Abstract
The antimicrobial activity of human β-defensin 3 (hBD-3) against multidrug-resistant clinical isolates of Staphylococcus aureus, Enterococcus faecium, Pseudomonas aeruginosa, Stenotrophomonas maltophilia, and Acinetobacter baumannii was evaluated. A fast bactericidal effect (within 20 min) against all bacterial strains tested was observed. The presence of 20% human serum abolished the bactericidal activity of hBD-3 against gram-negative strains and reduced the activity of the peptide against gram-positive strains.
The evolution and rapid spread of clinical bacterial strains resistant to the available antimicrobial agents are significant nosocomial problems and are of increasing importance in community-acquired infections (11). Besides being a growing cause of morbidity and mortality worldwide, the spread of multiresistant strains is associated with an increased rate of hospitalization, increased lengths of hospital stay, and consequently, increased health care costs (11). The progressive reduction of the therapeutic efficacies of the available antibiotics due to the spread of antimicrobial resistance underlines the urgency for the development of new classes of drugs for the treatment of infectious diseases (11). In light of this situation, antimicrobial peptides, alone or combined with conventional antibiotics, are attractive candidates as therapeutic agents for bacterial infections because of their selectivity, speed of action, relative difficulty in selection of resistant mutants in vitro, and inherent immunological compatibility (2, 3). In humans, an important family of antimicrobial peptides is represented by defensins; they are small cationic and Cys-rich peptides with molecular masses ranging from 3 to 5 kDa (1). On the basis of sequence homology and the localization of six conserved cysteyl residues, human defensins are classified into the α and the β families (6). In particular, the β-defensin 3 (hBD-3), which is widely expressed in skin, respiratory epithelium, and oral tissues, has generated interest because it shows strong, broad-spectrum, and salt-insensitive antimicrobial activity at concentrations noncytotoxic for human cells (4, 7, 9). A previous report demonstrated that hBD-3 is bactericidal against a panel of reference strains of clinically relevant bacterial species (12). No data on the effects of hBD-3 against multidrug-resistant strains isolated from hospitalized patients are yet available.
The aims of this study, therefore, were (i) to evaluate the bactericidal activity of hBD-3 against multidrug-resistant clinical isolates of common nosocomial pathogens, such as Staphylococcus aureus, Enterococcus faecium, and Pseudomonas aeruginosa, and clinical isolates of emergent pathogens, such as Stenotrophomonas maltophilia and Acinetobacter baumannii; (ii) to investigate the kinetics of the bactericidal activity of hBD-3 against selected strains; and (iii) to evaluate the bactericidal activity of hBD-3 in the presence of human serum.
The bactericidal activity of hBD-3 against all clinical isolates was evaluated by a liquid microdilution assay in 10 mM sodium phosphate buffer (SPB; pH 7.4) (7). The bactericidal activity of the peptide was also tested against a representative gram-positive strain and a representative gram-negative strain in 50 and 150 mM SPB. Identification and susceptibility testing of clinical isolates, collected from distinct patients at the Microbiology Unit of the University Hospital of Pisa, were performed by using VITEK 2 automatic instruments (Bio-Merieux, Lyon, France). The drug susceptibilities of S. maltophilia and A. baumannii and the vancomycin susceptibility of E. faecium were confirmed by using Etest (AB Biodisk, Solna, Sweden). Clinical isolates were considered multidrug resistant if they were resistant to at least three agents from distinct classes of antibiotics. According to this definition only A. baumannii strains 4, 5, and 6 were not multidrug resistant (Table 1). The bacterial strains were grown in tryptone soy broth (TSB; Oxoid, Basingstoke, United Kingdom). Synthetic hBD-3 (Sigma-Genosys) was diluted in 0.1% acetic acid to obtain a stock solution of 10 mg/ml. Exponentially growing bacteria were resuspended in SPB to reach a density of 1 × 107 CFU/ml. Ten microliters of each bacterial suspension was exposed at 37°C for various times (from 1 min to 3 h) to different concentrations of hBD-3 in 100 μl of SPB alone or in the presence of 20% heat-inactivated (56°C for 30 min) pooled human sera obtained from five healthy donors. Following incubation, the samples were diluted 10-fold in TSB, and 0.2 ml of each dilution was plated onto tryptone soy agar. The number of CFU was determined after 24 h of incubation at 37°C. Bactericidal activity was defined as a reduction in the numbers of viable bacteria of ≥3 log10 CFU/ml at any of the incubation times tested.
