Abstract
Our group and others have shown in vitro that repeated exposure of human mononuclear cells (MNC) to lipopolysaccharide can induce endotoxin tolerance, evidenced by downregulation of TLR2 and TLR4 mRNA and surface protein; moreover, the ability of the MNC to secrete inflammatory cytokines is reduced. In situ studies performed on diseased and healthy gingiva suggest that a similar pattern of endotoxin tolerance occurs in human oral mucosa with chronic periodontitis (CP). We hypothesized that this represents a fundamental immunoregulatory mechanism to restore immune homeostasis and protect the host from further tissue damage. In the current study, we extend these published studies by providing evidence that Src homology 2 containing inositol phosphatase, an inhibitor of NF-κB activation and a negative regulator of the immune response, is upregulated in the oral mucosa during CP compared to its level during gingival health. We have also isolated MNC from patients with CP and those with healthy gingiva and show that MNC from CP subjects have a reduced capacity to upregulate TLR2, TLR4, and interleukin-1β in response to endotoxin. Thus, we provide more definitive evidence for a basic mechanism of immunoregulation in the oral mucosa.
The ability to discriminate harmful microbes from commensal species is probably the most important property of the mucosal immune system, essential for maintaining a healthy host. Mucosal diseases, where the host inappropriately responds to commensal gut flora and to food antigens, are exemplified by Crohn's disease (1) and celiac disease (12), respectively. The Toll-like receptors (TLRs) are pattern recognition receptors that can rapidly identify microbial structures (e.g., peptidoglycan and lipopolysaccharide [LPS]) (2, 26, 30) and provoke an appropriate adaptive immune response (19). Chronic exposure to microbial structures such as LPS can lead to a selective and a transient hyporesponsive state called endotoxin tolerance. Antigen-presenting cells made tolerant (“tolerized”) in this manner have a reduced capacity to initiate an adaptive immune response (7, 8, 29). The molecular mechanisms involved in endotoxin tolerance are still unclear but include downregulation of TLRs (17, 20), alterations in signaling events downstream of Toll-like receptor (TLR) signaling (7, 16), and the induction of immune regulatory molecules such as Src homology 2 (SH2)-containing inositol phosphatase (SHIP), an inhibitor of NF-κB signaling (14, 23, 24).
Chronic periodontitis (CP) is an inflammatory disease of the oral mucosa (22) mediated by exposure to dental plaque, which contains >500 taxa of oral bacteria, ∼60% of which are cultivable (15, 21). Nonetheless, the disease appears to be associated with a small subset of species that share the common property of being gram negative, i.e., producing LPS (4, 9, 25). Our published results suggest that the host may respond to the products of these gram-negative species by downregulation of TLR2 and TLR4 at the level of transcription (17). This observation was corroborated using an in vitro endotoxin tolerance model, wherein endotoxin-sensitized mononuclear cells were rechallenged with LPS, inducing downregulation of TLR expression at both the transcriptional and protein levels. Moreover, tumor necrosis factor alpha and interleukin-1β (IL-1β) are downmodulated, but anti-inflammatory cytokines such as IL-10 were relatively resistant to the tolerizing effect of LPS exposure.
We present here the first evidence that SHIP, an immunoregulatory molecule involved in endotoxin tolerance, in negative regulation of T-cell responsiveness, and in osteoclast activation, is upregulated in CP compared to its levels under conditions of gingival health. We also show that mononuclear cells (MNC) isolated from tissues diseased from CP are in a state of endotoxin tolerance, evidenced by their resistance to upregulation of TLR2 and TLR4 expression, relative to cells from healthy patients. Interestingly, MNC from healthy gingival specimens, when stimulated with Porphyromonas gingivalis LPS, upregulated the expression of IL-1β, whereas the cells from CP tissues downregulated the expression of IL-1β during sensitization in vitro with P. gingivalis LPS. In summary, we substantiate our previous hypothesis that the oral mucosa is in a tolerized state, using isolated mononuclear cells; moreover, we provide further evidence for the potential immunoregulatory mechanisms involved by demonstrating elevated expression of SHIP-1 in diseased oral mucosa in chronic periodontitis.
(The work of M. Muthukuru on this study was in partial fulfillment of the Ph.D. degree.)
Clinical diagnoses and tissue collection.
