Abstract
Infectious mononucleosis (IM) is an acute sporadic infection that usually affects young adults, and during infection a massive expansion of CD8 T cells is generally considered to occur. However, CD28 expression of the expanded cells has not been characterized. When peripheral blood mononuclear cells of acute IM (AIM) patients were analyzed by flow cytometry, a continuous spectrum of CD28 intensity ranging from negative to high, which could be separated into CD28 negative, intermediate (int), and positive, was seen for CD8 T cells. We studied 26 IM patients who were diagnosed on the basis of standard methods and found that all patients had the continuous CD28 spectrum. CD28 is a costimulatory molecule on T cells, and its expression is associated with the subdivision of CD8 cells into cytotoxic (CD28-positive) and suppressor (CD28-negative) T cells. After 24 h of ex vivo culturing, however, the continuous spectrum was found to consist of only CD28-positive and CD28-negative CD8 T cells, because the CD28-int cells had disappeared due to apoptosis. The CD28-int T cells have several cytotoxic functions, suggesting that CD28-int T cells are effectors. Examination of other costimulatory markers in AIM patients showed that CD80 and CD152 were not affected. In patients with other viral infections, such as measles or rubella, however, the continuous spectrum was not detected. These results suggest that there is an unusual CD28 expression pattern in patients with AIM, namely, the presence of a functional CD28-int subset among CD8 T cells. These findings are of special importance for clarifying the defense mechanism against Epstein-Barr virus infection, and the role of CD28 molecules in humans and should also be helpful for the diagnosis of AIM.
Epstein-Barr virus (EBV) is a ubiquitous gammaherpesvirus that actively infects epithelial cells and establishes latency in B cells (4, 6). Most infections occur in young children and are asymptomatic. Adolescents and young adults infected with EBV often present with infectious mononucleosis (IM). Thereafter, in humans the virus maintains a latent infection without symptoms despite a vigorous immune response (8).
Acute IM (AIM), a manifestation of primary infection with EBV, is characterized by viral replication in the oropharynx, latent virus infection in a fraction of B cells, and massive expansion of T lymphocytes (9, 20, 28). Although there is a strong antibody (Ab) response to EBV, T cells play the main role in controlling both the primary and persistent phases of infection and in preventing the development of immunoblastic B-cell lymphomas (4). The expanded lymphocytes are mainly CD8 cells and are an activated effector population that arises in response to, and is directed against, the invading pathogen (2, 3, 7). Several CD8 subsets, such as HLA-DR and CD45RO, are reportedly increased in AIM (3, 7, 27). However, these are general activation markers, and an effector subset specific for IM has not been clarified among CD8 T cells.
CD28 is a homodimeric glycoprotein member of the immunoglobulin (Ig) supergene family of costimulatory molecules (5). Optimal stimulation of T cells requires engagement of T-cell receptors in conjugation with a second signal: ligation of the CD28-related molecules (14). The interaction of CD28 on the surface of T cells with members of the B7 receptor family on the surface of antigen presenting cells provides an important type of costimulation (5, 14). The expression of the CD28 molecule is limited mostly to T cells, and CD28 is expressed on virtually all resting CD4 T cells and on about 50% of resting CD8 T cells (14). Furthermore, according to their CD28 expression, CD8 T cells can be subdivided into cytotoxic (CD28-positive) and suppressor (CD28-negative) T cells (10). At present, however, the mechanism of regulation of CD28 expression and the role of CD28 molecules are unknown.
The present study aimed to evaluate CD28 expression in the activated populations of CD8 T cells that appear during AIM. We found the emergence of a novel, functional CD28-intermediate (int) subset among CD8 T cells of AIM patients.
MATERIALS AND METHODS
Patients and controls.
Clinical diagnosis of IM was based on fever, lymphocytosis, splenomegaly, and atypical lymphocytes in the peripheral blood. Patients who showed a positive IgM Ab titer for EBV capsid antigen (4) were selected for the present study. A total of 26 AIM patients (9 males and 17 females; age range, 15 to 33 years) with recent onset were examined.
Forty-five patients (19 males and 26 females; all under 35 years old) with several other viral infections (measles, human immunodeficiency virus [HIV], rubella, erythema infectiosum, hand-foot-and-mouth disease, hepatitis B and C, influenza, or mumps) who were diagnosed on the basis of serological tests and clinical symptoms were also examined.
Uninfected individuals (under 40 years old, more than 100 subjects) served as healthy controls. Blood was taken immediately after diagnosis, and informed consent was obtained.
Isolation of PBMC.
Human peripheral heparinized blood was diluted with equal parts of RPMI 1640 medium (Nikken Bio-medical, Kyoto, Japan). Peripheral blood mononuclear cells (PBMC) were obtained by centrifuging the diluted blood over Ficoll-Hypaque (Pharmacia, Uppsala, Sweden) and were washed three times with RPMI 1640 medium (21).
Flow cytometric analysis of CD8/CD28-related molecules.
Cells (106/tube) were incubated on ice for 30 min with fluorescein isothiocyanate (FITC)-conjugated anti-human CD8 monoclonal Ab (MAb) (Becton Dickinson & Co., Mountain View, Calif.) and phycoerythrin (PE)-conjugated anti-human CD28 MAb (Becton Dickinson) or FITC-conjugated Annexin-V (CALTAG) in RPMI 1640 supplemented with 5% fetal calf serum (FCS; Sterile System, Logan, Utah) and 0.02% NaN3 as described previously (21).
