcta3′ for B1; 5′cccAAACATGCCGtagctgaatcacACGCTAGTCTtat3′ and 5′ataAGACTAGCGTgtgattcagctaCGGCATGTTTggg3′ for B2; 5′cgagAGACATGCCCAGGCATGCCT3′ and 5′AGGCATGCCTGGGCATGTCTctcg3′ for consensus p53 binding site (14). (The underlined sequences are presumptive p53 binding site sequences, and uppercase letters indicate the half-sites of the p53 binding site sequence.) The oligonucleotide pairs (equal moles of each strand) were annealed and 5′ end labeled with [γ-32P]ATP using T4 polynucleotide kinase. The reaction mixtures were applied to a 4% nondenaturing polyacrylamide gel, and protein-DNA complexes were separated from free probe by electrophoresis at 100 V for 45 min in 1× Tris-borate-EDTA buffer. For the supershift assay, the nuclear extract was preincubated with 1 μg of mouse monoclonal antibody specific for p53 (DO1 [catalog no. sc-126]; Santa Cruz) for 30 min at room temperature before addition of γ-32P-labeled oligonucleotide probe.
Generation of mRTVP-1 antibody and Western blotting.
A polypeptide representing amino acid residues 27 to 43 of the hRTVP-1 sequence, DIENEDFIKDCVRIHNK, was used to immunize rabbits. The anti-RTVP-1 serum was affinity purified and characterized by Western blotting as described previously (40). Antibodies against mouse p53 (CM5), human p53 (DO7), or human and mouse p21 (SX118) were from Novocastra Laboratories Ltd or BD Pharmingen. The antibody for β-actin was from Sigma. To monitor the loading of protein lysates, the transferred membranes for Western blotting analysis were stained with Ponseau S before blocking with Tris-buffered saline containing 5% nonfat milk and 0.1% Tween 20.
Apoptosis analysis.
Adenoviral vectors Adβgal and AdmRTVP-1 were generated as previously described (22). Cells were infected with Adβgal or AdmRTVP-1 in serum-free medium for 3 h at a multiplicity of infection of 100, after which the medium was replaced with fresh complete medium and the cells were incubated for the indicated time period. Annexin V binding and flow cytometry were carried out as described previously (40). Apoptotic morphology was analyzed with phase-contrast or fluorescence microscopy following incubation with 0.2 μg of 4′,6′-diamidino-2-phenylindole (DAPI) per ml. DNA fragmentation analysis was performed with a Cell Death ELISA (enzyme-linked immunosorbent assay) kit (Boehringer Mannheim) according to the manufacturer's protocol, with the following modification. Cells were infected in six-well plates and continued to grow for the indicated time. After centrifugation of plates at 200 × g for 10 min, media were removed by aspiration, and the cells were lysed in 3 ml of lysis buffer at room temperature for 30 min. Two hundred microliters of lysis buffer containing cytosolic DNA fragments were transferred from each well to a microcentrifuge tube and centrifuged at 1,500 × g for 5 min. Triplicate samples of 20 μl were placed in a 96-well plate for analysis. DNA fragmentation was quantified by measuring absorbance at 405 nm with a reference wavelength at 490 nm. Data presented are representative of three or more independent experiments.
Construction of full-length RTVP-1 and signal peptide deletion mutants.
Full-length mRTVP-1 cDNA was obtained from a 148-1PA library by the digestion with SpeI and XbaI and was inserted into pcDNA3.1. Full-length hRTVP-1 was generated by PCR and inserted into the EcoRI site of pcDNA3.1 (+). An mRTVP-1 construct lacking the signal peptide sequence (mRTVP-1 Δ2-17) was generated by PCR amplification with a sense primer comprising a BamHI site, a Kozak sequence, and nt 203 to 220 corresponding to 18 to 23 amino acids (aa) of mRTVP-1 (see Fig. 3) (CGGGATCCAGAGAGACCATGTCTTCATTTACAGCAAGC) and an antisense primer including an EcoRI site and nt 926 to 944 (GGAATTCAGTTGTCCCCCAACCA). Similarly, to generate an mRTVP-1 construct with a partial deletion of the signal peptide sequence (mRTVP-1 Δ2-9), a sense primer containing a BamHI site, a Kozak sequence, and nt 179 to 196 corresponding to 10 to 15 aa (CGGGATCCAGAGAGACCATGTGGATGGCTTCGTCTGTG) was used with the same antisense primer as for mRTVP-1 Δ2-17. The hRTVP-1 construct lacking the signal peptide sequence (hRTVP-1 Δ2-21) was generated by using a sense primer comprising a BamHI site, a Kozak sequence, and nt 192 to 212 corresponding to 22 to 28 aa of hRTVP-1 (see Fig. 3) (CGGGATCCAGACAAAGCATGGCAAATATTTTGCCAGATATC) together with an antisense primer including a stop codon and nt 923 to 944 with alteration of a nucleotide after the stop codon to introduce an EcoRI site (CTTTCCTGAATTcTATTAGTCC) (the lowercase letter indicates the altered nucleotide). To generate an hRTVP-1 construct with a partial deletion of signal peptide (hRTVP-1 Δ2-6), a sense primer consisting of a BamHI site, a Kozak sequence, and nt 147 to 167 corresponding to 7 to 13 aa of hRTVP-1 (CGGGATCCAGACAAAGCATGACAATAGCCTGGATGGTTTCT) was used with the same antisense primer. The PCRs were performed using vent DNA polymerase (New England Biolabs). Cloned hRTVP-1 cDNA from human normal prostate and mRTVP-1 cDNA from the 148-1PA library were used as templates. The PCR fragments were then digested with BamHI and EcoRI and inserted into pcDNA3.1 (+). All constructs were confirmed by DNA sequencing, purified, and transfected into TSU-Pr1 cells with Lipofectamine Plus reagent.
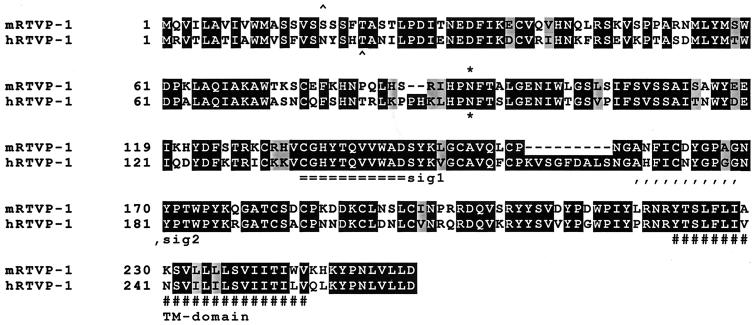
FIG. 3.
Comparison of deduced protein sequences for mouse and human RTVP-1. Identical amino acids are enclosed in dark boxes, conserved amino acid substitutions are lightly shaded, and gaps in the alignment are indicated by a dash. A potential N glycosylation site is indicated by an asterisk. The putative signal sequence precedes a cleavage site indicated by the symbol ∧. Two conserved regions known as extracellular protein signature motifs 1 and 2 (sig1 and sig2) and a transmembrane domain (TM) are indicated. The human RTVP-1 nucleotide and deduced protein sequences have been previously reported (32).
