Abstract
Popeye (Pop) genes are a novel gene family encoding putative transmembrane proteins predominantly present in striated and smooth muscle cells. In this study, a null mutation of Pop1 was generated by replacing the first coding exon of the Pop1 gene with the lacZ reporter gene. Homozygous mice lacking Pop1 were fertile and had a normal life span without any apparent phenotype. LacZ staining of tissues of heterozygous and homozygous Pop1-LacZ mice revealed strong expression in embryonic and fetal hearts. Pop1-LacZ was also expressed in the myotome and in myogenic progenitor cells within the limb and in smooth muscle cells of various organs. In the heart, Pop1-LacZ activity was downregulated postnatally in heterozygous mice but not in homozygous mice. Administration of the β-adrenergic agonist isoproterenol led to a rapid increase in Pop1-LacZ activity in heterozygotes without induction at the transcriptional level, suggesting stabilization of the protein. No difference, however, was observed between homozygous and heterozygous mice in the ability to develop cardiac hypertrophy in response to isoproterenol. The capacity to regenerate skeletal muscle was tested after cardiotoxin injection into the hind limbs of hetero- and homozygous mice. In activated satellite cells of both genotypes, rapid activation of Pop1-LacZ expression was observed. In heterozygous animals, LacZ activity was only transiently elevated in muscle precursor cells undergoing fusion and in newly formed myotubes. In homozygotes, persistence of LacZ expression and a retarded ability to regenerate skeletal muscle were apparent, suggesting that Pop1 plays a role in muscle regeneration.
Popeye (Pop) genes represent a novel gene family encoding proteins with three putative transmembrane domains and no other recognizable domain of known structure (2). In mammals, three family members, Pop1 to -3, have been identified that are differentially expressed in heart and skeletal muscles. At the mRNA level, Pop1 and Pop3 are expressed in both muscle types while Pop2 is expressed predominantly in cardiac muscle (2). In the chick, several Pop1 and Pop3 transcripts have been isolated that are presumably generated by alternative splicing from a single gene (2). In contrast, in mammals, the different Pop transcripts are transcribed from individual genes. In situ hybridization of embryonic chicken and mouse hearts revealed that Pop genes are expressed in the myocardial layer and not in the endocardium or epicardium (2). Pop1 was independently isolated by others and referred to as bves (17). In contrast to the expression pattern observed by in situ hybridization, immunohistochemistry analysis employing antibodies raised against synthetically made portions of the chicken bves protein revealed expression in the proepicardial organ and later in the epicardium and the developing coronary vascular system (17, 25). The reason for the contradictory descriptions of mRNA and protein localization of Pop1/bves is not known. Recently, it was reported that Pop1/bves encodes the prototype of a novel class of cell adhesion molecules mediating cell-cell contacts in a calcium-independent manner (25).
In order to begin to genetically define the function of this gene family, we ablated the Pop1 gene in mice by replacing the first coding exon with a lacZ reporter gene. In heterozygous animals, LacZ activity was first detectable at embryonic day 7.5 (E7.5) in the cardiac crescent. Subsequently, myocytes in the entire myocardium were Pop1-LacZ positive. At E11.5, Pop1-LacZ became downregulated in the trabecular layer but expression was maintained in the compact layer myocardium. In postnatal hearts of heterozygous but not homozygous mice, Pop1-LacZ activity was downregulated, despite the continuous presence of Pop1 transcripts. Isoproterenol (Iso) infusion induced cardiac hypertrophy with no apparent difference between the two genotypes. However, in heterozygous animals, induction of nuclear LacZ expression was observed. In order to induce skeletal muscle regeneration, cardiotoxin was injected into the hind limb. Both mononucleated activated satellite cells and newly regenerated myofibers showed prominent nuclear Pop1-LacZ activity in heterozygous animals. In null mutants, persistence of nuclear LacZ staining and an impaired ability to regenerate skeletal muscle were seen after cardiotoxin injection. The ability to undergo myogenic differentiation was also analyzed in isolated satellite cells in culture. Satellite cells of both genotypes were equally able to undergo myotube formation; however, differentiation appeared to be retarded. Nuclear LacZ activity was downregulated immediately after myotube formation in heterozygous satellite cells, while it persisted in homozygous cells. Our results suggest that Pop1 is involved in the regeneration of adult skeletal muscle.
MATERIALS AND METHODS
Generation of Pop1-LacZ mice.
Parts of the Pop1 gene were isolated from a genomic phage library (mouse genomic λ-Fix II library; Stratagene, Heidelberg, Germany) and a cosmid library (library no. 121 Mus musculus 129/ola; RZPD, Berlin, Germany) by screening with a full-length Pop1 cDNA probe (2). A 1-kb 3" Pop1 sequence was amplified from the genomic clone by PCR and cloned into NotI-XhoI-cut transfer vector pPNT (24). A 6.2-kb EcoRI fragment was identified that contained the first exon (5" untranslated region [UTR]) and parts of the second exon. This fragment was fused in frame to a lacZ reporter gene carrying a nuclear localization signal (NLS) by subcloning it into the pPD46.21 vector (6). The NotI restriction site 3" of the lacZ gene was modified with SalI linkers. A SalI fragment containing the 5" fragment of the Pop1 gene and the lacZ reporter gene was cloned into the BamHI restriction site of the pPNT vector that had been modified with SalI linkers. After electroporation into J1 embryonic stem (ES) cells, colonies were selected with G418 and ganciclovir. Homologous recombinants were identified by digesting genomic DNA from individual ES cell clones with XbaI and analyzing them on Southern blots with a 200-bp 3"-flanking probe amplified by PCR. This probe detects 5- and 8-kb XbaI fragments in mutant and wild-type cells, respectively. Several randomly chosen recombinant ES cell clones were injected into C57/BL6 mouse blastocysts. Two independent mouse strains were generated.