The Institutional Review Board approved this protocol. The clinical criteria for CP and subject gingival health were as previously described (13, 17). Briefly, in CP subjects, the sextant from which tissue was harvested exhibited more than four teeth, with probing depths of 5 to 10 mm, attachment loss of 5 to 10 mm, alveolar bone loss present, and bleeding on probing present. Subjects with gingival health exhibited probing depths of less than 4 mm, attachment loss of 0 mm, no alveolar bone loss, and no bleeding on probing. Based on the clinical criteria of gingival health and CP, a total of 21 samples from the interproximal papillae were collected, under informed consent, for PCR analysis. The mean ages (and standard errors of the means) of the CP and control subjects were 55 (±14.0) and 47.3 (±10.9), respectively, while the distribution of males in the CP and control subject groups was 57% and 36%, respectively; however, no significant differences were detected in either the age or gender distributions of subjects in the CP and control groups (P < 0.05; Student's t test).
Gingival mononuclear cell isolation.
For gingival mononuclear cell isolation, gingival tissues from a subset of the patient population previously characterized (17) were subjected to overnight 2-mg/ml dispase (Invitrogen) treatment. On the following day, epithelial sheets were removed with a sharp forceps and the connective tissue components were incubated in Hanks-based tissue dissociation buffer with 4 mg/ml type II collagenase (Invitrogen) for about 4 h. The tissues were then mechanically disaggregated by passing them through a stainless steel tissue sieve (Fisher Scientific) and the single-cell fractions were collected. Cells were placed on a Ficoll-Hypaque gradient (Invitrogen) and centrifuged at 400 × g for 30 min, and the mononuclear fractions of the gingival cells were collected.
LPS isolation and gingival MNC stimulation.
LPS was isolated from P. gingivalis strain 381 by hot phenol-water extraction followed by isopycnic density gradient centrifugation and was further purified of contaminating nucleic acids, proteins, and lipoproteins as previously described (5). Mononuclear fractions of the healthy and CP gingival tissues were placed in RPMI 1640 supplemented with 10% heat-inactivated fetal calf serum. Cells from the tissues were brought to the same concentration by appropriately adjusting the amount of cell culture medium. Cells from each tissue were divided into two fractions, and one fraction served as a control to determine constitutive gene expression. P. gingivalis LPS (1 μg/ml) was added to the other fraction and incubated for 1 h. The viability of the cells after LPS stimulation (typically >90%) was determined by trypan blue exclusion. The cells were collected by centrifugation, washed three times with cold phosphate-buffered saline, and stored in RNA-stabilizing solution at −80°C for RNA extraction and PCR analysis.
Real-time PCR. (i) RNA extraction and cDNA synthesis.
Gingival tissues and ex vivo isolated mononuclear cells were placed in RNAlater RNA-stabilizing reagent (QIAGEN) and frozen at −80°C for later use. Once the required sample size was obtained, frozen gingival tissues and cells were then ground and homogenized, and total RNA was extracted using QIAGEN RNeasy midi kits for gingival tissues and mini kits for gingival mononuclear cells, according to the manufacturer's instructions. Avian reverse transcriptase (RT) PCR first-strand kits (Sigma) were used to synthesize cDNA from total RNA. The concentration of total RNA was determined by optical density at 260 nm (OD260), and small discrepancies in the amount of gingival tissue collected were corrected by loading the same concentration of RNA for cDNA synthesis. The purity of cDNA was determined by analysis of the OD260/OD280 ratio.
(ii) Primers for PCR.
Nucleotide sequences were determined from NCBI PubMed, and the primers were custom designed using Primer3 software. Primers for SHIP (also known as SHIP-1), one of the two isoforms, whose levels were demonstrated to be upregulated during endotoxin stimulation and also essential for induction of tolerance during a second stimulation (24), were designed from Genbank accession number U84400. Table 1 shows the sequences and the product sizes of the various primers used for PCR quantitation.
TABLE 1.