RESULTS
RTVP-1 is upregulated by p53, gamma irradiation, and Dox.
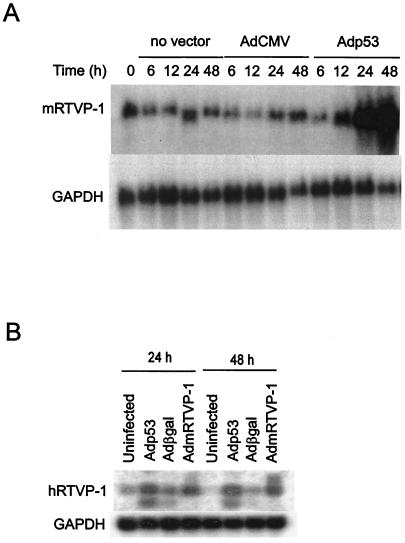
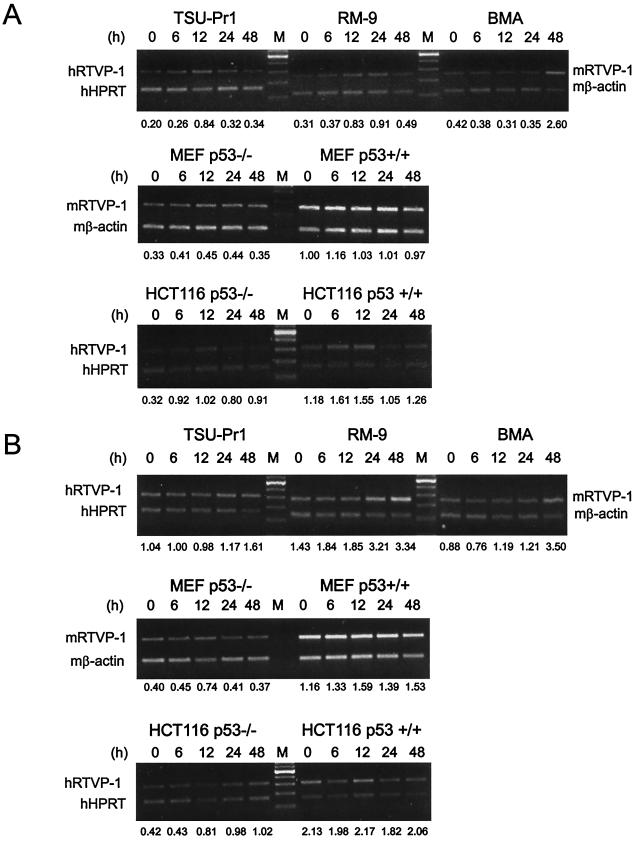
To identify prostate cancer-related genes under the transcriptional regulation of p53, we established a model system using adenoviral vector delivery of wild-type p53 compared to β-galactosidase or an empty cassette together with differential display PCR in the p53-null mouse prostate cancer cell line 148-1PA (38). Using this approach, we isolated numerous sequences that were known to be under p53 control including cyclin G, epoxide hydrolase, and MDM2 (not shown). In addition, we isolated genes that had not been previously associated with p53 regulation. One of the sequences that we identified encoded the mouse homologue for RTVP-1 (21, 32). The RTVP-1 cDNA was originally cloned from human glioma tissue and was subsequently reported to be expressed in differentiated macrophages (12). Using the differential display PCR fragment as a probe, we determined that mRTVP-1 mRNA (∼1.1-kb transcript) was induced at 24 and 48 h after Adp53 infection in 148-1PA cells (Fig. 1A). Additional studies demonstrated that hRTVP-1 mRNAs (∼0.8- to 1.3-kb transcripts) were also induced at 24 and 48 h after Adp53 infection in p53 mutant human prostate cancer cell line TSU-Pr1 (Fig. 1B). To test whether DNA damage can induce mRTVP-1 expression in the presence or absence of endogenous p53, specific transformed and nontransformed cell lines were subjected to gamma irradiation or Dox treatment, and the expression of mRTVP-1 mRNA was analyzed by RT-PCR. Figure 2A and B (upper panels) show that mRTVP-1 was induced approximately twofold within 12 to 24 h following gamma irradiation in RM-9 mouse prostate cancer cells that contain wild-type p53 (18) and more than twofold within 24 to 48 h after treatment with Dox. Interestingly, gamma irradiation and Dox treatment also induced the expression of RTVP-1 in TSU-Pr1, a p53 mutant human prostate cancer cell line, and 178-2 BMA, a p53-null mouse prostate cancer cell line derived from a bone metastasis (33) (Fig. 2A and B, upper panels). mRNA levels of hRTVP-1 in TSU-Pr1 were induced approximately threefold by gamma irradiation and ∼60% by Dox, while mRNA levels of mRTVP-1 in 178-2 BMA were induced more than fourfold by gamma irradiation and nearly threefold by Dox. In two isogenic human colon cancer cell lines hRTVP-1 was induced approximately twofold by gamma irradiation and more than onefold by Dox in HCT116 p53−/−, while in HCT116 p53+/+ hRTVP-1 was induced only marginally by gamma irradiation or Dox (Fig. 2A and B, lower panels). Interestingly, the same treatments did not induce mRTVP-1 in the nontransformed, isogenic pair of mouse embryo fibroblast cell lines MEF p53+/+ and MEF p53−/− (Fig. 2A and B, middle panels). These data suggest that RTVP-1 is upregulated by p53 and also by stress-related factors, such as gamma irradiation or Dox in a p53-independent manner in multiple cancer cell lines. The induction of mRTVP-1 is cell type specific, and nontransformed cells may be less responsive.
FIG. 1.
RTVP-1 is upregulated by p53. (A) Kinetic analysis of mRTVP-1 mRNA expression following infection of the p53-null mouse prostate cancer cell line 148-1 PA with no adenoviral vector, control adenoviral vector (AdCMV), or p53-expressing vector Adp53. (B) Induction of RTVP-1 at 24 and 48 h by Adp53 in the human prostate cancer cell line TSU-Pr1 relative to uninfected, control vector Adβgal, or AdmRTVP-1.
FIG. 2.
Time course of RTVP-1 upregulation by gamma irradiation and Dox. (A). Induction of RTVP-1 by gamma irradiation in p53 wild-type mouse prostate cancer cell line RM9, p53 mutant human prostate cancer cell line TSU-Pr1, and p53-null mouse prostate cancer cell line 178-2 BMA (upper panel); isogenic pairs of mouse embryo fibroblast cell lines (MEF p53+/+ and MEF p53−/−) (middle panel); and isogenic human colon cancer cell lines (HCT116 p53+/+ and HCT116 p53−/−) (lower panel). (B) Induction of RTVP-1 by Dox in the same cells as for panel A. The numbers at the bottom of gels are the ratios of hRTVP-1 to human HPRT or mRTVP-1 to mouse β-actin.
Comparison of mRTVP-1 and hRTVP-1 proteins.
