Abstract
To increase the frequency of homologous recombination, we inactivated the KU80 homologue in Aspergillus fumigatus (named akuBKU80). Homologous integration reached about 80% for both calcineurin A (calA) and polyketide synthase pksP (alb1) genes in the akuBKU80 mutant to 3 and 5%, respectively, when using a wild-type A. fumigatus strain. Deletion of akuBKU80 had no influence on pathogenicity in a low-dose murine infection model.
Double-strand breaks are the most severe form of DNA damage. Eukaryotes have two main pathways to deal with this type of DNA damage: (i) homologous recombination, involving interaction between homologous sequences, and (ii) nonhomologous end joining, involving direct ligation of the strand ends independently of DNA homology (17). The most important double-strand break repair pathway in Saccharomyces cerevisiae is homologous recombination (18, 20), while other organisms, such as humans, preferentially use nonhomologous end joining. The nonhomologous end-joining process is mediated by the DNA-dependent protein kinase catalytic subunit, the Ku70-Ku80 heterodimer, and the DNA ligase IV-Xrcc4 complex (17). Recently, Ninomiya et al. (13) disrupted Neurospora crassa genes homologous to human KU70 and KU80. Transformation of the N. crassa KU disruption strains yielded 100% transformants exhibiting integration at the homologous site, compared to 10 to 30% for a wild-type recipient.
Aspergillus fumigatus causes life-threatening pulmonary disease in severely immunocompromised patients (3). The completion of the A. fumigatus genome sequence has offered an opportunity to investigate complete pathways and classes of genes and do high-throughput gene tagging (12). New methodologies for rapid construction of gene replacements in Aspergillus have been proposed (21, 22). The rate-limiting step for further progress in this area is the percentage of homologous integration in A. fumigatus, which in some cases can be below 5%. Thus, we decided to delete the A. fumigatus KU80 gene and verify whether, as in N. crassa (13), the absence of nonhomologous end joining could increase the integration of introduced exogenous DNA fragments by homologous recombination.
To evaluate whether the deletion of these genes could increase homologous recombination, we selected calA and pksP (alb1), which encode the calcineurin A catalytic subunit and a polyketide synthase, respectively. Calcineurin is a serine-threonine-specific phosphatase heterodimer consisting of a catalytic subunit A and a Ca2+-binding regulatory B subunit. The association of the two subunits is essential for activity (7). The polyketide synthase is involved in the melanin biosynthesis required for conidial pigmentation (9, 19), so that the conidia of the ΔpksP mutant obtained by gene replacement will be white instead of green like the parental strain (11).
A. fumigatus strain CEA17 (pyrG) (2) was used throughout this work. The media used were YAG containing 2% (wt/vol) glucose, 0.5% (wt/vol) yeast extract, 2% (wt/vol) agar, and trace elements; YUU (YAG medium supplemented with 1.2 g/liter each of uracil and uridine), YG medium (same composition, but without agar) or minimal medium (MM: 1% glucose, nitrate salts, trace elements, 2% agar, pH 6.5). Trace elements, vitamins, and nitrate salts are described in the appendix to Kafer (5). DNA manipulations were according to Sambrook et al. (15). DNA fragment probes for Southern blots were labeled with [α-32P]dCTP using the RTS Rad Prime DNA labeling system kit (Gibco-BRL).
PCR primers were designed for amplifying each DNA fragment necessary for the PCR-mediated technique (8). In all the deletion constructs, the A. fumigatus pyrG gene was amplified from the pCDA21 plasmid (1) and is referred to as the zeo-pyrG cassette because the amplified fragment also contains the zeocin resistance gene. For DNA fragments containing the gene-flanking regions, genomic DNA was used as the template. For a description of the oligonucleotides used in this work and the strategy for PCR-mediated amplification, see Table 1.
TABLE 1.
