Abstract
Conventional kinesin is a ubiquitous organelle transporter that moves cargo toward the plus-ends of microtubules. In addition, several in vitro studies indicated a role of conventional kinesin in cross-bridging and sliding microtubules, but in vivo evidence for such a role is missing. In this study, we show that conventional kinesin mediates microtubule-microtubule interactions in the model fungus Ustilago maydis. Live cell imaging and ultrastructural analysis of various mutants in Kin1 revealed that this kinesin-1 motor is required for efficient microtubule bundling and participates in microtubule bending in vivo. High levels of Kin1 led to increased microtubule bending, whereas a rigor-mutation in the motor head suppressed all microtubule motility and promoted strong microtubule bundling, indicating that kinesin can form cross-bridges between microtubules in living cells. This effect required a conserved region in the C terminus of Kin1, which was shown to bind microtubules in vitro. In addition, a fusion protein of yellow fluorescent protein and the Kin1tail localized to microtubule bundles, further supporting the idea that a conserved microtubule binding activity in the tail of conventional kinesins mediates microtubule-microtubule interactions in vivo.
INTRODUCTION
Conventional kinesin is an ATP-dependent motor enzyme that supports the unidirectional movement of membranous vesicles along microtubules (Brady, 1985; Scholey et al., 1985; Vale et al., 1985). The heavy chain of conventional kinesin is an elongated molecule with a globular motor domain (head) at the N terminus, a stalk consisting of several coiled-coil segments, and a small C-terminal globular domain (Yang et al., 1989). In animal kinesin, two paired heavy chains interact at their C-terminal portions with two light chains (Diefenbach et al., 1998). Light chains are not essential for motor activity (Yang et al., 1990), but they are required for in vivo function (Gindhart et al., 1998; Rahmann et al., 1999). Conventional kinesins isolated from various fungal species (Steinberg and Schliwa, 1995; Lehmler et al., 1997; Steinberg, 1997; Grummt et al., 1998; Wu et al., 1998) share significant sequence homology and a similar domain structure with animal conventional kinesins (Kirchner et al., 1999). However, fungal kinesin purifies without light chains (Steinberg and Schliwa, 1996; Steinberg, 1997; Steinberg et al., 1998), and no conserved light chains are present in the genome of fungi, such as Neurospora crassa and Ustilago maydis (our unpublished data). In both fungal and animal kinesins, the motor domain contains a microtubule binding interface and a nucleotide binding site that couples ATP hydrolysis to movement along microtubules. The tail of the molecule is thought to mediate binding to organelles or to other substrates moved by kinesin (Skoufias et al., 1994). In addition, a highly conserved site in the globular C-terminal domain was determined to be involved in self-inactivation of conventional kinesin that is characterized by a folded conformation (Coy et al., 1999; Friedman and Vale, 1999; Stock et al., 1999).
Although numerous evidence exists for a role of kinesin-1 in organelle transport (Allan, 1995; Hirokawa, 1998), in vitro experiments demonstrated that purified kinesin-1 is able to promote microtubule bundling and sliding (Vale et al., 1985; Urrutia et al., 1991). Furthermore, ultrastructural analysis of microtubule bundles induced by incubation with squid brain kinesin and the nonhydrolyzable ATP analogue AMP-PNP revealed that single kinesin molecules can cross-bridge microtubules, which involves the motor domain as well as the C-terminal end of the molecule (Andrews et al., 1993). This suggests that the tail of kinesin-1 contains a second microtubule binding site, as was indicated by Navone et al. (1992) and Hackney and Stock (2000). Although the ability of purified conventional kinesins to cross-bridge microtubules is known for ∼20 years now, no physiological role in microtubule organization has been reported to date.
In this study, we use a simple eukaryotic model system to investigate the function of conventional kinesin in the organization of interphase microtubules in the living cell. In growing interphase cells of the fungus Ustilago maydis, microtubules are thought to be nucleated at a microtubule organizing center near the constriction between mother and daughter cell (Straube et al., 2003). On average, four microtubule tracks span the length of a cell, some of which represent bundles (Steinberg et al., 2001). Dynamic rearrangements such as the movement of short microtubules along the cell cortex and sliding and looping of microtubules are frequently observed in living U. maydis cells and occur at an average speed above 30 μm/min, suggesting that molecular motors are involved in these processes (Steinberg et al., 2001). Thus, U. maydis is an excellent model system to study the mechanisms underlying microtubule organization in vivo. Kin1, the conventional kinesin of U. maydis, was reported to be involved in polar hyphal growth (Lehmler et al., 1997, named Kin2 therein; Schuchardt et al., 2005) and organization of the vacuolar compartment (Steinberg et al., 1998), both cellular roles that are consistent with its assumed role in membrane traffic. Here, we provide strong indications that Kin1 also mediates microtubule-microtubule interactions in vivo, suggesting that it helps to organize the interphase microtubule arrays in U. maydis.
MATERIALS AND METHODS
Strains and plasmids
All U. maydis strains were in the genetic background of FB2 (Table 1). Microtubules were stained with green fluorescent protein (GFP)-α-tubulin (Steinberg et al., 2001) or cyan fluorescent protein (CFP)-tubulin (Wedlich-Soldner et al., 2002). Generation of kin1 mutant strains were described previously (named Kin2 in Lehmler et al., 1997). Point mutation G96E was introduced to kin1 in a PCR-based approach. The rigor mutation G105E in kin3 was described previously (Wedlich-Soldner et al., 2002). The fusion protein of Kin3rigor head and Kin1 tail was generated using the BssHII site at the head-neck border in kin1 and introducing a BssHII site by PCR to the 3′ end of kin3 head sequence. Deletions in the kin1 tail were chosen according predictions with Coils (http://www.ch.embnet.org/software/COILS_form.html). To delete C2, an NgoMIV site was introduced by PCR after the codon for S553 and fused to the NgoMIV site at A740. Deletion of C3 and G2, the EcoRI site in the 3′-untranslated region of kin1 was filled with Klenow and fused to Mung-bean-Nuclease-blunted NgoMIV site to create truncated protein, where T739 were followed by amino acids E and F with the COOH terminus. To create the yellow fluorescent protein (YFP)-Kin1T fusion protein, the YFP coding region was amplified by PCR with an NcoI and a BssHII site at the flanks and used to replace the Kin3 portion in the Kin3rigorK1T construct. For recombinant expression of UmC3G2 and HsC3G2, appropriate regions (Table 1) were amplified from pKin2 (Lehmler et al., 1997) or random primed cDNA from human neuroblastoma cells (a gift from W. Lutz, IMT, Marburg, Germany), respectively, thereby introducing nucleotides CACC just before the start codon. This allowed directional cloning into pBAD102-TOPO (Invitrogen, Carlsbad, CA) and subsequently the expression as C-terminally V5- and 6His-tagged proteins in Escherichia coli. Kinesin tail fragments were expressed in TOP10 cells (Invitrogen) after induction with 0.0025% arabinose for 4 h. All motor constructs were expressed in U. maydis cells, grown overnight in complete medium (CM) supplemented with 1% arabinose.
Table 1.
