Abstract
There is a continuing search for better ways to use existing drugs against human immunodeficiency virus (HIV). One idea is to use short therapy interruptions to “autovaccinate” HIV-infected patients. A group of 13 chronically HIV-infected patients enrolled in a trial of such so-called structured treatment interruptions (STIs) were intensively studied with respect to their viral load (VL) and HIV-specific CD8+ T-cell (cytotoxic T-lymphocyte [CTL]) responses. We found that 10 of the 13 patients had plateau VLs after STIs that were lower than their pretreatment VLs. While viral rebound rates became lower over STIs, there were no changes in clearance rates. Although numbers of CTLs did increase over the same time that viral rebounds decreased, there was no correlation between CTL count and either viral rebound rates or clearance rates. Finally, we asked whether absolute numbers of or changes in numbers of CTLs predict plateau VLs after STIs. No measure of CTLs was able to predict plateau VLs. Thus, there was no signature in these data of an important contribution to virological control from HIV-specific CD8+ T lymphocytes.
After primary human immunodeficiency virus (HIV) infection, patients' plasma viral loads (VLs) reach a set point, which is predictive of further clinical progression (19). For this reason, strategies to decrease viral set points are warranted. Recent studies suggested that this viral set point might be lowered in HIV-infected individuals by structured treatment interruptions (STIs), in which antiretroviral therapy (ART) is transiently and repeatedly discontinued with the aim of “autovaccinating” the individual through controlled exposure to autologous virus (8, 12, 23, 24, 28, 33). A decrease in viral growth kinetics over three treatment interruptions has been reported in some patients and was paralleled by increasing HIV-specific CD8+ cytotoxic T-cell (cytotoxic T-lymphocyte [CTL]) and transient HIV-specific CD4 proliferative responses. This lead to the hypothesis that STIs may enhance HIV-specific immunity, resulting in better control of viral replication on subsequent discontinuation of ART (7, 8, 16-18, 24, 33). However, at present it is still unclear to what extent frequencies of HIV-specific CD8+ T lymphocytes and CD4 proliferative responses correlate with control of viremia (2, 10, 15, 21, 24, 26, 30, 31, 34).
The plasma VL has been widely used to reveal underlying cellular events governing production and loss of HIV-infected cells. Its level in a patient is a function of the activity of all factors influencing viral replication in vivo, such as HIV-specific CD8+ cytotoxic T-cell and CD4 T-helper-cell responses, neutralizing antibodies, antiviral chemokine activity, host genetic factors such as chemokine receptor polymorphisms and HLA types, and viral factors (e.g., deletions in the nef region or coreceptor usage) (14, 36). Frequent sequential measures of plasma VL are the appropriate parameter when studying in vivo events influencing HIV replication.
A large clinical trial of STIs presented an opportunity to study 13 HIV-infected patients experiencing sequential episodes of viral growth and clearance (C. Fagard, M. LeBraz, H. F. Günthard, C. Tortajada, F. Garcia, M. Battegay, H. J. Furrer, P. Vernazza, E. Bernasconi, L. Ruiz, A. Telenti, A. Oxenius, R. E. Phillips, S. Yerly, J. M. Gatell, T. Perneger, P. Erb, L. Perrin, B. Hirschel, and the Swiss HIV Cohort Study, 8th Conf. Retroviruses Opportunistic Infect., abstr. G16e, 2001). The trial's protocol consisted of four cycles of STI (2 weeks off and 8 weeks on ART), and then, after 40 weeks, treatment was stopped completely. Intensive serial sampling from these patients allowed simultaneous quantification of their HIV VLs and HIV-specific CD8+ cells. This unique set of data allowed the first detailed comparison of patients' CD8+ T-cell responsiveness with their viral rebound rate upon interruption of therapy. Several important questions were addressed. (i) Do STIs lead to decreased VLs compared with pretreatment levels? (ii) Do viral rebound rates change with STIs? (iii) Do viral clearance rates change with STIs? (iv) Do STIs lead to increased numbers of circulating HIV-specific CD8+ T cells? (v) Do numbers of circulating HIV-specific CD8+ T cells correlate with viral rebound rates upon interruption of therapy? (vi) Do numbers of circulating HIV-specific CD8+ T cells correlate with viral clearance rates upon restarting therapy? (vii) Do absolute numbers of or changes in numbers of HIV-specific CD8+ T cells predict plateau VL after STIs?
The combination of a set of patients involved in a trial of STIs with very frequent quantification of their VLs and CTL populations gave a unique opportunity to address fundamental questions in HIV biology.
(This work was presented in part at the 9th Conference on Retroviruses and Opportunistic Infections, Seattle, Wash., 24 to 28 February 2002 [abstr. 495-M].)
MATERIALS AND METHODS
Patients and study design.
Fifteen of 29 patients enrolled from the Zurich cohort of the Swiss-Spanish Intermittent Therapy Trial (SSITT) (Fagard et al., 8th Conf. Retroviruses Opportunistic Infect.) participated in an extended-study protocol with frequent blood sampling. Thirteen patients with sufficient time points were eligible for mathematical analysis. Baseline characteristics are summarized in Table 1. To be eligible for the SSITT, patients had to be on combination ART with two or three drugs (nonnucleoside reverse transcription inhibitors excluded) for >6 months, the plasma viremia had to be <50 HIV RNA copies/ml for >6 months, and the CD4+ T-cell count had to exceed 300 cells/ml. Twelve patients started treatment during chronic HIV infection with one protease and two nucleoside reverse transcriptase inhibitors (Table 1), whereas patient 1 was treated with zidovudine-lamivudine alone for 5 months before indinavir was added. Subjects with change of treatment because of virological failure were excluded. The trial consisted of four cycles of a 2-week treatment interruption (STI) followed by 8 to 10 weeks of retreatment. Before therapy was stopped again, plasma viremia had to reach <50 HIV RNA copies/ml. After these four cycles, at week 40, treatment was stopped for ≥3 months unless symptoms of acute HIV infection occurred, the CD4 count dropped to <300 CD4 cells/ml, or the VL exceeded the following predetermined values: three consecutive measurements of >50,000/ml, two of >100,000/ml, or one of >500,000/ml. Between weeks 40 and 52, treatment had to be reinitiated in patients 1, 3, and 6. At week 52, three patients had viremia of below 5,000 copies/ml. The other eight patients did not resume treatment despite a VL of >5,000 copies/ml because the CD4 count was stable and did not warrant immediate reintroduction of therapy according to new guidelines for antiretroviral treatment (27, 38). In none of the 13 patients was ART started during primary HIV infection. Written informed consents for the SSITT and for the extended protocol were obtained separately from each patient.