After the cells were washed and resuspended in the RPMI 1640 medium, flow cytometric analysis was done using a FACScan flow cytometer (21) (Becton Dickinson). Subsequently, the expression of HLA-DR, CD16, CD56, CD45RA, CD45RO, CD80, and CD152 was also examined. Usually, 50,000 viable cells were examined, and the findings were analyzed by using CELLQuest software (Becton Dickinson) (21).
Assay of cytotoxic function in three CD8 subsets.
After CD8 cells were separated into three fractions (CD28 positive, CD28 int, and CD28 negative [see Fig. 4]), CD28-positive cells were first electronically sorted with a FACSVantage cell sorter (Becton Dickinson) and then the CD28-int and -negative fractions were sorted. Throughout the culture period the cells were cultured in the presence of interleukin-2 (IL-2) (100 U/ml; Takeda) and IL-10 (100 U/ml; Genzyme) to prevent apoptosis (25). At the end of sorting, the CD28-positive, -int, and -negative expression patterns of the sorted cell fractions were confirmed.
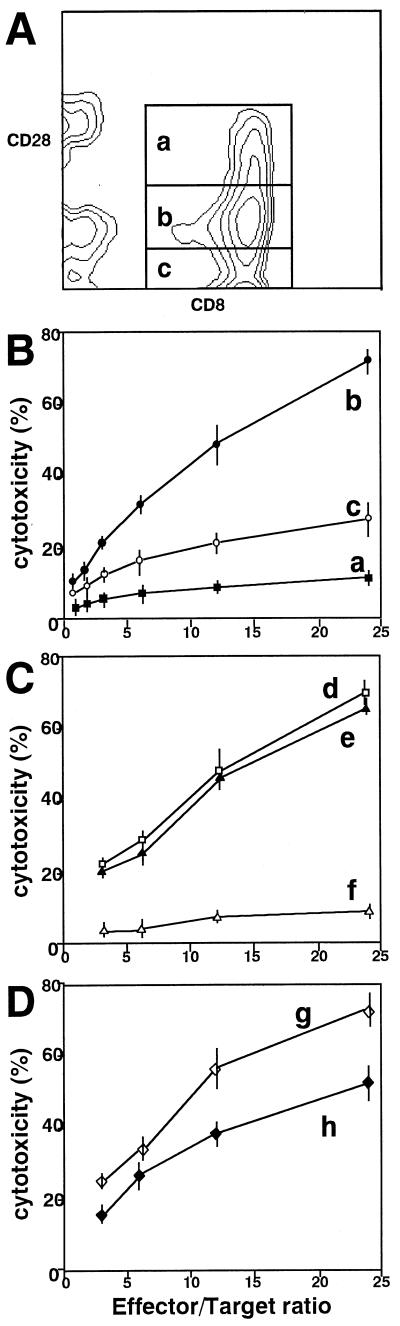
FIG. 4.
The CD28-int CD8 T cells have cytotoxic activity specific for autologous EBV-transformed B cells. (A) Separation of CD8 T cells from a patient with AIM into CD28-positive, -int, and -negative fractions. Flow cytometric analyses of CD8 and CD28 expressions showed that CD8 T cells could be fractionated into CD28-positive (a), -int (b), and -negative (c) CD8 subsets. Throughout the culture period the cells were incubated in the presence of IL-2 (100 U/ml) and IL-10 (100 U/ml) to prevent apoptosis (23). Typical results of three different AIM patients are shown. (B) Comparison of cytotoxic activities among CD8/CD28 subsets. Each CD8/CD28 subset was separately isolated by two cycles of electronic sorting. The three fractionated CD8 subsets (a, b, and c) were tested in triplicate for cytotoxicity against 51Cr-labeled autologous BLC with various effector/target ratios (1). After 5 h of incubation, the radioactivity was counted and the percentage of specific lysis was calculated (means ± standard deviations). (C) Inhibition of the cytotoxicity with Ab to HLA class I in CD28-int T cells. The fractionated CD28-int CD8 T cells (Fig. 4A, section b) and radiolabeled autologous BLC were incubated with an anti-class I monoclonal Ab (line f), isotype-matched mouse IgG Ab (line e), or medium alone (line d). After 5 h of incubation, the radioactivity was counted and the percentage of specific lysis was calculated. (D) Cytotoxic functions against other targets. The fractionated CD28-int subsets (HLA-A11/A26 and B52/B56) were tested in triplicate for cytotoxicity against 51Cr-labeled allogeneic BLC (HLA-A2/A24 and B35/B46; line g) and K562 (line h) cells with various effector/target ratios (1). After 5 h of incubation, the radioactivity was counted and the percentage of specific lysis was calculated (means ± standard deviations).
Autologous and HLA-mismatched EBV-transformed B lymphoblastoid cells (BLC) were prepared by coculturing PBMC of the AIM patients with the culture supernatant of the B95-8 cell line, as described previously (7). BLC were cultured in RPMI 1640 with 10% FCS.