TABLE 1.
Bactericidal activity of hBD-3 against nosocomial strains with defined resistance phenotypes
| Organism | Strain | Resistance phenotypea | hBD-3 concnb (μg/ml) |
|---|---|---|---|
| S. aureus | 1 | AMC AMP CIP ERY GEN OXA RIF | 4 |
| S. aureus | 2 | AMC AMP CIP ERY GEN LVX OXA RIF TET | 4 |
| S. aureus | 3 | AMC AMP CIP ERY GEN LVX OXA | 8 |
| S. aureus | 4 | AMC AMP AZM CIP CTX ERY IPM LVX OXA | 8 |
| S. aureus | 5 | AMC AMP CIP GEN IPM TOB SXT OXA AZM ERY CTX | 4 |
| S. aureus | 6 | AMP AMC AZM CIP CTX ERY GEN IPM LVX OXA RIF SXT | 4 |
| E. faecium | 1 | AMC CIP CTX ERY GEN SXT TEC VAN | 8 |
| E. faecium | 2 | AMP CIP CTX ERY GEN LVX TEC VAN | 8 |
| E. faecium | 3 | AMP CIP CTX ERY GEN LVX SXT TEC VAN | 8 |
| E. faecium | 4 | AMP CIP ERY GEN SXT TEC VAN | 8 |
| E. faecium | 5 | AMP AMC CIP CLI ERY GEN KAN LVX SXT TEC VAN | 8 |
| E. faecium | 6 | AMP AMC CLI CRO CTX CIP ERY GEN KAN LVX SXT VAN | 8 |
| P. aeruginosa | 1 | AMP CFZ CIP CTX CXM GEN LVX SXT TET | 8 |
| P. aeruginosa | 2 | AMP CFZ CIP CTX CXM GEN LVX IPM MEM SXT TET | 8 |
| P. aeruginosa | 3 | AMP CFZ CTX CIP GEN IPM LVX SXT TET TOB | 8 |
| P. aeruginosa | 4 | AMP AMC CFZ CRO GEN NIT SXT | 8 |
| P. aeruginosa | 5 | AMP AMC CTX LVX TET TOB SXT | 4 |
| P. aeruginosa | 6 | AMP AMC CFZ CIP GEN IPM TOB SXT | 8 |
| S. maltophilia | 1 | AMK CIP CTX IPM MEM SAM TZP | 4 |
| S. maltophilia | 2 | AMK CIP CTX IPM MEM SAM TZP | 4 |
| S. maltophilia | 3 | AMK CIP CTX IPM MEM SAM TZP | 8 |
| S. maltophilia | 4 | AMK CAZ CIP FEP IPM SAM TZP | 8 |
| S. maltophilia | 5 | AMK CIP CTX IPM MEM SAM TZP | 4 |
| S. maltophilia | 6 | AMK CIP CTX IPM MEM SAM TZP | 4 |
| A. baumannii | 1 | AMP AMC CFZ CIP IPM LVX PIP SXT TET | 4 |
| A. baumannii | 2 | AMP CFZ CIP LVX PIP SXT | 4 |
| A. baumannii | 3 | AMP AMC CFZ CIP IPM SXT TET TOB | 4 |
| A. baumanniic | 4 | AMP CFZ CTX TET | 4 |
| A. baumanniic | 5 | CFZ CFM CXM MEZ | 4 |
| A. baumanniic | 6 | AMP CFZ CFM | 4 |
AMC, amoxycillin; AMP, ampicillin; CIP, ciprofloxacin; ERY, erythromycin; GEN, gentamicin; OXA, oxacillin; RIF, rifampin; LVX, levofloxacin; TET, tetracycline; AZM, azithromycin; CTX, cefotaxime; IPM, imipenem; TOB, tobramycin; SXT, trimethoprim-sulfamethoxazole; TEC, teicoplanin; VAN, vancomycin; CLI, clindamycin; CRO, ceftriaxone; NIT, nitrofurantoin; CFZ, cefazolin; MEM, meropenem; SAM, ampicillin-sulbactam; TZP, piperacillin-tazobactam; AMK, amikacin; CAZ, ceftazidime; FEP, cefepime; PIP, piperacillin; TOB, tobramycin; MEZ, mezlocillin.