TABLE 1.
Patient characteristics
| Patient no. | Age (yr) | HIV subtypea | Time virus undetectable before STI (mo) | Before ART
|
Baseline of STI trial
|
Wk 52 HIV RNA (copies/ml) | ||
|---|---|---|---|---|---|---|---|---|
| Plasma HIV RNA (copies/ml) | CD4 cell count (cells/mm3) | CD4 cell count (cells/mm3) | ARTd | |||||
| 1 | 41 | B | 32 | 561,831 | 280 | 723 | ZDV-3TC-IDV | —e |
| 2 | 52 | B | 26 | 128,555c | 794 | 1,269 | ZDV-3TC-IDV | 41,316 |
| 3 | 38 | B | 31 | 34,752 | 1,010 | 1,115 | ZDV-3TC-RTV | — |
| 4 | 38 | B | 11 | 122,902c | 259 | 422 | DDI-D4T-NFV | 19,127 |
| 5 | 46 | B | 25 | 32,608c | 388 | 347 | ZDV-3TC-IDV | 8,757 |
| 6 | 53 | B | 32 | 467,593 | 11 | 350 | ZDV-3TC-IDV | — |
| 7 | 33 | B | 30 | 16,927 | 573 | 832 | ZDV-3TC-IDV | 4,560 |
| 8 | 55 | B | 28 | 162,701c | 461 | 766 | ZDV-3TC-IDV | 21,848 |
| 9 | 38 | B | 12 | 164,772 | 272 | 591 | D4T-3TC-NFV | 51,008 |
| 10 | 35 | E/CRF1b | 23 | 11,298 | 495 | 777 | DDI-D4T-NFV | 3,466 |
| 11 | 52 | B | 22 | 25,417 | 302 | 839 | D4T-3TC-NFV | 189,929 |
| 12 | 43 | B | 25 | 9,404 | 327 | 749 | ZDV-DDI-NFV | 24,587 |
| 13 | 68 | A | 31 | 821c | 650 | 670 | ZDV-3TC-NFV | 286 |
| Mean (range) | 46 (33-68) | 25 (11-32) | 133,814 (821-561,831) | 448 (11-1,010) | 739 (347-1,269) | 36,488 (286-189,929) | ||
Determined as described previously (39).
CRF1, circulating recombinant form 1.
Geometric mean, because two pretreatment HIV RNA values were available before initiation of ART.
ZDV, zidovudine; 3TC, lamivudine; IDV, indinavir; DDI, didanosine; RTV, ritonavir; D4T, stavudine; NFV, nelvinafir.
—, treatment had to be reinitiated between weeks 40 and 52.
Blood sampling.
Blood samples were obtained on days 0, 4, 8, 14, 18, 22, 24, 28, 35, 42, 56, and 70 of each cycle; every week from week 40 to 52; and every 1 to 2 weeks to week 64. Blood was processed within 2 h after blood was drawn.
HIV quantitation.
Plasma HIV RNA was quantified by using the Amplicor HIV-1 Monitor test, version 1.5 (Roche, Rotkreuz, Switzerland) with ultrasensitive modifications resulting in a level of detection of 50 HIV RNA copies/ml or lower (6, 35). To minimize assay variability, for each patient, batchwise HIV RNA testing was performed on frozen (−80°C) plasma samples for each complete cycle (2 weeks off and 8 weeks on ART).
IFN-γ ELISPOT.
For analysis of HIV-specific CD8+ T-cell frequencies, peripheral blood lymphocytes (PBL) were stimulated directly ex vivo with synthetic peptides corresponding to previously described HLA class I-restricted optimal CTL epitopes at a concentration of 2 μM in gamma interferon (IFN-γ) ELISPOT assays (25, 26). Results are expressed as spot-forming cells (SFC) per 106 PBL; background values were subtracted from the specific response before normalization to 106 PBL. All assays were performed in duplicate.
HLA typing.
The HLA class I and II genotypes of each patient were determined by PCR using sequence-specific primers (4).
Statistical analyses.
For each patient, 15 viral life history parameters were calculated (see Appendix for an explanation of terms): the pretreatment VL, the post-STI plateau VL, five upslopes for the five events of cessation of therapy, four fast downslopes for the first-phase decay upon restarting therapy, and four slow downslopes for the second-phase decay upon the restarting of therapy.
The pretreatment VL corresponded to the last plasma HIV RNA value recorded before any ART was started, or to the geometric mean of the last two such values when both were obtained in the last 6 months before start of ART. The post-STI plateau VL was the geometric mean of VLs in weeks 46 to 64 (or the first data points thereafter) for the majority of patients. However, three patients (patients 1, 3, and 6) restarted therapy before week 50 and had plateau VLs calculated from the two or three time points just prior to restarting therapy. In two patients (patients 2 and 13), the week 46 data point was omitted from the estimation of the plateau because it clearly represented part of a peak. For the 10 patients who remained off therapy for extended periods, an average of 9.4 data points were used to calculate the plateau VL (range, 7 to 12).
Rates of increase and decrease in VL were calculated from the natural logarithm of the RNA data. Upslopes were calculated from day 0 to 14 off therapy. The first-phase, fast downslopes were from day 0 to 14 on therapy. The second-phase, slow downslopes were from day 15 to 56 on treatment. In some patients, the VL fell below the threshold of detection. In all such cases, an estimated limit of detection was available. When runs of undetectable virus were present in the data, only the first or last such data point was used, and it was set to one below the limit of detection. This compromise allowed us to use the information that undetectable VLs are clearly very low, but it avoids systematic bias that would lead to underestimates of slopes if more than one undetectable VL is set to just below the limit of detection.