Cytotoxicity assays were performed with 51Cr release as described previously (25). Briefly, fractionated CD8 T cells were seeded in the wells of V-bottomed 96-well plates (Falcon) at the desired cell concentration. BLC and an NK-sensitive K562 cell line (23) were incubated with 51CrO4 for 90 min, and the labeled cells were washed and resuspended in the culture medium. BLC or K562 cells were then added to each well (2,500 cells/well). After 5 h of incubation, the supernatants were collected and the radioactivity was counted with a gamma counter (Beckman). Each assay was performed in triplicate. The percentages of specific cytotoxicity were calculated as 100 × ([experimental release − spontaneous release]/[total release − spontaneous release]). In all experiments the spontaneous release was less than 20% of the maximum release.
For blocking experiments, an anti-class I MAb (Clone W6/32; Cederlane, Hornby, Ontario, Canada) or isotype-matched mouse IgG antibodies were used at a final concentration of 50 μg/ml.
RESULTS
Appearance of a continuous feature of CD28 expression among CD8 T cells in AIM.
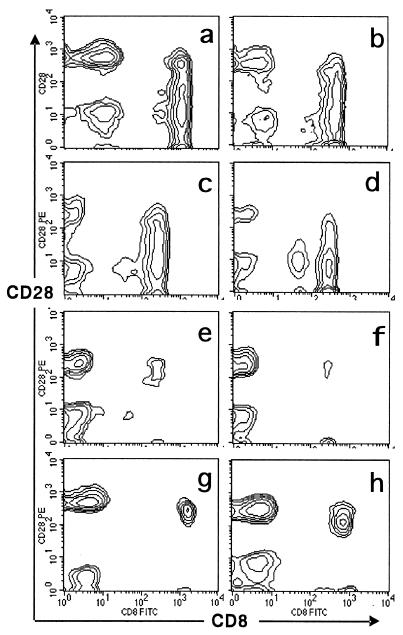
Analysis of CD8/CD28 expression was carried out by flow cytometry immediately after diagnosis of patients who were suspected of having AIM on the basis of their clinical symptoms. An unusual feature, a continuous spectrum of CD28 intensity ranging from negative to high, was seen for CD8 T cells (typical results are shown in Fig. 1a through d). The percentage of cells in the quadrants of Fig. 1 is also shown in Table 1. Massive expansion of CD8 T cells was observed as reported previously (2, 7). After diagnosis of AIM was confirmed on the basis of serum tests and clinical symptoms, every patient who showed the continuous CD28 expression was diagnosed with AIM. Subsequently, 22 additional AIM patients were examined, and all of them showed the continuous spectrum of CD28 intensity in CD8 but not CD4 T cells (four cases of the latter are shown in Fig. 3a, 4A, and 5). CD8 T cells from 45 patients with other viral infections, such as measles or rubella, however, were clearly separated into two subsets according to their CD28 intensity (Fig. 1e and f), as they generally also were for healthy controls (Fig. 1g and h). The present findings suggest that AIM patients specifically show a unique, continuous spectrum of CD28 intensity in CD8 T cells.
FIG. 1.
Emergence of a continuous figure among CD8 T cells in patients with AIM. After PBMC were separated by Ficoll-Hypaque, the expression of CD28 and CD8 was examined by flow cytometry. Twenty-six AIM patients were examined at diagnosis, and results of four typical samples are shown (a through d). The expression was also examined for 45 patients with other viral infections (measles, HIV, rubella, erythema infectiosum, hand-foot-and-mouth disease, hepatitis B and C, influenza, or mumps) and for healthy controls. Typical findings for viral infections (sample e, hand-foot-and-mouth disease, and sample f, measles) and healthy controls (g and h) are also shown.
TABLE 1.
Expansion of CD8/CD28 T cells in patients with AIMa
| T-cell type | % T cells from sample:
|
|||||||
|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | |
| CD8−/CD28+ | 31 | 21 | 14 | 10 | 35 | 60 | 61 | 53 |
| CD8−/CD28− | 13 | 12 | 16 | 16 | 54 | 30 | 20 | 22 |
| CD8+/CD28+ | 20 | 27 | 28 | 14 | 8 | 5 | 1 | 20 |
| CD8+/CD28− | 36 | 40 | 42 | 60 | 9 | 5 | 2 | 5 |
After PBMC were separated by Ficoll-Hypaque, the expression of CD28 and CD8 in patients with several viral infections was examined by flow cytometry. The percentages of cells in the quadrants of Fig. 1 were calculated. The results of four typical samples of AIM (a through d), other viral infections (e and f), and healthy controls (g and h) from Fig. 1 are shown.
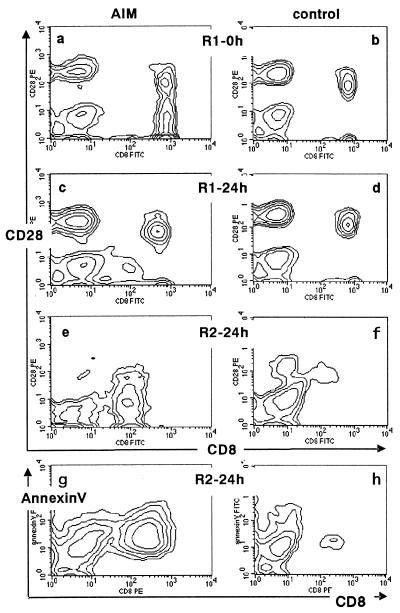
FIG. 3.