To further study this p53-regulated gene, we used the differential display PCR fragment to screen a mouse prostate cancer cDNA library generated from 148-1 PA cells (38) and isolated multiple cDNAs containing the complete open reading frame of mRTVP-1 that encoded 255 aa. DNA sequence analysis of cDNAs from normal prostate demonstrated sequences completely identical to that from the 148-1PA library (data not shown). Comparison of the deduced mRTVP-1 protein sequence with known protein sequences revealed that mRTVP-1 has 68% identity and 75% homology to the human RTVP-1 protein (Fig. 3). But, notably, mRTVP-1 contains two short in-frame deletions of 2 aa (PH) at positions 86 and 87 and a 9-aa deletion (KVSGFDALS) from aa 158 to 166 relative to hRTVP-1. Both mouse and human proteins contain a transmembrane domain, a putative N-linked glycosylation site and a hydrophobic region near the carboxyl terminus. Interestingly, both proteins also contain a putative N-terminal signal peptide and extracellular protein signature motifs, which suggests that both proteins are located on the surface of the cell membrane or potentially secreted.
Identification of p53 binding site.
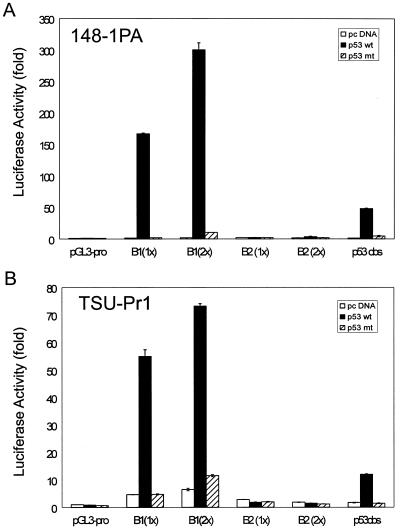
To explore the regulation of mRTVP-1 by p53, we used a 500-nt sequence from the mRTVP-1 cDNA as a probe to isolate genomic mRTVP-1 and sequenced a large portion of the mRTVP-1 gene, including 3.6 kb upstream of the initial ATG, exon 1 (325 nt), intron 1 (3.2 kb), exon 2 (242 nt), and intron 2 (4.6 kb). (See schematic of mRTVP-1 in Fig. 4A). Four potential p53 binding sites (>90% identity compared to the p53 consensus site RRRCWWGYYY nRRRCWWGYYY, where R is purine, Y is pyrimidine, W is A or T, and n is 0 to 13 bases [any base] [8]) were documented in the mRTVP-1 sequence (Fig. 4A). Site A, (−136) GcACTTGTCTagcatAAACAAcCCT (90% homology; n = 5), is located in exon 1; site B1, (+962) AGACAAGCCCAAACATGCCg (95% homology; n = 0), site B2, (+972) AAACATGCCgtagctgaatcacAcGCTAGTCT (90% homology; n = 12), which overlaps with site B1 (Fig. 4A), and site C, (+2888) AGGCTTGTgCGAGCAAGCCa (90% homology; n = 0), are located in intron 1. To test these four potential p53 binding sites, we first produced a series of mRTVP-1 DNA fragments with deletions of potential p53 binding sites and constructed pGL3-basic luciferase reporter constructs. (Fig. 4B). In 148-1 PA and TSU-Pr1 cells (Fig. 4C and D), cotransfection of wild-type or mutant p53-expressing plasmids (2) with 1.4k pro-Luc, which contains 1.4 kb of mRTVP-1 promoter sequence and site A, did not show p53-specific activity, although substantially increased p53-independent basal activity compared with that of the pGL3-basic control vector was demonstrated. However, when the intron 1 sequence containing site B1, site B2, and site C was added to 1.4k pro-Luc (1.4k pro+int1-Luc), p53-specific reporter activity increased approximately 20- and 5-fold in 148-1 PA and TSU-Pr1 cells, respectively, compared with control vector pcDNA-transfected cells. Mutant p53 failed to produce such p53-specific activity. Interestingly, the reporter with intron 1 only (int1-Luc) also exhibited about a 23-fold increase in p53-specific reporter activity in 148-1 PA and about a 5-fold increase in TSU-Pr1 along with a reduction of basal activity. To further clarify which potential binding site(s) mediates the p53-specific response, we produced three deletion and/or mutation constructs from 1.4k pro+int1-Luc (Fig. 4B). Deletion of site C (del C-Luc) did not alter p53-upregulated luciferase activity. However, deletion of site B (including both B1 and B2) (del B-Luc) completely abolished all p53-specific transactivation activity. Four point mutations in the p53 binding core sequence of site B1 (B1*4p-Luc) (Fig. 4B) also completely abolished p53-specific activity (Fig. 4C and D). These data strongly suggest that the B1 and/or B2 site is responsible for the p53-specific transactivation of mRTVP-1. However, whether B1, B2, or both are responsible for the p53-specific transactivation was not resolved in this experiment, since the mutation in the second half-site of B1 could also abolish B2 activity. To clearly discriminate between B1 and B2 activities, we directly tested synthetic B1 or B2 binding site sequences by using the simian virus 40 minimal promoter-controlled luciferase reporter (pGL3-promoter). A p53 consensus binding sequence was also inserted into pGL3-promoter and served as a positive control. The results shown in Fig. 5A and B demonstrated that wild-type p53 strongly activated the transcription of B1-mediated (both 1x and 2x copies) luciferase activity (118- and 165-fold, respectively, in 148-1 PA; and ∼12-fold for both in TSU-Pr1), however, wild-type p53 failed to activate the transcription of B2-mediated (both 1x and 2x copies) luciferase activity. These data clearly indicate that B1 is the p53 binding site responsible for the transactivation of mRTVP-1.
FIG. 5.
Verification of p53 binding function of B1 and B2 sites by luciferase reporter constructs containing either 1x or 2x copies of B1 or B2 sequence in pGL3-promoter as described in Materials and Methods. Luciferase activities in 148-1PA (A) and TSU-Pr1 (B) were determined as described above and in Materials and Methods. Symbols are as defined for Fig. 4.
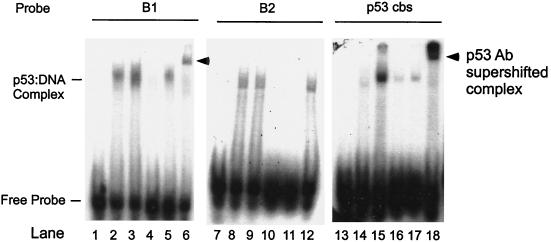
We also performed EMSA using synthetic B1 and B2 binding site sequences as probes. A synthetic p53 consensus binding sequence served as a positive control. Figure 6 shows that p53-specific protein-DNA complex (pCMVp53-transfected nuclear extract, lanes 3 and 15) compared to nonspecific protein-DNA complex (pCMVβgal-transfected nuclear extract, lanes 2 and 14) was present only when B1 or p53 consensus but not B2 binding site sequences were used as probes (compare lanes 3 and 15 to lane 9). Cold (1,000×) B1 or p53 consensus binding site sequences effectively competed with radiolabeled B1 probes (lanes 4 and 5). In addition, the p53-DNA complex was supershifted by a p53-specific antibody (DO1) (lanes 6 and 18). Notably this p53-specific supershifted complex was absent when B2 was used as a probe (lane 12). These results confirm that site B1 is responsible for p53 binding.
FIG. 6.