Tests to search for trends in slopes over the rounds of STI were performed by using tests for homogeneity of slopes in general linear models (37). Such statistical tests address the question of whether the slopes are different during different rounds. Models took the general form log10 RNA = patient identification number + round + day of cycle + patient identification number × day of cycle + round × day of cycle. These tests were performed separately for RNA values from the days off therapy (upslopes) and the two phases on therapy (fast and slow downslopes). The day of cycle was calculated to count days since the relevant change in therapy (starting or stopping therapy for up- and downslopes, respectively). In these analyses, the round and day of cycle were treated as covariates.
To test whether absolute HIV-specific CD8+ T-cell counts were predictive of viral up- and downrates during STIs, we performed tests for homogeneity of slopes (37). This set of statistical tests ask if changes in slopes can be explained by different CTL counts. Models took the form log10 RNA = patient identification number + CTL + day of cycle + patient identification number × day of cycle + CTL × day of cycle, where CTL was either the total number of SFC or the number of epitope-specific responses per patient at the beginning of the time period in question. In these analyses, CTL and day of cycle were treated as covariates.
All statistical analyses were performed using the statistical package Minitab, release 12 (Minitab Inc., State College, Pa.).
RESULTS
Virological analysis. (i) Patients.
All 13 patients underwent four STI cycles (2 weeks off therapy followed by 8 weeks on therapy). At week 40, treatment was stopped in all subjects, but it was restarted in patient 6 at week 45, patient 1 at week 47, and patient 3 at week 50 because of a rapid drop of CD4 counts in the first patient; low-grade fevers, night sweats, and symptomatic lymphadenopathy in the second patient; and symptomatic lymphadenopathy in the third patient. At week 52, 3 of the 13 patients (patient 7, 4,560 copies/ml; patient 10, 3,466 copies/ml; patient 13, 286 copies/ml) controlled plasma viremia at levels of <5,000 copies/ml. The other seven untreated patients showed higher levels of HIV RNA (8,757 to 189,929 copies/ml) but did not want to resume ART because CD4 counts were stable at levels which, according to new guidelines and studies (27, 29, 38), did not warrant immediate antiretroviral treatment.
(ii) Plateau VLs after four STIs were lower than HIV RNA levels before ART.
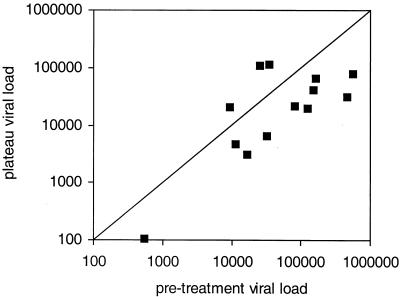
To test whether STIs have an impact on individual viral set points, we compared pretreatment VLs with plateau VLs reached after having stopped ART at week 40. Ten of 13 patients showed a reduction in plateau VLs compared with pretreatment VLs (Fig. 1). Overall plateau VLs were 0.4-log-unit lower than pretreatment VLs, at a mean of 4.2 log units compared to a mean of 4.6 log units (P = 0.01 in a one-sided paired t test).
FIG. 1.
For each individual patient, the plateau VL is shown as a function of the pretreatment VL. Both axes show plasma HIV RNA levels as copies per milliliter of plasma. The line dividing the square indicates parity. The plateau VLs for 10 patients are below parity, indicating a decrease in viral replication compared with pretreatment VLs. In three patients, the plateau VLs lie above parity, indicating higher viral replication compared to pretreatment VLs.
(iii) Plasma viral rebound during STIs.
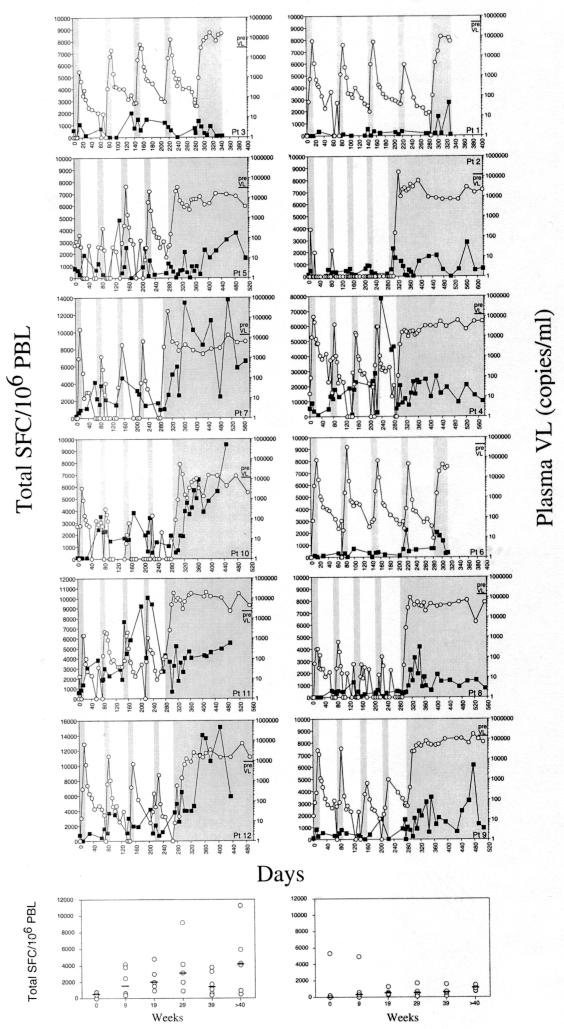
Plasma virus became detectable (>50 HIV RNA copies/ml) in 11 of the 13 patients during the 2-weeks interruption, but only once during the first interruption in patient 2 (see Fig. 3) and not at all in patient 13. However, when therapy was stopped at week 40 for at least 3 months, these two patients showed viral rebounds at weeks 44 and 46, respectively. To ensure that drugs were not taken during STIs, plasma protease inhibitors levels were randomly determined for each patient once during one STI, and all were negative (data not shown).
FIG. 3.
Individual plasma HIV RNA levels and HIV-specific CTLs in 12 patients during STIs. The courses of plasma HIV RNA and of HIV-specific CTLs are demonstrated for 12 patients with HIV subtype B individually. Each of the four 2-week treatment interruptions is marked by a grey bar. Intervals of retreatment are marked as white areas. Graphs on the left represent patients with pretreatment VLs below the median of all pretreatment VLs. Graphs on the right indicate patients with pretreatment VLs above the median. The left y axis reflects total SFC per 106 PBL and the right y axis represents HIV RNA copies per milliliter of plasma. Data for patient 13 are not shown because CTL responses could not be determined due to infection with HIV subtype A.