Disappearance of the CD28-int subset in ex vivo cultures due to apoptosis. After PBMC of an AIM patient (a, c, e, and g) and a healthy control (b, d, f, and h) were separated, the PBMC were stained for either CD8 and CD28 (a through f) or CD8 and Annexin-V (g and h), and two-color flow cytometric analyses were carried out before and after 24 h of ex vivo culturing. PBMC were cultured in RPMI 1640 with 10% FCS at 37°C. For this analysis, the R1 (a through d) and R2 (e through h) fractions shown in Fig. 2 were examined. The values are representative of those obtained in five independent experiments.
The reason why the continuous figures are seen in AIM patients is not clear, but our findings suggest that a novel, third population of CD8 T cells, a CD28-int subset, whose level of CD28 intensity is between those of fully positive and negative cells, might have emerged during AIM infection. We identified the third population as a CD28-int CD8 T-cell subset.
Appearance of the R2 fraction after 24 h of ex vivo culturing.
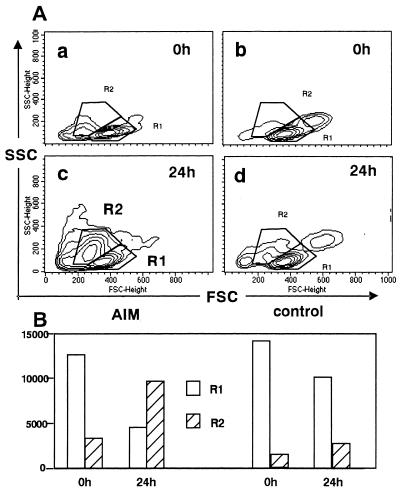
Another striking feature of AIM T cells is that they rapidly die via apoptosis when incubated ex vivo (1, 26, 27). After PBMC of AIM patients were cultured for 24 h ex vivo, flow cytometric light scattering analyses were carried out; typical examples are shown in Fig. 2. When PBMC were freshly isolated and subjected to forward and side scattering (FSC and SSC) analyses, most cells were found in the R1 fraction both in AIM patients and healthy controls (Fig. 2A, samples a and b). FSC and SSC reflect the approximate cell size and density, respectively. Usually, most lymphocytes are included in the R1 fraction. After 24 h of ex vivo culturing, a unique R2 fraction emerged in the AIM patients (Fig. 2A, sample c) and the proportion of cells included in the R1 fraction was decreased markedly (Fig. 2B), so that the majority of the cells were found in the R2 fraction (Fig. 2B). In contrast, the proportion of cells included in the R2 fraction did not increase markedly in the healthy controls (Fig. 2A, sample d, and B).
FIG. 2.
A flow cytometric light scattering analysis before and after ex vivo culturing. (A) After the PBMC of an AIM patient and a healthy control were separated, flow cytometric light scattering analyses were carried out immediately and after 24 h of ex vivo culturing (21). PBMC were selectively gated with FSC and SSC analysis, and the staining patterns were compared before and after 24 h of ex vivo culturing. PBMC were cultured in RPMI 1640 with 10% FCS at 37°C. During the analyses, two typical fractions, R1 and R2, appeared and are marked. Most lymphocytes were found in the R1 fraction. After 24 h of ex vivo culturing, a unique R2 fraction emerged in the AIM patient. The values are representative of those obtained in five independent experiments. (B) Comparison of cell numbers included in the R1 and R2 fractions. The number of cells in the R1 and R2 fractions were compared before and after 24 h of ex vivo culturing for the AIM patient and a healthy control.
Disappearance of the CD28-int subset due to apoptosis.
The expression of CD8 and CD28 in the R1 and R2 fractions was examined. In CD8 T cells in the R1 fraction of the AIM patients (Fig. 2A), continuous spectrums were seen on day 0 (Fig. 3a). After 24 h of ex vivo culturing, however, these continuous spectrums disappeared in the R1 fraction, and only CD28-positive and -negative CD8 T cells were detected (Fig. 3c), as was generally observed for the healthy controls (Fig. 3b). Even after 24 h of culturing, the expression pattern of CD8/CD28 did not change significantly in healthy controls (Fig. 3d). In AIM patients, analysis of the R2 fraction of the 24 h ex vivo cultures revealed that CD28-int cells were found in abundance (Fig. 3e), suggesting that the CD28-int cells were converted from the R1 to the R2 fraction during the 24 h of culturing, which would explain why the continuous spectrum disappeared from the R1 fraction. Analysis of the R2 fraction of the healthy controls, however, revealed that few CD8 cells were present in the 24-h cultures (Fig. 3f).
When the cells that appeared in the R2 fraction after 24 h were stained with Annexin-V, which detects early signs of apoptosis (11), most of the CD8 cells from the AIM patients were apoptotic (Fig. 3g). In contrast, few CD8 cells were apoptotic in the healthy controls (Fig. 3h). Taken together, these findings suggest that a CD28-int subset emerges among CD8 T cells during AIM infection and undergoes apoptosis during 24 h of culturing, and that the ex vivo cultures can discriminate this newly expanded cell population from the ubiquitous CD8 T-cell subsets. The present findings support previous observations that the expanded cells in AIM undergo apoptosis in ex vivo cultures (1, 26) and suggest that the expanded population is mainly a CD28-int subset.
CD28-int T cells have a cytotoxic function for EBV-related antigen.
