Abstract
The inability of replication-defective viral vectors to efficiently transduce tumor cells in vivo has prevented the successful application of such vectors in gene therapy of cancer. To address the need for more efficient gene delivery systems, we have developed replication-competent retroviral (RCR) vectors based on murine leukemia virus (MLV). We have previously shown that such vectors are capable of transducing solid tumors in vivo with very high efficiency. While the natural requirement of MLV infection for cell division imparts a certain degree of specificity for tumor cells, additional means for confining RCR vector replication to tumor cells are desirable. Here, we investigated the parameters critical for successful tissue-specific transcriptional control of RCR vector replication by replacing various lengths of the MLV enhancer/promoter with sequences derived either from the highly prostate-specific probasin (PB) promoter or from a more potent synthetic variant of the PB promoter. We assessed the transcriptional specificity of the resulting hybrid long terminal repeats (LTRs) and the cell type specificity and efficiency of replication of vectors containing these LTRs. Incorporation of PB promoter sequences effectively restricted transcription from the LTR to prostate-derived cells and imparted prostate-specific RCR vector replication but required the stronger synthetic promoter and retention of native MLV sequences in the vicinity of the TATA box for optimal replicative efficiency and specificity. Our results have thus identified promoter strength and positioning within the LTR as important determinants for achieving both high transduction efficiency and strict cell type specificity in transcriptionally targeted RCR vectors.
Defective vectors derived from oncoretroviruses possess a number of properties that have made them useful tools for therapeutic and experimental gene delivery, including low immunogenicity, integration into host cell DNA, a simple, well-characterized genome, and ease of production. It has become clear from a large number of studies, however, that defective retroviral vectors are generally incapable of efficient transduction when administered in vivo (14, 25, 33-35, 39, 40, 49). Although certain gene therapy applications, such as those in which transduction confers a selective advantage to target cells (3, 24) or those requiring only relatively low levels of expression of a secreted product (23), may not require high gene transfer efficiency, others, such as cytoablative strategies for genetic cancer therapy, generally require high transduction efficiency (45).
As a means for attaining more efficient gene delivery, we have constructed a series of replication-competent retrovirus (RCR) vectors derived from murine leukemia virus (MLV) (26). We demonstrated that an RCR vector containing an internal ribosome entry site (IRES)-green fluorescent protein (GFP) cassette between the env gene and the 3′ untranslated region can transduce solid tumors in vivo with a level of efficiency much greater than that possible using standard defective retrovirus vectors (27). However, while replication competence imparts greatly enhanced gene transfer power to retroviral vectors, it also represents a potential increase in the risk of oncogenesis, particularly under conditions of severe immunosuppression (6). Confining viral replication exclusively to targeted cell types would be one way of minimizing this risk.
An approach to the targeting of defective retroviral vectors that has been utilized previously is that of replacement of the promiscuous transcriptional control elements within the U3 region of the long terminal repeat (LTR) with promoter sequences that are active only in certain cell types (5, 8, 18, 30). This strategy has consistently allowed the generation of vectors with transgene expression limited in a cell-type-specific manner. Applied to an RCR vector, such a targeting strategy could be used to target both vector replication and transgene expression. Thus far, however, there have been no studies examining the parameters critical for achieving efficient and tissue-specific transcriptional control of retrovirus replication.
In this study, we have characterized a series of RCR vectors targeted to prostate cells by replacement of various segments of the U3 region with sequences from the probasin (PB) promoter or a recombinant variant of the PB promoter. Transcription of the PB gene is regulated by androgen and is highly specific to prostate epithelium in rodents (20). A 450-bp fragment of the proximal rat PB promoter has been used to generate mice whose transgene expression is limited exclusively to the prostate epithelium (12, 13). In vitro studies have also demonstrated that although there is no human homolog for this gene, the proximal PB promoter retains high levels of activity and prostate specificity in human cells (22, 36). ARR2PB is a synthetic variant of the proximal PB promoter that possesses levels of strength significantly greater than and prostate specificity equal to the parental promoter (47, 50) and has been used previously to target transgene expression in adenovirus vectors (1, 48). Our results demonstrate that both replication and transgene expression of an RCR vector can be transcriptionally targeted to particular cell types with high specificity and that efficient replication requires optimization of promoter strength and the retention of specific sequences within the native MLV promoter.
MATERIALS AND METHODS
Cell lines and virus production.
LNCaP (16), MDA PCa 2b (31), DU145 (42), and PC-3 (19) human prostate carcinoma cells, NMU rat mammary carcinoma cells (4), and HeLa human cervical carcinoma cells (38) were obtained from the American Type Culture Collection. PC-3(AR)2 cells (15) were kindly provided by Theodore Brown (University of Toronto). LNCaP, PC-3, and PC-3(AR)2 cells were cultivated in RPMI 1640 medium with 10% fetal bovine serum (FBS). MDA PCa 2b cells were grown in Ham's F12K medium with 20% FBS and supplements as described previously (31). HeLa and NMU cells were cultivated in minimum essential Eagle's medium with 10% FBS. 293T cells (7) were grown in Dulbecco's modified Eagle's medium with 10% FBS. For luciferase assays, all media were formulated using FBS treated with charcoal-dextran (HyClone) to remove steroids. Vector stocks were produced by transfection of 293T cells with vector plasmid by using Lipofectamine Plus (Invitrogen).
Construction of plasmids.
All vector plasmids described in this study were derived from pACE-GFP-dm, which contains a full-length amphotropic RCR vector encoding GFP and the cytomegalovirus (CMV) promoter in place of the 5′ U3 region. To construct pACE-GFP-dm, the CMV promoter was amplified from pEGFP-N1 (Clontech) and introduced into pAZE-GFP (26) in place of the 5′ U3 region by using overlap extension PCR (17). The resulting plasmid, pACE-GFP, was then subjected to site-directed mutagenesis (QuikChange kit; Stratagene) to introduce a PmeI site into the 3′ U3 region and remove the SacI site in the pol gene, creating pACE-GFP-dm. The PmeI site was created at position −92 (relative to the transcription start site) by two point mutations, and the SacI site was removed by the introduction of a silent point mutation. These mutations had no discernible effect on in vitro replication of the vector (data not shown). The introduced PmeI site and the remaining SacI site at −30 in the 3′ U3 region are both unique in pACE-GFP-dm.
To create plasmids pACE-GFP-Pr and pACE-GFP-Ar, we used overlap extension PCR to replace the 3′ U3 sequences in pACE-GFP-dm, from the unique NheI site to the 5′ border of the R region, with the proximal rat PB promoter or the synthetic PB promoter variant ARR2PB, respectively. Plasmids pACE-GFP-Pt and pACE-GFP-At were constructed by first amplifying the proximal PB promoter and ARR2PB (from their 5′ termini to the 5′ borders of their TATA boxes) with primers that introduce a 5′-terminal NheI site and a 3′-terminal SacI site. These PCR products were then used to replace a large stretch of the 3′ U3 region of pACE-GFP-dm (i.e., from the NheI site to the SacI site just upstream of the MLV TATA box). Plasmids pACE-GFP-Pc and pACE-GFP-Ac were constructed by replacement of the 3′ U3 region of pACE-GFP-dm from the NheI site to the PmeI site with sequences of the proximal PB promoter or ARR2PB PCR amplified from their 5′ ends to the 5′ borders of their CAAT boxes at position −48.