(iv) Uprates during viral rebound decrease over cycles.
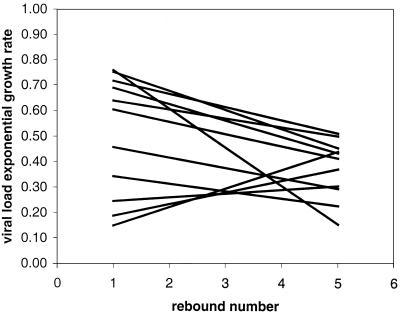
From 11 patients, 5 uprates were available for comparison of changes over time. These five uprates included the four 2-week interruptions and the first 2-week off period after week 40. A formal test for changes in upslopes with round of STI was performed using RNA data from days 0 to 14 off therapy in a test for homogeneity of slopes, with log10 VL as the response variable, day of cycle and round as covariates, and patient number as a factor. The interaction of round and day of cycle was significant, as was the interaction of patient number and day. This analysis indicated that different patients have significantly different upslopes and that overall the slopes decrease significantly with round (P = 0.039).
Figure 2 represents these trends in growth rates with one regression line for each of the 11 patients in whom HIV rebounded during STIs. In eight patients exponential growth rates decreased, and in three patients they increased. In Table 2 doubling times for each patient and each round are given numerically. They are in the same range as described previously (7, 9, 33).
FIG. 2.
Slope of the rebound rate for each patient. Trends of VL exponential growth rates during STI cycles are expressed by linear regression lines fitted for each of the 11 patients presenting viral rebounds during STI cycles through the five VL exponential growth rates, which were determined for each treatment interruption: first, second, third, and fourth STIs and first 2 weeks of the final cessation.
TABLE 2.
Doubling times of plasma HIV RNA during STIs and first- and second-phase decay half-lifes after resumption of treatment
| Patient no. | VL (copies/ml)
|
Doubling time (days) during STI:
|
First-phase decay half-life (days) after STI:
|
Second-phase decay half-life (days) after STI:
|
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pretreatment | Plateau | 1 | 2 | 3 | 4 | 5 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | |
| 1 | 561,831 | 76,805 | 1.18 | 1.24 | 1.15 | 2.10 | 1.49 | 1.48 | 1.42 | 1.52 | NAa | 15.53 | 18.85 | 27.31 | 15.11 |
| 2 | 80,600 | 21,393 | NA | NA | NA | NA | 1.33 | NA | NA | NA | NA | NA | NA | NA | NA |
| 3 | 34,752 | 115,546 | 0.78 | 1.31 | 1.26 | 1.33 | 1.33 | 3.06 | 1.98 | 2.26 | 1.77 | 16.32 | 16.35 | 10.57 | 15.19 |
| 4 | 122,729 | 19,344 | 1.04 | 1.21 | 1.25 | 1.29 | 1.35 | 2.26 | 1.23 | 2.21 | 1.93 | 12.06 | 8.80 | 10.05 | 11.14 |
| 5 | 32,140 | 6,459 | 10.26 | 4.89 | 1.52 | 1.13 | 3.56 | 2.10 | 1.42 | 1.49 | 1.78 | NA | NA | 18.19 | 14.01 |
| 6 | 467,598 | 31,500 | 1.06 | 0.85 | 1.28 | 1.38 | 1.25 | 1.61 | 1.18 | 1.59 | 1.26 | 13.12 | 10.92 | 14.83 | 10.65 |
| 7 | 16,927 | 3,140 | 0.95 | 1.01 | 1.99 | 1.21 | 1.48 | 1.16 | 1.94 | NA | 1.07 | 1,664.09 | 6.05 | NA | 14.08 |
| 8 | 150,390 | 40,176 | 3.67 | 1.74 | 8.00 | 3.31 | 1.35 | 5.63 | 2.05 | 14.27 | 1.88 | 32.94 | −95.31 | 11.20 | NA |
| 9 | 164,772 | 65,941 | 1.38 | 1.46 | 3.18 | 2.36 | 1.78 | 1.94 | 1.02 | 17.69 | NA | 9.70 | 18.88 | 12.40 | 11.76 |
| 10 | 11,298 | 4,623 | 1.97 | 7.86 | 3.67 | 1.26 | 3.58 | 2.00 | 2.13 | 1.38 | 1.24 | −48.28 | NA | NA | NA |
| 11 | 25,417 | 106,928 | 2.01 | 2.15 | 2.73 | 2.46 | 3.16 | 2.22 | 8.48 | 1.98 | 3.54 | −72.05 | 19.87 | −20.33 | −57.25 |
| 12 | 9,404 | 20,236 | 1.00 | 1.25 | 0.99 | 2.86 | 7.65 | 1.17 | 1.37 | NA | 1.88 | 14.87 | 13.17 | 21.65 | 21.15 |
| 13 | 537 | 105 | NA | NA | NA | NA | 1.86 | NA | NA | NA | NA | NA | NA | NA | NA |
| Mean | 2.30 | 2.27 | 2.46 | 1.88 | 2.40 | 2.24 | 2.20 | 4.93 | 1.82 | 165.83 | 1.96 | 11.76 | 6.20 | ||
NA, not available (either no rebound was observed during the 2-week STI or not enough data points were available to calculate first- and second-phase half-lives).
(v) Downrates after reinitiation of ART do not change over cycles.
Neither high downrates (first-phase decay, days 0 to 14 on therapy) nor low downrates (second-phase decay, days 15 to 56 on therapy) showed significant changes with round or with patient in tests for homogeneity of slopes. This implied that there were no systematic changes in these clearance rates over the cycles of STIs. First-phase half-lives, as calculated during the initial 2 weeks after reinitiation of treatment after an STI, and second-phase half-lives, reflecting the decay of plasma HIV RNA from week 2 up to week 8 of retreatment after STI, are shown in Table 2. Overall, the first- and second-phase decay rates were in the range previously described (13, 39).
Immunological analysis. (i) HIV-specific cytotoxic CD8+ T-cell responses were strongly enhanced by STI.
