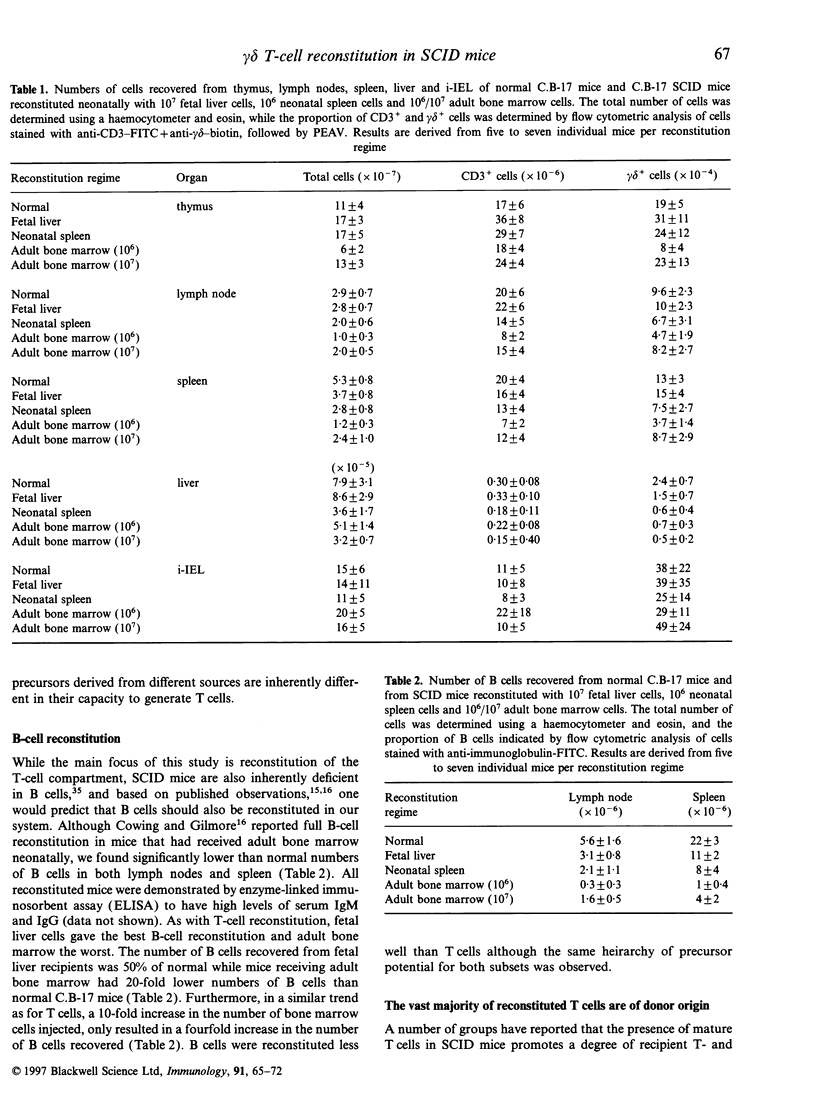
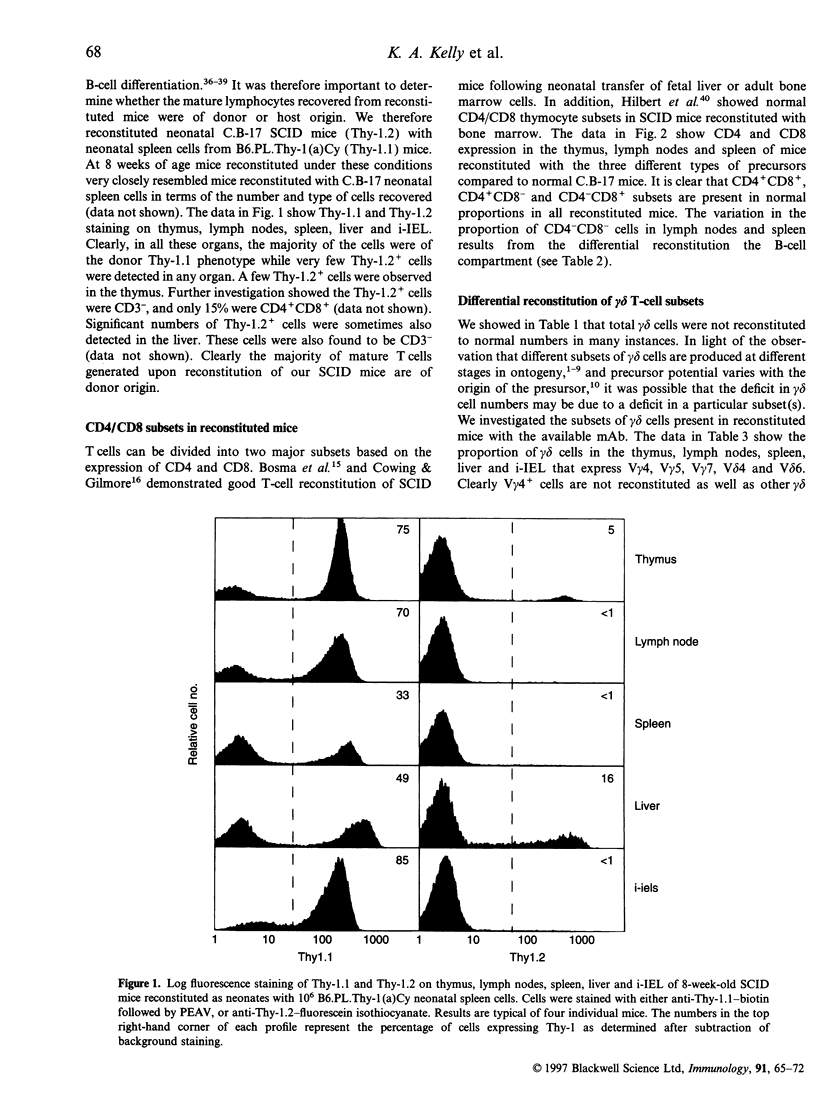
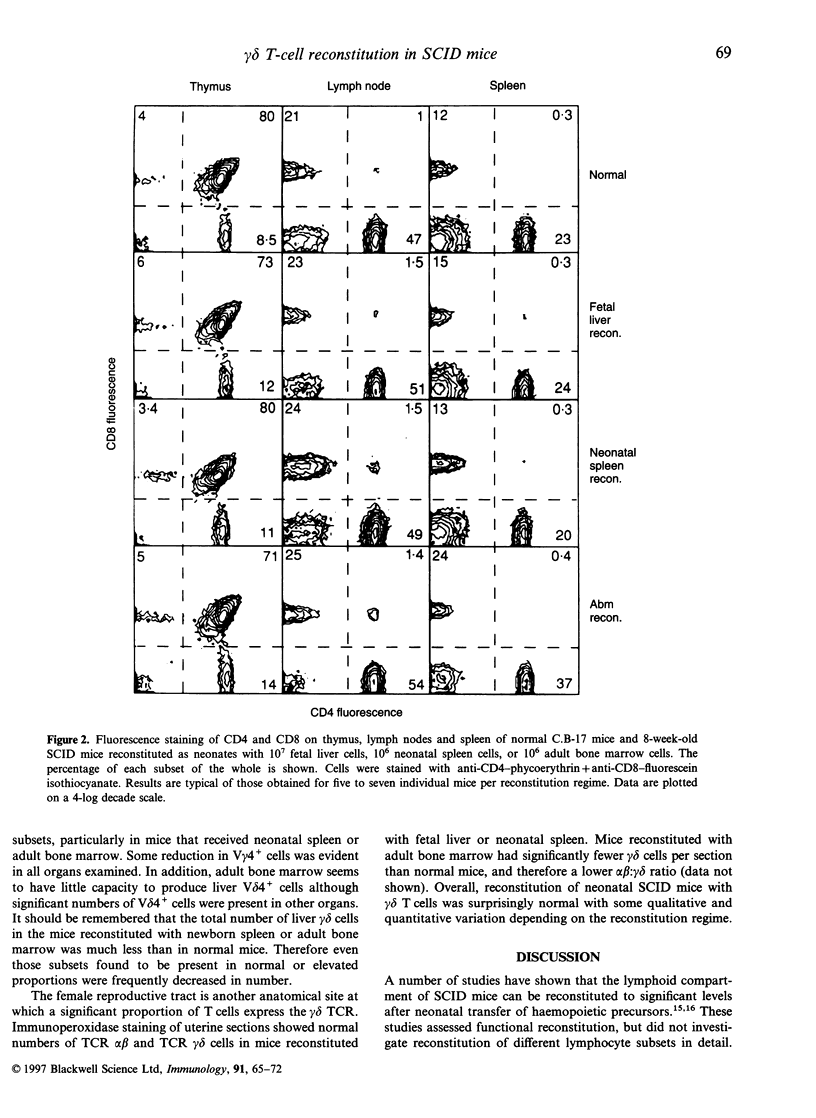
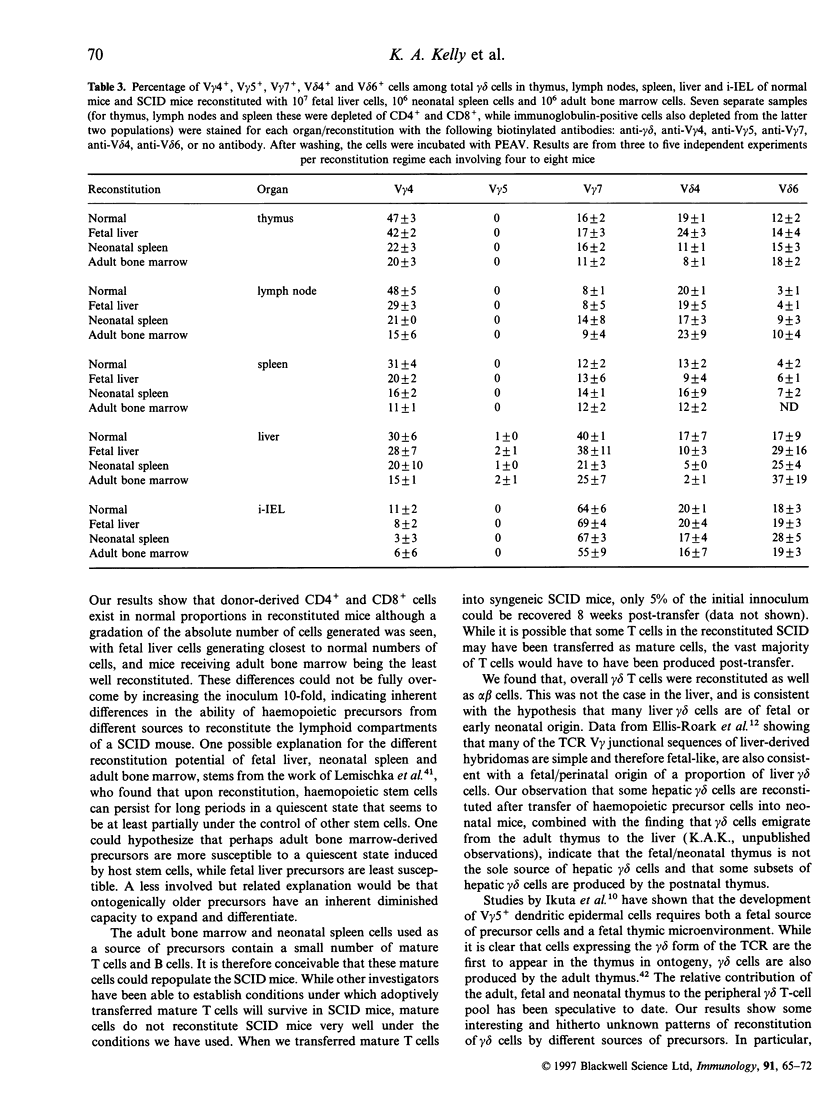
Abstract
A well-known characteristic of gamma delta T cells is that they are produced in waves during ontogeny, with cells expressing T-cell receptor V gamma 5 appearing early in fetal thymic ontogeny, followed by V gamma 6, then by other gamma delta T-cell types. In addition, evidence exists to suggest that the potential of haemopoietic precursors to generate different types of gamma delta T cells changes in ontogeny. We have used these observations as the basis for an extensive study of the potential for haemopoietic precursors isolated from fetal liver, neonatal spleen and adult bone marrow to reconstitute severe combined immunodeficient (SCID) mice. Mice that were reconstituted as newborns with fetal liver cells most closely resembled normal C.B-17 mice with respect to both lymphocyte numbers and subsets, while mice reconstituted with adult bone marrow had fewer cells than normal mice. This deficit spanned both T and B cells in all organs examined. Among the gamma delta T-cell subsets examined, the ability to reconstitute V gamma 4+ cells was particularly dependent on the ontogenic age of the reconstituting presursors, with fetal liver cells having the greatest capacity to generate V gamma 4+ cells, and adult bone marrow cells the least. The vast majority of the T cells produced in the reconstituted mice were of donor origin, and the level of reconstitution was found to be dependent upon some factor other than the presursor frequency.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Asarnow D. M., Kuziel W. A., Bonyhadi M., Tigelaar R. E., Tucker P. W., Allison J. P. Limited diversity of gamma delta antigen receptor genes of Thy-1+ dendritic epidermal cells. Cell. 1988 Dec 2;55(5):837–847. doi: 10.1016/0092-8674(88)90139-0. [DOI] [PubMed] [Google Scholar]