List of primers used in this worka
| Primer | Sequence | PCR conditions |
|---|---|---|
| KU80-1 | 5′-CTGACTGATTCGACCGAG-3′ | (i) 5′-Flanking region about 2,000 bp (KU80-1 × KU80-2) |
| KU80-2 | 5′-CTGAGAATTCCGATCAAGATGCTCTAGAATA-3′ | (ii) zeo-pyr cassette (KU80-A × KU80-B) |
| KU80-3 | 5′-TGAGGCGAATTCAGTCGCTCCTCTAAATGG-3′ | (iii) 3′-Flanking region 1,997 bp (KU80-3 × KU80-4) |
| KU80-4 | 5′-ATGAAACTCTTGAACCCGA-3′ | (iv) Fusion PCR fragment about 6.4 kb (KU80-1 × KU80-4) |
| KU80-A | 5′-GAGCATCTTGATCGGAATTCTCAGTCCTGCT-3′ | |
| KU80-B | 5′-AGAGGAGCGACTGAATTCGCCTCAAACAAT-3′ | |
| Calcineurin1 | 5′-GGTTCATTGGGACCAGACC-3′ | (i) 5′-Flanking region about 2,000 bp (calcineurin1 × calcineurin2) |
| Calcineurin2 | 5′-CTGAGAATTCCCTTGCGCAGTGTGAACTAAG-3′ | (ii) zeo-pyrG cassette (calcin-ZeoF × calcin-PyrR) |
| Calcineurin3 | 5′-TGAGGCGAATTCAATTGCTTCGGTCACGTCCA-3′ | (iii) 3′-Flanking region about 2,000 bp (calcineurin3 × calcineurin4) |
| Calcineurin4 | 5′-GCGAACTCTGAAGCTCGGT-3′ | (iv) Fusion PCR fragment about 6.4 kb (calcineurin1 × calcineurin4) |
| Calcin-ZeoF | 5′-CACACTGCGCAAGGGAATTCTCAGTCCTGCTC-3′ | |
| Calcin-PyrR | 5′-GACCGAAGCAATTGAATTCGCCTCAAACAATGC-3′ | |
| ALB-1 | 5′-AAGCCAAGAAGCCTTACTG-3′ | (i) 5′-Flanking region about 2,000 bp (ALB-1 × ALB-2) |
| ALB-2 | 5′-CTGGAATTCCTGACTGCAAGGAGTAGTG-3′ | (ii) zeo-pyrG cassette (ALB-A × ALB-B) |
| ALB-3 | 5′-AAGAGATGGAGGACGAC-3′ | (iii) 3′-Flanking region about 2,000 bp (ALB-3 × ALB-4) |
| ALB-4 | 5′-TGAGGCGAATTCTTGTGCATACCTAAATTGG-3′ | (iv) Fusion PCR fragment about 6.4 kb (ALB-1 × ALB-4). |
| ALB-A | 5′-CTCCTTGCAGTCAGGAATTCTCAGTCCTGCT-3′ | |
| ALB-B | 5′-AGGTATGCACAAAATTCGCCTCAAACAAT-3′ |
The 50-μl amplification mixture included 1× Platinum Taq DNA polymerase high-fidelity buffer (Invitrogen), 20 pmol of each primer, 0.4 mM deoxynucleotide triphosphate mix, 1.0 U of Taq DNA HiFi platinum polymerase (Invitrogen), and 500 ng of genomic DNA or 100 ng plasmid. PCR amplification was carried out in a PTC100 96-well thermal cycler (MJ Research) at 94°C for 2 minutes and 30 cycles of 94°C for 1 minute, 52 to 60°C (depending on the fragment) for 1 minute, and 68°C for 7 minutes, followed by an extension step at 68°C for 7 minutes. After the reaction, the PCR products were purified with a Qiagen PCR cleanup kit according to the manufacturer's instructions.