Strains and plasmids used in this study
| Strains and plasmids | Genotype | Reference |
|---|---|---|
| FB2 | a2b2 | Banuett and Herskowitz (1989) |
| FB2GT | a2b2/potefGFPTub1 | Adamikova et al. (2004) |
| AB33G3Kin1 | a2 PnarbW2 PnarbE1, bleR/pGFP3Kin1 | Schuchardt et al. (2005) |
| ΔKin1YK1TCT | a2b2 Δkin1::hphR/pYFPKin1T/potefCFPTub1 | This study |
| ΔKin1GT | a2b2 Δkin1::hphR/potefGFPTub1 | This study |
| rKin1GT | a2b2 Pcrg-kin1, natR/potefGFPTub1 | =FB2rKin2GT (Straube et al., 2003) |
| FB2ΔKin1 | a2b2 Δkin1::hphR | =FB2ΔKin2 (Lehmler et al., 1997) |
| FB2rKin1 | a2b2 Pcrg-kin1, natR | =FB2rKin2 (Straube et al., 2003) |
| FB2rGFPTub1 | a2b2/prGFPTub1 | Steinberg et al. (2001) |
| FB2Peb1RGT | a2b2 peb1-mrfp, bleR/potefGFPTub1 | This study |
| rKin1rigorGT | a2b2/potefGFPTub1/pHcrgKin1G96E | This study |
| rKin3rigorGT | a2b2/potefGFPTub1/pHcrgKin3G105E | This study |
| rK3rigorK1TGT | a2b2/potefGFPTub1/pHcrgK3G105E-K1T | This study |
| rKin1rigor | a2b2/pHcrgKin1G96E | This study |
| rK3rigorK1T | a2b2/pHcrgK3G105E-K1T | This study |
| rK3rigorK1TΔC2GT | a2b2/potefGFPTub1/pHcrgK3G105E-K1TΔC2 | This study |
| rK3rigor-K1TΔC3G2GT | a2b2/potefGFPTub1/pHcrgK3G105E-K1TΔC3G2 | This study |
| potefCFPTub1 | Potef-cfp-tub1, hphR | Wedlich-Soldner et al. (2002) |
| prGFPTub1 | Pcrg-egfp-tub1, cbxR | Steinberg et al. (2001) |
| pYFPKin1T | Pcrg-yfp-kin1336-968, cbxR | This study |
| potefGFPTub1 | Potef-egfp-tub1, cbxR | Steinberg et al. (2001) |
| pHcrgKin1G96E | Pcrg-kin1G96E, hphR | This study |
| pHcrgKin3G105E | Pcrg-kin3G105E, hphR | This study |
| pHcrgK3G105E-K1T | Pcrg-kin31-363 G105E-kin1336-968, hphR | This study |
| pHcrgK3G105E-K1TΔC2 | Pcrg-kin31-363 G105E-kin1336-968 Δ554-739, hphR | This study |
| pHcrgK3G105E-K1TΔC3G2 | Pcrg-kin31-363 G105E-kin1336-739, hphR | This study |
| pBAD-HsC3G2 | PBAD-hpt-khc808-963-V5-6H, blaR | This study |
| pBAD-UmC3G2 | PBAD-hpt-kin1740-968-V5-6H, blaR | This study |
Strain nomenclature is as follows: GT, GFP-tubulin; H, head; T, tail; r, regulated expression; Δ, deletion; rigor, not ATP cleavage and tight binding to microtubules; Genotype nomenclature: a, b, mating type loci; Δ, deletion; P, promoter; ::, homologous replacement;-, fusion; hphR, hygromycine resistance; cbxR, carboxin resistance; /, ectopically integrated; yfp, yellow-shifted derivative of green fluorescent protein; and mrfp, monomeric red fluorescein.
Antibody Generation
Rabbit antibodies against Kin1 were raised against the oligopeptides C-220QQRNTETGSAKTGNL234 and C-951SLGENSPKARSSWF964 (Eurogentec, Herstal, Belgium). Rabbit anti-Kin3 antibodies were raised against recombinant Kin31-431 (Davids Biotechnologie, Regensburg, Germany). Both sera were affinity purified against the recombinant Kin31-431 fragment and full-length Kin1 protein (kindly provided by C. Horn and M. Schliwa, Institute for Cell Biology, Munich, Germany) following described protocols (Steinberg and Schliwa, 1995).
Western Blot Analysis and Microtubule Pull-Down Assay
Cell extracts of U. maydis and E. coli cells were prepared in PMEGI (100 mM PIPES, pH 6.9, 2 mM MgCl2, 1 mM EDTA, 1 mM EGTA, 0.9 M glycerol, and complete protease inhibitor; Roche Diagnostics, Mannheim, Germany) and processed for Western analysis as described previously (Straube et al., 2001). Kin1 and Kin3 were detected with specific affinity-purified antibodies (see above). Tubulin antibodies were from Oncogene Science (Cambridge, MA). Cell extracts were cleared by high-speed centrifugation at 200,000 × g for 1 h, supplemented with 2 mM adenylyl imidodiphosphate (AMP-PNP) and 10 μM taxol (both from Sigma, Taufkirchen, Germany), and incubated with taxol-stabilized microtubules (tubulin kindly provided by T. Surrey, EMBL, Heidelberg, Germany) for 1 h at 4°C. Microtubules were sedimented at 40,000 × g for 30 min, subsequently resuspended in PMEGI with 0.5 mM AMP-PNP and 10 μM taxol, and centrifuged through a 20% sucrose cushion. Release was done in PMEGI with 10 μM taxol and 10 mM MgATP. Pellets were resuspended in one-fourth of input volume. UmC3G2 and HsC3G2 were detected on Western blots with anti-His-tag antibodies (Sigma).
Light and Electron Microscopy and Analysis of Microtubule Bundling and Bending
Cells from logarithmically growing cultures were prepared and observed as described previously (Steinberg et al., 2001; Straube et al., 2003). In brief, logarithmically growing cells were embedded in 1% low melt agarose and observed using a Zeiss Axioplan-Imaging II microscope and the standard fluorescein isothiocyanate filter set. To minimize the risk of artifacts because of oxygen depletion or radiation, each preparation was used for 5-6 min only. Image acquisition started at 40-50% lamp intensity (AttoArc 100-W HBO; Carl Zeiss, Jena, German). For speckle analysis, strain FB2rGFPTub1 (Steinberg et al., 2001) was grown in complete medium supplemented with 1% arabinose (CM-A). Under these conditions, cells express an additional copy of α-tubulin that is fused to GFP, whereas the crg-promoter is repressed in the presence of 1% glucose (CM-G; Bottin et al., 1996). After shift to CM-G, the amount of GFP-α-tubulin decreases with time. GFP speckles in the microtubule become visible after ∼4 h. For quantitative analysis of microtubule bending and bundling, timed image stacks of 60 frames at an interval of 500 ms were taken using a cooled charge-coupled device (CCD) camera (C4742-95; Hamamatsu, Bridgewater, NJ) controlled by Image-Pro Plus (Media Cybernetics, Silver Spring, MD). A microtubule bending event was defined as the process of bending or straightening of a microtubule that changed the angle of the microtubule relative to the cell axis >30°. To be counted as separate events, such bending had to occur either in different cell regions or be separated temporally by >2 s. To calculate the number of microtubules present per bundles, we measured the highest average fluorescence intensity of 9-10 microtubule signals and divided it by the mean average intensity of 9-10 individual microtubules. For each strain analyzed, microtubule signals were chosen from 40 to 50 cells and measured using the MetaMorph software (Universal Imaging, Downingtown, PA). For ultrastructural studies, cells were high pressure frozen (HPM 010, BAL-TEC, Liechtenstein), cryosubstituted in 0.25% glutaraldehyde (Sigma) and 0.1% uranyl acetate (Chemapol, Prague, Czech Republic) in acetone for 5 d using cryosubstitution equipment (FSU; BAL-TEC). This was followed by embedding in HM20 (Polysciences Europe, Eppelheim, Germany) at -20°C. Sections were poststained with uranyl acetate and lead citrate in an EMstain apparatus (Leica, Bensheim, Germany) and subsequently observed with an EM 900 transmission electron microscope (LEO, Oberkochen, Germany). Micrographs were taken with a Variospeed slow scan CCD camera (TRS, Moorenweis, Germany). For the quantification of microtubule bundling, cross-sections of U. maydis cells were searched for perpendicular-sectioned microtubules. Microtubules closer than 100 nm to each other were considered as being bundled, although we never observed distances between 20 and 100 nm in control cells. However, groups of microtubules were embedded in a fine matrix in some mutant strains (for an example, see Figure 3, D2) and therefore represented bundles. Distances of up to 100 nm were observed between neighboring microtubules in such bundles.