- Bandeira A., Itohara S., Bonneville M., Burlen-Defranoux O., Mota-Santos T., Coutinho A., Tonegawa S. Extrathymic origin of intestinal intraepithelial lymphocytes bearing T-cell antigen receptor gamma delta. Proc Natl Acad Sci U S A. 1991 Jan 1;88(1):43–47. doi: 10.1073/pnas.88.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosma G. C., Gibson D. M., Custer R. P., Bosma M. J. Reconstitution of scid mice by injection of varying numbers of normal fetal liver cells into scid neonates. Curr Top Microbiol Immunol. 1989;152:151–159. doi: 10.1007/978-3-642-74974-2_18. [DOI] [PubMed] [Google Scholar]
- Ceredig R., Lowenthal J. W., Nabholz M., MacDonald H. R. Expression of interleukin-2 receptors as a differentiation marker on intrathymic stem cells. Nature. 1985 Mar 7;314(6006):98–100. doi: 10.1038/314098a0. [DOI] [PubMed] [Google Scholar]
- Cowing C., Gilmore G. L. Allogeneic chimerism in scid mice after neonatal transfer of bone marrow. J Immunol. 1992 Feb 15;148(4):1072–1079. [PubMed] [Google Scholar]
- De Geus B., Van den Enden M., Coolen C., Nagelkerken L., Van der Heijden P., Rozing J. Phenotype of intraepithelial lymphocytes in euthymic and athymic mice: implications for differentiation of cells bearing a CD3-associated gamma delta T cell receptor. Eur J Immunol. 1990 Feb;20(2):291–298. doi: 10.1002/eji.1830200210. [DOI] [PubMed] [Google Scholar]
- Dent A. L., Matis L. A., Hooshmand F., Widacki S. M., Bluestone J. A., Hedrick S. M. Self-reactive gamma delta T cells are eliminated in the thymus. Nature. 1990 Feb 22;343(6260):714–719. doi: 10.1038/343714a0. [DOI] [PubMed] [Google Scholar]
- Dialynas D. P., Wilde D. B., Marrack P., Pierres A., Wall K. A., Havran W., Otten G., Loken M. R., Pierres M., Kappler J. Characterization of the murine antigenic determinant, designated L3T4a, recognized by monoclonal antibody GK1.5: expression of L3T4a by functional T cell clones appears to correlate primarily with class II MHC antigen-reactivity. Immunol Rev. 1983;74:29–56. doi: 10.1111/j.1600-065x.1983.tb01083.x. [DOI] [PubMed] [Google Scholar]
