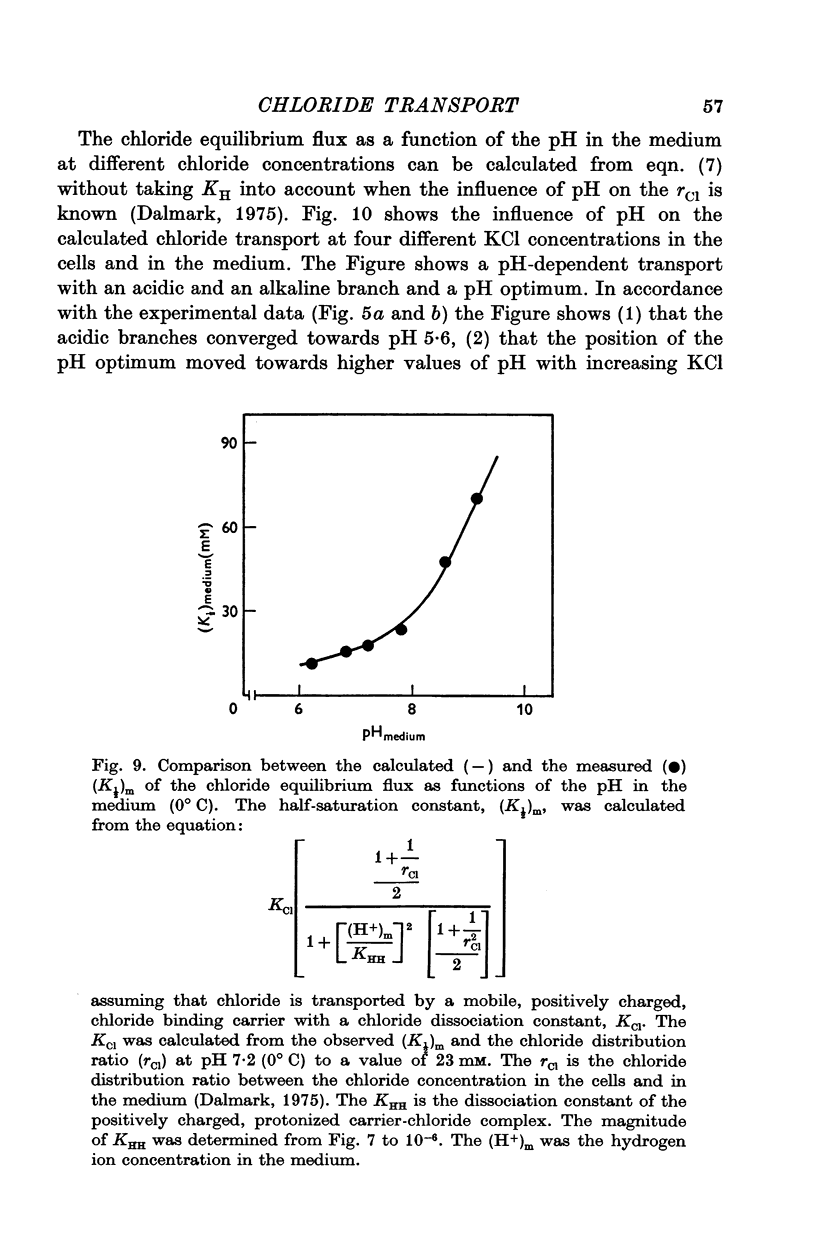
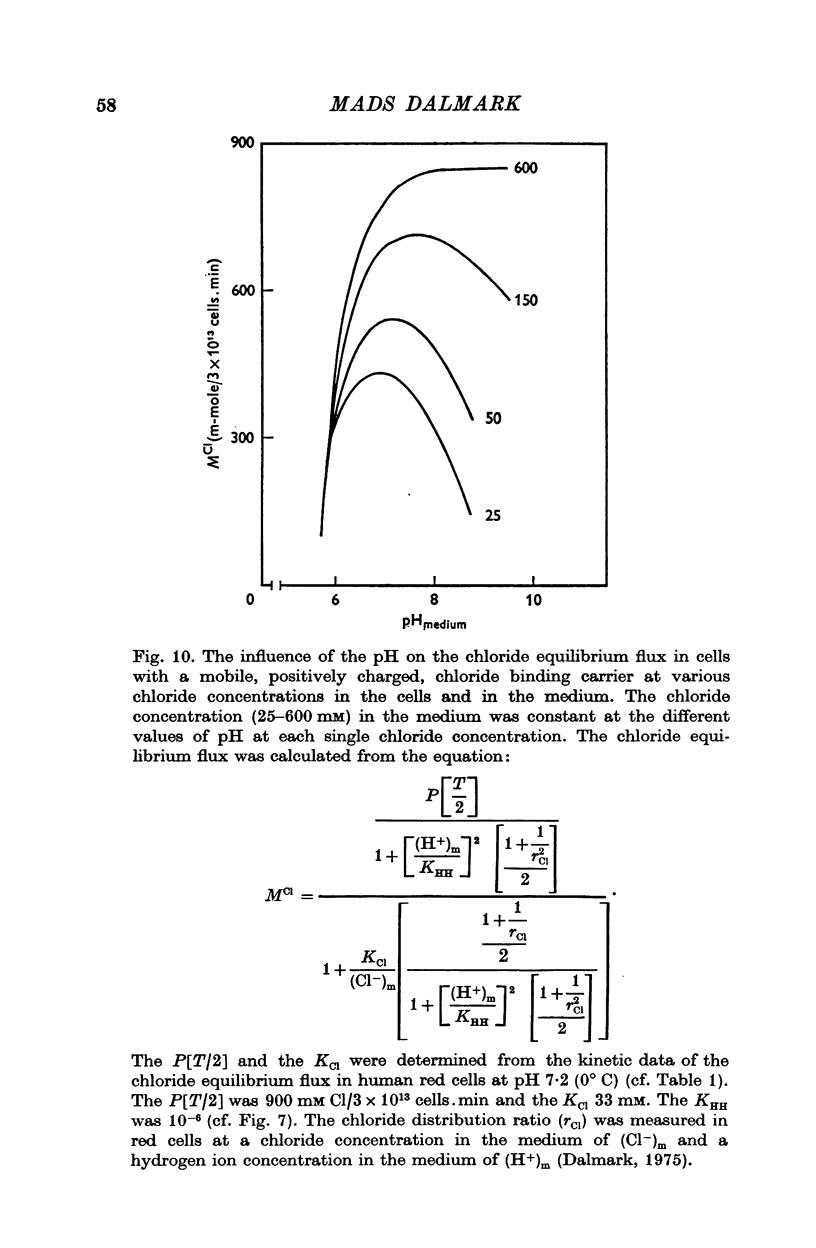
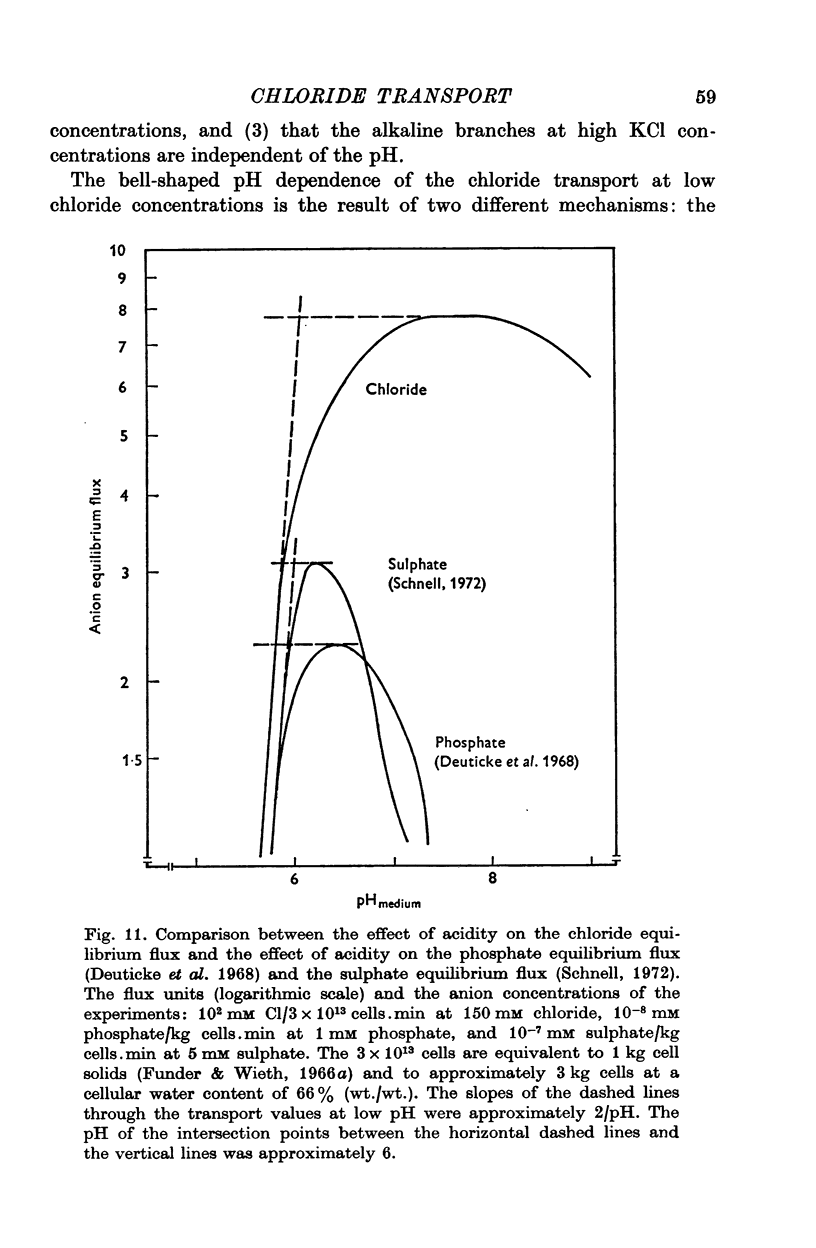
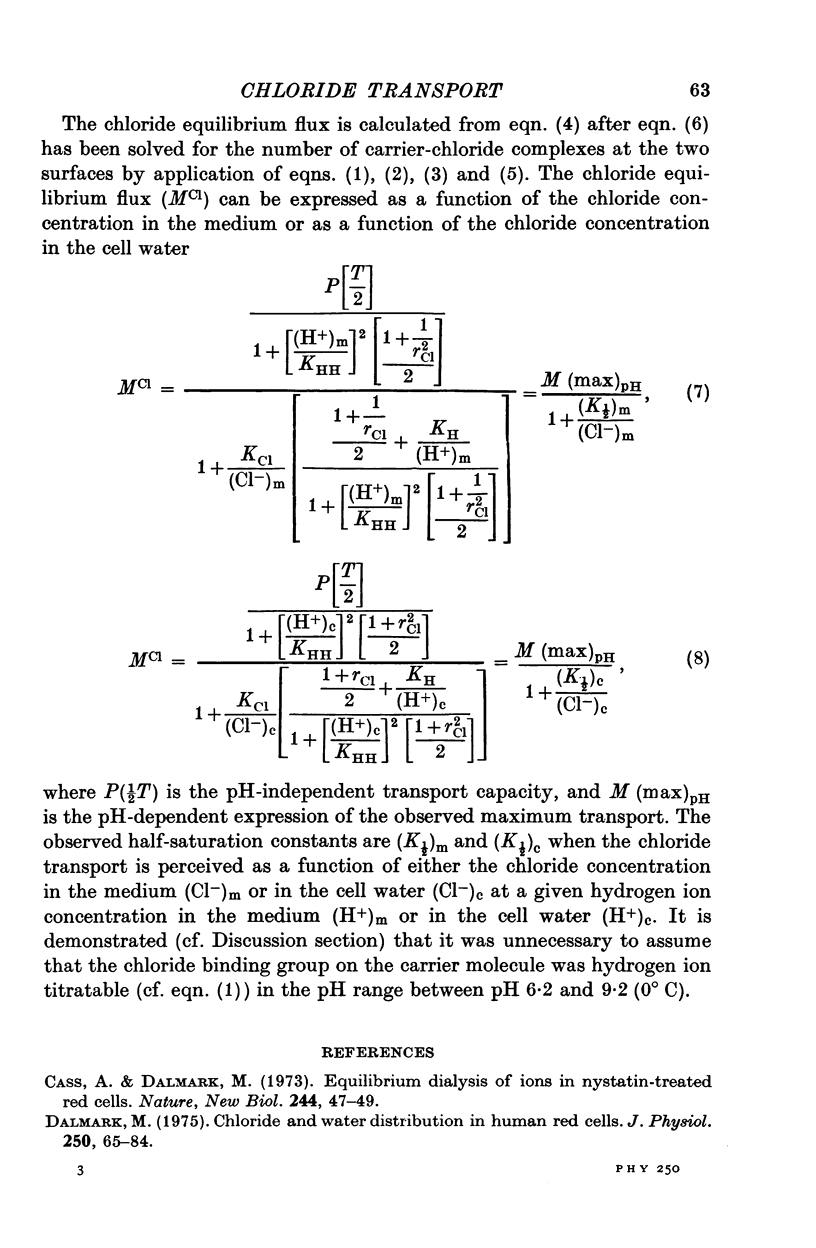
Abstract
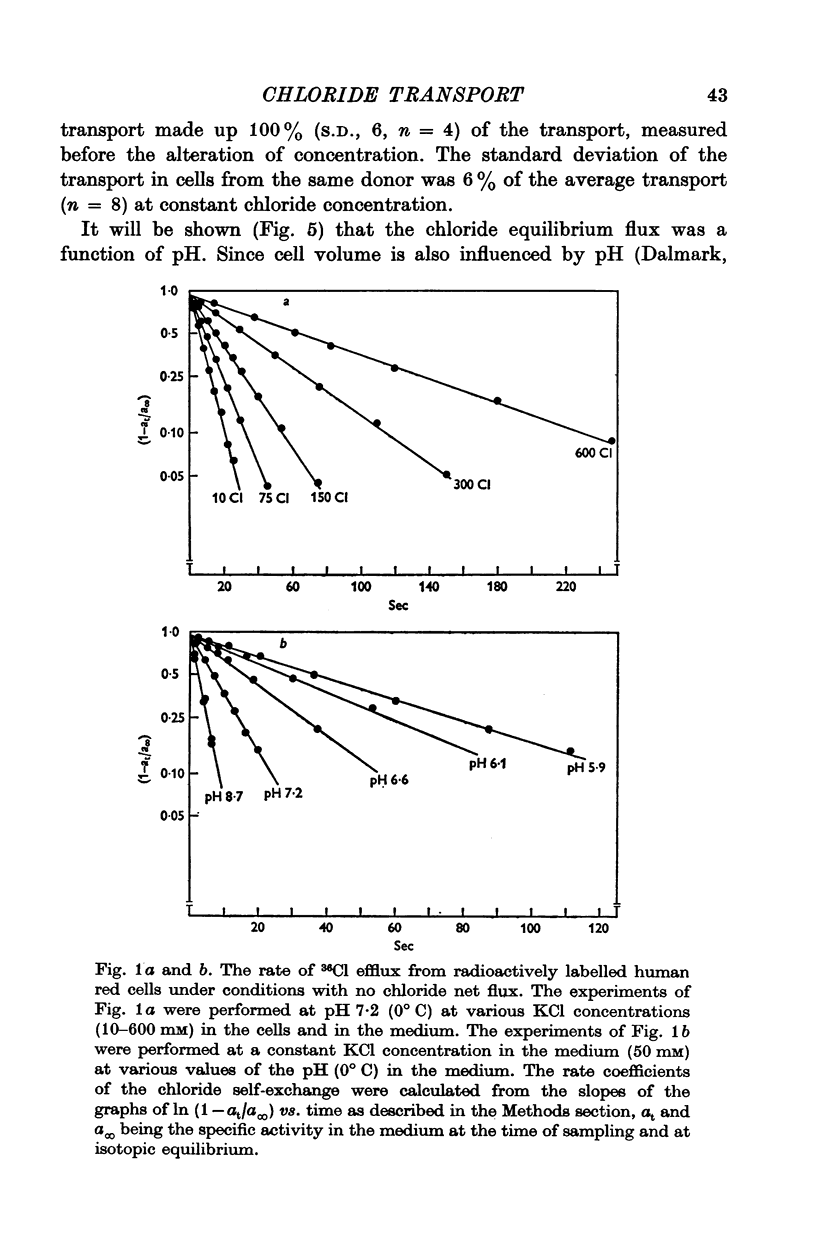
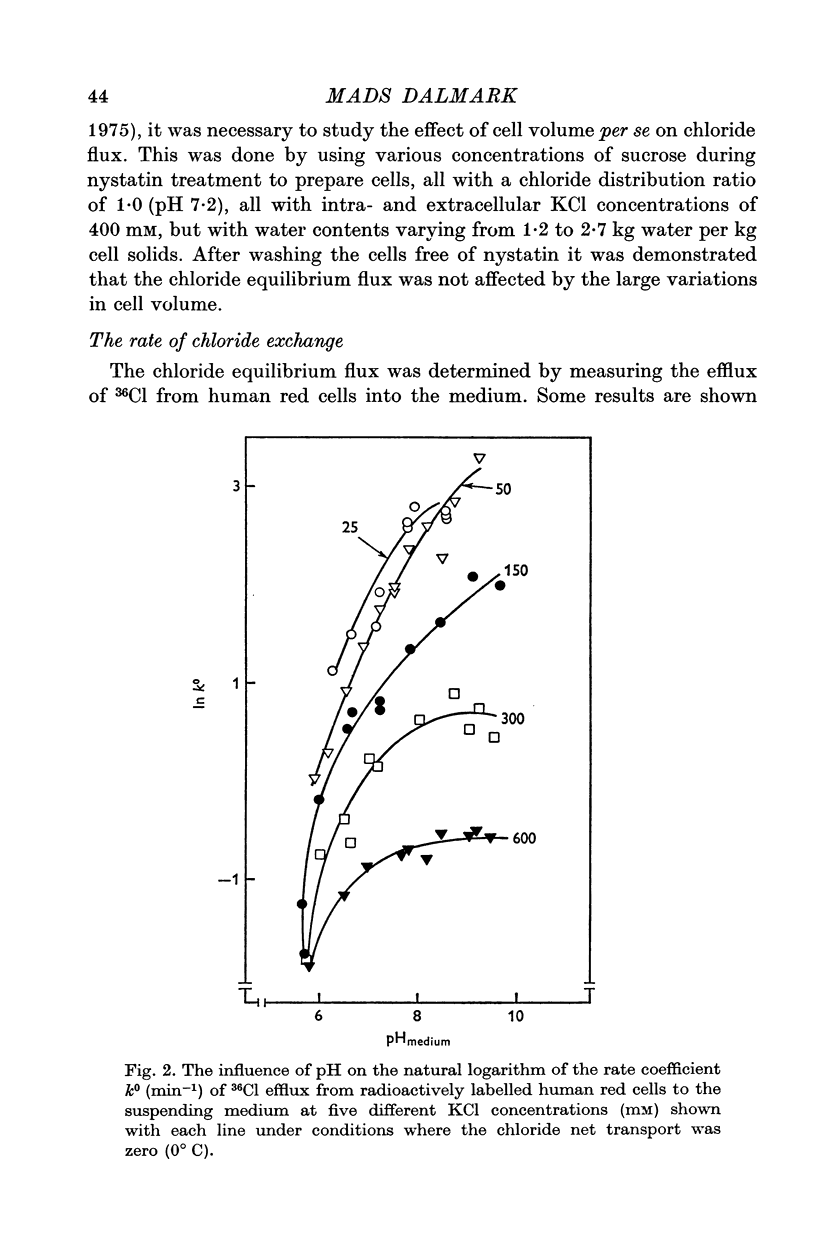
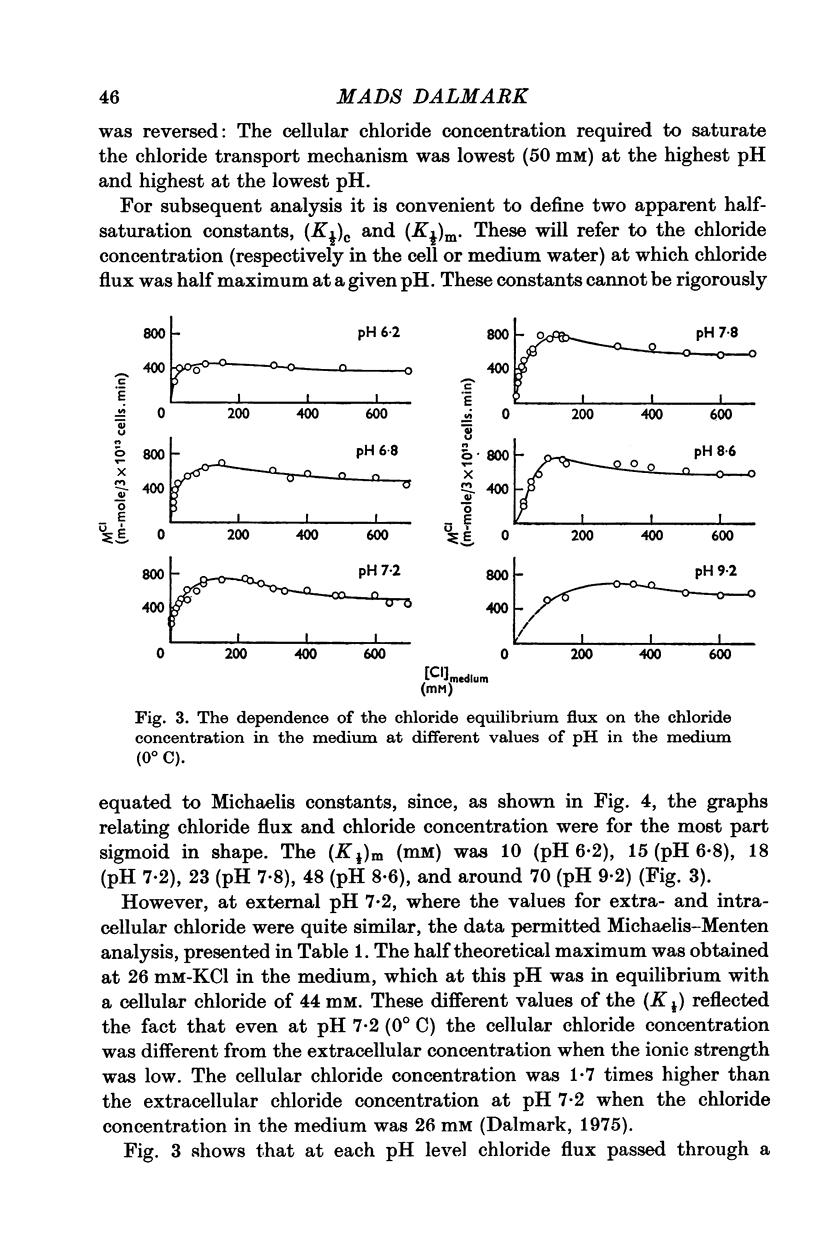
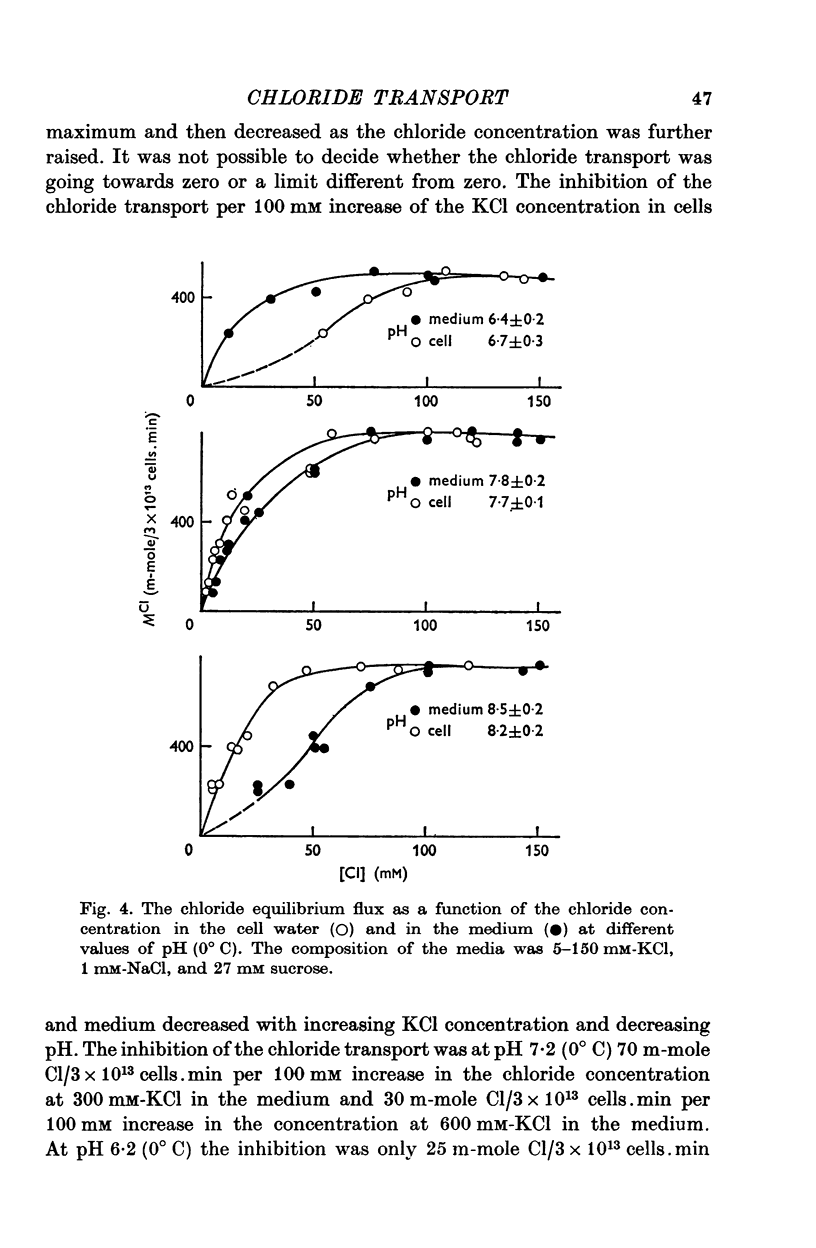
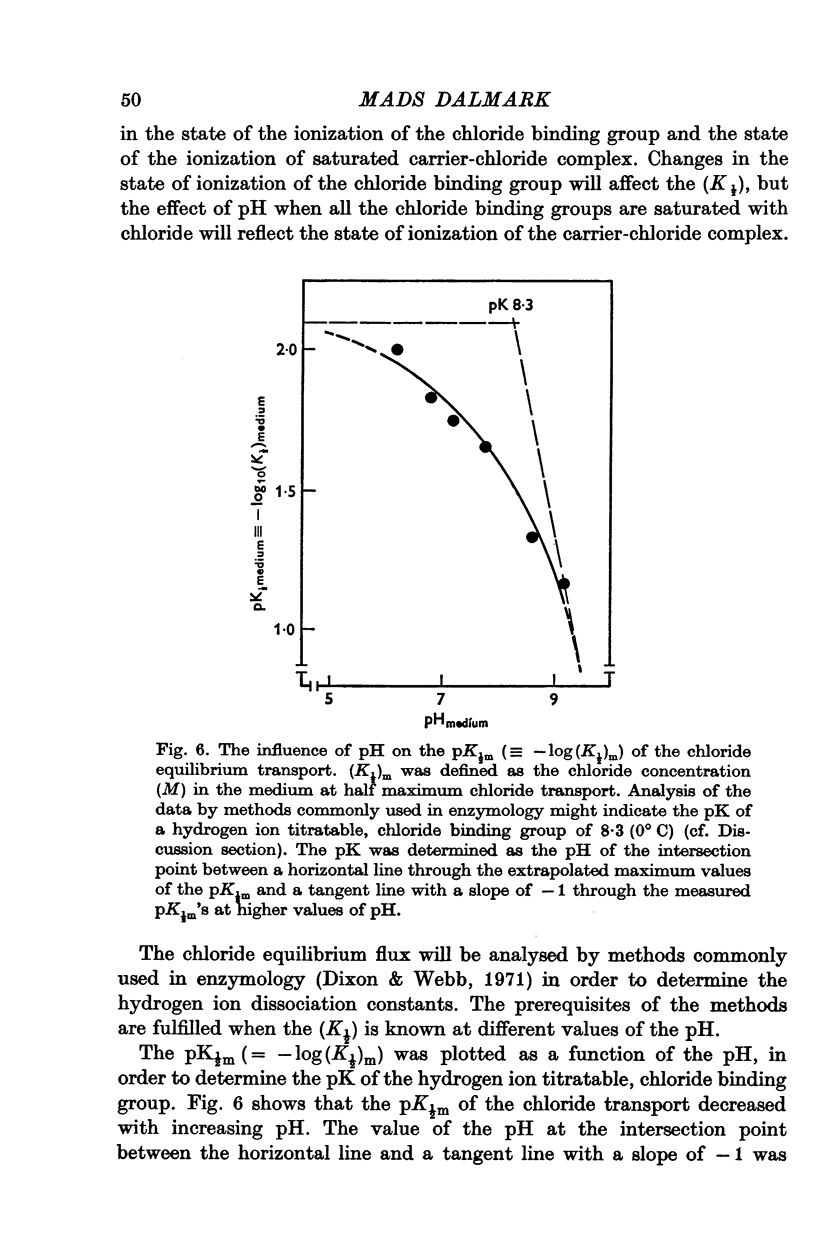
1. The chloride equilibrium flux (chloride self-exchange) was determined by measuring the rate