Primer sequence and product size for PCR quantitation
| Gene | Left primer (from 5′ to 3′) | Right primer (from 5′ to 3′) | Product size (bp) |
|---|---|---|---|
| SHIP | CTGAGGCTGGAAGAAAAACG | GCACTTAGCACAGCACTTGG | 222 |
| TLR2 | GGAGGCTGCATATTCCAAGG | GCCAGGCATCCTCACAGG | 216 |
| TLR4 | CTTGACCTTCCTGGACCTCTC | ACTTGGAAAATGCTGTAGTTCC | 217 |
| IL-1β | GGCAGAAAGGGAACAGAAAGG | AGTGAGTAGGAGAGGTGAGAGAGG | 200 |
| β-Actin | ACTCTTCCAGCCTTCCTTCC | GTTGGCGTACAGGTCTTTGC | 204 |
(iii) Real-time RT-PCR quantitation.
Real-time reverse transcriptase (RT)-PCR analysis was performed using the iCycler (Bio-Rad, Hercules, CA) with SYBR green kits (Bio-Rad, Hercules, CA) and mRNA quantitation by the standard curve method as previously described (17). For each transcript analyzed, a standard curve with predetermined concentrations and serially diluted respective PCR amplification products from 0.1 to 0.00001 ng was constructed. This approach allows the standards to be amplified the same way as the template cDNA in the unknown samples, as the product sequences and sizes are identical. Levels of β-actin mRNA served as an internal control to normalize samples for variations in sample volume loading, presence of inhibitors, and nucleic acid recovery during extraction and cDNA synthesis procedures (as discussed in online Roach PCR protocols). The normalized initial concentration of each transcript in every sample was converted to an initial copy number using the following formula: amount (copies/μl) = [6 × 1023 (copies/mol) × concentration (g/μl)]/molecular weight (g/mol), where the average molecular weight of double-stranded DNA is the number of base pairs times 660 Da/base pair. All analyses were performed in triplicate.
Upregulation of SHIP-1 in the oral mucosa during CP.
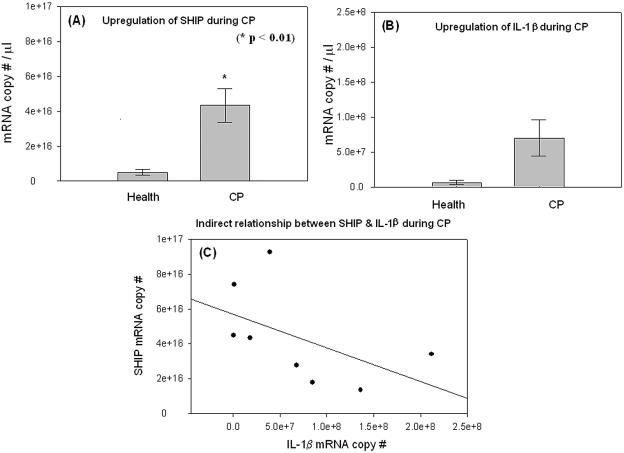
Gingival specimens were analyzed by quantitative real-time RT-PCR for the expression of SHIP-1. Expression of SHIP-1 was upregulated (Fig. 1A) in CP by approximately ninefold compared to levels in healthy tissue, and this difference was statistically significant (P < 0.01; Student's t test). As previously reported, the inflammatory state of the tissues in CP, relative to health, was confirmed by expression of IL-1β mRNA (Fig. 1B). Linear regression analysis (Fig. 1C), although not statistically significant, suggests an inverse relationship between IL-1β and SHIP-1 expression in the CP specimens.
FIG. 1.
Upregulation of SHIP-1 in the oral mucosa during CP. Healthy (n = 8) and CP (n = 8) human gingival specimens were collected and total RNA was extracted and quantitated and its purity confirmed as described in the text. cDNA was synthesized from identical quantities of total RNA from all samples. The purity of cDNA was determined by analysis of the OD260/OD280 ratio. mRNA quantitation was performed by real-time RT-PCR analysis. Levels of β-actin mRNA served as an internal control to normalize samples. All analyses were performed in triplicate. (A) Upregulation of SHIP mRNA in CP compared to gingival health by about ninefold (Student's t test; P < 0.01). (B) Upregulation of IL-1β mRNA in CP by about 11-fold compared to health, establishing the inflamed state of the CP specimens. (C) A trend to an inverse relationship between IL-1β and SHIP-1 expression was observed, although it was not statistically significant.
Impaired ability of ex vivo isolated MNC from CP subjects to upregulate TLRs and IL-1β.