Luciferase reporter plasmids were generated by using pGL2-Basic (Promega). The entire length of each hybrid LTR was amplified from the vector plasmids by using a 5′ primer containing an SmaI site and a 3′ primer containing an MluI site. These products were then introduced into pGL2-Basic at the SmaI and MluI sites upstream of the luciferase cDNA.
Luciferase assays.
Cells at 70 to 80% confluence in 6-well plates were transfected with 3 μg of luciferase reporter plasmid containing the hybrid LTR by using Lipofectamine Plus. Parallel cultures were transfected with plasmids pGL2-Control (Promega), which contains the luciferase gene under the control of the simian virus 40 (SV40) promoter and enhancer, or pGL2-Basic, which contains the luciferase gene but no promoter. Each culture was cotransfected with 2 μg of the β-galactosidase expression plasmid pCH110 to control for transfection efficiency. For cultures subjected to androgen induction, 5-α-dihydrotestosterone (DHT) was added to the media to a concentration of 1 nM at 24 h posttransfection. At 48 h posttransfection, reporter lysis buffer (Promega) was used to prepare extracts from the cells. Luciferase and β-galactosidase activities were measured with the Luciferase assay system and the β-Galactosidase enzyme assay system (Promega), respectively.
Infections with replicating vectors.
LNCaP, MDA PCa 2b, HeLa, or NMU cells at 20 to 30% confluence in 6-cm-diameter dishes were infected with equal amounts of stock vector, corresponding to a multiplicity of infection of 0.05, in the presence of 4 μg of Polybrene/ml. At 3, 8, and 21 days postinfection, the cells were analyzed for GFP expression by using flow cytometry on a FACScan II apparatus (Becton Dickinson). Serial infections with the ACE-GFP-At virus were carried out by passaging vector taken from LNCaP cultures at day 21 postinfection through fresh LNCaP cultures every week thereafter for 7 weeks, using 20-fold dilutions of supernatant for each infection cycle. Genomic DNA from each infection cycle was isolated by using the GenomicPrep kit (Amersham-Pharmacia).
Vector titrations.
The stock vector titer was initially determined by RNA dot blot analysis as described previously (32). Blots were probed with a gag-pol fragment of MLV labeled with [32P]dCTP by using the Prime-It II kit (Stratagene), and titers were estimated by comparison to virion RNA of an ACE-GFP stock of known biological titer. Biological titers of ARR2PB-targeted vectors were determined by exposing 20 to 30% confluent cultures in the presence of 4 μg of Polybrene/ml to dilutions of the vector produced by LNCaP, MDA PCa 2b, HeLa, or NMU cultures infected 30 to 32 days previously. The following day, the medium on the cells was replaced with fresh medium containing 50 μM azidothymidine (Sigma) to block the secondary spread of the vector. On day 3, the cells were analyzed by using flow cytometry to determine the number of cells expressing GFP. The titer of the vector produced from each infected cell type was determined by using the same cell type.
Southern blot analysis.
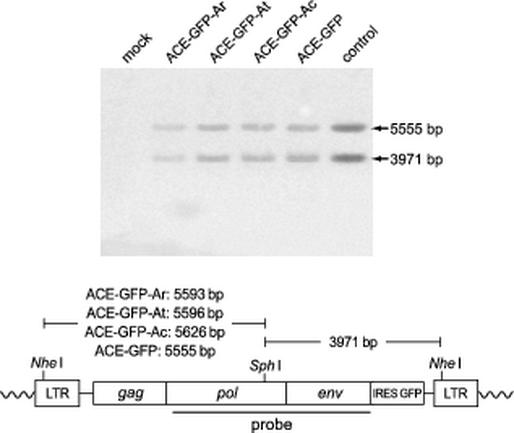
Genomic DNA was isolated from infected LNCaP cells by using the GenomicPrep kit (Amersham-Pharmacia Biotech). The DNA was digested with NheI and SphI and then repurified by ethanol precipitation. A total of 10 μg of each DNA was fractionated on an agarose gel and blotted onto a Zeta-Probe nylon membrane (Bio-Rad) by using a Stratagene Posiblot manifold. A pol-env fragment of pACE-GFP was random prime labeled and hybridized to the membrane in Stratagene QuickHyb hybridization buffer. The hybridized blot was visualized using a Storm PhosphorImager (Molecular Dynamics).
PCR analysis and sequencing.
The 5′ LTR of the ACE-GFP-At vector provirus was amplified by PCR by using an upstream primer that binds the 22 5′-terminal nucleotides of the U3 region, a downstream primer that binds just upstream of gag, and Pfu polymerase (Stratagene). Genomic DNA from infected LNCaP cells or a molecularly cloned copy of the 5′ region of ACE-GFP-At was used as the template. The amplified products were separated by gel electrophoresis, and the predominant species were extracted with a QIAquick gel extraction kit (Qiagen) and directly sequenced by using the downstream PCR primer.
RESULTS
Construction of hybrid PB-MLV LTRs.
The proximal PB promoter contains elements that direct prostate-specific transcription, including a 148-bp sequence (from −244 to −96) called the androgen-responsive region (ARR; Fig. 1A). Within the ARR are two androgen receptor binding sites, which work cooperatively in the induction of promoter activity by androgens (21). A recently described synthetic variant of the proximal PB promoter, ARR2PB, contains two copies of the ARR and exhibits more robust transcriptional activity than the wild-type (wt) promoter in vitro and in vivo (50). The PB promoter, ARR2PB, and the MLV U3 region each contain CAAT and TATA boxes.
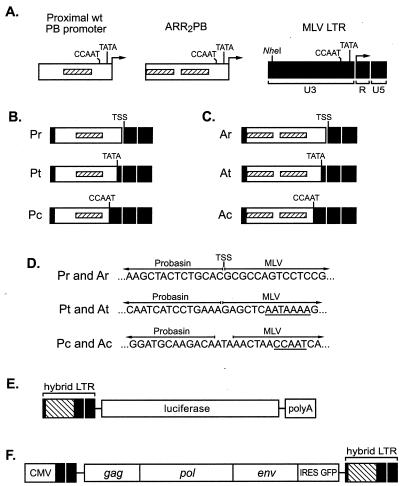
FIG. 1.