Demonstration of p53 binding to site B1 by EMSA. Nuclear extracts from 148-1 PA cells (p53 null) transfected with pCMVp53 or pCMVβgal were incubated with γ-32P-labeled oligonucleotide probes B1, B2, or p53cbs. Lanes 1, 7, and 13, free probe; lanes 2, 8, and 14, pCMV-βgal-transfected nuclear extract; lanes 3 to 6, 9 to 12, and 15 to 18, pCMV-p53-transfected nuclear extract. Lanes 4 and 16, competition with cold B1 probe; lane 10, competition with cold B2 probe; lanes 5, 11, and 17, competition with p53 cbs probe; lanes 6, 12, and 18, supershift with p53 specific antibody DO1.
Evaluation of RTVP-1 expression in mouse and human cells.
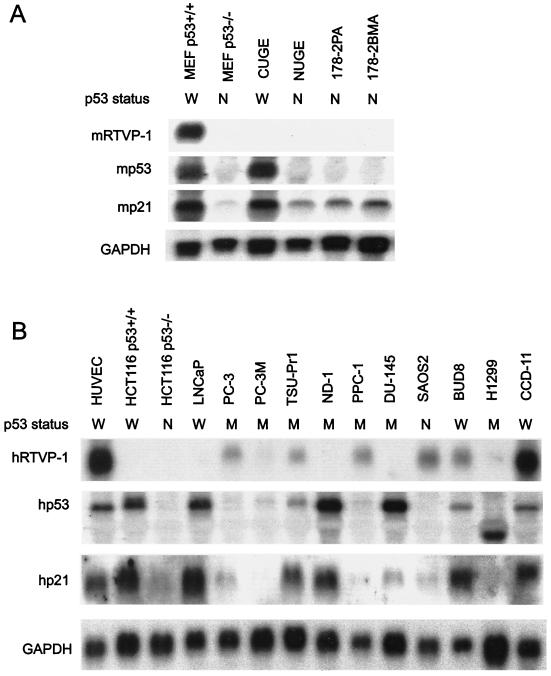
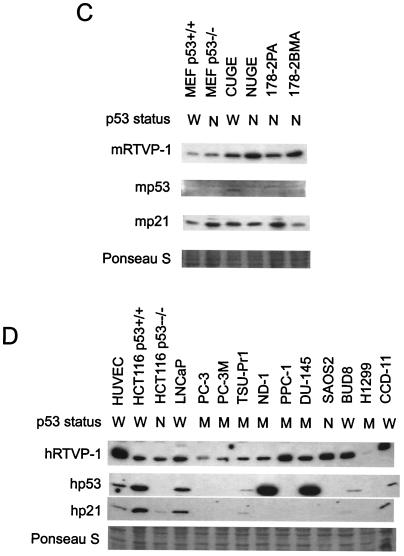
To evaluate the pattern of RTVP-1 expression we analyzed RTVP-1 mRNA and protein levels in selected nontransformed and transformed mouse and human cell lines (Fig. 7). MEF p53+/+ contained an mRTVP-1 transcript of approximately 1.1 kb, whereas in MEF p53−/−, CUGE (nontransformed urogenital sinus epithelial cells from p53+/+ mice [3]), NUGE (nontransformed urogenital sinus epithelial cells from p53−/− mice [38]), and mouse prostate cancer cell lines derived from a primary tumor (178-2 PA) and a bone metastasis (178-2 BMA) generated from p53−/− prostatic tissues (33) mRTVP-1 mRNA levels were undetectable by Northern blotting (Fig. 7A). Mouse p53 mRNA was detected in MEF p53+/+ and CUGE cells but not in the four p53-null cell lines analyzed (Fig. 7A). Relatively high levels of p21 mRNA were observed in the two p53+/+ cell lines, compared to the p53−/− mouse cell lines that demonstrated lower levels of p21 (Fig. 7A). Both prostate and nonprostate human cancer cell lines contained low to undetectable levels of RTVP-1 mRNA with the exception of CCD-11 cells, where significant levels of hRTVP-1 mRNA were apparent. Relatively high levels of hRTVP-1 mRNA were demonstrated in nontransformed HUVEC (Fig. 7B). The levels of p53 and p21 mRNAs in these cells were also determined (Fig. 7B). The results were consistent with the known p53 gene status of each specific cell line and showed coordinate regulation of p53 and p21 mRNA levels. Western blotting analysis revealed that all mouse and human cancer cell lines expressed RTVP-1 protein although the levels were lower than those of HUVEC (Fig. 7C and D). Among the mouse cell lines, p53 protein was detected in CUGE but not in other cell lines, whereas p21 was present in all cell lines (Fig. 7C). Among the human cell lines (Fig. 7D), two cancer cell lines with p53 mutations, ND1 and DU145, demonstrated high levels of p53 protein, presumably due to higher protein stability. p53 protein was not detected in p53-null cell lines HCT116 p53−/− and SAOS2 or in p53 mutant cell lines PC-3, PC-3 M, PPC-1, and H1299. P21 was present in HUVEC, HCT116 p53+/+, HCT116 p53−/−, LNCaP, TSU-Pr1, BUD8, and CCD-11. The data suggest that RTVP-1 is expressed in normal and cancer cells in a p53-independent manner. All human cancer cell lines expressed lower levels of RTVP-1 than did nontransformed HUVEC.
FIG. 7.
RTVP-1 expression in mouse and human cell lines. Northern blot analysis of mouse (A) and human (B) cell lines. Cells were grown, RNA was isolated, and Northern blotting was performed as described in Materials and Methods. GAPDH was monitored as a loading control. Western blot analysis of mouse (C) and human (D) cell lines. The same membranes used for Western blotting were prestained with Ponseau S, and a portion of each membrane was included at the bottom of each panel as loading control. p53 status is indicated as follows: W, wild type (+/+); M, mutant; and N, null (−/−).
mRTVP-1 induces apoptosis in cancer cells.
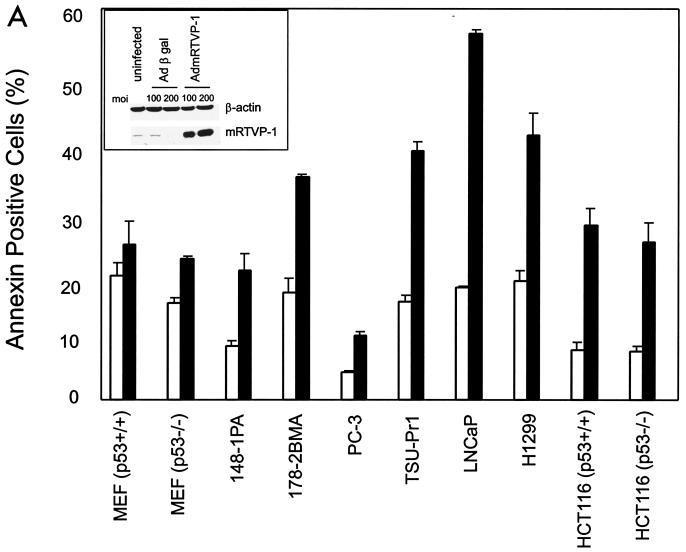
To analyze the biological activities of mRTVP-1, a panel of nontransformed and transformed cells were infected with AdmRTVP-1 or Adβgal and subsequently analyzed for apoptotic activities. Western blotting analysis demonstrated high levels of mRTVP-1 protein 48 h after infection of TSU-Pr1 cells with AdmRTVP-1 (Fig. 8A, inset). AdmRTVP-1 induced apoptosis as demonstrated by significant increases in annexin V positivity (Fig. 8A) in 148-1PA, 178-2 BMA, PC-3, TSU-Pr1, LNCaP, H1299, HCT116 p53+/+, and HCT116 p53−/− cell lines. Notably, there was less effect of AdmRTVP-1 on nontransformed MEF cells (p53+/+ and p53−/−).