The KU80 gene (here named akuBKU80; Afu2g02620, accession number EAL87359) was identified in the A. fumigatus genome (http://www.tigr.org/tdb/e2k1/afu1/) by similarity to N. crassa mus-52 (42% identity, 60% similarity, e-value = 7e-117). Routinely, in PCR-mediated deletions for A. fumigatus, flanking regions of about 1.5 to 2.0 kb were used. This size increased the frequency of homologous integration (data not shown). Flanking sequences shorter than 500 bp did not yield any homologous integration (data not shown).
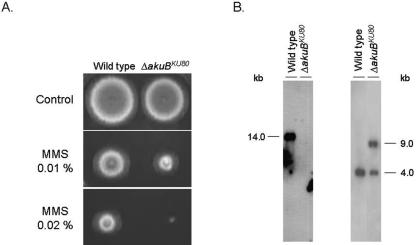
Transformation of A. fumigatus strain CEA17 (mutant pyrG recipient strain) was according to the procedure of Osmani et al. (14) using 5 μg of linear DNA fragments. A. fumigatus was transformed with the PCR-mediated deletion cassette for the akuBKU80 gene. Several transformants were obtained and identified by their ability to grow in YAG. These transformants were screened for sensitivity to hydroxyurea, camptothecin, 4-nitroquinoline oxide, and methane methyl sulfonate (MMS). Only one transformant in 100 displayed decreased growth in the presence of MMS (a frequency of 1%) (Fig. 1A). The N. crassa mus-52 mutant also showed mild sensitivity to MMS (13). The pyrG+ strain KU80Δ was point inoculated on YUU plates containing 5-fluoroorotic acid in a subinhibitory concentration of 0.55 mg/ml, resulting in the recovery of a sector that was auxotrophic to uridine and uracil and resistant to up to 0.75 mg/ml of 5-fluoroorotic acid. This strain was named KU80ΔpyrG. The allelic replacement of the akuBKU80 gene in this transformant (KU80ΔpyrG strain) and the presence of a single integration event were confirmed by Southern blot analysis (Fig. 1B).
FIG. 1.
ΔakuBKU80 strain displays increased sensitivity to MMS. (A) Growth phenotype of the A. fumigatus wild-type and KU80ΔpyrG strains grown for 48 h at 37°C in YUU medium in the presence or absence of different concentrations of MMS. (B) Southern blot analysis of the KU80ΔpyrG strain. At the left panel, genomic DNA from the wild-type and KU80ΔpyrG strains was isolated and digested with EcoRI and the entire akuBKU80 open reading frame was used as a hybridization probe. The akuBKU80 gene probe recognized a single band of approximately 14.0 kb only in the wild-type strain, indicating the akuBKU80 gene was deleted in the KU80ΔpyrG strain. In the right panel, genomic DNA from the wild-type and KU80ΔpyrG strains was isolated and cleaved with the enzyme KpnI and the pyrG gene was used as a hybridization probe. The pyrG gene recognized one band in the wild-type strain (approximately 4.0 kb) and two bands in the akuBKU80 deletion strain (around 4.0 and 9.0 kb). This additional band in the KU80ΔpyrG strain indicates a single event of integration of the transforming deletion cassette.
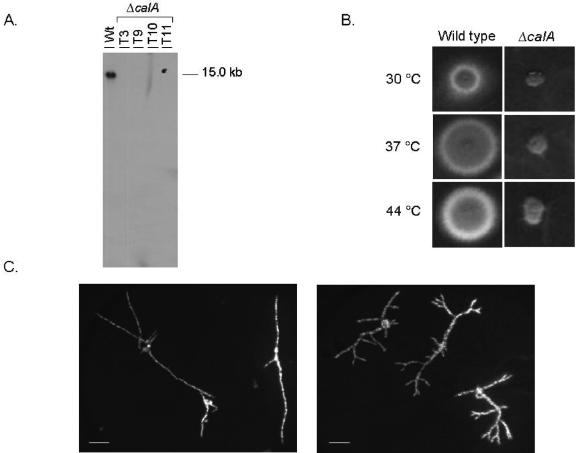
Next, the effects of deleting the akuBKU80 gene on A. fumigatus homologous recombination integration of the calA and pksP genes were investigated. Therefore, the A. fumigatus calA and pksP deletion cassettes were amplified by using PCR-mediated deletion (Table 1). One hundred transformants were obtained and 15 of them were evaluated by Southern blotting for the absence of the calA gene; 80% of them showed homologous integration of the deletion cassette at the calA locus and a single event of integration, as shown by four representative transformants in Fig. 2A. The transformants that displayed nonhomologous integration at the calA locus were also not integrated at the pyrG locus (data not shown), suggesting that there are other mechanisms of nonhomologous recombination in A. fumigatus besides nonhomologous end joining. When the wild-type strain was transformed with the calA deletion cassette, the frequency of homologous integration was 3%.