HIV-specific CD8+ T-cell responses were serially measured by IFN-γ ELISPOT analysis. Each patient was HLA typed, and, according to the HLA class I genotype, a series of previously defined optimal CTL epitopes were screened at each time point. PBL samples were analyzed at baseline, at multiple time points (median, 18; range, 15 to 20) during STI and resumption of ART, and at multiple time points (median, 18; range, 6 to 21) after final cessation of therapy (week 40). A median of 18 (range, 7 to 29) peptide epitopes were screened per patient.
The total SFC (defined as the sum of individual epitope-specific responses) and numbers of recognized epitopes increased significantly during STIs. Over the four treatment rounds, the total SFC increased by a mean of 500 SFC/106 PBL per round and patient (P = 0.026, excluding one extreme outlier in a linear regression of total SFC against round number), and the number of epitopes recognized increased by 0.5 response every round (P = 0.002 in a regression of number of responses against round number). The correlation between change in total SFC and the change in numbers of epitopes recognized after STIs was very high (r2 = 0.73; P = 0.0004). There were further increases once patients ceased therapy. Responses measured at or near week 52 were significantly larger and broader than average responses during the STIs. Total SFC increased by a median of 448/106 PBL (range, −530 to 11,602), and the number of responses increased by a median of 1 extra response (range, 1 to 7) when week 39 was compared with week 52. The dynamics of VL rebound, decay, and HIV-specific CTL responses are given for each patient in Fig. 3.
(ii) Do CTLs correlate with viral rebound and clearance rates?
To investigate the relationship between CTL response and viral rebound and clearance rates, we performed tests for homogeneity of slopes, searching for an effect of CTL responses on uprates when off therapy and downrates when on therapy. No such effects could be detected.
(iii) Post-STI VL plateaus do not correlate with changes in CTLs.
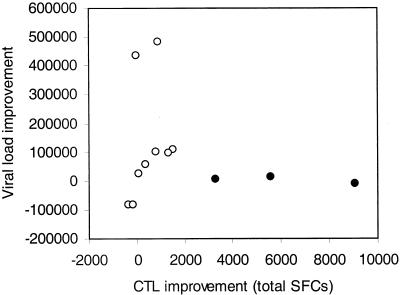
Figure 4 depicts the relationship between the difference of pretreatment VL and post-STI VL plateaus and change in CTL total SFC for each patient between baseline and week 52. There is no correlation between these two measures of viral and immune system changes. It may be of interest that the three patients with the lowest pretreatment VLs on this graph are the three who, despite large gains in CTL responses, did not show a matching large improvement in control of viremia. (Patient 13, who showed the lowest pretreatment and also the lowest VL plateau, could not be included in this analysis because no CTL response at all was detected, most likely because the virus was subtype A.)
FIG. 4.
No correlation between changes in VL and changes in immune response. This figure demonstrates a nonlinear relationship between VL improvement (y axis) (difference between VL plateau after final cessation of ART and pretreatment VL) and CTL improvement (x axis) (difference between the mean of the total number of SFC after cessation of therapy and the total SFC at baseline of the first STI). Closed circles depict data for three patients with the strongest increase of CTL responses but no improvement in VL.
To further investigate the hypothesis that pretreatment VL might be predictive of the size of the CTL response, we stratified the patients according to pretreatment VLs. Patients with pretreatment VLs below the median pretreatment VL of 78,827 HIV RNA copies/ml showed a tendency towards stronger CTL responses over the interruption cycles, expressed as total SFC (Fig. 3), but statistical significance was not achieved.
DISCUSSION
Repeated STIs have been associated with an increase in viral doubling time and with a reduction of plasma HIV RNA levels after final cessation of therapy in some patients enrolled in various studies (3, 7-9, 23, 24, 28, 33). It has been hypothesized that such reduction in viral replication was causally related to the induction of HIV-specific CD8+ T-cell responses (8, 31, 33). Yet, in our study, despite a unique density of virological and immunological data allowing detailed analysis and long-term follow-up, no direct relationship between changes in viral rebound and clearance rates and HIV-specific CTL responses could be established.
In the 13 patients studied we found a significant decrease in plateau VLs compared with pretreatment HIV plasma RNA levels (Fig. 1). A reduction in viral replication rate was also observed as a decrease in the viral exponential growth rates over STI cycles (Fig. 2). We then investigated whether reductions in VL plateaus were correlated with changes in frequencies of HIV-specific CD8+ T lymphocytes. Total SFC and the number of recognized epitopes increased per cycle and were highest after week 40, when patients were exposed to HIV antigens for a prolonged period of time. Yet, the increase of HIV-specific CD8+ T cells correlated neither with the lowering of uprates during STIs nor with VL plateaus after cessation of therapy after week 40. Similarly, no correlations could be found between the change in numbers of recognized epitopes, viral dynamics, and VL plateau. However, comparison of changes of the magnitude of HIV-specific CD8+ T cells and changes in VL plateau, as depicted in Fig. 4, suggests that there is a nonlinear relationship between increase in CTL activity and VL: the three patients (patients 7, 10, and 12) who were able to mount the strongest CTL responses over the STI cycles were also the patients who controlled their viruses at the lowest levels before starting ART (Table 1). A further stratification of patients with regard to their pretreatment VL, however, did not reveal a significantly stronger CTL response in patients with lower pretreatment VLs.
The fact that we could not demonstrate a linear relationship between frequencies of HIV-specific CD8+ T lymphocytes and an inhibitory effect on viral replication is in conflict with published reports which have speculated that increases in HIV-specific CTLs would result in a reduction of viral rebound and VL plateaus (7, 8, 33) and which have described an inverse relationship between HIV-specific T-cell responses and plasma HIV RNA levels in untreated patients (22). What could explain such discrepancies? First, our study is different from others in that it is a long-term longitudinal study with a uniquely high density of virological and immunological data for each patient. Thus, limitations of cross-sectional studies could be eliminated; longitudinal data at such a high frequency allow each patient to serve as his own control with regard to antigen-CTL interaction. Second, methodological differences may potentially be of importance. Measuring the frequency of IFN-γ-producing CD8 cells stimulated with a panel of synthetic peptides against HLA class I-restricted optimal CTL epitopes allowed us to determine the frequencies of many more HIV-specific CD8+ T cells, directed to a whole range of CTL epitopes, than would be possible with tetramer technology. At the same time, quantification of antigen-specific T-cell responses to single peptides by these two technologies correlate well (11, 20). Furthermore, Betts et al. used overlapping peptide pools encompassing all products of the HIV-1 genome in 23 untreated patients and did not find any inverse correlation but rather a positive correlation between HIV-specific CD8+ T-cell frequencies and plasma HIV RNA levels (2). Third, discrepancies in the function of effector cells may also have accounted for the differences: in our study, we did not perform assays determining direct antiviral effector function such as cytotoxicity or chemokine release (1, 2), nor did we frequently measure T-helper-cell proliferative responses, which have been associated with control of HIV replication in untreated patients (31, 32).