of 36Cl efflux from radioactively labelled human red cells. The cellular chloride concentration was varied between 5 and 700 mM by the nystatin technique (Cass & Dalmark, 1973). The chloride transport capacity was not affected by the nystatin technique. 2. The chloride equilibrium flux showed saturation kinetics in the pH range between 6-2 and 9-2 (0 degrees C). The chloride transport decreased at chloride concentrations higher than those which gave the maximum transport. 3. The apparent half-saturation constant, (K1/2), depended on the pH and whether the chloride transport was perceived as a function of the chloride concentration in the medium or in the cell water. The (K1/2)m increased and the (K1/2)c decreased with increasing pH. The dependence of the chloride transport on the chloride concentration was described by Michaelis-Menten kinetics at pH 7-2, but at values of pH outside pH 7-8 S-shaped or steeper graphs were observed. 4. The chloride equilibrium flux varied with the pH at constant chloride concentration in the medium (pH 5-7-9-5). The transport had a bell-shaped pH dependence at chloride concentrations below 200 mM. At chloride concentrations between 300 and 600 mM the chloride transport increased with increasing pH to reach a plateau around pH 8. The position of the acidic branches of the pH graphs was independent of the chloride concentration (25-600 mM), but the position of the alkaline branches moved towards higher values of pH with increasing chloride concentration (5-150 mM). Thus, the position of the pH optimum increased with increasing chloride concentration. The chloride transport at low pH values was a function of the inverse second power of the hydrogen ion concentration. The pK of the groups which caused the inhibition was approximately 6 and independent of the temperature (0-18 degrees C). 5. The chloride equilibrium flux as a function of chloride concentration, pH, and temperature could be described by a transport model with a mobile, positively charged, chloride binding carrier with a single chloride dissociation constant of 33 mM, a transport capacity of 900 m-mole/3 x 10(13) cells.min (pH 7-2, 0 degrees C), and an Arrhenius activation energy of 30 kcal/mole. The pH dependence of the transport of inorganic monovalent and divalent anions is discussed in relation to the suggested model.