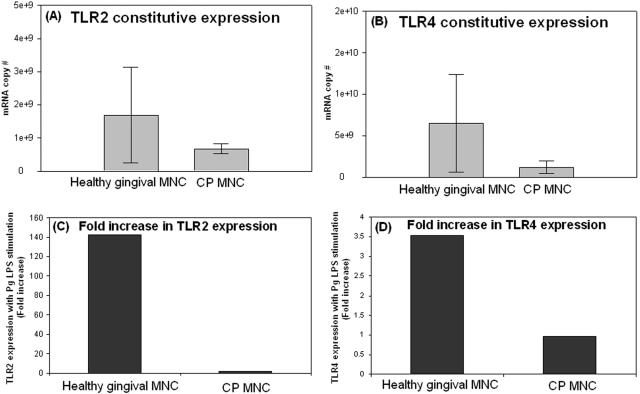
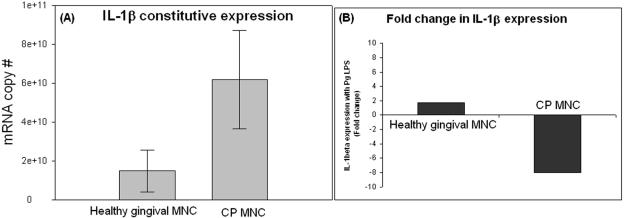
We have previously established, using whole gingival tissue, that tissues of CP patients have reduced levels of in situ transcripts for TLRs relative to healthy tissue and, moreover, that monocytes stimulated and then challenged in vitro with LPS are similarly impaired in TLR expression and inflammatory-cytokine production (17). Here, we isolated MNC from healthy and CP gingival tissues and determined constitutive expression of TLRs (Fig. 2) and IL-1β (Fig. 3). We then subjected the MNC to stimulation with P. gingivalis LPS. The results show that constitutive TLR2 (Fig. 2A) and TLR4 (Fig. 2B) mRNA expression was lower in MNC from CP patients than in those from healthy subjects. Moreover, when stimulated with P. gingivalis LPS, CP MNC upregulate TLR2 by only 2-fold (Fig. 2C), while healthy MNC upregulate TLR2 by 143-fold (Fig. 2C). Interestingly, TLR4 expression was unaltered in CP cells (Fig. 2D); however, healthy gingival cells upregulated TLR4 by about fourfold during P. gingivalis LPS stimulation (Fig. 2D). Figure 3A demonstrates that constitutive expression of IL-1β during CP is higher than that in gingival health; moreover, while healthy gingival MNC responded to LPS with a twofold increase in IL-1β (Fig. 3B), CP MNC downregulated the IL-1β expression eightfold (Fig. 3B).
FIG. 2.
Ex vivo isolated gingival cells from healthy specimens upregulate TLRs but not so those from CP tissues. Gingival MNC from healthy (n = 3) and CP (n = 2) gingival specimens were isolated as described in the text. MNC were divided into two fractions from each of the healthy and CP specimens. One of the cellular fractions served as an unstimulated control to determine the constitutive expression of TLR2 (A) and TLR4 (B), and the other fraction was stimulated for 1 hour with 1,000 ng/ml of P. gingivalis LPS, as described in the text, to determine the ability to respond. (C) Healthy MNC, upon in vitro stimulation with P. gingivalis LPS, upregulate TLR2 by 143-fold, and in contrast, CP MNC upregulate TLR2 by only 2-fold relative to the unstimulated control. (D) Healthy MNC, upon stimulation with P. gingivalis LPS, upregulate TLR4 by fourfold and CP MNC do not respond to P. gingivalis LPS (TLR4 expression is unaltered). Pg, P. gingivalis.
FIG. 3.
Differential regulation of IL-1β expression upon LPS stimulation in healthy and CP gingival mononuclear cells. Gingival mononuclear cells were isolated from healthy (n = 3) and CP (n = 2) gingival specimens and stimulated with P. gingivalis LPS for 1 hour as described in the text and the legend to Fig. 2. (A) Constitutive expression of IL-1β during CP is higher constitutive expression by three times during gingival health, which establishes the inflammatory state of CP gingivae. (B) Healthy MNC from healthy gingival specimens, upon stimulation with P. gingivalis LPS, upregulate IL-1β mRNA by twofold, while CP MNC, upon P. gingivalis LPS stimulation, downregulate the expression of IL-1β by eightfold relative to the constitutive expression (A). Pg, P. gingivalis.