Taken together, neither absolute frequencies nor changes in frequencies of HIV-specific CD8+ T cells as measured in this study were able to explain significant changes in viral dynamics observed during STI in humans. In theory, limited target cell availability induced by repeated STIs could result in a decrease of viral uprates. However, total CD4 T-cell counts were remarkably stable over the four STIs. Moreover, HIV-specific CD4+ T-cell frequencies, representing a preferential target cell population for HIV infection (5), did not decrease over the four STIs but rather increased. HIV-specific CD4+ T-cell frequencies were measured by IFN-γ ELISPOT assay at weeks 0 and 39 (data not shown). Thus, factors which serve as correlates of protection in chronically HIV-infected patients remain to be identified.
Finally, it must be clearly stated that despite statistical significance, the virological effects induced in our STI study do not support clinical relevance of the autovaccination hypothesis, and similar conclusions have been reached for the entire SSITT (Fagard et al., 8th Conf. Retroviruses Opportunistic Infect.).
In summary, STIs resulted in reduced uprates of viral rebound and were followed by decreased plateau VLs compared to pretreatment VLs. Yet, these improvements in VL were modest and of unknown clinical relevance. Significant increases in HIV-specific CTL responses were observed but correlated neither with changes in up- and downrates of viral growth and decay nor with plateau VLs post-STI. These results suggest that factors other than CTLs as measured by IFN-γ ELISPOT were responsible for the decreases in viral replication observed in these patients.
Acknowledgments
We are grateful to the patients who participated in this very demanding study for their contribution to the better understanding of HIV pathogenesis. Moreover, we thank Friederike Burgener, Erika Schlaepfer-Nadal, Esther Beerli, Herbert Kuster, and Doris Russenberger for prompt and high-quality processing of clinical samples and Alexandra Trkola, Joseph Wong, and Beda Joos for stimulating discussions and review of the manuscript.
This study has been financed in the framework of the Swiss HIV Cohort Study, supported by the Swiss National Science Foundation (grant 3345-062041), and was also supported by SHCS grant 290 (to H.F.G.), by SNF grant 3345-65168.01 (to H.F.G.), by a research grant from the Kanton of Zürich, and by the Schweizerische Stiftung für medizinisch-biologische Stipendien and the Novartis Foundation (to A.O.), the Wellcome Trust (to R.E.P. and S.J.D.), and the Medical Research Council (to D.A.P.).
APPENDIX
The following terms describing viral kinetics are used in this paper.
Upslope (uprate). The upslope (uprate) is the linear regression fitted through plasma HIV RNA levels arising during 2-week treatment interruptions.
Fast downslope (fast downrate). The fast downslope (fast downrate) is the linear regression fitted through plasma HIV RNA levels measured during the first 2 weeks after ART was resumed. This describes first-phase decay of plasma virus.
Slow downslope (slow downrate). The slow downslope (slow downrate) is the linear regression fitted through plasma HIV RNA levels from week 2 to 10 after treatment was resumed. This describes second-phase decay of plasma virus.
Viral set point. The viral set point is the plasma HIV RNA plateau reached by an individual after primary HIV infection. It reflects the steady state of HIV replication in a person after primary HIV infection and is predictive for further clinical progression.
Pretreatment VL. The pretreatment VL is the last pretreatment plasma HIV RNA value before ART or, if two measurements within 6 months before initiation of ART were available, the geometric mean of those levels.
VL plateau. The VL plateau is the geometric mean of plasma HIV RNA levels measured after cessation of therapy, when a steady state was reached. For most patients, this was between weeks 46 and 64.
REFERENCES
- 1.Appay, V., D. F. Nixon, S. M. Donahoe, G. M. Gillespie, T. Dong, A. King, G. S. Ogg, H. M. Spiegel, C. Conlon, C. A. Spina, D. V. Havlir, D. D. Richman, A. Waters, P. Easterbrook, A. J. McMichael, and S. L. Rowland-Jones. 2000. HIV-specific CD8(+) T cells produce antiviral cytokines but are impaired in cytolytic function. J. Exp. Med. 192:63-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Betts, M. R., D. R. Ambrozak, D. C. Douek, S. Bonhoeffer, J. M. Brenchley, J. P. Casazza, R. A. Koup, and L. J. Picker. 1991. 2001. Analysis of total human immunodeficiency virus (HIV)-specific CD4+ and CD8+ T-cell responses: relationship to viral load in untreated HIV infection. J. Virol. 75:11983-11991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Binley, J. M., D. S. Schiller, G. M. Ortiz, A. Hurley, D. F. Nixon, M. M. Markowitz, and J. P. Moore. 2000. The relationship between T cell proliferative responses and plasma viremia during treatment of human immunodeficiency virus type 1 infection with combination antiretroviral therapy. J. Infect. Dis. 181:1249-1263. [DOI] [PubMed] [Google Scholar]