Full text
PDF





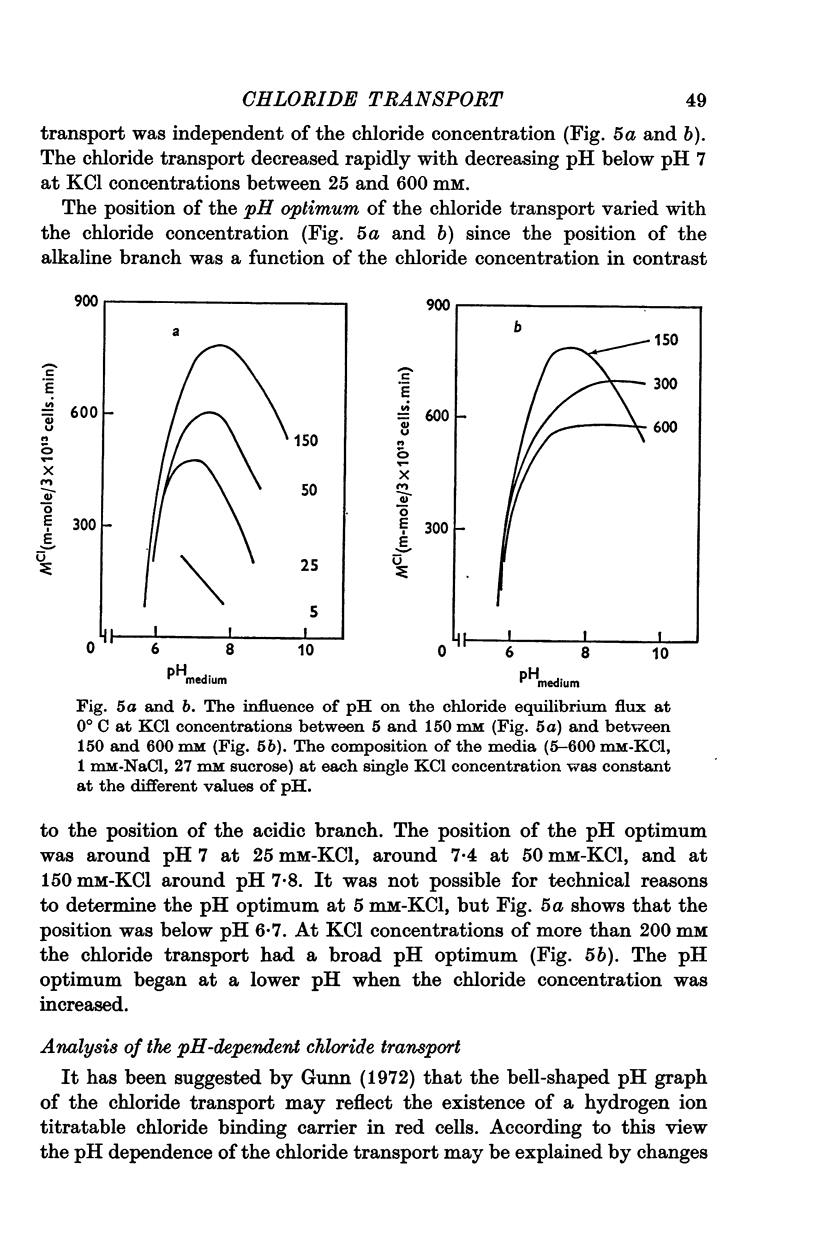
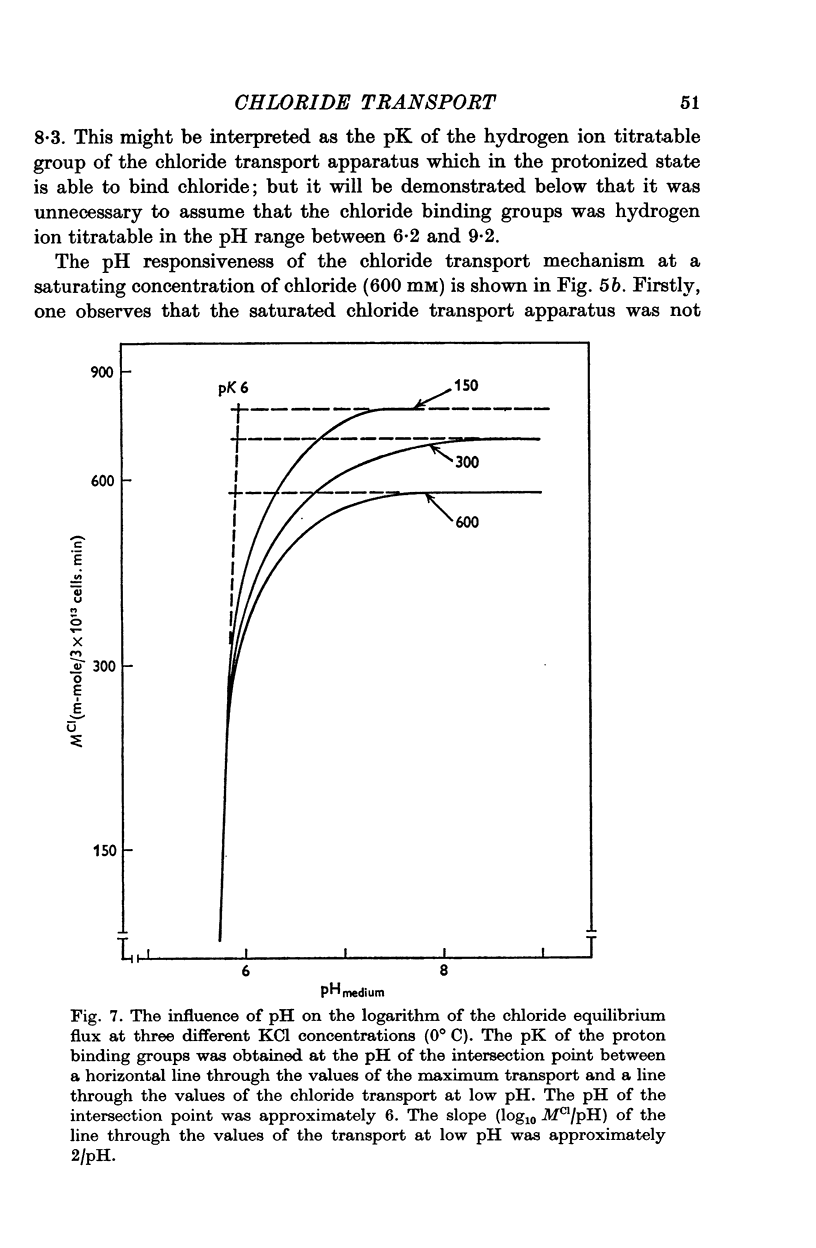

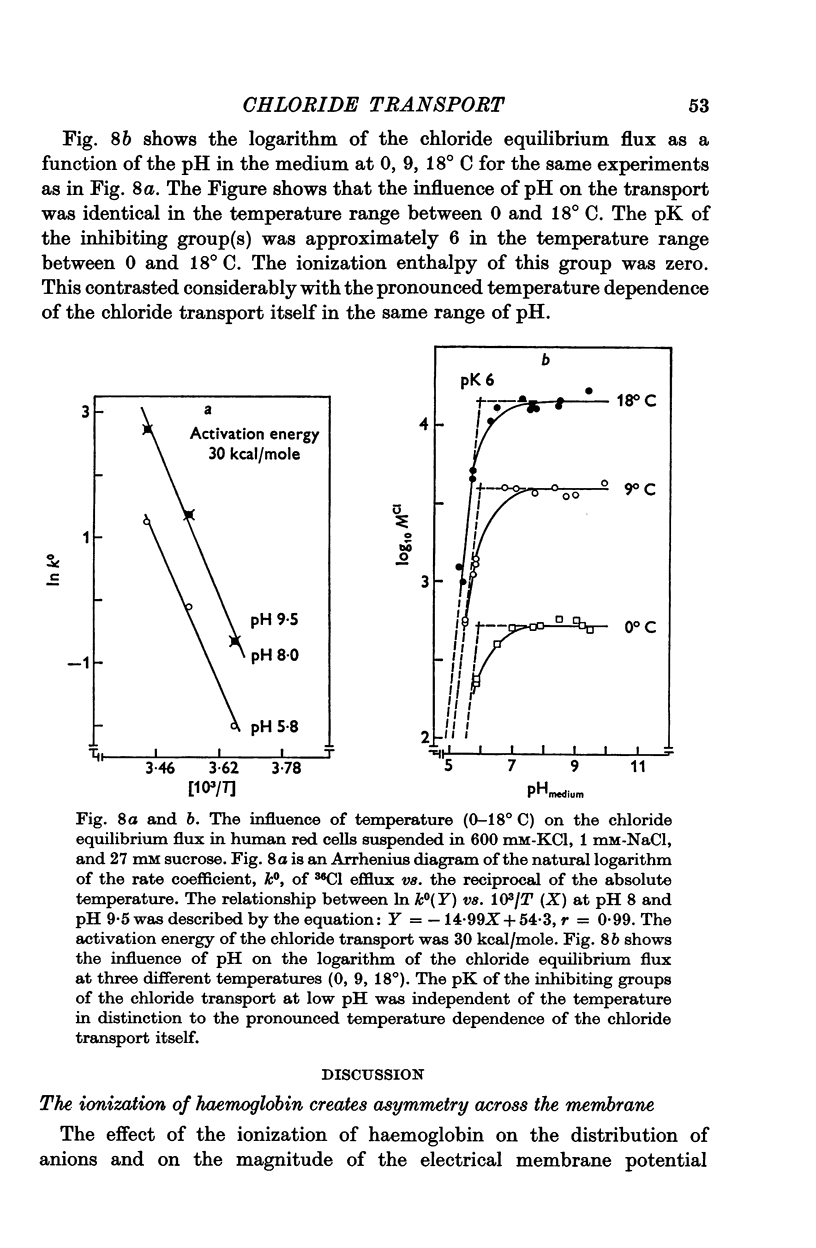







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cass A., Dalmark M. Equilibrium dialysis of ions in nystatin-treated red cells. Nat New Biol. 1973 Jul 11;244(132):47–49. doi: 10.1038/newbio244047a0. [DOI] [PubMed] [Google Scholar]
- Dalmark M. Chloride and water distribution in human red cells. J Physiol. 1975 Aug;250(1):65–84. doi: 10.1113/jphysiol.1975.sp011043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Funder J., Wieth J. O. Potassium, sodium, and water in normal human red blood cells. Scand J Clin Lab Invest. 1966;18(2):167–180. doi: 10.3109/00365516609051812. [DOI] [PubMed] [Google Scholar]
- Gunn R. B., Dalmark M., Tosteson D. C., Wieth J. O. Characteristics of chloride transport in human red blood cells. J Gen Physiol. 1973 Feb;61(2):185–206. doi: 10.1085/jgp.61.2.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnell K. F. On the mechanism of inhibition of the sulfate transfer across the human erythrocyte membrane. Biochim Biophys Acta. 1972 Sep 1;282(1):265–276. doi: 10.1016/0005-2736(72)90333-1. [DOI] [PubMed] [Google Scholar]
- Shean G. M., Sollner K. Carrier mechanisms in the movement of ions across porous and liquid ion exchanger membranes. Ann N Y Acad Sci. 1966 Jul 14;137(2):759–776. doi: 10.1111/j.1749-6632.1966.tb50198.x. [DOI] [PubMed] [Google Scholar]