Figure 3.
Kinesin rigor mutants induce rigid microtubule cross-bridges. (A) A point mutation in the P-loop of kinesins interferes with ATP hydrolysis and confers “rigorous” binding to microtubules (Wedlich-Soldner et al., 2002). The fusion protein K3rigorK1T consists of the tail of Kin1 and the rigor mutated motor domain of Kin3, a Kif1a/Unc104 homologue of U. maydis. (B) Mutated Kin1 protein (Kin1rigor) but not Kin3rigor led to thicker microtubule bundles. Cells expressing the K3rigorK1T fusion protein contained long and very bright microtubule bundles (K3rigorK1T). Arrowheads mark thick microtubule bundles. Bar, 5 μm. (C) Both Kin1rigor and K3rigorK1T inhibited microtubule bending, whereas Kin3rigor was without effect. See supplemental movies (Videos 5 and 6). (D) In cells expressing Kin1rigor (D1) or K3rigorK1T (D2 and D3), microtubule bundles of up to nine microtubules were found that were surrounded by a fine matrix. Bars, 100 nm. (E) 69% and 86% of cross-sectioned microtubules were bundled in Kin1rigor- and K3rigorK1T-expressing cells, respectively. (F) Fluorescence intensity measurement of GFP-labeled microtubule bundles in various kinesin mutants demonstrated that bundles in control cells contain on average 3 microtubules. In kin1 deletion mutants, the number of microtubules in the remaining bundles is significantly reduced. Expression of Kin1 at a high level significantly enhances microtubule bundling (p values are indicated by asterisks). The rigor constructs Kin1rigor and Kin3rigorK1T raise the number per bundle to approximately 5 and approximately 7.5, respectively, whereas Kin3rigor was without effect. (G) The mean distance between neighboring microtubules was determined to be ∼10 nm for control cells, ∼41 nm for Kin1rigor, and ∼45 nm for K3rigorK1T.
RESULTS
A Putative Microtubule Binding Site in the Conventional Kinesin Tail
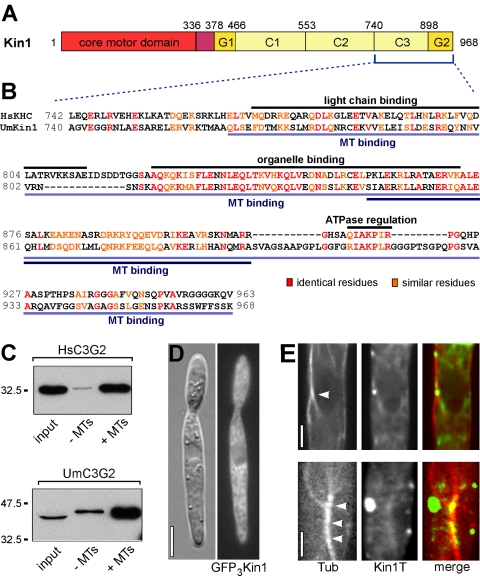
Kin1 shares significant sequence homology and a similar domain structure with animal conventional kinesins (Lehmler et al., 1997). The N-terminal motor domain (Figure 1A, red) is separated from the stalk by a neck linker (Figure 1A, purple). The stalk is predicted to contain three coiled-coil regions (C1-C3) and a C-terminal globular domain (Figure 1A, yellow, G2). Functional regions for organelle binding and ATPase regulation have both been mapped to the C-terminal one-third of the kinesin stalk (Figure 1B, black bars above sequence alignment; Diefenbach et al., 1998; Stock et al., 1999; Seiler et al., 2000). Expression of a human kinesin tail fragment in CV-1 monkey kidney epithelial cells (Figure 1B; light blue bar below alignment) demonstrated that the C-terminal 196 amino acids of human kinesin heavy chain (KHC) localize to microtubules in vivo (Navone et al., 1992). Interestingly, Hackney and Stock (2000) assigned an in vitro microtubule binding activity to a stretch of 55 amino acids between the putative organelle binding region and the proposed ATPase regulation site, which is highly conserved between human and U. maydis kinesin-1 (65% similarity; Figure 1B, dark blue bar below alignment). This suggested that the tail of Kin1 could also be able to bind microtubules in vitro. To test for such an activity of fungal kinesin-1, we expressed C-terminal kinesin fragments from U. maydis Kin1 as 6xHis-tagged polypeptides in E. coli and performed microtubule pull-down experiments. As a control, we included the respective tail fragment of human KHC. Indeed, both proteins specifically coprecipitated with pig brain microtubules (Figure 1C), indicating that the tail of Kin1 binds microtubules in vitro. To gain first evidence for such a binding activity in living cells, we attempted to localize Kin1 in vivo. A fusion of triple GFP and full-length Kin1 resulted in an even cytoplasmic background that did not allow for any subcellular localization (Figure 1D). However, when we expressed the YFP fused to Kin1 that was truncated for the motor domain (Kin1336-968), it partially colocalized with CFP-labeled microtubules. This was done in kin1-null mutants to avoid dimerization with endogenous Kin1 (strain ΔKin1YK1TCT; see Table 1 for details). Expression of the fusion protein led to strong accumulations at the cell poles (our unpublished data) and numerous faint dots in the cytoplasm. Quantitative analysis revealed that 70.2% of these YFP-Kin1T signals colocalized with microtubules (n = 11 cells, 47 signals; Figure 1E), and colocalization was mainly seen at microtubule bundles (Figure 1E, arrowheads), suggesting that the Kin1 tail is able to bind microtubules, most likely via a motor domain-independent microtubule binding site.
Figure 1.
A second microtubule binding site in conventional kinesin. (A) Kin1 is the conventional kinesin from U. maydis (Lehmler et al., 1997). It has an N-terminal motor core. The Kin1 tail is predicted to consist of a globular domain (G1), three coiled-coils (C1-C3), and a C-terminal globular domain (G2). Amino acids are given above the bar. (B) The C-terminal portion of conventional kinesin tails contains binding sites for light chains (Diefenbach et al., 1998) and organelles (Seiler et al., 2000) as well as a conserved motif that is implied in regulation of motor activity (Stock et al., 1999; Seiler et al., 2000). In addition, the C-terminal 196 amino acids of human kinesin heavy chain (HsKHC) localize to microtubules in vivo (Navone et al., 1992); region indicated by light blue bars). A microtubule binding activity was mapped to 55 amino acids between organelle binding and ATPase regulation sites (Hackney and Stock, 2000). Sequence similarity between human and fungal (UmKin1) kinesin is significantly higher within the functional regions. Note that fungal kinesins do not possess light chains. (C) Recombinant tail polypeptides from human kinesin-1 (HsC3G2) and U. maydis kinesin-1 (UmC3G2) cosedimented specifically with taxol-stabilized microtubules. Compare pellets after centrifugation with and without microtubules (+MT and -MT) from E. coli extracts (input). Note that microtubules were limiting in that experiment, thus similar amounts of both polypeptides were pulled down. (D) Expression of full-length Kin1 tagged with triple GFP resulted in an even cytoplasmic background. Bar, 5 μm. (E) A fusion protein of YFP and the Kin1 tail (amino acids 336-968) localized to microtubules (CFP-α-tubulin, red) in U. maydis Δkin1 cells. Note that colocalization occurs mainly within microtubule bundles (marked by arrowheads). Bars, 1 μm.