Concentration of hBD-3 sufficient to obtain a bactericidal effect after 1.5 h of incubation in SPB.
Bacterial strains resistant to one or two classes of antibiotics.
hBD-3 demonstrated bactericidal activity against all clinical isolates tested (six strains of every species). The concentrations of peptide sufficient to obtain a bactericidal effect after 1.5 h of incubation in SPB were 4 μg/ml for A. baumannii; 8 μg/ml for E. faecium; and from 4 to 8 μg/ml for S. aureus, P. aeruginosa, and S. maltophilia (Table 1).
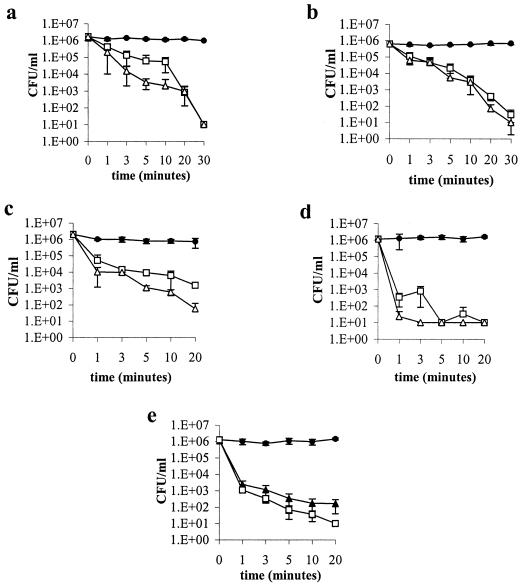
The killing kinetics of hBD-3 were evaluated with a representative isolate of each bacterial species arbitrarily chosen from among those that turned out to be less susceptible to the peptide (Table 1). In these experiments the peptide was tested at concentrations equal to or twofold greater than those that resulted in bactericidal activity after 1.5 h of incubation. hBD-3 showed fast bactericidal activity against every bacterial strain tested (Fig. 1). In particular, hBD-3 used at a concentration of 4 μg/ml was bactericidal against A. baumannii (strain 3) after 3 min of incubation (Fig. 1e). At a concentration of 8 μg/ml, the peptide caused a bactericidal effect within 20 min of incubation against S. aureus (strain 3), E. faecium (strain 3), and P. aeruginosa (strain 3) and within 1 min against S. maltophilia (strain 4) and A. baumannii (Fig. 1a to e). At a concentration of 16 μg/ml, the peptide produced a bactericidal effect against S. aureus, E. faecium, P. aeruginosa, and S. maltophilia within 10, 20, 5, and 1 min, respectively (Fig. 1a to d, respectively). In order to evaluate the impact of high ionic strength on the activity of hBD-3, the killing kinetics of hBD-3 against A. baumannii (strain 3) and S. aureus (strain 3) were also determined in the presence of 50 and 150 mM SPB. The peptide showed a bactericidal effect in 50 mM SPB against A. baumannii at 8 μg/ml after 3 min of incubation and against S. aureus at 16 μg/ml after 1.5 h of incubation. In the presence of 150 mM SPB, hBD-3 was bactericidal against A. baumannii at 64 μg/ml after 1.5 h of incubation, while it was shown to be inactive against S. aureus (data not shown).