It has been suggested that in AIM infection the expanded T-cell population has several cytotoxic functions (2, 23, 24, 25). However, the specificity and functions of these expanded T cells have been difficult to assess in vitro because most of the expanded cells die via apoptosis (1, 26). In the following experiments, the function of the CD28-int subset was examined in the presence of IL-2 and IL-10, which prevent apoptosis in long-term cultures (1, 25). After the CD8-positive fraction of AIM patients was separated into CD28-positive, CD28-int, and CD28-negative fractions (Fig. 4A), each of these fractions was sorted and the cytotoxic activity against autologous EBV-transformed B cells was measured (1). Cytotoxic activity was predominantly detected in the CD28-int population, and little cytotoxic activity was detected in the CD28-positive fraction (Fig. 4B). Subsequently, we examined the class I restriction of the response. The cytotoxic assay was carried out in the presence of anti-class I MAb or isotype-matched mouse Ab as a control. The cytotoxic function was drastically reduced with anti-class I Ab but not with control Ab (Fig. 4C). The results show that the cytotoxic function of CD28-int T cells was through authentic recognition of antigens presented by class I molecules.
In further experiments, we examined whether CD28-int T cells can kill other targets, such as HLA-mismatched BLC which were reportedly susceptible to being killed by AIM T cells (23, 25). The labeled allogeneic BLC and K562 cells were added to fractionated CD28-int T cells, and cytotoxic functions were examined. As shown in a representative experiment, CD28-int T cells showed cytotoxic effects against K562 cells and allogeneic BLC. These findings suggest that the unique CD28-int subset of CD8 T cells, expanded in AIM, might be effector cells that function in the defense against EBV infection.
Time course of emergence and disappearance of CD28-int T cells in AIM.
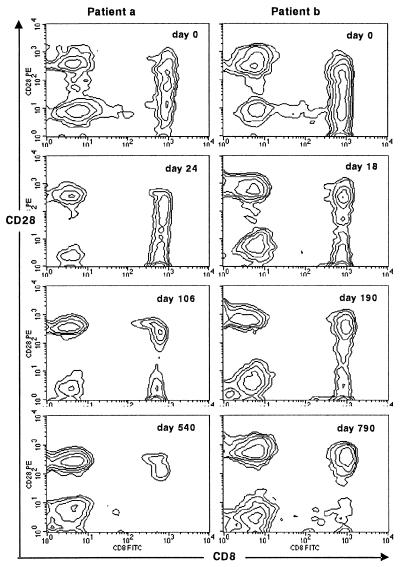
Upon the resolution of AIM, the lymphadenopathy disappears and the absolute number of circulating T cells, as well as the relative proportions of CD4 and CD8 cells, return to normal (3, 4). Next we examined the time course of the expression of the CD28-int subset. Flow cytometric analysis of CD28 and CD8 revealed that the massive expansion of the CD28-int CD8 T cells diminished gradually after the clinical symptoms disappeared (typical figures are shown in Fig. 5). The percentage of cells in the quadrants of Fig. 5 are shown in Table 2. The continuous spectrum of CD28 expression persisted for more than 1 month, and the inversion of the CD4/CD8 ratio lasted as long as 6 months. Surprisingly, it took more than 1 year to regain a normal CD4/CD8 ratio in the peripheral blood. These findings suggest that CD28-int CD8 T cells emerge during AIM and have profound effects on lymphocytes, especially CD4 and CD8 T cells, in vivo after the clinical symptoms have disappeared.
FIG. 5.
Time course of the CD28-int CD8 T-cell subset during the disease course in AIM patients. After PBMC was isolated, CD8 and CD28 analyses were carried out by using flow cytometry (21). The patterns of CD8 and CD28 expression were determined at the diagnosis of AIM and after resolution of the illness. Typical findings of two AIM patients (patients a and b) are shown. The CD4/CD8 ratios were 0.71 (0 days), 0.45 (24 days), 1.05 (106 days), and 3.33 (540 days) for patient a and 0.43 (0 days), 1.21 (18 days), 0.99 (190 days), and 1.75 (790 days) for patient b.
TABLE 2.
Time course of CD8/CD28 expression in AIM patientsa
| T-cell type | % T cells in patient on day:
|
|||||||
|---|---|---|---|---|---|---|---|---|
| a
|
b
|
|||||||
| 0 | 24 | 106 | 540 | 0 | 18 | 190 | 790 | |
| CD8−/CD28+ | 25 | 26 | 41 | 53 | 27 | 40 | 34 | 41 |
| CD8−/CD28+ | 37 | 12 | 18 | 32 | 10 | 25 | 32 | 35 |
| CD8+/CD28+ | 15 | 28 | 23 | 9 | 27 | 19 | 22 | 18 |
| CD8+/CD28− | 23 | 34 | 18 | 6 | 36 | 16 | 12 | 6 |
Expression of other phenotypes in AIM.