Myogenic regeneration model.
Three-month-old female homo- or heterozygous Pop1-LacZ mice were injected with 0.1 ml of 10 μM cardiotoxin (Sigma, Deisenhofen, Germany), diluted in phosphate-buffered saline (PBS), in the right hind limb muscles (gastrocnemius and soleus muscles) (4, 7). The left side was injected with PBS and served as a control. The muscles from two or three animals per time point were harvested at 6 and 12 h and 1, 2, 5, 10, and 20 days following injection. For histological analysis, muscles were infiltrated with 30% sucrose at 4°C overnight, embedded in Polyfreeze tissue freezing medium (Polyscience), cryosectioned, and stained for β-galactosidase activity.
Induction of cardiac hypertrophy.
Three-month-old male homo- or heterozygous Pop1-LacZ mice (five per genotype) were infused with Iso (Sigma) diluted in PBS containing 0.5 mM ascorbic acid (Sigma) at 30 mg kg−1 day−1 for 3 days via osmotic minipumps (model 2001; Alza Pharmaceuticals, Palo Alto, Calif.) (19). Control animals received pumps filled with the vehicle. Pumps were implanted subcutaneously in the backs of mice that had been anesthetized with Avertin (tribromoethanol in tert-amyl alcohol; Aldrich).
X-Gal staining and immunohistochemistry.
Embryos, cryosections, and satellite cells were fixed with 0.2% glutaraldehyde, permeabilized with 0.01% sodium deoxycholate-0.02% Nonidet P-40, and stained with 5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside (X-Gal) at 0.5 mg/ml-10 mM K3[Fe(CN)6]-10 mM K4[Fe(CN)6]. In some cases, sections were counterstained with eosin. For immunohistochemical staining, cells were refixed with acetone for 10 min at −20°C and blocked with 0.1% bovine serum albumin-20% fetal calf serum-1.5% goat serum in PBS containing 0.1% Triton X-100. A polyclonal antibody for M-cadherin (18) and a monoclonal antibody for desmin (5) were used. Antibody binding was detected by using an avidin-biotin peroxidase system (Vectastain ABC kit; Vector Laboratories). The D3 monoclonal antibody against desmin developed by Donald A. Fishman was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the National Institute of Child Health and Human Development and maintained by the Department of Biological Sciences, University of Iowa, Iowa City.
RT-PCR.
For reverse transcription (RT)-PCR analysis, total RNA was isolated from regenerating hind limb muscles and from various tissues of homozygous, heterozygous, and wild-type animals as previously described (21). cDNA was synthesized from DNase-treated total RNA by using avian myeloblastosis virus reverse transcriptase. PCR was performed with the primer pairs described in Table 1.
TABLE 1.
Primer pairs used in this study
| Product | Primer sequences |
|---|---|
| Pop1 | 5"-CGCAAGGACTTTTCAACATG-3" |
| 5"-ATCTGCGTTGATCTGAATTCGG-3" | |
| Pop3 | 5"-TATCTACTCTTTGCGCAGCATCGG-3" |
| 5"-ATGGGGTGATCTGGACAAGTCTGG-3" | |
| Pop1-LacZ | 5"-CGCAAGGACTTTTCAACATG-3" |
| 5"-CTCTTCGCTATTACGCCAGC-3" | |
| ANF | 5"-GAGAGACGGCAGTGCTTCTAGGC-3" |
| 5"-CGTGACACACCACAAGGGCTTAGG-3" | |
| GATA4 | 5"-GCACAGCCTGCCTGGACG-3" |
| 5"-GCTGCTGCTGCTGCTAGTGG-3" | |
| Myf5 | 5"-TGCCATCCGCTACATTGAGAG-3" |
| 5"-CCGGGGTAGCAGGCTGTGAGTTG-3" | |
| MyoD | 5"-GCAGGCTCTGCTGCGCGACC-3" |
| 5"-TGCAGTCGATCTCTCAAAGCACC-3" | |
| Myogenin | 5"-GAGCGCGATCTCCGCTACAGAGG-3" |
| 5"-CTGGCTTGTGGCAGGCCCAGG-3" | |
| MRF4 | 5"-CTGCGCGAAAGGAGGAGACTAAAG-3" |
| 5"-ATGGAAGAAAGCCGCTGAAGACTG-3" | |
| RPL7 | 5"-GAAGCTCATCTATGAGAAGGC-3" |
| 5"-AAGACGAAGGAGCTGCAGAAC-3" | |
| GAPDHa | 5"-AACTTTGGCATTGTGGAAGG-3" |
| 5"-ATCGAAGGTGGAAGAGTGGG-3" |
GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Satellite cell isolation and culture.
