Abstract
Vancomycin-tolerant Streptococcus pneumoniae is a growing problem among drug-resistant human pathogens. Some vancomycin-tolerant pneumococci have been reported to carry mutations in loci encoding a two-component regulatory system designated VncRS or in a proximal ABC transporter, Vex. A model was advanced proposing that the tolerance phenotype resulted from the inability of a vncS mutant to respond to the Vex-transported Pep27 “death peptide” signal and dephosphorylate VncR, thereby preventing relief of repression of autolytic and other cell death functions in response to antibiotics. To explore this hypothesis, we constructed mutations in vncS, vncR, vex3, and pep27 in S. pneumoniae strain R6 and two additional genetic backgrounds. The lytic responses of the isogenic ΔvncS, Δvex3, ΔvncR, and Δpep27 mutants, but not a ΔlytA strain, to vancomycin were indistinguishable from that of the parent strain. ΔvncS strains also failed to exhibit tolerance to vancomycin at various doses in multiple media and showed wild-type sensitivity to other classes of autolysis-inducing antibiotics. In contrast, addition of subinhibitory levels of the antibiotic erythromycin led to tolerance to vancomycin during late, but not early, exponential-phase growth in a ΔvncS strain, in the parent strain R6, and in two other strains bearing erythromycin resistance markers, namely, a ΔvncR strain and an unrelated ΔcomD strain that is defective in competence-quorum sensing. Thus, this tolerance effect resulted from changes in cell growth or other erythromycin-dependent phenomena and not inactivation of vncS per se. Consistent with these results, and in contrast to a previous report, we found that a synthetic form of Pep27 did not elicit lytic or nonlytic killing of pneumococci. Finally, microarray transcriptional analysis and β-galactosidase reporter assays revealed VncS-dependent regulation of the vex123 gene cluster but did not support a role for VncRS in the regulation of autolytic or other putative cell death loci. Based on these findings, we propose that vancomycin tolerance in S. pneumoniae does not result from loss of vncS function alone.
Antimicrobial tolerance (43) is defined as the capacity of bacteria to survive, but not grow, during antibiotic therapy (22). Tolerant bacteria cease active growth and are not killed by normally lethal doses of a given antibiotic, yet the MICs for tolerant bacteria remain identical to those seen for their nontolerant counterparts. This mechanism for persistence in the presence of antibiotics is distinct from that of antimicrobial resistance, in which bacteria are insensitive to the antibiotic and continue to multiply in the presence of the inhibitor (22). Given the insidious nature of tolerant bacteria, it is thought that replication of these organisms will renew once the drug is removed. In this way, tolerance to antimicrobial agents may promote the failure of antibiotic chemotherapy and may lead to the acquisition of bona fide drug resistance (23, 29, 43).
The gram-positive diplococcus Streptococcus pneumoniae is a common cause of bacterial invasive disease and is a leading cause of morbidity and mortality, especially among the elderly and the very young (13). The incidence of antibiotic resistance in pneumococci has increased dramatically in the past decade, and multiple-antibiotic-resistant strains are now being reported worldwide (13, 42). As a result, vancomycin is now recommended for initial treatment of pneumococcal meningitis in children (33). Thus, the recent recovery of clinical isolates of S. pneumoniae exhibiting tolerance to vancomycin (1, 10, 20, 22, 44) is of utmost concern, as this may be indicative of selective pressure for the emergence of true vancomycin resistance in this devastating pathogen.
The emergence of drug tolerance in S. pneumoniae has been proposed to result from mutations in several distinct genetic loci (4, 26-29, 46), although more recent evidence has called into question the role that certain gene products (i.e., ZmpB, PsaA, and ClpC) play in tolerance to antibiotic challenge (3, 5, 7, 36). Some tolerant laboratory strains (29), as well as a handful of clinical isolates (10), have been reported to carry mutations in a single locus encoding a two-component regulatory system (TCS) designated vncRS. TCSs are employed by bacteria to sense and respond to environmental stimuli and typically consist of a membrane-bound histidine protein kinase (e.g., VncS) and a cytoplasmic effector termed a response regulator (e.g., VncR) (reviewed in reference 38). Genomic sequence analysis has revealed the presence of vncRS, 12 additional TCSs, and one orphan response regulator in multiple strains of S. pneumoniae (12, 41). The functions of most of these TCSs in pneumococci are largely unknown. It was recently reported that SPSJ01, an R6 pneumococcal strain with an insertion of the vector pJDC9 into vncS, is tolerant to vancomycin as well as to beta-lactams, cephalosporins, aminoglycosides, and quinolones (29). A later study suggested that a secreted peptide, Pep27, encoded upstream of the vncRS locus, was the effector molecule sensed by VncS to trigger the cell death pathway (28). Together, these studies led to a model for Pep27-VncRS signal transduction (28). According to the model described previously (28), phosphorylated VncR functions as a repressor of autolytic and other cell death functions under normal growth conditions. As the cells enter stationary phase or following drug treatment, Pep27 is secreted through the Vex ABC transporter and accumulates in the medium. Accumulated Pep27 is then sensed by VncS, converting this histidine protein kinase into a phosphatase that dephosphorylates phosphorylated VncR, allowing derepression of autolytic pathways and resulting in the loss of viability and lysis of pneumococcal cells.
Because TCSs are thought to represent potential targets for broad-spectrum antibiotics (19), we further evaluated the role of vncS in antibiotic tolerance in S. pneumoniae and tested the proposed Pep27-VncRS signal transduction model. We constructed several defined vncS mutations in multiple genetic backgrounds and evaluated antibiotic tolerance in the presence of multiple classes of antibiotics and under different culture conditions. In addition, we constructed defined mutations in genes encoding other reported components of this signal transduction pathway, including the ABC transporter vex3, the response regulator vncR, the major autolysin lytA, and the peptide effector pep27. Herein, we report the effects of these mutations on the drug tolerance of S. pneumoniae and describe microarray transcriptional profiling and β-galactosidase reporter assays of some of these strains as a first step in understanding the genetic pathways regulated by the VncRS-Pep27 signal transduction system. Our results show that tolerance to vancomycin is induced following treatment of pneumococci with erythromycin but that inactivation of VncRS function is not required for this effect.
MATERIALS AND METHODS
Bacterial strains, growth, and transformation conditions.
Bacterial strains employed in this study were derived from S. pneumoniae CP1250 (17), D39, and the unencapsulated D39 derivative R6 (12, 37) (Table 1). R6 and D39 were assigned unique strain designations to track isogenic derivatives. Bacteria were cultivated statically in either brain heart infusion broth (BHI; Becton Dickinson BBL), a semisynthetic casein acid hydrolysate medium supplemented with 0.5% (wt/vol) yeast extract (C+Y broth) (14), Todd-Hewitt broth (Difco) supplemented with 0.5% (wt/vol) yeast extract (THB-Y), or chemically defined medium (CDM) (36, 40), as indicated. For enumeration of bacteria and for transformation recovery, bacteria were plated on Trypticase soy agar II blood agar plates (TSAII BA; Becton Dickinson BBL) or on CAT agar (24) at 37°C in an atmosphere of 5% CO2.
TABLE 1.
Bacterial strains, DNA amplicons, and plasmids used in strain construction
| Bacterial strain, amplicon, or plasmid | Relevant characteristicsa | Genotype and/or phenotypeb | Source or reference |
|---|---|---|---|
| Bacterial strains | |||
| CP1250 | hex malM511 str-1 bgl-1 | Derivative of strain Rx | 17 |
| CP1257 | CP1250 ΔvncS41::Pc::ermAM Err | CP1250 transformed with linear vncS(Δ41-729)::Pc::ermAM amplicon | This study |
| CP1260 | Avirulent, unencapsulated | R6 parentc, derivative of D39 | J.-P. Claverys |
| CP1261 | CP1260 ΔvncS41::Pc::ermAM Err | CP1260 transformed with linear vncS (Δ41-729)::Pc::ermAM amplicon | This study |
| CP1292 | CP1250 ΔvncS78::Pc::aphIII Kmr | CP1250 transformed with linear vncS (Δ78-1224)::Pc::aphIII amplicon | This study |
| EL23 | EL59 vncS::(pCZA342)::vncS Err | EL59 transformed with vncS::(pCZA342)::vncS | This study |
| EL59 | Avirulent, unencapsulated | R6 parent,d derivative of D39 | A. Tomasz; 12 |
| EL117 | EL59 ΔcomD::ermAM Err | EL59 transformed with linear comD(Δ325-1014)::ermAM amplicon | This study |
| EL121 | EL59 ΔvncS273::ermAM Err | EL59 transformed with linear vncS (Δ273-1021)::ermAM amplicon | This study |
| EL161 | Virulent, encapsulated type 2 | D39 parent, subclone of clinical isolate | J. Yother; 37 |
| EL381 | EL161 ΔvncS273::aad9 Spr | EL161 transformed with linear vncS (Δ273-1021)::aad9 amplicon | This study |
| EL395 | EL161 ΔvncR::aad9 Spr | EL161 transformed with linear vncR(Δ147-511)::aad9 amplicon | This study |
| EL555 | EL59 ΔbgaA::ermAM Err | EL59 transformed with linear ΔbgaA::ermAM | 36 |