FIG. 8.
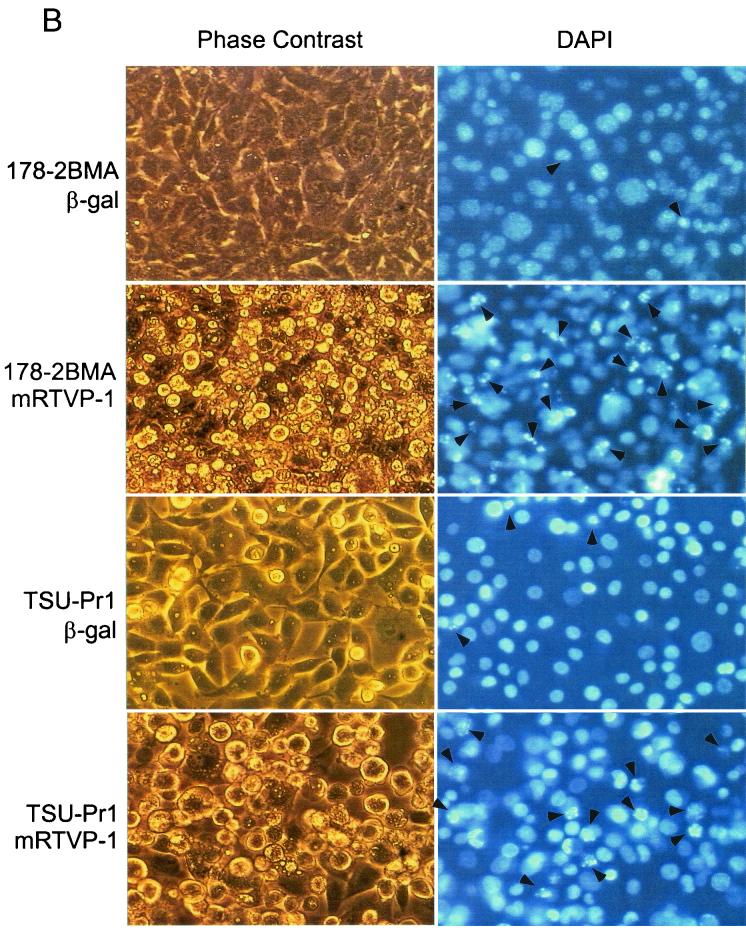
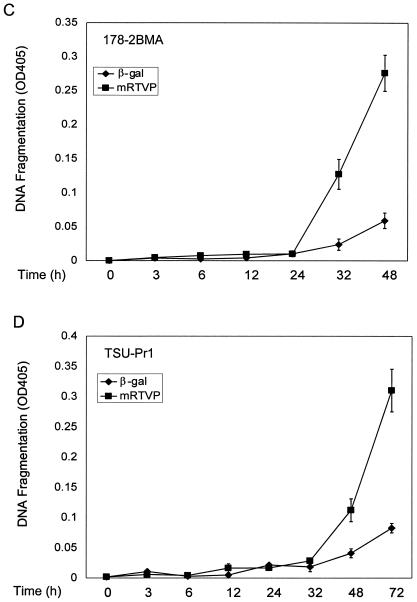
Overexpression of mRTVP-1 induces apoptosis. (A) Apoptosis was determined by annexin V binding and flow cytometry on cells infected with Adβgal (open boxes) or AdmRTVP-1 (closed boxes) at a multiplicity of infection of 100. Protein levels of mRTVP-1 in TSU-Pr1 cells were determined by Western blotting (inset) using an affinity-purified antibody to mRTVP-1. The levels of β-actin were also monitored as a loading control. (B) Morphological changes documented by phase-contrast microscopy (left panels) and fluorescence microscopy after DAPI staining (right panels). Arrows indicate apoptotic nuclei. 178-2 BMA images were taken 48 h postinfection; TSU-Pr1 images were taken 72 h postinfection. (C and D) Time courses of DNA fragmentation in 178-2 BMA (C) and TSU-Pr1 (D) after infection by Adβgal or AdmRTVP-1. DNA fragmentation was analyzed at indicated time points by using a Cell Death ELISA kit (Boehringer Mannheim).
More-detailed analyses of 178-2 BMA cells showed that extensive apoptosis occurred in AdmRTVP-1-infected cells 48 h postinfection as documented by phase-contrast microscopy and DAPI staining (Fig. 8B). In the human prostate cancer cell line TSU-Pr1, similar results were observed 72 h after infection (Fig. 8B). Analysis using DNA fragmentation ELISA revealed that DNA fragmentation was initiated 24 h after infection by AdmRTVP-1 and markedly increased during 24 to 48 h postinfection in 178-2 BMA cells. Greater-than-fourfold-increased DNA fragmentation was observed in AdmRTVP-1-infected cells compared with Adβgal-infected cells (Fig. 8C, upper panel). In TSU-Pr1 cells, the initiation of DNA fragmentation was delayed approximately 8 h compared with 178-2 BMA and by 72 h after infection the extent of DNA fragmentation was similar to that observed in 178-2 BMA 48 h postinfection. These data provide definitive evidence that adenoviral vector-transduced mRTVP-1 has proapoptotic activities in malignant cells.
Deletion of the signal peptide from RTVP-1 reduces its proapoptotic activities.
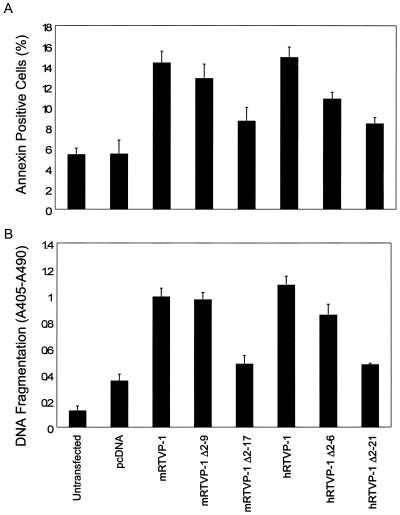
Signal peptides are essential to target proteins to the secretory pathway (24, 30). The deduced mRTVP-1 protein sequence and reported hRTVP-1 protein sequence (Fig. 3) (21, 32) show that both proteins contain a putative N-terminal signal peptide. A cleavage site for the signal peptide of mRTVP-1 is predicted to be located between aa 17 and 18 (VSS ^ SS) of mRTVP-1 and that of hRTVP-1 is predicted to be located between aa 21 and 22 (SHT ^ AN) of hRTVP-1 according to a program provided by the SignalP V1.1 World Wide Web server. To explore the potential role of predicted signal peptides in RTVP-1-mediated apoptotic activities, RTVP-1 lacking the signal peptide (mRTVP-1 Δ2-17 and hRTVP-1 Δ2-21) and RTVP-1 with partial deletion of signal peptide (mRTVP-1 Δ2-9 and hRTVP-1 Δ2-9) were constructed. Human prostate cancer cells TSU-Pr1 were transfected with full-length RTVP-1, or deletion mutant, or control plasmid vector pcDNA. The comparison of these transfected cells revealed that mRTVP-1 Δ2-17 or hRTVP-1 Δ2-21 (full deletion of signal peptide sequence) suppressed RTVP-1-mediated apoptotic activities to levels that approached those of control vector and mRTVP-1 Δ2-9 or hRTVP-1 Δ2-9 (deletion of a portion of the signal peptide) had less effect as demonstrated by both annexin V positivity analysis and DNA fragmentation analysis (Fig. 9). These data indicate that the signal peptide is important for RTVP-1-mediated proapoptotic activities. In addition they demonstrate that either transfected mouse or human RTVP-1 can induce apoptosis.