FIG. 2.
Calcineurin A deletion mutant (ΔcalA) showed increased branching and a phenotype of reduced growth. (A) Southern blot analysis of the ΔcalA strain. Genomic DNA from wild-type and four ΔcalA strains (T3, T9, T10, and T11) was isolated and digested with SfiI and the entire calA open reading frame was used as a hybridization probe. The calA gene probe recognized a single band of approximately 15.0 kb only in the wild-type strain, indicating the calA gene was deleted in all four transformants. (B) Growth phenotype of the A. fumigatus wild-type (left panel) and ΔcalA (right panel) strains grown for 48 h at 30, 37, and 44°C in YUU medium. (C) Conidia of the wild-type and ΔcalA strains were inoculated onto coverslips in YG+UU medium. After 10 h incubation at 37°C, coverslips with adherent germlings were transferred to fixative solution (3.7% formaldehyde, 50 mM sodium phosphate buffer, pH 7.0, 0.2% Triton X-100) for 30 min at room temperature. Then, they were briefly rinsed with PBS buffer (140 mM NaCl, 2 mM KCl, 10 mM NaHPO4, 1.8 mM KH2PO4, pH 7.4) and incubated for 5 min in a solution with 100 ng/ml of DAPI (4′,6′-diamino-2-phenylindole, Sigma Chemical Co.). After incubation with the dyes, they were washed with phosphate-buffered saline for 5 min at room temperature and then rinsed in distilled water and mounted on the slides. The material was photographed using a Zeiss epifluorescence microscope. The number of nuclei was assessed by DAPI staining. Graph bars indicate 10 μm.
The primary transformants already displayed a pronounced phenotypic defect, i.e., small sporulating colonies that, when purified, showed comparable growth at different temperatures (Fig. 2B). The morphology of the ΔcalA germlings showed increased branching compared to the corresponding wild-type strain (Fig. 2C). Calcineurin A deletion was also found to affect colony morphology in several media known to modulate the C. albicans dimorphic switch (16). These results suggest that the calA gene is not essential in A. fumigatus, but its deletion confers a severe growth defect that is probably due to abnormal branching.
To further confirm homologous integration in a high frequency in the ΔakuBKU80 strain, the pksP deletion cassette was introduced into strain KU80ΔpyrG. Transformants were checked for the percentage with white spores. Flanking regions of about 0.25, 0.5, 1.5, and 2.0 kb were used. Flanking sequences shorter than 500 bp did not yield any homologous integration in either the wild-type or akuB deletion strains (about 30 transformants each were observed for flanking regions of 0.25 and 0.5 kb). However, flanking regions of 1.5 to 2.0 kb yielded about 80 and 5% transformants that produced white spores in the akuB deletion and wild-type strains, respectively (about 30 transformants each were observed for flanking regions of 1.5 and 2.0 kb). Taken together, these results suggest that deletion of the akuBKU80 gene in A. fumigatus provides a much higher frequency of homologous recombination.