| EL556 | EL59 ΔlytA::aad9 Spr | EL59 transformed with linear ΔlytA::aad9 | 36 |
| EL562 | CP1250 ΔvncS273::ermAM Err | CP1250 transformed with linear vncS (Δ273-1021)::ermAM amplicon | This study |
| EL822 | EL59 Δvex3::ermAM Err | EL59 transformed with linear vex3(Δ1-1377)::ermAM amplicon | This study |
| EL842 | EL59 Δpep27::ermAM Err | EL59 transformed with linear pep27 (Δ1-81)::ermAM amplicon | This study |
| EL898 | EL59 ΔvncR::ermAM Err | EL161 transformed with linear vncR(Δ147-511)::ermAM amplicon | This study |
| EL1066 | EL555 vex2::(pEVP3)::vex2 Err Cmr | EL555 transformed with pEL48 | |
| EL1069 | EL1066 ΔvncS273::aad9 Err Cmr Spr | EL1066 transformed with linear vncS(Δ273-1021)::aad9 amplicon | This study |
| Amplicons | |||
| vncS(131-626) | EL59 genomic DNA, (PS198, PS199), partial construct internal bp 131 to 626 of 1,329-bp vncS, for insertion into the BamHI site of pCZA342 | This study | |
| vncS273e-a | EL59 genomic DNA, (PS481-a1, PS483-a2E), partial construct | This study | |
| S-ermAM-b | pVA838, (PS485-b1E, PS487-b2E), partial construct | This study | |
| VncS273e-c | EL59 genomic DNA, (PS489-c1E, PS490-c2), partial construct | This study | |
| vncS(Δ273-1021) ::ermAM | vncS273e-a, S-ermAM-b, vncS273e-c, (PS481-a1, PS490-c2), completed construct, ermAM replacing bp 273 to 1021 | This study | |
| of 1,329-bp vncS | |||
| vncS273s-a | EL59 genomic DNA, (PS481-a1, PS482-a2SP), partial construct | This study | |
| S-aad9-b | pDL278, (PS484-b1SP, PS486-b2SP), partial construct | This study | |
| vncS273s-c | EL59 genomic DNA, (PS488-c1SP, vncSc2), partial construct | This study | |
| vncS(Δ273-1021) ::aad9 | vncS273s-a, S-aad9-b, vncS273s-c, (PS481-a1, PS490-c2), completed construct, aad9 replacing bp 273 to 1021 of 1,329-bp vncS | This study | |
| vncS41-a | CP1250 genomic DNA, (UpL41, UpR41), partial construct | This study | |
| 41-Pc::ermAM-b | M13EryAD, (DAM212, DAM213), partial construct | This study | |
| vncS41-c | CP1250 genomic DNA, (DnL41, DnR41), partial construct | This study | |
| vncS(Δ41-729) ::Pc::ermAM | vncS41-a, 41-Pc::ermAM-b, vncS41-c, (UpL41, DnR41) completed construct, Pc::ermAM replacing bp 41 to 729 of the 1,329-bp vncS | This study | |
| vncS78-a | CP1250 genomic DNA, (UpL78, UpR78), partial construct | This study | |
| 78-Pc::aphIII-b | pR410, (DAM301, DAM302), partial construct | This study | |
| vncS78-c | CP1250 genomic DNA, (DnL78, DnR78), partial construct | This study | |
| vncS(Δ78-1224) ::Pc ::aphIII | Ligation of vncS78-a (BamHI), 78-Pc::aphIII-b(BamHI, Apal) | This study | |
| vncS78-c (ApaI), completed construct, Pc::aphIII replacing bp 78 to 1224 of the 1,329-bp vncS by ligation of restricted DNA fragments | |||
| vncRs-a | EL59 genomic DNA, (PS291-a1, PS292a-a2SP), partial construct | This study | |
| R-aad9-b | pDL278, (PS293a-b1SP, PS294a-b2SP), partial construct | This study | |
| vncRs-c | EL59 genomic DNA, (PS295a-c1SP, PS296-c2), partial construct | This study | |
| vncR(Δ147-511):: aad9 | vncRs-a, R-aad9-b, vncRs-c, (PS291-a1, PS296-c2), completed construct, aad9 replacing bp 147 to 511 of the 657-bp vncR | This study | |
| vncRe-a | EL59 genomic DNA, (PS291-a1, PS292b-a2E), partial construct | This study | |
| R-ermAM-b | pVA838, (PS293b-b1E, PS294b-b2E), partial construct | This study/PICK> | |
| vncRe-c | EL59 genomic DNA, (PS295b-c1E, PS296-c2), partial construct | This study | |
| vncR(Δ147-511)::ermAM | vncRc-a, R-ermAM-b, vncRe-c, (PS291-a1, PS296-c2), completed construct, ermAM replacing bp 147 to 511 of the 657-bp vncR | This study | |
| vex3-a | EL59 genomic DNA, (JZ109-a1, JZ 110-a2), partial construct | This study | |
| X-ermAM-b | pVA838, (JZ111-b1er, JZ112-b2er), partial construct | This study | |
| vex3-c | EL59 genomic DNA, (JZ113-c1, JZ114-c2), partial construct | This study | |
| vex3(Δ1-1377)::ermAM | vex3-a, X-ermAM-b, vex3-c, (JZ109-a1, JZ114-c2), completed construct, ermAM replacing bp 1 to 1377 of the 1,377-bp vex3 | This study | |
| pep27-a | EL59 genomic DNA, (JZ115-a1, JZ116-a2), partial construct | This study | |
| 27-ermAM-b | pVA838, (JZ117-b1er, JZ118-b2er), partial construct | This study | |
| pep27-c | EL59 genomic DNA, (JZ119-c1, JZ120-c2), partial construct | This study | |
| pep27(Δ1-81)::ermAM | pep27-a, 27-ermAM-b, pep27-c, (JZ115-a1, JZ120-c2), completed construct, ermAM transcribed in the opposite orientation and replacing bp 1 to 81 of the 81-bp pep27 | This study | |
| comD-a | EL59 genomic DNA, (PS431-a1, PS433-a2E), partial construct | This study | |
| D-ermAM-b | pVA838, (PS435-b1eE, PS437-b2E), partial construct | This study | |
| comD-c | EL59 genomic DNA, (PS439-c1E, PS440-c2), partial construct | This study | |
| comD(Δ325-1014)::ermAM | comD-a, D-ermAM-b, comD-c, (PS431-a1, PS440-c2), completed construct, ermAM replacing bp 325 to 1014 of the 1,323-bp comD | This study | |
| Plasmids | |||
| pEVP3 | Insertion duplication lacZ reporter plasmid, Cmr | 6 | |
| pCZA342 | Cloning vector for insertion duplication, Emr Amr | 2 | |
| pGD024 | BamHI restricted vncS(131-626) (above), ligated to BamHI | This study | |
| pCZA342, Err | |||
| pEL48 | PCR blunt, BamHI restricted vex3-a (above), ligated to SmaI, BamHI pEVP3, Cmr | This study |
Err, resistant to erythromycin; Kmr, resistant to kanamycin; Spr, resistant to spectinomycin; Cmr, resistant to chloramphenicol; and Amr, resistant to apramycin. Strain construction was carried out by transformation of the indicated recipient with a linear double-stranded synthetic PCR amplicon or circular plasmid DNA, as indicated.
For amplicons, the source of template DNA is indicated followed in parentheses by the primer pair sets used to generate the given amplicon.
R6 isolate R800 from J. Claverys.
R6 isolate ATCC BAA 255, laboratory stock.
For transformation of S. pneumoniae, cells were grown to an optical density at 620 nm (OD620) of ≤0.1, diluted 1:10 in BHI containing 10% heat-inactivated horse serum (Sigma), 10 mM glucose, and 100 ng of synthetic competence stimulatory peptide 1 per ml, and incubated for 15 min at 37°C in an atmosphere of 5% CO2 (17). Following the addition of 0.2 to 1.0 μg of DNA per ml, the cells were allowed to incubate for an additional 1.5 h to allow phenotypic expression of antibiotic resistance markers. Transformants were recovered on TSAII BA containing appropriate antibiotics in nutrient broth soft agar overlays (0.8% [wt/vol] Bacto Nutrient Broth, 0.4% [wt/vol] Bacto Agar) or by using the double-overlay method (30) at 37°C in an atmosphere of 5% CO2. For antibiotic selection, media were supplemented with 200 μg of kanamycin per ml, 100 μg of spectinomycin per ml, 2.5 μg of chloramphenicol per ml, or 0.3 or 1.0 μg of erythromycin per ml, as indicated. To test the effect of erythromycin on vancomycin tolerance, media were supplemented with 1.0 μg of erythromycin per ml for strains carrying an ermAM (erythromycin resistance) marker and 0.002 and 0.004 μg of erythromycin per ml for R6. In other instances, media were not routinely supplemented with antibiotics.
Construction of insertion-duplication and deletion-replacement mutants of S. pneumoniae.
Insertion-duplication mutants were constructed by using suicide vector pCZA342 (2), and deletion-replacement mutants were constructed by using synthetic amplicons generated by overlap extension PCR (15) or ligation of BamHI- and ApaI-restricted PCR fragments (see below and Table 1). For insertion-duplication mutagenesis, a 495-bp 5′ internal fragment of vncS, spanning nucleotides 131 to 626 of the 1,329-bp vncS open reading frame (ORF), was inserted into the BamHI site of vector pCZA342 (Fig. 1 and Table 1). For deletion-replacement mutagenesis, each amplicon was designed such that all or most of the target gene was deleted and replaced with the spectinomycin resistance marker aad9 (16), the erythromycin resistance marker ermAM (18), or derivatives of the erythromycin and kanamycin resistance genes under control of a robust synthetic Pc promoter to give Pc::ermAM (6) and Pc::aphIII (39), respectively (Fig. 1 and Table 1). Methods for amplicon generation by overlap extension PCR (15) were as described previously (36). The vncS(Δ78-1224)::Pc::aphIII linear-deletion amplicon was generated by incorporating unique heterologous BamHI or ApaI restriction sites into the oligonucleotide primers used to generate the flanking DNA fragments (Table 2). Following restriction with BamHI and ApaI, these fragments were ligated to a similarly digested PCR-amplified kanamycin resistance cassette (39). The resultant linear-deletion amplicons or the insertion-duplication vector was used to transform S. pneumoniae as described above. All mutations were confirmed by PCR analysis of genomic DNA based on diagnostic changes in DNA fragment size following recombination of plasmids or amplicons or by the use of primer sets specific for the mutagenesis cassette and flanking DNA sequences.
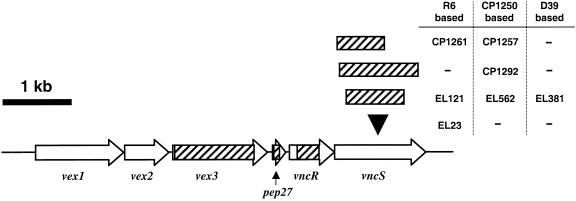
FIG. 1.
Organization of the vncRS gene cluster in S. pneumoniae R6. The genes encoding the Vex1, Vex2, and Vex3 ABC transporter, the putative peptide Pep27, the VncR response regulator, and the VncS histidine protein kinase are indicated with arrows (drawn to scale). The black arrowhead indicates the site of pCZA342 insertion in EL23; the hatched areas indicate the regions of DNA that were replaced in individual deletion-replacement mutants. For vncS strains, the name of the corresponding mutant is adjacent to the region deleted.
TABLE 2.