Satellite cells were isolated from hind limb muscles from 2- to 3-month-old heterozygous and homozygous Pop1-LacZ mice as previously described (1, 20). Briefly, hind limb muscles were dissected and dissociated with protease type XIV (Sigma) at 1.25 mg/ml. The suspension was incubated at 37°C for 45 min and triturated every 15 min. The suspension was filtered through 50- and 200-mesh sieves and centrifuged for 15 min at 1,500 × g. The pellet was resuspended in satellite cell medium (Dulbecco modified Eagle medium, 20% fetal calf serum, heparin σ at 5 ng/ml, hepatocyte growth factor [R&D Systems] at 10 ng/ml, penicillin at 200 U/ml, streptomycin at 200 μg/ml, 0.002% amphotericin B [Fungizone]) and preplated for 3 h to remove fibroblasts. Satellite cells in the supernatant were plated on collagen-coated cell culture dishes, and after 48 h, the medium was supplemented with basic fibroblast growth factor at 5 ng/ml. For the induction of muscle differentiation, satellite cells were plated at 100,000 cells per 35-mm-diameter dish. After 24 h, the medium was changed to differentiation medium (Dulbecco modified Eagle medium, 5% horse serum, penicillin at 200 U/ml, streptomycin at 200 μg/ml, 0.002% amphotericin B). Cells were analyzed at 1, 2, 3, 4, and 5 days after serum withdrawal.
RESULTS
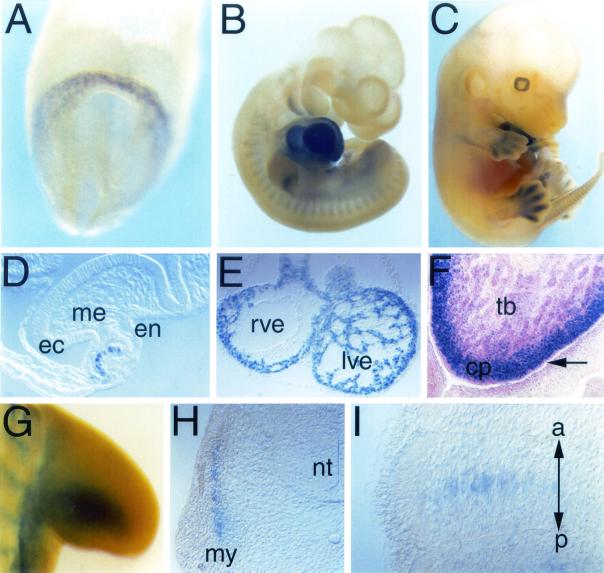
To assess the function of Pop genes in striated muscle, we used homologous recombination to generate a null allele at the Pop1 locus. Gene targeting deleted part of the second (first coding) exon and placed a β-galactosidase-encoding gene containing a simian virus 40 NLS downstream of the ATG of the Pop1 gene (Fig. 1A). Thus, the transcript generated by the Pop1 mutant allele contains the 5" UTR of Pop1 (encoded by the first Pop1 exon) and NLS-LacZ. Homozygous Pop1-LacZ offspring were viable and born with Mendelian frequency (Fig. 1B). No Pop1 mRNA was present in homozygous mice (Fig. 1C). The Pop1 and Pop3 genes are colocalized on chromosome 10 in mice (2) and are organized in a tandem array, with the Pop3 gene located 5" to the Pop1 gene (B. Andrée et al., unpublished observation). We therefore investigated the expression of Pop3 in the mutant background. In skeletal and cardiac muscles, the level of Pop3 mRNA was unaltered in homozygous animals compared to that in heterozygous mutants or wild-type animals. However, a slight increase in Pop3 expression was seen in mutant bladder tissue (Fig. 1C). In order to analyze the developmental expression of Pop1 in further detail, we monitored lacZ reporter gene activity during mouse embryogenesis. Pop1-LacZ was first detected at E7.5 in the mesoderm of the cardiac crescent (Fig. 2A). At E10.5, Pop1-LacZ was prominently expressed in cardiac myocytes of both the atrial and ventricular chambers (Fig. 2B and E). Additional expression was found in the posterior part of the branchial arches, in the myotome, and in a posterior domain in the limb (Fig. 2B and G to I). At E13.5, Pop1-LacZ was still strongly expressed in the heart, mainly in the compact layer myocardium, whereas the trabecular myocardium expressed the gene very weakly (Fig. 2C and F). No expression was present in the endocardium or epicardium. At this stage, Pop1-LacZ expression was also found in the peridigital mesenchyme, the somites of the tail bud (Fig. 2C), the smooth muscle cells of the trachea and the developing bronchial tree, the smooth muscle cells lining the digestive tract, the dorsal root ganglia, and the pancreas anlage (data not shown). Some of these expression domains became visible only after prolonged staining of homozygous embryos.
FIG. 1.