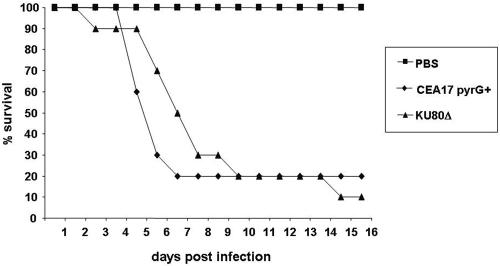
To determine a possible influence on pathogenicity resulting from deletion of the akuBKU80 gene, the corresponding deletion mutant was tested in an animal model. The virulence of A. fumigatus strain KU80Δ in comparison to strain CEA17pyrG+, derived from its parental strain CEA17(pyrG) by reversion to uracil prototrophy, which was used as the wild type, was determined in a murine low-dose model for invasive aspergillosis as optimized by Liebmann et al. (10). The results of a representative experiment are shown in Fig. 3. They reveal no differences in virulence between the wild-type and the akuBKU80 deletion strain. This is true for both the overall mortality and the curve shape representing the time course of survival after infection. Infection with either the wild-type or the akuBKU80 deletion strain resulted in a mortality rate of 80% 10 days after inoculation of the conidia. These results indicate that the akuBKU80 mutation is not causing any loss of virulence in the KU80Δ strain compared to the corresponding wild-type strain.
FIG. 3.
Virulence of A. fumigatus KU80Δ mutant strain. Survival of mice infected intranasally with conidia of A. fumigatus CEA17pyrG+ and KU80Δ was determined. As shown by d'Enfert et al. (3) uracil auxotrophic A. fumigatus strains show attenuated virulence. Therefore, the uracil prototroph strain CEA17pyrG+ was isolated from strain CEA17 by spontaneous reversion of the point mutation in the pyrG locus. CEA17pyrG+ shows the same virulence in comparison to the A. fumigatus wild-type strain ATCC 46645 (data not shown). For the same reason the uracil prototrophic strain KU80Δ was used instead of KU80ΔpyrG. In brief, female BALB/c mice (Harlan Winkelmann, Borchen, Germany) of 18 to 20 g body weight were immunosuppressed by intraperitoneal injection of cyclophosphamide (100 mg/ml; Sigma, Taufkirchen, Germany) on days −4, −1, 2, 5, 8, 11, and 14 prior to and after infection on day 0. A single dose of cortisone acetate (200 mg/kg of body weight; Sigma) was injected subcutaneously on day −1. Suspensions of A. fumigatus conidia were harvested with phosphate-buffered saline containing 0.1% (vol/vol) Tween 80 (Merck) and filtered through a 40-μm nylon cell strainer (BD Biosciences Europe, Belgium). Mice were anesthetized by intraperitoneal injection of 200 μl of 1% (vol/vol) ketamine (Velonarcon, Berlin Chemie, Germany) and 0.02% (vol/vol) xylazine (Rompun, Bayer Leverkusen, Germany) and intranasally infected with a 25-μl drop of a fresh suspension containing 2 × 104 conidia. Survival was monitored daily, and moribund animals were sacrificed by intraperitoneal injection of 200 μl of 3.2% (vol/vol) narcoren (Rhone Merieux, Laupheim, Germany). The drinking water was supplemented with 0.5 mg of tetracycline (Sigma) per ml to prevent opportunistic bacterial infections. Mice were tested in cohorts of 10 animals. A control group (inhalation of phosphate-buffered saline [PBS]) remained uninfected to monitor the influence of the immunosuppression procedure on vitality. Survival was monitored for 16 days. Data are representative of several independent experiments.
Taken together, our results strongly indicate that the akuBKU80 deletion strain could be a powerful tool for high-throughput gene replacement in A. fumigatus. Once the target gene has been deleted, the wild-type akuB gene might be restored by complementation studies to allow analysis of the deletion strain in an otherwise wild-type background. The recognition of the complementation of the akuBKU80 deletion mutation might be simple considering that the complemented strain will be resistant to MMS. Moreover, we have used a homologous auxotrophic marker pyrG gene for our transformation studies, but it is also possible to use dominant markers such as hygromycin resistance, as shown in an A. fumigatus akuAKU70 deletion mutation background (6).