Synthetic oligonucleotides used in these studies
| Name and descriptiona | Sequence (5′→3′)b |
|---|---|
| Primer set used for vncS insertion-duplication cassette construction | |
| vncS::(pCZA342)::vncS (Emr) | |
| PS198 | GTCATCGTCAggatccCATTGGTCAAAAGGC |
| PS199 | AACAGTCAAGggatccTGGTAGAGGCTATTG |
| Primer sets used for deletion replacement cassette construction | |
| vncS(Δ273-1021)::ermAM (Err) and vncS (Δ273-1021)::aad9 (Spr) | |
| PS481-a1 | GGCTCAAGCCTCTAACCTTGGTGGT |
| PS482-a2SP | CGAACGAAAATCGATGAGGAAGACTGTCCTTGAC |
| PS483-a2E | TTTCCATAACTTCTTGAGGAAGACTGTCCTTGAC |
| PS484-b1SP | GTCAAGGACAGTCTTCCTCATCGATTTTCGTTCGTGAAT |
| PS485-b1E | GTCAAGGACAGTCTTCCTCAAGAAGTTATGGAAA |
| PS486-b2SP | GGTTTAGATAAGCCTGCTGATTTAGAATGAATATTTCC |
| PS487-b2E | GGTTTAGATAAGCCTGCTGATTTAGCTCCTTGGAAGC |
| PS488-c1SP | GGAAATATTCATTCTAAATCAGCAGGCTTATCTAAACC |
| PS489-c1E | GCTTCCAAGGAGCTAAATCAGCAGGCTTATCTAAACC |
| PS490-c2 | GCAGTTATGTTGTCCTGTTC |
| vncS(Δ41-729)::Pc::ermAM (Err) (CP1261) | |
| UpL41 | AGATGAGTGCCTTTGCCTCTTTGG |
| UpR41 | ATCAAACAAATTTTGGGCCCGGTAAACCTGTTCGTTTCATTTTCGC |
| DnL41 | AATTCTATGAGTCGCTGCCGACTGAAAACACCGCTGGCTAGTTTGA |
| DnR41 | TTCTCCTTCTCTTTCTCCAATTCG |
| DAM212 | CCGGGCCCAAAATTTGTTTGAT |
| DAM213 | AGTCGGCAGCGACTCATAGAAT |
| vncS(Δ78-1224)::Pc::aphIII (Kmr) (CP1292) | |
| UpL78 | CTCTAACCTTGGTGGTGGTGCAGAAGTA |
| UpR78 | ATggatccTGAAGGCAGATAACCAGAACACTAA |
| DnL78 | ATgggcccTTTGTGGTTAAGAGTCTATTAGAA |
| DnR78 | ATGTTACCGATACCAGCTGCCAAGAAGT |
| DAM301 | CGCGCAAGCTGGggatccG |
| DAM302 | ACGTgggcccTAGGTACTAAAACAATTCATCCAGTAA |
| vncR(Δ147-511)::aad9 (Spr) and vncR (Δ147-511)::ermAM (Err) | |
| PS291-a1 | GAAACGCCAGAAACCAAGAAGAATC |
| PS292a-a2SP | CGAACGAAAATCGATGCATCTGGATATCCAG |
| PS292b-a2E | TTTCCATAACTTCTTGCATCTGGATATCCAG |
| PS293a-b1SP | CTGGATATCCAGATGCATCGATTTTCGTTCGTGAAT |
| PS293b-b1E | CTGGATATCCAGATGCAAGAAGTTATGGAAA |
| PS294a-b2SP | GAGACCGAGTCAAGGCCTTAGAATGAATATTTCC |
| PS294b-b2E | GAGACCGAGTCAAGGCCTTAGCTCCTTGGAAGC |
| PS295a-c1SP | GGAAATATTCATTCTAAGGCCTTGACTCGGTCTC |
| PS295b-c1E | GCTTCCAAGGAGCTAAGGCCTTGACTCGGTCTC |
| PS296-c2 | TCCCATTTACATTGTTCATTTTATCA |
| vex3(Δ1-1377)::ermAM (Err) | |
| JZ109-a1 | AATCCTTATCCACGAAGAATTGGCTA |
| JZ110-a2 | TTTCCATAACTTCTTAGATTCCCTTTCTAGATTTTGTTTTAATC |
| JZ111-b1E | CTAGAAAGGGAATCTAAGAAGTTATGGAAATAAGACTTAGAAGCAAAC |
| JZ112-b2E | CATTTTTTCAAATTTATTAGCTCCTTGGAAGCTGTCAGTAGTATACCT |
| JZ113-c1 | GCTTCCAAGGAGCTAATAAATTTGAAAAAATGAGTCTGGAATAAAG |
| JZ114-c2 | TGGTTTTACGAATCTCAGCTAGAACTTC |
| pep27(Δ1-81)::ermAM (Err) | |
| JZ115-a1 | ACGATGCAGATAATGAAAAAGGAGCC |
| JZ116-a2 | CTTCCAAGGAGCTAAAAATTCCATAGCTTTAGTATATTATATTTA |
| JZ117-b1er | TAAAGCTATGGAATTTTTAGCTCCTTGGAAGCTGTCAGTAGTATAC |
| JZ118-b2er | TAAATACCTACTCTAAAGAAGTTATGGAAATAAGACTTAGAAGCA |
| JZ119-c1 | TTTCCATAACTTCTTTAGAGTAGGTATTTATTCTAAGAAAAAT |
| JZ120-c2 | GGCGGTCTGTGTCCAGAGGAAGACTG |
| comD(Δ325-1014)::ermAM (Err) | |
| PS431-a1 | GTTGTAACAGTTGGGAAACTG |
| PS433-a2E | TTTCCATAACTTCTTGAGGAGAAAGTTACGG |
| PS435-b1eE | CCGTAACTTTCTCCTCAAGAAGTTATGGAAA |
| PS437-b2E | CGCTCATGATACGAACCTTAGCTCCTTGGAAGC |
| PS439-c1E | GCTTCCAAGGAGCTAAGGTTCGTATCATGAGCG |
| PS440-c2 | GCAACTGAATCTAAATACCACCG |
Err, resistant to erythromycin; Spr, resistant to spectinomycin; and Kmr, resistant to kanamycin.
Lowercase letters indicate heterologous restriction sites engineered into the oligonucleotide to facilitate ligation. Underlined portions of oligonucleotides represent short heterologous extensions that are complementary to the fragments of DNA to be linked by overlap extension PCR.
Vancomycin-induced autolysis.
The methods and materials for measuring the sensitivity of S. pneumoniae to the cell wall synthesis inhibitor vancomycin were those described previously (29), unless otherwise stated in the text. Briefly, cells were inoculated directly from frozen glycerol stocks into 5 ml of fresh C+Y broth and serially diluted in C+Y broth for static overnight growth at 37°C in an atmosphere of 5% CO2. Cultures with equivalent OD620s from the dilution series were then diluted in fresh C+Y broth to an OD620 of 0.02 (∼5 × 106 CFU per ml) and incubated at 37°C in an atmosphere of 5% CO2 until the OD620 reached 0.15 to 0.20 by direct measurement in 1.4-cm-path-length tubes. This absorbance corresponds to 0.25 to 0.30 when converted to the standard 1-cm path length, as was reported previously (29). Vancomycin was added at 5 μg per ml (10 times the MIC), and incubation of cultures was continued. At various times after the addition of vancomycin, cell death and autolysis were measured by viable cell counts and direct measurement of the change in the OD620, respectively. For viable cell counts, samples of cultures were aseptically removed and serially diluted in C+Y broth. Portions of the diluted cultures were immediately spread onto TSAII BA, which were incubated overnight at 37°C in an atmosphere of 5% CO2. Bacterial colonies were enumerated the following day. The OD620 was determined with a Spectronic 20 Genesys spectrometer (Spectronic Instruments, Rochester, N.Y.) fitted for direct measurement of 1.4-cm capped tubes.
Nonionic detergent-induced autolysis.
Cells grown in BHI as described above were diluted in 8 ml of fresh BHI to an OD620 of ∼0.02 (∼4 × 106 CFU per ml). These cells were incubated at 37°C in an atmosphere of 5% CO2 until the OD620 reached ∼0.13 (∼2 × 107 CFU per ml). Each culture was divided into prewarmed tubes and mixed with 0.4% (wt/vol) deoxycholate to a final concentration of 0.05% or with an equal volume of water. Cultures were returned to 37°C, and growth or autolysis was measured as changes in OD620 over time. An R6 lytA deletion mutant (EL556) (Table 1), which lacks the major autolysin of S. pneumoniae, was employed as a control for deoxycholate-induced autolysis.
In vivo assay for vancomycin tolerance.
Experiments with animals were conducted strictly according to the Guide for the Care and Use of Laboratory Animals prepared by the Committee on Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, National Research Council (25). Vancomycin tolerance was monitored in immunosuppressed outbred ICR mice in a thigh model of infection (45). Briefly, neutropenia was induced in female ICR mice (Harlan Sprague Dawley), each weighing ∼20 g, by intraperitoneal injection with 150 and 100 mg cyclophosphamide per kg of body weight at 4 days and 1 day prior to infection, respectively. S. pneumoniae D39 and derivatives were harvested from TSAII BA after overnight incubation, diluted in sterile 0.9% (wt/vol) NaCl (saline) to yield an OD580 of ∼0.3, and then further diluted 1:100 in BHI to yield ∼106 CFU per ml. Following brief isoflurane anesthetization, mice were infected by intramuscular inoculation of 0.1 ml of bacterial suspension into each thigh. One mouse from each group was immediately sacrificed by CO2 asphyxiation, and the mean infecting dose per thigh was determined (see below). Of the remaining animals, half were treated by subcutaneous injection with 10 mg of vancomycin per kg of body weight at 1 and 5 h postinfection. At 8 and 24 h postinfection, animals were sacrificed by CO2 asphyxiation and the thighs were removed and homogenized in 9 ml of saline. Homogenized tissues were serially diluted in saline and immediately plated onto TSAII BA. Bacterial colonies were enumerated following overnight incubation at 37°C in an atmosphere of 5% CO2.
Synthesis of Pep27.
Synthetic peptides corresponding to either the predicted (MRKEFHNVLSSDQLLTDKRPARDYNRK; Lilly-Pep27) (12) or published (MRKEFHNVLSSGQLLADKRPARDYNRK; Pub-Pep27) (28) sequences of Pep27 were prepared on an Applied Biosystems ABI433A peptide synthesizer with 9-fluorenylmethoxy-carbonyl amino acids. Each synthesis was started from 0.1 mmol of 9-fluorenylmethoxy-carbonyl Lys(Boc) WANG resin (Midwest Biotech) by using dicyclohexyl carbodiimide/hydroxybenzotriazole-activated single couplings (10-fold excess). The completed peptidyl resins were cleaved with 5 ml of reagent K (0.75 g of phenol, 0.5 ml of thioanisole, 0.25 ml of ethanedithiol, and 0.5 ml of water, each per 10 ml of trifluoroacetic acid [TFA]). The TFA filtrates were added to 40 ml of ethyl ether, and the precipitates were centrifuged. The pellets were washed with ether and dried in vacuo. Each crude product was dissolved in 10 ml of aqueous acetic acid and loaded onto a 2.2- by 25-cm Vydac C18 column. Peptide was eluted with a gradient of 15 to 35% (vol/vol) acetonitrile containing 0.1% (vol/vol) TFA on a fast protein liquid chromatograph (Pharmacia) at 4 ml per min while the UV absorption at 214 nm was monitored and 5-min fractions were collected. The appropriate fractions were combined, frozen, and lyophilized to give 38 mg of Lilly-Pep27 and 28 mg of Pub-Pep27. The peptide purities were ∼90% according to high-performance liquid chromatography and were verified by mass spectrometry by using the electrospray source of a PESciex API III mass spectrometer in the positive-ion-detection mode.