Constructs used in this study. (A) Sequences used in generating hybrid LTRs. The proximal rat PB promoter (left) contains CAAT and TATA box homologies and an ARR (shown as a hatched box) important for androgen induction of transcription. ARR2PB (center) is a synthetic variant of the PB promoter and contains two copies of the ARR. The MLV LTR (right) comprises the U3, R, and U5 regions. The transcriptional control sequences of MLV are located primarily in the U3 region, which also contains CAAT and TATA box sequences. (B) Hybrid LTRs containing the wt PB promoter. LTRs Pr, Pt, and Pc contain PB promoter sequences from position −383 to the transcription start site (TSS), TATA box, and CAAT box, respectively. (C) Hybrid LTRs containing ARR2PB. LTRs Ar, At, and Ac contain ARR2PB sequences from the 5′ end of the upstream ARR to the TSS, TATA box, and CAAT box, respectively. In each of the six hybrid LTRs, MLV U3 sequences from the NheI site to the TSS, TATA box, or CAAT box were replaced with the corresponding PB or ARR2PB sequences. (D) Sequence details of the hybrid LTRs. Shown are the nucleotide sequences at the 3′ borders between the PB and MLV sequences. TATA and CAAT boxes are underlined. TSS, transcription start site. (E) Luciferase reporter constructs containing hybrid LTRs. (F) Structure of replication-competent MLV vectors containing hybrid LTRs. Each vector contains an IRES-GFP cassette positioned immediately downstream of the env gene and a 5′ LTR in which the U3 region was replaced by the CMV immediate-early promoter. The 3′ LTR is used to form the 5′ LTR during MLV replication. We therefore replaced the 3′ LTR of the RCR vector with the hybrid LTRs.
We constructed a series of six hybrid MLV LTRs incorporating sequences from the PB promoter. The hybrid LTRs were generated by replacement of the MLV U3 sequence (from the NheI site near the 5′ end to the CAAT box, TATA box, or transcription start site) with sequences from the wt PB promoter or ARR2PB (Fig. 1B and C). Each of these LTRs was designed such that transcription would be initiated at the 5′ border of the R region, as occurs in wt MLV. LTRs Pr and Ar contain the wt PB promoter and ARR2PB, respectively, with their transcription start sites fused precisely at the 5′ border of the MLV R region. LTRs Pt and At contain the wt PB promoter and ARR2PB, respectively (from their 5′ ends to the 5′ ends of their TATA boxes), fused at the MLV TATA box. LTRs Pc and Ac contain the wt PB promoter and ARR2PB, respectively (from their 5′ ends to the 5′ ends of their CAAT boxes), fused to the MLV CAAT box. A series of six reporter plasmids in which each of the hybrid LTRs controls expression of the luciferase cDNA were constructed (Fig. 1E).
Transcriptional activity of hybrid promoters in prostate and nonprostate cells.
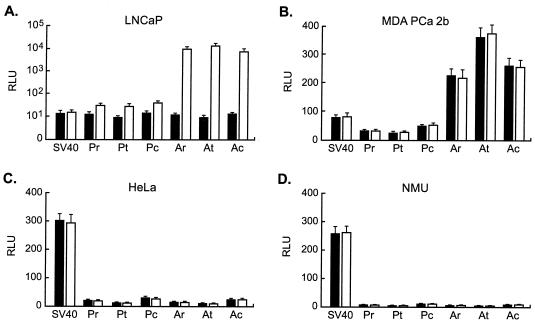
LNCaP and MDA PCa 2b prostate carcinoma, HeLa cervical carcinoma, and NMU mammary carcinoma cells were transiently transfected with the reporter plasmids containing the hybrid LTRs to assess transcriptional activity and cell type specificity. Transfections were carried out both in the presence and absence of androgen to evaluate the androgen inducibility of the hybrid LTRs. As a standard, a reporter plasmid containing the SV40 early promoter was used in parallel transfections.
In LNCaP cells, the three wt PB promoter-targeted LTRs and the SV40 promoter exhibited similar levels of strength in the absence of androgen (Fig. 2A). Upon androgen induction, activity levels of these hybrid LTRs increased three- to fourfold, while the SV40 promoter activity level remained unchanged. The ARR2PB-targeted LTRs also exhibited, in the absence of androgen, a level of transcriptional strength similar to that of the SV40 promoter, although upon androgen induction, transcription from each of these LTRs increased roughly 1,000-fold. In MDA PCa 2b cells, all three wt PB promoter hybrid LTRs exhibited somewhat lower levels of strength than the SV40 promoter and were not induced by androgen (Fig. 2B). The LTRs containing ARR2PB were significantly more potent in these cells than in those containing the wt PB promoter, driving luciferase expression to levels three- to fourfold higher than that by the SV40 promoter. Androgen also had no significant effect on transcription from the ARR2PB-containing LTRs in MDA PCa 2b cells. The absence of induction in MDA PCa 2b cells was most likely due to the presence in these cells of a doubly mutated androgen receptor that exhibits greatly reduced affinity to DHT and increased affinity to other steroids compared to both the wt receptor and the singly mutated receptor present in LNCaP cells (51).
FIG. 2.
Cell type specificity and androgen inducibility of transcription from hybrid LTRs containing wt PB promoter or ARR2PB. Each of the hybrid LTRs was cloned upstream of the luciferase gene in plasmid pGL2 and transfected into two prostate (A and B) and two nonprostate (C and D) cell lines as indicated. Transcriptional activity was determined in the absence (black bars) and presence (open bars) of androgen. All cells were cultured in media containing serum that was charcoal stripped to remove endogenous steroids, and androgen induction was carried out by the addition of 1 nM DHT. Arbitrary relative light unit (RLU) levels were normalized for transfection efficiency, and the level determined for the promoterless control reporter plasmid in each cell line was assigned a value of 1. Results are the means obtained from at least three independent experiments. Error bars indicate standard deviations.
In the two nonprostate cell lines, the level of activity of each of the hybrid LTRs was much lower than that of the SV40 promoter, and androgen had no discernible effect on luciferase expression (Fig. 2C and D). Of the six hybrid LTRs, the two constructed using the CAAT box fusion design, Pc and Ac, exhibited the highest activity levels in HeLa and NMU cells. The higher nonspecific transcriptional activity level of these LTRs was most likely due to the retention of more of the MLV U3 region in these sequences than in the other LTRs. Surprisingly, the LTRs employing the TATA box fusion design consistently displayed the lowest activity levels in the nonprostate cell lines but levels of strength comparable to those of the other LTRs in the prostate lines. Use of the MLV promoter downstream from and including the TATA box in place of the corresponding region of the PB promoter therefore slightly improved, rather than impaired, prostate specificity. Taken together, these results show that, within the context of the hybrid LTRs, both the proximal PB promoter and ARR2PB retain androgen inducibility and high specificity for cells derived from prostate epithelium.
Construction of replication-competent vectors containing hybrid LTRs.
To assess the ability of the hybrid LTRs to support replication of a nondefective MLV and to impart prostate cell-specific replication, we replaced the 3′ LTR in an amphotropic, GFP-encoding RCR vector with each of these LTRs (Fig. 1F). The hybrid LTRs were introduced into the 3′ LTR position, as this copy is used as the template in the formation of both LTRs during reverse transcription. The vector plasmid we used to construct the targeted vectors also contained the CMV promoter in place of the 5′ U3 region. In pilot studies, we found that when the 5′ LTR of the plasmid contained the wt U3, the 5′ LTR in the plasmid recombined during transfection with other plasmid molecules at the 3′ LTR to reconstitute an RCR vector with wt LTRs at both termini (data not shown). This resulted in contamination of the targeted vector preparation with an untargeted vector that exhibited a replicative advantage and eventually came to dominate the virus population. In contrast, no such revertants emerged in the vector-encoding plasmids after replacement of the U3 region of the 5′ LTR with the CMV promoter. Stocks of the targeted RCR vectors were generated by transient transfection of the vector plasmids, and their titers were initially determined by RNA dot blot analysis (data not shown).