- Dorshkind K., Keller G. M., Phillips R. A., Miller R. G., Bosma G. C., O'Toole M., Bosma M. J. Functional status of cells from lymphoid and myeloid tissues in mice with severe combined immunodeficiency disease. J Immunol. 1984 Apr;132(4):1804–1808. [PubMed] [Google Scholar]
- Elliott J. F., Rock E. P., Patten P. A., Davis M. M., Chien Y. H. The adult T-cell receptor delta-chain is diverse and distinct from that of fetal thymocytes. Nature. 1988 Feb 18;331(6157):627–631. doi: 10.1038/331627a0. [DOI] [PubMed] [Google Scholar]
- Fazekas de St Groth B., Gallagher P. F., Miller J. F. Involvement of Lyt-2 and L3T4 in activation of hapten-specific Lyt-2+ L3T4+ T-cell clones. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2594–2598. doi: 10.1073/pnas.83.8.2594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fulop G. M., Phillips R. A. Full reconstitution of the immune deficiency in scid mice with normal stem cells requires low-dose irradiation of the recipients. J Immunol. 1986 Jun 15;136(12):4438–4443. [PubMed] [Google Scholar]
- Garman R. D., Doherty P. J., Raulet D. H. Diversity, rearrangement, and expression of murine T cell gamma genes. Cell. 1986 Jun 6;45(5):733–742. doi: 10.1016/0092-8674(86)90787-7. [DOI] [PubMed] [Google Scholar]
- Goodman T., Lefrancois L. Intraepithelial lymphocytes. Anatomical site, not T cell receptor form, dictates phenotype and function. J Exp Med. 1989 Nov 1;170(5):1569–1581. doi: 10.1084/jem.170.5.1569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Havran W. L., Allison J. P. Developmentally ordered appearance of thymocytes expressing different T-cell antigen receptors. Nature. 1988 Sep 29;335(6189):443–445. doi: 10.1038/335443a0. [DOI] [PubMed] [Google Scholar]
- Havran W. L., Allison J. P. Origin of Thy-1+ dendritic epidermal cells of adult mice from fetal thymic precursors. Nature. 1990 Mar 1;344(6261):68–70. doi: 10.1038/344068a0. [DOI] [PubMed] [Google Scholar]