Targeted disruption of Pop1. (A) Restriction map of the Pop1 locus and gene-targeting strategy. The targeting vector was constructed by replacing the coding region of the first coding exon with an NLS-lacZ cassette (β-galactosidase-encoding gene with a simian virus 40 NLS at the 5" end). 3" of the NLS-lacZ gene was a neomycin resistance-encoding gene (neo) in the opposite orientation with respect to the coding region of the Pop1 gene. The left homology arm was a 6.2-kb EcoRI genomic fragment. For the right homology arm, a 1-kb genomic fragment was amplified by PCR. Homologous recombinants were identified by Southern blot analysis using a 200-bp fragment (So) amplified by PCR from genomic DNA (hybridizing to the Pop1 gene 3" to the targeted region). Hybridization of DNA digested with XbaI with So generated 5- and 8-kb fragments from the mutant and wild-type alleles, respectively. Exons are represented by filled boxes. E, EcoRI; X, XbaI; hsv-tk, herpes simplex virus thymidine kinase. (B) Offspring were genotyped by Southern blot analysis. Lanes 1, 4, and 8 contained DNAs from homozygotes, lanes 2, 3, 5, and 6 contained samples from heterozygotes, and lane 7 contained DNA from a wild-type animal. (C) RT-PCR analysis of adult heart, skeletal muscle (skm), and bladder reveals an absence of the Pop1 transcript in mutant mice. Pop3 expression appeared not to be altered in cardiac and skeletal muscle in the absence of Pop1; however, Pop3 was slightly upregulated in the homozygote bladder.
FIG. 2.
Pop1-LacZ expression in heterozygous Pop1-LacZ mouse embryos. LacZ expression was detected after whole-mount staining (A to C and G) or after cryosectioning (F). (D and E) Cryosections of the cardiac region of the embryos shown in panels A and B, respectively. (A and D) E7.5 embryo. LacZ activity is detectable in the cardiogenic mesoderm of the anterior (presumptive ventricular) heart field. (B) E10.5 embryo. Expression is detectable in the heart, branchial arches, and somites and in a posterior domain within the limb. (C) E13.5 embryo. Expression is found in the heart and the peridigital mesenchyme. (E) Section through the heart of the embryo shown in panel B. Myocytes were LacZ positive, while endocardial and epicardial cells were devoid of Pop1-LacZ staining. (F) Section through the heart of an E13.5 embryo stained for Pop1-LacZ activity and counterstained with eosin. Pop1-LacZ activity was confined to the compact layer (cp) myocardium and was strongly diminished or absent from myocytes in the trabecular layer (tb). (G) E10.5 embryo. Pop1-LacZ activity was present within the limb in a posterior domain. (H and I) Transverse (H) and sagittal (I) sections through somites of a E10.5 embryo. Staining is confined to the myotome. The two-headed arrow in panel I indicates the anterior and posterior borders of the somite. Pop1-LacZ appears to label forming myotubes in the center of the somite. ec, ectoderm; en, endoderm; me, mesoderm; my, myotome; nt, neural tube; rve, right ventricle; lve, left ventricle.
At postnatal day 1, the entire myocardium expressed the Pop1-lacZ transgene (Fig. 3A). No expression was found in coronary arteries, cardiac valves, and the vasculature. Expression in atria was weaker than in ventricles (Fig. 3A and A"). Interestingly, Pop1-LacZ expression was rapidly lost from the postnatal myocardium in heterozygous animals but not in homozygous animals. At postnatal day 3, the number of myocyte nuclei that were LacZ positive was reduced and, in addition, LacZ was present in cytoplasmic vesicular structures (Fig. 3B and B"). At postnatal day 8, nuclear LacZ activity was further reduced and the number of LacZ-stained cytoplasmic vesicles increased (Fig. 3C and C"). In the adult heart, only faint cytoplasmic LacZ activity was detectable (Fig. 3D and D"). In contrast to the observed loss of Pop1-LacZ activity in heterozygous hearts, the transcripts of both the endogenous Pop1 gene and the targeted allele were present in hearts of neonatal and postnatal day 8 and 14 mice (Fig. 3E). At the ages of 3 and 8 months, a reduced level of expression of both genes was observed. Evidently, the time course of Pop1-LacZ activity did not correlate with the transcript levels of endogenous Pop1 and Pop1-LacZ hybrid mRNAs. In order to compare LacZ expression directly between heterozygous and homozygous animals, sections of adult heart and skeletal muscles were stained for LacZ activity. In contrast to the faint expression in heterozygous animals, homozygotes showed strong nuclear LacZ activity in both the atrium and ventricle (Fig. 4A to D). Quantification of β-galactosidase enzyme activity revealed approximately ninefold higher levels in tissue extracts from homozygous hearts than in those from heterozygous hearts. Nuclear LacZ staining was also observed in skeletal muscle of homozygous adult animals, albeit at a very low frequency, but not in that of heterozygous adult animals (Fig. 4E and F). Taken together, these observations suggest that Pop1-LacZ expression in the postnatal heart may be under posttranscriptional control.
FIG. 3.