Conventional Kinesin in Microtubule Bending and Bundling
To analyze the role of Kin1 in cross-bridging and organizing microtubules in vivo, we deleted and overexpressed kin1 (Figure 2A). Absence of Kin1 had no obvious effects on the organization of GFP-labeled interphase microtubules (Figure 2B). However, electron microscopic analysis revealed that microtubules bundling was reduced in Δkin1 mutants. In wild-type cells, 25% of all sectioned microtubules (n = 77) formed bundles of up to three microtubules (Figure 2, C and D). In contrast, only 8% of all cross-sectioned microtubules were found to be bundled in Kin1-deficient cells (n = 73; Figure 2D), demonstrating that kinesin-1 promotes microtubule bundling in vivo. Growing interphase cells of U. maydis contain long microtubules that are nucleated at a microtubule organizing center near the constriction between mother and daughter cell (Straube et al., 2003). These microtubules form bundles that are predominantly (although not exclusively) parallel (Figure 2G). Occasionally, individual microtubules within such a bundle become bent (Steinberg et al., 2001). This bending is often due to sliding of microtubules along each other, which is best illustrated by speckle analysis (Figure 2E; GFP-Tub1 speckle in inverted image indicated by arrow; for details, see Materials and Methods). To determine the direction of microtubule sliding during these events, we generated strain FBPeb1R_GT that contains GFP-labeled microtubules and also expresses a fusion of Peb1, the EB1-homologue of U. maydis (Straube et al., 2003) with monomeric red fluorescent protein (Campbell et al., 2002) that labels growing microtubule plus-ends. A quantitative analysis of 74 bending events in >50 cells in this strain revealed that ∼45% of all bending motility seems to depend on microtubule-microtubule interaction. Of these bending events (that were set to 100%), 21% take place in unipolar microtubule bundles, whereas 9% occurred in antipolar microtubule bundles (examples of anti- and unipolar bundles are given in Figure 2G, plus-ends are indicated by arrows). The remaining events could not be clearly categorized. Plus-ends of looping microtubules indicated that most microtubule-microtubule bending events were either driven by minus-motors or involved both motor types. Only 4% of all bending motility seemed to be supported by a plus-end-directed kinesin (Figure 2F1, arrowhead marks a red fluorescent protein [RFP]-Peb1-labeled plus-end). Note that the RFP-Peb1 signal disappears before the bending event, suggesting that the microtubule stopped growth). Such bending events can be explained by the activity of a kinesin that cross-bridges two microtubules and stays stationary on one microtubule, whereas it moves toward the plus end of the other. This movement pushes the microtubule backward and results in bending of the polymer (Figure 2F2). In general, bending of microtubules occurred an average frequency of one event per 40 s (Figure 2H, control). Although the portion of kinesin-driven bending motility was relatively low, bending events in Δkin1 mutants were reduced to 60% compared with wild-type cells (Figure 2, H and I). In contrast, overexpression of Kin1 (Figure 2A) led to a twofold increase in bending frequency (Figure 2, H and K) and resulted in less ordered and curled microtubules (Figure 2B). Together, these data demonstrate that Kin1 is required for efficient microtubule bundling and promotes microtubule bending in vivo.
Figure 2.
Kin1 in microtubule bending and bundling. A, In kin1-null mutants no Kin1 was found in Western analysis, whereas expression of Kin1 under control of the strong crg-promoter (Bottin et al., 1996) led to ∼100-fold overexpression. Note that a 1:50 dilution was loaded for the overexpressing strain as seen in tubulin loading control panel. (B) Control cells contain microtubules that are partially bundled (control). Deletion of kin1 results in less bundling and shorter microtubules (ΔKin1). In contrast, overexpression of Kin1 led to less organized microtubules that were longer and formed more intensively stained bundles (Kin1↑). Bar, 5 μm. (C) Ultrastructural analysis of U. maydis wild-type cells shows long bundles of microtubules. Cross-sections reveal that bundles consist of up to three microtubules. Bars, 100 nm. (D) In electron microscopy (EM) images, 25% of all cross-sectioned microtubules were found in bundles of two (light gray) or three (dark gray) in control cells. Bundling was threefold reduced in the Kin1 deletion mutant (ΔKin1), but EM analysis failed to detect increased bundling in the Kin1-overexpressing strain (Kin1↑). Note that significantly more bundling was found during analysis of GFP-fluorescence in this strain (Figure 3F). (E1) Repressed expression of GFP-Tub1, which was integrated into the genome as an additional copy (strain FB2rGFPTub1) led to decreased microtubule labeling. Movement of speckles (arrow) that serve as structural landmarks illustrate that bending results from sliding of a microtubule over another microtubule. Bar, 2 μm. (F1) Image series showing bending of GFP-labeled microtubules in a strain that also expresses a fusion of Peb1, the EB1-homologue of U. maydis (Straube et al., 2003) with monomeric red fluorescent protein (Campbell et al., 2002). A microtubule slides over a second microtubule (arrows) and forms a loop. RFP-Peb1 (arrowhead) indicates that the microtubule slides with plus-end trailing. Elapsed time is given in seconds. Bar, 2 μm. (F2) The cartoon illustrates how a plus-end-directed kinesin molecule could accomplish such a bending event. The motor (red) cross-bridges two microtubules in a bundle. Although it walks toward the plus-end of one microtubule, this microtubule is pushed backward and bent. (G) Microtubule polarity within microtubule bundles is determined using Peb1-RFP marked growing microtubule plus-ends within microtubule bundles (green, GFP-tubulin). An example for a parallel and an antiparallel microtubule bundle is shown. Bar, 5 μm. (H) In control cells, microtubule bending motility was observed with a frequency of one event per 40 s. Deletion of Kin1 reduced this frequency to below one event per minute, whereas high levels of Kin1 doubled the bending activity. See supplemental movies (Videos 2-4). (I) Time-lapse images illustrate the reduced motility of the microtubule cytoskeleton in the absence of Kin1. The false-colored image merges two images that span a time interval of 18 s (T, 0 in red; T, 18 s in green; stationary microtubules in yellow). Elapsed time is given in seconds. Bar, 3 μm. (K) Time-lapse images of a Kin1-overexpressing cell reveal an enhance microtubule bending activity. Bending events are marked with arrowheads. The false-colored image merges two images that span a time interval of 18 s (T, 0 in red; T, 18 s in green; stationary microtubules in yellow). Elapsed time is given in seconds. Bar, 3 μm.