FIG. 9.
Deletion of the signal peptide reduces RTVP-1-mediated apoptotic activities. Annexin V positivity (A) and DNA fragmentation (B) were analyzed 72 h after TSU-Pr1 cells were transfected with full-length RTVP-1 or the signal peptide deletion mutants described in Materials and Methods.
DISCUSSION
Over the last decade, numerous studies have established p53 mutations as being paramount to the development and progression of various malignancies. The p53 tumor suppressor protein has been associated with specific tumor suppressor functions that include growth suppression, apoptosis, and antiangiogenic activities. The majority of p53 mutations in human tumors have shown decreased sequence-specific DNA binding and transactivation activity (6). It has been estimated that 200 to 300 genes are under the transcriptional regulation of p53 (41). However, there is substantial heterogeneity in the expression of p53 target genes among colorectal cancer cell lines (45), and presumably within other malignant cells. Because loss of p53 function is associated with prostate cancer metastasis (7, 19, 23, 28, 35, 38, 43), we developed molecular approaches to identify p53 target genes that might suppress prostate cancer progression. In this study, we identified another gene that is upregulated and directly targeted by p53, mRTVP-1. RTVP-1 mRNA levels were shown to be induced by adenoviral vector-mediated p53 overexpression. We also demonstrated that gamma irradiation or Dox induced mRTVP-1 mRNA levels in cancer cells both in the presence and in the absence of endogenous p53, indicating p53-independent, DNA damage-related pathways of RTVP-1 regulation.
In our efforts to understand the regulation of the mRTVP-1 gene by p53, we isolated genomic mRTVP-1 and sequenced a significant portion of this gene (∼11 kb), including 3.5 kb of the promoter region, exon 1, intron 1, exon 2, and intron 2. Sequence analysis revealed four potential p53 binding sites that were located in exon 1 and intron 1. Cotransfection studies and EMSA led to the detection of site B1 in intron 1 as the sole p53-specific binding site. These results conclusively determine that mRTVP-1 is a direct p53 target gene.
Northern blotting analyses demonstrated that multiple nontransformed and transformed mouse and human cell lines expressed relatively low levels of RTVP-1 mRNA. Three cell lines including two nontransformed cell lines (MEF p53+/+ and HUVEC) and a transformed human cell line (CCD-11) that contain wild-type p53 expressed significant levels of RTVP-1 mRNA. However, in general there was no evidence for linkage between the expression of wild-type p53 and RTVP-1 in contrast to wild-type p53 and p21 mRNA, which appeared to be linked. Western blotting analyses revealed higher levels of RTVP-1 protein than expected from Northern blotting, suggesting independent levels of regulation for RTVP-1 expression. As found with RTVP-1 mRNA levels, RTVP-1 protein levels were not related to p53 or p21 protein levels. Overall, the results of our expression analysis were consistent with the induction studies and define this gene as being capable of p53-independent regulation. Our results did not reveal expression patterns unique to prostate cancer cells or to malignant cells in general. Further studies that include analysis of RTVP-1 expression in various normal and malignant tissues will be necessary to detect any cell or organ type specificity in RTVP-1 expression during the transitions from nontransformed to malignant to fully metastatic. It is of interest that multiple RTVP-1 transcripts were apparent in human cells whereas only a single major transcript was expressed in mouse tissue and cell lines. This may reflect different gene structure and/or regulatory mechanisms between mouse and human. However, RTVP-1 mRNA levels were responsive to p53, gamma irradiation, and Dox in both mouse and human cell lines.
Additional studies showed that overexpression of mRTVP-1 induces apoptosis in cancer cells in vitro as demonstrated by annexin V positivity, phase microscopy, DAPI staining, and DNA fragmentation ELISA. There are numerous studies that have documented the tumor-suppressing activities of p53 in various systems, and clear evidence has been found for a role for p53 as a tumor suppressor gene in human cancer. The induction of apoptosis is an important function of p53 that has profound relevance to the tumor suppressor role of p53 (5, 13) and potentially for its putative antimetastatic functions in prostate cancer (7, 28, 35, 38). Although in some cases p53-mediated transcriptional activity is not required for apoptosis, sequence-specific transactivation of downstream target genes by p53 is essential for apoptosis in many systems (reviewed in references 5, 9, 11, and 42). Better-characterized p53-induced proapoptotic genes include bax, Fas/APO1, Killer/DR5, PIG3, and IGF-BP3. Bax binds to Bcl2 and antagonizes its ability to block apoptosis (15). Fas/APO1 and Killer/DR5 are cell surface death receptors that transmit rapid apoptotic signals initiated by the binding to death ligands (reviewed in reference 1). PIG3 is one of several p53-induced genes (PIGs) identified by Polyak et al. (27). IGF-BP3 inhibits signaling by insulin-like growth factor 1 and therefore exerts both antimitogenic and proapoptotic effects (29). Although we clearly demonstrated that adenoviral vector-mediated overexpression of mRTVP-1 induced apoptosis in multiple cancer cell lines, the downstream pathways involved remain to be fully elucidated. However, we have shown that RTVP-1 levels are induced by p53 and by gamma irradiation or Dox in a p53-independent fashion. Furthermore, based on the deducted amino acid sequence and our own results it appears that the RTVP-1 protein is secreted or cleaved at the extracellular-transmembrane domain junction and that extracellular RTVP-1 is important in RTVP-1-mediated apoptosis. The capacity to induce apoptosis as a secreted and soluble protein would place RTVP-1 in a unique category. Since it has been previously shown that RTVP-1 expression is associated with macrophage differentiation (12), it's conceivable that extracellular RTVP-1 may have widespread biological effects through cell type-specific activities.
In summary, the present study demonstrates that mRTVP-1 is a p53 target gene and is capable of inducing apoptosis in various malignant cells. The discovery of mRTVP-1 as a p53-regulated proapoptotic gene not only adds mRTVP-1 to the list of p53 target genes but also represents the first report on the function of RTVP-1. The elucidation of this proapoptotic function for RTVP-1 combined with the observation that a secreted and soluble form of RTVP-1 is important for its apoptotic activities places this gene in a unique niche. Its complex regulation that includes induction by p53 and by gamma irradiation or Dox in a p53-independent fashion further distinguishes RTVP-1. Clearly, additional studies concentrating on the molecular mechanisms that underlie the expression, regulation, and functions of mRTVP in normal and malignant cells are indicated.