- Havran W. L., Grell S., Duwe G., Kimura J., Wilson A., Kruisbeek A. M., O'Brien R. L., Born W., Tigelaar R. E., Allison J. P. Limited diversity of T-cell receptor gamma-chain expression of murine Thy-1+ dendritic epidermal cells revealed by V gamma 3-specific monoclonal antibody. Proc Natl Acad Sci U S A. 1989 Jun;86(11):4185–4189. doi: 10.1073/pnas.86.11.4185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayakawa K., Hardy R. R., Herzenberg L. A., Herzenberg L. A. Progenitors for Ly-1 B cells are distinct from progenitors for other B cells. J Exp Med. 1985 Jun 1;161(6):1554–1568. doi: 10.1084/jem.161.6.1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilbert D. M., Holmes K. L., Anderson A. O., Rudikoff S. Long-term thymic reconstitution by peripheral CD4 and CD8 single-positive lymphocytes. Eur J Immunol. 1993 Oct;23(10):2412–2418. doi: 10.1002/eji.1830231006. [DOI] [PubMed] [Google Scholar]
- Houston L. L., Nowinski R. C., Bernstein I. D. Specific in vivo localization of monoclonal antibodies directed against the Thy 1.1 antigen. J Immunol. 1980 Aug;125(2):837–843. [PubMed] [Google Scholar]
- Ikuta K., Kina T., MacNeil I., Uchida N., Peault B., Chien Y. H., Weissman I. L. A developmental switch in thymic lymphocyte maturation potential occurs at the level of hematopoietic stem cells. Cell. 1990 Sep 7;62(5):863–874. doi: 10.1016/0092-8674(90)90262-d. [DOI] [PubMed] [Google Scholar]
- Ito K., Bonneville M., Takagaki Y., Nakanishi N., Kanagawa O., Krecko E. G., Tonegawa S. Different gamma delta T-cell receptors are expressed on thymocytes at different stages of development. Proc Natl Acad Sci U S A. 1989 Jan;86(2):631–635. doi: 10.1073/pnas.86.2.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itohara S., Farr A. G., Lafaille J. J., Bonneville M., Takagaki Y., Haas W., Tonegawa S. Homing of a gamma delta thymocyte subset with homogeneous T-cell receptors to mucosal epithelia. Nature. 1990 Feb 22;343(6260):754–757. doi: 10.1038/343754a0. [DOI] [PubMed] [Google Scholar]
- Kalataradi H., Eyster C. L., Fry A., Vollmer M. K., Fu Y. X., Born W. K., O'Brien R. L. Allelic differences in TCR gamma-chains alter gamma delta T cell antigen reactivity. J Immunol. 1994 Aug 15;153(4):1455–1465. [PubMed] [Google Scholar]