Pop1-LacZ expression in postnatal hearts from heterozygous Pop1-LacZ mice. LacZ expression was detected in frozen sections of postnatal day 1 (A and A"), 3 (B and B"), and 8 (C and C") and adult (3 months old; D and D") hearts. Panels A" to D" are high-power views of sections of hearts of the same ages as those shown in panels A to D that were stained for β-galactosidase activity. The section in panel A" was incubated for 5 h, while the sections shown in panels B" to D" were incubated for 16 h in order to visualize staining in myocyte nuclei (arrows) and cytoplasmic vesicles (arrowheads), respectively. (E) RT-PCR analysis of Pop1, Pop1-LacZ, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) expression in cardiac muscle at postnatal days 1, 8, 14, 90, and 240.
FIG. 4.
Pop1-LacZ activity in heart and skeletal muscles of heterozygous (A, C, and E) and homozygous (B, D, and F) Pop1-LacZ animals. Panels: A and B, ventricle; C and D, atrium; E and F, skeletal muscle.
The high level of Pop1 expression in developing skeletal and cardiac muscles and its downregulation in adult organs prompted us to examine Pop1 expression under conditions of inducible growth. Infusion of the β-adrenergic agonist Iso induces myocardial hypertrophy (3, 19). In order to examine whether Pop1-LacZ may respond to hypertrophic signals, homo- and heterozygous mutants were infused for 3 days with Iso at 30 mg kg−1 day−1. A 30% increase in the heart-to-body weight ratio was found in treated animals. This hypertrophy was independently of the genotype. Nuclear Pop1-LacZ activity was strongly elevated in hearts of Iso-treated heterozygous animals (Fig. 5A and B). However, no increase in Pop1-LacZ mRNA was observed although transcription of GATA4 and ANF was induced, as expected in hypertrophic hearts (Fig. 5C).
FIG. 5.
Pop1-LacZ activity in ventricular tissue of heterozygous Pop1-LacZ animals after 3 days of infusion of PBS (A) and Iso (B). (C) RT-PCR analysis of Pop1-LacZ. Pop1, GATA4, ANF, and RPL7 mRNA expression in ventricular tissue of PBS- and Iso-infused animals 3 days after pump implantation. The arrows in panel B indicate nuclear LacZ activity.
Injection of cardiotoxin is an established model for induction of lesions in skeletal muscle and its regeneration (4, 7). In order to compare the regenerative capacities of homozygous and heterozygous Pop1-LacZ mice, cardiotoxin was injected into the hind limbs and muscle tissue was isolated 6 and 12 h and 1, 2, 5, 10, and 20 days later. Two days after cardiotoxin injection, mononuclear cells and small myotubes were found to be LacZ positive independently of the genotype. Costaining with M-cadherin revealed that cells positively stained for LacZ were also labeled by the M-cadherin antibody (data not shown). Five days after cardiotoxin injection, many regenerated fibers appeared and only few fibers remained LacZ positive in heterozygous animals whereas only small myotubes with prominent Pop1-LacZ activity were present in homozygous Pop1-LacZ mice (Fig. 6A). After 10 days, heterozygous mice had fully regenerated the muscle with the lesion, with very few nuclei still LacZ positive, while the tissue architecture in homozygous mutant mice still appeared markedly disorganized (Fig. 6A). New myotubes were still small or intermediate in size and strongly expressed Pop1-LacZ. Twenty days after injury, LacZ-positive nuclei were centrally positioned in all regenerated myofibers and the tissue architecture appeared essentially indistinguishable in homozygous and heterozygous mice (Fig. 6A). We also studied expression of myogenic determination factors (MyoD, myogenin, Myf5, and MRF4) in regenerating muscle of homo- and heterozygous animals (Fig. 6B). In homozygous Pop1-LacZ mice, expression of MyoD, Myf5, and particularly myogenin appeared to be prolonged, with considerable transcript levels still present at 10 and 20 days after cardiotoxin injection. This confirms the retarded regeneration in homozygous Pop1-LacZ mice that was also seen histologically. Expression of Pop1-LacZ hybrid mRNA was similar in mice of both genotypes and did not correlate with the time course of Pop1-LacZ activity (Fig. 6B).
FIG. 6.
Morphological and molecular analysis of skeletal muscle after injection of cardiotoxin. (A) Pop1-LacZ activity in heterozygous (+/−) and homozygous (−/−) mice at 5, 10, and 20 days after cardiotoxin injection. (B) RT-PCR analysis of myogenin, MyoD, Myf5, MRF4, Pop1, Pop1-LacZ, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) in skeletal muscle tissue that was excised at the indicated times after cardiotoxin injection.
Myotube formation was further studied in isolated satellite cells of heterozygous and homozygous Pop1-LacZ animals. No apparent difference in yield and proliferative capacity was observed for either genotype. Satellite cells cultured in growth medium displayed nuclear Pop1-LacZ expression (Fig. 7A and E), and costaining with M-cadherin revealed that the majority of cells positively stained for both LacZ and M-cadherin (Fig. 7B and F). Satellite cells of both genotypes were equally able to undergo myotube formation; however, differentiation appeared to be retarded. Nuclear LacZ activity was downregulated immediately after myotube formation in heterozygous satellite cells, while it persisted in homozygous cells (Fig. 7C and G). After 5 days in differentiation medium, nuclear Pop1-LacZ activity was also downregulated in homozygous myotubes (Fig. 7D and H).