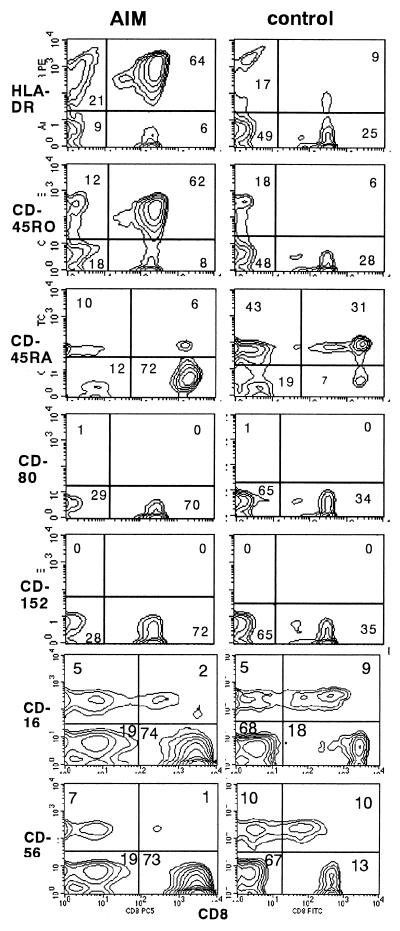
Other phenotypes, such as HLA-DR, also were examined in cells from AIM patients, and typical findings are shown in Fig. 6. In the AIM patient samples, about 90% of CD8 T cells expressed HLA-DR and CD45RO (markers of activation), which is in agreement with reported results (3, 7, 27). In contrast, less than 10% of CD8 cells were CD45RA positive. In healthy controls, most CD8 cells were both HLA-DR and CD45RO negative and CD45RA positive. These findings suggest that the massively expanded CD8 cells were HLA-DR and CD45RO positive and CD45RA negative.
FIG. 6.
Expression of CD8 and other phenotypes in AIM patients. After PBMC were isolated by Ficoll-Hypaque, the expression of CD8 and other activation markers (HLA-DR, CD45-RO, and CD45-RA), NK cell markers (CD16 and CD56), and costimulatory molecules (CD80 and CD152) was examined by flow cytometry (21). Typical results obtained with samples of an AIM patient and a healthy control are shown. The percentages of cells in quadrants are given in each panel.
Subsequently, NK cell markers were examined. In the AIM patient samples, CD16- and CD56-positive CD8 T cells were less than 2% of total cells examined, suggesting that the expanded CD8 T cells were different from that of NK cells.
Other costimulatory molecules, CD152 and CD80, were also examined. Few lymphocytes, including CD8 T cells, were CD152 and CD80 positive in either the patients or the healthy controls. These findings suggest that the CD28-int subset emerged without affecting other costimulatory molecules, such as CD80 and CD152, in AIM patients.
DISCUSSION
The present findings provide evidence suggesting that a novel CD28-int subset emerges among CD8 T cells but not among CD4 T cells and that this subset accounts for an unusual continuous spectrum of CD28 intensity that could be separated into the classifications of negative, int, and positive for patients with AIM.
It is generally accepted that a clonal expansion of T cells occurs during AIM infection and that the characteristic features of the expanded cells are as follows. (i) Most expanded cells are CD8 T cells (2, 3, 7). (ii) Most of the expanded CD8 T cells are HLA-DR positive, CD45RO positive, CD45RA negative, CD38 positive, and CD62 negative (3, 7, 27). (iii) The expanded cells undergo apoptosis upon ex vivo culturing (1, 27). (iv) The expanded cells have cytotoxicity specific for EBV-related antigens (2, 3, 7). However, the CD28 T-cell subset responsible for these effects in IM has not been identified.
The CD28-int T cells described above have these characteristic features, suggesting that they may be the cells that act as defensive effectors in EBV infection. Indeed, most of the expanded CD8 T cells are HLA-DR and CD45-RO positive, but the expression of CD28 is quite variable, and it does not follow that all of the expanded cells have cytotoxic function (3, 7). It has been reported that one-third of HLA-DR- and CD45-RO-positive cells have cytotoxic function (7). In the present paper the cytotoxic function was through nonspecific mechanisms and was detected predominantly in CD28-int T cells lacking the NK cell markers, such as CD16 and CD56. The present findings are in agreement with previous observations (23, 24, 25) and, furthermore, suggest that the effector cells are mainly a CD28-int subset. These observations may lead to the hypothesis that the expansion is not antigen-specific but rather represents standard activation and/or cross-reactive stimulation of nonspecific cells.
The massive expansion of CD8 T cells among PBMC is not specific to AIM but is a common feature during the primary response to several viral infections (19, 29). Several CD8 subsets, such as HLA-DR- and CD45RO-positive cells, are reportedly increased in AIM infection (3, 7, 27), but an increase in these markers is also observed for many other diseases, suggesting that these characteristics are not specific to AIM.
The regulation of CD28 expression is poorly understood, perhaps because murine cells do not lose CD28 when activated or energized (5). The generation of a CD28-int population has been reported only after in vitro culturing (12, 16). The emergence of a CD28-int subset has not hitherto been reported for any human disease, and the functional role of the CD28-int subset remains unknown.
During HIV and human T-cell leukemia virus infections, the expanded CD8 T cells are reportedly CD28 negative (13, 15). A massive expansion of HLA-DR- and CD45-RO-positive T cells has been confirmed during AIM infection (2, 3, 7, 20, 27), but the CD28 expression pattern remains controversial. Callan et al. reported that the levels of CD28 positivity were highly variable (3). In another study, CD28 expression was lower in AIM than in normal controls (1), and in yet another study 66% of CD8 T cells were CD28 positive (2).
The present findings suggest that the CD28-int subset is selectively expanded during AIM, and these findings are of importance for clarifying the mechanism of defense against EBV infection and the role of CD28 in humans. These findings may also be useful for assays for the diagnosis of IM based on examination for the presence of the CD28-int subset.