- Kelly K. A., Pearse M., Lefrancois L., Scollay R. Emigration of selected subsets of gamma delta + T cells from the adult murine thymus. Int Immunol. 1993 Apr;5(4):331–335. doi: 10.1093/intimm/5.4.331. [DOI] [PubMed] [Google Scholar]
- Klein J. R. Ontogeny of the Thy-1-, Lyt-2+ murine intestinal intraepithelial lymphocyte. Characterization of a unique population of thymus-independent cytotoxic effector cells in the intestinal mucosa. J Exp Med. 1986 Jul 1;164(1):309–314. doi: 10.1084/jem.164.1.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuziel W. A., Takashima A., Bonyhadi M., Bergstresser P. R., Allison J. P., Tigelaar R. E., Tucker P. W. Regulation of T-cell receptor gamma-chain RNA expression in murine Thy-1+ dendritic epidermal cells. Nature. 1987 Jul 16;328(6127):263–266. doi: 10.1038/328263a0. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., Herzenberg L. A. Xenogeneic monoclonal antibodies to mouse lymphoid differentiation antigens. Immunol Rev. 1979;47:63–90. doi: 10.1111/j.1600-065x.1979.tb00289.x. [DOI] [PubMed] [Google Scholar]
- Lemischka I. R., Raulet D. H., Mulligan R. C. Developmental potential and dynamic behavior of hematopoietic stem cells. Cell. 1986 Jun 20;45(6):917–927. doi: 10.1016/0092-8674(86)90566-0. [DOI] [PubMed] [Google Scholar]
- Lynch F., Shevach E. M. Gamma delta T cells promote CD4 and CD8 expression by SCID thymocytes. Int Immunol. 1993 Aug;5(8):991–995. doi: 10.1093/intimm/5.8.991. [DOI] [PubMed] [Google Scholar]
- Reimann J., Rudolphi A., Claesson M. H. CD3+ T-cells in severe combined immunodeficiency (scid) mice. III. Transferred congenic, selfreactive CD4+ T cell clones rescue IgM-producing, scid-derived B cells. Int Immunol. 1991 Jul;3(7):657–663. doi: 10.1093/intimm/3.7.657. [DOI] [PubMed] [Google Scholar]
- Riggs J. E., Stowers R. S., Mosier D. E. Adoptive transfer of neonatal T lymphocytes rescues immunoglobulin production in mice with severe combined immune deficiency. J Exp Med. 1991 Jan 1;173(1):265–268. doi: 10.1084/jem.173.1.265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roark C. E., Vollmer M. K., Cranfill R. L., Carding S. R., Born W. K., O'Brien R. L. Liver gamma delta T cells. TCR junctions reveal differences in heat shock protein-60-reactive cells in liver and spleen. J Immunol. 1993 Jun 1;150(11):4867–4875. [PubMed] [Google Scholar]