Rigor Mutations Demonstrate Microtubule Cross-linking Capacity of Kin1
In vitro data demonstrated that purified conventional kinesin is able to cross-bridge microtubules and slide microtubules along each other (Vale et al., 1985; Urrutia et al., 1991; Andrews et al., 1993). This suggests that microtubule bundling and bending in U. maydis might be caused directly by Kin1. Based on the results described above, we considered it possible that Kin1-heads walk along microtubules, whereas the putative C-terminal microtubule-binding site attaches to another microtubule, thereby promoting microtubule motility and bundling. To test this hypothesis in the living cell, we expressed a mutated Kin1 protein that contained a single amino acid exchange in the ATPase site (Figure 3A, G96E). This modification was shown to result in rigorous binding of the kinesin motor head to microtubules (Meluh and Rose, 1990; Wedlich-Soldner et al., 2002). Assuming that Kin1 moves microtubules along each other, we expected that this rigor-protein should tightly cross-link microtubules. Indeed, expression of Kin1rigor in strain rKin1GT led to thick microtubule bundles (Figure 3B) and strongly suppressed microtubule bending (Figure 3C). Kin1 is known to inhibit itself by switching into a folded conformation where the ATPase regulatory domain in the tail binds to the motor head (Coy et al., 1999; Stock et al., 1999). To circumvent this autoinhibition, we fused the Kin1 tail to an accordingly rigor-mutated motor head of Kin3, a Kif1A/Unc104-like kinesin involved in endosome motility (Wedlich-Soldner et al., 2002). Expression of the point-mutated Kin3rigor protein (Figure 3A) was without significant effect on microtubule bundling (Figure 3B) and bending (Figure 3C). In contrast, the fusion protein of the mutated Kin3 head and the Kin1 tail (Figure 3A, K3rigorK1T) led to heavy bundling and abolished microtubule bending almost completely (Figure 3C). These observations were confirmed by electron microscopy of cells expressing Kin1rigor (strain rKin1rigor; Table 1) and Kin3rigorKin1tail (strain rK3rigorK1T; Table 1). Both fusion proteins cross-linked microtubules and promoted the formation of bundles (Figure 3D) with up to nine microtubules per bundle. In cells expressing Kin1rigor, 69% (n = 195) of all microtubules were found in bundles, and this number increased to 86% (n = 111) in cells expressing Kin3rigorK1tail (Figure 3E). Fluorescence intensity measurements of GFP-labeled microtubules supported the electron microscopic data. Although a microtubule bundle in control cells contained on average three microtubules, high expression of Kin1 raised this number to four microtubules per bundle (Figure 3F). Introduction of the rigor mutation resulted in a further increase to five microtubules per bundle and bundles in Kin3rigorK1tail expressing strains contained more than seven microtubules on average (Figure 3F). The distances between microtubules in bundles were similar between microtubules in Kin1rigor (41.2 ± 19.4 nm, n = 44) and Kin3rigorK1tail-induced bundles (43.0 ± 12.1 nm; n = 47; Figure 3G). Although this distance corresponds well to the length of the kinesin-1 stalk (Amos, 1987; Hirokawa et al., 1989; Hisanaga et al., 1989; Scholey et al., 1989), it is remarkably different from that found in wild-type U. maydis cells (10.1 ± 3.8 nm; n = 16). However, microtubule bundles were rendered immobile in the rigor mutants (Figure 3C), which could allow the cross-bridging kinesin molecules to stretch out perpendicular to the bundle. In contrast, an active kinesin molecule exerting sideways forces in a sliding microtubule bundle is likely to bend at the various hinge points in the stalk, hence pulling the microtubules closer together.
Deletion in the Kin1 Tail Results in Loss of Cross-linking Activity
The ability of Kin1rigor and K3rigorK1T to cross-bridge microtubules requires two microtubule binding sites, and we supposed that the putative microtubule binding site in the Kin1 tail is required for this activity. To confirm this assumption, we constructed truncated K3rigor-K1T proteins that were deleted for the second coiled-coil (C2; Figure 4A) and the region that showed microtubule binding activity in vitro (C3G2; Figure 4A). Western analysis confirmed that both truncated proteins were expressed at similar levels (Figure 4B). Expression in strains that contain GFP-labeled microtubules demonstrated that the second coiled-coil (C2) was not the required for the ability to bundle microtubules (Figure 4, C and D; compare K3rigorK1T with K3rigorK1TΔC2; arrowheads mark bundles). Consequently, microtubule bending was still abolished when K3rigorK1TΔC2 was expressed (Figure 4E, compare K3rigorK1T with K3rigorK1TΔC2). In contrast, deletion of the third coiled-coil and the globular domain (ΔC3G2) resulted in loss of the bundling activity, led to normal microtubule arrays (Figure 4, C and D, compare control and K3rigorK1TΔC3G2), and almost normal microtubule bending (Figure 4, E and G, compare to the full-length chimera in 4F). These results demonstrate that the C-terminal region that covers the putative microtubule binding site is essential for the cross-linking activity of the mutant proteins in vivo, which adds further support to the notion that Kin1 is able to cross-bridge microtubules and to promote microtubule bending in living cells.
Figure 4.
The bundling activity in the Kin1 tail is located at its C terminus. (A) Constructs used to identify the region in the Kin1 tail that mediates heavy bundling and suppression of bending in K3rigorK1T. (B) Western analysis demonstrates comparable expression levels of the mutant proteins. (C) Deletion of the second coiled-coil (ΔC2) was without effect on microtubule bundling. However, deletion of the third coiled-coil alone or together with the C-terminal globular domain (ΔC3G2) eliminated the bundling activity of K3rigorK1T. Images for control and K3rigorK1T are included for comparison. To visualize single microtubules as well, images appear saturated where thick microtubule bundles occur. Asterisks mark the bud neck. Bars, 5 μm. (D) Fluorescence intensity measurement of GFP-labeled microtubule bundles in cells expressing Kin1 tail deletion constructs showed that the second coiled-coil domain is dispensable for the microtubule bundling activity in the Kin1 tail (Kin3rigorK1TΔC2). In contrast, deletion of the third coiled-coil and the C-terminal globular domain (ΔC3G2) from the Kin3rigorK1T chimera disabled microtubule bundling completely. (E) Deletion of C3 and G2 restored bending activity to the level of the Kin1 deletion mutant, whereas the mutant construct with deletion of C2 suppressed microtubule bending as effective as the full-length Kin3rigorK1T construct did. Data for control, ΔKin1 and K3rigorK1T are included for comparison. See supplemental movies (Videos 2, 6, and 7). (F) Time-lapse images illustrate the strongly reduced motility of the microtubule cytoskeleton in Kin3rigorK1T-expressing cells. The false-colored image merges two images that span a time interval of 18 s (T, 0 in red; T, 18 s in green; stationary microtubules in yellow). Elapsed time is given in seconds. Bar, 3 μm. (G) Time-lapse images illustrate the recovered motility of the microtubule cytoskeleton in cells expressing the deletion construct Kin3rigorK1TΔC3G2. The false-colored image merges two images that span a time interval of 18 s (T, 0 in red; T, 18 s in green; stationary microtubules in yellow). Elapsed time is given in seconds. Bar, 3 μm.