Replication of targeted vectors in prostate and nonprostate cells.
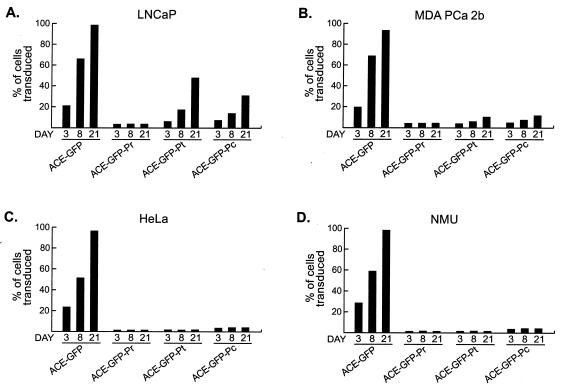
LNCaP, MDA PCa 2b, HeLa, and NMU cultures were inoculated with stock vector at a multiplicity of infection of 0.05, and vector replication was followed by flow cytometric analysis of the cultures at 3, 8, and 21 days postinfection. Although no exogenous androgen was added to these cultures, all media used during infections were made with nonstripped serum and thus contained endogenous androgen.
LNCaP cells infected with either ACE-GFP-Pt or ACE-GFP-Pc exhibited progressively increasing transduction levels over the 21-day period (Fig. 3A). The replication of these two vectors was significantly delayed relative to that of the untargeted parental vector ACE-GFP and spread to no more than half of the cells in each LNCaP culture after 21 days. In contrast, while the number of cells transduced by ACE-GFP-Pr was comparable to that by ACE-GFP-Pt and ACE-GFP-Pc by the 3-day time point, no spread was observed thereafter. ACE-GFP-Pt and ACE-GFP-Pc also replicated in MDA PCa 2b cells, although only very slowly, transducing approximately 10% of the cells in each culture after 21 days (Fig. 3B). ACE-GFP-Pr did not replicate in these cells beyond an initial low level of transduction. In HeLa and NMU cells, none of the three wt PB promoter-targeted vectors exhibited evidence of replication, while untargeted vector spread through both cell lines very efficiently. A small percentage (1.3 to 2.8%) of cells in HeLa and NMU cultures exposed to the wt PB promoter-targeted vectors expressed detectable levels of GFP at each time point, although these percentages were stable over several weeks and expression levels were extremely low (Fig. 3C and D and data not shown). As the vectors are targeted only at the transcriptional level, an initial low level of transduction by the vector stocks was not surprising. Of the HeLa and NMU cultures infected with the wt PB promoter-targeted vectors, those infected with ACE-GFP-Pc exhibited the highest percentage of cells expressing detectable levels of GFP, which is a finding consistent with the higher nonspecific transcriptional activity of the Pc LTR.
FIG. 3.
Replication of wt PB promoter-targeted vectors in prostate and nonprostate cells. GFP expression in infected cells was assessed by flow cytometry on the indicated days postinfection. Results from infections of LNCaP (A), MDA PCa 2b (B), HeLa (C), and NMU (D) cells are shown. All cells were grown in nonstripped serum, and no exogenous androgen was added. Values are the means of three experiments.
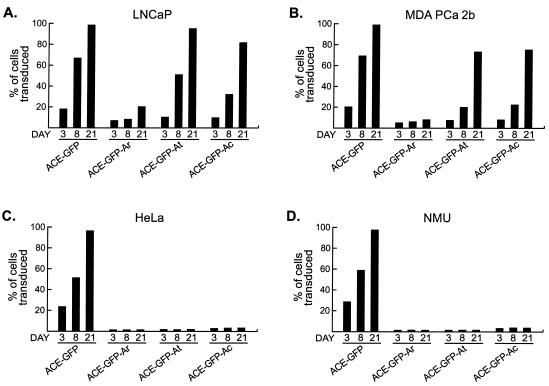
In LNCaP cells, both ACE-GFP-At and ACE-GFP-Ac replicated with kinetics markedly faster than the corresponding wt PB promoter-targeted vectors and only moderately slower than the untargeted vector (Fig. 4A), indicating that the greater transcriptional strength of ARR2PB relative to that of the wt PB promoter enabled much more efficient vector propagation. Additionally, ACE-GFP-Ar, unlike ACE-GFP-Pr, was capable of replication in LNCaP cells, transducing on average approximately 20% of each infected culture after 21 days. Thus, the defect in replication of ACE-GFP-Pr was due at least in part to the insufficient strength of the wt PB promoter. MDA PCa 2b cells supported levels of replication of the three ARR2PB-targeted vectors that were similar but slightly lower overall (Fig. 4B), with ACE-GFP-Ar again spreading much more slowly than the other two. The slower spread of the targeted vectors in MDA PCa 2b cells compared to LNCaP cells is consistent with the relative levels of transcriptional strength of the targeted LTRs in these cell lines. While the ARR2PB hybrid LTRs mediated transcription levels roughly 10,000-fold higher than those of the promoterless control in LNCaP cells in the presence of androgen, transcription from these LTRs in MDA PCa 2b cells in the presence of androgen was by comparison 200- to 400-fold higher than that from the promoterless control (Fig. 2A and B). The disparity in the sequences of the androgen receptors in LNCaP and MDA PCa 2b cells may be responsible in part for these differences. Despite the greatly improved replication kinetics of the vectors containing ARR2PB compared to that of the vectors containing the wt PB promoter in prostate cells, GFP expression in infected HeLa and NMU cells remained restricted to an equally low, stable number of cells (Fig. 4C and D). Continued propagation of the HeLa and NMU cells infected with the ARR2PB-targeted vectors for 6 weeks after inoculation did not result in an increase in the percentage of infected cells (data not shown).
FIG. 4.
Replication of ARR2PB-targeted vectors in prostate and nonprostate cells. GFP expression in infected cells was assessed by flow cytometric analysis on the indicated days postinfection. Results from infections of LNCaP (A), MDA PCa 2b (B), HeLa (C), and NMU (D) cells are shown. All cells were grown in medium containing nonstripped serum, and no exogenous androgen was added. Values are the means of three experiments.
Three additional prostate-derived cell lines were tested for their ability to support both transcription from the At LTR and replication of ACE-GFP-At. Two of these cell lines, DU145 and PC-3, do not express androgen receptor, while the third, PC-3(AR)2, a PC-3 derivative, expresses androgen receptor from an episomal plasmid. Each of these cell lines was efficiently infected with untargeted vector (data not shown). However, neither DU145 nor PC-3 cells mediated either efficient transcription from the At LTR or replication of ACE-GFP-At (Table 1). In PC-3(AR)2 cells, by contrast, transcription from the At LTR was highly efficient and ACE-GFP-At was capable of replication. Thus, we observed a strong correlation in prostate cell lines between efficiency of transcription from the At LTR, replication of ACE-GFP-At, and expression of androgen receptor, further corroborating the transcriptional specificity of this vector.
TABLE 1.