FIG. 7.
Differentiation of adult satellite cells isolated from skeletal muscle of heterozygous (+/−) and homozygous (−/−) Pop1-LacZ mice. (A and E) Nuclear Pop1-LacZ activity in satellite cells cultured in growth medium. (B and F) Expression of M-cadherin (brown staining) and Pop1-LacZ in satellite cells cultured in growth medium. Arrowheads demarcate cells that express M-cadherin but not Pop1-LacZ. (C and G) Pop1-LacZ expression in cells cultured for 3 days in differentiation medium. In satellite cells derived from heterozygous mice (C), LacZ activity is downregulated in myotubular nuclei (arrowhead) while myoblasts display nuclear LacZ (arrows). In contrast, in satellite cells derived from homozygous muscle (G), nuclear LacZ activity is present in both unfused myoblasts (arrow in the middle) and myotubes (arrows on the left and right). (D and H) Satellite cells cultured for 5 days in differentiation medium and stained for MF20 and Pop1-LacZ. Irrespective of the genotype, nuclear LacZ staining is lost from myotubes (arrowheads); however, myoblasts (arrows) display nuclear lacZ expression.
DISCUSSION
Targeted disruption of the Pop1 gene reveals its critical role during muscle regeneration in the adult mouse. Pop1 seems not to be essential for correct development of cardiac and skeletal muscles or of any other organ in which Pop1 is expressed. Pop1 is also not required for induction of cardiac hypertrophy or for establishment of the satellite cell population within adult muscle tissues. However, Pop1 seems to be essential for molecular and cellular events during skeletal muscle regeneration. In the absence of Pop1, skeletal muscle regeneration appears to be less efficient and delayed. At 5 and 10 days after cardiotoxin injection, the muscle architecture remains more disorganized in homozygous than in heterozygous mutants. While there is a kinetic difference in muscle regeneration, at 20 days after muscle injury, lesions were essentially repaired in mice of both genotypes.
Pop1-LacZ activity and expression of Pop1 mRNA do not always correlate.
The lacZ reporter gene was placed downstream of the endogenous ATG in the second exon of Pop1. The rest of the second exon was deleted; however, no intronic gene sequences were missing and thus, all of the regulatory elements controlling the expression should be present in the targeted locus. We therefore expected that Pop1-LacZ distribution would largely coincide with the Pop1 expression pattern. Pop1-LacZ was first detected at E7.5 in the anterior heart field and between E7.5 and E10.5 in the entire myocardium. In contrast, Pop1 mRNA was first detectable at E9.5 in atrial tissue and in a few ventricular cells restricted to the dorsal side (2). Possible explanations for this difference include the higher sensitivity of enzyme detection or a difference in stability between LacZ protein and Pop1 mRNA.
At midgestation, Pop1-LacZ was differentially expressed in the ventricular wall; i.e., expression was prominent in the compact layer myocardium and faint in trabecular myocytes. This was also observed for Pop1 mRNA (2). Maturation of the ventricular wall involves the formation of the trabecular layer. Myocytes in this tissue are more differentiated than myocytes of the subepicardial compact layer (9). Cardiac trabeculation is dependent on interaction of the endocardium and myocardium and involves the endocardium-derived neuregulin signal, which is received by a myocardium-expressed receptor complex of erbB2 and erbB4 (8, 11, 14). The subepicardial compact layer shows severalfold higher rates of proliferation than the trabecular layer (23). Maintenance of the proliferative activity and inhibition of myocardial differentiation of myocytes in the compact layer are probably controlled by the epicardium and involve, among other signals, retinoic acid. Mutations of both the retinoic acid receptor RXRα and RALDH2 cause premature myocardial differentiation and result in ventricular malformations (9, 16). Whether the differential expression of Pop1-LacZ in the midgestation ventricle is the result of an inductive signal, possibly from the epicardium, or is due to repression in the trabecular layer remains to be clarified.
Despite prominent expression of Pop1 mRNA in neonatal and adult hearts, Pop1-LacZ was downregulated during the first week of postnatal life. These data suggest that Pop1 is under posttranscriptional control. This hypothesis was corroborated by the fact that in heterozygous mice, LacZ activity was upregulated by Iso treatment with no alterations at the transcript level. In the Pop1-LacZ hybrid mRNA, the 5" UTR of the endogenous Pop1 transcript is present while the 3" UTR is not. The 5" UTR may therefore be a target for posttranscriptional control. Based on a computer algorithm (mfold [http://bioinfo.math.rpi.edu/∼mfold/rna/form1.cgi]) (13), a stem-loop structure with a free enthalpy of −108.2 kcal mol−1 is predicted for the 5" UTR of mouse Pop1. It has been proposed that stem-loop structures have a repressive influence on translation (10). For example, biosynthesis of the tumor suppressor gene that encodes p53 is regulated at the translational level and a stem-loop structure is found in the 5" UTR of p53 (15). Interestingly, the p53 protein is involved in an autoregulatory negative feedback loop that inhibits its own translation. Based on the observation that, in homozygous animals, Pop1-LacZ activity remained high in the postnatal heart, one can hypothesize that endogenous Pop1 protein negatively controls its own translation.