We show here that transcripts for the SHIP-1 isoform are upregulated in gingival tissues from CP subjects (Fig. 1A); moreover, levels of IL-1β mRNA are inversely related to those of SHIP-1 mRNA in CP subjects (Fig. 1C), although this was not statistically significant. These data confirm and expand upon our published evidence that the oral mucosa in CP is in a state of endotoxin tolerance (17). Although endotoxin tolerance was previously documented by analysis of TLR transcripts in CP and healthy tissue and correlated with an in vitro endotoxin tolerance model using peripheral blood monocytes in vitro (17), lacking was more definitive evidence, i.e., that using mechanistic markers of endotoxin tolerance. Upregulation of SHIP-1 has recently been shown to mediate endotoxin tolerance by inhibiting activation of NF-κB, and hence, transcription of inflammatory cytokines is reduced (24). SHIP-1, or SH2 domain containing 5′-inositol phosphatase, is one of several inhibitory phosphatases that are recruited by the Lyn tyrosine kinase to the plasma membrane in response to specific stimuli (10). Others include SH2 domain-containing phosphatase-1 (SHP-1), SHP-2, and SHIP-2. The SHPs belong to a protein family which includes two vertebrate SHPs (SHP-1 and SHP-2) and invertebrate SHP orthologs (Cork-screw and Ptp-2) in Drosophila and Caenorhabditis elegans, respectively (reviewed in reference 18). All of these isoforms attenuate the phosphorylation level of proteins with tyrosine kinase activity and thus contribute to homeostasis by keeping tyrosine kinases at a dynamic equilibrium within biological systems (28, 31). Sustained activation of Lyn tyrosine kinase in vivo (i.e., in targeted gain-of-function mutation Lynup/up mice) leads to autoimmunity (11). SHIP expression is dependent on the autocrine action of transforming growth factor beta, upregulation of suppressor of cytokine secretion (SOCS), and the inhibitory interleukin-1 receptor-associated kinase-M (IRAK-M) (24). SHIP may play an important role as a potential negative regulator of autoimmune diseases and as a target for immunotherapy (3, 10). Moreover, SHIP is expressed by T cells and plays a role in antigen receptor signal transduction, which is central to the development and function of T and B lymphocytes in generating a productive immune response (27). Interestingly, overexpression of SHP-1 has been shown to enhance the number of tartrate-resistant acid phosphatase-positive multinuclear osteoclast-like cells in response to the receptor activator of NF-κB ligand (RANKL) in a dose-dependent manner (31), suggesting an intriguing link between elevated expression of these inhibitory phosphatases, as shown here, and in osteoclastic bone loss in periodontal disease.
Further lacking was evidence that the mononuclear cell fraction, among the many other cell types that populate the gingiva (6), was tolerized by exposure to endotoxin. This was demonstrated by the isolation of MNC from CP and healthy tissues, which indicates that, indeed, MNC of CP subjects show (i) decreased (constitutive) expression of TLR2 and TLR4 in CP MNC (Fig. 2A through D) and (ii) decreased responsiveness to in vitro LPS challenge, as determined by impaired induction of TLR (Fig. 2C and D) and IL-1β (Fig. 3B) transcripts. Such a regulatory mechanism may be important for the host to establish immune homeostasis during inflammatory conditions like chronic periodontitis and may offer clues for the development of immune therapies. This needs to be weighed against the roles of tyrosine phosphatases in impairment of specific protective immunity and in induction of osteoclastogenesis.
Acknowledgments
This study was supported by a U.S. Public Health Service grant from the NIH/NIDCR (R01 DE14328).