DISCUSSION
Conventional kinesin (=kinesin-1, Lawrence et al., 2004) is the founding member of the large superfamily of kinesins and was initially identified in neurons (Brady, 1985; Vale et al., 1985) and sea urchin eggs (Scholey et al., 1985). Since these early reports, a role of this motor in transport of membranous cargo was confirmed in numerous cell types (Bloom and Endow, 1995; Hirokawa, 1998). Consequently, kinesin-1 is the best-understood organelle transporter, which is found in amoeba (Klopfenstein et al., 2002), filamentous fungi (Steinberg and Schliwa, 1995; Lehmler et al., 1997; Schoch et al., 2003), and animal cells (Hirokawa, 1998). However, a few reports exist that also imply kinesin-1 in microtubule-microtubule interactions. Most strikingly, in vitro experiments demonstrated that purified kinesin-1 is able to bundle and slide microtubules along each other (Vale et al., 1985; Amos, 1987; Urrutia et al., 1991; Andrews et al., 1993). Such an activity requires a second microtubule binding site in kinesin-1 and it was reported that the kinesin tail is able to bind microtubules (Navone et al., 1992; Hackney and Stock, 2000). However, an in vivo function in microtubule organization could never be assigned for kinesin-1 family members. Here, we provide the first in vivo evidence for the involvement of the kinesin-1 tail in microtubule bundling. We took advantage of U. maydis, a well-suited eukaryotic model organism that combines numerous technical advantages with a relatively simple microtubule cytoskeleton, which contains bundles and shows microtubule sliding and bending in vivo (Steinberg et al., 2001). Furthermore, this fungus contains 10 kinesins (Schuchardt et al., 2005), including the conventional kinesin Kin1 (Lehmler et al., 1997), which makes it a well suited model to analyze the role of kinesin-1 in microtubule motility and organization. Indeed, we describe here that kin1 deletion mutants showed a threefold reduction in microtubule bundling, and live cell imaging revealed that overexpression of Kin1 significantly increases microtubule bending activity. A point mutation in the motor domain led to rigorous binding of the head to microtubules and resulted in heavy bundling of microtubules. Because the deletion of the C-terminal coiled-coil domain abolished this effect, we conclude that the tail of Kin1 is involved in microtubule bundling. This is surprising, because the tail of conventional kinesin binds to membranes (Skoufias et al., 1994). In fact, the putative organelle binding region (Seiler et al., 2000) overlaps with a conserved amino acid stretch that was shown to bind to microtubules in vitro (Hackney and Stock, 2000), suggesting that simultaneous binding to membranes and microtubules is prevented. It remains to be seen whether the overlap of binding sites might be relevant for the regulation of cargo loading and unloading.
Interaction of the kinesin tail with microtubules has to be tightly regulated in vivo to avoid kinesin sticking to microtubules. It has been reported that this microtubule binding site is masked when kinesin is in its folded, inactive conformation (Hackney and Stock, 2000). Accordingly, we observed a stronger effect on the microtubule cytoskeleton when the Kin1 tail was fused to another kinesin's head. This most probably prevents folding of the molecule that involves interaction of the IAK domain with the motor head (Hackney and Stock, 2000), and the head of Kin3 should not contain the respective binding site. In addition, it was reported that the light chains fulfill regulatory functions in animal kinesin-1 and help to keep kinesin in an inactive state (Verhey et al., 1998). Fungal kinesins do not possess light chains (Steinberg and Schliwa, 1995; Lehmler et al., 1997; Steinberg, 1997), but organelle binding, ATPase regulation, and microtubule binding activity seem to be conserved in the tail of kinesin (Seiler et al., 2000). It will be a future challenge to unravel the mechanism by which kinesins organelle and microtubule binding properties are regulated in the cell. We therefore expect that fungal model organisms will prove to be well suited to further investigate the regulation of kinesin heavy chain function.
What role does Kin1 have in microtubule organization? Self-organization of microtubules involves molecular motors that exert force to generate ordered microtubule arrays (Mitchison, 1992). Such motor activity underlies flagellar bending (Woolley, 2000), mitotic spindle assembly (Walczak et al., 1998), and most likely supports motility of interphase microtubules, observed in animals (Cassimeris et al., 1988; Tanaka and Kirschner, 1991; Baas, 2002), protozoa (Koonce et al., 1987; Koonce and Khodjakov, 2002), and the fungus U. maydis (Steinberg et al., 2001). Analysis of a deletion mutant showed that Kin1 is required for microtubule bundling and high levels of Kin1 resulted in increased microtubule bundling and bending, suggesting that Kin1 participates in self-organizing microtubule arrays in U. maydis by bundling microtubules. Moreover, we found that YFP-Kin1tail fusion proteins localize preferentially to microtubule bundles, which is consistent with a role in mediating microtubule-microtubule interactions. We consider it most likely that microtubule bundling is mediated directly by kinesin dimers that cross-bridge microtubules by binding one microtubule with their motor heads and a second via the microtubule binding sites in their tails. In vitro experiments have shown that stabilized microtubule cross-bridges contain only a single kinesin molecule rather than an aggregate of several kinesin molecules (Andrews et al., 1993). In this study, the distance between bundled microtubules observed in cells expressing rigor mutated kinesin-1 also corresponds well to the length of a straightened kinesin molecule, indicating that kinesin cross-bridges microtubules directly rather than multiple kinesins on a vesicle span the space between two microtubules. Moreover, electron microscopic observation did not reveal any vesicles between microtubule bundles (our unpublished data). The distance between bundled microtubules was shorter in wild-type cells than in the rigor mutants. This apparent discrepancy is most likely because of sideways forces generated by active kinesin within a motile microtubule bundle that does not allow the kinesin to stretch out perpendicular to the length of the microtubule. Thus, the microtubule spacing will seem reduced. In addition, other microtubule-associated proteins might be required to maintain the proper spacing of microtubules within a bundle, and the binding of Kin1rigor or Kin3rigorK1T along the length of microtubules excluded such proteins from the bundles. The high number of microtubules per bundle in cells expressing Kin1rigor or Kin3rigorK1T suggests that the overall number of microtubules per cell might have changed. However, we could not observe obvious changes in the tubulin content between different strains on Western blots (our unpublished data). Furthermore, cells never contained more than one thick bundle, which included most or all microtubules in that area. It was reported previously that wild-type cells of U. maydis contain up to seven microtubule tracks (Steinberg et al., 2001) and in this study we show that ∼25% of these represent bundles of up to three microtubules. Thus, control cells contain up to 10 microtubules, which correlates well to the maximal number of microtubules observed in a bundle in Kin3rigorK1T-expressing cells. Hence, we conclude that the number of microtubules per cell is not altered in the rigor mutants.
In U. maydis, microtubules undergo bending and are translocated within the cell (Steinberg et al., 2001). Although the bending motility is increased in cells that express high levels of Kin1, most bending activity persists in kin1 deletion mutants, indicating that other motors are involved in microtubule motility. Indeed, our quantitative analysis of bending motility in strain FB2Peb1R_GT reveals that only a small portion of all events could be explained by the activity of a plus-end-directed kinesin. A high portion of all bending was indicative of the activity of a minus-motor, whereas the rest could not be assigned or involved both activities at the same time. In animal cells, cytoplasmic dynein participates in microtubule transport (Baas, 2002). Consistently, in dynein mutants of U. maydis, a reduction in microtubule motility was observed (our unpublished data). Therefore, we speculate that other motors such as cytoplasmic dynein participate in arranging interphase microtubules in U. maydis, whereas Kin1-based motility mediates only a minority of all observed bending events. Surprisingly, in kin1 null mutants bending frequency dropped to ∼60%, suggesting that the absence of kinesin-1 affects the activity of other motors involved in bending. Presently, it is unknown how this is achieved. Moreover, it must be considered that other kinesin motors might be involved as well. Microtubule binding sites in the tail domain have been detected in other kinesins, including Ncd (Karabay and Walker, 1999) and Kid (Shiroguchi et al., 2003), and bipolar BimC-like kinesins cross-bridge microtubules via their antipolar-oriented motor heads (Kashina et al., 1997). The genome of U. maydis contains 10 kinesins, including orthologues of BimC and Ncd (Schuchardt et al., 2005), but most of them have no obvious phenotype. It will be challenge for the nearer future to further elucidate their role in self-organization of microtubules in U. maydis. The present study demonstrates a significant role of conventional kinesin, currently thought to be primarily, if not exclusively, an organelle motor, in the self-organization and dynamics of microtubule arrays in eukaryotic cells.