The origin and function of these T cells is now under investigation. Although the expansion appears to be antigen driven (2, 7, 20), its functional importance in controlling viral infection is not clear. It is likely that EBV latent or early lytic cycle antigens stimulate organic lymphoid systems (4, 6, 8). Consequently, CD8 T cells may undergo clonal and/or oligoclonal expansion (2, 3, 7, 20). It was reported that clonally expanded CD8 T cells frequently lack expression of CD28 (17, 18). Clonally expanded CD28-negative CD8 T cells have undergone many more rounds of replication than CD28-positive CD8 T cells, and this is consistent with the loss of CD28 expression (18). It is possible that during the clonal expansion of CD8 T cells, the expression of CD28 may decrease, which might facilitate the emergence of the CD28-int subset. It has been suggested that in AIM the antigenic stimulation is extremely strong and specific (4, 8, 22), and we speculate that this might result in the intermediate level of CD28 expression observed here. The CD28-int T cells expanded in AIM would function as an effector subset and would be destined to die by apoptosis. However, the massively expanded CD28-int CD8 T cells presently observed in PBMC during AIM were clearly different from the CD28-negative CD8 T cells usually observed in healthy controls, since 1 day of ex vivo culturing produced CD28-int cells that were distinguishable from CD28-negative cells, as shown in Fig. 3e and f. These findings are consistent with the idea that clonal expansion of CD8 T cells is the result of cell replication but that the expanded cells are not able to divide further (17, 18).
The ligands of CD28 are CD80 and CD86. It is known that CD28 and CD152 cross-talk with CD80 and CD86 (5, 14). This costimulatory pathway has the ability to regulate immune responses both positively and negatively. However, its physiological role in viral infection is unknown. Infection with HIV leads to constitutive expression of CD80 and CD86, concomitant with down-regulation of CD28 on T cells (13). However, there was no significant change in the expression of these other costimulatory molecules in the AIM patients we examined (Fig. 6), suggesting that the mechanism of defense against EBV infection might be regulated differently from that against HIV infection.
We hope that the present observations will provide the basis for further and more sophisticated elucidation of the immunoregulatory mechanisms of viral infections.
Acknowledgments
This work was supported by a grant from the Ministry of Health and Welfare of Japan and the Osaka Foundation for Promotion of Clinical Immunology.
We thank T. Kinoshita (Osaka University of Medical School) for comments on the manuscript. We also thank S. Hamagami (Habikino Hospital) for excellent technical support.
REFERENCES
- 1.Borthwick, N. J., M. Bofill, I. Hassan, P. Panayiotidis, G. Janossy, M. Salmon, and A. N. Akbar. 1996. Factors that influence activated CD8+ T-cell apoptosis in patients with acute herpesvirus infections: loss of costimulatory molecules CD28, CD5 and CD6 but relative maintenance of Bax and Bcl-X expression. Immunology 88:508-515. [PMC free article] [PubMed] [Google Scholar]
- 2.Callan, M. F. C., N. Steven, P. Krausa, J. D. K. Wilson, P. A. H. Moss, G. M. Gillespie, J. I. Bell, A. B. Rickinson, and A. J. McMichael. 1996. Large clonal expansions of CD8+ T cells in acute infectious mononucleosis. Nat. Med. 2:906-911. [DOI] [PubMed] [Google Scholar]
- 3.Callan, M. F. C., L. Tan, N. Annels, G. S. Ogg, J. D. K. Wilson, C. A. O'Callaghan, N. Steven, A. J. McMichael, and A. B. Rickinson. 1998. Direct visualization of antigen-specific CD8+ T cells during the primary immune response to Epstein-Barr virus in vivo. J. Exp. Med. 187:1395-1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cohen, J. I. 1999. The biology of Epstein-Barr virus: lessons learned from the virus and the host. Curr. Opin. Immunol. 11:365-370. [DOI] [PubMed] [Google Scholar]
- 5.Greenfield, E. A., K. A. Nguyen, and V. K. Kuchroo.1998. CD28/B7 costimulation: a review. Crit. Rev. Immunol. 18:389-418. [DOI] [PubMed] [Google Scholar]
- 6.Greenspan, J. S., D. Greenspan, E. T. Lennette, D. I. Abrams, M. A. Conant, V. Petersen, and U. K. Freese.1985. Replication of Epstein-Barr virus within the epithelial cells of oral “hairy” leukoplakias, an AIDS-associated lesion. N. Engl. J. Med. 313:1564-1571. [DOI] [PubMed] [Google Scholar]
- 7.Hoshino, Y., T. Morishima, H. Kimura, K. Noshikawa, T. Tsurumi, and K. Kuzushima. 1999. Antigen-driven expansion and contraction of CD8+-activated T cells in primary EBV infection. J. Immunol. 163:5735-5740. [PubMed] [Google Scholar]