Pep27 challenge.
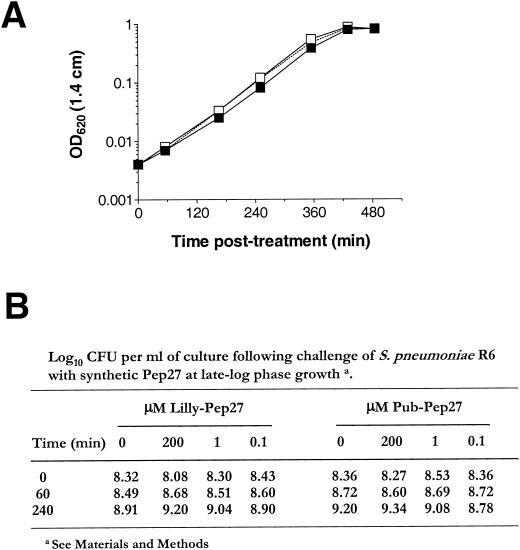
Cells grown overnight in CDM were diluted in fresh CDM containing either 50 μM Lilly-Pep27 or 50 μM Pub-Pep27 or a volume of sterile water equivalent to that added with each peptide to yield an OD620 of ∼0.004 (∼2 × 105 CFU per ml) in a 1.4-cm path length. Cultures were placed at 37°C in an atmosphere of 5% CO2 and monitored for changes in OD620 over time. To exclude the possibility that Pep27 was active only at a high cell density, a late-exponential-phase CDM culture (OD620, ∼0.45 in a 1.4-cm path length; ∼2 × 108 CFU per ml) was split into 12 equal 0.5-ml aliquots and challenged with a 0, 0.1, 1.0, or 200 μM concentration of either Lilly-Pep27 or Pub-Pep27. Cultures were placed at 37°C in 5% CO2, and viability was determined at 0, 60, and 240 min after peptide addition by serial dilution in saline and plating on TSAII BA.
RNA isolation and microarray analysis.
Microarray transcription profiles were determined for S. pneumoniae EL59 (R6 parent), EL121 (R6 ΔvncS273::ermAM), EL822 (Δvex3::ermAM), and EL898 (ΔvncR::ermAM) grown in CDM to an OD620 of 0.3 (mid-exponential phase; ∼1 × 108 CFU per ml) and 0.7 (late-exponential phase; ∼3 × 108 CFU per ml). Total RNA was extracted, purified, and labeled as described previously (36). Relative transcript amounts were estimated by hybridizing labeled RNA to low-density custom Affymetrix S. pneumoniae R6 microarrays covering >95% of predicted S. pneumoniae ORFs (36). Data were analyzed by using Affymetrix Microarray Suite version 5.0 and Data Mining Tool version 3.0. The signal log ratio algorithm (details available at the Affymetrix website) was used to estimate the total number of transcriptional changes observed between a control and an experimental microarray. For these studies, changes in the expression of an experimental array probe relative to the baseline that were greater or equal to a signal log ratio of 1 (log2 of 1 equals an ∼2-fold change) and that were observed in two or more independent experiments were considered significant. For the select gene changes shown herein (i.e., vex123, vncRS, lytA, lytB, lytC, and murMN), a ratio of the average difference call, which represents the relative hybridization signal above that of the mismatch controls, was used to estimate the change in target transcript amounts between RNA from the experimental strains (i.e., vncR, vncS, or vex3 strains) and the control strains (R6 parent), where a ratio of 1 indicated no change.
β-Galactosidase reporter assays.
A transcriptional fusion between vex2 and the Escherichia coli lacZ gene was generated by ligation of PCR-amplified vex3-a (Table 1) following digestion with BamHI to create a 1.3-kb fragment bearing a 3′ overhang for directional cloning into the SmaI and BamHI sites preceding lacZ in the pneumococcal reporter vector pEVP3 (6). The resultant pneumococcal insertion-duplication plasmid pEL48 (Table 1) was propagated in E. coli HB101 and introduced by transformation into S. pneumoniae EL555 (R6 ΔbgaA::ermAM) to yield strain EL1066 (Table 1). The ΔvncS273::aad9 mutation was introduced into this background to yield an isogenic reporter for vex gene expression in the absence of vncS (EL1069). For assay of vex2::lacZ reporter activity, bacterial cultures with an OD620 of ∼0.6 (∼5 × 108 CFU per ml) after overnight incubation in BHI at 37°C were diluted in 11 ml of BHI to yield an OD620 ∼0.03 (∼5 × 106 CFU per ml). In all cases, media were supplemented with 2.5 μg of chloramphenicol per ml to ensure maintenance of the vex2::(pEVP3)::vex2 insertion-duplication. Cultures were placed at 37°C in an atmosphere of 5% CO2, and growth was monitored by direct measurement of changes in OD620 in a 1.5-cm path length. At 60, 180, 270, and 400 min postinoculation, 1 ml of cell suspension was removed, lysed with 0.1% (vol/vol) Triton X-100 at 37°C for 2 min, and assayed for β-galactosidase activity with o-nitrophenyl-β-d-galactopyranoside as the substrate. Results of β-galactosidase assays are expressed in Miller units normalized to the OD620 of the culture at each time point (21).
RESULTS
Addition of erythromycin, but not inactivation of vncS, promotes tolerance to vancomycin in S. pneumoniae R6.
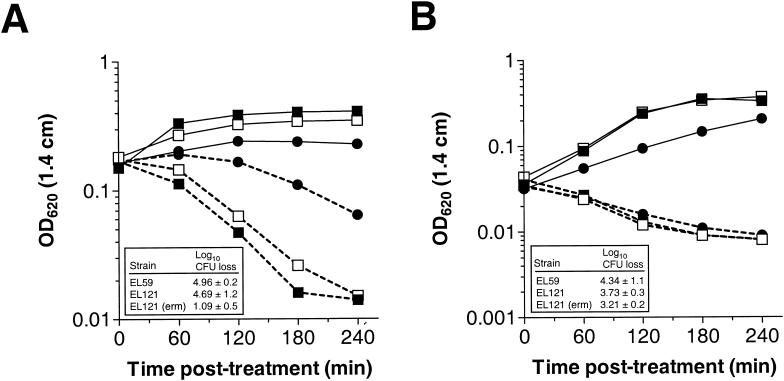
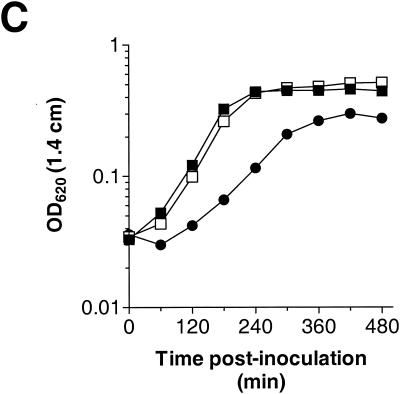
To address whether the VncS histidine protein kinase is required for tolerance to vancomycin, we constructed a defined deletion-replacement mutation in vncS (EL121) of S. pneumoniae R6 (Table 1). The vncRS locus from S. pneumoniae R6 and the site of DNA insertion or replacement in the mutants constructed in this study are schematically represented in Fig. 1. The ΔvncS strain (EL121) and its isogenic vncS+ R6 parent (EL59) were tested for tolerance to 10 times the MIC of vancomycin (5 μg of vancomycin per ml) at an OD620 equivalent to that reported previously (29). Based on numerous repetitions of the tolerance assay, we failed to demonstrate reproducible tolerance to vancomycin for our vncS deletion strain (EL121) either by measuring the change in absorbance at 620 nm (Fig. 2A) or by direct measurement of cellular viability following 4 h of treatment with this cell wall biosynthesis inhibitor (Fig. 2A, inset). We also failed to demonstrate tolerance when cells were challenged with vancomycin at a significantly lower cell density (OD620, 0.05 to 0.06) to reflect the actual number of viable CFU per ml (∼0.5 × 107 to 1.0 × 107 CFU per ml) reported previously (29) (Fig. 2B; also data not shown). Similar results were observed for a vncS insertion-duplication mutant (EL23) of S. pneumoniae R6 (Table 1 and Fig. 1) challenged with various concentrations of vancomycin in BHI medium (data not shown). This indicates that the loss of vncS function alone does not confer a vancomycin tolerance phenotype to S. pneumoniae. In contrast, reproducible tolerance to vancomycin was observed for the ΔvncS strain (EL121) when erythromycin was added to the C+Y medium at ∼3.3 times our normal selection dose for the erythromycin marker derived from pVA838 (1.0 versus 0.3 μg of erythromycin per ml [18]) to reflect the methods reported previously (29) (Fig. 2A). This was not due to selection for ΔvncS isolates that have retained their ermAM marker, as serial dilution and duplicate plating revealed equivalent numbers of bacteria on both selective and nonselective media and PCR analysis revealed no loss of the ermAM antibiotic resistance marker in EL121 (data not shown). The ability of erythromycin to promote tolerance of the ΔvncS strain (EL121) to vancomycin was growth stage dependent, as challenge of cells during early exponential growth (OD620, 0.05 to 0.06) failed to produce a similar result (Fig. 2B). This concentration of erythromycin did, however, reduce the overall yield of the ΔvncS strain (EL121) and increased the doubling time by 78% (∼46 min versus ∼82 min) in C+Y medium, extending the time required to reach an equivalent OD620 by as much as 2 h (Fig. 2C).
FIG. 2.
(A and B) Lack of vancomycin tolerance in a defined deletion-replacement mutant of vncS during mid-exponential to late exponential (OD620, ∼0.16 to 0.2) (A) or early exponential (OD620, ∼0.05 to 0.06) (B) growth. Duplicate samples grown in C+Y medium without supplementation or C+Y medium supplemented with 1 μg of erythromycin per ml were left untreated (growth control) or were treated with 5 μg of vancomycin per ml (10 times the MIC). Growth or autolysis was measured as a change in OD620 in a 1.4-cm-path-length tube. Data are representative of at least two independent experiments. Solid lines, untreated growth control; dashed lines, treated with 5 μg of vancomycin per ml. Insets show log10 CFU loss (mean ± standard deviation of results from two independent experiments) following 4-h challenge with vancomycin as determined by serial dilution in C+Y medium and plating on TSAII BA. (C) Growth kinetics of the S. pneumoniae vncS strain altered in the presence of high levels of erythromycin. Cells were inoculated into C+Y medium with no supplementation or C+Y medium supplemented with 1 μg of erythromycin per ml, and growth was measured as changes in the OD620 over time. Symbols: closed squares, R6 parent strain (EL59); open squares, R6 ΔvncS273::ermAM (EL121); closed circles, R6 ΔvncS273::ermAM (EL121) supplemented with 1 μg of erythromycin per ml.