Correlation of transcriptional activity of hybrid LTR At, replication of ACE-GFP-At, and presence of androgen receptor in human prostate carcinoma cell lines
| Cell line | Androgen receptor expression | Efficient transactivation of hybrid LTR Ata | Replication of ACE-GFP-Atb |
|---|---|---|---|
| LNCaP | + | + | + |
| MDA PCa 2b | + | + | + |
| DU145 | − | − | − |
| PC-3 | − | − | − |
| PC-3(AR)2 | + | + | + |
Cell lines denoted as positive exhibited luciferase expression levels 200-fold or more higher than those of promoterless control plasmids; cell lines denoted as negative exhibited levels less than 10-fold higher than those of promoterless controls. Data used were taken from transfections in charcoal-stripped serum containing 1 nM DHT.
Determined by flow cytometric analysis of infected cells over a 21-day period following exposure to stock vector.
Determination of biological titers of targeted vectors.
The titers of the three ARR2PB-targeted vectors replicating in LNCaP, MDA PCa 2b, HeLa, and NMU cells were determined by using a flow cytometric assay. LNCaP cultures chronically infected by ACE-GFP, ACE-GFP-At, or ACE-GFP-Ac produced infectious vector at comparable levels of between 3 × 104 and 10 × 104 GFP-transducing units/ml (Table 2). In contrast, titers of ACE-GFP-Ar produced by the same cells were more than 10-fold lower than those of the other two targeted vectors, which is a finding consistent with the slower replication kinetics of this vector. ACE-GFP, ACE-GFP-At, and ACE-GFP-Ac each replicated on MDA PCa 2b cultures at titers roughly three- to sixfold lower than on LNCaP cells. In contrast, on the two nonprostate cell lines, the targeted and untargeted vectors were produced at very different levels. On NMU cells, average titers of ACE-GFP-At and ACE-GFP-Ac were 3 and 8%, respectively, of those of ACE-GFP. On HeLa cells, while the levels of ACE-GFP-At and ACE-GFP-Ac remained below the detection limit of the assay (30 GFP-transducing units/ml), ACE-GFP was produced at 1.8 × 103 GFP-transducing units/ml.
TABLE 2.
Titers of ARR2PB-targeted vectors
| Vector | Titer (GFP-transducing units/ml) for cell type:
|
|||
|---|---|---|---|---|
| LNCaP | MDA PCa 2b | NMU | HeLa | |
| ACE-GFP | 9.9 × 104 | 2.9 × 104 | 1.0 × 104 | 1.8 × 103 |
| ACE-GFP-Ar | 3.0 × 103 | NDa | ND | ND |
| ACE-GFP-At | 4.8 × 104 | 7.6 × 103 | 300 | <30 |
| ACE-GFP-Ac | 3.3 × 104 | 8.7 × 103 | 800 | <30 |
ND, not determined.
Structural analysis of proviral vector DNA.
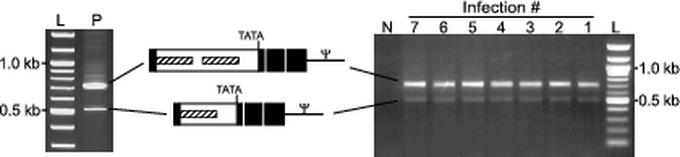
To confirm that the ARR2PB vectors did not undergo genomic alterations during the infection of LNCaP cells, we analyzed integrated provirus from infected LNCaP cells by using Southern hybridization. For each of the vectors, only bands of the sizes expected for full-length proviruses were obtained (Fig. 5), demonstrating that the GFP transgene had been transmitted as part of the intact vectors. To assess the stability of the hybrid LTR sequences over multiple replication cycles, we subjected ACE-GFP-At to seven serial passages through LNCaP cultures and isolated genomic DNA from each passage. We utilized PCR to amplify the 5′ LTR from integrated vector provirus and sequenced the resulting products. The sequences of the PCR products from each of the seven vector passages were found to be identical to those obtained by PCR using an intact, molecularly cloned ACE-GFP-At provirus control (Fig. 6). The amplifications of both the control and experimental templates generated two products. One represented the full-length At LTR, and the other represented a variant form of the At LTR containing only one ARR. The presence of this variant in the control reaction product indicates that it was an artifact of the PCR. Thus, the integrity of the hybrid LTR of ACE-GFP-At appears to have been maintained through prolonged propagation of the vector. The retention of the prostate specificity of serially passaged ACE-GFP-At was further demonstrated by the absence of vector spread in NMU cells exposed to conditioned medium from each of the seven ACE-GFP-At-infected cultures (data not shown).
FIG. 5.
Southern blot analysis of genomic DNA from LNCaP cells infected with ARR2PB-targeted vectors. Each DNA sample was digested with NheI and SphI, separated on an agarose gel, and blotted onto a nylon membrane. The membrane was hybridized to a radiolabeled pol-env fragment of MLV. mock, DNA from mock-infected cells; control, NheI/SphI-digested pAZE-GFP plasmid with DNA from mock-infected cells. A schematic diagram of integrated vector provirus indicating the size of restriction fragments for each vector and the location of the probe is shown.
FIG. 6.
Hybrid LTR of ACE-GFP-At remains unchanged through multiple serial passages. A PCR-based assay designed to assess the stability of the hybrid LTRs by amplification and sequencing of the 5′ LTR from proviral DNA was utilized. Amplification of a cloned proviral copy of ACE-GFP-At as a positive control resulted in products of approximately 720 and 510 bp (left gel). As depicted schematically, sequencing demonstrated that these products corresponded to the full-length, intact At LTR and an artifactual form of the At LTR missing one of the two ARRs, respectively. ACE-GFP-At was subjected to seven cell-free serial passages through LNCaP cultures, and genomic DNA from each culture was used as the template in PCR amplification and sequencing of the LTR (right gel). Sequencing of the two resulting products from selected passages demonstrated that their sequences were identical to those of the products generated by amplification of the positive control template. Abbreviations: L, 100-bp ladder; P, amplification using a plasmid containing a cloned proviral copy of ACE-GFP-At as the template; N, amplification using DNA from uninfected cells.
DISCUSSION
Virus vectors that replicate exclusively in targeted tissues would represent a valuable addition to the tools available for use in therapeutic and experimental gene transfer. In this study, we evaluated the possibility of targeting replication of a nondefective retrovirus vector to prostate carcinoma cells by replacement of transcriptional control sequences of the vector's native U3 region with sequences from the rat PB promoter. Our results demonstrate that this approach can be used to generate RCR vectors that replicate in a highly efficient and cell type-specific manner.
Much interest in replication-competent forms of a number of virus species for use in cancer therapy has arisen in recent years (37, 46). RCR vectors derived from oncoretroviruses such as MLV possess unique properties that may provide significant advantages for use in cancer gene therapy. First, most oncoretroviruses, including MLV, are not inherently cytopathic. Thus, the inclusion of a conditionally cytotoxic transgene such as thymidine kinase would allow strict exogenous control of cell-killing activity through administration of prodrug. Second, oncoretroviruses infect only dividing cells (29) and therefore possess, as do some other oncolytic viruses (28, 41, 43), a level of built-in specificity for the actively dividing tissue characteristic of malignancy. Third, the biology and genomes of simple retroviruses make these viruses ideal for transcriptionally targeting replication to tumor cells. The oncoretroviral genome consists of a single transcriptional unit controlled primarily by elements collected in a single area within the virus, namely, the U3 region. Thus, transcription of all viral genes as well as of the RNA genome itself is determined by U3 sequences. Previous studies have shown that the transcriptional control sequences of U3 within defective vectors can be replaced with cell type-specific promoter elements to target transgene expression to defined cell types (5, 8, 18, 30).