FIG. 1.
Time-kill curves of S. aureus (strain 3) (a), E. faecium (strain 3) (b), P. aeruginosa (strain 3) (c), S. maltophilia (strain 4) (d), and A. baumannii (strain 3) (e) in SPB. The concentrations of hBD-3 are in μg/ml. The control consisted of bacteria incubated in the absence of hBD-3 but in the presence of the appropriate amount of the solvent (0.1% acetic acid). Data are means ± standard deviation of three independent experiments. •, control; ▴, 4 μg/ml; □, 8 μg/ml; Δ, 16 μg/ml.
In order to evaluate whether hBD-3 retains activity against selected drug-resistant strains in the presence of biological fluids, bactericidal assays were performed in the presence of human serum. Heat-inactivated human serum diluted at 20% abolished the bactericidal activity of hBD-3 (assayed to 256 μg/ml) against P. aeruginosa (strain 3), S. maltophilia (strain 4), and A. baumannii (strain 3) within 3 h of incubation, while the peptide maintained its bactericidal activity against S. aureus (strain 3) and E. faecium (strain 3) at concentrations of 128 and 64 μg/ml, respectively (Table 2).
TABLE 2.
Effect of human serum on bactericidal activity of hBD-3
| Organism | Strain no. | hBD-3 concna (μg/ml) after:
|
|
|---|---|---|---|
| 1.5 h | 3 h | ||
| S. aureus | 3 | 128 | 128 |
| E. faecium | 3 | 64 | 64 |
| P. aeruginosa | 3 | >256 | >256 |
| S. maltophilia | 4 | >256 | >256 |
| A. baumannii | 3 | >256 | >256 |
Concentrations of hBD-3 necessary to obtain a bactericidal effect in the presence of 20% heat-inactivated human serum.
This study demonstrates that hBD-3 at relatively low concentrations is bactericidal against multidrug-resistant nosocomial strains, independently of their resistance phenotypes. It is noteworthy that a significant variability of the bactericidal concentrations of hBD-3 was not observed among clinical isolates belonging to the same species, while a strain selectivity of hBD-3, when it was tested against periodontal pathogens, has recently been reported (5). Those authors argue that such a strain selectivity of the peptide can be due to the variable compositions of lipopolysaccharide, which represents an important microbial target of the peptide (5). Moreover, hBD-3 exhibited rapid bactericidal activity against every bacterial strain tested, suggesting that the drug resistance does not interfere with the mode of action of hBD-3. Although hBD-3 is insensitive to physiologic concentrations of NaCl (150 mM) (4), the strong inhibition of bactericidal activity of hBD-3 in the presence of 150 mM SPB was observed. This finding suggests that hBD-3 is not insensitive to all types of salts, although the concentrations of phosphate salts tested were much higher than those detectable in normal serum (13). In accordance with our previous findings, the bactericidal activity of hBD-3 was inhibited in the presence of human serum (8). In particular, while the bactericidal activity of hBD-3 against gram-negative strains was completely abolished in the presence of serum, the peptide remained active against gram-positive isolates when it was used at high concentrations. Altogether these results suggest that, in addition to the presence of proteases or molecules that reduce peptide bioavailability (14), other serum-mediated inhibitory mechanisms, such as high ionic strength or salt properties, might be important in hBD-3 inhibition. Moreover, the differential susceptibilities to hBD-3 of gram-positive and gram-negative isolates observed in serum suggests that human serum may contain components that could mask the binding sites for the peptide as a result of their interaction with the surfaces of gram-negative bacterial microorganisms (10). Further studies aimed at elucidating the mechanisms involved in the inhibitory effect of serum on hBD-3 and the identification of derivatives of the peptide less sensitive to the inhibitory action of serum are required. Nevertheless, the high susceptibility to hBD-3 of bacterial strains resistant to most currently available antibiotics makes such a peptide an interesting antimicrobial agent for potential use for the therapy of nosocomial infections.