Acknowledgments
This work was supported by grants from the NCI (RO1-50588) and Specialized Program of Research Excellence (SPORE) (P50-58204).
REFERENCES
- 1.Ashkenazi, A., and V. M. Dixit. 1998. Death receptors: signaling and modulation. Science 281:1305-1308. [DOI] [PubMed] [Google Scholar]
- 2.Baker, S. J., S. Markowitz, E. R. Fearon, J. K. Willson, and B. Vogelstein. 1990. Suppression of human colorectal carcinoma cell growth by wild-type p53. Science 249:912-915. [DOI] [PubMed] [Google Scholar]
- 3.Baley, P. A., K. Yoshida, W. Qian, I. Sehgal, and T. C. Thompson. 1995. Progression to androgen insensitivity in a novel in vitro mouse model for prostate cancer. J. Steroid Biochem. Mol. Biol. 52:403-413. [DOI] [PubMed] [Google Scholar]
- 4.Bunz, F., P. M. Hwang, C. Torrance, T. Waldman, Y. Zhang, L. Dillehay, J. William, C. Lengauer, K. W. Kinzler, and B. Vogelstein. 1999. Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J. Clin. Investig. 104:263-269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Burns, T. F., and W. S. El-Deiry. 1999. The p53 pathway and apoptosis. J. Cell. Physiol. 181:231-239. [DOI] [PubMed] [Google Scholar]
- 6.Cho, Y., S. Gorina, P. D. Jeffrey, and N. P. Pavletich. 1994. Crystal structure of a p53 tumor suppressor-DNA complex: understanding tumorigenic mutations. Science 265:346-355. [DOI] [PubMed] [Google Scholar]
- 7.Eastham, J. A., A. M. Stapleton, A. E. Gousse, T. L. Timme, G. Yang, K. M. Slawin, T. M. Wheeler, P. T. Scardino, and T. C. Thompson. 1995. Association of p53 mutations with metastatic prostate cancer. Clin. Cancer Res. 1:1111-1118. [PubMed] [Google Scholar]
- 8.el-Deiry, W. S., S. E. Kern, J. A. Pietenpol, K. W. Kinzler, and B. Vogelstein. 1992. Definition of a consensus binding site for p53. Nat. Genet. 1:45-49. [DOI] [PubMed] [Google Scholar]
- 9.el-Deiry, W. S. 1998. Regulation of p53 downstream genes. Semin. Cancer Biol. 8:345-357. [DOI] [PubMed] [Google Scholar]
- 10.Furukawa, M., T. Uchiumi, M. Nomoto, H. Takano, R. I. Morimoto, S. Naito, M. Kuwano, and K. Kohno. 1998. The role of an inverted CCAAT element in transcriptional activation of the human DNA topoisomerase IIα gene by heat shock. J. Biol. Chem. 273:10550-10555. [DOI] [PubMed] [Google Scholar]
- 11.Giaccia, A. J., and M. B. Kastan. 1998. The complexity of p53 modulation: emerging patterns from divergent signals. Genes Dev. 12:2973-2983. [DOI] [PubMed] [Google Scholar]
- 12.Gingras, M. C., and J. F. Margolin. 2000. Differential expression of multiple unexpected genes during U937 cell and macrophage differentiation detected by suppressive subtractive hybridization. Exp. Hematol. 28:65-76. [DOI] [PubMed] [Google Scholar]
- 13.Gottlieb, T. M., and M. Oren. 1998. p53 and apoptosis. Semin. Cancer Biol. 8:359-368. [DOI] [PubMed] [Google Scholar]
- 14.Gupta, S., V. Radha, Y. Furukawa, and G. Swarup. 2001. Direct transcriptional activation of human caspase-1 by tumor suppressor p53. J. Biol. Chem. 276:10585-10588. [DOI] [PubMed] [Google Scholar]
- 15.Kelekar, A., and C. B. Thompson. 1998. Bcl-2-family proteins: the role of the BH3 domains in apoptosis. Trends Cell Biol. 8:324-330. [DOI] [PubMed] [Google Scholar]
- 16.Levine, A. J. 1997. p53, the cellular gatekeeper for growth and division. Cell 88:323-331. [DOI] [PubMed] [Google Scholar]
- 17.Li, L., G. Yang, S. Ebara, T. Sato, Y. Nasu, T. L. Timme, C. Ren, J. Wang, S. A. Tahir, and T. C. Thompson. 2001. Caveolin-1 mediates testosterone-stimulated survival/clonal growth and promotes metastatic activities in prostate cancer cells. Cancer Res. 61:4386-4392. [PubMed] [Google Scholar]
- 18.Lu, X., S. H. Park, T. C. Thompson, and D. P. Lane. 1992. Ras-induced hyperplasia occurs with mutation of p53, but activated ras and myc together can induce carcinoma without p53 mutation. Cell 70:153-161. [DOI] [PubMed] [Google Scholar]
- 19.Mirchandani, D., J. Zheng, G. J. Miller, A. K. Ghosh, D. K. Shibata, R. J. Cote, and P. Roy-Burman. 1995. Heterogeneity in intratumor distribution of p53 mutations in human prostate cancer. Am. J. Pathol. 147:92-101. [PMC free article] [PubMed] [Google Scholar]
- 20.Miyashita, T., and J. C. Reed. 1995. Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 80:293-299. [DOI] [PubMed] [Google Scholar]
- 21.Murphy, E. V., Y. Zhang, W. Zhu, and J. Biggs.. 1995. The human glioma pathogenesis-related protein is structurally related to plant pathogenesis-related proteins and its gene is expressed specifically in brain tumors. Gene 159:131-135. [DOI] [PubMed] [Google Scholar]
- 22.Nasu, Y., T. L. Timme, G. Yang, C. H. Bangma, L. Li, C. Ren, S. H. Park, M. DeLeon, J. Wang, and T. C. Thompson. 1998. Suppression of caveolin expression induces androgen sensitivity in metastatic androgen-insensitive mouse prostate cancer cells. Nat. Med. 4:1062-1064. [DOI] [PubMed] [Google Scholar]
- 23.Navone, N. M., P. Troncoso, L. L. Pisters, T. L. Goodrow, J. L. Palmer, W. W. Nichols, A. C. von Eschenbach, and C. J. Conti. 1993. p53 protein accumulation and gene mutation in the progression of human prostate carcinoma. J. Natl. Cancer Inst. 85:1657-1669. [DOI] [PubMed] [Google Scholar]
- 24.Nielsen, H., J. Engelbrecht, S. Brunak, and G. von Heijne. 1997. Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng. 10:1-6. [DOI] [PubMed] [Google Scholar]
- 25.Oda, K., H. Arakawa, T. Tanaka, K. Matsuda, C. Tanikawa, T. Mori, H. Nishimori, K. Tamai, T. Tokino, Y. Nakamura, and Y. Taya. 2000. p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by Ser-46-phosphorylated p53. Cell 102:849-862. [DOI] [PubMed] [Google Scholar]