- Schrader J. W., Scollay R., Battye F. Intramucosal lymphocytes of the gut: Lyt-2 and thy-1 phenotype of the granulated cells and evidence for the presence of both T cells and mast cell precursors. J Immunol. 1983 Feb;130(2):558–564. [PubMed] [Google Scholar]
- Scollay R., Shortman K. Thymocyte subpopulations: an experimental review, including flow cytometric cross-correlations between the major murine thymocyte markers. Thymus. 1983 Sep;5(5-6):245–295. [PubMed] [Google Scholar]
- Shores E. W., Sharrow S. O., Uppenkamp I., Singer A. T cell receptor-negative thymocytes from SCID mice can be induced to enter the CD4/CD8 differentiation pathway. Eur J Immunol. 1990 Jan;20(1):69–77. doi: 10.1002/eji.1830200111. [DOI] [PubMed] [Google Scholar]
- Shortman K., Williams N., Adams P. The separation of different cell classes from lymphoid organs. V. Simple procedures for the removal of cell debris. Damaged cells and erythroid cells from lymphoid cell suspensions. J Immunol Methods. 1972 May;1(3):273–287. doi: 10.1016/0022-1759(72)90005-1. [DOI] [PubMed] [Google Scholar]
- Tomonari K. A rat antibody against a structure functionally related to the mouse T-cell receptor/T3 complex. Immunogenetics. 1988;28(6):455–458. doi: 10.1007/BF00355379. [DOI] [PubMed] [Google Scholar]
- Viney J. L., MacDonald T. T., Kilshaw P. J. T-cell receptor expression in intestinal intra-epithelial lymphocyte subpopulations of normal and athymic mice. Immunology. 1989 Apr;66(4):583–587. [PMC free article] [PubMed] [Google Scholar]
- von Boehmer H., Shortman K. The separation of different cell classes from lymphoid organs. IX. A simple and rapid method for removal of damaged cells from lymphoid cell suspensions. J Immunol Methods. 1973 Apr;2(3):293–301. doi: 10.1016/0022-1759(73)90055-0. [DOI] [PubMed] [Google Scholar]