Acknowledgments
This work was supported by grants from “PRIN” protocols 2002067349-001 and 2004067822-001, Rome, Italy.
REFERENCES
- 1.Ganz, T. 2003. Defensins: antimicrobial peptides of innate immunity. Nat. Rev. 3:710-720. [DOI] [PubMed] [Google Scholar]
- 2.Hancock, R. E. W. 2005. Mechanism of action of newer antibiotics for gram-positive pathogens. Lancet Infect. Dis. 5:209-218. [DOI] [PubMed] [Google Scholar]
- 3.Hancock, R. E. W. 2001. Cationic peptides: effectors in innate immunity and novel antimicrobials. Lancet Infect. Dis. 1:156-164. [DOI] [PubMed] [Google Scholar]
- 4.Harder, J., J. Bartels, E. Christophers, and J. M. Schroder. 2001. Isolation and characterization of human β-defensin 3, a novel human inducible peptide antibiotic. J. Biol. Chem. 276:5707-5713. [DOI] [PubMed] [Google Scholar]
- 5.Joly, S., C. Maze, P. B. McCray, Jr., and J. M. Guthmiller. 2004. Human β-defensin 2 and 3 demonstrate strain-selectivity against oral microorganisms. J. Clin. Microbiol. 42:1024-1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lehrer, R. I. 2004. Primate defensins. Nat. Rev. Microbiol. 2:727-738. [DOI] [PubMed] [Google Scholar]
- 7.Maisetta, G., G. Batoni, S. Esin, F. Luperini, M. Pardini, D. Bottai, W. Florio, M. R. Giuca, M. Gabriele, and M. Campa. 2003. Activity of human β-defensin 3 alone or combined with other antimicrobial agents against oral bacteria. Antimicrob. Agents Chemother. 47:3349-3351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Maisetta, G., G. Batoni, S. Esin, G. Raco, D. Bottai, F. Favilli, W. Florio, and M. Campa. 2005. Susceptibility of Streptococcus mutans and Actinobacillus actinomycetemcomitans to bactericidal activity of human β-defensin 3 in biological fluids. Antimicrob. Agents Chemother. 49:1245-1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nishimura, M., Y. Abiko, Y. Kurashige, M. Takeshima, M. Yamazaki, K. Kusano, M. Saitoh, K. Nakashima, T. Inoue, and T. Kaku. 2004. Effect of defensin peptides on eukaryotic cells: primary epithelial cells, fibroblast and squamous cell carcinoma cell lines. J. Dermatol. Sci. 36:87-95. [DOI] [PubMed] [Google Scholar]
- 10.Nyberg, P., M. Rasmussen, and L. Bjorck. 2004. α2-macroglobulin-proteinase complex protect Streptococcus pyogenes from killing by the antimicrobial peptide LL-37. J. Biol. Chem. 51:52820-52823. [DOI] [PubMed] [Google Scholar]
- 11.Rice, L. B. 2003. Do we really need new anti-infective drugs? Curr. Opin. Pharmacol. 5:459-463. [DOI] [PubMed] [Google Scholar]
- 12.Sahly, H., S. Shubert, J. Harder, P. Rautenberg, U. Ulmann, J. Schroder, and R. Podschun. 2003. Burkholderia is highly resistant to human beta-defensin 3. Antimicrob. Agents Chemother. 5:1739-1741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shiber, J. R., and A. Mattu. 2002. Serum phosphate abnormalities in the emergency department. J. Emerg. Med. 23:395-400. [DOI] [PubMed] [Google Scholar]
- 14.Wang, Y., B. Agerberth, A. Lothgren, A. Almstedt, and J. Johansson. 1998. Apolipoprotein A-I binds and inhibits the human antibacterial/cytotoxic peptide LL-37. J. Biol. Chem. 273:33115-33118. [DOI] [PubMed] [Google Scholar]