In skeletal muscle, Pop1-LacZ activity was downregulated during myogenic differentiation in the embryo. Only a few muscle nuclei were labeled in homozygous adult skeletal muscle, possibly representing newly regenerated muscle fibers. After cardiotoxin injection, transient Pop1-LacZ expression in activated satellite cells and in newly forming myotubes was observed in heterozygous mice. In homozygous mice, however, nuclear Pop1-LacZ expression persisted for a much longer time. This observation is also compatible with the hypothesis that Pop1 negatively controls its own translation and therefore the protein persists in the null mutant. Alternatively, the prolonged Pop1-LacZ expression in the regenerating muscle of the null mutant might be caused by the slow progression of muscle regeneration.
The timing of repression of Pop1-LacZ activity in the postnatal heart correlates with the myocardial transition from hyperplastic to hypertrophic growth.
The postnatal myocardium showed rapid inactivation of Pop1-LacZ activity. The timing of downregulation of Pop1 correlates well with the transition from hyperplastic to hypertrophic growth in the myocardium (22). In the embryonic heart at E13.5, Pop1-LacZ expression largely overlapped the bromodeoxyuridine-positive, mitotically active myocardium, while the expression domains of Pop1-LacZ and the cyclin-dependent kinase (CDK) inhibitor p57Kip2 label nonoverlapping cell populations (A. Fleige et al. and B. Andrée et al., unpublished observations). Pop1 became reexpressed in the hypertrophic myocardium, and cardiac hypertrophy has several elements in common with hyperplastic growth, including the induction of cyclins D2 and D3 and CDK4/6 kinases and downregulation of CDK inhibitors p21Waf1 and p27Kip1 (12). It is unclear, however, whether Pop1 has any functional role in growth regulation or merely labels the immature myocardium.
The rather mild phenotype of the Pop1-LacZ mouse was unexpected given the strong expression of Pop1 in the developing heart and skeletal muscles. However, Pop1 is a member of the Pop gene family, which consists of three members (2). While the homology between the different family members is rather limited (at the protein level, Pop1 is 24 and 28% identical to Pop2 and -3, respectively, while Pop2 and -3 are approximately 50% identical [2]), it is still possible that the proteins can functionally substitute for each other. It will therefore be necessary to inactivate all three members separately and in combination to genetically define the function of the Pop gene family.
The function of the protein encoded by Pop1 is unknown. Pop genes encode membrane proteins with two or three predicted transmembrane domains. Recent biochemical and cell biological analysis indicates that the C terminus locates to the cytosol and the N terminus becomes glycosylated (Andrée et al., unpublished). Expression analysis of green fluorescent protein- and myc-tagged constructs in Cos7 and C2C12 cells indicated that Pop1 goes to the endoplasmic reticulum/Golgi compartment and to the cell membrane (2). Unfortunately, no Pop-interacting protein has been identified that could help to place Pop proteins in the context of known cellular pathways. A recent report proposed that Pop1/bves might be a cell adhesion molecule that mediates cell-cell interaction in a calcium-independent manner (25). This adhesion function would be consistent with the observed impairment of skeletal muscle regeneration in the null mutant. However, in cell culture, we found no difference between satellite cells derived from the null mutant and wild-type cells in the ability to undergo myotube formation. An important goal for the future is to further define the cellular and biochemical activities of Pop1 and the other members of the Pop gene family.
Acknowledgments
We gratefully acknowledge the expert technical assistance of Kerstin Zander and Stefanie Willenzon. We thank Anna Starzinski-Powitz for donation of the M-cadherin antibody and the Resource Center of the German Human Genome Project at the Max Planck Institute for Molecular Genetics for providing high-density filters.
This work was supported by Sonderforschungsbereich 271, TP B10.