- 26.Osoegawa, K., M. Tateno, P. Y. Woon, E. Frengen, G. A. Mammoser, J. J. Catanese, Y. Hayashizaki, and P. J. de Jong. 2000. Bacterial artificial chromosome libraries for mouse sequencing and functional analysis. Genome Res. 10:116-128. [PMC free article] [PubMed] [Google Scholar]
- 27.Polyak, K., Y. Xia, J. L. Zweier, K. W. Kinzler, and B. Vogelstein. 1997. A model for p53 induced apoptosis. Nature 389:300-305. [DOI] [PubMed] [Google Scholar]
- 28.Quinn, D. I., S. M. Henshall, D. R. Head, D. Golovsky, J. D. Wilson, P. C. Brenner, J. J. Turner,W. Delprado, J. F. Finlayson, P. D. Stricker, J. J. Grygiel, and R. L. Sutherland. 2000. Prognostic significance of p53 nuclear accumulation in localized prostate cancer treated with radical prostatectomy. Cancer Res. 60:1585-1594. [PubMed] [Google Scholar]
- 29.Rajah, R., B. Valentinis, and P. Cohen. 1997. Insulin like growth factor (IGF)-binding protein 3 induces apoptosis and mediates the effects of transforming growth factor-beta 1 on programmed cell death through p53- and IGF-independent mechanism. J. Biol. Chem. 272:12181-12188. [DOI] [PubMed] [Google Scholar]
- 30.Rapoport, T. A. 1992. Transport of proteins across the endoplasmic reticulum membrane. Science 258:931-936. [DOI] [PubMed] [Google Scholar]
- 31.Ren, C., G. Yang, T. L. Timme, T. M. Wheeler, and T. C. Thompson. 1998. Reduced lysyl oxidase messenger RNA levels in experimental and human prostate cancer. Cancer Res. 58:1285-1290. [PubMed] [Google Scholar]
- 32.Rich, T., P. Chen, F. Furman, N. Huynh, and M. A. Israel. 1996. RTVP-1, a novel human gene with sequence similarity to genes of diverse species, is expressed in tumor cell lines of glial but not neuronal origin. Gene 180:125-130. [DOI] [PubMed] [Google Scholar]
- 33.Shaker, M. M., G. Yang, T. L. Timme, S. H. Park, D. Kadmon, C. Ren, X. Ji, H.-L. Lee, I. Sehgal, M. Anzano, M. B. Sporn, and T. C. Thompson. 2001. Dietary 4-HPR suppresses the development of bone metastasis in vivo in a mouse model of prostate cancer progression. Clin. Exp. Metastasis 18:429-438. [DOI] [PubMed] [Google Scholar]
- 34.Sheikh, M. S., T. F. Burns, Y. Huang, G. S. Wu, S. Amundson, K. S. Brooks, A. J. Fornace, Jr., and W. S. el-Deiry. 1998. p53-dependent and -independent regulation of the death receptor KILLER/DR5 gene expression in response to genotoxic stress and tumor necrosis factor alpha. Cancer Res. 58:1593-1598. [PubMed] [Google Scholar]
- 35.Stapleton, A. M., T. L. Timme, A. E. Gousse, Q. F. Li, A. A. Tobon, M. W. Kattan, K. M. Slawin, T. M. Wheeler, P. T. Scardino, and T. C. Thompson. 1997. Primary human prostate cancer cells harboring p53 mutations are clonally expanded in metastases. Clin. Cancer Res. 3:1389-1397. [PubMed] [Google Scholar]
- 36.Stapleton, A. M., P. Zbell, M. W. Kattan, G. Yang, T. M. Wheeler, P. T. Scardino, and T. C. Thompson.. 1998. Assessment of the biologic markers p53, Ki-67, and apoptotic index as predictive indicators of prostate carcinoma recurrence after surgery. Cancer 82:168-175. [DOI] [PubMed] [Google Scholar]
- 37.Tahir, S. A., G. Yang, S. Ebara, T. L. Timme, T. Satoh, L. Li, A. Goltsov, M. Ittmann, J. D. Morrisett, and T. C. Thompson. 2001. Secreted caveolin-1 stimulates cell survival/clonal growth and contributes to metastasis in androgen-insensitive prostate cancer. Cancer Res. 61:3882-3885. [PubMed] [Google Scholar]
- 38.Thompson, T. C., S. H. Park, T. L. Timme, C. Ren, J. A. Eastham, L. A. Donehower, A. Bradley, D. Kadmon, and G. Yang. 1995. Loss of p53 function leads to metastasis in ras+myc-initiated mouse prostate cancer. Oncogene 10:869-879. [PubMed] [Google Scholar]
- 39.Thompson, T. C., T. L. Timme, and I. Sehgal. 1998. Oncogenes, growth factors, and hormones in prostate cancer, p. 327-359. In R. B. Dickson and D. S. Salomon (ed.), Hormones and growth factors in development and neoplasia. Wiley-Liss, Inc., New York, N.Y.
- 40.Timme, T. L., A. Goltsov, S. Tahir, L. Li, J. Wang, C. Ren, R. N. Johnston, and T. C. Thompson. 2000. Caveolin-1 is regulated by c-myc and suppresses c-myc-induced apoptosis. Oncogene 19:3256-3265. [DOI] [PubMed] [Google Scholar]
- 41.Tokino, T., S. Thiagalingam, W. S. el-Deiry, T. Waldman, K. W. Kinzler, and B. Vogelstein. 1994. p53 tagged sites from human genomic DNA. Hum. Mol. Genet. 3:1537-1542. [DOI] [PubMed] [Google Scholar]
- 42.Vogelstein, B., and K. W. Kinzler. 1992. p53 function and dysfunction. Cell 70:523-526. [DOI] [PubMed] [Google Scholar]
- 43.Yang, G., A. M. Stapleton, T. M. Wheeler, L. D. Truong, T. L. Timme, P. T. Scardino, and T. C. Thompson. 1996. Clustered p53 immunostaining: a novel pattern associated with prostate cancer progression. Clin. Cancer Res. 2:399-401. [PubMed] [Google Scholar]
- 44.Yang, G., L. D. Truong, T. L. Timme, C. Ren, T. M. Wheeler, S. H. Park, Y. Nasu, C. H. Bangma, M. W. Kattan, P. T. Scardino, and T. C. Thompson. 1998. Elevated expression of caveolin is associated with prostate and breast cancer. Clin. Cancer Res. 4:1873-1880. [PubMed] [Google Scholar]
- 45.Yu, J., L. Zhang, P. M. Hwang, C. Rago, K. W. Kinzler, and B. Vogelstein. 1999. Identification and classification of p53-regulated genes. Proc. Natl. Acad. Sci. USA 96:14517-14522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhu, J., and X. Chen. 2000. MCG10, a novel p53 target gene that encodes a KH domain RNA-binding protein, is capable of inducing apoptosis and cell cycle arrest in G(2)-M. Mol. Cell. Biol. 20:5602-5618. [DOI] [PMC free article] [PubMed] [Google Scholar]