Acknowledgments
This research was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil, and the Deutsche Forschungsgemeinschaft (Priority Program 1160).
We thank Ursula Stöckel and Birgit Weber for excellent technical assistance.
REFERENCES
- 1.Chaveroche, M.-K., J.-M. Ghigo, and C. D'Enfert. 2000. A rapid method for efficient gene replacement in the filamentous fungus Aspergillus nidulans. Nucleic Acids Res. 28:e97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.D'Enfert, C. 1996. Selection of multiple disruption events in Aspergillus fumigatus using the orotidine-5′-decarboxylase gene, pyrG, as a unique transformation marker. Curr. Genet. 30:76-82. [DOI] [PubMed] [Google Scholar]
- 3.D'Enfert, C., M. Diaquin, A. Delit, N. Wuscher, J. P. Debeaupuis, M. Huerre, and J. P. Latge. 1996. Attenuated virulence of uridine-uracil auxotrophs of Aspergillus fumigatus. Infect. Immun. 64:4401-4405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Denning, D. W. 1996. Diagnosis and management of invasive aspergillosis. Curr. Clin. Top. Infect. Dis. 16:277-299. [PubMed] [Google Scholar]
- 5.Kafer, E. 1977. Meiotic and mitotic recombination in Aspergilllus and its chromosomal aberrations. Adv. Genet. 19:33-131. [DOI] [PubMed] [Google Scholar]
- 6.Krappmann, S., C. Sasse, and G. H. Braus. 2006. Gene targeting in Aspergillus fumigatus by homologous recombination is facilitated in a nonhomologous end-joining-deficient genetic background. Eukaryot. Cell 5:223-226, this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kraus, P. R., and J. Heitman. 2003. Coping with stress: calmodulin and calcineurin in model and pathogenic fungi. Biochem. Biophys. Res. Commun. 311:1151-1157. [DOI] [PubMed] [Google Scholar]
- 8.Kuwayama, H., S. Obara, T. Morio, M. Katoh, H. Urushihara, and Y. Tanaka. 2002. PCR-mediated generation of a gene disruption construct without the use of DNA ligase and plasmid vectors. Nucleic Acids Res. 30:e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Langfelder, K., B. Jahn, H. Gehringer, A. Schmidt, G. Wanner, and A. A. Brakhage. 1998. Identification of a polyketide synthase gene (pksP) of Aspergillus fumigatus involved in conidial pigment biosynthesis and virulence. Med. Microbiol. Immunol. 187:72-89. [DOI] [PubMed] [Google Scholar]
- 10.Liebmann, B., T. W. Muhleisen, M. Muller, M. Hecht, G. Weidner, A. Braun, M. Brock, and A. A. Brakhage. 2004. Deletion of the Aspergillus fumigatus lysine biosynthesis gene lysF encoding homoaconitase leads to attenuated virulence in a low-dose mouse infection model of invasive aspergillosis. Arch. Microbiol. 181:378-383. [DOI] [PubMed] [Google Scholar]
- 11.Mouyna, I., C. Henry, T. L. Doering, and J.-P. Latgé. 2004. Gene silencing with RNA interference in the human pathogenic fungus Aspergillus fumigatus. FEMS Microbiol. Lett. 237:317-324. [DOI] [PubMed] [Google Scholar]