Although transcriptionally targeted RCR vectors have not been previously described, a replication-competent MLV containing sequences from the mouse transthyretin promoter in place of the U3 enhancer repeats was previously characterized (9). This promoter possesses specificity for cells of the liver and choroid plexus and was thus introduced into the LTR as a possible means of targeting viral replication to these cell types. The resulting virus, however, exhibited tissue tropism as broad as that of wt MLV. This was most likely due to the retention of a large stretch (−152 to −1) of transcriptionally active U3 sequence in the vector (11).
The results presented here help to define how a transcriptionally targeted MLV-based RCR vector can be optimally constructed. First, we have shown that the entire U3 region except for approximately 30 bp at each end can be replaced with sequences from a heterologous promoter without significantly reducing the vector's replicative ability. The sequence contained within this region therefore does not, at least in vitro, appear to play an important role in viral replication other than that of providing transcriptional regulation. Inclusion of any of part of this 380-bp region in a transcriptionally targeted vector appears to be unnecessary and would likely only serve to increase nonspecific transcription, as observed with the vectors and LTRs constructed using the CAAT box fusion design.
It is notable that replication of both of the vectors in which the targeting promoter was fused at the LTR's transcriptional start site was greatly impaired relative to that of the vectors having the CAAT or TATA box fusion designs. Given that the LTRs of all three designs exhibited very similar levels of transcriptional strength in prostate cells, these results imply that the 3′-terminal 30 bp of U3 are involved in some stage of viral replication other than transcription. In human immunodeficiency virus, the sequence of U3 downstream from the TATA box has been shown to play a role in 3′-end formation (10, 44). It has been proposed that the corresponding region of MLV might also play some role in 3′-end formation, through a hairpin loop formed in the viral RNA at the U3-R border (2). Our results provide evidence for some secondary, albeit unknown, function of this sequence.
Our results also demonstrate that a strong promoter is required to support efficient virus replication. Despite the fact that the Pt and Pc wt PB promoter-targeted LTRs possessed levels of transcriptional strength in LNCaP cells similar to that of the SV40 enhancer/promoter, the corresponding vectors only replicated with very slow kinetics. The use of the hybrid LTRs containing the much stronger ARR2PB resulted in greatly improved replicative efficiency. This improvement in efficiency was not accompanied by an increase in replication in nonprostate cells.
The stability of the targeting sequences during vector replication would be of central importance in any consideration of the therapeutic use of a transcriptionally targeted RCR vector. Our results indicate that the sequence of an ARR2PB-targeted LTR is retained over multiple serial vector passages through human prostate carcinoma cells. Additionally, the specificity of ARR2PB-targeted vector for prostate cells was found to be unchanged after multiple passages, further confirming the stability of the hybrid LTR.
One potential use of a transcriptionally targeted RCR vector would be that of delivering a suicide transgene to tumor cells. Results published previously by Logg et al. (26, 27) demonstrate that RCR vectors containing insertions at the env-3′ untranslated region boundary can retain inserted transgenes over multiple cell-free passages and are stable enough to spread within and transmit inserted transgenes throughout solid tumors with very high efficiency. Delivery of a suicide transgene by an RCR vector into tumors should allow levels of transduction sufficient to achieve a potent antitumor effect following the administration of a prodrug. While the presence of a prodrug would result in the selection of variants of the vector from which the transgene has been deleted, if the prodrug is administered only after sufficient virus spread within the tumor has occurred, such selection pressure should not hinder the therapeutic effectiveness of this approach. Furthermore, the inclusion of a suicide transgene would provide a safety mechanism, as cells harboring the vector would be killed after exposure to the prodrug, thereby attenuating further vector spread. One motivation behind the present study was to examine whether vector replication could be restricted to particular target cell types so as to better confine the effects of a prodrug as well as the continued replication of potential transgene-deleted variants.
These studies indicate that replication of MLV can be directed with high specificity to particular cell types by replacement of the virus's nonspecific enhancer/promoter region with an exogenous promoter. An increasingly large number of cellular promoter sequences with various cell type specificities are becoming available, and many of these should be adaptable for use in generating RCR vectors capable of both highly efficient transduction and target-specific replication.
Acknowledgments
This work was supported in part by NIH grants R01 CA85908 and P01 CA59318, Department of Defense Prostate Cancer Research Program grant DAMD17-00-1-0044 (N.K. and B.H.B.), and the Francis Williams Preston Laboratories of the T.J. Martell Foundation (R.J.M.).
REFERENCES
- 1.Andriani, F., B. Nan, J. Yu, X. Li, N. L. Weigel, M. J. McPhaul, S. Kasper, S. Kagawa, B. Fang, R. J. Matusik, L. Denner, and M. Marcelli. 2001. Use of the probasin promoter ARR2PB to express Bax in androgen receptor-positive prostate cancer cells. J. Natl. Cancer Inst. 93:1314-1324. [DOI] [PubMed] [Google Scholar]
- 2.Benz, E. W., Jr., R. M. Wydro, B. Nadal-Ginard, and D. Dina. 1980. Moloney murine sarcoma proviral DNA is a transcriptional unit. Nature 288:665-669. [DOI] [PubMed] [Google Scholar]
- 3.Cavazzana-Calvo, M., S. Hacein-Bey, G. de Saint Basile, F. Gross, E. Yvon, P. Nusbaum, F. Selz, C. Hue, S. Certain, J. L. Casanova, P. Bousso, F. L. Deist, and A. Fischer. 2000. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 288:669-672. [DOI] [PubMed] [Google Scholar]
- 4.Cohen, L. A. 1982. Isolation and characterization of a serially cultivated, neoplastic, epithelial cell line from the N-nitrosomethylurea induced rat mammary adenocarcinoma. In Vitro 18:565-575. [DOI] [PubMed] [Google Scholar]