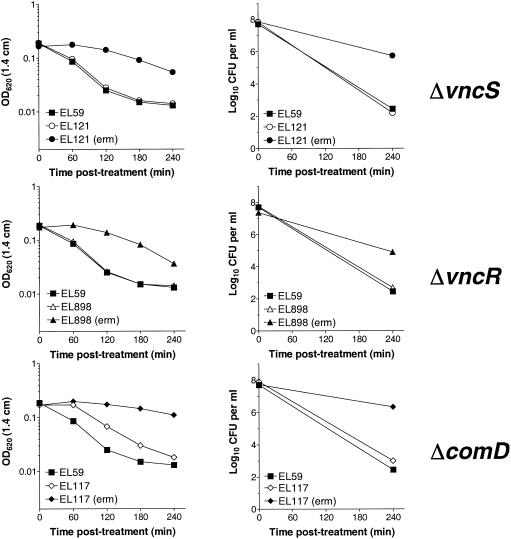
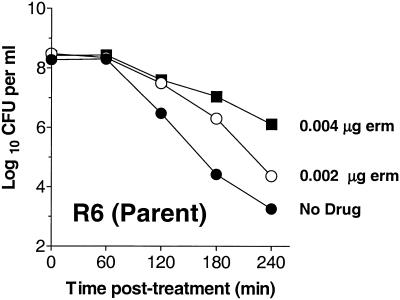
To test whether erythromycin alone at the cell density reported previously (29) can promote a drug tolerance phenotype, the effect of high levels of erythromycin on growth and tolerance of the ΔvncS (EL121), ΔvncR (EL898), and the unrelated vncS+ ΔcomD (EL117) strains was evaluated. These bacteria were grown overnight in C+Y medium, diluted to an OD620 of 0.03 (∼5 × 106 CFU per ml) in fresh C+Y or in C+Y supplemented with 1.0 μg of erythromycin per ml of medium, and compared to the untreated R6 parental control (EL59). At an OD620 of ∼0.2 (∼5 × 107 CFU per ml), cultures were challenged with 5 μg of vancomycin per ml of medium and monitored for changes in OD and viable counts through 4 h of vancomycin treatment (Fig. 3). Comparison to control cultures to which vancomycin was not added confirmed that the addition of 1.0 μg of erythromycin per ml of medium reduced the growth rate of erythromycin-resistant bacteria in C+Y medium by a factor of ∼1.8 to 2.3 (data not shown). This change in growth rate coincided with a dramatic increase in tolerance to vancomycin in all strains tested, including the unrelated vncS+ ΔcomD strain (EL117) (Fig. 3). This indicates that the addition of high levels of erythromycin to S. pneumoniae expressing erythromycin resistance from the ermAM marker (18) can in and of itself promote a vancomycin tolerance phenotype. To determine if this effect could be reproduced in erythromycin-susceptible bacteria, our erythromycin-sensitive vncS+ R6 parent strain (EL59) was treated with 0.002 to 0.004 μg of erythromycin per ml of medium and assayed for phenotypic tolerance to vancomycin in C+Y medium. At the highest concentration of erythromycin, the R6 parent strain continued to grow but had a doubling time of ∼1.7 times that of the untreated control (data not shown) and showed greatly reduced susceptibility to lysis in the presence of vancomycin (Fig. 4). These findings are consistent with a reduction in the sensitivity of S. pneumoniae to the action of the cell wall synthesis inhibitor vancomycin following changes in cell growth or other erythromycin-associated effects, but not by the loss of vncS. It is not yet known whether this is an erythromycin-specific effect or if other inhibitors of cell growth or protein synthesis would confer a similar result. Clearly, additional experiments are necessary to more fully address this question.
FIG. 3.
Addition of erythromycin to S. pneumoniae vncS, vncR, and comD strains promotes a vancomycin tolerance phenotype. Cells grown in C+Y medium with no supplementation or C+Y medium supplemented with 1 μg of erythromycin per ml [indicated by (erm)] were treated with 5 μg of vancomycin per ml (10 times the MIC). Growth or autolysis was measured as the change in OD620 (left panels), and loss of viability (right panels) was monitored by serial dilution and plating on TSAII BA. Viability data are presented as log10 CFU per milliliter of medium.
FIG. 4.
Addition of sublethal concentrations of erythromycin promotes a vancomycin tolerance phenotype in the erythromycin-sensitive R6 parent strain (EL59). R6 grown in C+Y medium with no supplementation or in C+Y medium supplemented with 0.002 or 0.004 μg of erythromycin (erm) per ml was challenged with 5 μg of vancomycin per ml (10 times the MIC). Data are presented as log10 CFU per ml of medium. Symbols: closed circles, EL59 (R6 parent) without erythromycin supplementation; open circles, EL59 (R6 parent) supplemented with 0.002 μg of erythromycin per ml; closed squares, EL59 (R6 parent) supplemented with 0.004 μg of erythromycin per ml.
Inactivation of vncS in different genetic backgrounds does not result in tolerance to vancomycin or other antibiotics in vitro or in vivo.
It is possible that subtle unknown genetic differences might exist between our R6 parent strain and the strain employed in a previous study (29). Thus, our inability to demonstrate vancomycin tolerance in the absence of erythromycin could be due to unknown differences between these separate clones of the common laboratory strain R6. To eliminate this possibility, we compared the effects of ΔvncS deletion-replacement mutations on tolerance to vancomycin challenge in C+Y medium in three common genetic backgrounds of S. pneumoniae, namely, R6, D39, and CP1250 (Table 1). As shown in Table 3, none of the ΔvncS strains derived from the R6, D39, or CP1250 parental strains exhibited any degree of tolerance to 10 times the MIC of vancomycin in C+Y medium (Table 3). By comparison, a deletion-replacement mutation in lytA (EL556), which encodes the major pneumococcal autolysin, was highly tolerant to vancomycin and, as expected, did not undergo autolysis in the presence of this antibiotic (Table 3). These findings strongly argue that LytA activity is essential for lysis in the presence of 10 times the MIC of vancomycin but that the VncS histidine protein kinase is not required for this process.
TABLE 3.
Vancomycin-induced autolysis is not altered in vncS mutants constructed in different genetic backgrounds and is not dependent on vex3 or pep27 functiona
| Strain | Description | Log10 CFU per ml at:
|
Log10CFU lossb | |
|---|---|---|---|---|
| 0 h | 4 h | |||
| EL59 | R6 parent | 7.86 ± 0.16 | 3.20 ± 0.45 | 4.66 ± 0.39 |
| EL121 | R6 vncS::ermAM | 8.07 ± 0.07 | 3.15 ± 0.45 | 4.92 ± 0.52 |
| EL822 | R6 vex3::ermAM | 8.00 ± 0.08 | 4.40 ± 0.33 | 3.60 ± 0.26 |
| EL842 | R6 pep27::ermAM | 7.76 | 4.40 | 3.36 |
| EL556 | R6 lytA::aad9 | 8.48 | 7.77 | 0.71 |
| EL161 | D39 parent | 8.15 | 4.11 | 4.03 |
| EL381 | D39 vncS::aad9 | 7.95 | 4.45 | 3.51 |
| CP1250 | Rx-based parent | 8.26 | 2.90 | 5.35 |
| EL562 | CP1250 vncS::ermAM | 8.26 | 2.04 | 6.21 |
Values with standard errors represent means of results of three independent experiments. Values without standard errors are the results of a single set of experiments.
Log10 CFU loss of ≥ 2.0 is not considered tolerant (29).
In a previous study (29), a vncS insertion-duplication mutant derived from D39 was reported to exhibit little to no killing during vancomycin therapy in a rabbit meningitis model and was found to have a selective advantage over an otherwise isogenic vncS+ parental stain. This finding led to the proposal that the loss of VncS function may lead to the failure of vancomycin therapy in vivo (29). To test the generality of this proposal, we employed an established murine neutropenic thigh model of infection to evaluate the resistance of vncS-deficient pneumococci to vancomycin therapy in vivo (see Materials and Methods) (45). In control mice that were not treated with vancomycin, the vncS strain (EL381), the vncR strain (EL395), and the D39 parent strain (EL161) were found to replicate extensively and to nearly equivalent levels (data not shown). However, in mice that received subcutaneous vancomycin therapy at 1 and 5 h postinfection, significant clearance (>4 log reduction in CFU) of all strains was observed at 8 and 24 h postinfection, and the kinetics of clearance of the vncS strain (EL381) was found to be nearly identical to that of either the vncR strain (EL395) or the D39 parent (EL161) (data not shown). Thus, inactivation of vncS appears to afford no obvious protection from vancomycin therapy in vivo in this established murine infection model system.
Our failure to demonstrate tolerance of S. pneumoniae vncS mutants to vancomycin prompted an investigation of the role of this gene product in resistance to other antibiotics to which the inactivation of vncS was reported to confer tolerance (29). Again, to ensure that the lack of observed vancomycin tolerance was not due to the nature of our previous mutations, an additional vncS deletion-replacement mutation was generated and introduced into a derivative of strain Rx (CP1250) and an independent R6 isolate (CP1260) obtained from another laboratory (Table 1). In these strains, the front half of vncS, including the active histidine, was removed and replaced with a region encoding an erythromycin resistance gene expressed from a robust synthetic promoter (Pc::ermAM) (6) transcribed in the same orientation as the original gene (CP1257 and CP1261) (Fig. 1 and Table 1). Using these strains, we found that while the kinetics of vancomycin killing for the R6-based vncS mutant (CP1261) and its R6 parent (CP1260) were somewhat more rapid than that observed for the Rx-based pair (CP1250 and CP1257), again there was no significant difference between the vncS mutant strains and their vncS+ parents (data not shown). The R6-based vncS strain (CP1261) and its isogenic parent (CP1260) were further tested for sensitivity to an aminoglycoside (streptomycin), which inhibits protein synthesis at the level of the ribosome, a fluoroquinolone (ciprofloxacin), which targets toposiomerase IV and DNA gyrase A, and two cell wall inhibitory agents, a beta-lactam (ampicillin) and cephalosporin (11). In each of the four cases, the bactericidal effect on the vncS strain (CP1261) was the same as that on its isogenic vncS+ parent (CP1260) (data not shown). Thus, in contrast to what was reported previously for the SPSJ01 vncS insertion-duplication mutant (29), these data indicate that the loss of VncS function affords pneumococci no protection from killing by these diverse autolysis-inducing antibiotics.