- 12.Nierman, W., A. Pain, M. J. Anderson, J. Wortman, H. S. Kim, J. Arroya, M. Berriman, K. Abe, D. B. Archer, C. Bermejo, J. Bennett, P. Bowyer, D. Chen, M. Collins, R. Coulsen, R. Davies, P. S. Dyer, M. Farman, N. Fedorova, T. V. Feldblyum, R. Fischer, N. Fosker, A. Fraser, J. L. Garcia., M. J. Garcia, A. Goble, G. H. Goldman, K. Gomi, S. Griffith-Jones, R. Gwilliam, B. Haas, H. Haas, D. Harris, H. Horiuchi, J. Huang, S. Humphrey, J. Jimenez, J., N. Keller, H. Khouri, K. Kitamoto, T. Kobayashi, R. Kulkarni, T. Kumagai, A. Lafton, J.-P. Latgé, W. Li, A. Lord, A., C. Lu, W. H. Majoros, G. S. May, B. L. Miller, Y. Mohamoud, M. Molina, M. Monod, I. Mouyna, S. Mulligan, L. Murphy, S. O'Neil, I. Paulsen, M. A. Penalva, M. Pertea, C. Price, B. L. Pritchard, M. A. Quail, E. Rabbinowitsch, N. Rawlins, M.-A. Rajandream, U. Reichard, H. Renauld, G. D. Robson, S. Rodriguez de Cordoba, J. M. Rodriguez-Pena, J. M. Ronning, C. M. Rutter, S. S. L. Salzberg, M. Sanchez, J. C. Sanchez-Ferrero, D. Saunders, K. Seeger, R. Squares, S. Squares, M. Takeuchi, F. Tekaia, G. Turner, C. R. Vazquez de Aldana, J. Weidman, O. White, J. Woodward,J., J.-H. Yu, C. Fraser, J. E. Galagan, K. Asai, M. Machida, N. Hall, B. Barrell, and D. W. Denning. Genomic sequence of the pathogenic and allergenic filamentous fungus Aspergillus fumigatus. Nature, in press. [DOI] [PubMed]
- 13.Ninomiya, Y., K. Suzuki, C. Ishii, and H. Inoue. 2004. Highly efficient gene replacements in Neurospora strains deficient for nonhomologous end joining. Proc. Natl. Acad. Sci. USA 101:12248-12253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Osmani, S. A., G. S. May, and N. R. Morris. 1987. Regulation of the mRNA levels of nimA, a gene required for the G2-M transition in Aspergillus nidulans. J. Cell Biol. 104:1495-1504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
- 16.Sanglard, D., F. Ischer, O. Marchetti, J. Enenza, and J. Bille. 2003. Calcineurin A of Candida albicans: involvement in antifungal tolerance, cell morphogenesis and virulence. Mol. Microbiol. 48:959-976. [DOI] [PubMed] [Google Scholar]
- 17.Tachibana, A. 2004. Genetic and physiological regulation of non-homologous end-joining in mammalian cells. Adv. Biophys. 38:21-44. [PubMed] [Google Scholar]
- 18.Takita, Y., M. Takahara, S. Nogami, Y. Anraku, and Y. Oya. 1997. Applications of the long and accurate polymerase chain reaction method in yeast molecular biology: direct sequencing of the amplified DNA and its introduction into yeast. Yeast 13:763-768. [DOI] [PubMed] [Google Scholar]
- 19.Tsai, H. F., Y. C. Chang, R. G. Washburn, M. H. Wheeler, and K. J. Kwon-Chung. 1998. The developmentally regulated alb1 gene of Aspergillus fumigatus: its role in modulation of conidial morphology and virulence. J. Bacteriol. 180:3031-3038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wach, A., A. Brachat, R. Pohlmann, and P. Philippsen. 1994. New heterologous modules for classical or PCR-based gene disruptions in Saccharomyces cerevisiae. Yeast 10:1793-1808. [DOI] [PubMed] [Google Scholar]
- 21.Yang, L., L. Ukil, A. Osmani, F. Nahm, J. Davies, C. P. C. de Souza, X. Dou, A. Perez-Balaguer, and S. A. Osmani. 2004. Rapid production of gene replacement constructs and generation of a green fluorescent protein-tagged centromeric marker in Aspergillus nidulans. Eukaryot. Cell 3:1359-1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yu, J.-H., Z. Hamari, K.-H. Han, J.-A. Seo, Y. Reyes-Dominguez, and C. Scazzocchio. 2004. Double-joint PCR: a PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet. Biol. 41:973-981. [DOI] [PubMed] [Google Scholar]