Editor: J. D. Clements
REFERENCES
- 1.Baumgart, D. C., D. Metzke, J. Schmitz, A. Scheffold, A. Sturm, B. Wiedenmann, and A. U. Dignass. 2005. Patients with active inflammatory bowel disease lack immature peripheral blood plasmacytoid and myeloid dendritic cells. Gut 54:228-236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beutler, B., K. Hoebe, X. Du, and R. J. Ulevitch. 2003. How we detect microbes and respond to them: the toll-like receptors and their transducers. J. Leukoc. Biol. 74:479-485. [DOI] [PubMed] [Google Scholar]
- 3.Bowcock, A. M., and J. G. Krueger. 2005. Getting under the skin: the immunogenetics of psoriasis. Nat. Rev. Immunol. 5:699-711. [DOI] [PubMed] [Google Scholar]
- 4.Cohen, N., J. Morisset, and D. Emilie. 2004. Induction of tolerance by Porphyromonas gingivalis on APCs: a mechanism implicated in periodontal infection. J. Dent. Res. 83:429-433. [DOI] [PubMed] [Google Scholar]
- 5.Cutler, C. W., P. I. Eke, C. A. Genco, T. E. Van Dyke, and R. R. Arnold. 1996. Hemin-induced modifications of the antigenicity and hemin-binding capacity of Porphyromonas gingivalis lipopolysaccharide. Infect. Immun. 64:2282-2287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cutler, C. W., and R. Jotwani. 2004. Antigen-presentation and the role of dendritic cells in periodontitis. Periodontology 2000 35:135-157. [DOI] [PubMed] [Google Scholar]
- 7.Dobrovolskaia, M. A., A. E. Medvedev, K. E. Thomas, N. Cuesta, V. Toshchakov, T. Ren, M. J. Cody, S. M. Michalek, N. R. Rice, and S. N. Vogel. 2003. Induction of in vitro reprogramming by toll-like receptor (TLR) 2 and TLR4 agonists in murine macrophages: effects of TLR “homotolerance” versus “heterotolerance” on NF-kappa B signaling pathway components. J. Immunol. 170:508-519. [DOI] [PubMed] [Google Scholar]
- 8.Dobrovolskaia, M. A., and S. N. Vogel. 2002. Toll receptors, CD14, and macrophage activation and deactivation by LPS. Microbes Infect. 4:903-914. [DOI] [PubMed] [Google Scholar]
- 9.Ezzo, P. J., and C. W. Cutler. 2003. Microorganisms as risk indicators for periodontal disease. Periodontol. 2000 32:24-35. [DOI] [PubMed] [Google Scholar]
- 10.Harder, K. W., C. Quilici, E. Naik, M. Inglese, N. Kountouri, A. Turner, K. Zlatic, D. M. Tarlinton, and M. L. Hibbs. 2004. Perturbed myelo/erythropoiesis in Lyn-deficient mice is similar to that in mice lacking the inhibitory phosphatases SHP-1 and SHIP-1. Blood 104:3901-3910. [DOI] [PubMed] [Google Scholar]
- 11.Hibbs, M. L., K. W. Harder, J. Armes, N. Kountouri, C. Quilici, F. Casagranda, A. R. Dunn, and D. M. Tarlinton. 2002. Sustained activation of Lyn tyrosine kinase in vivo leads to autoimmunity. J. Exp. Med. 196:1593-1604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.James, S. P. 2005. Prototypic disorders of gastrointestinal mucosal immune function: celiac disease and Crohn's disease. J. Allergy Clin. Immunol. 115:25-30. [DOI] [PubMed] [Google Scholar]
- 13.Jotwani, R., and C. W. Cutler. 2003. Multiple dendritic cell (DC) subpopulations in human gingiva and association of mature DCs with CD4+ T-cells in situ. J. Dent. Res. 82:736-741. [DOI] [PubMed] [Google Scholar]
- 14.Kalesnikoff, J., N. Baur, M. Leitges, M. R. Hughes, J. E. Damen, M. Huber, and G. Krystal. 2002. SHIP negatively regulates IgE + antigen-induced IL-6 production in mast cells by inhibiting NF-kappa B activity. J. Immunol. 168:4737-4746. [DOI] [PubMed] [Google Scholar]
- 15.Kroes, I., P. W. Lepp, and D. A. Relman. 1999. Bacterial diversity within the human subgingival crevice. Proc. Natl. Acad. Sci. USA 96:14547-14552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Medvedev, A. E., K. M. Kopydlowski, and S. N. Vogel. 2000. Inhibition of lipopolysaccharide-induced signal transduction in endotoxin-tolerized mouse macrophages: dysregulation of cytokine, chemokine, and toll-like receptor 2 and 4 gene expression. J. Immunol. 164:5564-5574. [DOI] [PubMed] [Google Scholar]