REFERENCES
- 1.Allen, R., C. Temm-Grove, S. Sheehan, and G. Rice. 1997. Methods in muscle biology. Methods Cell Biol. 52:155-176. [DOI] [PubMed] [Google Scholar]
- 2.Andrée, B., T. Hillemann, G. Kessler-Icekson, T. Schmitt-John, H. Jockusch, H. H. Arnold, and T. Brand. 2000. Isolation and characterization of the novel popeye gene family expressed in skeletal muscle and heart. Dev. Biol. 223:371-382. [DOI] [PubMed] [Google Scholar]
- 3.Brand, T., H. S. Sharma, and W. Schaper. 1993. Expression of nuclear proto-oncogenes in isoproterenol-induced cardiac hypertrophy. J. Mol. Cell. Cardiol. 25:1325-1337. [DOI] [PubMed] [Google Scholar]
- 4.d'Albis, A., R. Couteaux, C. Janmot, and J. C. Mira. 1989. Myosin isoform transitions in regeneration of fast and slow muscles during postnatal development of the rat. Dev. Biol. 135:320-325. [DOI] [PubMed] [Google Scholar]
- 5.Danto, S. I., and D. A. Fischman. 1984. Immunocytochemical analysis of intermediate filaments in embryonic heart cells with monoclonal antibodies to desmin. J. Cell Biol. 98:2179-2191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fire, A., S. W. Harrison, and D. Dixon. 1990. A modular set of lacZ fusion vectors for studying gene expression in Caenorhabditis elegans. Gene 93:189-198. [DOI] [PubMed] [Google Scholar]
- 7.Garry, D. J., Q. Yang, R. Bassel-Duby, and R. S. Williams. 1997. Persistent expression of MNF identifies myogenic stem cells in postnatal muscles. Dev. Biol. 188:280-294. [DOI] [PubMed] [Google Scholar]
- 8.Gassmann, M., F. Casagranda, D. Orioli, H. Simon, C. Lai, R. Klein, and G. Lemke. 1995. Aberrant neural and cardiac development in mice lacking the ErbB4 neuregulin receptor. Nature 378:390-394. [DOI] [PubMed] [Google Scholar]
- 9.Kastner, P., N. Messaddeq, M. Mark, O. Wendling, J. M. Grondona, S. Ward, N. Ghyselinck, and P. Chambon. 1997. Vitamin A deficiency and mutations of RXRalpha, RXRbeta and RARalpha lead to early differentiation of embryonic ventricular cardiomyocytes. Development 124:4749-4758. [DOI] [PubMed] [Google Scholar]
- 10.Kozak, M. 1986. Influences of mRNA secondary structure on initiation by eukaryotic ribosomes. Proc. Natl. Acad. Sci. USA 83:2850-2854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee, K.-F., H. Simon, H. Chen, B. Bates, M.-C. Hung, and C. Hauser. 1995. Requirement for neuregulin receptor erbB2 in neural and cardiac development. Nature 378:394-398. [DOI] [PubMed] [Google Scholar]
- 12.Li, J. M., and G. Brooks. 1997. Downregulation of cyclin-dependent kinase inhibitors p21 and p27 in pressure-overload hypertrophy. Am. J. Physiol. 273:H1358-H1367. [DOI] [PubMed] [Google Scholar]
- 13.Mathews, D., J. Sabina, M. Zuker, and D. Turner. 1999. Expanded sequence dependence of thermodynamic parameters improves prediction of RNA secondary structure. J. Mol. Biol. 288:911-940. [DOI] [PubMed] [Google Scholar]
- 14.Meyer, D., and C. Birchmeier. 1995. Multiple essential functions of neuregulin in development. Nature 378:386-390. [DOI] [PubMed] [Google Scholar]
- 15.Mosner, J., T. Mummenbrauer, C. Bauer, G. Sczakiel, F. Grosse, and W. Deppert. 1995. Negative feedback regulation of wild-type p53 biosynthesis. EMBO J. 14:4442-4449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Niederreither, K., J. Vermot, N. Messaddeq, B. Schuhbaur, P. Chambon, and P. Dolle. 2001. Embryonic retinoic acid synthesis is essential for heart morphogenesis in the mouse. Development 128:1019-1031. [DOI] [PubMed] [Google Scholar]
- 17.Reese, D. E., and D. M. Bader. 1999. Cloning and expression of hbves, a novel and highly conserved mRNA expressed in the developing and adult heart and skeletal muscle in the human. Mammal. Genome 10:913-915. [DOI] [PubMed] [Google Scholar]
- 18.Rose, O., J. Rohwedel, S. Reinhardt, M. Bachmann, M. Cramer, M. Rotter, A. Wobus, and A. Starzinski-Powitz. 1994. Expression of M-cadherin protein in myogenic cells during prenatal mouse development and differentiation of embryonic stem cells in culture. Dev. Dyn. 201:245-259. [DOI] [PubMed] [Google Scholar]
- 19.Saadane, N., L. Alpert, and L. E. Chalifour. 1999. Expression of immediate early genes, GATA-4, and Nkx-2.5 in adrenergic-induced cardiac hypertrophy and during regression in adult mice. Br. J. Pharmacol. 127:1165-1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sabourin, L. A., A. Girgis-Gabardo, P. Seale, A. Asakura, and M. A. Rudnicki. 1999. Reduced differentiation potential of primary MyoD−/− myogenic cells derived from adult skeletal muscle. J. Cell Biol. 144:631-643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schlange, T., B. Andree, H. H. Arnold, and T. Brand. 2000. BMP2 is required for early heart development during a distinct time period. Mech. Dev. 91:259-270. [DOI] [PubMed] [Google Scholar]
- 22.Soonpaa, M. H., and L. J. Field. 1994. Assessment of cardiomyocyte DNA synthesis during hypertrophy in adult mice. Am. J. Physiol. 266:H1439-H1445. [DOI] [PubMed] [Google Scholar]
- 23.Tokuyasu, K. 1990. Co-development of embryonic myocardium and myocardial circulation, p. 205-218. In E. Clark and A. Takao (ed.), Developmental cardiology: morphogenesis and function. Futura, Mt. Kisco, N.Y.
- 24.Tybulewicz, V. L., C. E. Crawford, P. K. Jackson, R. T. Bronson, and R. C. Mulligan. 1991. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65:1153-1163. [DOI] [PubMed] [Google Scholar]
- 25.Wada, A., D. Reese, and D. Bader. 2001. Bves: prototype of a new class of cell adhesion molecules expressed during coronary artery development. Development 128:2085-2093. [DOI] [PubMed] [Google Scholar]