Supplementary Material
Acknowledgments
We thank Daniela Aβmann for expert technical assistance and Roland Wedlich-Söldner for help with raising the Kin3 antibody. We are much obliged to Thomas Surrey for purified pig-brain tubulin, to Werner Lutz for cDNA from human neuronal tissues, and to C. Horn and M. Schliwa for recombinant Kin1 protein. This work was supported by the Deutsche Forschungsgemeinschaft (SPP1111).
This article was published online ahead of print in MBC in Press (http://www.molbiolcell.org/cgi/doi/10.1091/mbc.E05-06-0542) on December 7, 2005.
The online version of this article contains supplemental material at MBC Online (http://www.molbiolcell.org).
References
- Adamikova, L., Straube, A., Schulz, I., and Steinberg, G. (2004). Calcium signaling is involved in dynein-dependent microtubule organization. Mol. Biol. Cell 15, 1969-1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allan, V. (1995). Membrane traffic motors. FEBS Lett 369, 101-106. [DOI] [PubMed] [Google Scholar]
- Amos, L. A. (1987). Kinesin from pig brain studied by electron microscopy. J. Cell Sci. 87, 105-111. [DOI] [PubMed] [Google Scholar]
- Andrews, S. B., Gallant, P. E., Leapman, R. D., Schnapp, B. J., and Reese, T. S. (1993). Single kinesin molecules crossbridge microtubules in vitro. Proc. Natl. Acad. Sci. USA 90, 6503-6507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baas, P. W. (2002). Microtubule transport in the axon. Int. Rev. Cytol. 212, 41-62. [DOI] [PubMed] [Google Scholar]
- Banuett, F., and Herskowitz, I. (1989). Different a alleles of Ustilago maydis are necessary for maintenance of filamentous growth but not for meiosis. Proc. Natl. Acad. Sci. USA 86, 5878-5882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloom, G. S., and Endow, S. A. (1995). Motor proteins 1, kinesins. Protein Profile 2, 1105-1171. [PubMed] [Google Scholar]
- Bottin, A., Kamper, J., and Kahmann, R. (1996). Isolation of a carbon source-regulated gene from Ustilago maydis. Mol. Gen. Genet. 253, 342-352. [DOI] [PubMed] [Google Scholar]
- Brady, S. T. (1985). A novel brain ATPase with properties expected for the fast axonal transport motor. Nature 317, 73-75. [DOI] [PubMed] [Google Scholar]
- Campbell, R. E., Tour, O., Palmer, A. E., Steinbach, P. A., Baird, G. S., Zacharias, D. A., and Tsien, R. Y. (2002). A monomeric red fluorescent protein. Proc. Natl. Acad. Sci. USA 99, 7877-7882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassimeris, L., Pryer, N. K., and Salmon, E. D. (1988). Real-time observations of microtubule dynamic instability in living cells. J. Cell Biol. 107, 2223-2231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coy, D. L., Hancock, W. O., Wagenbach, M., and Howard, J. (1999). Kinesin's tail domain is an inhibitory regulator of the motor domain. Nat. Cell Biol. 1, 288-292. [DOI] [PubMed] [Google Scholar]
- Diefenbach, R. J., Mackay, J. P., Armati, P. J., and Cunningham, A. L. (1998). The C-terminal region of the stalk domain of ubiquitous human kinesin heavy chain contains the binding site for kinesin light chain. Biochemistry 37, 16663-16670. [DOI] [PubMed] [Google Scholar]
- Friedman, D. S., and Vale, R. D. (1999). Single-molecule analysis of kinesin motility reveals regulation by the cargo-binding tail domain. Nat. Cell Biol. 1, 293-297. [DOI] [PubMed] [Google Scholar]
- Gindhart, J. G., Desai, C. J., Beushausen, S., Zinn, K., and Goldstein, L.S.B. (1998). Kinesin light chains are essential for axonal transport in Drosophila. J. Cell Biol. 141, 443-454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grummt, M., Pistor, S., Lottspeich, F., and Schliwa, M. (1998). Cloning and functional expression of a 'fast' fungal kinesin. FEBS Lett. 427, 79-84. [DOI] [PubMed] [Google Scholar]
- Hackney, D. D., and Stock, M. F. (2000). Kinesin's IAK tail domain inhibits initial microtubule-stimulated ADP release. Nat. Cell Biol. 2, 257-260. [DOI] [PubMed] [Google Scholar]
- Hirokawa, N. (1998). Kinesin and dynein superfamily proteins and the mechanism of organelle transport. Science 279, 519-526. [DOI] [PubMed] [Google Scholar]
- Hirokawa, N., Pfister, K. K., Yorifuji, H., Wagner, M. C., Brady, S. T., and Bloom, G. S. (1989). Submolecular domains of bovine brain kinesin identified by electron microscopy and monoclonal antibody decoration. Cell 56, 867-878. [DOI] [PubMed] [Google Scholar]
- Hisanaga, S., Murofushi, H., Okuhara, K., Sato, R., Masuda, Y., Sakai, H., and Hirokawa, N. (1989). The molecular structure of adrenal medulla kinesin. Cell Motil. Cytoskeleton 12, 264-272. [DOI] [PubMed] [Google Scholar]
- Karabay, A., and Walker, R. A. (1999). Identification of microtubule binding sites in the Ncd tail domain. Biochemistry 38, 1838-1849. [DOI] [PubMed] [Google Scholar]
- Kashina, A. S., Rogers, G. C., and Scholey, J. M. (1997). The bimC family of kinesins: essential bipolar mitotic motors driving centrosome separation. Biochim. Biophys. Acta 1357, 257-271. [DOI] [PubMed] [Google Scholar]
- Kirchner, J., Woehlke, G., and Schliwa, M. (1999). Universal and unique features of kinesin motors: insights from a comparison of fungal and animal conventional kinesins. Biol. Chem. 380, 915-921. [DOI] [PubMed] [Google Scholar]
- Klopfenstein, D. R., Holleran, E. A., and Vale, R. D. (2002). Kinesin motors and microtubule-based organelle transport in Dictyostelium discoideum. J. Muscle Res. Cell Motil. 23, 631-638. [DOI] [PubMed] [Google Scholar]
- Koonce, M. P., and Khodjakov, A. (2002). Dynamic microtubules in Dictyostelium. J. Muscle Res. Cell Motil. 23, 613-619. [DOI] [PubMed] [Google Scholar]
- Koonce, M. P., Tong, J., Euteneuer, U., and Schliwa, M. (1987). Active sliding between cytoplasmic microtubules. Nature 328, 737-739. [DOI] [PubMed] [Google Scholar]
- Lawrence, C. J., et al. (2004). A standardized kinesin nomenclature. J. Cell Biol. 167, 19-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehmler, C., Steinberg, G., Snetselaar, K. M., Schliwa, M., Kahmann, R., and Bolker, M. (1997). Identification of a motor protein required for filamentous growth in Ustilago maydis. EMBO J. 16, 3464-3473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meluh, P. B., and Rose, M. D. (1990). KAR3, a kinesin-related gene required for yeast nuclear fusion. Cell 60, 1029-1041. [DOI] [PubMed] [Google Scholar]
- Mitchison, T. J. (1992). Self-organization of polymer-motor systems in the cytoskeleton. Phil. Trans. R. Soc. Lond. B Biol. Sci. 336, 99-106. [DOI] [PubMed] [Google Scholar]