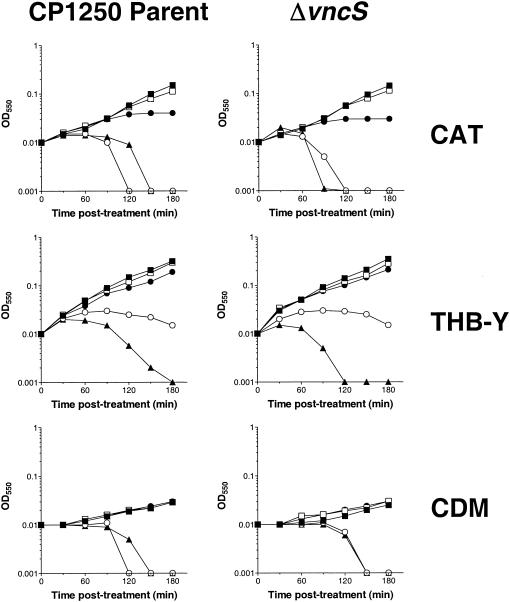
Given the surprising sensitivity of multiple vncS strains constructed in distinct genetic backgrounds to killing by vancomycin and four additional antibiotics, we evaluated whether the inactivation of vncS might cause a subtle degree of tolerance, observable in specific media at inhibitor levels close to the MIC of vancomycin. To eliminate any possibility of unknown polar effects of our previous mutations on downstream genes, an additional larger deletion of vncS was constructed; in this deletion, the majority of the coding sequence was removed and replaced with a kanamycin resistance gene expressed from the Pc synthetic promoter (Pc::aphIII) (39) transcribed in an orientation opposite to that of the original gene (strain CP1292) (Fig. 1 and Table 1). For these experiments, early-exponential-phase cultures (OD550, 0.1) of the ΔvncS::Pc::aphIII strain (CP1292) and its parent strain (CP1250) were diluted 1:10 into fresh media (i.e., CDM, THB-Y, and CAT agar) containing various concentrations of vancomycin and monitored for changes in OD550 over the course of 3 h of incubation at 37°C. Interestingly, while the sensitivity of S. pneumoniae to vancomycin differed slightly in various media, the responses of the vncS strain (CP1292) and its vncS+ parent strain (CP1250) to this inhibitor at various doses of drug were indistinguishable (Fig. 5). Susceptibility to vancomycin was also unaffected by growth of these strains in CDM or THB-Y in an atmosphere of CO2 (data not shown), and in no case was the delay in vancomycin-induced autolysis at ≥2 times the MIC extended for the vncS mutant (CP1292) relative to that of its parent strain (CP1250). Thus, mutation of vncS does not appear to afford any obvious protection against vancomycin, even at graded doses of inhibitor in rich or defined media.
FIG. 5.
Response of S. pneumoniae to challenge with vancomycin in three different growth media. CP1250 parent cells (left panels) and ΔvncS cells (right panels) grown in media specified on the right were challenged with various doses of vancomycin. Cell growth or autolysis was measured by the change in OD550. Symbols: closed squares, no vancomycin; open squares, 0.25 μg of vancomycin per ml; closed circles, 0.5 μg of vancomycin per ml; open circles, 1.0 μg of vancomycin per ml; closed triangles, 5.0 μg of vancomycin per ml.
Normal autolytic functions are unperturbed in vex3, pep27, and vncR mutants.
Our inability to reproduce the reported tolerance phenotype of vncS-deficient pneumococci led us to examine whether other components of the proposed VncRS-Pep27 signal transduction pathway were required for drug tolerance. To test this proposal, defined deletion-replacement mutants of vncR, vex3, and pep27, the latter two having been previously implicated in the reported tolerance phenomenon (28), were constructed and assayed for tolerance to vancomycin at 10 times the MIC in C+Y medium. None of the individual mutations tested afforded any protection to vancomycin challenge (Table 3). Again, all strains evaluated exhibited similar kinetics of autolysis following challenge with vancomycin (Table 3) or with another well-characterized autolysin trigger for pneumococci, namely, 0.05% deoxycholate (data not shown) (3). In contrast, the lytA control strain (EL556) stopped growing but failed to undergo autolysis under either condition (Table 3 and data not shown). In total, these results indicate that the normal autolysis-signaling pathway is unperturbed in the absence of any one component of the proposed VncRS-Pep27 signal transduction pathway.
Synthetic Pep27 does not induce cell autolytic or other death functions in pneumococci.
A small peptide, Pep27, encoded upstream of the vncRS locus (Fig. 1), was reported to function as a “death peptide,” signaling both autolytic and nonautolytic cell death functions following its accumulation in the surrounding growth medium (28). As our studies indicate that mutation of the gene responsible for synthesis of Pep27 (pep27) or of those encoding the putative transporter (vex) or sensor (vncS) did not have any discernible effect on tolerance to vancomycin, we tested whether Pep27 exhibited any inhibitory activity against S. pneumoniae. Comparison of the pep27 sequence obtained from the R6 genomic sequencing project (12) and that previously reported for R6 (28) predicted two nonconserved amino acid substitutions within Pep27 (see Materials and Methods). This finding is consistent with the reported heterogeneity of peptide quorum-sensing systems in S. pneumoniae (32, 34). Therefore, to be consistent with the previous study (28), two versions of synthetic Pep27, reflecting both the published (Pub-Pep [28]) and predicted (Lilly-Pep [12]) Pep27 sequences, were synthesized and tested for their killing effects on S. pneumoniae R6 (EL59). Using peptide that was predicted to be intact and more than 90% pure (see Materials and Methods), we challenged S. pneumoniae with 50 μM peptide at a low cell density (Fig. 6A) or with graded doses of peptide at a high cell density (Fig. 6B). In both cases, we observed no negative effect on bacterial cell growth, lysis, or viability following Pep27 treatment. Indeed, a previously described disk sensitivity assay (36) using concentrations of synthetic Pep27 ranging from 3 to 300 μM failed to reveal any detectable zone of inhibition for our R6 (EL59) or D39 (EL161) parent strains on TSAII BA (data not shown). These results are consistent with the lack of tolerance observed for our pep27 deletion-replacement mutant (EL842) (Table 3) and indicate that it is unlikely that Pep27 functions as a general effector of cell death in pneumococci.
FIG. 6.
Treatment of S. pneumoniae EL59 (R6 parent strain) with synthetic Pep27 does not inhibit growth or induce autolysis. (A) S. pneumoniae grown in CDM medium was diluted to an OD620 of ∼0.004 in CDM containing 50 μM synthetic Pep27. Growth was monitored by the change in the OD620 over time. Symbols: closed squares, no peptide control; open squares, 50 μM Pub-Pep27; open squares with dashed line, 50 μM Lilly-Pep27. (B) Cells grown in CDM to an OD620 of ∼0.45 (late exponential phase) were challenged with various doses of the two synthetic forms of Pep27. Viability was monitored by serial dilution and plating on TSAII BA. Data are presented as log10 CFU per milliliter of medium.
Expression of vex123 is derepressed in the absence of VncS.
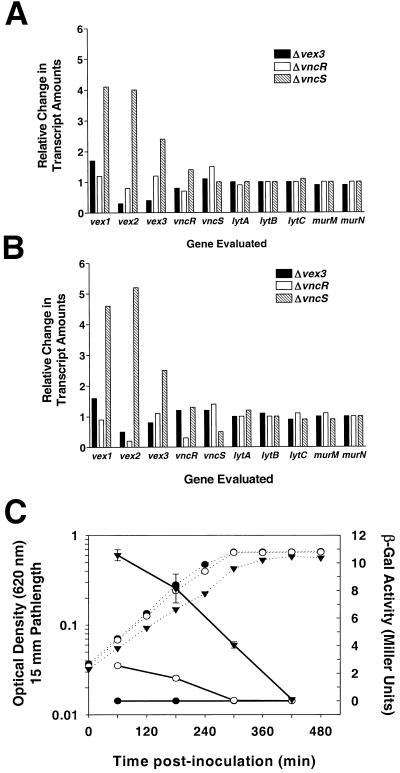
As a first step in determining the physiologic function of the VncRS signal transduction pathway, we employed microarray global transcriptional analysis to map changes in gene expression in the absence of vncS, vncR, or vex3 during mid- or late-exponential-phase growth in CDM (see Materials and Methods). By this approach, we found that very few (19 to 35) genes showed expression changes of ≥2-fold (data not shown). This is surprisingly low compared to results of similar studies evaluating the competence TCS pathways of S. pneumoniae (31, 35) and may indicate that the true physiologic inducing condition for this TCS is not accurately achieved under these experimental conditions. However, in no case was the transcription of known autolysis-related genes or genes that may regulate tolerance and cell death, such as the recently described murMN operon (9), altered in the absence of individual components of the VncRS signal transduction system (Fig. 7A and B). However, under both experimental growth conditions, we did observe induction of the vex123 gene cluster in the S. pneumoniae vncS strain (EL121) but not in other strains evaluated (Fig. 7A and B). Derepression of vex123 in the absence of VncS was highly reproducible, being observed in four additional microarray experiments, and was found to occur irrespective of the growth stage of the bacteria evaluated (Fig. 7A and B; also data not shown). To further investigate this relationship and to examine the kinetics of vex gene expression, we constructed a vex2 transcriptional fusion with the E. coli lacZ gene by insertion-duplication of the pneumococcal suicide plasmid pEVP3 in the native S. pneumoniae vex locus (see Materials and Methods) (6). In contrast with the parent strain (EL555), which contains a deletion of the bgaA gene to eliminate endogenous β-galactosidase activity (47), strain EL1066 showed β-galactosidase activity resulting from the strain's vex2::lacZ fusion that was highest during early-exponential-phase growth in BHI and that declined steadily as the cultures reached mid-exponential to late exponential growth and stationary phases relative to the corresponding increase in OD620 (Fig. 7C). Introduction of a vncS(Δ273-1021)::aad9 mutation into the EL1066 background (EL1069) did not alter the overall pattern of vex gene expression but did result in a four- to fivefold increase in β-galactosidase activity at all time points(Fig. 7C). This result is consistent with that of our global transcriptional analysis (Fig. 7A and B) and strongly argues that expression of the vex gene cluster is induced in the absence of VncS.
FIG. 7.