- 8.Khanna, R., S. R. Burrows, and D. J. Moss.1995. Immune regulation in Epstein-Barr virus-associated diseases. Microbiol. Rev. 59:387-405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Klein, G. 1989. Viral latency and transformation: the strategy of Epstein-Barr virus. Cell 58:5-8. [DOI] [PubMed] [Google Scholar]
- 10.Koide, J., and E. G. Engleman. 1990. Differences in surface phenotype and mechanism of action between alloantigen-specific CD8+ cytotoxic and suppressor T cell clones. J. Immunol. 144:32-40. [PubMed] [Google Scholar]
- 11.Koopman, G., C. P. Reutelingsperger, G. A. Kuijten, R. M. Keehnen, S. T. Pals, and M. H. Vanoers. 1994. Annexin V for flow cytometric detection of phosphatidylserine expression on B cells undergoing apoptosis. Blood 84:1415-1420. [PubMed] [Google Scholar]
- 12.Labalette, M., E. Leteurtre, C. Thumerelle, C. Grutzmacher, B. Tourvieille, and J. P. Dessaint. 1999. Peripheral human CD8+ CD28+ T lymphocytes give rise to CD28− progeny, but IL-4 prevents loss of CD28 expression. Int. Immunol. 11:1327-1335. [DOI] [PubMed] [Google Scholar]
- 13.Lai, R. B., D. L. Rudolph, C. S. Dezzutti, P. S. Linsley, and H. E. Prince. 1996. Costimulatory effects of T cell proliferation during infection with human T lymphotropic virus type I and II are mediated through CD80 and CD86 ligands. J. Immunol. 157:1288-1296. [PubMed] [Google Scholar]
- 14.Lenschow, D. J., T. L. Walunas, and J. A. Bluestone. 1996. CD28/B7 system of T cell costimulation. Annu. Rev. Immunol. 14:233-258. [DOI] [PubMed] [Google Scholar]
- 15.Lewis, D. E., D. S. Tang, X. Wang, and C. Kozinetz. 1999. Costimulatory pathways mediate monocyte-dependent lymphocyte apoptosis in HIV. Clin. Immunol. 90:302-312. [DOI] [PubMed] [Google Scholar]
- 16.Lloyd, T. E., L. Tang, D. N. Tang, T. Bennett, W. Schober, and D. E. Lewis. 1997. Regulation of CD28 costimulation in human CD8+ T cells. J. Immunol. 158:1551-1558. [PubMed] [Google Scholar]
- 17.Monteiro, J., F. Batliwalla, H. Ostrer, and P. K. Gregersen. 1996. Shortened telomeres in clonally expanded CD28− CD8+ T cells imply a replicative history that is distinct from their CD28+ CD8+ counterparts. J. Immunol. 156:3587-3590. [PubMed] [Google Scholar]
- 18.Morley, J., F. Batliwalla, R. Hingorani, and P. K. Gregersen.1995. Oligoclonal CD8+ T cells are preferentially expanded in the CD57+ subset. J. Immunol. 154:6182-6190. [PubMed] [Google Scholar]
- 19.Pantaleo, B., J. F. Demarest, H. Soudeyns, C. Graziosi, F. Denis, J. W. Adelsberger, P. Borrow, M. S. Saag, G. M. Shaw, and R. P. Sekaly. 1994. Major expansions of CD8+ T cells with a predominant Vβ usage during the primary immune response to HIV. Nature 370:463-467. [DOI] [PubMed] [Google Scholar]
- 20.Rickinson, A. B., and D. J. Moss. 1997. Human cytotoxic T lymphocyte responses to Epstein-Barr virus infection. Annu. Rev. Immunol. 15:405-431. [DOI] [PubMed] [Google Scholar]
- 21.Saiki, O., M. Kawamoto, M. Fukuzumi, M. Kanou, and S. Utsumi. 1993. Staphylococcus aureus Wood 46 strain activates human B cells without affecting DNA synthesis or tyrosine phosphorylation. J. Immunol. 150:3224-3229. [PubMed] [Google Scholar]
- 22.Steven, N. M, N. E. Annels, A. Kumar, A. M. Leese, M. G. Kurilla, and A. B. Richinson. 1997. Immediate early and early lytic cycle proteins are frequent targets of the Epstein-Barr virus-induced cytotoxic T cell response. J. Exp. Med. 185:1605-1617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Strang, G., and B. Rickinson. 1987. Multiple HLA class I-dependent cytotoxicities constitute the “non-HLA-restricted” response in infectious mononucleosis. Eur. J. Immunol. 17:1007-1013. [DOI] [PubMed] [Google Scholar]
- 24.Svedmyr, E., and M. Jondal. 1975. Cytotoxic effector cells specific for B cell lines transformed by Epstein-Barr virus are present in patients with infectious mononucleosis. Proc. Natl. Acad. Sci. USA 72:1622-1626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taga, K., J. Chretien, B. Cherney, L. Diaz, M. Brown, and G. Tosato. 1994. Interleukin-10 inhibits apoptotic cell death in infectious mononucleosis T cells. J. Clin. Investig. 94:251-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tarner, J. E., and C. Alfieri. 1999. Epstein-Barr virus induces fas (CD95) in T cells and fas ligand in B cells leading to T cell apoptosis. Blood 94:3439-3447. [PubMed] [Google Scholar]
- 27.Uehara, T., T. Miyawaki, K. Ohta, Y. Tamaru, T. Yokoi, S. Nakamura, and N. Taniguchi. 1992. Apoptotic cell death of primed CD45RO+ T lymphocytes in Epstein-Barr virus-induced infectious mononucleosis. Blood 80:452-458. [PubMed] [Google Scholar]
- 28.Waele, M. D., C. Thielemans, and B. K. V. Camp. 1981. Characterization of immunoregulatory T cells in EBV-induced infectious mononucleosis by monoclonal antibodies. N. Engl. J. Med. 304:460-462. [DOI] [PubMed] [Google Scholar]
- 29.Williams, M. G. M, and M. M. Davis. 1995. Antigen specific development of primary and memory T cells in vivo. Science 268:106-111. [DOI] [PubMed] [Google Scholar]