- 4.Bunce, M., C. M. O'Neill, M. C. Barnardo, P. Krausa, M. J. Browning, P. J. Morris, and K. I. Welsh. 1995. Phototyping: comprehensive DNA typing for HLA-A, B, C, DRB1, DRB3, DRB4, DRB5 & DQB1 by PCR with 144 primer mixes utilizing sequence-specific primers (PCR-SSP). Tissue Antigens 46:355-367. [DOI] [PubMed] [Google Scholar]
- 5.Douek, D. C., J. M. Brenchley, M. R. Betts, D. R. Ambrozak, B. J. Hill, Y. Okamoto, J. P. Casazza, J. Kuruppu, K. Kunstman, S. Wolinsky, Z. Grossman, M. Dybul, A. Oxenius, D. A. Price, M. Connors, and R. A. Koup. 2002. HIV preferentially infects HIV-specific CD4+ T cells. Nature 417:95-98. [DOI] [PubMed] [Google Scholar]
- 6.Fischer, M., W. Huber, A. Kallivroussis, P. Ott, M. Opravil, R. Luthy, R. Weber, and R. W. Cone. 1999. Highly sensitive methods for quantitation of human immunodeficiency virus type 1 RNA from plasma, cells, and tissues. J. Clin. Microbiol. 37:1260-1264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Frost, S. D., J. Martinez-Picado, L. Ruiz, B. Clotet, and A. J. Leigh Brown. 2002. Viral dynamics during structured treatment Interruptions of chronic human immunodeficiency virus type 1 infection. J. Virol. 76:968-979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Garcia, F., M. Plana, G. M. Ortiz, S. Bonhoeffer, A. Soriano, C. Vidal, A. Cruceta, M. Arnedo, C. Gil, G. Pantaleo, T. Pumarola, T. Gallart, D. F. Nixon, J. M. Miro, and J. M. Gatell. 2001. The virological and immunological consequences of structured treatment interruptions in chronic HIV-1 infection. AIDS 15:F29-F40. [DOI] [PubMed] [Google Scholar]
- 9.Garcia, F., M. Plana, C. Vidal, A. Cruceta, W. A. O'Brien, G. Pantaleo, T. Pumarola, T. Gallart, J. M. Miro, and J. M. Gatell. 1999. Dynamics of viral load rebound and immunological changes after stopping effective antiretroviral therapy. AIDS 13:F79-F86. [DOI] [PubMed] [Google Scholar]
- 10.Gea-Banacloche, J. C., S. A. Migueles, L. Martino, W. L. Shupert, A. C. McNeil, M. S. Sabbaghian, L. Ehler, C. Prussin, R. Stevens, L. Lambert, J. Altman, C. W. Hallahan, J. C. de Quiros, and M. Connors. 2000. Maintenance of large numbers of virus-specific CD8+ T cells in HIV-infected progressors and long-term nonprogressors. J. Immunol. 165:1082-1092. [DOI] [PubMed] [Google Scholar]
- 11.Goulder, P. J., Y. Tang, C. Brander, M. R. Betts, M. Altfeld, K. Annamalai, A. Trocha, S. He, E. S. Rosenberg, G. Ogg, C. A. O'Callaghan, S. A. Kalams, R. E. McKinney, Jr., K. Mayer, R. A. Koup, S. I. Pelton, S. K. Burchett, K. McIntosh, and B. D. Walker. 2000. Functionally inert HIV-specific cytotoxic T lymphocytes do not play a major role in chronically infected adults and children. J. Exp. Med. 192:1819-1832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Haslett, P. A., D. F. Nixon, Z. Shen, M. Larsson, W. I. Cox, R. Manandhar, S. M. Donahoe, and G. Kaplan. 2000. Strong human immunodeficiency virus (HIV)-specific CD4+ T cell responses in a cohort of chronically infected patients are associated with interruptions in anti-HIV chemotherapy. J. Infect. Dis. 181:1264-1272. [DOI] [PubMed] [Google Scholar]
- 13.Ho, D. D., A. U. Neumann, A. S. Perelson, W. Chen, J. M. Leonard, and M. Markowitz. 1995. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature 373:123-126. [DOI] [PubMed] [Google Scholar]
- 14.Hogan, C. M., and S. M. Hammer. 2002. Host determinants in HIV infection and disease. Ann. Intern. Med. 134:761-776. [DOI] [PubMed] [Google Scholar]
- 15.Kuroda, M. J., J. E. Schmitz, W. A. Charini, C. E. Nickerson, M. A. Lifton, C. I. Lord, M. A. Forman, and N. L. Letvin. 1999. Emergence of CTL coincides with clearance of virus during primary simian immunodeficiency virus infection in rhesus monkeys. J. Immunol. 5127-5133. [PubMed]
- 16.Lori, F., M. G. Lewis, J. Xu, G. Varga, D. E. Zinn, Jr., C. Crabbs, W. Wagner, J. Greenhouse, P. Silvera, J. Yalley-Ogunro, C. Tinelli, and J. Lisziewicz. 2000. Control of SIV rebound through structured treatment interruptions during early infection. Science 290:1591-1593. [DOI] [PubMed] [Google Scholar]
- 17.Lori, F., and J. Lisziewicz. 2001. Structured treatment interruptions for the management of HIV infection. JAMA 286:2981-2987. [DOI] [PubMed] [Google Scholar]
- 18.Lori, F., R. Maserati, A. Foli, E. Seminari, J. Timpone, and J. Lisziewicz. 2000. Structured treatment interruptions to control HIV-1 infection. Lancet 355:287-288. [DOI] [PubMed] [Google Scholar]
- 19.Mellors, J. W., C. R. Rinaldo, Jr., P. Gupta, R. M. White, J. A. Todd, and L. A. Kingsley. 1996. Prognosis in HIV-1 infection predicted by the quantity of virus in plasma. Science 272:1167-1170. [DOI] [PubMed] [Google Scholar]
- 20.Murali-Krishna, K., J. D. Altman, M. Suresh, D. J. Sourdive, A. J. Zajac, J. D. Miller, J. Slansky, and R. Ahmed. 1998. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8:177-187. [DOI] [PubMed] [Google Scholar]
- 21.Ogg, G. S., X. Jin, S. Bonhoeffer, P. R. Dunbar, M. A. Nowak, S. Monard, J. P. Segal, Y. Cao, S. L. Rowland-Jones, V. Cerundolo, A. Hurley, M. Markowitz, D. D. Ho, D. F. Nixon, and A. J. McMichael. 1998. Quantitation of HIV-1-specific cytotoxic T lymphocytes and plasma load of viral RNA. Science 279:2103-2106. [DOI] [PubMed] [Google Scholar]