- 5.Diaz, R. M., T. Eisen, I. R. Hart, and R. G. Vile. 1998. Exchange of viral promoter/enhancer elements with heterologous regulatory sequences generates targeted hybrid long terminal repeat vectors for gene therapy of melanoma. J. Virol. 72:789-795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Donahue, R. E., S. W. Kessler, D. Bodine, K. McDonagh, C. Dunbar, S. Goodman, B. Agricola, E. Byrne, M. Raffeld, R. Moen, et al. 1992. Helper virus induced T cell lymphoma in nonhuman primates after retroviral mediated gene transfer. J. Exp. Med. 176:1125-1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.DuBridge, R. B., P. Tang, H. C. Hsia, P. M. Leong, J. H. Miller, and M. P. Calos. 1987. Analysis of mutation in human cells by using an Epstein-Barr virus shuttle system. Mol. Cell. Biol. 7:379-387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ferrari, G., G. Salvatori, C. Rossi, G. Cossu, and F. Mavilio. 1995. A retroviral vector containing a muscle-specific enhancer drives gene expression only in differentiated muscle fibers. Hum. Gene Ther. 6:733-742. [DOI] [PubMed] [Google Scholar]
- 9.Feuer, G., and H. Fan. 1990. Substitution of murine transthyretin (prealbumin) regulatory sequences into the Moloney murine leukemia virus long terminal repeat yields infectious virus with altered biological properties. J. Virol. 64:6130-6140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gilmartin, G. M., E. S. Fleming, and J. Oetjen. 1992. Activation of HIV-1 pre-mRNA 3′ processing in vitro requires both an upstream element and TAR. EMBO J. 11:4419-4428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Graves, B. J., R. N. Eisenman, and S. L. McKnight. 1985. Delineation of transcriptional control signals within the Moloney murine sarcoma virus long terminal repeat. Mol. Cell. Biol. 5:1948-1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Greenberg, N. M., F. DeMayo, M. J. Finegold, D. Medina, W. D. Tilley, J. O. Aspinall, G. R. Cunha, A. A. Donjacour, R. J. Matusik, and J. M. Rosen. 1995. Prostate cancer in a transgenic mouse. Proc. Natl. Acad. Sci. USA 92:3439-3443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Greenberg, N. M., F. J. DeMayo, P. C. Sheppard, R. Barrios, R. Lebovitz, M. Finegold, R. Angelopoulou, J. G. Dodd, M. L. Duckworth, J. M. Rosen, et al. 1994. The rat probasin gene promoter directs hormonally and developmentally regulated expression of a heterologous gene specifically to the prostate in transgenic mice. Mol. Endocrinol. 8:230-239. [DOI] [PubMed] [Google Scholar]
- 14.Harsh, G. R., T. S. Deisboeck, D. N. Louis, J. Hilton, M. Colvin, J. S. Silver, N. H. Qureshi, J. Kracher, D. Finkelstein, E. A. Chiocca, and F. H. Hochberg. 2000. Thymidine kinase activation of ganciclovir in recurrent malignant gliomas: a gene-marking and neuropathological study. J. Neurosurg. 92:804-811. [DOI] [PubMed] [Google Scholar]
- 15.Heisler, L. E., A. Evangelou, A. M. Lew, J. Trachtenberg, H. P. Elsholtz, and T. J. Brown. 1997. Androgen-dependent cell cycle arrest and apoptotic death in PC-3 prostatic cell cultures expressing a full-length human androgen receptor. Mol. Cell. Endocrinol. 126:59-73. [DOI] [PubMed] [Google Scholar]
- 16.Horoszewicz, J. S., S. S. Leong, T. M. Chu, Z. L. Wajsman, M. Friedman, L. Papsidero, U. Kim, L. S. Chai, S. Kakati, S. K. Arya, and A. A. Sandberg. 1980. The LNCaP cell line—a new model for studies on human prostatic carcinoma, p. 115-132. In G. P. Murphy (ed.), Models for prostate cancer, vol. 37. Alan R. Liss, New York, N.Y. [PubMed]
- 17.Horton, R. M., H. D. Hunt, S. N. Ho, J. K. Pullen, and L. R. Pease. 1989. Engineering hybrid genes without the use of restriction enzymes: gene splicing by overlap extension. Gene 77:61-68. [DOI] [PubMed] [Google Scholar]
- 18.Jager, U., Y. Zhao, and C. D. Porter. 1999. Endothelial cell-specific transcriptional targeting from a hybrid long terminal repeat retrovirus vector containing human prepro-endothelin-1 promoter sequences. J. Virol. 73:9702-9709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kaighn, M. E., K. S. Narayan, Y. Ohnuki, J. F. Lechner, and L. W. Jones. 1979. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Investig. Urol. 17:16-23. [PubMed] [Google Scholar]
- 20.Kasper, S., and R. J. Matusik. 2000. Rat probasin: structure and function of an outlier lipocalin. Biochim. Biophys. Acta 1482:249-258. [DOI] [PubMed] [Google Scholar]
- 21.Kasper, S., P. S. Rennie, N. Bruchovsky, L. Lin, H. Cheng, R. Snoek, K. Dahlman-Wright, J. A. Gustafsson, R. P. Shiu, P. C. Sheppard, and R. J. Matusik. 1999. Selective activation of the probasin androgen-responsive region by steroid hormones. J. Mol. Endocrinol. 22:313-325. [DOI] [PubMed] [Google Scholar]
- 22.Kasper, S., P. S. Rennie, N. Bruchovsky, P. C. Sheppard, H. Cheng, L. Lin, R. P. Shiu, R. Snoek, and R. J. Matusik. 1994. Cooperative binding of androgen receptors to two DNA sequences is required for androgen induction of the probasin gene. J. Biol. Chem. 269:31763-31769. [PubMed] [Google Scholar]
- 23.Kay, M. A., C. S. Manno, M. V. Ragni, P. J. Larson, L. B. Couto, A. McClelland, B. Glader, A. J. Chew, S. J. Tai, R. W. Herzog, V. Arruda, F. Johnson, C. Scallan, E. Skarsgard, A. W. Flake, and K. A. High. 2000. Evidence for gene transfer and expression of factor IX in haemophilia B patients treated with an AAV vector. Nat. Genet. 24:257-261. [DOI] [PubMed] [Google Scholar]
- 24.Kohn, D. B., M. S. Hershfield, D. Carbonaro, A. Shigeoka, J. Brooks, E. M. Smogorzewska, L. W. Barsky, R. Chan, F. Burotto, G. Annett, J. A. Nolta, G. Crooks, N. Kapoor, M. Elder, D. Wara, T. Bowen, E. Madsen, F. F. Snyder, J. Bastian, L. Muul, R. M. Blaese, K. Weinberg, and R. Parkman. 1998. T lymphocytes with a normal ADA gene accumulate after transplantation of transduced autologous umbilical cord blood CD34+ cells in ADA-deficient SCID neonates. Nat. Med. 4:775-780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kruse, C. A., M. D. Roper, B. K. Kleinschmidt-DeMasters, S. J. Banuelos, W. R. Smiley, J. M. Robbins, and F. J. Burrows. 1997. Purified herpes simplex thymidine kinase retrovector particles. I. In vitro characterization, in situ transduction efficiency, and histopathological analyses of gene therapy-treated brain tumors. Cancer Gene Ther. 4:118-128. [PubMed] [Google Scholar]