Transcriptional expression of vex123 is induced in the absence of VncS. (A and B) Relative changes in amounts of mRNA from select genes from microarray gene expression profiling experiments using vex3 (EL822) (closed bars), vncR (EL898) (open bars), and vncS (EL121) (hatched bars) mutants during mid-exponential-phase growth (OD620, ∼0.3) (A) or late-exponential-phase growth (OD620, ∼0.7) (B) are shown in comparison to that of the R6 parent strain (EL59) grown under the same conditions. Data are presented as ratios of the hybridization signals (see Materials and Methods) from the experimental strains (i.e., vex3, vncR, or vncS mutant) to those of the control (i.e., R6 parent). (C) Expression of a vex2::lacZ fusion in the presence or absence of VncS. Symbols: dashed lines, OD620; solid lines, β-galactosidase activity; closed circles, ΔbgaA parent strain (EL555); open circles, vex2::lacZ reporter strain (EL1066); closed triangles, ΔvncS vex2::lacZ reporter strain (EL1069); vertical bars, standard errors of the means. The experiment was conducted in triplicate and was performed twice.
DISCUSSION
The VncRS signal transduction system of S. pneumoniae has been suggested to play a key role in the regulation of cell death and autolysis in response to vancomycin challenge (10, 29). By using four distinct mutations in vncS, in some cases in multiple genetic backgrounds of S. pneumoniae, we have demonstrated that the loss of VncS function alone does not result in tolerance to vancomycin challenge or to other autolysis-inducing antibiotics to which the inactivation of vncS was reported to confer a selective advantage (28, 29). We did observe a reproducible tolerance of S. pneumoniae to vancomycin challenge during mid-exponential-phase to late exponential-phase growth (OD620, >0.15 in a 1.4-cm path length) following exposure of erythromycin-resistant or -susceptible bacteria to sublethal concentrations of erythromycin (Fig. 2A, 3, and 4). This was not dependent on mutation of vncS, as it could be reproduced in a ΔvncR strain (EL898), a ΔcomD strain (EL117) (Fig. 3), or in the vncS+ erythromycin-sensitive parent strain R6 (EL59) (Fig. 4). This could partly explain the discrepancy between our results and those reported previously (29), because in the prior study, vancomycin tolerance was routinely assayed in C+Y medium containing a concentration of erythromycin that was found to promote tolerance to vancomycin challenge in our studies (Fig. 2A and 3). Interestingly, we also found that this concentration of erythromycin impairs growth of pneumococci in C+Y medium (Fig. 2B), a property not similarly reported in the earlier study (29). Thus, whether the erythromycin-induced tolerance to vancomycin is due solely to changes in cell growth and division or to other unknown erythromycin-related effects is not yet clear. The proposition that erythromycin increases tolerance to vancomycin by altering cell growth is a testable hypothesis; however, it could also be the case that specific, as yet unidentified physiologic changes resulting from erythromycin treatment are responsible for this effect. One serious but as yet untested implication of this observation is that sublethal concentrations of erythromycin, or possibly of other static antibiotics, may promote slowed growth and phenotypic tolerance of the pneumococcus to secondary autolysis-inducing antimicrobials (i.e., vancomycin) in the clinical setting.
Growth in the presence of sublethal concentrations of erythromycin clearly induces tolerance of pneumococci to vancomycin, yet this phenomenon does not reconcile why Novak and associates (29) did not similarly observe tolerance for other erythromycin-resistant mutants [i.e., vncR::(pJDC9)] in vitro (Fig. 3) nor does it explain the discrepancies between their in vivo assays in a rabbit meningitis model and ours in neutropenic mice (see Materials and Methods). Given the number of diverse mutations constructed for this study, we find it highly unlikely that the vncS mutants evaluated here are not functionally equivalent to those reported elsewhere (10, 29). Thus, in addition to the effect of erythromycin on the tolerance of S. pneumoniae, additional factors may contribute to the phenotypes observed in previous studies (29), such as polarity or other effects of the insertion mutation or some special features of the strain pool from which SPSJ01 was isolated.
In addition to our inability to reproduce antibiotic tolerance in vncS-deficient S. pneumoniae, we were unable to demonstrate vancomycin tolerance in strains deficient in the proposed peptide signal for this response (e.g., Δpep27 [EL842]) or deficient in a component of the proposed Pep27 ABC transporter (e.g., Δvex3 [EL822]) (Table 3). Moreover, a synthetic form of the Pep27 death peptide failed to elicit a negative physiologic response in our R6 parent strain (EL59) at concentrations reported to promote both lytic and nonlytic killing of this bacterium (28). Both results argue against the model for vancomycin tolerance in S. pneumoniae that was advanced previously (28), and taken together, they support the lack of tolerance observed for our ΔvncS mutants. Microarray global transcription analysis of S. pneumoniae ΔvncR, ΔvncS, and Δvex3 mutants failed to identify altered expression of genes known to contribute to autolysis (i.e., lytA, lytB, and lytC) or tolerance to vancomycin (i.e., murMN). These studies did reveal a strong correlation between the loss of vncS function and the induction of the upstream vex123 gene cluster (Fig. 7A and B). These microarray data were supported by direct measurement of vex2 gene expression with β-galactosidase reporter assays in vncS+ and ΔvncS S. pneumoniae strains (Fig. 7C) and are consistent with a role for the VncRS signal transduction system in the transcriptional regulation of vex gene expression. This is not uncommon for TCSs whose targets are often proximal to the genes encoding the corresponding histidine protein kinase and response regulator (8).
As transcriptional changes of the vex gene cluster were not similarly altered in the absence of vncR or vex3 itself, the effect of the vncS deletion on vex123 may be either indirect and dependent on VncR being locked in a nonphosphorylated state or, alternatively, dependent on other properties resulting from the loss of VncS activity. We propose either that VncR in its nonphosphorylated state functions as an activator of vex123 gene expression or, alternatively, that phosphorylated VncR is required for repression of these genes. Both models can adequately explain the induction of vex123 transcription in the absence of vncS function. However, given the lack of vex induction for the vncR mutant (EL898), we favor the former model. Additional experiments are needed to confirm such a relationship. It also remains to be determined what represents the true physiologic signal(s) for VncRS signal transduction and what roles this poorly characterized TCS plays in pneumococcal biology. If, as was previously reported (28), Pep27 promotes dephosphorylation of VncR through stimulation of VncS phosphatase activity, we would anticipate seeing the induction of vex gene expression following challenge with synthetic forms of Pep27. However, preliminary experiments using microarrays and vex2::lacZ reporter assays have failed to establish such a correlation (data not shown). It is noteworthy that while inactivation of vncR or vncS does not alter vancomycin-mediated clearance of pneumococci from a neutropenic murine thigh model (see Results), this TCS has been implicated in the colonization of the mouse lung (41). Thus, while inactivation of the vncRS signal transduction system does not confer a general vancomycin tolerance phenotype, this TCS may contribute to the capacity of pneumococci to produce disease in a host. Given the small number of transcriptional changes observed in our microarray studies (Fig. 7A and B and data not shown), it is not possible to speculate on the basis of this attenuation. Indeed, this may suggest that the activation signals for this TCS are observed only during colonization of a susceptible host. Regardless, given that antibiotic tolerance is not a general property of strains lacking VncS activity and the requirement for this and other TCSs in pathogenesis and viability of S. pneumoniae (41; G.T. Robertson, unpublished data), TCSs may still represent potential targets for broad-spectrum antibiotics.
Acknowledgments
We thank J. P. Claverys, A. Tomasz, and J. Yother for bacterial strains, D. Smiley and J. Richardson for synthesis and purification of synthetic Pep27, P. M. Sun for initial mutagenesis work, C. M. Morris for initial MIC testing, J. Foley for assistance with animal studies, and W.-L. Ng for helpful discussions regarding microarray experiments.
This work was supported by resources provided by Lilly Research Laboratories and Proctor and Gamble Pharmaceuticals and by grant MCB-9722821 from the National Science Foundation to D.A.M. Gregory Robertson was supported by a Lilly Postdoctoral Fellowship.
REFERENCES
- 1.Anton, N., R. Blazquez, J. L. Gomez-Garces, and J. I. Alos. 2001. Study of vancomycin tolerance in 120 strains of Streptococcus pneumoniae isolated in 1999 in Madrid, Spain. J. Antimicrob. Chemother. 47:902-903. [DOI] [PubMed] [Google Scholar]
- 2.Baltz, R. H., F. H. Norris, P. Matsushima, B. S. Dehoff, P. Rockey, G. Porter, S. Burgett, R. B. Peery, J. Hoskins, L. Braverman, I. Jenkins, P. Solenberg, M. Young, M. A. McHenney, P. R. Rosteck, Jr., and P. L. Skatrud. 2000. DNA sequence sampling and gene disruption for identification of new antibacterial targets in Streptococcus pneumoniae, p. 33-44. In A. Tomasz (ed.), Streptococcus pneumoniae: molecular biology and mechanisms of disease. Mary Ann Liebert, Inc., Larchmont, N.Y.