- 22.Ogg, G. S., X. Jin, S. Bonhoeffer, P. Moss, M. A. Nowak, S. Monard, J. P. Segal, Y. Cao, S. L. Rowland-Jones, A. Hurley, M. Markowitz, D. D. Ho, A. J. McMichael, and D. F. Nixon. 1999. Decay kinetics of human immunodeficiency virus-specific effector cytotoxic T lymphocytes after combination antiretroviral therapy. J. Virol. 73:797-800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ortiz, G. M., D. F. Nixon, A. Trkola, J. Binley, X. Jin, S. Bonhoeffer, P. J. Kuebler, S. M. Donahoe, M. A. Demoitie, W. M. Kakimoto, T. Ketas, B. Clas, J. J. Heymann, L. Zhang, Y. Cao, A. Hurley, J. P. Moore, D. D. Ho, and M. Markowitz. 1999. HIV-1-specific immune responses in subjects who temporarily contain virus replication after discontinuation of highly active antiretroviral therapy. J. Clin. Invest 104:R13-R18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ortiz, G. M., M. Wellons, J. Brancato, H. T. Vo, R. L. Zinn, D. E. Clarkson, K. Van Loon, S. Bonhoeffer, G. D. Miralles, D. Montefiori, J. A. Bartlett, and D. F. Nixon. 2001. Structured antiretroviral treatment interruptions in chronically HIV-1-infected subjects. Proc. Natl. Acad. Sci. USA 98:13288-13293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Oxenius, A., H. F. Gunthard, B. Hirschel, S. Fidler, J. N. Weber, P. J. Easterbrook, J. I. Bell, R. E. Phillips, and D. A. Price. 2001. Direct ex vivo analysis reveals distinct phenotypic patterns of HIV-specific CD8(+) T lymphocyte activation in response to therapeutic manipulation of virus load. Eur. J. Immunol. 31:1115-1121. [DOI] [PubMed] [Google Scholar]
- 26.Oxenius, A., D. A. Price, P. J. Easterbrook, A. O'Callaghan, A. D. Kelleher, J. A. Whelan, G. Sontag, A. K. Sewell, and R. E. Phillips. 2000. Early highly active antiretroviral therapy for acute HIV-1 infection preserves immune function of CD8+ and CD4+ T lymphocytes. Proc. Natl. Acad. Sci. USA 97:3382-3387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Panel on Clinical Practices for Treatment of HIV Infection. 2001. Guidelines for the use of antiretroviral agents in HIV-infected adults and adolescents. [Online.] http://www.hivatis.org.
- 28.Papasavvas, E., G. M. Ortiz, R. Gross, J. Sun, E. C. Moore, J. J. Heymann, M. Moonis, J. K. Sandberg, L. A. Drohan, B. Gallagher, J. Shull, D. F. Nixon, J. R. Kostman, and L. J. Montaner. 2000. Enhancement of human immunodeficiency virus type 1-specific CD4 and CD8 T cell responses in chronically infected persons after temporary treatment interruption. J. Infect. Dis. 182:766-775. [DOI] [PubMed] [Google Scholar]
- 29.Phillips, A. N., S. Staszewski, R. Weber, O. Kirk, P. Francioli, V. Miller, P. Vernazza, J. D. Lundgren, and B. Ledergerber. 2001. HIV viral load response to antiretroviral therapy according to the baseline CD4 cell count and viral load. JAMA 286:2560-2567. [DOI] [PubMed] [Google Scholar]
- 30.Pitcher, C. J., C. Quittner, D. M. Peterson, M. Connors, R. A. Koup, V. C. Maino, and L. J. Picker. 1999. HIV-1-specific CD4+ T cells are detectable in most individuals with active HIV-1 infection, but decline with prolonged viral suppression. Nat. Med. 5:518-525. [DOI] [PubMed] [Google Scholar]
- 31.Rosenberg, E. S., M. Altfeld, S. H. Poon, M. N. Phillips, B. M. Wilkes, R. L. Eldridge, G. K. Robbins, R. T. D'Aquila, P. J. Goulder, and B. D. Walker. 2000. Immune control of HIV-1 after early treatment of acute infection. Nature 407:523-526. [DOI] [PubMed] [Google Scholar]
- 32.Rosenberg, E. S., J. M. Billingsley, A. M. Caliendo, S. L. Boswell, P. E. Sax, S. A. Kalams, and B. D. Walker. 1997. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science 278:1447-1450. [DOI] [PubMed] [Google Scholar]
- 33.Ruiz, L., G. Carcelain, J. Martinez-Picado, S. Frost, S. Marfil, R. Paredes, J. Romeu, E. Ferrer, K. Morales-Lopetegi, B. Autran, and B. Clotet. 2001. HIV dynamics and T-cell immunity after three structured treatment interruptions in chronic HIV-1 infection. AIDS 15:F19-F27. [DOI] [PubMed] [Google Scholar]
- 34.Schmitz, J. E., M. J. Kuroda, S. Santra, V. G. Sasseville, M. A. Simon, M. A. Lifton, P. Racz, K. Tenner-Racz, M. Dalesandro, B. J. Scallon, J. Ghrayeb, M. A. Forman, D. C. Montefiori, E. P. Rieber, N. L. Letvin, and K. A. Reimann. 1999. Control of viremia in simian immunodeficiency virus infection by CD8+ lymphocytes. Science 283:857-860. [DOI] [PubMed] [Google Scholar]
- 35.Schockmel, G. A., S. Yerly, and L. Perrin. 1997. Detection of low HIV-1 RNA levels in plasma. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 14:179-183. [DOI] [PubMed] [Google Scholar]
- 36.Sewell, A. K., D. A. Price, A. Oxenius, A. D. Kelleher, and R. E. Phillips. 2000. Cytotoxic T lymphocyte responses to human immunodeficiency virus: control and escape. Stem Cells 18:230-244. [DOI] [PubMed] [Google Scholar]
- 37.Sokal, R. R., and F. J. Rohlf. 1987. Biometry: the principles and practise of statistics in biological research. Freeman and Co., New York, N.Y.
- 38.Subkommission Klinik (SKK) der Eidg. Kommission für Aids-Fragen (EKAF). 2000. Empfehlungen zur antiretroviralen HIV-Therapie 2001. Bulletin Bundesamt Gesundheit 51:994-1000. [Google Scholar]
- 39.Wei, X., S. K. Ghosh, M. E. Taylor, V. A. Jonson, E. A. Emini, P. Deutsch, J. D. Lifson, S. Bonhoeffer, M. A. Nowak, B. H. Hahn, M. S. Saag, and G. M. Shaw. 1995. Viral dynamics in human immunodeficiency virus type 1 infection. Nature 373:117-122. [DOI] [PubMed] [Google Scholar]