- 17.Muthukuru, M., R. Jotwani, and C. W. Cutler. 2005. Oral mucosal endotoxin tolerance induction in chronic periodontitis. Infect. Immun. 73:687-694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Neel, B. G., H. Gu, and L. Pao. 2003. The ‘Shp'ing news: SH2 domain-containing tyrosine phosphatases in cell signaling. Trends Biochem. Sci. 28:284-293. [DOI] [PubMed] [Google Scholar]
- 19.Netea, M. G., C. Van der Graaf, J. W. Van der Meer, and B. J. Kullberg. 2004. Toll-like receptors and the host defense against microbial pathogens: bringing specificity to the innate-immune system. J. Leukoc. Biol. 75:749-755. [DOI] [PubMed] [Google Scholar]
- 20.Nomura, F., S. Akashi, Y. Sakao, S. Sato, T. Kawai, M. Matsumoto, K. Nakanishi, M. Kimoto, K. Miyake, K. Takeda, and S. Akira. 2000. Endotoxin tolerance in mouse peritoneal macrophages correlates with down-regulation of surface toll-like receptor 4 expression. J. Immunol. 164:3476-3479. [DOI] [PubMed] [Google Scholar]
- 21.Paster, B. J., S. K. Boches, J. L. Galvin, R. E. Ericson, C. N. Lau, V. A. Levanos, A. Sahasrabudhe, and F. E. Dewhirst. 2001. Bacterial diversity in human subgingival plaque. J. Bacteriol. 183:3770-3783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Seymour, G. J. 1991. Importance of the host response in the periodontium. J. Clin. Periodontol. 18:421-426. [DOI] [PubMed] [Google Scholar]
- 23.Sly, L. M., M. J. Rauh, J. Kalesnikoff, T. Buchse, and G. Krystal. 2003. SHIP, SHIP2, and PTEN activities are regulated in vivo by modulation of their protein levels: SHIP is up-regulated in macrophages and mast cells by lipopolysaccharide. Exp. Hematol. 31:1170-1181. [DOI] [PubMed] [Google Scholar]
- 24.Sly, L. M., M. J. Rauh, J. Kalesnikoff, C. H. Song, and G. Krystal. 2004. LPS-induced upregulation of SHIP is essential for endotoxin tolerance. Immunity 21:227-239. [DOI] [PubMed] [Google Scholar]
- 25.Socransky, S. S., A. D. Haffajee, M. A. Cugini, C. Smith, and R. L. Kent, Jr. 1998. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 25:134-144. [DOI] [PubMed] [Google Scholar]
- 26.Takeuchi, O., K. Hoshino, T. Kawai, H. Sanjo, H. Takada, T. Ogawa, K. Takeda, and S. Akira. 1999. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11:443-451. [DOI] [PubMed] [Google Scholar]
- 27.Tomlinson, M. G., V. L. Heath, C. W. Turck, S. P. Watson, and A. Weiss. 2004. SHIP family inositol phosphatases interact with and negatively regulate the Tec tyrosine kinase. J. Biol. Chem. 279:55089-55096. [DOI] [PubMed] [Google Scholar]
- 28.Tonks, N. K., and B. G. Neel. 2001. Combinatorial control of the specificity of protein tyrosine phosphatases. Curr. Opin. Cell Biol. 13:182-195. [DOI] [PubMed] [Google Scholar]
- 29.West, M. A., and W. Heagy. 2002. Endotoxin tolerance: a review. Crit. Care Med. 30:S64-S73. [PubMed] [Google Scholar]
- 30.Zhang, F. X., C. J. Kirschning, R. Mancinelli, X. P. Xu, Y. Jin, E. Faure, A. Mantovani, M. Rothe, M. Muzio, and M. Arditi. 1999. Bacterial lipopolysaccharide activates nuclear factor-κB through interleukin-1 signaling mediators in cultured human dermal endothelial cells and mononuclear phagocytes. J. Biol. Chem. 274:7611-7614. [DOI] [PubMed] [Google Scholar]
- 31.Zhang, Z., E. Jimi, and A. L. Bothwell. 2003. Receptor activator of NF-kappa B ligand stimulates recruitment of SHP-1 to the complex containing TNFR-associated factor 6 that regulates osteoclastogenesis. J. Immunol. 171:3620-3626. [DOI] [PubMed] [Google Scholar]