- 26.Logg, C. R., A. Logg, C. K. Tai, P. M. Cannon, and N. Kasahara. 2001. Genomic stability of murine leukemia viruses containing insertions at the Env-3′ untranslated region boundary. J. Virol. 75:6989-6998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Logg, C. R., C. K. Tai, A. Logg, W. F. Anderson, and N. Kasahara. 2001. A uniquely stable replication-competent retrovirus vector achieves efficient gene delivery in vitro and in solid tumors. Hum. Gene Ther. 12:921-932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Martuza, R. L. 2000. Conditionally replicating herpes vectors for cancer therapy. J. Clin. Investig. 105:841-846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Miller, D. G., M. A. Adam, and A. D. Miller. 1990. Gene transfer by retrovirus vectors occurs only in cells that are actively replicating at the time of infection. Mol. Cell. Biol. 10:4239-4242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Moore, K. A., M. Scarpa, S. Kooyer, A. Utter, C. T. Caskey, and J. W. Belmont. 1991. Evaluation of lymphoid-specific enhancer addition or substitution in a basic retrovirus vector. Hum. Gene Ther. 2:307-315. [DOI] [PubMed] [Google Scholar]
- 31.Navone, N. M., M. Olive, M. Ozen, R. Davis, P. Troncoso, S. M. Tu, D. Johnston, A. Pollack, S. Pathak, A. C. von Eschenbach, and C. J. Logothetis. 1997. Establishment of two human prostate cancer cell lines derived from a single bone metastasis. Clin. Cancer Res. 3:2493-2500. [PubMed] [Google Scholar]
- 32.Onodera, M., A. Yachie, D. M. Nelson, H. Welchlin, R. A. Morgan, and R. M. Blaese. 1997. A simple and reliable method for screening retroviral producer clones without selectable markers. Hum. Gene Ther. 8:1189-1194. [DOI] [PubMed] [Google Scholar]
- 33.Puumalainen, A. M., M. Vapalahti, R. S. Agrawal, M. Kossila, J. Laukkanen, P. Lehtolainen, H. Viita, L. Paljarvi, R. Vanninen, and S. Yla-Herttuala. 1998. Beta-galactosidase gene transfer to human malignant glioma in vivo using replication-deficient retroviruses and adenoviruses. Hum. Gene Ther. 9:1769-1774. [DOI] [PubMed] [Google Scholar]
- 34.Rainov, N. G. 2000. A phase III clinical evaluation of herpes simplex virus type 1 thymidine kinase and ganciclovir gene therapy as an adjuvant to surgical resection and radiation in adults with previously untreated glioblastoma multiforme. Hum. Gene Ther. 11:2389-2401. [DOI] [PubMed] [Google Scholar]
- 35.Ram, Z., K. W. Culver, E. M. Oshiro, J. J. Viola, H. L. DeVroom, E. Otto, Z. Long, Y. Chiang, G. J. McGarrity, L. M. Muul, D. Katz, R. M. Blaese, and E. H. Oldfield. 1997. Therapy of malignant brain tumors by intratumoral implantation of retroviral vector-producing cells. Nat. Med. 3:1354-1361. [DOI] [PubMed] [Google Scholar]
- 36.Rennie, P. S., N. Bruchovsky, K. J. Leco, P. C. Sheppard, S. A. McQueen, H. Cheng, R. Snoek, A. Hamel, M. E. Bock, B. S. MacDonald, et al. 1993. Characterization of two cis-acting DNA elements involved in the androgen regulation of the probasin gene. Mol. Endocrinol. 7:23-36. [DOI] [PubMed] [Google Scholar]
- 37.Russell, S. J. 1994. Replicating vectors for gene therapy of cancer: risks, limitations and prospects. Eur. J. Cancer 8:1165-1171. [DOI] [PubMed] [Google Scholar]
- 38.Scherer, W. F., J. T. Syverton, and G. O. Gey. 1953. Viral multiplication in a stable strain of human malignant epithelial cells (strain HeLa) derived from an epidermoid carcinoma of the cervix. J. Exp. Med. 97:695-709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Short, M. P., B. C. Choi, J. K. Lee, A. Malick, X. O. Breakefield, and R. L. Martuza. 1990. Gene delivery to glioma cells in rat brain by grafting of a retrovirus packaging cell line. J. Neurosci. Res. 27:427-439. [DOI] [PubMed] [Google Scholar]
- 40.Smiley, W. R., B. Laubert, B. D. Howard, C. Ibanez, T. C. Fong, W. S. Summers, and F. J. Burrows. 1997. Establishment of parameters for optimal transduction efficiency and antitumor effects with purified high-titer HSV-TK retroviral vector in established solid tumors. Hum. Gene Ther. 8:965-977. [DOI] [PubMed] [Google Scholar]
- 41.Stojdl, D. F., B. Lichty, S. Knowles, R. Marius, H. Atkins, N. Sonenberg, and J. C. Bell. 2000. Exploiting tumor-specific defects in the interferon pathway with a previously unknown oncolytic virus. Nat. Med. 6:821-825. [DOI] [PubMed] [Google Scholar]
- 42.Stone, K. R., D. D. Mickey, H. Wunderli, G. H. Mickey, and D. F. Paulson. 1978. Isolation of a human prostate carcinoma cell line (DU 145). Int. J. Cancer 21:274-281. [DOI] [PubMed] [Google Scholar]
- 43.Strong, J. E., M. C. Coffey, D. Tang, P. Sabinin, and P. W. Lee. 1998. The molecular basis of viral oncolysis: usurpation of the Ras signaling pathway by reovirus. EMBO J. 17:3351-3362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Valsamakis, A., S. Zeichner, S. Carswell, and J. C. Alwine. 1991. The human immunodeficiency virus type 1 polyadenylylation signal: a 3′ long terminal repeat element upstream of the AAUAAA necessary for efficient polyadenylylation. Proc. Natl. Acad. Sci. USA 88:2108-2112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vile, R. G., S. J. Russell, and N. R. Lemoine. 2000. Cancer gene therapy: hard lessons and new courses. Gene Ther. 7:2-8. [DOI] [PubMed] [Google Scholar]
- 46.Wildner, O. 2001. Oncolytic viruses as therapeutic agents. Ann. Med. 33:291-304. [DOI] [PubMed] [Google Scholar]
- 47.Wu, X., J. Wu, J. Huang, W. C. Powell, J. Zhang, R. J. Matusik, F. O. Sangiorgi, R. E. Maxson, H. M. Sucov, and P. Roy-Burman. 2001. Generation of a prostate epithelial cell-specific Cre transgenic mouse model for tissue-specific gene ablation. Mech. Dev. 101:61-69. [DOI] [PubMed] [Google Scholar]
- 48.Xie, X., X. Zhao, Y. Liu, J. Zhang, R. J. Matusik, K. M. Slawin, and D. M. Spencer. 2001. Adenovirus-mediated tissue-targeted expression of a caspase-9-based artificial death switch for the treatment of prostate cancer. Cancer Res. 61:6795-6804. [PubMed] [Google Scholar]
- 49.Yang, L., R. Hwang, L. Pandit, E. M. Gordon, W. F. Anderson, and D. Parekh. 1996. Gene therapy of metastatic pancreas cancer with intraperitoneal injections of concentrated retroviral herpes simplex thymidine kinase vector supernatant and ganciclovir. Ann. Surg. 224:405-414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang, J., T. Z. Thomas, S. Kasper, and R. J. Matusik. 2000. A small composite probasin promoter confers high levels of prostate-specific gene expression through regulation by androgens and glucocorticoids in vitro and in vivo. Endocrinology 141:4698-4710. [DOI] [PubMed] [Google Scholar]
- 51.Zhao, X. Y., P. J. Malloy, A. V. Krishnan, S. Swami, N. M. Navone, D. M. Peehl, and D. Feldman. 2000. Glucocorticoids can promote androgen-independent growth of prostate cancer cells through a mutated androgen receptor. Nat. Med. 6:703-706. [DOI] [PubMed] [Google Scholar]