- 3.Berge, M., P. Garcia, F. Iannelli, M. F. Prere, C. Granadel, A. Polissi, and J. P. Claverys. 2001. The puzzle of zmpB and extensive chain formation, autolysis defect and non-translocation of choline-binding proteins in Streptococcus pneumoniae. Mol. Microbiol. 39:1651-1660. [DOI] [PubMed] [Google Scholar]
- 4.Charpentier, E., R. Novak, and E. Tuomanen. 2000. Regulation of growth inhibition at high temperature, autolysis, transformation and adherence in Streptococcus pneumoniae by clpC. Mol. Microbiol. 37:717-726. [DOI] [PubMed] [Google Scholar]
- 5.Chastanet, A., M. Prudhomme, J. P. Claverys, and T. Msadek. 2001. Regulation of Streptococcus pneumoniae clp genes and their role in competence development and stress survival. J. Bacteriol. 183:7295-7307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Claverys, J. P., A. Dintilhac, E. V. Pestova, B. Martin, and D. A. Morrison. 1995. Construction and evaluation of new drug-resistance cassettes for gene disruption mutagenesis in Streptococcus pneumoniae, using an ami test platform. Gene 164:123-128. [DOI] [PubMed] [Google Scholar]
- 7.Claverys, J. P., C. Granadel, A. M. Berry, and J. C. Paton. 1999. Penicillin tolerance in Streptococcus pneumoniae, autolysis and the Psa ATP-binding cassette (ABC) manganese permease. Mol. Microbiol. 32:881-883. [DOI] [PubMed] [Google Scholar]
- 8.de Saizieu, A., C. Gardes, N. Flint, C. Wagner, M. Kamber, T. J. Mitchell, W. Keck, K. E. Amrein, and R. Lange. 2000. Microarray-based identification of a novel Streptococcus pneumoniae regulon controlled by an autoinduced peptide. J. Bacteriol. 182:4696-4703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Filipe, S. R., E. Severina, and A. Tomasz. 2002. The murMN operon: a functional link between antibiotic resistance and antibiotic tolerance in Streptococcus pneumoniae. Proc. Natl. Acad. Sci. USA 99:1550-1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Henriques Normark, B., R. Novak, A. Ortqvist, G. Kallenius, E. Tuomanen, and S. Normark. 2001. Clinical isolates of Streptococcus pneumoniae that exhibit tolerance of vancomycin. Clin. Infect. Dis. 32:552-558. [DOI] [PubMed] [Google Scholar]
- 11.Hooper, D. C. 2001. Mechanisms of action of antimicrobials: focus on fluoroquinolones. Clin. Infect. Dis. 32(Suppl. 1):S9-S15. [DOI] [PubMed] [Google Scholar]
- 12.Hoskins, J., W. E. Alborn, Jr., J. Arnold, L. C. Blaszczak, S. Burgett, B. S. DeHoff, S. T. Estrem, L. Fritz, D. J. Fu, W. Fuller, C. Geringer, R. Gilmour, J. S. Glass, H. Khoja, A. R. Kraft, R. E. Lagace, D. J. LeBlanc, L. N. Lee, E. J. Lefkowitz, J. Lu, P. Matsushima, S. M. McAhren, M. McHenney, K. McLeaster, C. W. Mundy, T. I. Nicas, F. H. Norris, M. O'Gara, R. B. Peery, G. T. Robertson, P. Rockey, P. M. Sun, M. E. Winkler, Y. Yang, M. Young-Bellido, G. Zhao, C. A. Zook, R. H. Baltz, S. R. Jaskunas, P. R. Rosteck, Jr., P. L. Skatrud, and J. I. Glass. 2001. Genome of the bacterium Streptococcus pneumoniae strain R6. J. Bacteriol. 183:5709-5717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Klugman, K. P., and C. Feldman. 2001. Streptococcus pneumoniae respiratory tract infections. Curr. Opin. Infect. Dis. 14:173-179. [DOI] [PubMed] [Google Scholar]
- 14.Lacks, S., and R. D. Hotchkiss. 1960. A study of the genetic material determining an enzyme activity in pneumococcus. Biochim. Biophys. Acta 39:508-518. [DOI] [PubMed] [Google Scholar]
- 15.Lau, P. C., C. K. Sung, J. H. Lee, D. A. Morrison, and D. G. Cvitkovitch. 2002. PCR ligation mutagenesis in transformable streptococci: application and efficiency. J. Microbiol. Methods 49:193-205. [DOI] [PubMed] [Google Scholar]
- 16.LeBlanc, D. J., L. N. Lee, and A. Abu-Al-Jaibat. 1992. Molecular, genetic, and functional analysis of the basic replicon of pVA380-1, a plasmid of oral streptococcal origin. Plasmid 28:130-145. [DOI] [PubMed] [Google Scholar]
- 17.Lee, M. S., and D. A. Morrison. 1999. Identification of a new regulator in Streptococcus pneumoniae linking quorum sensing to competence for genetic transformation. J. Bacteriol. 181:5004-5016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Macrina, F. L., J. A. Tobian, K. R. Jones, R. P. Evans, and D. B. Clewell. 1982. A cloning vector able to replicate in Escherichia coli and Streptococcus sanguis. Gene 19:345-353. [DOI] [PubMed] [Google Scholar]
- 19.Matsushita, M., and K. D. Janda. 2002. Histidine kinases as targets for new antimicrobial agents. Bioorg. Med. Chem. 10:855-867. [DOI] [PubMed] [Google Scholar]
- 20.McCullers, J. A., B. K. English, and R. Novak. 2000. Isolation and characterization of vancomycin-tolerant Streptococcus pneumoniae from the cerebrospinal fluid of a patient who developed recrudescent meningitis. J. Infect. Dis. 181:369-373. [DOI] [PubMed] [Google Scholar]
- 21.Miller, J. H. 1972. Experiments in molecular genetics. Cold Spring Harbor Laboratories, Cold Spring Harbor, N.Y.
- 22.Mitchell, L., and E. Tuomanen. 2001. Vancomycin-tolerant Streptococcus pneumoniae and its clinical significance. Pediatr. Infect. Dis. J. 20:531-533. [DOI] [PubMed] [Google Scholar]
- 23.Moreillon, P., and A. Tomasz. 1988. Penicillin resistance and defective lysis in clinical isolates of pneumococci: evidence for two kinds of antibiotic pressure operating in the clinical environment. J. Infect. Dis. 157:1150-1157. [DOI] [PubMed] [Google Scholar]
- 24.Morrison, D. A., S. A. Lacks, W. R. Guild, and J. M. Hageman. 1983. Isolation and characterization of three new classes of transformation-deficient mutants of Streptococcus pneumoniae that are defective in DNA transport and genetic recombination. J. Bacteriol. 156:281-290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.National Research Council. 1985. Guide for the care and use of laboratory animals. NIH publication no. 86-23. National Institutes of Health, Bethesda, Md.
- 26.Novak, R., J. S. Braun, E. Charpentier, and E. Tuomanen. 1998. Penicillin tolerance genes of Streptococcus pneumoniae: the ABC-type manganese permease complex Psa. Mol. Microbiol. 29:1285-1296. [DOI] [PubMed] [Google Scholar]
- 27.Novak, R., E. Charpentier, J. S. Braun, E. Park, S. Murti, E. Tuomanen, and R. Masure. 2000. Extracellular targeting of choline-binding proteins in Streptococcus pneumoniae by a zinc metalloprotease. Mol. Microbiol. 36:366-376. [DOI] [PubMed] [Google Scholar]
- 28.Novak, R., E. Charpentier, J. S. Braun, and E. Tuomanen. 2000. Signal transduction by a death signal peptide: uncovering the mechanism of bacterial killing by penicillin. Mol. Cell 5:49-57. [DOI] [PubMed] [Google Scholar]
- 29.Novak, R., B. Henriques, E. Charpentier, S. Normark, and E. Tuomanen. 1999. Emergence of vancomycin tolerance in Streptococcus pneumoniae. Nature 399:590-593. [DOI] [PubMed] [Google Scholar]
- 30.Pestova, E. V., and D. A. Morrison. 1998. Isolation and characterization of three Streptococcus pneumoniae transformation-specific loci by use of a lacZ reporter insertion vector. J. Bacteriol. 180:2701-2710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Peterson, S., R. T. Cline, H. Tettelin, V. Sharov, and D. A. Morrison. 2000. Gene expression analysis of the Streptococcus pneumoniae competence regulons by use of DNA microarrays. J. Bacteriol. 182:6192-6202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pozzi, G., L. Masala, F. Iannelli, R. Manganelli, L. S. Havarstein, L. Piccoli, D. Simon, and D. A. Morrison. 1996. Competence for genetic transformation in encapsulated strains of Streptococcus pneumoniae: two allelic variants of the peptide pheromone. J. Bacteriol. 178:6087-6090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reese, R. E., R. F. Betts, and B. Gumustop. 2000. Handbook of antibiotics, 3rd ed. Lippincott, Williams & Wilkins, Philadelphia, Pa.
- 34.Reichmann, P., and R. Hakenbeck. 2000. Allelic variation in a peptide-inducible two-component system of Streptococcus pneumoniae. FEMS Microbiol. Lett. 190:231-236. [DOI] [PubMed] [Google Scholar]
- 35.Rimini, R., B. Jansson, G. Feger, T. C. Roberts, M. de Francesco, A. Gozzi, F. Faggioni, E. Domenici, D. M. Wallace, N. Frandsen, and A. Polissi. 2000. Global analysis of transcription kinetics during competence development in Streptococcus pneumoniae using high density DNA arrays. Mol. Microbiol. 36:1279-1292. [DOI] [PubMed] [Google Scholar]
- 36.Robertson, G. T., W.-L. Ng, J. Foley, R. Gilmour, and M. E. Winkler. 2002. Global transcriptional analysis of clpP mutations of type 2 Streptococcus pneumoniae and their effects on physiology and virulence. J. Bacteriol. 184:3508-3520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Smith, M. D., and W. R. Guild. 1979. A plasmid in Streptococcus pneumoniae. J. Bacteriol. 137:735-739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Stock, A. M., V. L. Robinson, and P. N. Goudreau. 2000. Two-component signal transduction. Annu. Rev. Biochem. 69:183-215. [DOI] [PubMed] [Google Scholar]
- 39.Sung, C. K., H. Li, J. P. Claverys, and D. A. Morrison. 2001. An rpsL cassette, janus, for gene replacement through negative selection in Streptococcus pneumoniae. Appl. Environ. Microbiol. 67:5190-5196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Talkington, D. F., D. C. Voellinger, L. S. McDaniel, and D. E. Briles. 1992. Analysis of pneumococcal PspA microheterogeneity in SDS polyacrylamide gels and the association of PspA with the cell membrane. Microb. Pathog. 13:343-355. [DOI] [PubMed] [Google Scholar]
- 41.Throup, J. P., K. K. Koretke, A. P. Bryant, K. A. Ingraham, A. F. Chalker, Y. Ge, A. Marra, N. G. Wallis, J. R. Brown, D. J. Holmes, M. Rosenberg, and M. K. Burnham. 2000. A genomic analysis of two-component signal transduction in Streptococcus pneumoniae. Mol. Microbiol. 35:566-576. [DOI] [PubMed] [Google Scholar]
- 42.Tomasz, A. 1999. New faces of an old pathogen: emergence and spread of multidrug-resistant Streptococcus pneumoniae. Am. J. Med. 107:55S-62S. [DOI] [PubMed] [Google Scholar]
- 43.Tomasz, A., A. Albino, and E. Zanati. 1970. Multiple antibiotic resistance in a bacterium with suppressed autolytic system. Nature 227:138-140. [DOI] [PubMed] [Google Scholar]
- 44.Tuomanen, E., H. Pollack, A. Parkinson, M. Davidson, R. Facklam, R. Rich, and O. Zak. 1988. Microbiological and clinical significance of a new property of defective lysis in clinical strains of pneumococci. J. Infect. Dis. 158:36-43. [DOI] [PubMed] [Google Scholar]
- 45.Vogelman, B., S. Gudmundsson, J. Turnidge, J. Leggett, and W. A. Craig. 1988. In vivo postantibiotic effect in a thigh infection in neutropenic mice. J. Infect. Dis. 157:287-298. [DOI] [PubMed] [Google Scholar]
- 46.Williamson, R., and A. Tomasz. 1980. Antibiotic-tolerant mutants of Streptococcus pneumoniae that are not deficient in autolytic activity. J. Bacteriol. 144:105-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zahner, D., and R. Hakenbeck. 2000. The Streptococcus pneumoniae beta-galactosidase is a surface protein. J. Bacteriol. 182:5919-5921. [DOI] [PMC free article] [PubMed] [Google Scholar]