- Navone, F., Niclas, J., Hom-Booher, N., Sparks, L., Bernstein, H. D., McCaffrey, G., and Vale, R. D. (1992). Cloning and expression of a human kinesin heavy chain gene: interaction of the COOH-terminal domain with cytoplasmic microtubules in transfected CV-1 cells. J. Cell Biol. 117, 1263-1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahmann, A., Kamal, A., Roberts, E. A., and Goldstein, L. S. (1999). Defective kinesin heavy chain behaviour in mouse kinesin light chain mutants. J. Cell Biol. 146, 1277-1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoch, C. L., Aist, J. R., Yoder, O. C., and Gillian Turgeon, B. (2003). A complete inventory of fungal kinesins in representative filamentous ascomycetes. Fungal Genet. Biol. 39, 1-15. [DOI] [PubMed] [Google Scholar]
- Scholey, J. M., Heuser, J., Yang, J. T., and Goldstein, L.S.B. (1989). Identification of globular mechanochemical heads of kinesin. Nature 338, 355-357. [DOI] [PubMed] [Google Scholar]
- Scholey, J. M., Porter, M. E., Grissom, P. M., and McIntosh, J. R. (1985). Identification of kinesin in sea urchin eggs, and evidence for its localization in the mitotic spindle. Nature 318, 483-486. [DOI] [PubMed] [Google Scholar]
- Schuchardt, I., Aβmann, D., Thines, E., Schuberth, C., and Steinberg, G. (2005). Myosin-V, kinesin-1 and kinesin-3 cooperate in long-distance transport in hyphal growth of the fungus Ustilago maydis. Mol. Biol. Cell 16, 5191-5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seiler, S., Kirchner, J., Horn, C., Kallipolitou, A., Woehlke, G., and Schliwa, M. (2000). Cargo binding and regulatory sites in the tail of fungal conventional kinesin. Nat. Cell Biol. 2, 333-338. [DOI] [PubMed] [Google Scholar]
- Shiroguchi, K., Ohsugi, M., Edamatsu, M., Yamamoto, T., and Toyoshima, Y. Y. (2003). The second microtubule-binding site of monomeric kid enhances the microtubule affinity. J. Biol. Chem. 278, 22460-22465. [DOI] [PubMed] [Google Scholar]
- Skoufias, D. A., Cole, D. G., Wedaman, K. P., and Scholey, J. M. (1994). The carboxyl-terminal domain of kinesin heavy chain is important for membrane binding. J. Biol. Chem. 269, 1477-1485. [PubMed] [Google Scholar]
- Steinberg, G. (1997). A kinesin-like mechanoenzyme from the zygomycete Sycephalastrum racemosum shows biochemical similarities with conventional kinesin from Neurospora crassa. Eur. J. Cell Biol. 73, 124-131. [PubMed] [Google Scholar]
- Steinberg, G., and Schliwa, M. (1995). The Neurospora organelle motor: a distant relative of conventional kinesin with unconventional properties. Mol. Biol. Cell 6, 1605-1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg, G., and Schliwa, M. (1996). Characterization of the biophysical and motility properties of kinesin from the fungus Neurospora crassa. J. Biol. Chem. 271, 7516-7521. [DOI] [PubMed] [Google Scholar]
- Steinberg, G., Schliwa, M., Lehmler, C., Bolker, M., Kahmann, R., and McIntosh, J. R. (1998). Kinesin from the plant pathogenic fungus Ustilago maydis is involved in vacuole formation and cytoplasmic migration. J. Cell Sci. 111, 2235-2246. [DOI] [PubMed] [Google Scholar]
- Steinberg, G., Wedlich-Soldner, R., Brill, M., and Schulz, I. (2001). Microtubules in the fungal pathogen Ustilago maydis are highly dynamic and determine cell polarity. J. Cell Sci. 114, 609-622. [DOI] [PubMed] [Google Scholar]
- Stock, M. F., Guerrero, J., Cobb, B., Eggers, C. T., Huang, T. G., Li, X., and Hackney, D. D. (1999). Formation of the compact confomer of kinesin requires a COOH-terminal heavy chain domain and inhibits microtubule-stimulated ATPase activity. J. Biol. Chem. 274, 14617-14623. [DOI] [PubMed] [Google Scholar]
- Straube, A., Brill, M., Oakley, B. R., Horio, T., and Steinberg, G. (2003). Microtubule organization requires cell cycle-dependent nucleation at dispersed cytoplasmic sites: polar and perinuclear microtubule organizing centers in the plant pathogen Ustilago maydis. Mol. Biol. Cell 14, 642-657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straube, A., Enard, W., Berner, A., Wedlich-Soldner, R., Kahmann, R., and Steinberg, G. (2001). A split motor domain in a cytoplasmic dynein. EMBO J. 20, 5091-5100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka, E. M., and Kirschner, M. W. (1991). Microtubule behavior in the growth cones of living neurons during axon elongation. J. Cell Biol. 115, 345-363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urrutia, R., McNiven, M. A., Albanesi, J. P., Murphy, D. B., and Kachar, B. (1991). Purified kinesin promotes vesicle motility and induces active sliding between microtubules in vitro. Proc. Natl. Acad. Sci. USA 88, 6701-6705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vale, R. D., Reese, T. S., and Sheetz, M. P. (1985). Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell 42, 39-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verhey, K. J., Lizotte, D. L., Abramson, T., Barenboim, L., Schnapp, B. J., and Rapoport, T. A. (1998). Light chain-dependent regulation of kinesin's interaction with microtubules. J. Cell Biol. 143, 1053-1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walczak, C. E., Vernos, I., Mitchison, T. J., Karsenti, E., and Heald, R. (1998). A model for the proposed roles of different microtubule-based motor proteins in establishing spindle bipolarity. Curr. Biol. 8, 903-913. [DOI] [PubMed] [Google Scholar]
- Wedlich-Soldner, R., Straube, A., Friedrich, M. W., and Steinberg, G. (2002). A balance of KIF1A-like kinesin and dynein organizes early endosomes in the fungus Ustilago maydis. EMBO J. 21, 2946-2957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolley, D. (2000). The molecular motors of cilia and eukaryotic flagella. Essays Biochem. 35, 103-115. [DOI] [PubMed] [Google Scholar]
- Wu, Q., Sandrock, T. M., Turgeon, B. G., Yoder, O. C., Wirsel, S. G., and Aist, J. R. (1998). A fungal kinesin required for organelle motility, hyphal growth, and morphogenesis. Mol. Biol. Cell 9, 89-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, J. T., Laymon, R. A., and Goldstein, L. S. (1989). A three-domain structure of kinesin heavy chain revealed by DNA sequence and microtubule binding analyses. Cell 56, 879-889. [DOI] [PubMed] [Google Scholar]
- Yang, J. T., Saxton, W. M., Stewart, R. J., Raff, E. C., and Goldstein, L.S.B. (1990). Evidence that the head of kinesin is sufficient for force generation and motility in vitro. Science 249, 42-47. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.